Abstract
The Cosmetic Ingredient Review Expert Panel (Panel) reviewed the product use, formulation, and safety data of 115 amino acid alkyl amides, which function as skin and hair conditioning agents and as surfactants—cleansing agents in personal care products. Safety test data on dermal irritation and sensitization for the ingredients with the highest use concentrations, lauroyl lysine and sodium lauroyl glutamate, were reviewed and determined to adequately support the safe use of the ingredients in this report. The Panel concluded that amino acid alkyl amides are safe in the present practices of use and concentration in cosmetics, when formulated to be nonirritating.
Introduction
This safety assessment summarizes the available data relevant to assessing the safety of 115 amino acid alkyl amides as used in cosmetics. These ingredients mainly function as skin and hair conditioning agents and as surfactants—cleansing agents in personal care products. The list of ingredients in this report is found in Table 1.
Abbreviations: CIR, Cosmetic Ingredient Review; misc, miscellaneous; TEA, triethanolamine.
aThe italicized text represents additions made by CIR staff.
By and large, the ingredients in this report will not rapidly dissociate (beyond zwitterion formation) in the presence of water, but action by amidases is the most likely first step of metabolism if dermal penetration occurs. The relative exposure, hence, would also include amino acids and fatty acids. The Cosmetic Ingredient Review Expert Panel (Panel) previously has reviewed the safety of α-amino acids and animal- and plant-derived amino acids and concluded that these ingredients are safe for use as cosmetic ingredients. 1,2 The Panel also reviewed the following fatty acid constituents and concluded that these fatty acids are safe for use as cosmetic ingredients: coconut acid, olive acid, sunflower seed acid, palm acid, acetic acid, lauric acid, oleic acid, palmitic acid, stearic acid, and myristic acid. 3 –9 The Panel concluded that malic acid was safe for use as a pH adjuster, but the data were insufficient to determine safety for any other functions. 10 The maximum concentrations of use along with summaries of the data included in those existing safety assessments are provided in Table 2.
Constituent Acids With CIR Conclusions.

Abbreviations: CIR, Cosmetic Ingredient Review.
Chemistry
The amino acid alkyl amides in this report are comprised of amino acids acylated with acids or acid chlorides at the amino acid nitrogen to form amides (except for lauroyl lysine, which is formed by acylation at the epsilon nitrogen). For example, capryloyl glycine is the N-acylation product of glycine with caprylic acid chloride.
A likely metabolic pathway for these ingredients includes reactions catalyzed by amidases, should the ingredients penetrate the skin. The net result would be the release of the amino acid (glycine in the example above) and a fatty acid (caprylic acid in the example). The definitions of the amino acid alkyl amides can be found in Table 1, and the structures can be found in Table 3.
Abbreviations: CIR, Cosmetic Ingredient Review; TEA, triethanolamine.
aThe asterisk marked structures represent additions made by CIR staff.
Physical and Chemical Properties
The ingredients in this report are typically water-soluble, waxy solids. Available chemical properties can be found in Table 4.
Chemical Properties of Amino Acids Alkyl Amides.

Method of Manufacturing
As shown in Figure 1, the ingredients in this report are most commonly manufactured by the acylation of a free amine of an amino acid with an acyl chloride, a reaction known as the Schotten-Baumann reaction. 11 –13 A major side product for this reaction is hydrochloric acid, which can be easily removed.
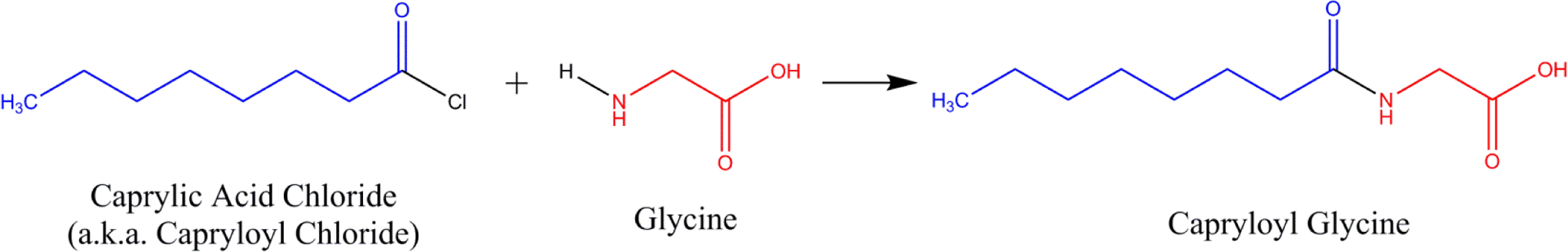
Synthesis of the amino acid alkyl amide, capryloyl glycine.
Disodium capryloyl glutamate, sodium cocoyl glutamate, and sodium lauroyl glutamate
According to a supplier, disodium capryloyl glutamate, sodium cocoyl glutamate, and sodium lauroyl glutamate are produced via the Schotten-Baumann reaction. 14 –16 The supplier also described the origin of starting materials: glutamic acid is obtained through formation of glucose/molasses or from wheat and capryloyl chloride, cocoyl chloride, and lauroyl chloride are obtained from caprylic acid, coconut acid, and lauric acid that come from cleavage and distillation of coconut oil. The respective resultant materials are aqueous solutions comprised of 37% to 41% disodium capryloyl glutamate, 32.6% to 38% sodium cocoyl glutamate, and 36% to 40% sodium lauroyl glutamate.
Sodium lauroyl silk amino acids
A supplier of sodium lauroyl silk amino acids reports that the material is prepared by acylation of a free amine of silk amino acid obtained by silk protein hydrolysis. The final product is a 20% water solution of sodium lauroyl silk amino acids. 17
Impurities
Disodium capryloyl glutamate
A supplier has reported that disodium capryloyl glutamate may contain 4% to 6% propylene glycol, 3% caprylic acid (maximum), 5% disodium glutamate (maximum), and 6% to 8% sodium chloride. 14 Disodium capryloyl glutamate contains <2 ppm arsenic, <5 ppm antimony, <1 ppm lead, <2 ppm cadmium, <2 ppm mercury, <1 ppm nickel, <2 ppm chromium, and <10 ppm total heavy metals (as iron).
Sodium cocoyl glutamate
The same supplier has reported that sodium cocoyl glutamate may contain 4% to 6% propylene glycol, 5% (max) sodium glutamate, 3% coconut acid, and 4% to 5.5% sodium chloride. 16 Sodium cocoyl glutamate contains <2 ppm arsenic, <5 ppm antimony, <1 ppm lead, <2 ppm cadmium, <2 ppm mercury, <1 ppm nickel, <2 ppm chromium, and <10 ppm total heavy metals (as iron).
Sodium lauroyl glutamate
A supplier has reported that sodium lauroyl glutamate may contain 4% to 6% propylene glycol, 5% (maximum) glutamic acid, 3% (maximum) lauric acid, and 3% to 4.5% sodium chloride. 15 Sodium lauroyl glutamate contains <2 ppm arsenic, <5 ppm antimony, <1 ppm lead, <2 ppm cadmium, <2 ppm mercury, <1 ppm nickel, <2 ppm chromium, and <10 ppm total heavy metals (as iron).
Sodium lauroyl silk amino acids
A supplier of sodium lauroyl silk amino acids reports that the material has heavy metals and arsenic ≤20 and ≤2 ppm, respectively. 17
Triethanolamine-containing ingredients
The issue of levels of free diethanolamine (DEA) that could be present as an impurity in the ingredients containing triethanolamine (TEA), and the potential of TEA to act as a precursor in nitrosamine formation by undergoing nitrosative cleavage, has been previously reviewed by the Panel. 18,19
Use
Cosmetic
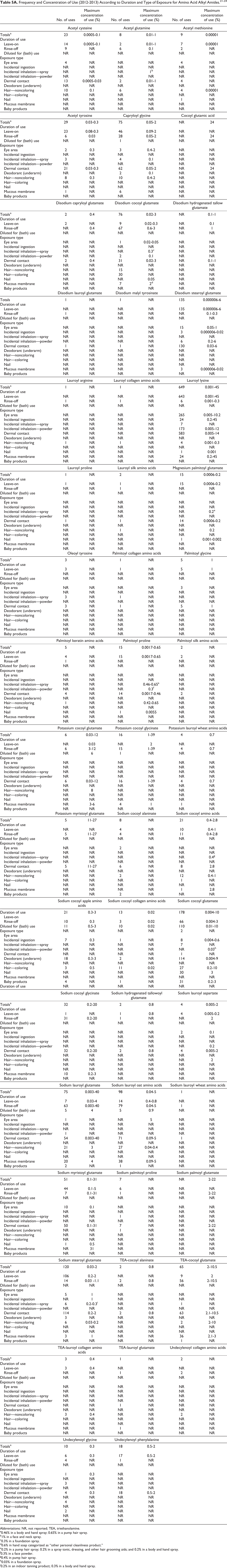
Table 5A contains the current product formulation data for amino acid alkyl amides. These ingredients function primarily as skin and hair conditioning agents and surfactants. 20 The safety of amino acid alkyl amides included in this safety assessment is evaluated based on data received from the US Food and Drug Administration (FDA) and the cosmetic industry on the expected use of these ingredients in cosmetics. Use frequencies of individual ingredients in cosmetics are collected from manufacturers and reported by cosmetic product category in FDA’s Voluntary Cosmetic Registration Program (VCRP) database. Use concentration data are submitted by the industry in response to surveys conducted by the Personal Care Products Council (Council) of maximum reported use concentrations by product category.
Abbreviations: NR, not reported; TEA, triethanolamine.
a0.46% in a body and hand spray; 0.65% in a pump hair spray.
b1% in a face and neck spray.
c0.3% in a foundation spray.
d0.6% in hand soap categorized as “other personal cleanliness product.”
e0.2% in a pump hair spray; 0.2% in a spray tonic, dressing, and other hair grooming aids; and 0.2% in a body and hand spray.
f0.3% in a face powder.
g0.4% in pump hair spray.
h0.03% in a foundation spray.
i0.2% in an indoor tanning product; 0.3% in a body and hand spray.
According to VCRP data, lauroyl lysine has the most reported uses in cosmetic and personal care products, with a total of 649; most uses are in leave-on eye and facial makeup. 21 Sodium cocoyl glutamate has the second greatest number of overall uses reported, with a total of 178; more than half of those uses are in rinse-off products.
In the Council’s use concentration survey, lauroyl lysine had a wide maximum use concentration range of 0.001% to 45%, with the 45% reported in lipsticks. 22 –24 Sodium lauroyl glutamate also had a wide maximum use concentration range of 0.003% to 40%, with the 40% reported in skin cleansing agents. All other use concentrations that were reported had similar ranges.
In some cases, reports of uses were received from the VCRP, but no concentrations of use data were available. For example, palmitoyl keratin amino acids are reported to be used in 5 formulations, but no use concentration data were available. In other cases, no reported uses were received from the VCRP, but a use concentration was provided in the industry survey. For example, cocoyl glutamic acid was not reported in the VCRP database to be in use, but the industry survey indicated that it is used in leave-on formulations at a maximum concentration of 24%. Cocoyl glutamic acid is used presumably in at least 1 cosmetic formulation. Ingredients with no reported uses or use concentrations are listed in Table 5B.
Amino Acid Alkyl Amides Not Reported in Use

Abbreviation: TEA, triethanolamine.
Several of the amino acid alkyl amides described in this report are used in cosmetic sprays, including pump hair, face, and body spray products, foundation spray products, and indoor tanning spray products, and could possibly be inhaled. The maximum concentration of amino acid alkyl amide reported to be used in a spray product is 0.65% palmitoyl proline in a pump hair spray. In practice, 95% to 99% of the droplets/particles released from cosmetic sprays have aerodynamic equivalent diameters >10 µm, with propellant sprays yielding a greater fraction of droplets/particles <10 µm compared with pump sprays. 25,26 Therefore, most droplets/particles incidentally inhaled from cosmetic sprays would be deposited in the nasopharyngeal and bronchial regions and would not be respirable (ie, able to enter the lungs) to any appreciable amount. 27,28
In the European Union, trialkylamines, trialkanolamines, and their salts (ingredients containing TEA) may only be used up to 2.5%, must be at least 99% pure, are not to be used with nitrosating systems, must have ≤5% secondary amine content and ≤50 µg/kg nitrosamine, and must be kept in nitrite-free containers. 29 The use of the remaining ingredients is not restricted under the rules governing cosmetic products in the European Union.
Noncosmetic
Amino acid alkyl amides are used in household detergents. 30 Acetyl cysteine has been approved by the FDA to treat acetaminophen overdose via intravenous injection and for mucolytic therapy. 31 Acetyl methionine is an approved direct food additive (21 CFR §172.372).
Toxicokinetics
Absorption, Distribution, Metabolism, and Excretion
Acetyl tyrosine
A percutaneous absorption study of 3 formulations containing 1.75% acetyl tyrosine was performed in vitro on human trunk skin using the finite dose technique and Franz diffusion cells. 32 The formulations were a gel, a cream, and a water solution in silicone. Each formulation was evaluated on 3 replicate sections from 2 different donors of ex vivo human trunk skin. At dosing, 10 mg formulation/cm2/skin section equivalent volume was dispensed by pipette and a glass rod was used to evenly distribute the formulation into the skin. The percutaneous absorption of the test material was determined over a 48-hour dose period. At 6, 12, 32, and 48 hours after application, the dermal receptor solution was removed in its entirety, replaced with stock receptor solution, and 4 mL aliquot was saved for subsequent analysis. After the last receptor solution collection, the skin surface was washed twice with 50:50 methanol:water to collect unabsorbed formulation from the skin. The glass rod used for dosing, the surface wash, stratum corneum, epidermis, and dermis were recovered and evaluated for compound content. The samples were analyzed for test material content using high-performance liquid chromatography method.
In the formulation with water, the test material was found in the following mean distribution: 0.48% in the receptor solution, 0.04% in the dermis, 1.25% in the epidermis, 4.64% in the stratum corneum, and 83.15% in the surface wash (total recovery was 89.55%). For the gel formulation, the test material was found in the following mean distribution: 1.03% in the receptor solution, 0.07% in the dermis, 1.15% in the epidermis, 0. 70% in the stratum corneum, and 88.59% in the surface wash (total recovery was 91.53%). Finally, in the cream formulation, the test material was found in the following mean distribution: 2.70% in the receptor solution, 0.39% in the dermis, 15.96% in the epidermis, 11.91% in the stratum corneum, and 54.34% in the surface wash (total recovery was 85.30%). The authors of the study concluded that acetyl tyrosine in all 3 formulations evaluated does penetrate into and through ex vivo human skin using the in vitro finite dose. 32
Toxicological Studies
The amino acid alkyl amides in this assessment will not rapidly dissociate (beyond zwitterion formation) in the presence of water, but action by amidases is the most likely first step of metabolism if dermal penetration occurs. Exposure to these ingredients, hence, would also involve exposures to amino acid and fatty acid metabolites of these ingredients. Because most of these amino acids and fatty acids are found in the foods we consume daily, systemic toxicity is not expected. Systemic toxicity following dermal exposure is not expected to differ from that of oral exposure. This report is focused on irritation and sensitization. Although most surfactants have the potential for some irritation, data from the previous safety assessments on α-amino acids and fatty acids support that these ingredients, as used in cosmetics, would not likely be irritants or sensitizers.
Reproductive and Developmental Toxicity
Acetyl Cysteine
In teratology studies, no evidence of impaired fertility or toxicity to fetuses was observed in rats at oral doses up to 2,000 mg/kg/d acetyl cysteine and in rabbits at oral doses up to 1,000 mg/kg/d acetyl cysteine. 33 No further details were provided.
Male rats treated orally with 250 mg/kg/d acetyl cysteine for 15 weeks did not experience adverse effects to fertility or reproductive performance. 33 No further details were provided.
Genotoxicity
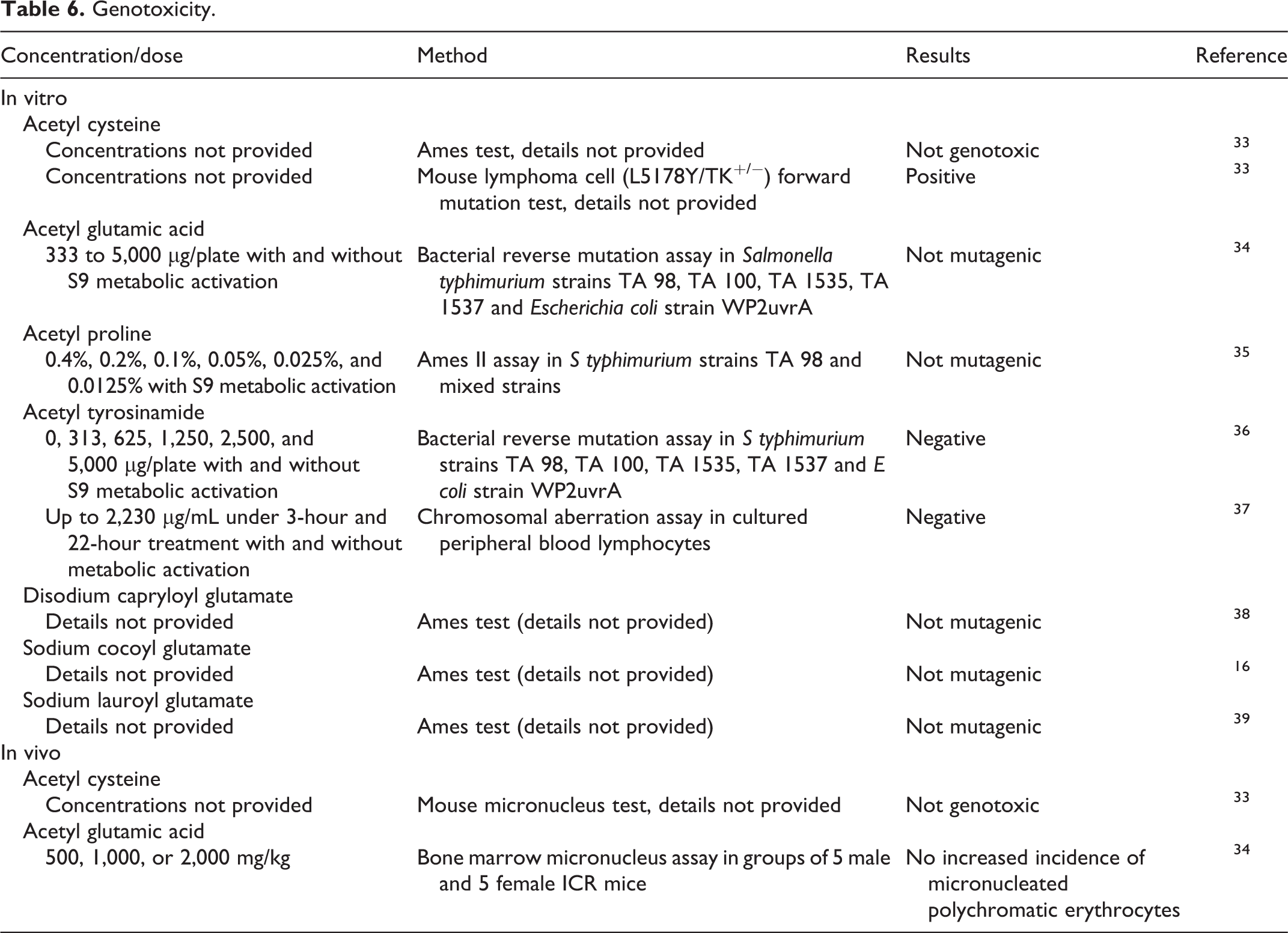
In vitro and in vivo genotoxicity studies are presented in Table 6. In in vitro studies, acetyl glutamic acid, acetyl proline, acetyl tyrosine, disodium capryloyl glutamate, sodium cocoyl glutamate, and sodium lauroyl glutamate were negative for genotoxicity. Acetyl cysteine was not genotoxic in an Ames test but had positive results in in vitro mouse lymphoma test. Acetyl cysteine and acetyl glutamic acid were negative in in vivo mouse studies. 16,33 –39
Genotoxicity.
Carcinogenicity
No published carcinogenicity studies on amino acid alkyl amides were identified by a literature search for these ingredients and no unpublished data were submitted.
Irritation and Sensitization
Data from a previous CIR safety assessment of α-amino acids 2 indicate that cysteine HCl and methionine were used as negative controls in in vitro assays to predict potential skin irritants; arginine (up to 5%), aspartic acid (up to 0.2%), cysteine (up to 5%), glycine (up to 2%), magnesium aspartate (up to 0.1%), serine (up to 0.3%), and tyrosine (up to 1%) did not produce any adverse effects in rats, guinea pigs, or mouse skin models in separate dermal and ocular studies; and glutamic acid was used as a negative control in an in vitro study to identify skin sensitizers. The data also showed that products containing amino acid ingredients at concentrations up to 2.784% were not dermal irritants or sensitizers in HRIPT studies, and in several validation studies for in vitro phototoxicity assays, histidine was used as a negative control. Neither magnesium aspartate up to 0.5% nor 1% tyrosine was phototoxic in assays using yeast.
Irritation
Dermal
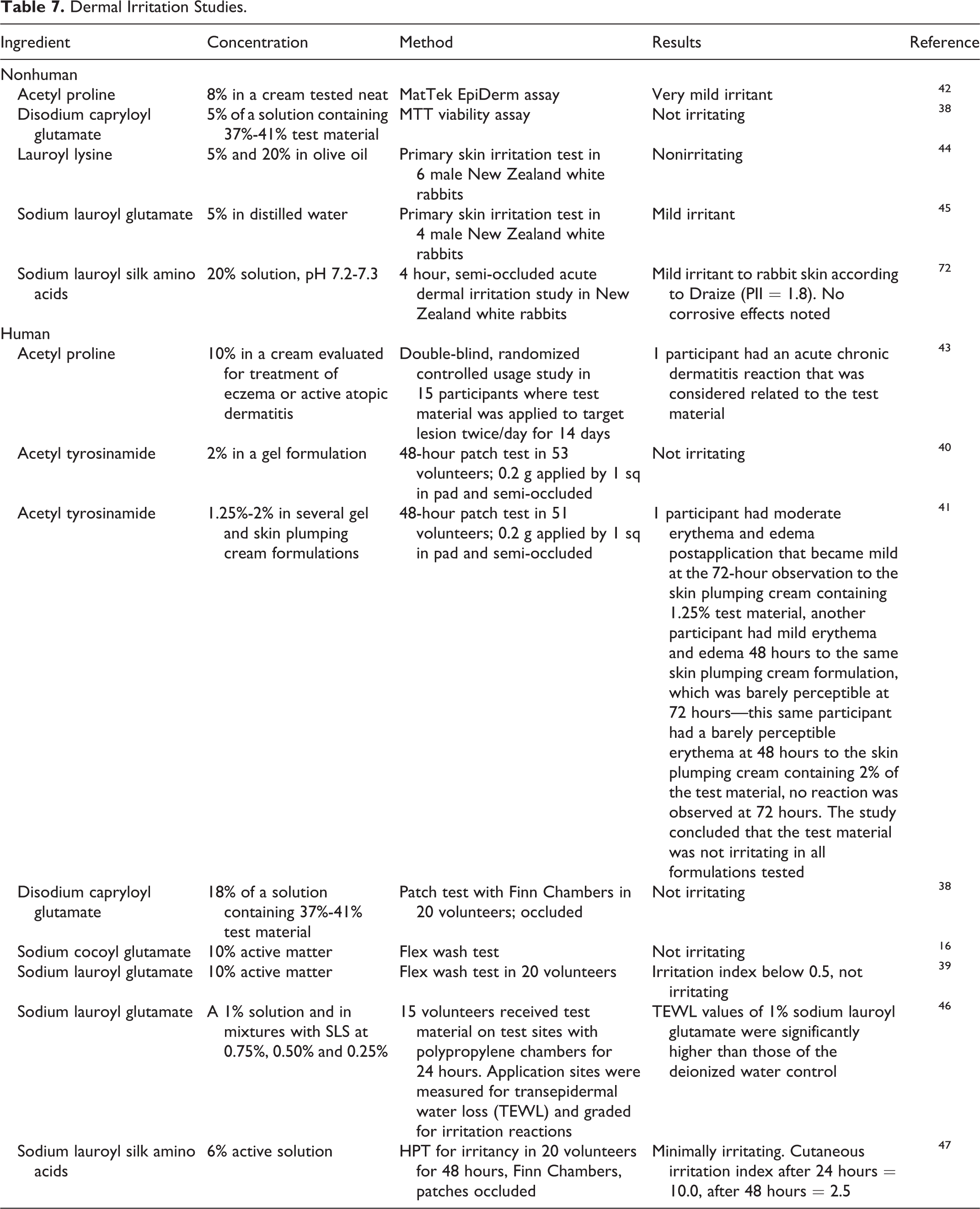
In vitro and human dermal irritation studies are presented in Table 7. No irritation was observed in in vitro studies with disodium capryloyl glutamate when tested at concentrations of 1.85% to 2.05%. A cream containing 8% acetyl proline was a mild irritant in another in vitro study. In rabbits, lauroyl lysine was nonirritating when tested at a concentration of 20%, whereas sodium lauroyl glutamate was a mild irritant at a concentration of 5%. In human studies, acetyl proline (up to 10%), acetyl tyrosine (up to 2%), disodium capryloyl glutamate (up to 7.38%), sodium cocoyl glutamate (up to 10%), sodium lauroyl glutamate (up to 10%), and sodium lauroyl silk amino acids (up to 6%) were not dermal irritants. 16,38 –47
Dermal Irritation Studies.
Ocular
Nonhuman in vitro and in vivo and human ocular irritation studies are presented in Table 8. No ocular irritation was observed in in vitro studies of acetyl tyrosine (up to 1.25%), disodium capryloyl glutamate (up to 2%), and sodium lauroyl glutamate (up to 5%). Severe irritation was observed with sodium cocoyl glutamate at 5% using the hen’s egg test chorioallantoic membrane (HET-CAM) method, but it was not irritating in another study with an unknown concentration. Slight irritation was observed with sodium lauroyl silk amino acids at 0.5% using the HET-CAM method. Lauroyl arginine in a mixture at 10% was not irritating to rabbit eyes. No adverse effects were observed during in-use studies of eye-area products containing acetyl hydroxyproline (up to 2%) and acetyl tyrosine (up to 2%) in human participants. 15,16,38,39,48 –55
Ocular Irritation Studies.
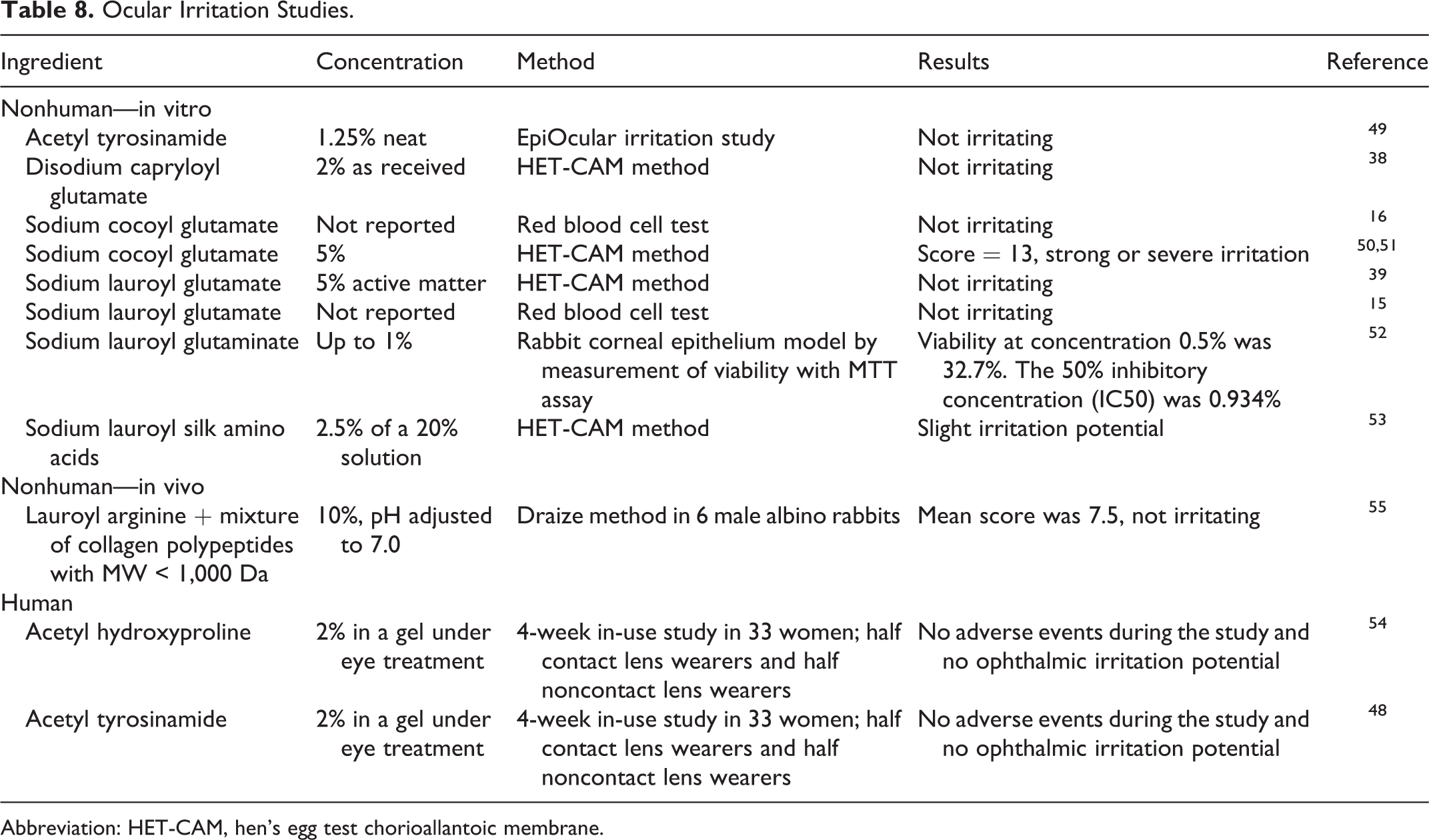
Abbreviation: HET-CAM, hen’s egg test chorioallantoic membrane.
Sensitization
Nonhuman and human dermal sensitization studies are presented in Table 9. Sodium lauroyl silk amino acids when tested in a 20% solution were not sensitizing in an LLNA. In guinea pigs, lauroyl lysine and sodium lauroyl glutamate were not sensitizers at concentrations of 50% and 5%, respectively. No sensitization was observed in human studies with products containing acetyl hydroxyproline (up to 2%), acetyl proline (up to 10%), acetyl tyrosine (up to 2%), disodium capryloyl glutamate (up to 7.38%), lauroyl lysine (up to 12.5%), sodium cocoyl glutamate (up to 5%), and sodium lauroyl glutamate (up to 5%). 16,38,39,56 –66
Dermal Sensitization Studies.
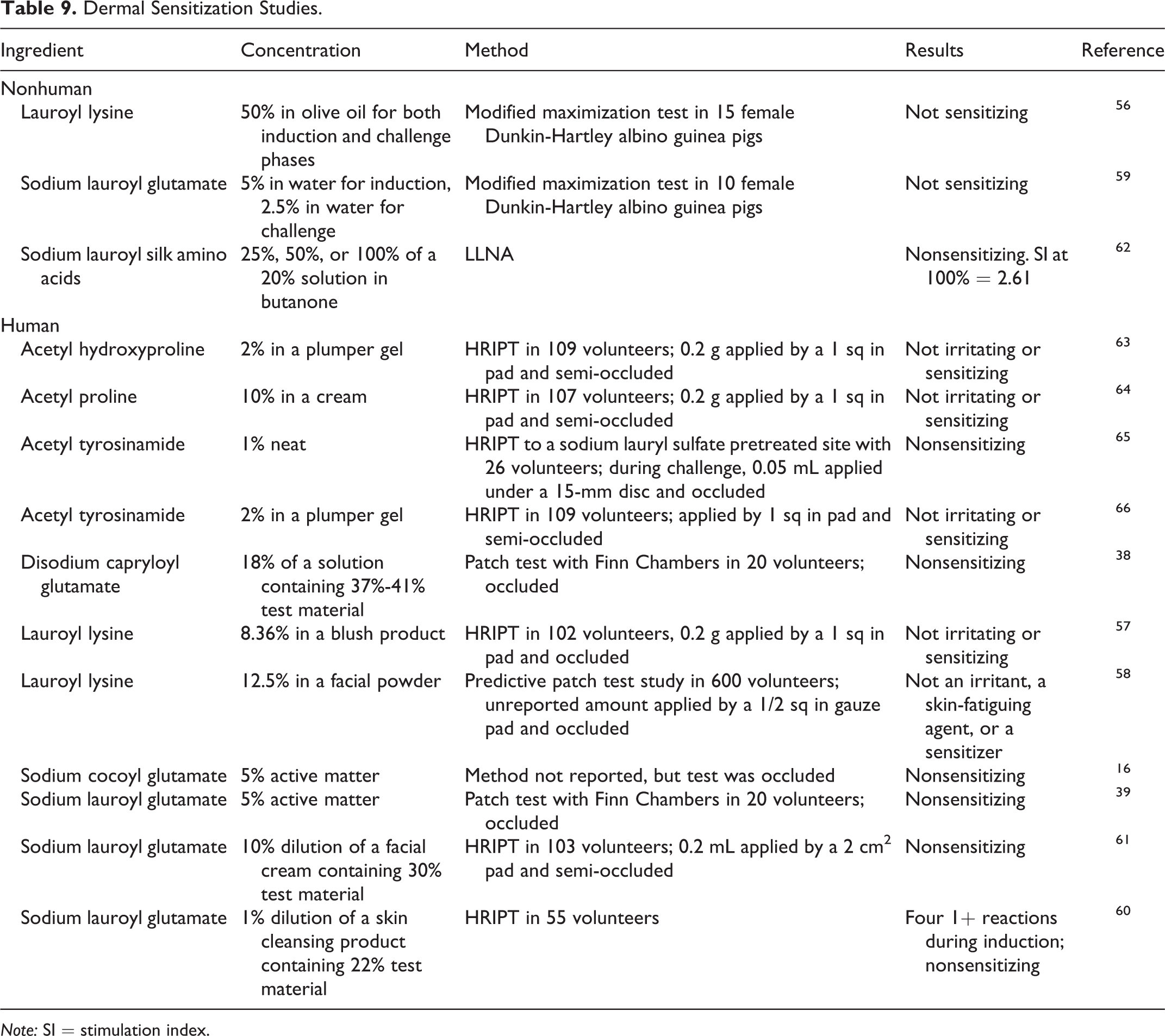
Note: SI = stimulation index.
Phototoxicity
Nonhuman and human phototoxicity studies are presented in Table 10. In nonhuman and human studies, acetyl tyrosine was not phototoxic at concentrations up to 1 and 10 mg/mL, respectively. Sodium cocoyl glutamate (up to 5%) and sodium lauroyl glutamate (up to 5%) were not phototoxic in human studies. 15,16,67,68
Phototoxicity and Photosensitization.

Summary
The 115 amino acid alkyl amides in this report mainly function as skin and hair conditioning agents and as surfactants—cleansing agents in personal care products. These ingredients are comprised of amino acids acylated with acids or acid chlorides at the amino acid nitrogen to form amides (except for lauroyl lysine, which is formed by acylation at the epsilon nitrogen). By and large, the ingredients in this report will not rapidly dissociate (beyond zwitterion formation) in the presence of water, but action by amidases is the most likely first step of metabolism if dermal penetration occurs. The relative exposure, hence, could include exposure to amino acid and fatty acid metabolites of these ingredients.
Lauroyl lysine has the most reported uses in cosmetic and personal care products, with a total of 649; most uses are in leave-on eye and facial makeup. Sodium cocoyl glutamate has the second greatest number of overall uses reported, with a total of 178; more than half of those uses are in rinse-off products. Lauroyl lysine is used at maximum concentrations up to 45% in lipsticks.
In the European Union, trialkylamines, trialkanolamines, and their salts (ingredients containing TEA) may be used only up to 2.5%, must be at least 99% pure, are not to be used with nitrosating systems, must have secondary amine content no greater than 0.5% and nitrosamine content no greater than 50 μg/kg, and must be kept in nitrite-free containers. The remaining ingredients are not restricted under the rules governing cosmetic products in the European Union.
Amino acid alkyl amides are used in household detergents. The FDA has approved acetyl cysteine in drug therapies. Acetyl methionine is an approved direct food additive.
In a study of 3 formulations containing 1.75% acetyl tyrosine, the test material was found to penetrate into and through ex vivo human skin, with the greatest penetration (approximately 30%) from a cream formulation.
In teratology studies, no evidence of impaired fertility or toxicity to fetuses was observed in rats at oral doses up to 2,000 mg/kg/d acetyl cysteine and in rabbits at oral doses up to 1,000 mg/kg/d acetyl cysteine. Male rats treated orally with 250 mg/kg/d acetyl cysteine for 15 weeks did not experience adverse effects to fertility or reproductive performance.
In in vitro studies, acetyl glutamic acid, acetyl proline, acetyl tyrosine, disodium capryloyl glutamate, sodium cocoyl glutamate, and sodium lauroyl glutamate were negative for genotoxicity. Acetyl cysteine was not genotoxic in an Ames test but had positive results in in vitro mouse lymphoma test. Acetyl cysteine and acetyl glutamic acid were negative in in vivo mouse studies.
No irritation was observed in in vitro studies with disodium capryloyl glutamate when tested at concentrations of 1.85% to 2.05%. A cream containing 8% acetyl proline was a mild irritant in another in vitro study. In rabbits, lauroyl lysine was nonirritating when tested at a concentration of 20%, whereas sodium lauroyl glutamate was a mild irritant at a concentration of 5%. In human studies, acetyl proline (up to 10%), acetyl tyrosine (up to 2%), disodium capryloyl glutamate (up to 7.38%), sodium cocoyl glutamate (up to 10%), sodium lauroyl glutamate (up to 10%), and sodium lauroyl silk amino acids (up to 6%) were not dermal irritants.
No ocular irritation was observed in in vitro studies of acetyl tyrosine (up to 1.25%), disodium capryloyl glutamate (up to 2%), and sodium lauroyl glutamate (up to 5%). Severe irritation was observed with sodium cocoyl glutamate at 5% using the HET-CAM method, but it was not irritating in another study with an unknown concentration. Slight irritation was observed with sodium lauroyl silk amino acids at 0.5% using the HET-CAM method. Lauroyl arginine in a mixture at 10% was not irritating to rabbit eyes. No adverse effects were observed during in-use studies of eye-area products containing acetyl hydroxyproline (up to 2%) and acetyl tyrosine (up to 2%) in human participants.
Sodium lauroyl silk amino acids when tested in a 20% solution was not sensitizing in an LLNA. No sensitization was observed in human studies with products containing acetyl hydroxyproline (up to 2%), acetyl proline (up to 10%), acetyl tyrosine (up to 2%), disodium capryloyl glutamate (up to 7.38%), sodium cocoyl glutamate (up to 5%), and sodium lauroyl glutamate (up to 5%).
Acetyl tyrosine was not phototoxic in a nonhuman study at up to 1000 µg/mL, nor was it phototoxic in a human study at 1%.
No published carcinogenicity studies on amino acid alkyl amides were identified by a literature search for these ingredients and no unpublished data were submitted.
Discussion
The Panel acknowledged that the safety of α-amino acids, acetyl methionine, and acetyl cysteine has been well supported by extensive studies and evaluation of acute and chronic dietary exposures (α-amino acids, acetyl methionine), use as approved food additives (α-amino acids, acetyl methionine), or therapeutic use (acetyl cysteine). The Panel determined that this body of research, coupled with irritation and sensitization data and the expectation that exposure from cosmetics is lower than from food consumed daily in the diet and would not result in significant systemic exposure (as discussed in the current report on amino acids), provides sufficient basis for determining the safety of amino acid–derived ingredients in cosmetic products.
Safety test data on dermal irritation and sensitization for the ingredients with the highest use concentrations, lauroyl lysine and sodium lauroyl glutamate, adequately supported the safety of the use of these ingredients in cosmetics. These ingredients function primarily as skin and hair conditioning agents and surfactants. The Panel noted that most surfactants exhibit some irritancy, as was the case with sodium lauroyl glutamate at 5% in a guinea pig study. Products using these ingredients should be formulated to be nonirritating.
The Panel discussed the issue of incidental inhalation exposure from hair sprays, face and body sprays, foundation sprays, and indoor tanning sprays. No inhalation data were identified or provided. These ingredients reportedly are used at concentrations up to 0.65% in cosmetic products that may be aerosolized. The Panel noted that 95% to 99% of droplets/particles would not be respirable to any appreciable amount. Coupled with the small actual exposure in the breathing zone and the concentrations at which the ingredients are used, the available information indicates that incidental inhalation would not be a significant route of exposure that might lead to local respiratory or systemic toxic effects. The Panel considered other data available to characterize the potential amino acid alkyl amides to cause systemic toxicity, irritation, sensitization, or other effects. They noted that numerous studies and reviews have been published in the literature regarding the safety of dietary exposure to amino acids, including studies on oral acute and chronic toxicity, carcinogenicity, and genotoxicity, which found no safety concerns for these substances in the amounts at which they are consumed in flavoring agents. Additionally, little or no irritation was observed in multiple tests of dermal and ocular exposure. A detailed discussion and summary of the Panel’s approach to evaluating incidental inhalation exposures to ingredients in cosmetic products is available at http://www.cir-safety.org/cir-findings.
The Panel expressed concern about animal-derived ingredients, namely the transmission of infectious agents. They stressed that these ingredients must be free of detectible pathogenic viruses or infectious agents (eg, bovine spongiform encephalopathy). These ingredients should be produced according to good manufacturing procedures and should conform to regulations for producing substances from animal-derived materials.
The Panel also expressed concern regarding pesticide residues and heavy metals that may be present in botanical ingredients. Because the plant proteins from which amino acids alkyl amides are produced are extensively processed, it is unlikely that these impurities would remain.
The Panel raised the issue of levels of free DEA that could be present as an impurity in the ingredients containing TEA and indicated that the levels of free DEA must not exceed those considered safe by the Panel, as stated in the current report on DEA, which is up to 0.64%. The Panel also discussed that tertiary alkyl amines such as TEA do not react with N-nitrosating agents directly to form N-nitroso compounds, but they can act as precursors in nitrosamine formation by undergoing nitrosative cleavage. The resulting secondary amine can then be N-nitrosated to products that may be carcinogenic.
The Panel noted the uncertainty regarding method of manufacturing. The Panel stated that industry should manufacture amino acid alkyl amides in a way that minimizes residual peptide content.
Conclusion
The Panel concluded that the 115 amino acid alkyl amides listed below are safe in the present practices of use and concentration in cosmetics, when formulated to be nonirritating. acetyl arginine* acetyl cysteine acetyl glutamic acid* acetyl glutamine acetyl histidine* acetyl methionine acetyl proline* acetyl tyrosine capryloyl collagen amino acids* capryloyl glycine capryloyl gold of pleasure amino acids* capryloyl keratin amino acids* capryloyl pea amino acids* capryloyl quinoa amino acids* capryloyl silk amino acids* cocoyl glutamic acid dipalmitoyl cysteine* dipotassium capryloyl glutamate dipotassium undecylenoyl glutamate* disodium capryloyl glutamate disodium cocoyl glutamate disodium hydrogenated tallow glutamate disodium N-lauroyl aspartate* disodium lauroyl glutamate disodium malyl tyrosinate disodium stearoyl glutamate disodium undecylenoyl glutamate* lauroyl arginine lauroyl collagen amino acids lauroyl glutamic acid* lauroyl lysine lauroyl proline lauroyl silk amino acids magnesium palmitoyl glutamate myristoyl glutamic acid* oleoyl tyrosine palmitoyl alanine* palmitoyl arginine* palmitoyl collagen amino acids palmitoyl glutamic acid* palmitoyl glycine palmitoyl gold of pleasure amino acids* palmitoyl isoleucine* palmitoyl keratin amino acids palmitoyl millet amino acids* palmitoyl oat amino acids* palmitoyl pea amino acids* palmitoyl proline palmitoyl quinoa amino acids* palmitoyl silk amino acids potassium caproyl tyrosine* potassium capryloyl glutamate* potassium cocoyl glutamate potassium cocoyl glycinate potassium cocoyl rice amino acids* potassium lauroyl collagen amino acids* potassium lauroyl glutamate* potassium lauroyl oat amino acids* potassium lauroyl pea amino acids* potassium lauroyl silk amino acids* potassium lauroyl wheat amino acids potassium myristoyl glutamate potassium olivoyl/lauroyl wheat amino acids* potassium stearoyl glutamate* potassium undecylenoyl glutamate* propionyl collagen amino acids* sodium caproyl prolinate* sodium capryloyl glutamate* sodium cocoyl alaninate sodium cocoyl amino acids sodium cocoyl apple amino acids sodium cocoyl barley amino acids* sodium cocoyl collagen amino acids sodium cocoyl glutamate sodium cocoyl glutaminate* sodium cocoyl glycinate sodium cocoyl/hydrogenated tallow glutamate* sodium cocoyl oat amino acids* sodium cocoyl/palmoyl/sunfloweroyl glutamate* sodium cocoyl proline* sodium cocoyl threoninate* sodium cocoyl wheat amino acids* sodium hydrogenated tallowoyl glutamate sodium lauroyl aspartate sodium lauroyl collagen amino acids* sodium lauroyl glutamate sodium lauroyl millet amino acids* sodium lauroyl/myristoyl aspartate* sodium lauroyl oat amino acids sodium lauroyl silk amino acids* sodium lauroyl wheat amino acids sodium myristoyl glutamate sodium olivoyl glutamate* sodium palmitoyl proline sodium palmoyl glutamate sodium stearoyl glutamate sodium/TEA-lauroyl collagen amino acids* sodium/TEA-lauroyl keratin amino acids* sodium/TEA-undecylenoyl collagen amino acids* sodium undecylenoyl glutamate* stearoyl glutamic acid* stearoyl leucine* TEA-cocoyl alaninate TEA-cocoyl glutamate TEA-cocoyl glutaminate* TEA-hydrogenated tallowoyl glutamate* TEA-lauroyl collagen amino acids TEA-lauroyl glutamate TEA-lauroyl keratin amino acids* TEA-lauroyl/myristoyl aspartate* undecylenoyl collagen amino acids undecylenoyl glycine undecylenoyl phenylalanine undecylenoyl wheat amino acids* zinc lauroyl aspartate*
*Not in current use. Were ingredients in this group not in current use to be used in the future, the expectation is that they would be used in product categories and at concentrations comparable to others in this group.
Footnotes
Authors’ Note
Unpublished sources cited in this report are available from the Director, Cosmetic Ingredient Review, Suite 1200, 1620 L Street, NW, Washington, DC 20036, USA.
Author Contributions
Christina L. Burnett contributed to conception and design, contributed to acquisition, analysis, and interpretation, and drafted the manuscript. Bart Heldreth contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the manuscript, and critically revised the manuscript. Lillian J. Gill, F. Alan Andersen, Wilma F. Bergfeld, Donald V. Belsito, Ronald A. Hill, Curtis D. Klaassen, Daniel C. Liebler, James G. Marks, Ronald C. Shank, Thomas J. Slaga, and Paul W. Snyder contributed to conception and design, contributed to analysis and interpretation, and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The articles in this supplement were sponsored by the Cosmetic Ingredient Review. The Cosmetic Ingredient Review is financially supported by the Personal Care Products Council.