Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the safety of α-amino acids, which function primarily as hair- and skin-conditioning agents in cosmetic products. The safety of α-amino acids as direct food additives has been well established based on extensive research through acute and chronic dietary exposures. The Panel focused its review on dermal irritation and sensitization data relevant to the use of these ingredients in topical cosmetics. The Panel concluded that α-amino acids were safe as cosmetic ingredients in the practices of use and concentration of this safety assessment.
Introduction
Amino acids and their salts are widely used as cosmetic ingredients and function primarily as hair-conditioning agents and skin-conditioning agents (humectant and miscellaneous).
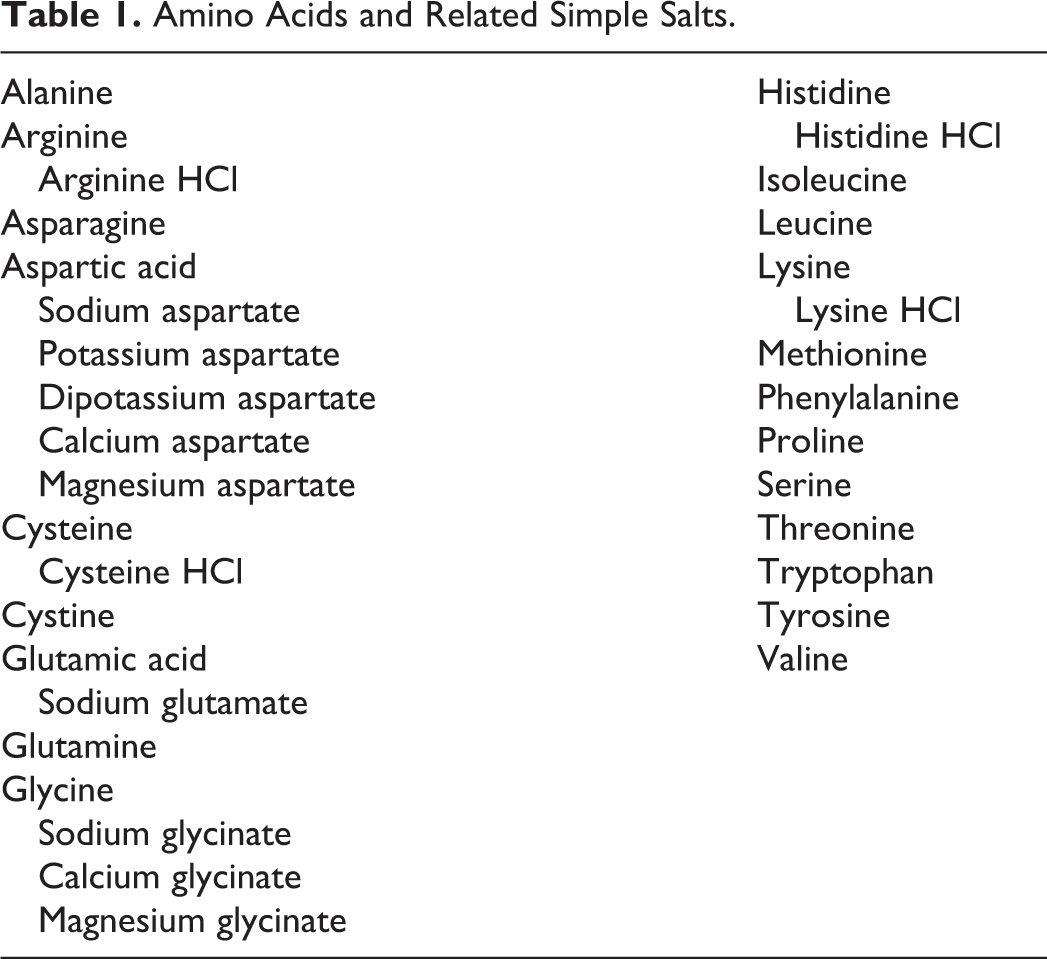
The 21 most common naturally occurring amino acids are building blocks of proteins. As such, amino acids are critical to life and metabolic function. Eight of these amino acids, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine, are essential and must be obtained through nutrients, as they cannot be synthesized by human cells. Because amino acids are present in all living organisms and their general biology is well characterized, they are not considered to pose any significant safety concern following oral exposure, except to individuals with certain genetic disorders. Accordingly, this safety assessment focuses on the basic chemistry, uses as cosmetic ingredients, and, because of the importance for products that will be applied to the skin, addresses all irritation and sensitization data available on these 21 amino acids and their simple salts. The full list of the ingredients in this report is found in Table 1.
Amino Acids and Related Simple Salts.
The naturally occurring protein amino acids are all α-amino acids and, with the exception of glycine and methionine, have
Monosodium glutamate (MSG) has been reported to be associated with a human condition known as “MSG symptom complex” in which symptoms such as a burning sensation of the face, neck, and chest; headache; and nausea occur after consumption of large amounts of this amino acid salt in some foods.1–3
A rare genetic disorder, phenylketonuria, caused by a mutation in the gene that encodes phenylalanine hydroxylase, prevents affected individuals from converting phenylalanine to tyrosine. 4 If this disorder is not detected by blood testing during the first few days after birth and proper dietary management is not observed, irreversible neurological effects may occur.
Chemistry
The definition and structure of these ingredients are presented in Table 2, and available information on the physical and chemical properties of amino acids and their salts is presented in Table 3.
Definitions, Functions, and Idealized Structures of α-amino Acid Ingredients in This Safety Assessment.a
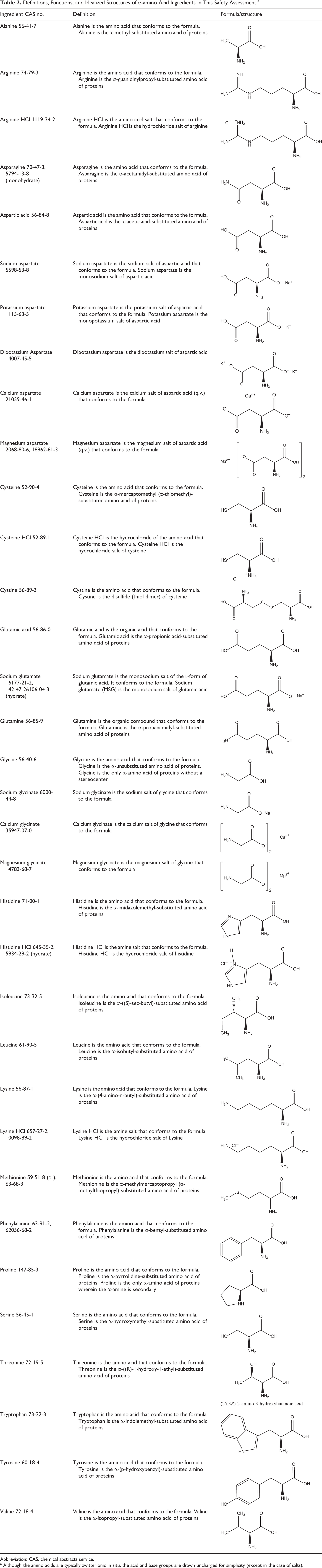
Abbreviation: CAS, chemical abstracts service.
a Although the amino acids are typically zwitterionic in situ, the acid and base groups are drawn uncharged for simplicity (except in the case of salts).
Physical and Chemical Properties.

Abbreviation: alc, alcohol; dil, dilute; soln, solution.
The generic term “amino acid” is commonly considered shorthand for α-amino acid. This designates a carboxylic acid with an amine group on the immediately adjacent (α) carbon as shown in Figure 1.
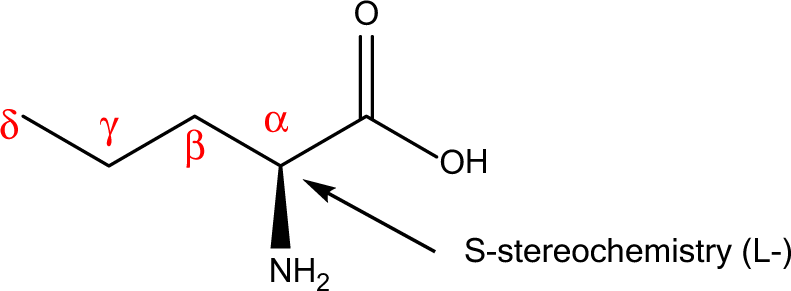
General structure for α-amino acids (see Discussion section).
The “natural” amino acids present in proteins are all α-amino acids, with S-stereochemistry at the α-carbon, except glycine and cysteine. Glycine is achiral (no stereochemistry).
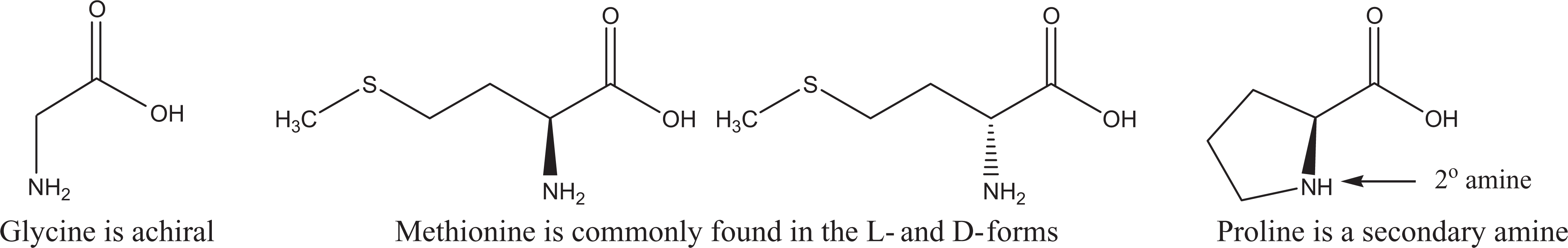
Exceptions in natural amino acid chirality and amine substitution—glycine, methionine, and proline.
Cysteine and cystine are related as thiol (reduced monomer) and the disulfide (oxidized dimer) forms of the same structure. These 2 molecules play major roles in reversible cellular redox chemistry as shown in Figure 3 and can serve a similar function in hairdressings, such as permanent waves.
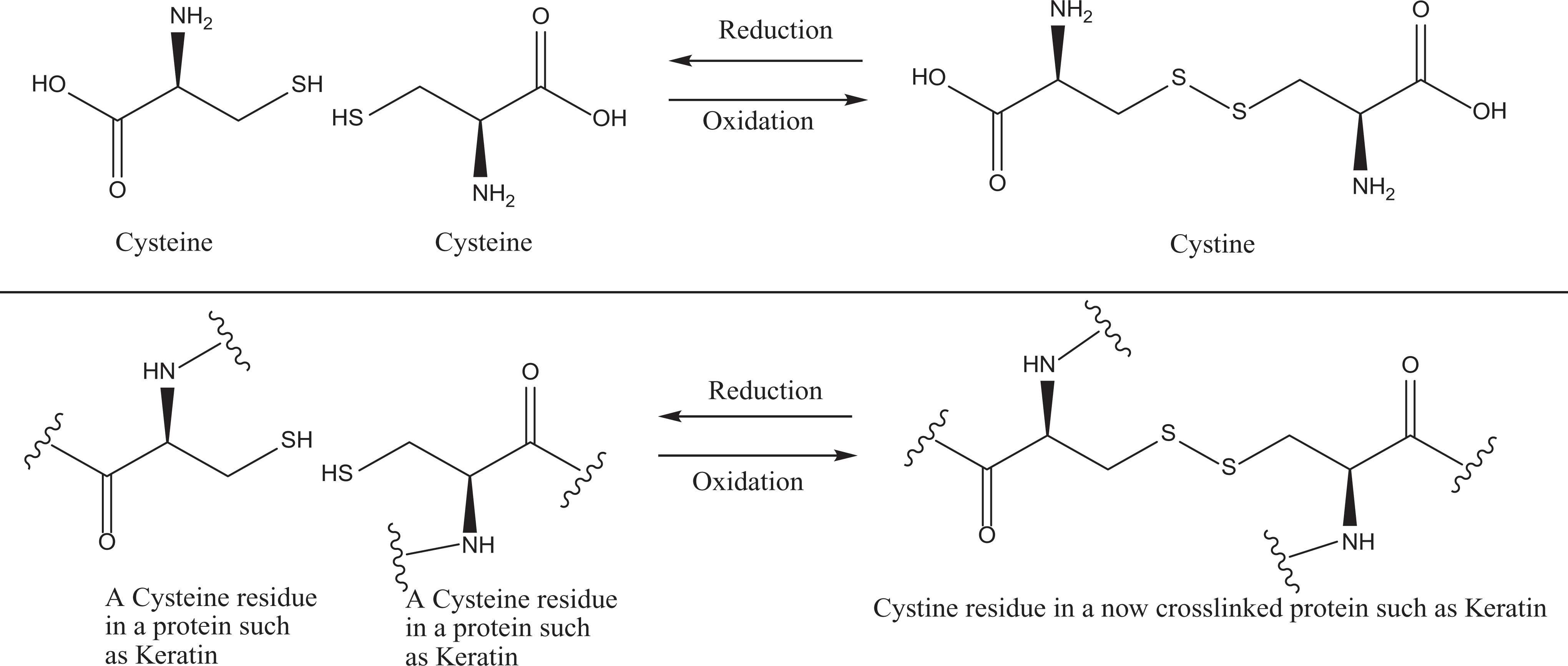
Cysteine/cystine redox chemistry.
Method of Manufacturing
The most common manufacture methodology for glutamine, histidine, leucine, isoleucine, proline, serine, arginine, tryptophan, phenylalanine, threonine, glutamic acid, and lysine is fermentation. 5 This method utilizes bacterial strains that overproduce and release, extracellularly, the desired amino acids during carbohydrate metabolism. Cell separation and crystallization of the amino acids remove any concern of residual organisms or proteins in the end product. For example, glutamate can be produced in a fermentation tank charged with a culture medium including sugar and a culture of Corynebacterium glutamicum. The extracellularly released glutamate is then separated from the biomass and crystallized.
Alanine, methionine, valine, and aspartic acid, on the other hand, are most commonly manufactured via enzymatic catalysis. 5 This method utilizes active cell components in continuously operating reactors. Often, these biocatalysts can be immobilized to provide more efficient separations. This methodology also involves separation techniques that negate any concern of organism or protein contamination in the end product. For example, methionine can be produced in an enzyme membrane reactor with an acylase from Aspergillus oryzae and easily crystallized from the reaction mixture.
Cystine (and cysteine by reduction of cystine), asparagine, and tyrosine, however, are primarily obtained by extraction from the complete hydrolysis of proteins. 5 Glycine, the achiral amino acid, is typically synthesized from chloroacetic acid and ammonia.
Impurities
A supplier to the cosmetics industry reported that
According to the Food Chemicals Codex, United States Pharmacopeia (USP)-grade amino acids (“
Use
Cosmetic
The amino acids and the salts discussed in this safety assessment function primarily as hair-conditioning agents and skin-conditioning agents (humectant and miscellaneous) in cosmetic formulations. 8 Additional functions may include the use as oral care agents (arginine), antioxidants (cysteine and cysteine HCl), hair waving/straightening agents (cysteine and cysteine HCl), reducing agents (cysteine and cysteine HCl), fragrance ingredients (cystine), buffering agents (glycine and its calcium and magnesium salts), and pH adjusters (glycine and its calcium and magnesium salts).
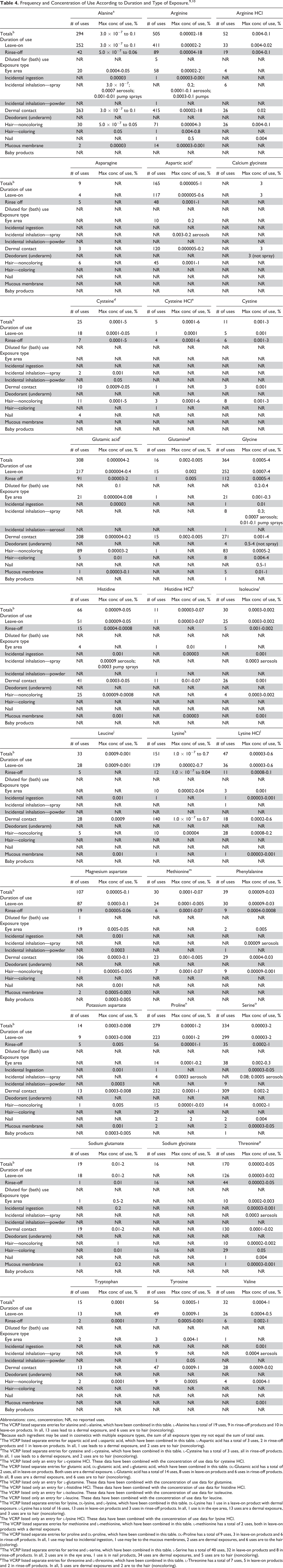
Table 4 presents the frequency and maximum use concentration ranges for amino acids. 9,10 According to information supplied to the FDA by industry as part of the Voluntary Cosmetic Registration Program (VCRP), arginine has the most reported uses in cosmetic and personal care products, with a total of 505 uses; 411 of those uses are in leave-on formulations. 9 Glycine has the second greatest number of overall uses reported, with a total of 364; 252 of those uses are in leave-on formulations. No uses were reported to the VCRP for sodium aspartate, dipotassium aspartate, calcium aspartate, calcium glycinate, or magnesium glycinate. In a recent survey of use concentrations, arginine had a maximum use concentration range of 2.0 × 10−5% to 18%, with the 18% reported in paste masks and mud packs. 10 Glycine had a maximum use concentration range of 5.0 × 10−4% to 4%, with the 4% reported in hair dyes and nonspray deodorants. No use concentrations were reported for asparagine, sodium aspartate, dipotassium aspartate, calcium aspartate, sodium glycinate, and magnesium glycinate. In some cases, reports of uses were received in the VCRP, but no concentration of use data were available. For example, asparagine is reported to be used in 9 formulations, but no use concentration data were available. In other cases, no reported uses were received in the VCRP, but a use concentration was provided in the industry survey. For example, calcium glycinate was not reported in the VCRP to be in use, but the industry survey indicated that it is used in leave-on formulations at up to 3%. It should be presumed that calcium glycinate is used in at least one cosmetic formulation.
Abbreviations: conc, concentration; NR, no reported uses.
aThe VCRP listed separate entries for alanine and
bBecause each ingredient may be used in cosmetics with multiple exposure types, the sum of all exposure types my not equal the sum of total uses.
cThe VCRP listed separate entries for aspartic acid and
dThe VCRP listed separate entries for cysteine and
eThe VCRP listed only an entry for
fThe VCRP listed separate entries for glutamic acid,
gThe VCRP listed only an entry for
hThe VCRP listed only an entry for
iThe VCRP listed only an entry for
jThe VCRP listed only an entry for
kThe VCRP listed separate entries for lysine,
lThe VCRP listed only an entry for
mThe VCRP listed separate entries for methionine and
nThe VCRP listed separate entries for proline and
oThe VCRP listed separate entries for serine and
pThe VCRP listed separate entries for threonine and
Products containing amino acids are reported to be used on baby skin, may be applied to the eye area or mucous membranes, or could be incidentally ingested. Additionally, amino acids are used in cosmetic sprays, including hair and other propellant and pump spray products, and could possibly be inhaled. The maximum concentration of amino acids reported to be used in a spray product is 0.3% glycine in a face spray. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays.11,12 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, able to enter the lungs) to any appreciable amount.13,14
Noncosmetic
The “
In addition to food additives and supplements, amino acids also may be used in the production of pesticides as source materials of chemical synthesis.15,16
Toxicological Studies
The amino acids in this assessment are found in foods, and the daily exposure from food use would result in a much larger systemic dose than that resulting from use in cosmetic products. Numerous studies and reviews have been published in the literature about the safety of dietary exposure to amino acids, including a review by the Joint Food and Agriculture Organization of the United Nations/World Health Organization Expert Committee on Food Additives that summarized studies on oral acute and chronic exposure/carcinogenicity studies and genotoxicity and found no safety concerns for these substances at the amounts they are used in flavoring agents.
17
Also, as noted earlier, the “
Enzyme Regulation
In an in vitro study using human keratinocytes, continuous application of arginine (
Irritation and Sensitization
Irritation
Dermal
Non-human irritation studies are presented in Table 5. These data show tested ingredients to be nonirritating.
Dermal Non-Human Irritation Studies.

Ocular
Non-human and human ocular irritation studies are presented in Table 6. The findings range from nonirritating to moderately irritating.
Ocular Irritation Studies.
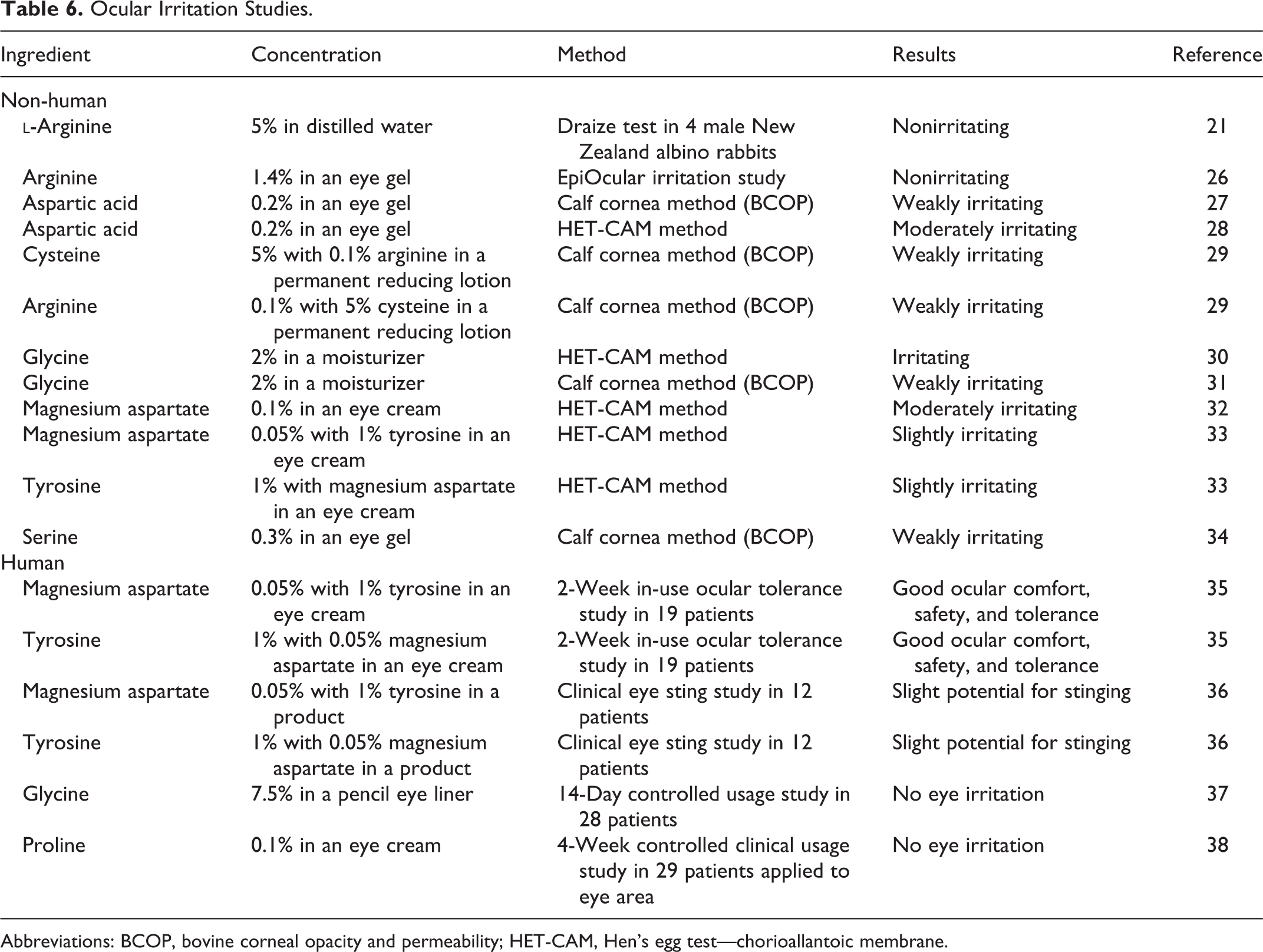
Abbreviations: BCOP, bovine corneal opacity and permeability; HET-CAM, Hen’s egg test—chorioallantoic membrane.
Sensitization
Non-human and human sensitization studies are presented in Table 7. No irritation was reported during induction, and no sensitization was reported at challenge.
Dermal Sensitization Studies.
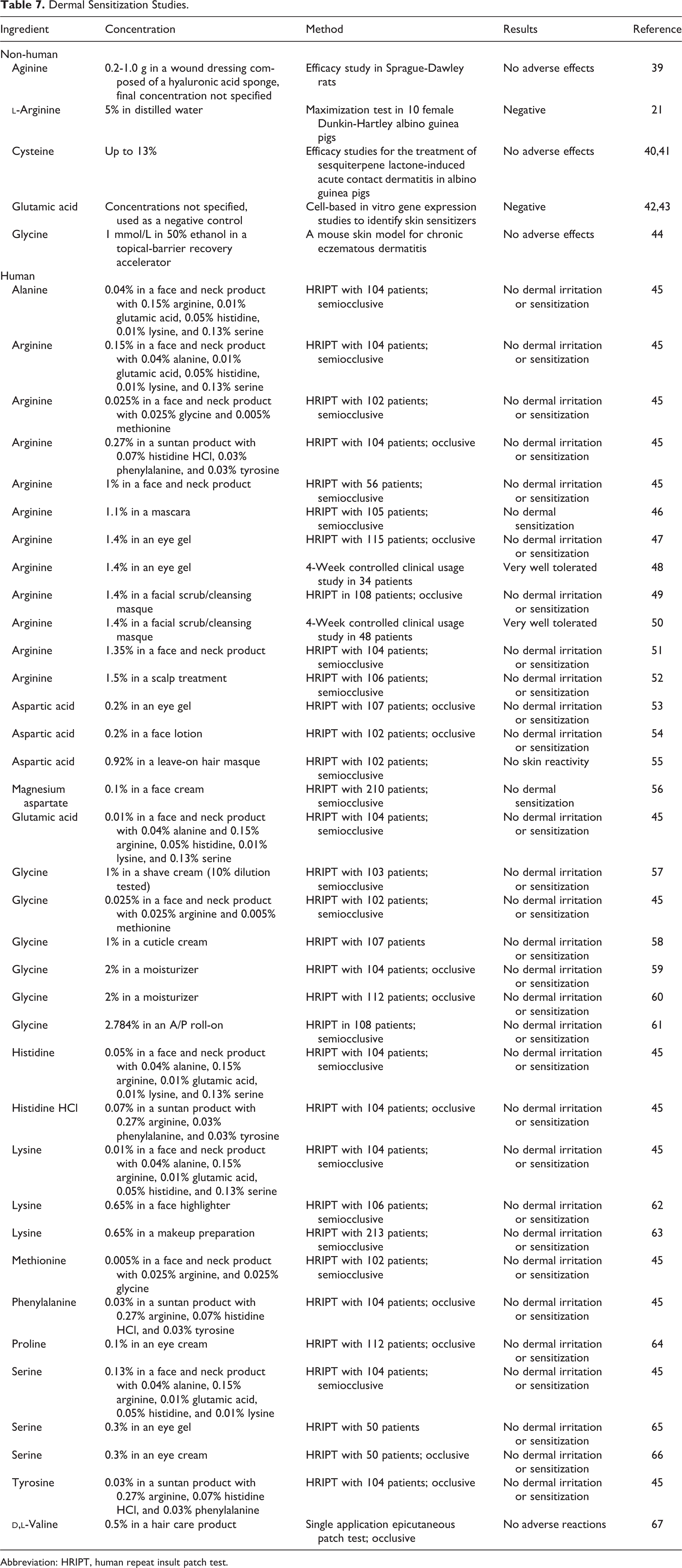
Abbreviation: HRIPT, human repeat insult patch test.
Phototoxicity
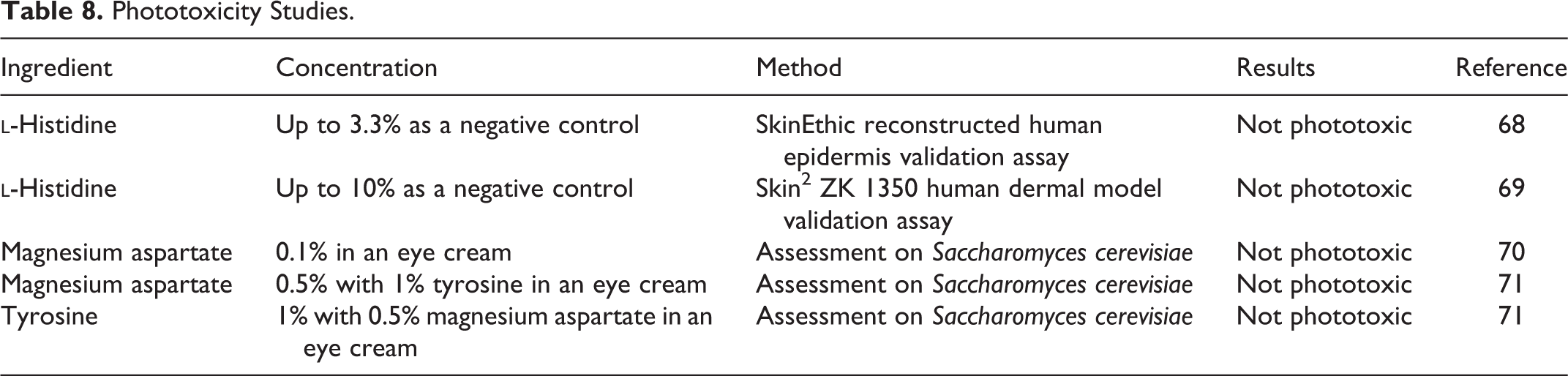
Phototoxicity studies are presented in Table 8. No phototoxicity was reported.
Phototoxicity Studies.
Summary
Amino acids are critical to life and metabolic function. Because amino acids are present in all living organisms, they are not considered to pose any significant safety concern following oral exposure except to individuals with certain genetic disorders, and their general biology is well characterized.
The amino acids and their salts are used primarily as hair-conditioning agents and skin-conditioning agents in cosmetic formulations. Arginine has the most reported uses in cosmetic and personal care products, with a total of 440. The maximum use concentration range for arginine is 2.0 × 10−5% to 18%. Glycine has the second greatest number of overall uses reported, with a total of 323, and has a maximum use concentration range of 5.0 × 10−4% to 4%. The maximum concentration of amino acids reported to be used in a spray product is 0.3% glycine in a face spray.
The “
An in vitro study using human keratinocytes found that continuous application of arginine (
Cysteine HCl and methionine were used as negative controls in in vitro assays to predict potential skin irritants.
In separate efficacy studies, arginine, cysteine, and glycine did not produce any adverse effects in rats, guinea pigs, or mouse skin models. Glutamic acid was used as a negative control in an in vitro study to identify skin sensitizers.
Human repeat insult patch test studies of many products containing amino acid ingredients concluded that products containing these ingredients were not dermal irritants or sensitizers.
In several validation studies for in vitro phototoxicity assays, histidine was used as a negative control. Magnesium aspartate up to 0.5% and 1% tyrosine were not phototoxic in assays using yeast.
Discussion
The Cosmetic Ingredient Review Expert Panel (Panel) acknowledged that the safety of α-amino acids as direct food additives has been well established based on extensive research through acute and chronic dietary exposures. The Panel determined that this body of research, coupled with the available irritation and sensitization data and use concentrations that are at levels much lower than those consumed daily in the diet, were a sufficient basis for determining the safety of α-amino acids in cosmetic products.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, face and neck sprays, and suntan sprays. No inhalation data were available. These ingredients reportedly are used at concentrations up to 0.3% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects. The Panel considered other data available to characterize the potential for α-amino acids to cause systemic toxicity, irritation, sensitization, or other effects. They noted that numerous studies and reviews have been published in the literature regarding the safety of dietary exposure to amino acids, including studies on oral acute and chronic toxicity, carcinogenicity, and genotoxicity, which found no safety concerns for these substances in the amounts at which they are consumed in flavoring agents. Additionally, little or no irritation or sensitization was observed in multiple tests of dermal and ocular exposure. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel recognized that there are issues with sodium glutamate and phenylalanine in the diet for certain individuals. However, the Panel determined that the concentrations of these amino acids in cosmetic products are at levels that would not be significantly absorbed through topical application or incidental ingestion and, thus, would not cause systemic reactions in individuals.
Although the International Cosmetic Dictionary and Handbook does not distinguish among the α-amino acids used in cosmetics that are
Conclusion
The Panel concluded that α-amino acids, listed below, are safe in the present practices of use and concentration in cosmetics.
Alanine
Arginine
Arginine HCl
Asparagine
Aspartic Acid
Calcium Aspartate*
Calcium Glycinate
Cysteine
Cysteine HCl
Cystine
Dipotassium Aspartate*
Glutamic Acid
Glutamine
Glycine
Histidine
Histidine HCl
Isoleucine
Leucine
Lysine
Lysine HCl
Magnesium Aspartate
Magnesium Glycinate*
Methionine
Phenylalanine
Potassium Aspartate
Proline
Serine
Sodium Aspartate*
Sodium Glutamate
Sodium Glycinate
Threonine
Tryptophan
Tyrosine
Valine
If the ingredients in this group not reported in current use (as indicated by *) are to be used in cosmetics in the future, the expectation is that they would be used at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, 1101 17th St, Suite 412, Washington, DC 20036, USA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.