Abstract
A 13-week dermal repeat-dose toxicity study was conducted with hydrodesulfurized (HDS) kerosene, a test material that also met the commercial specifications for aviation turbine fuel (jet A). The objectives were to assess the potential for target organ toxicity and neurotoxicity. The HDS kerosene was applied to the shaved backs of Sprague-Dawley CD rats, 12/sex/group, 6 h/d, 5 d/wk in doses of 0 (vehicle control), 165 mg/kg (20% HDS kerosene), 330 mg/kg (40% HDS kerosene), or 495 mg/kg (60% HDS kerosene). Additional rats (12/sex) from the control and the high-dose groups were held without treatment for 4 weeks to assess recovery. Standard parameters of toxicity were investigated during the in-life phase. At necropsy, organs were weighed and selected tissues were processed for microscopic evaluation. Neurobehavioral evaluations included tests of motor activity and functional observations that were conducted pretest, at intervals during the exposure period and after recovery. No test substance-related effects on mortality, clinical observations (except dermal irritation), body weight, or clinical chemistry values were observed. A dose-related increase in skin irritation, confirmed histologically as minimal, was evident at the dosing site. The only statistically significant change considered potentially treatment related was an increase in the neutrophil count in females at 13 weeks. No test article-related effects were observed in the neurobehavioral assessments or gross or microscopic findings in the peripheral or central nervous system tissues in any of the dose groups. Excluding skin irritation, the no observed adverse effect level value for all effects was considered 495 mg/kg/d.
Keywords
Introduction
The US Environmental Protection Agency (US EPA) announced a voluntary chemical data collection effort in 1998 called the High Production Volume (HPV) Challenge Program. 1 HPV chemicals are those produced or imported into the United States in aggregate quantities of at least 1 million pounds per year. Approximately 400 petroleum substances were sponsored in the EPA’s Challenge Program by companies belonging to the Petroleum HPV Testing Group. These 400 substances were organized into 13 categories to facilitate data sharing and to avoid redundant testing. The categories included crude oil, gases, gasoline, kerosene/jet fuel, gas oils, heavy fuel oils, lubricating oils, waxes, aromatic extracts, asphalts, grease thickeners, petroleum coke, and hydrocarbon wastes.
One goal of the HPV Challenge Program was to make previously unpublished data available to the public. As part of the HPV initiative, this previously unpublished report on the systemic and neurotoxic effects of kerosine (petroleum) and hydrodesulfurized (referred to in this article as HDS kerosene) is summarized subsequently for this special supplement.
Kerosene, also spelled “kerosine,” is used to describe the fraction of crude oil that boils approximately in the range of 302°F to 554°F (150°C-290°C) and is composed of hydrocarbons with carbon numbers primarily in the range of C9 to C16. The HDS kerosene that can contain thousands of individual chemical constituents is a class 2 UVCB 2 substance on the US Toxic Substances Control Act (TSCA) Chemical Inventory. Studies on the physical–chemical properties and composition of commercial kerosene products have demonstrated their complex chemistry. 3,4 There are several refinery processes that make hydrocarbon streams suitable for blending kerosene-range products. 5 Kerosene and derivative products have been used by consumers and industry for illumination and heating since the 1800s. However, today the predominant use of kerosene in the United States is in the production of aviation turbine fuel for civilian (jet A) and military (JP-8) aircraft. 6 Other fuel applications of kerosene are diesel fuel no. 1 and stationary turbine fuel no. 1. These kerosene-based fuels are generically similar but different technical requirements related to the specific end uses are associated with differing performance specifications and allowable additives. 6
Hydrodesulfurized kerosene and other types of kerosene cause dermal irritation in animals 7 and humans. 8,9 Since the discovery that aliphatic hydrocarbons from the kerosene boiling range could cause skin tumors in lifetime mouse skin painting studies, 10,11 the role of dermal irritation in tumor formation has been extensively studied. 12 -15 The HDS kerosene has been shown to be a mouse skin tumor promoter but not an initiating agent. Further, the promotional properties of these substances were shown to be associated with prolonged and repeated dermal irritation. If the equivalent dose of kerosene was applied to the skin under conditions that did not result in significant skin irritation (eg, dilution with a mineral oil), no skin tumors occurred. 14,15
The first studies to assess the potential for toxicity of kerosene following repeated inhalation exposure were conducted in 1976. 16 Rats and beagle dogs were exposed 6 h/d, 5 d/w for 12 weeks to kerosene vapors at levels up to 100 mg/m3 (the maximally attainable vapor only concentration). The authors reported that there were no ill effects associated with exposure at this level. The test material used was described as “deodorized kerosene,” boiling between 406°F and 522°F (208°C-272°C) and comprised primarily of aliphatic (96%) hydrocarbons. The aromatics present were primarily alkylbenzenes.
Due to the relatively low volatility of HDS kerosene, 2 studies were initiated utilizing dermal application as the route of test material exposure. The repeat-dose study presented here evaluated target organ effects and neurotoxicological properties. A previously reported study was done with the same HDS kerosene sample to evaluate the potential for developmental and/or reproductive effects. 17 Both studies were designed to limit the amount of dermal irritation in male and female rats. The screening study for reproduction and developmental effects (Organization for Economic Cooperation and Development [OECD] guideline 421) showed that 8 weeks of daily application (6 h/d) of HDS kerosene to the shaved backs of Sprague-Dawley rats produced only slight to moderate skin irritation at the high dose. 17 The study reported here, a 13-week repeat-dose study with neurotoxicity evaluations, supplemented the previous investigations of kerosene by assessing the potential for target organ effects and neurotoxicity and also investigated the potential for recovery by holding some control and high-dose group rats without treatment for 4 weeks after the scheduled termination.
Materials and Methods
Test Materials
Hydrodesulfurized kerosene (CAS number 64742-81-0) was obtained from BP Oil Co (Cleveland, Ohio) and was manufactured to meet the specifications for jet A. 18 Chemical analysis of hydrocarbon types was done by Mobil Research and Development Corp (Paulsboro, New Jersey). Other physical properties (ie, flash point, freezing point, sulfur, and distillation temperature) were done according to the American Society for Testing and Materials (ASTM) methods specified in D1655 by Chevron Research and Technology Company (Richmond, California). The vehicle used in this study was United States Pharmacopeia (USP)-grade mineral oil (Bristol-Myers Squibb Company, New York, New York) with a viscosity of 340 Saybolt Universal Seconds.
Animals
A total of 72 male and 72 female Sprague-Dawley (CD) rats, approximately 5 weeks of age, were purchased from Charles River Laboratories (Portage, Michigan). The rats were quarantined for 7 days for serological testing and observation. All animals were considered healthy and were approximately 7 to 9 weeks of age at the initiation of this study. The rats were housed individually in stainless steel wire-bottom cages during all study phases. Animals study rooms were set to maintain 18°C to 26°C temperature, 40% to 70% relative humidity, and 12-hour light–dark cycles. Except for overnight fasting periods prior to scheduled clinical pathology evaluations at necropsy, each rat was allowed ad libitum access to Certified Rodent Diet 5002 (PMI Feeds, Inc, St Louis, Missouri) and water during quarantine and study periods.
Experimental Design
The study was conducted in compliance with the US EPA's Good Laboratory Practice Regulations (TSCA, 40 CFR, Part 792) and was listed on Battelle Master List of regulated studies. The test procedures used in this study followed the US EPA Neurotoxicity Test Guidelines 40 CFR Ch 1 (7-1-94). Test procedures were also similar to those of the OECD test number 411: Subchronic Dermal Toxicity Study: 90 Days and Test number 424: Neurotoxicity in Rodents. Animals were randomized to treatment groups 4 days before initiation of dosing. The animals were randomly assigned by the PATH/TOX system (Xybion Corporation, Morris Plains, New Jersey) to treatment groups by body weight. The PATH/TOX software algorithm assures homogeneity of mean body weights across all treatment groups.
Dose Administration
Hydrodesulfurized kerosene was diluted in USP-grade mineral oil to obtain mixtures of 20%, 40%, and 60% (v/v) HDS kerosene and stored in amber glass bottles with magnetic stir bars at room temperature. All dosing bottles were mixed just prior to use, and all dosing formulations were used within 14 days of preparation. Stability and homogeneity of the formulated doses were determined by ultraviolet spectroscopy. Calibrated volumetric pipettes with disposable tips were used to deliver and evenly spread the dosing solution over a standard area (approximately 25 cm2) from the mid-back to the intrascapular area. The application site was clipped free of fur weekly (7 + 1 days). Dosing volumes were calculated based on the group mean body weights for each subgroup within a treatment group at 1 mL/kg/d, to a maximum of 0.3 mL/rat/d. Animals (12/sex/group) were dosed 5 consecutive days/wk for 13 consecutive weeks at doses of 0 (vehicle control), 165 mg/kg (20% HDS kerosene), 330 mg/kg (40% HDS kerosene), or 495 mg/kg (60% HDS kerosene). The high dose was chosen since previous studies had demonstrated that repeated dermal application of HDS kerosene when diluted to concentrations above 60% with mineral oil produced unacceptable dermal irritation. 17 Additional rats (12/sex) in the control and high-dose groups were held after final treatment for a 4-week recovery period. The site of application was not occluded during the 6-hour exposure period. Instead, animals were fitted with plastic Elizabethan collars to prevent them from disturbing the sites of test material application. After 6 hours, any remaining dose was removed with dry gauze. Necropsy animals received at least 2 consecutive doses and were dosed within 24 hours of necropsy. Dose concentrations of HDS kerosene were analyzed monthly over the course of the study for a total of 5 analyses. The results of analyses performed during the study indicate that HDS kerosene concentrations were within 9% of the target concentrations. Test article homogeneity was evaluated on the high and low concentration during the first week of dosing. Samples from the top, middle, and bottom of each batch demonstrated the formulations were homogeneous.
Homogeneity analyses performed on the low- and high-dose formulations indicated relative standard deviations of the determined concentration from the top, middle, and bottom of each batch to be less than 1%, demonstrating that the formulations were homogeneous.
In-Life Observations
Each animal was examined once daily for overt signs of toxicity and changes in general behavior and appearance during the study. Just prior to dosing, the application site was evaluated for dermal irritation. 19 Skin irritation scoring was also performed on nontreatment days during the dosing phase of the study. Body weights were measured at the time of randomization, on day 1 prior to dosing and weekly thereafter. Fasted body weights were measured at each scheduled necropsy. Ophthalmic examinations were performed on all animals prior to the start of dosing and prior to scheduled termination.
Neurobehavioral Assessment
During the week prior to the first dose, each rat was subjected to the functional observational battery (FOB) conducted according to the 1994 EPA Neurotoxicity Guidelines to obtain baseline data. The FOB alone was administered at 1, 6, and 24 hours postfirst dose and at 7 and 14 days. During study weeks 4, 8, and 12, FOB, motor activity, and startle response testing were conducted o\in all rats.
Half of the control and high-dose animals were allowed to recover for 30 days and then were retested on each of the tasks. The FOB began at the home cage prior to dosing (except for the 1 and 6 hours postdose evaluations on day 1). The FOB included a thorough description of the test animal’s appearance, behavior, and functional integrity. This was assessed through home cage observations, while the rat was moving freely in an open field and through manipulative tests. Testing proceeded from the least to the most interactive with the animal. The observations included posture, palpebral closure, convulsions, spontaneous vocalizations, piloerection, lacrimation, salivation, and appearance of the fur. Open field observations were done next and included counting number of rears, gait characteristics, and ease of mobility. Reflex testing began next and measured responses to the approach and touch of a blunt object, a click stimulus, tail pinch, constriction of the pupil to light, aerial righting, and grip strength performance. After body weights and rectal temperatures were recorded, measurements of landing foot-splay and paw-lick latency were evaluated. All observations were done by trained technicians who were not aware of the exposures that the animals had received.
Motor activity was measured in 8 animals simultaneously for the pretest and on-study test intervals. A Figure 8 Photobeam Activity System (San Diego Instruments, San Diego, California) was used to make the measurements. Motor activity was determined over six 5-minute intervals. The test sessions were controlled and monitored by a computer, and white noise projection was used to mask any sounds that could potentially influence behavior.
Startle responsiveness was measured using an SR-LAB startle Response System (San Diego Instruments). Response measurement took place within sound-isolated cubicles (Coulbourn Instruments Isolation Cubicle Model E10-20, Whitewall, Pennsylvania). Startle reflex was determined over 60 repeated trials, and each session took approximately 15 minutes to complete. An acclimation period of 5 minutes was followed by either startle trials with a tactile air-puff the main startle stimulus or prepulse trials in which an 80- to 90-db white noise prepulse preceded the tactile stimulation. A background level of 70-db white noise was present when the prepulse or main stimulus was off. Trials and intertrial intervals were randomized with a total of 30 main stimulus only trials and 30 prepulse plus main stimulus trials being presented. The average response amplitude at each trial presentation was the dependent variable for this study. Inclusion of a prepulse and measuring startle response over repeated trials provide data for initial reactivity, habituation, and prepulse inhibition of the startle response in the same test.
Hematology and Serum Chemistry
At the end of treatment and following the 4-week recovery period, blood was collected for hematology, coagulation determination, and serum chemistry evaluations. Blood for hematology evaluations was collected from 12 rats/sex/group at termination at day 90 and from the recovery rats in the vehicle control and high-dose groups (12 rats/sex/group) at day 120 of necropsy. Blood for determination of coagulation endpoints was collected from 6 rats/sex/group at necropsy at day 90 and from the recovery rats in the vehicle control and high-dose groups (6 rats/sex/group) at day 120 18 of necropsy. Blood for serum chemistry evaluations was collected from 12 rats/sex/group at necropsy at day 90 and from the recovery rats in the vehicle control and high-dose groups (12 rats/sex/group) at day 120 of necropsy. Except for blood collected at necropsy from the abdominal aorta for coagulation times, blood was obtained from the retro-orbital sinus. Blood for hematology evaluation was collected in microtubes containing EDTA as the anticoagulant. Blood for prothrombin and activated partial thromboplastin time determinations was collected in BD Vacutainers (Becton, Dickinson and Company, Franklin Lakes, New Jersey) with sodium citrate as the anticoagulant. Blood for serum chemistry determinations was collected in microtubes without anticoagulant, and the serum was separated by centrifugation after clotting.
Necropsy, Histology, and Pathology
Following an overnight fast from food (but not water), complete necropsies were performed on 6 rats/sex/group following 13 weeks of dosing and on 6 rats/sex/group (control and high dose only) following a 4-week nontreatment recovery period. Rats were routinely sacrificed (exsanguinated) following clinical pathology blood collection. All necropsies were attended by a board-certified veterinary pathologist experienced in small laboratory animal pathology. Each necropsy included examination of the external surface of the body, all orifices, cranial, thoracic, abdominal, and pelvic cavities, and their contents. Gross observations were initially recorded on each rat’s Individual Animal Necropsy Record and later entered into the PATH/TOX computer system for the purposes of generating individual and summarized necropsy data.
The tissues listed in Table 1 were collected for microscopic evaluation, and organ weights were obtained for the adrenal glands, brain, epididymides, heart, kidneys, liver, ovaries, prostate, spleen, testes, thymus, and uterus. Tissues were collected and fixed in 10% neutral-buffered formalin except for eyes, which were preserved in Bouin's fixative. The animals selected for neuropathological evaluation (6 rats/sex/group) were anesthetized by intraperitoneal injection of Pentothal (Hospira, Inc, Lake Forest, Illinois) and transcardially perfused in situ using 10% neutral-buffered formalin. Samples of brain, spinal cord, sciatic nerve, sural nerve, and tibial nerve were collected for microscopic evaluation.
Tissues Collected for Weight Determinations and/or Histological Evaluation.
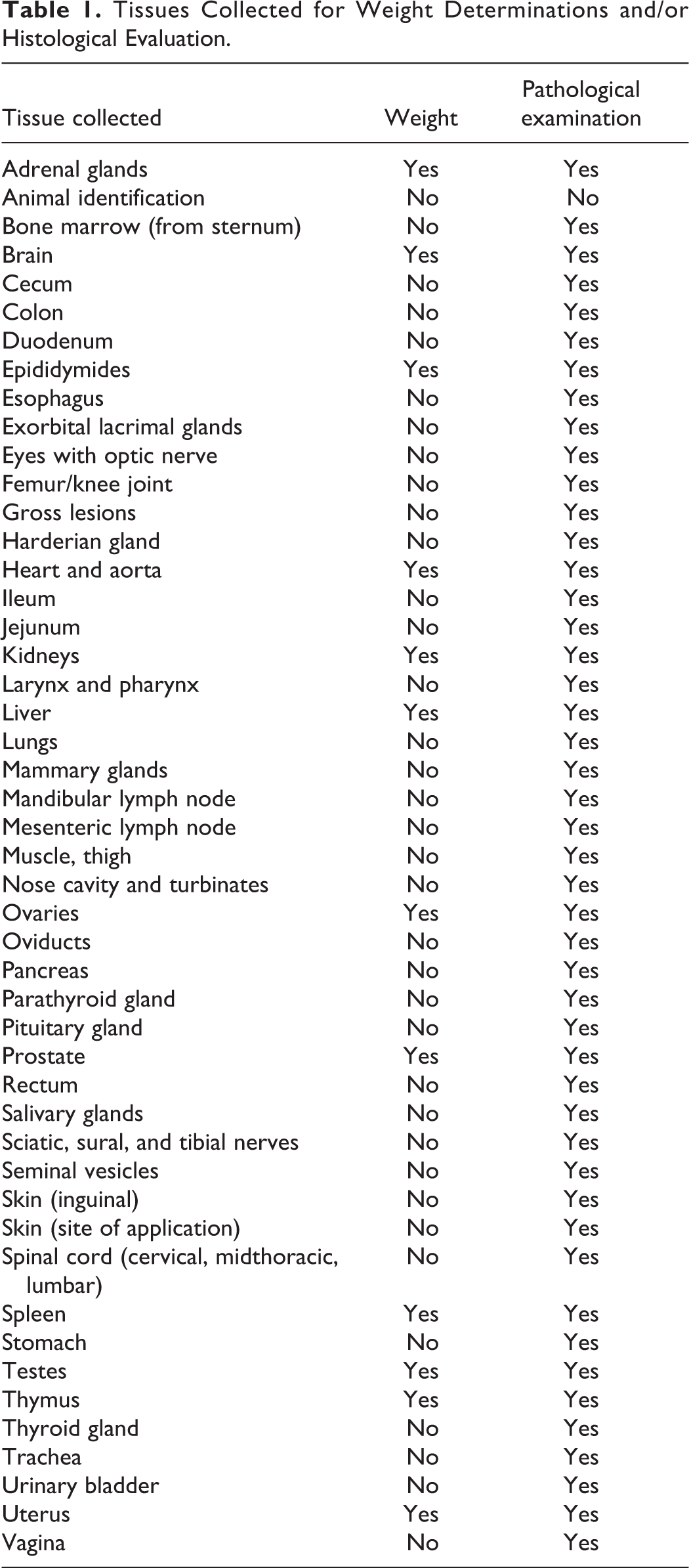
Following fixation, all tissues collected at necropsy (excluding animal identification, harderian glands, and nasal turbinates) from the 13-week control and high-dose-treated rats were trimmed, embedded, sectioned, and stained with hematoxylin and eosin for microscopic examination. In addition, any gross lesions from lower dose or recovery rats were evaluated. Since skin at the site of application was deemed to be a target tissue of kerosene, this tissue was also examined from all the low- and mid-dose 13-week rats and all recovery rats. In contrast, the brain (forebrain, cerebrum, midbrain, cerebellum, pons, and medulla oblongata), gasserian ganglia, dorsal root ganglia, dorsal and ventral root fibers, sural nerve, tibial nerve, spinal cord (cervical and lumbar areas), and sciatic nerve were examined from the designated neuropathology groups, consisting of control and high-dose rats. As there was no pathological evidence of neurological effects in animals at the end of the exposure period, these tissues were not evaluated histologically in the recovery group animals.
Statistics
Normally distributed in-life data were analyzed for test substance effects by analysis of variance (ANOVA) and pairwise comparisons made between groups using Dunnett test. A nonparametric 1-way ANOVA Kruskal-Wallis test was also calculated. A nonparametric procedure was used if the assumptions of normality and constant variance did not hold as indicated by the examination of the residuals from the parametric ANOVA fit. The Wilcoxon 2-sample rank sum procedure was then used for pairwise comparisons with the control group. Statistical significance was reported at the
Results
Analytical Characterization
The test material conformed to the key specifications for commercial aviation turbine fuel 18 (jet A) and Air Force jet propulsion fuel 20 (JP-8) that were evaluated as part of the test substance characterization work during a previous study with the test article. 17 Table 2 summarizes the most relevant specifications for those 2 fuels and the specific data on the HDS kerosene sample. The test material had similar distillation characteristics but higher (∼20%) aromatic levels than the deodorized kerosene that had been used in previous repeat-dose inhalation studies. 16
Comparison of the HDS Kerosene Sample to Jet Fuel Specifications.

Abbreviations: ASTM, American Society for Testing and Materials; HDS, hydrodesulfurized.
In-Life Observations and Dermal Histopathology
The incidence and severity of dermal irritation at the site of application were dose related in rats of both sexes. Daily scoring for irritation at the sites of application showed that erythema was first observed in low-dose males on day 4 with a mean score of 0.17, in mid-dose males on day 4 with mean score of 0.5, and in high-dose males on day 4 with score of 1.33. By day 5, males in the high-dose group had a mean score of 2 and by day 6 females in the high-dose group had a mean score of 1.33. These were the highest scores for any day of the study and indicate very slight to well-defined erythema (data not shown). Edema was first observed on day 10 in high-dose males and females with mean scores of 0.33 and 0.04 (very slight edema), respectively. Signs of dermal irritation receded as the study progressed. After the second week, no group mean score for erythema exceeded 1.0, and no sign of edema was recorded after week 5 (data not shown). At the end of the study, skin from the application site was removed, examined, and graded for irritant effects (Table 3).
Averagea Severity Scoreb of Skin Lesions in 13-Week-Treated Rats.
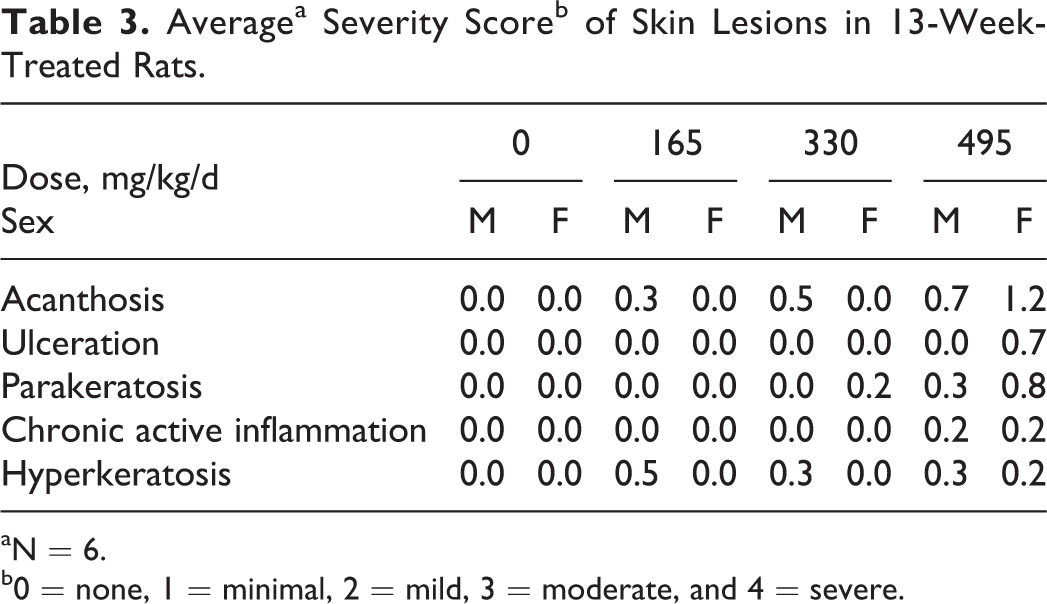
aN = 6.
b0 = none, 1 = minimal, 2 = mild, 3 = moderate, and 4 = severe.
All other observations noted during the dosing phase of the study, including alopecia, abrasions/lesions, and red eye discharge, were considered to be incidental in nature and unrelated to treatment. There were no statistically significant differences in mean absolute body weight or mean absolute weight gain when treated groups were compared to the control group for the duration of the study. There were no apparent test substance-related effects in the eyes as evidenced by ophthalmoscopic examination prior to necropsy (data not shown).
Functional Observational Battery
The parameters evaluated in the functional observation battery are listed in Table 4. Changes in autonomic response were not evident with exposure to HDS kerosene. Occasional lacrimation and totally constricted pupils were observed in all groups and were considered incidental (data not shown).
Parameters Evaluated as Part of the Functional Observation Battery.

Muscle tone and equilibrium end points were unaffected by exposure to HDS kerosene. No differences in gait, mobility, or righting reflex were observed (data not shown). Forelimb and hindlimb grip strength increased with time across all dose groups, consistent with body weight gain (data not shown). Prestudy forelimb grip strength ranged from 0.53 to 0.64 kg and increased to 0.82 to 1.31 kg at day 90. Hindlimb grip strength increased from a range of 0.21 to 0.25 to a range of 0.54 to 0.73 kg from prestudy to day 90. During the early test intervals, males and females had similar grip strength scores. By day 30, forelimb grip strength in males was greater than females and after day 60 hindlimb grip strength in males was greater than females. Data on landing foot-splay are shown in Table 5. Although there were 2 time points at which statistically significant differences in foot-splay between males in the high dose and control group were observed on days 1 and 120, these differences were not considered to have been treatment related as the absolute values are most likely due to preexisting, random difference in scores between the vehicle group and the high-dose group that were observed prior to the initiation of dosing, relatively preserved over the course of the experiment and were well within the range of experimental variability.
Mean Landing Footsplaya for Various Time Points During Dermal Exposure to HDS Kerosene.
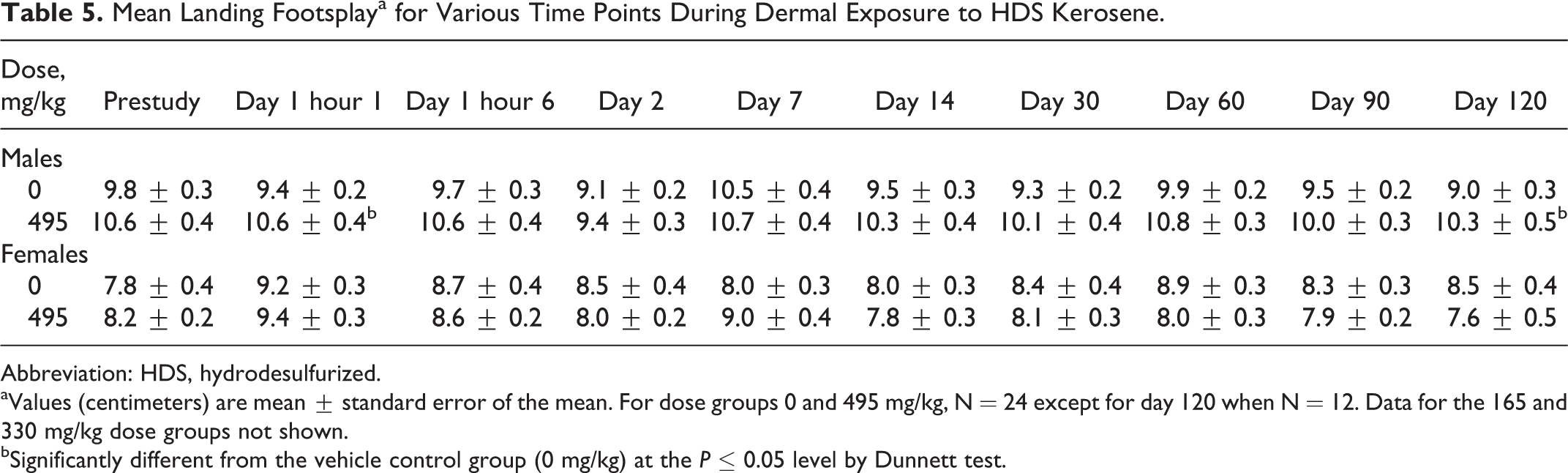
Abbreviation: HDS, hydrodesulfurized.
aValues (centimeters) are mean ± standard error of the mean. For dose groups 0 and 495 mg/kg, N = 24 except for day 120 when N = 12. Data for the 165 and 330 mg/kg dose groups not shown.
bSignificantly different from the vehicle control group (0 mg/kg) at the
Sensorimotor reactivity (motor responses that are normally elicited by sensory stimuli) and indices reflective of central nervous system integrity were unchanged during treatment (data not shown).
Body temperature ranged from 36.4°C to 38.3°C. During the various test intervals, body temperature fluctuated within that range without any dose–response relationship. Occasionally, statistically significant differences in body temperature from control were seen
Motor Activity
The mean total horizontal and total vertical activity data for each sex/dose group/study day combination are provided in Table 6. Female rats exhibited greater activity than did male rats at all testing intervals. Statistically significant differences attributed to HDS kerosene exposure were not evident.
Average Total Motor Activitya for Various Time Points During Dermal Exposure to HDS Kerosene.
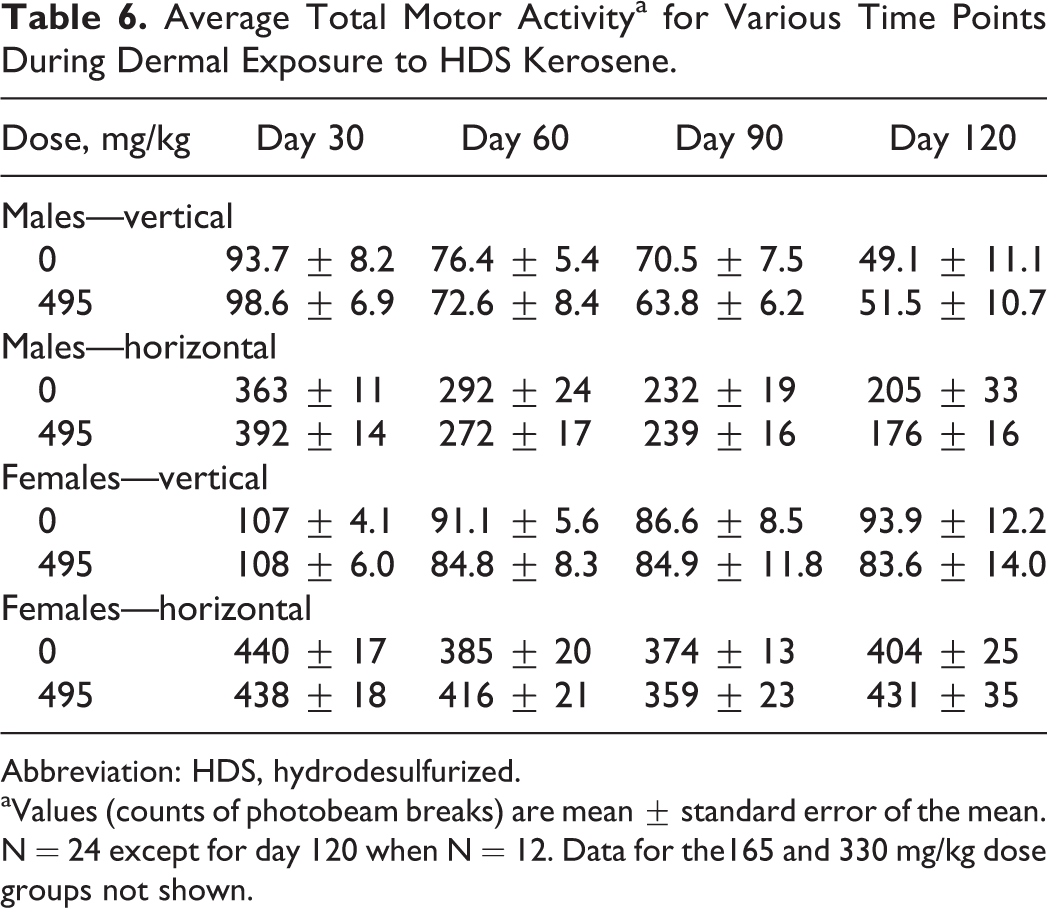
Abbreviation: HDS, hydrodesulfurized.
aValues (counts of photobeam breaks) are mean ± standard error of the mean. N = 24 except for day 120 when N = 12. Data for the165 and 330 mg/kg dose groups not shown.
For both horizontal and vertical activity within each time period, the percentage habituation (percentage change in activity from testing interval 1 to testing interval 6) was also calculated. No significant change was caused in this measure by repeated exposure to HDS kerosene (data not shown).
Startle Response
Dermal exposure to HDS kerosene at any of the test doses produced no statistically significant differences in startle response (Table 7). Only data from the vehicle control group and the 495 mg/kg (60% HDS kerosene) are displayed. The addition of a prepulse before the main tactile stimulus inhibited the startle response. No statistically significant difference in habituation of the startle response was found between dose groups across trials compared with controls (data not shown).
Mean Startle Response for Trials With and Without Prepulse During Dermal Exposure to HDS Kerosene.a
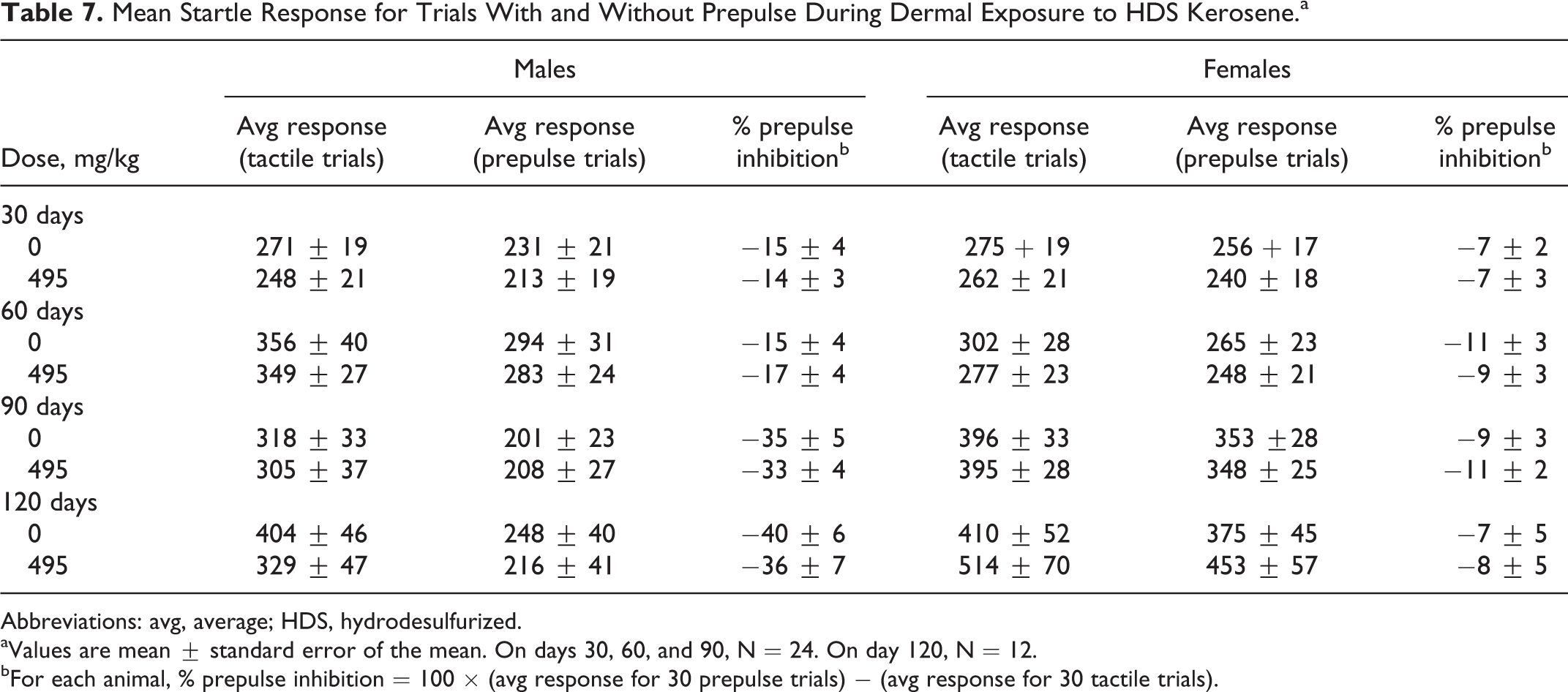
Abbreviations: avg, average; HDS, hydrodesulfurized.
aValues are mean ± standard error of the mean. On days 30, 60, and 90, N = 24. On day 120, N = 12.
bFor each animal, % prepulse inhibition = 100 × (avg response for 30 prepulse trials) − (avg response for 30 tactile trials).
Hematology and Serum Chemistry
None of the results of hematology tests performed in study indicated treatment-related effects with the exception of the neutrophil counts in the high-dose females (Table 8). Because of the magnitude of the difference from control values and the fact that neutrophil counts in the high-dose males and females were high compared to historical control data, the neutrophil counts were interpreted to have been increased (slightly) due to treatment with HDS kerosene. However, the differences were so small that they were not considered toxicologically important and not used in determining the overall no observed adverse effect level (NOAEL). Statistical evaluation of mean hematology data also revealed significant differences when treated groups were compared to respective control values for hematocrit and red cell count values (data not shown). Males treated with 495 mg/kg (60% HDS kerosene) had statistically significant decreased hematocrit values. Increased mean corpuscular hemoglobin concentration (MCHC) values were noted in males in the 330 and 495 mg/kg treatment groups on day 90 compared to controls. Due to the small magnitude of the changes, these hematology results are not considered to be treatment related. Qualitative evaluation of morphological features in erythrocytes and platelets was all within normal limits. No apparent test substance-related differences in coagulation times were observed when treated groups were compared to respective control values (data not shown).
Differential White Blood Cell Countsa in Females.
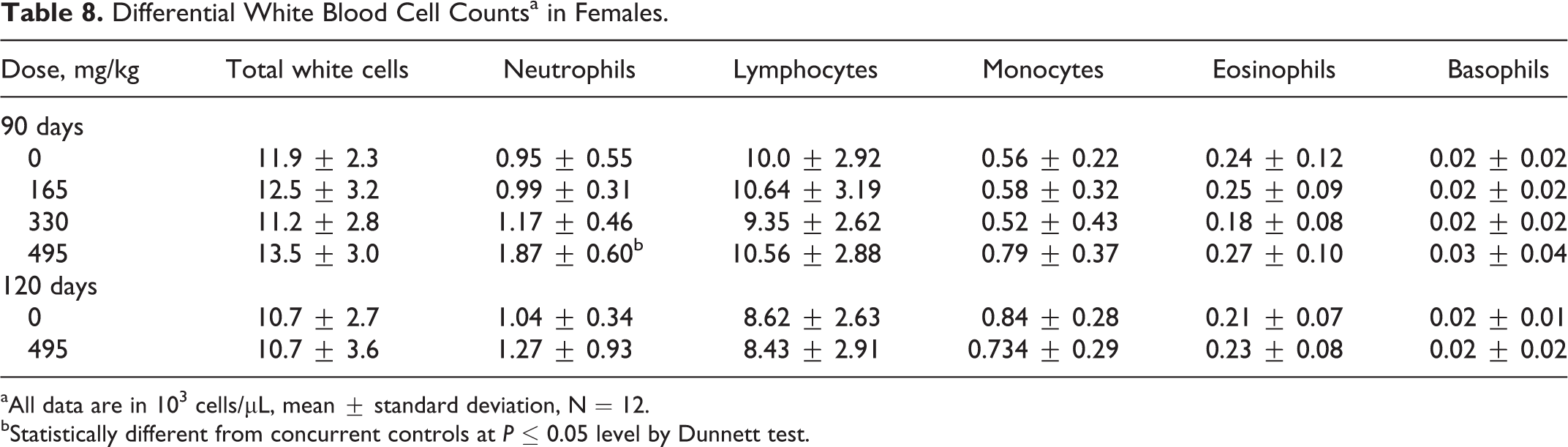
aAll data are in 103 cells/μL, mean ± standard deviation, N = 12.
bStatistically different from concurrent controls at
Evaluation of mean serum chemistry parameters revealed a number of statistically significant differences when treated groups were compared to respective control values (data not shown). These differences, however, were considered incidental to treatment based on the lack of a dose response, the small magnitude of difference, or biologically insignificant changes. Statistically significant differences included (1) an increase in sorbitol dehydrogenase in the 330 mg/kg (40% HDS kerosene)-treated males on day 90 and in the 495 mg/kg (60% HDS kerosene)-treated males on day 120 and (2) an increase in the day 90 chloride values in the 495 mg/kg (60% HDS kerosene)-treated males. Statistically significant increases in sodium values were noted in the 330- and 495 mg/kg-treated females on day 90, and statistically significant decreases in total protein, albumin, and calcium values were noted in the 495 mg/kg-treated females on recovery day 120.
Organ Weights and Neuropathology
At the 13-week necropsy, the statistically significant findings were only found in females in the 495 mg/kg (60% HDS kerosene) group (data not shown). This group had increased spleen-to-body weight and spleen-to-brain weight ratios compared to the vehicle control group. However, no gross or microscopic findings were observed in the spleens. After the 4-week recovery period, females in the 495 mg/kg group had an absolute spleen weight increase compared to the vehicle control group. No gross or microscopic findings in the spleens were observed and the effects were not considered to be treatment related.
The results of the neuropathology phase of the study indicate that dermal doses of HDS kerosene up to 495 mg/kg for 13 weeks did not cause treatment-related neurological effects. A control animal had a brain lesion that consisted of an incidental focus of perivascular lymphocytic inflammation. No other gross or microscopic findings in the peripheral or central nervous system were noted.
Discussion
This study was conducted in order to characterize the potential toxicity of topically applied HDS kerosene to male and female Sprague-Dawley rats. Dose levels of 0%, 20% (165 mg/kg), 40% (330 mg/kg), or 60% (495 mg/kg) HDS kerosene in mineral oil were applied 5 days/wk for 13 consecutive weeks to the intrascapular region of rats. Survival, clinical observations, body weight and food consumption values, ophthalmic examinations, FOB, motor activity and startle response, clinical pathology, and a gross and microscopic examination of protocol-specified tissues were performed to assess toxicity.
All animals survived until their day of scheduled sacrifice. No test substance-related effects on body weight were observed. High-dose females had a small but statistically significant increase in spleen weight absent corresponding pathological changes. No such increase in weight was noted in males or in any other organs of either sex for any of the dose groups. A general dose-related increase in the incidence of erythema, edema, epidermal scaling, scab formation, thickening of the skin, and ulceration of the dose site was observed for both sexes. Upon microscopic examination, the skin lesions at the application site were minimal in nature with an average severity score of less than 1 (minimal). Evaluation of the application sites of high-dose recovery animals revealed complete reversibility in the females and minimal hyperkeratosis in the males, an indication of recovery from dosing.
Dermal irritation from repeated exposure to middle distillate hydrocarbons like kerosene is recognized as a significant experimental toxicology issue. 15 Repeated application will cause severe skin injury in many common animal models. Some methods of mitigating jet A dermal irritation in rats were explored previously, 21 including rotating the site of application, diluting 50% with 4:1 acetone–olive oil, and diluting 50% with mineral oil. Only the use of mineral oil resulted in an acceptable degree of skin irritation over 28 days of repeated application. Similar experiences have been noted by other investigators. 14,17 The minimal dermal irritation observed over the 13-week duration of this study provides some assurance that any adverse findings were not secondary to stress or other factors unrelated to the test article.
No apparent test substance-related effects on neurobehavioral parameters were observed including autonomic function, muscle tone and equilibrium, sensorimotor responses, central nervous system, and physiologic responses. No gross or microscopic changes in peripheral or central nervous system tissues were seen, consistent with an absence of neurological clinical signs.
Hematological evaluations revealed some changes that were toxicologically insignificant based on their observed magnitude of change compared with control and the lack of biological relevance to kerosene exposure. None of the hematological results performed in the study indicated any treatment-related effects with the possible exception of the neutrophil counts in the high-dose females. However, the performing laboratory did not consider this finding to be significant enough to influence the determination of the NOAEL.
Based upon the data generated in this study, HDS kerosene applied dermally 5 days/wk up to 495 mg/kg/d (60%) to Sprague-Dawley rats for 13 weeks caused minimal skin irritation and no apparent target organ toxicity. No kerosene-induced gross or microscopic findings in peripheral or central nervous tissue were observed; these findings are consistent with and were supported by an absence of neurological clinical signs. Therefore, the NOAEL for target organ toxicity of dermally applied HDS kerosene was the highest dose tested, 495 mg/kg/d.
Footnotes
Acknowledgments
The authors would like to thank Wende S. Davis, Gary B. Freeman, Craig R. Mahon, John T. Yarrington, Michael J. Ryan, and Merrill R. Osheroff for their preparation of the laboratory report and Joseph M. Frasca, Richard H. McKee, and Daniel W. Kelly for their review of the article.
Authors’ Note
The study reported here was sponsored by the Petroleum Product Stewardship Council in 1997 as part of a testing program to generate hazard information on representative refinery streams used to blend transportation fuels.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Several of the authors are now or were previously employed by companies that manufacture kerosene and/or jet fuel or by the American Petroleum Institute, a trade organization that represents the interests of petroleum product manufacturers in the United States.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by the Petroleum Product Stewardship Council whose members where Amoco, Arco, BP, Chevron, Mobil, Texaco, and Unocal.
