Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of animal- and plant-derived amino acid mixtures, which function as skin and hair conditioning agents. The safety of α-amino acids as direct food additives has been well established, based on extensive research through acute and chronic dietary exposures and the Panel previously has reviewed the safety of individual α-amino acids in cosmetics. The Panel focused its review on dermal irritation and sensitization data relevant to the use of these ingredients in topical cosmetics. The Panel concluded that these 21 ingredients are safe in the present practices of use and concentration as used in cosmetics.
Keywords
Introduction
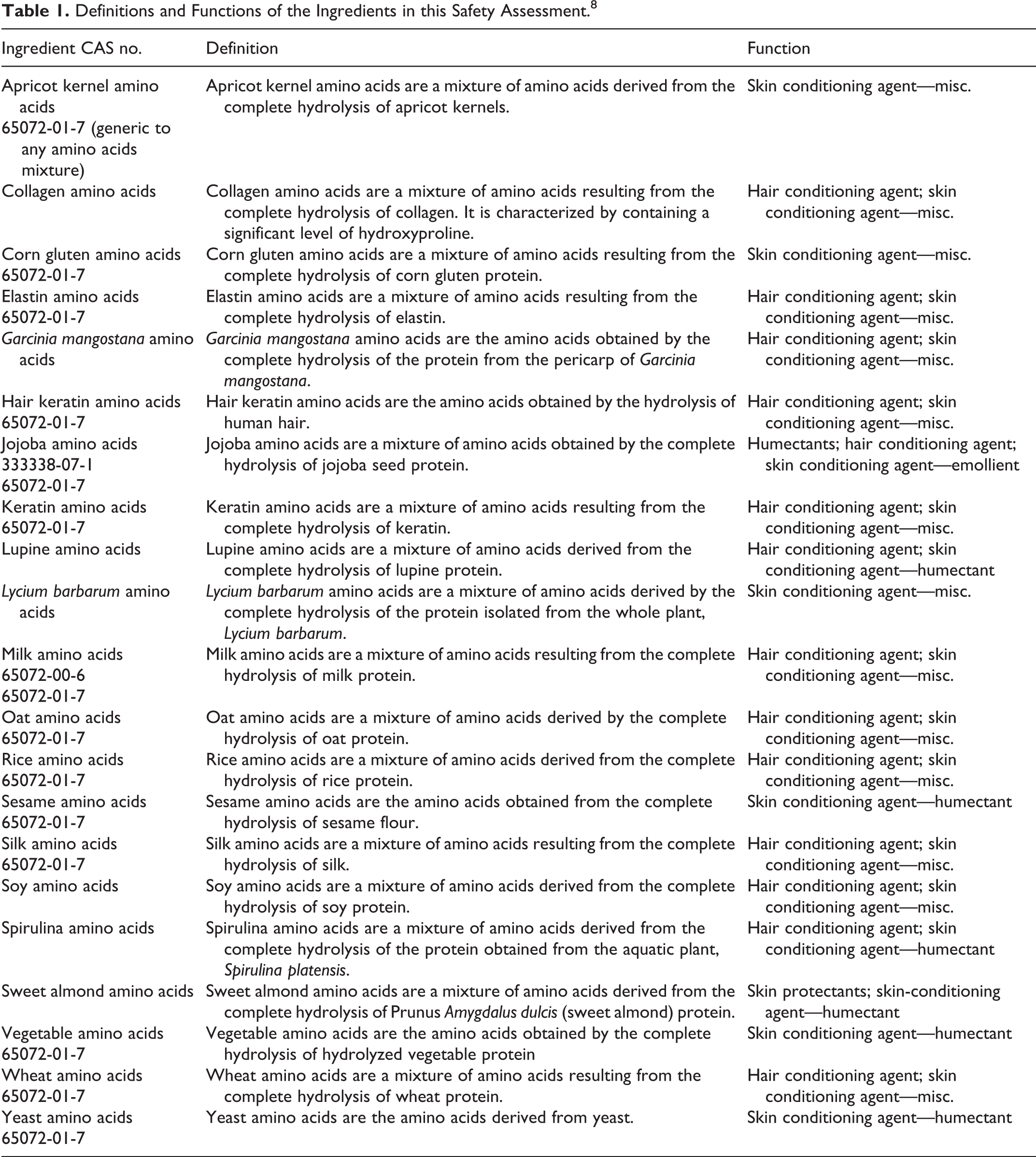
This safety assessment covers 21 ingredients, each of which is a mixture of amino acids derived from specific animal and plant sources. These ingredients function as skin and hair conditioning agents in personal care products. The full list of ingredients in this report is found in Table 1.
Definitions and Functions of the Ingredients in this Safety Assessment. 8
A concurrent review of the safety of hydrolyzed proteins from animal and plant sources is being performed by the Cosmetic Ingredient Review (CIR) Expert Panel. The Expert Panel previously has reviewed the safety of individual α-amino acids and concluded that these ingredients are safe for use in cosmetic ingredients. 1
Chemistry
The ingredients in this group are related because they are each prepared from proteins by complete hydrolysis to yield amino acid mixtures (Figure 1). The most frequently used method for hydrolyzing proteins to amino acids appears to be heating to at least 110°C with concentrated hydrochloric acid.
2,3
However, some amino acids, including tryptophan, are readily destroyed or are only partially hydrolyzed by hydrochloric acid. For these amino acids, alternate hydrolysis agents, like sulfonic acids, are used in place of hydrochloric acid, or alkaline hydrolysis is performed.
3
–5
While these amino acids are produced from proteins, the hydrolysis process commonly racemizes the amino acids and the end product is not primarily in the
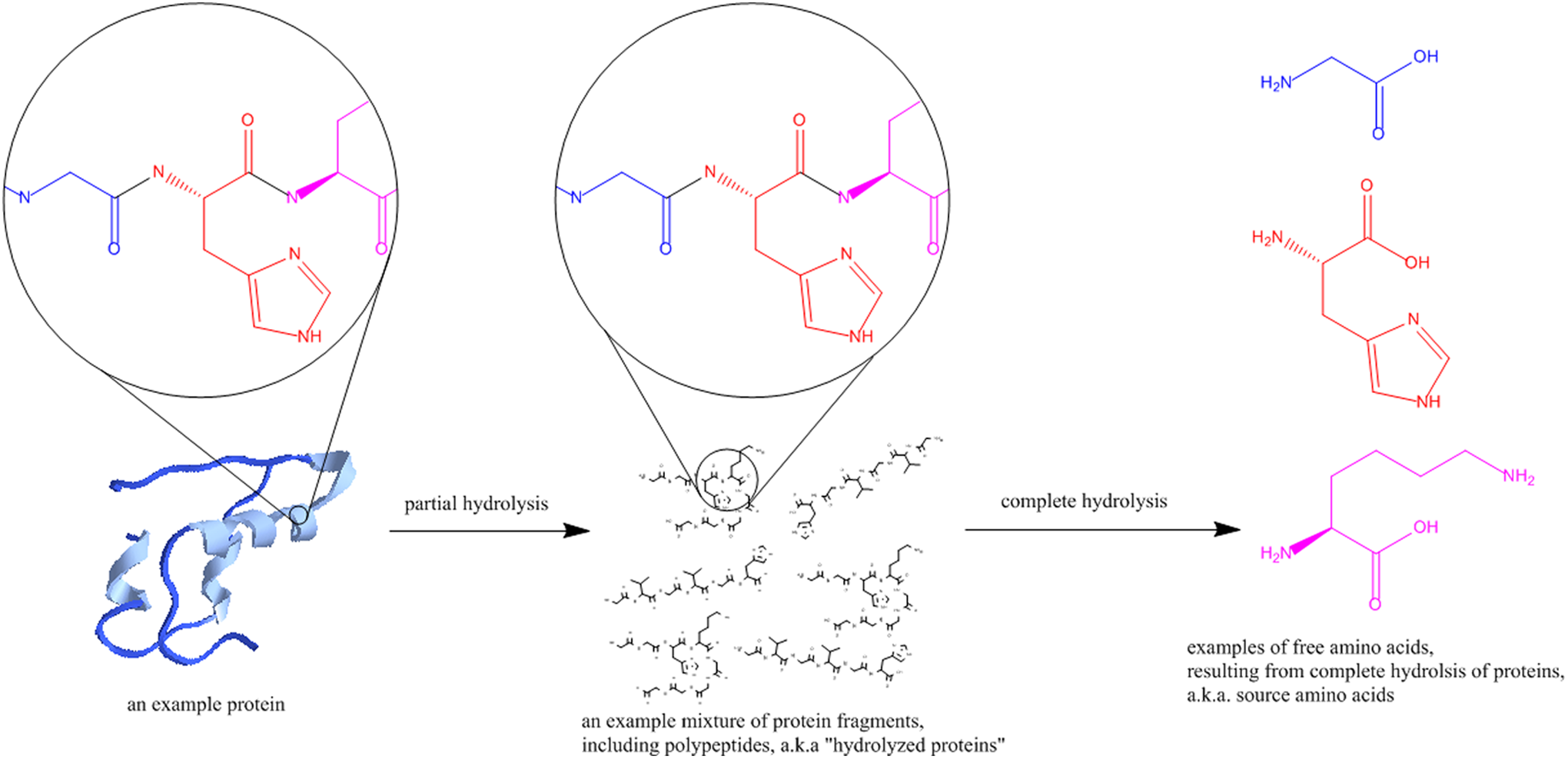
Production of animal- and plant-derived amino acids. 16
The composition of amino acids may be analyzed by chromatographic separation, such as separation with liquid–liquid chromatography ion-exchange columns, and detection, such as detection via mass spectrometry, of the residues after adequate hydrolysis of the substrate. 2,3
Method of Manufacturing
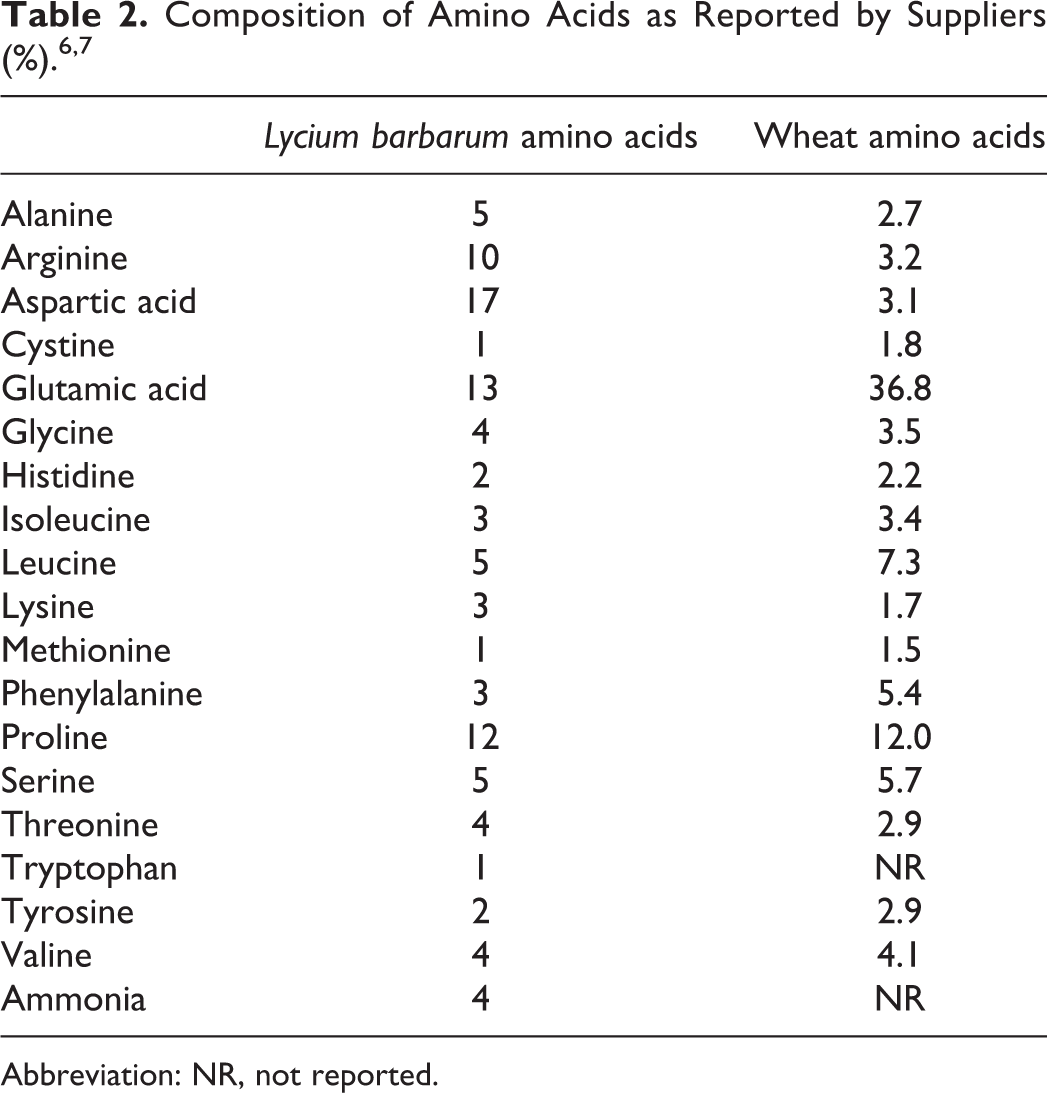
As noted previously, most methods utilized acid hydrolysis at elevated temperatures to hydrolyze proteins to their constituent amino acids. However, one supplier has reported that Lycium barbarum (wolfberry) amino acids are derived from the L barbarum fruit. 6 The fruit is autolyzed in water to free the cytoplasmic proteins. The proteins are then hydrolyzed with proprietary protease enzymes. The hydrolysis breaks down the fruit proteins into basic amino acid constituents that are water soluble. The product is then purified by filtration and spray-dried to obtain a powdered mixture.
Another supplier reported that amino acids from keratin, lupine, oat, rice, silk, and wheat proteins are manufactured by enzymatic hydrolysis for a specific duration of time and at an elevated temperature (details not provided). 7
Impurities
Several of the ingredients of this safety assessment, including collagen amino acids and elastin amino acids, may be bovine sourced. These ingredients are highly processed and, as such, Food and Drug Administration (FDA) does not consider them risk materials for transmission of infectious agents (21 CFR §700.27).
No data were found in the published literature on the impurities of animal- and plant-derived amino acids. A supplier has provided the composition data on amino acids from L barbarum and wheat proteins (see Table 2). 6,7 No composition data were discovered for the remaining animal- and plant-derived amino acid ingredients.
Abbreviation: NR, not reported.
Use
Cosmetic
The animal- and plant-derived amino acids in this safety assessment function primarily as hair conditioning and skin conditioning agents in cosmetic formulations. 8 These functions are similar to those of the α-amino acid ingredients. 1
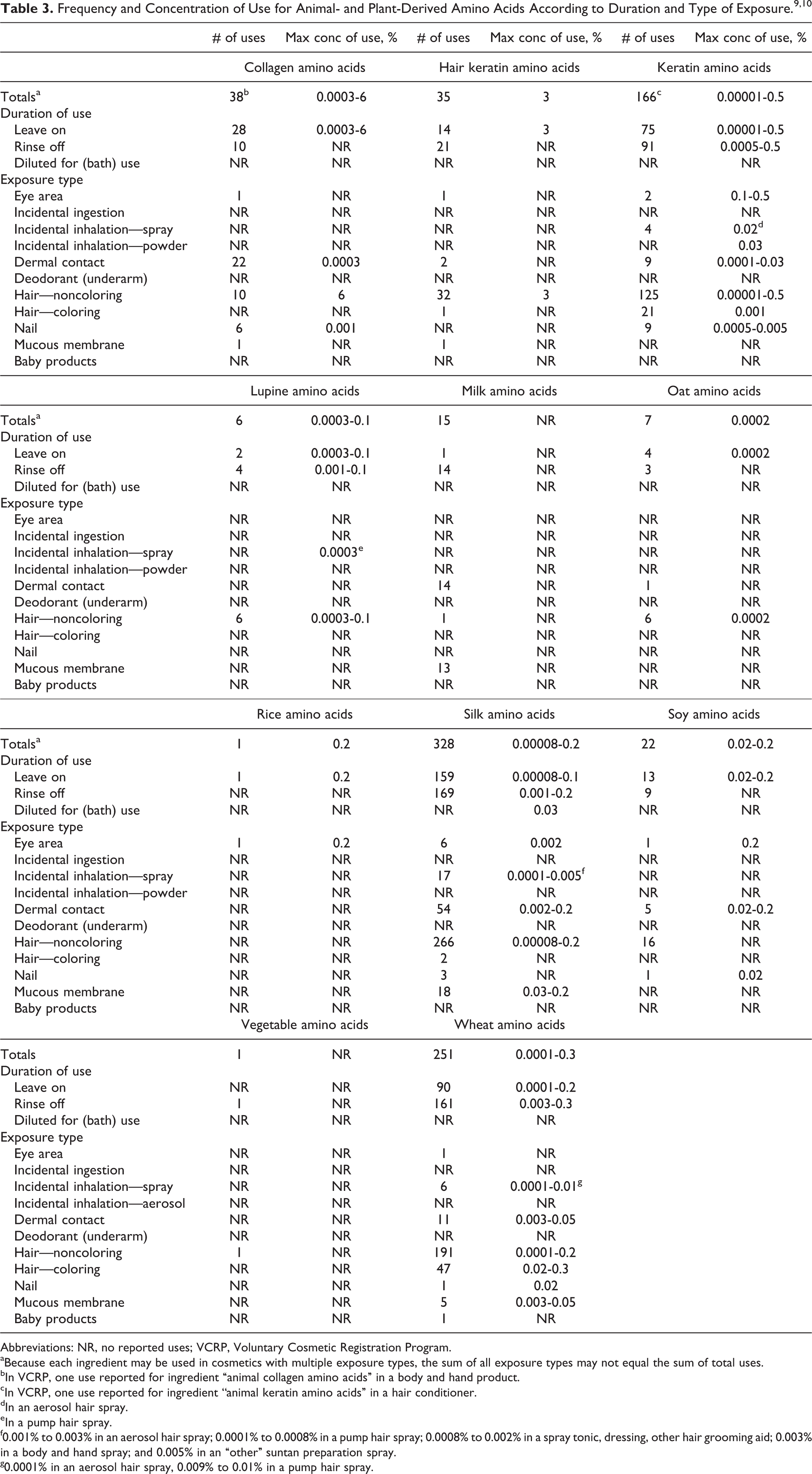
Table 3 presents the current product formulation data for animal- and plant-derived amino acids. According to information supplied to the FDA by industry as part of the Voluntary Cosmetic Registration Program, silk amino acids have the most reported uses in cosmetic and personal care products, with a total of 328; approximately half of those uses are in noncoloring hair products. 9 Wheat amino acids have the second greatest number of overall uses reported, with a total of 251; again, approximately half of those uses are in noncoloring hair products. The reported product categories of the animal- and plant-derived amino acid ingredients are comparable to those of the α-amino acid ingredients. 1
Abbreviations: NR, no reported uses; VCRP, Voluntary Cosmetic Registration Program.
aBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types may not equal the sum of total uses.
bIn VCRP, one use reported for ingredient “animal collagen amino acids” in a body and hand product.
cIn VCRP, one use reported for ingredient “animal keratin amino acids” in a hair conditioner.
dIn an aerosol hair spray.
eIn a pump hair spray.
f0.001% to 0.003% in an aerosol hair spray; 0.0001% to 0.0008% in a pump hair spray; 0.0008% to 0.002% in a spray tonic, dressing, other hair grooming aid; 0.003% in a body and hand spray; and 0.005% in an “other” suntan preparation spray.
g0.0001% in an aerosol hair spray, 0.009% to 0.01% in a pump hair spray.
In a recent survey of use concentrations by the Personal Care Products Council, silk amino acids had a maximum use concentration range of 8.0 × 10−5% to 0.2%, with the 0.2% reported in shampoos and other personal cleanliness products. 10 Wheat amino acids had a maximum use concentration range of 1.0 × 10−4% to 0.3%, with 0.3% reported in hair dyes. The use concentrations of the animal- and plant-derived amino acid ingredients are similar, if not lower in some cases, to those of the α-amino acid ingredients. 1 Those ingredients with no reported uses or use concentrations are listed in Table 4.
Several of the amino acids sourced from animals and plants described in this report are used in cosmetic sprays, including aerosol and pump hair and body spray products, and could possibly be inhaled. The maximum concentration of amino acids from animal and plant sources reported to be used in a spray product is 0.02% keratin amino acids in an aerosol hair spray. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 11,12 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, able to enter the lungs) to any appreciable amount. 13,14
The European Commission’s Scientific Committee on Consumer Products concluded that the risk of exposure to viruses, such as avian influenza virus or papillomavirus, is negligible through topical application of cosmetic products containing amino acids from the hydrolysis of human hair or chicken feathers. 15
Toxicological Studies
Many of the proteins that serve as the sources of the amino acids that are described in this safety assessment are found in the foods consumed daily. Toxicities from dermal exposure would not be expected to be different from oral exposures and, as such, not concerning to the CIR Expert Panel. Irritation and sensitization are of concern and are the focus in this report. Data from the previous safety assessment on α-amino acids support that these ingredients would not likely be irritants or sensitizers. 1
Irritation and Sensitization
As given in the Panel safety assessment of α-amino acids, 1 cysteine HCl and methionine were used as negative controls in in vitro assays to predict potential skin irritants. In separate efficacy studies, arginine, cysteine, and glycine did not produce any adverse effects in rats, guinea pigs, or mouse skin models. Glutamic acid was used as a negative control in an in vitro study to identify skin sensitizers. Products containing amino acid ingredients at concentrations up to 2.784% were not dermal irritants or sensitizers in Human Repeated Insult Patch Test (HRIPT) studies. In several validation studies for in vitro phototoxicity assays, histidine was used as a negative control. Neither magnesium aspartate up to 0.5% nor 1% tyrosine was phototoxic in assays using yeast.
Irritation
Dermal
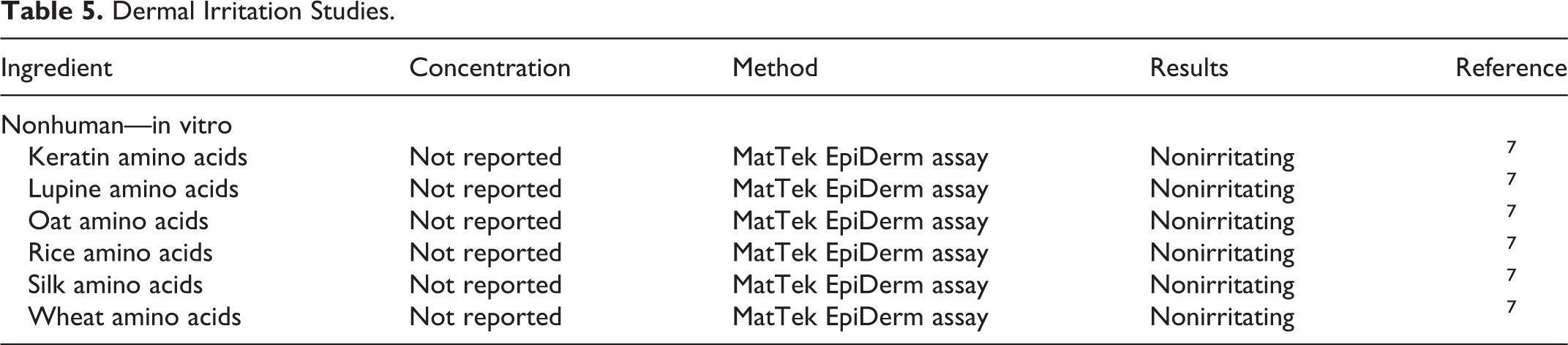
Nonhuman in vitro irritation studies are presented in Table 5. In these studies, amino acids derived from keratin, lupine, oat, rice, silk, and wheat sources were not dermal irritants.
Dermal Irritation Studies.
Ocular
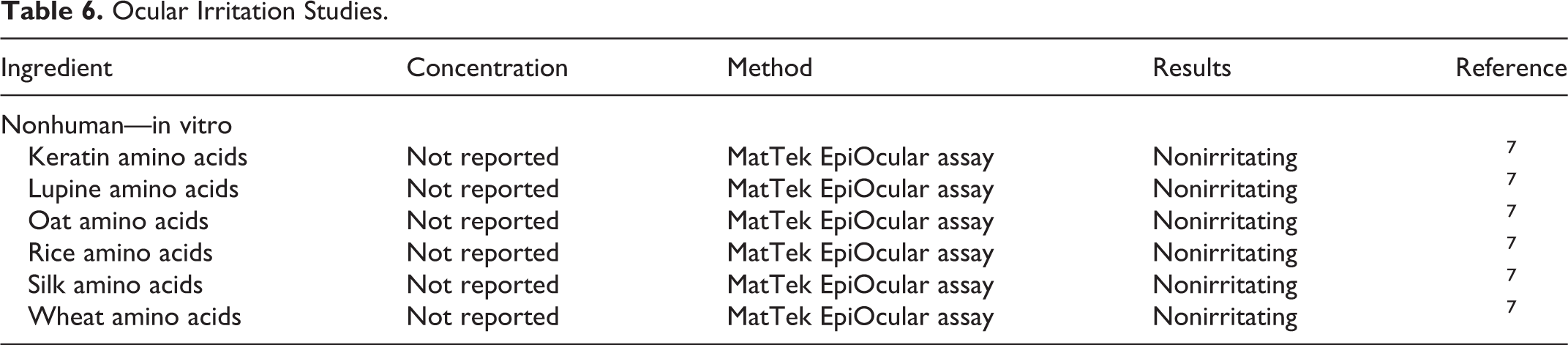
Nonhuman in vitro ocular irritation studies are presented in Table 6. In these studies, amino acids derived from keratin, lupine, oat, rice, silk, and wheat sources were not ocular irritants.
Ocular Irritation Studies.
Sensitization
No published sensitization studies were available for animal- and plant-derived amino acids and no unpublished data were provided.
Phototoxicity
No published phototoxicity studies were available for animal- and plant-derived amino acids and no unpublished data were provided.
Summary
Amino acids derived from animal and plant sources function primarily as skin and hair conditioning agents in personal care products. While the expectation is that most animal- and plant-derived amino acids will be produced by acid hydrolysis of proteins from the relevant source, enzymatic cleavage has been reported to be used.
Of the animal- and plant-derived amino acids, amino acids from silk have the most reported uses in cosmetic and personal care products, with a total of 328; the maximum use concentration range was reported to be 8.0 × 10−5% to 0.2%. The reported functions, product categories, and use concentrations of the animal- and plant-derived amino acid ingredients were comparable to those of the α-amino acid ingredients.
Many of the proteins that serve as the sources of the amino acids that are described in this safety assessment are found in the foods consumed daily. Toxicities from dermal exposure would not be expected to be different from oral exposures and, as such, are not concerning to the CIR Expert Panel. Irritation and sensitization are of concern and are the focus in this report. Data from the previous safety assessment on α-amino acids support that these ingredients would not likely be irritants or sensitizers. Additionally, nonhuman in vitro dermal and ocular irritation studies determined that amino acids derived from keratin, lupine, oat, rice, silk, and wheat sources were not irritants.
Discussion
The Panel acknowledged that the safety of α-amino acids as direct food additives has been well supported based on extensive research through acute and chronic dietary exposures. The Panel determined that this body of research, coupled with the available irritation and sensitization data and use concentrations much lower than those consumed daily in the diet, provides a sufficient basis for determining the safety of amino acids in cosmetic products.
The Panel recognized that there are issues, that is, MSG symptom complex and phenylketonuria, with sodium glutamate and phenylalanine, respectively, in the diet for certain individuals. However, the Panel concluded that the concentrations of these amino acids in cosmetic products are low, and would not be conducive to significant absorption through dermal application or incidental ingestion, and thus, would not cause systemic reactions in individuals.
While the International Cosmetic Dictionary and Handbook does not distinguish among the α-amino acids used in cosmetics that are
The Panel discussed the issue of incidental inhalation exposure from hair sprays, body and hand sprays, aftershave lotion sprays, and suntan sprays. No inhalation data were available. These ingredients reportedly are used at concentrations up to 0.02% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects. The Panel considered other data available to characterize the potential for animal- and plant-derived amino acids to cause systemic toxicity, irritation, sensitization, or other effects. They noted that numerous studies and reviews have been published in the literature regarding the safety of dietary exposure to amino acids, including studies on oral acute and chronic toxicity, carcinogenicity, and genotoxicity, which found no safety concerns for these substances in the amounts at which they are consumed in flavoring agents. Additionally, little or no irritation was observed in multiple tests of dermal and ocular exposure. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel expressed concern about the dangers inherent in using animal-derived ingredients, namely the transmission of infectious agents. They stressed that these ingredients must be free of detectible pathogenic viruses or infectious agents (eg, bovine spongiform encephalopathy). These ingredients should be produced according to current good manufacturing practices and should conform to regulations for producing substances from animal-derived materials.
The Panel also expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. Because the plant proteins from which plant-derived amino acids are produced are extensively processed, it is unlikely that these impurities would remain.
The Panel noted the uncertainty regarding method of manufacturing. The Panel considered that vigorous acid hydrolysis would yield amino acids with little or no residual peptides, because such acid hydrolysis attacks all the peptide bonds in a protein in a nonpreferential manner. The Panel was concerned that enzymatic hydrolysis may fail to completely hydrolyze the source proteins and, thus, may produce di-, tri-, or other peptides, as well as amino acids. Such residual peptide impurities may have the potential to cause allergic reactions in sensitive individuals. Further input on the usual methods used to produce amino acids from animal and plant proteins would be useful. If the methodology involves rigorous acid hydrolysis, there is less concern that residual small peptides will be present. In the absence of further clarification, the Panel stated that industry should manufacture animal- and plant-derived amino acids in a way that ensures that no residual peptides remain.
Conclusion
The CIR Expert Panel concluded that the animal- and plant-derived amino acids listed are safe in the present practices of use and concentration in cosmetics. The 21 ingredients included in this safety assessment are as follows: apricot kernel amino acids* collagen amino acids corn gluten amino acids* elastin amino acids*
Garcinia mangostana amino acids* hair keratin amino acids jojoba amino acids* keratin amino acids lupine amino acids
L barbarum amino acids* milk amino acids oat amino acids rice amino acids sesame amino acids* silk amino acids soy amino acids Spirulina amino acids* sweet almond amino acids* vegetable amino acids wheat amino acids yeast amino acids*
*Not reported to be in current use. Were ingredients in this group not reported to be in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Author Contributions
C. Burnett contributed to conception and design, acquisition, analysis, and interpretation; drafted the article; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. B. Heldreth contributed to conception and design, acquisition, analysis, and interpretation; drafted the article; critically revised the article; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. L. Gill, W. Bergfeld, D. Belsito, R. Hill, C. Klaassen, D. Liebler, J. Marks, R. Shank, T. Slaga, and P. Snyder contributed to conception and design, analysis and interpretation; critically revised the article; gave final approval; and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1620 L Street, NW, Suite 1200, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.