Abstract
The present study investigated the potential subchronic toxicity of self-assembled-micelle inhibitory RNA-targeting amphiregulin (SAMiRNA-AREG) in mice. The test reagent was administered once-daily by intravenous injection for 4 weeks at 0, 100, 200, or 300 mg/kg/day doses. Additional recovery groups (vehicle control and high dose groups) were observed for a 2-week recovery period. During the test period, mortality, clinical signs, body weight, food consumption, ophthalmology, urinalysis, hematology, serum biochemistry, gross pathology, organ weight, and histopathology were examined. An increase in the percentages of basophil and large unstained cells was observed in the 200 and 300 mg/kg/day groups of both sexes. In addition, the absolute and relative weights of the spleen were higher in males given 300 mg/kg/day relative to the concurrent controls. However, these findings were considered of no toxicological significance because the changes were minimal, were not accompanied by other relevant results (eg, correlating microscopic changes), and were not observed at the end of the 2-week recovery period indicating recovery of the findings. Based on the results, SAMiRNA-AREG did not cause treatment-related adverse effects at dose levels of up to 300 mg/kg/day in mice after 4-week repeated intravenous doses. Under these conditions, the no-observed-adverse-effect level of the SAMiRNA-AREG was ≥300 mg/kg/day in both sexes and no target organs were identified.
Keywords
Introduction
RNA interference (RNAi) is a natural process of double strand RNA regulating specific genes, which occurs in most eukaryotic cells. 1 RNAi uses synthetic small interfering RNA (siRNA) and microRNA. 2 Since the discovery of the RNAi mechanism in mammalian cells,3,4 it has been a therapeutic target for gene-associated diseases including cancer, viral infections, autoimmune diseases, and gene disorders.5-7
Compared to the traditional small molecules and protein/antibody-based drugs, siRNA therapeutic approaches are more advantageous owing to their design which allows them to knock down specific targets, including non-druggable targets. 6 In addition, the delivery of target gene-specific siRNAs is faster, and their selectivity and potency are much higher than traditional drugs. 8 Despite the great therapeutic potential demonstrated by clinical trials, RNAi application is limited by the safety, poor stability, and low efficacy in cellular uptake. 9 In addition, innate immune activation via siRNA represents undesirable side effects in vivo. 10 Recently, various approaches by chemical modification or liposome or lipid conjugation have been developed in an effort to increase the therapeutic potential of siRNA therapeutics such as decreasing toxicity, increasing cellular uptake, and more effective gene silencing.11-13
Among the diseases, one that reported to be amenable to siRNA-based therapy is pulmonary fibrosis, which is characterized by excessive fibroblast proliferation and extracellular matrix deposition leading to airway remodeling. 14 Transforming growth factor (TGF)-β plays a central role in the pathogenesis of pulmonary fibrosis.15,16 TGF-β has been shown to activate amphiregulin (AREG), an epithelial growth factor receptor ligand that plays an essential role in pathogenesis of TGF-β-induced pulmonary fibrosis in vitro and in vivo.17,18 Interestingly, a previous study showed that AREG-specific siRNA silencing attenuated collagen accumulation in a pulmonary fibrosis animal model. 18 Further, a recent study revealed that modified siRNA, self-assembled-micelle inhibitory RNA nanoparticles-targeted to AREG (SAMiRNA-AREG), effectively suppressed the collagen deposition and restored lung functions in a bleomycin-induced pulmonary fibrosis model. 9 More recently, it has been demonstrated that SAMiRNA-AREG has reno-protective effects by ameliorating renal fibrosis and inflammation. 19
Despite such promising outcomes with high stability and low immunogenicity, no animal experiments have been reported on the potential toxic effects of SAMiRNA-AREG. Therefore, as part of an extensive program of the safety evaluation studies, we conducted a 4-week repeated intravenous dose toxicity study in ICR mice and determined a no-observed-adverse-effect level (NOAEL). The present study was performed according to the test guidelines from the Ministry of Food and Drug Safety for the toxicity study of SAMiRNA-AREG under modern Good Laboratory Practice regulation.20,21
Materials and Methods
Animal Husbandry and Maintenance
Specific-pathogen-free CrlOri:CD (ICR) mice were purchased from Orient Bio (Seognam, Republic of Korea) at 6 weeks old and dosing started after 1 week of acclimatization. The mouse was selected for toxicological testing because it has been shown to be a pharmacologically relevant species following target engagement with as SAMiRNA-AREG.9,19 The animal rooms were maintained under a temperature of 23 ± 3°C, a relative humidity 50 ± 20%, with artificial lighting from 08:00 to 20:00 and with 10 to 20 air changes per hour and animals were housed individually in a polycarbonate cage (145 W × 295 L × 140 H mm). The animals were fed a commercial rodent chow (PMI Nutritional International Inc., Richmond, IN, USA) and sterilized tap water ad libitum. This experiment was conducted in facilities approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. All procedures were approved by the Institutional Animal Care and Use Committee of Korea Institute of Toxicology (Jeongeup, Republic of Korea).
Test Reagent and Treatment
SAMiRNA-AREG was provided by Bioneer Inc. (Daejeon, Republic of Korea) and manufactured as previously described. 9 The sizes of SAMiRNA-AREG nanoparticles are uniform and similar to each other (100 ± 20 nm and .2 ± .01 nm for average size and PDI, respectively). The stability of SAMiRNA nanoparticles was demonstrated to be 12 months as no significant degradation was noted in size distribution or polydispersion index in solution over this period under room temperature. The test reagent was dissolved in Dulbecco’s phosphate-buffered saline (PBS, pH 7.4; WelGENE Inc., Daegu, Republic of Korea) and prepared daily before treatment. After the tail was cleaned with 70% isopropyl alcohol, the test reagent was administered within 3 hours after test reagent preparation by intravenous injection through the tail vein for 4 weeks at dose levels of 0, 100, 200, or 300 mg/kg/day. SAMiRNA-AREG dosage (at a dose volume of 10 mL/kg) was calculated based on the most recently recorded body weight of each animal. The vehicle control group received an equivalent volume of PBS alone. The recovery group of animals were kept for 2 weeks after the last dose was administered.
Dose Selection
In a previous 2-week repeated intravenous dose toxicity study (data not shown), a significant decrease in the hemoglobin (HGB) and hematocrit (HCT) and significant increase in the absolute and relative spleen weights were observed in the male 300 mg/kg/day group, but not in the female 300 mg/kg/day group. Therefore, a dose of 300 mg/kg/day was selected as the highest dose in this current study, while 100 and 200 mg/kg/day were selected as the lowest and medium doses, respectively, using a common ratio of 1.5 or 2.
Experimental Groups
Healthy male and female mice were randomly assigned to 4 experimental groups: 3 SAMiRNA-AREG treatment groups receiving 100, 200, and 300 mg/kg/day and a vehicle control group. Each group was composed of 10 (low and middle dose groups) or 15 (vehicle control and high dose groups) mice from each sex. After 4 weeks of treatment, the animals were sacrificed; however, some animals in the vehicle control and high dose groups (n = 5, recovery groups) were retained and observed for the potential persistence, reversibility, and delayed occurrence of toxic effects.
Mortality and Clinical Observation
All animals were observed twice daily, and clinical characteristics and any mortality were recorded.
Body Weight and Food Consumption
The body weight and food consumption of each mouse was measured at the start of the treatment and once a week during the treatment and recovery periods. The weight gained was calculated as follows: body weight at weighing day − body weight at test initiation day. The amount of food was weighed before it was supplied to each cage and remains were weighed the next day. The difference was calculated and regarded as the daily food consumption (g/mice/day).
Ophthalmology
Ophthalmologic examinations were conducted once prior to treatment for all animals and before the terminal sacrifice for all surviving animals using a binocular indirect ophthalmoscope (Vantage Plus Digital, Keeler LTD., England) and a slit lamp (XL-1; Ohira Co., Niigata, Japan) after the animals were treated with a mydriatic (Mydrin-P, Santen Pharmaceutical Co., Japan).
Urinalysis
To conduct urinalysis, food was withheld from all animals during urine collection, but allowed to take water, and urine samples were collected approximately for 1 to 4 hours using a stainless-steel cage (130 W × 170 L × 100 H mm) on test day 26 or recovery day 12. Urinalysis was carried out to determine pH and specific gravity using a Cobas U411 urine analyzer (Roche Diagnostics, Mannheim, Germany) and Combur 10 TM urine sticks.
Hematology
The blood samples (approximately .5 mL) were collected from the posterior vena cava of mice (the first five animals/sex/group) using a 26-gauge syringe under isoflurane anesthesia on test day 29 or recovery day 15. The whole blood samples were collected into complete blood count bottles containing EDTA-2K (BD, Franklin Lakes, NJ, USA) and analyzed using an ADVIA 2120i hematology analyzer (Siemens Healthcare Diagnostics, Deerfield, IL, USA). The following parameters were measured: red blood cells count, HGB, HCT, mean corpuscular volume, mean corpuscular HGB, mean corpuscular HGB concentration, red blood cell distribution width, reticulocyte, platelet count, and white blood cells count with differential count of neutrophil (%), lymphocyte (LYM, %), eosinophil (%), monocyte (MON, %), basophil (BAS, %), and large unstained cell (LUC, %) as previously described. 22
Serum Biochemistry
The blood samples (approximately .5 mL) were collected from the posterior vena cava of mice (the last five animals/sex/group) using a 26-gauge syringe under isoflurane anesthesia and left at room temperature for at least 30 minute on test day 29 or recovery day 15. The blood samples were centrifuged at 3000 r/min for 10 minute at ambient temperature to separate the serum. The following parameters were evaluated using a Toshiba 120FR chemistry analyzer (Toshiba Medical System, Tokyo, Japan): aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase (ALP), gamma glutamyl transferase, blood urea nitrogen, creatinine, glucose, total cholesterol, total bilirubin, total protein, albumin, albumin/globulin ratio, creatine phosphokinase, triglyceride, calcium, inorganic phosphorus, phospholipid, sodium (Na), potassium, and chloride.
Necropsy and Gross Findings
After blood samples collection for hematology and serum biochemistry analysis, the mice were sacrificed by exsanguination from the abdominal aorta. All animals were subjected to full and detailed gross findings, which included a careful examination of the external surface of the body, all orifices, and the cranial, thoracic, and abdominal cavities and their contents.
Organ Weight
The following organs were weighed to determine the absolute and relative weights based on body weight ratios: salivary glands, adrenal glands, brain, heart, kidneys, liver, pituitary gland, spleen, thymus, lung, thyroid glands (including parathyroid), testes, epididymides, seminal vesicles (with coagulation glands), prostates, uteri (with cervix), and ovaries (with oviduct).
Histopathology
The following organs or tissues were obtained from all animals for histopathological analysis: skin, mammary gland, spleen, pancreas, tongue, esophagus, stomach, jejunum, duodenum, ileum, cecum, colon, rectum, mesenteric lymph node, mandibular lymph node, salivary glands, adrenal glands, pituitary glands, thyroid (including parathyroids), eyes, brain, thymus, heart, aorta, trachea, lung, liver, kidneys, sciatic nerve, spinal cord, skeletal muscle, urinary bladder, sternum, femur, Harderian glands, reproductive organs (testes, epididymides, seminal vesicles, prostates, vagina, uteri, and ovaries), and injection site. Testes and eyes were preserved in Bouin’s fixative and Davidson’s fixative, respectively. The other tissues were fixed with 10% neutral buffered formalin solution. The tissues were routinely processed, embedded in paraffin, and sectioned at 3 to 5 μm. The sections were stained with hematoxylin–eosin for microscopic examination as previously described. 23 All observations were made manually in a blinded manner using a light microscope with × 4, × 10, × 20, and × 40 objective lenses and a × 100 oil immersion lens. The severity of observed histological lesions was presented 5 grades as follows: minimal, slight, moderate, marked, and severe.
Statistical Analysis
Data are expressed as the means ± standard deviation. Variance homogeneity was done using Bartlett’s test. Homogeneous data were analyzed using one-way analysis of variance and the significance of inter-group differences were analyzed using post hoc Dunnett’s test. Heterogeneous data were analyzed using the Kruskal–Wallis test and the significance of inter-group differences between the vehicle control and treated groups was assessed using post hoc Dunn’s rank sum test. To compare the vehicle control and recovery groups, the data were analyzed for homogeneity of variance using the F-test. Homogeneous data were analyzed using the Student’s t-test, and significant differences between vehicle control and recovery groups were assessed using post hoc Dunnett’s test. Heterogeneous data were analyzed using the Kruskal–Wallis test, and the significance differences between vehicle control and recovery groups were assessed using post hoc Dunn’s rank sum test. Comparison analysis between the dose and vehicle control groups was done using the Pristima system (version 7.3., Xybion Medical System Co., NJ, USA). The comparison results were indicated as P values <.05 or .01.
Results
Mortality and Clinical Signs
There were no treatment-related deaths and clinical signs in any treatment groups during the treatment and recovery periods. In the male mice, a single mouse in the 100 mg/kg/day group showed enlarged lymph nodes (mandibular, mesenteric, hepatic, hilar, iliac, renal, and sciatic). Histopathological examination revealed that the lesion was non-Hodgkin’s lymphoma, which was considered as a spontaneous finding. Thus, the all test data for this animal were excluded in the results of the present study.
Body Weight Changes
As shown in Figure 1, there were no treatment-related effects on body weight with any dose during the treatment and recovery periods. Body weight changes in mice treated with self-assembled-micelle inhibitory RNA-targeting amphiregulin for 4 weeks. Values are presented as means ± SD.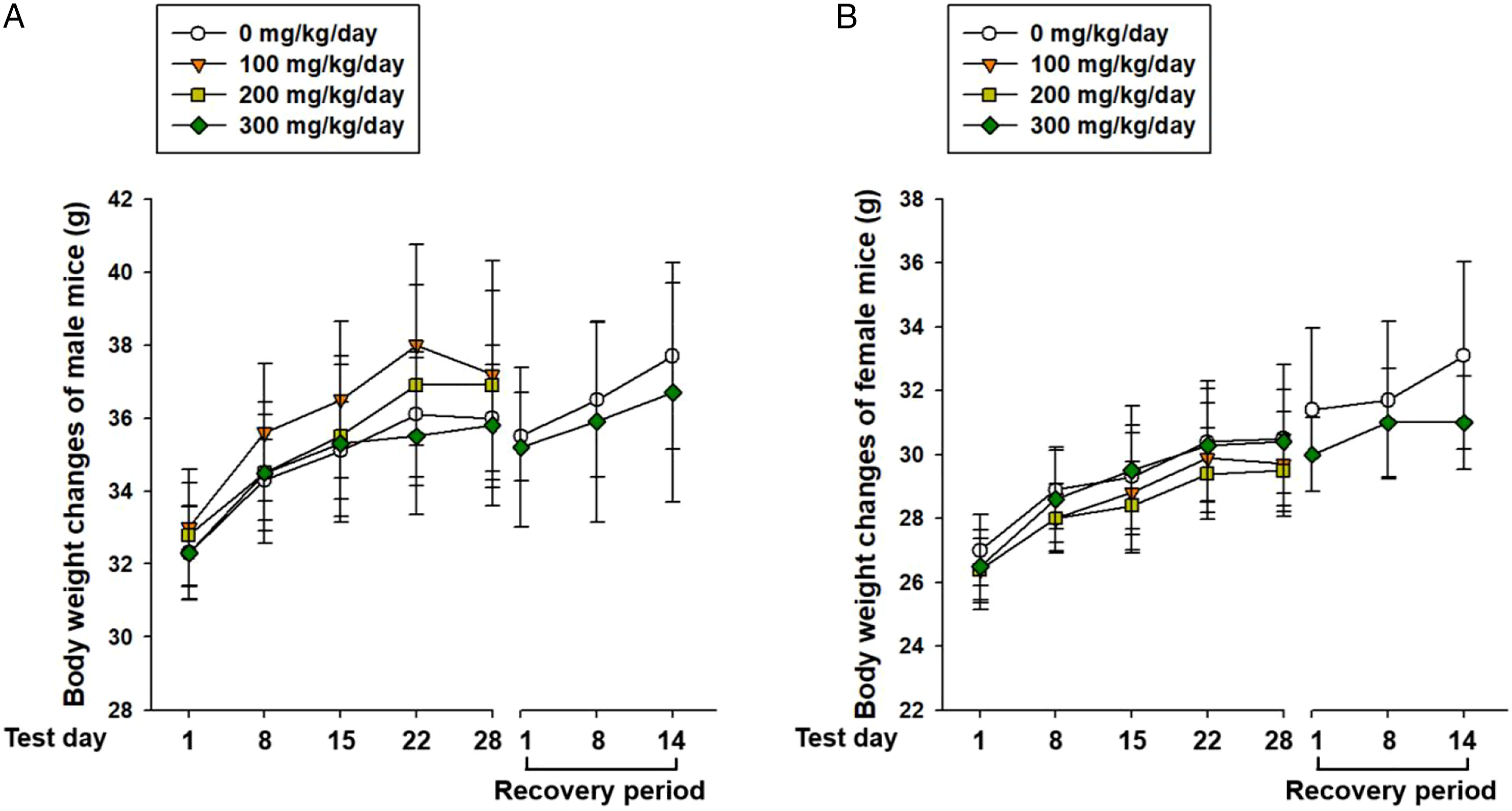
Food Consumption
Daily Food Consumption of Male and Female Mice Treated With SAMiRNA-AREG for 4 Weeks.
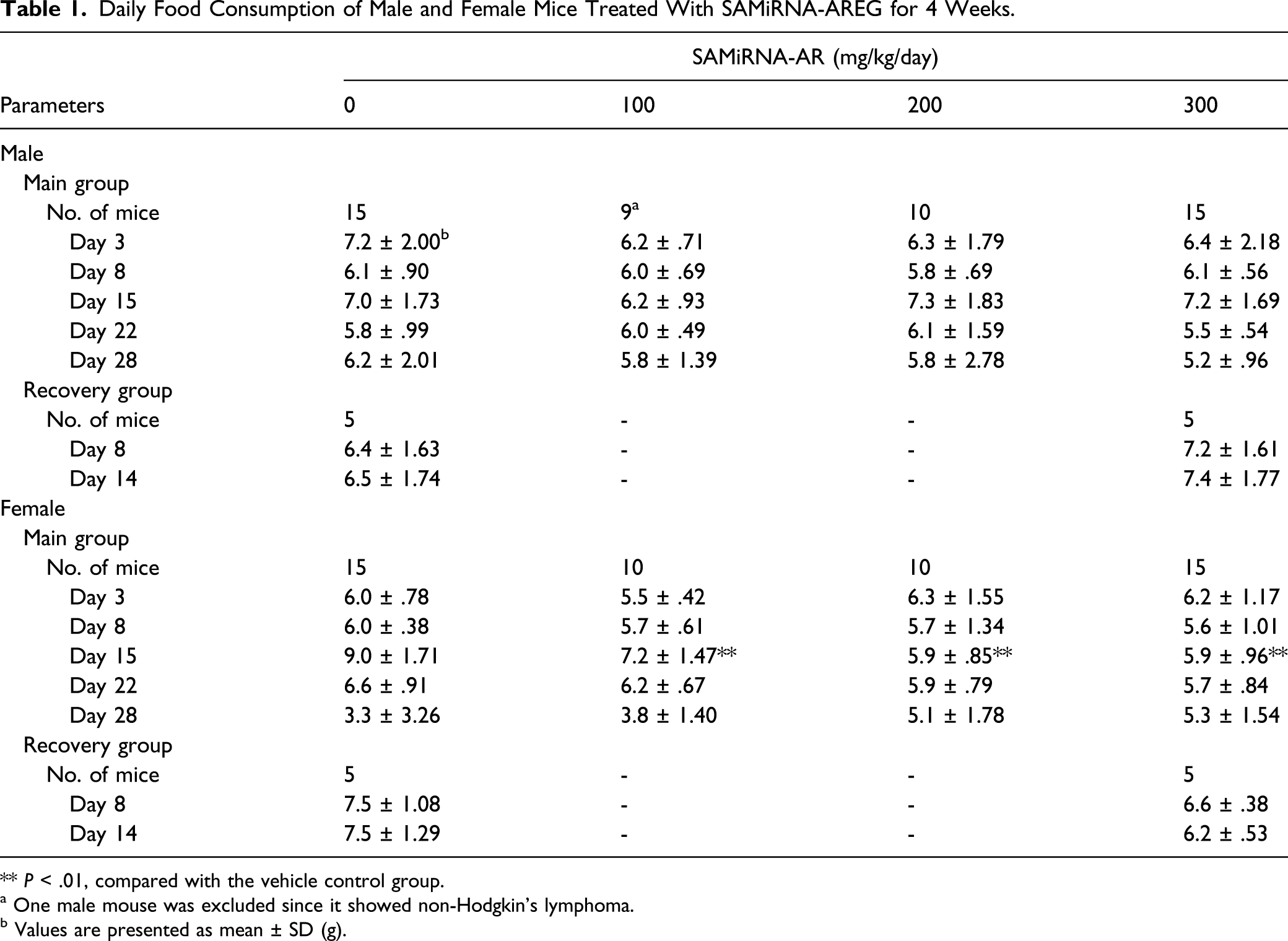
** P < .01, compared with the vehicle control group.
a One male mouse was excluded since it showed non-Hodgkin’s lymphoma.
b Values are presented as mean ± SD (g).
Ophthalmology
Ophthalmologic examination did not show any treatment-related ocular lesions in any of the mice treated with SAMiRNA-AREG (data not shown).
Urinalysis
Urinalysis did not show any treatment-related changes in any treatment groups for both sexes (data not shown).
Hematology
Hematological Values of Male and Female Mice Treated With SAMiRNA-AREG for 4 Weeks.
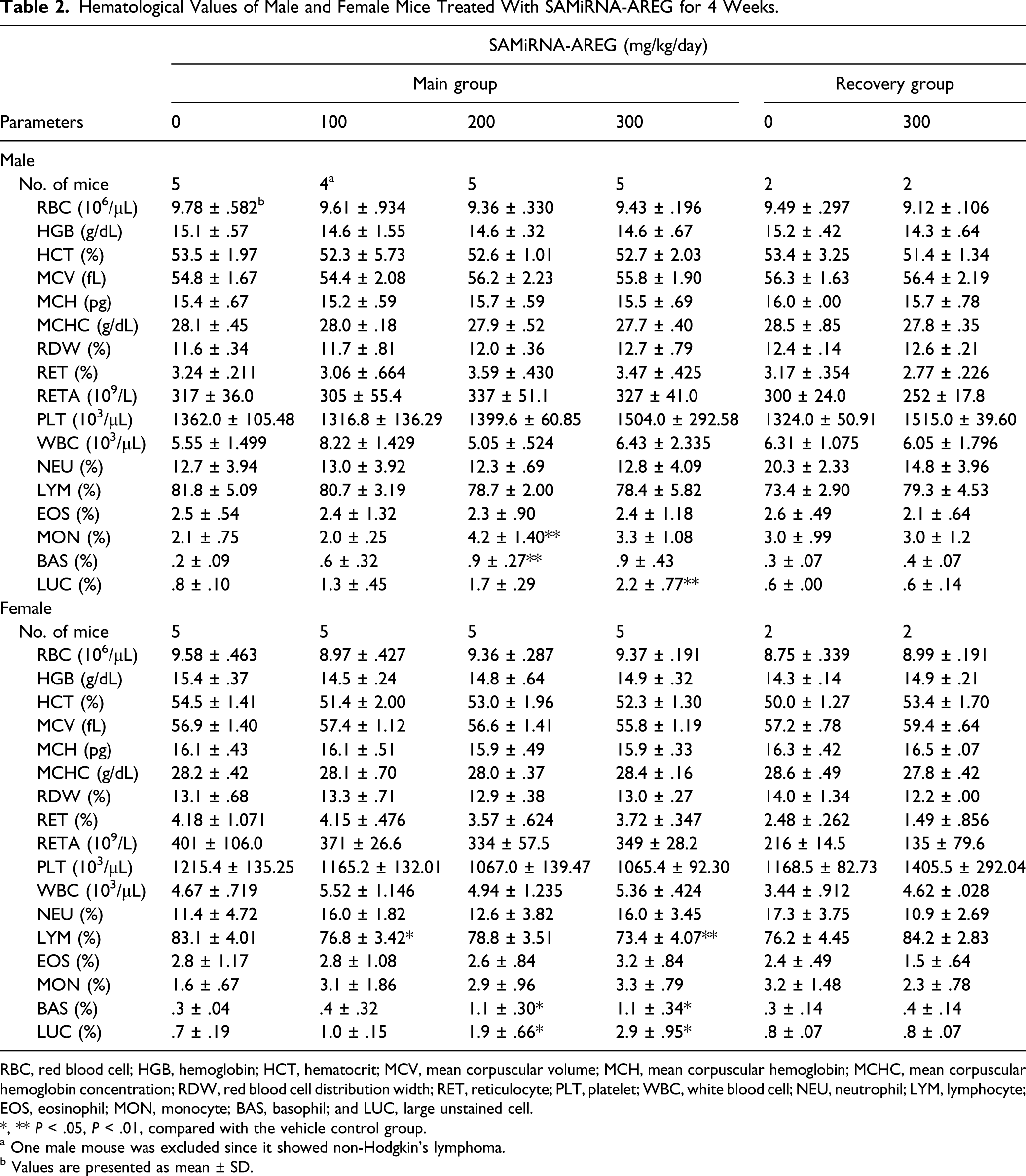
RBC, red blood cell; HGB, hemoglobin; HCT, hematocrit; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; RDW, red blood cell distribution width; RET, reticulocyte; PLT, platelet; WBC, white blood cell; NEU, neutrophil; LYM, lymphocyte; EOS, eosinophil; MON, monocyte; BAS, basophil; and LUC, large unstained cell.
*, ** P < .05, P < .01, compared with the vehicle control group.
a One male mouse was excluded since it showed non-Hodgkin’s lymphoma.
b Values are presented as mean ± SD.
Serum Biochemistry
Serum Biochemical Values of Male and Female Mice Treated With SAMiRNA-AREG for 4 Weeks.
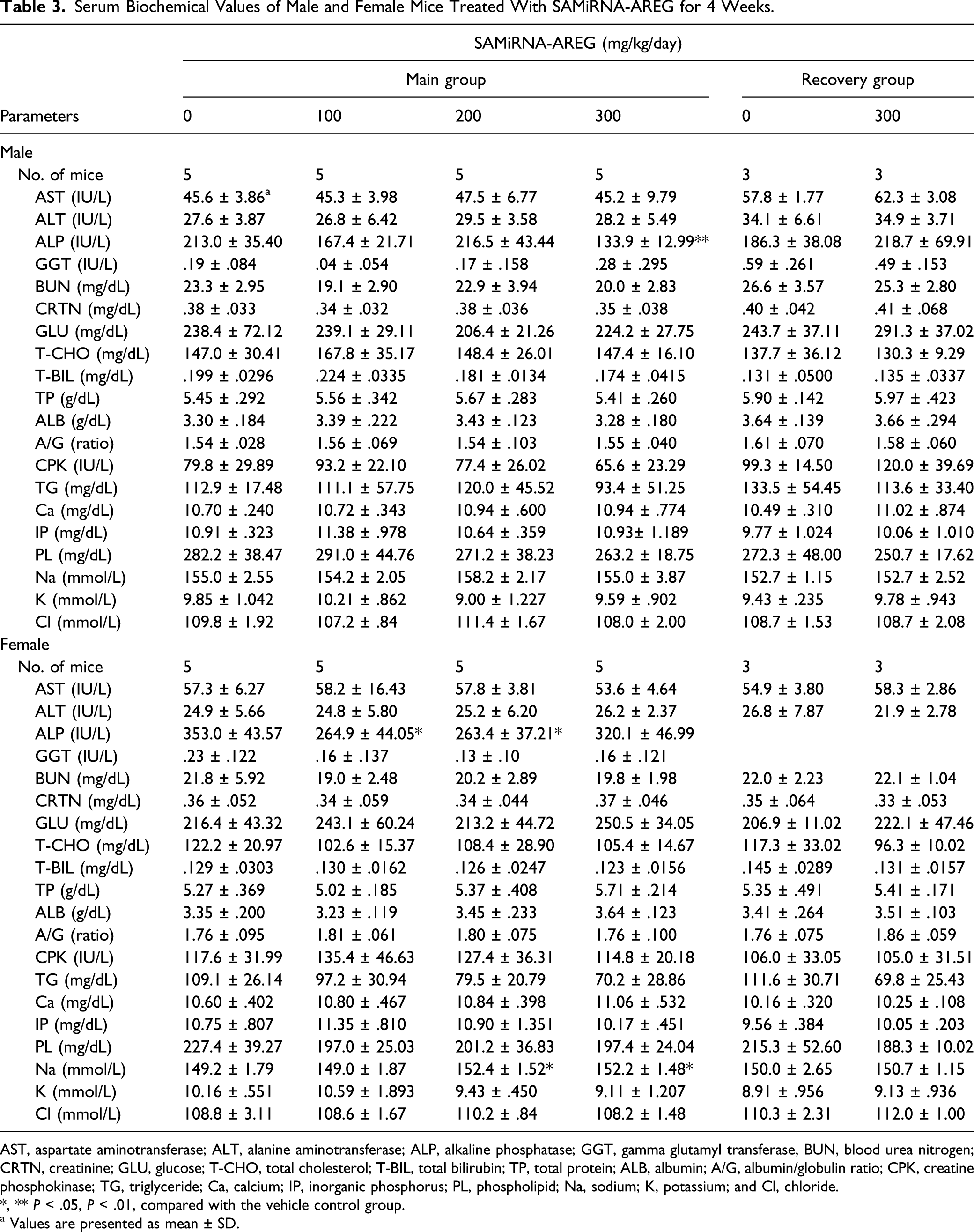
AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; GGT, gamma glutamyl transferase, BUN, blood urea nitrogen; CRTN, creatinine; GLU, glucose; T-CHO, total cholesterol; T-BIL, total bilirubin; TP, total protein; ALB, albumin; A/G, albumin/globulin ratio; CPK, creatine phosphokinase; TG, triglyceride; Ca, calcium; IP, inorganic phosphorus; PL, phospholipid; Na, sodium; K, potassium; and Cl, chloride.
*, ** P < .05, P < .01, compared with the vehicle control group.
a Values are presented as mean ± SD.
Gross Findings
The gross findings observed at the scheduled necropsy of male mice included pale discoloration in the kidneys (1/9) and enlarged liver (1/9) in the 100 mg/kg/day group (data not shown). The pale discoloration in the kidneys (1/10) and enlarged spleen (2/10) were also observed in the male 300 mg/kg/day group. For the female mice, cyst in the ovaries (1/10) was observed in the 100 mg/kg/day group. In the recovery groups, there were no notable changes in the vehicle control and high dose groups of both sexes (data not shown).
Organ Weight
Absolute and Relative Organ Weights of Male Mice Treated With SAMiRNA-AREG for 4 Weeks.
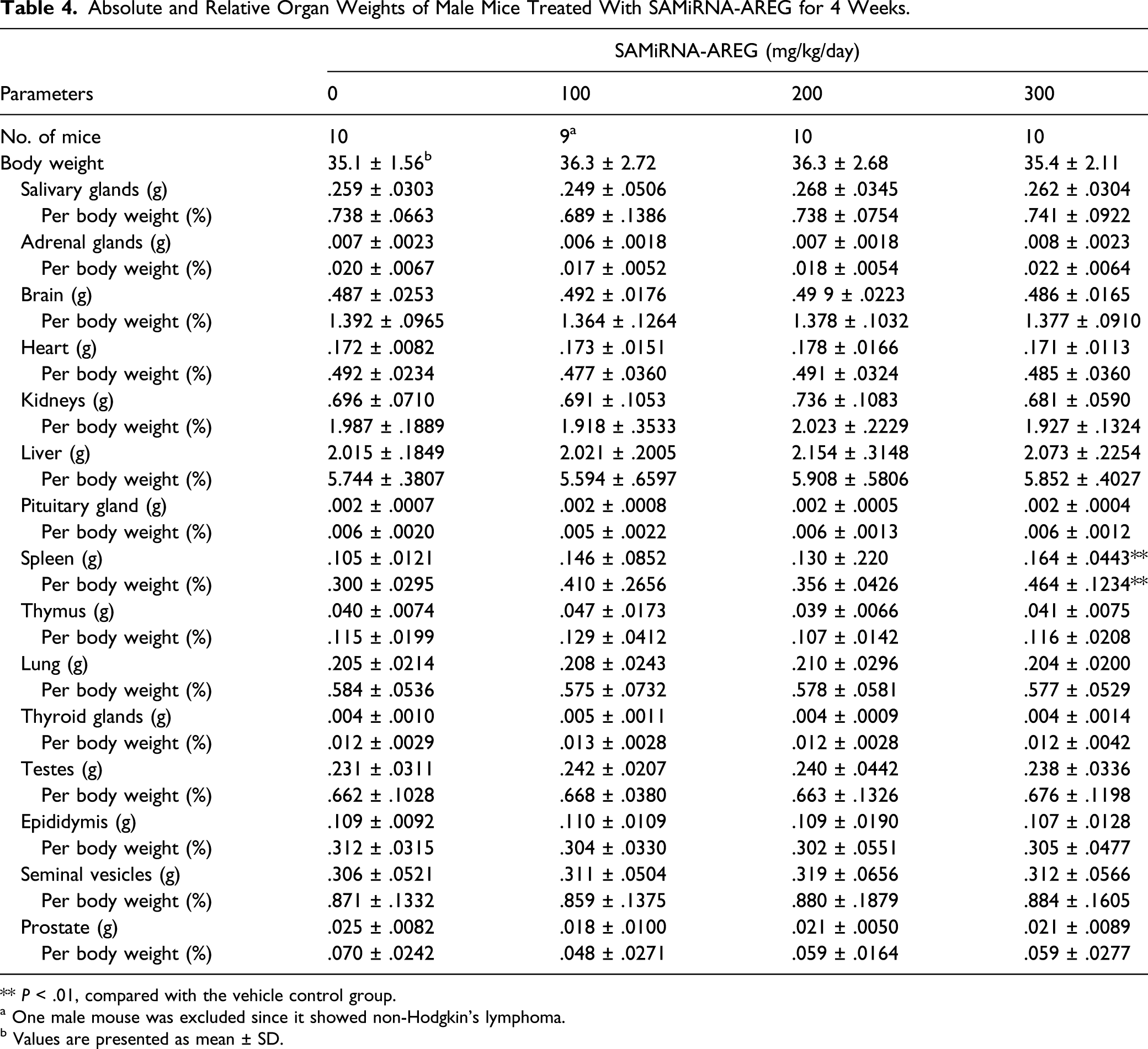
** P < .01, compared with the vehicle control group.
a One male mouse was excluded since it showed non-Hodgkin’s lymphoma.
b Values are presented as mean ± SD.
Absolute and Relative Organ Weights of Female Mice Treated With SAMiRNA-AREG for 4 Weeks.
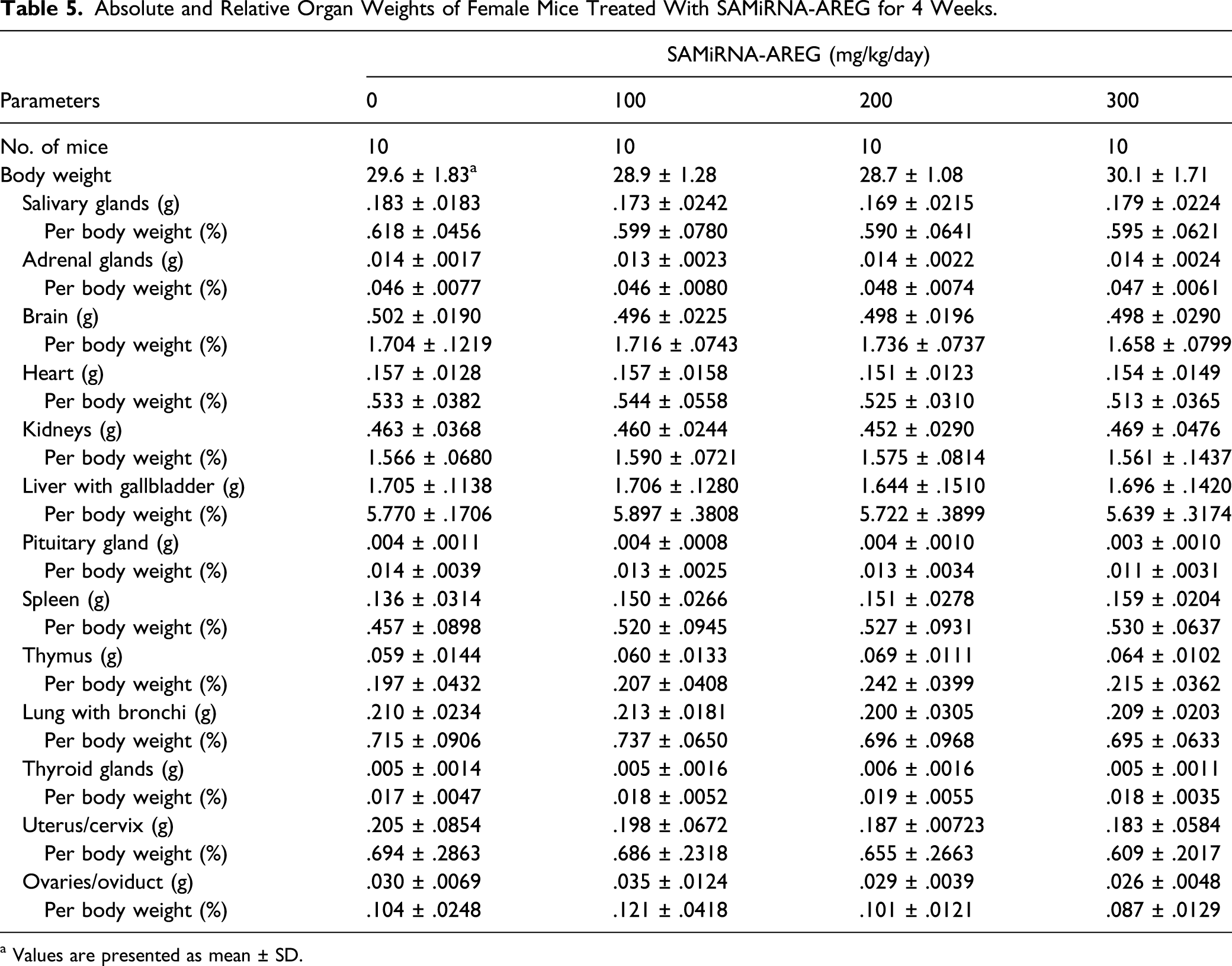
a Values are presented as mean ± SD.
Histopathological Findings
Histopathological Findings in Male Mice Treated With SAMiRNA-AREG for 4 Weeks.
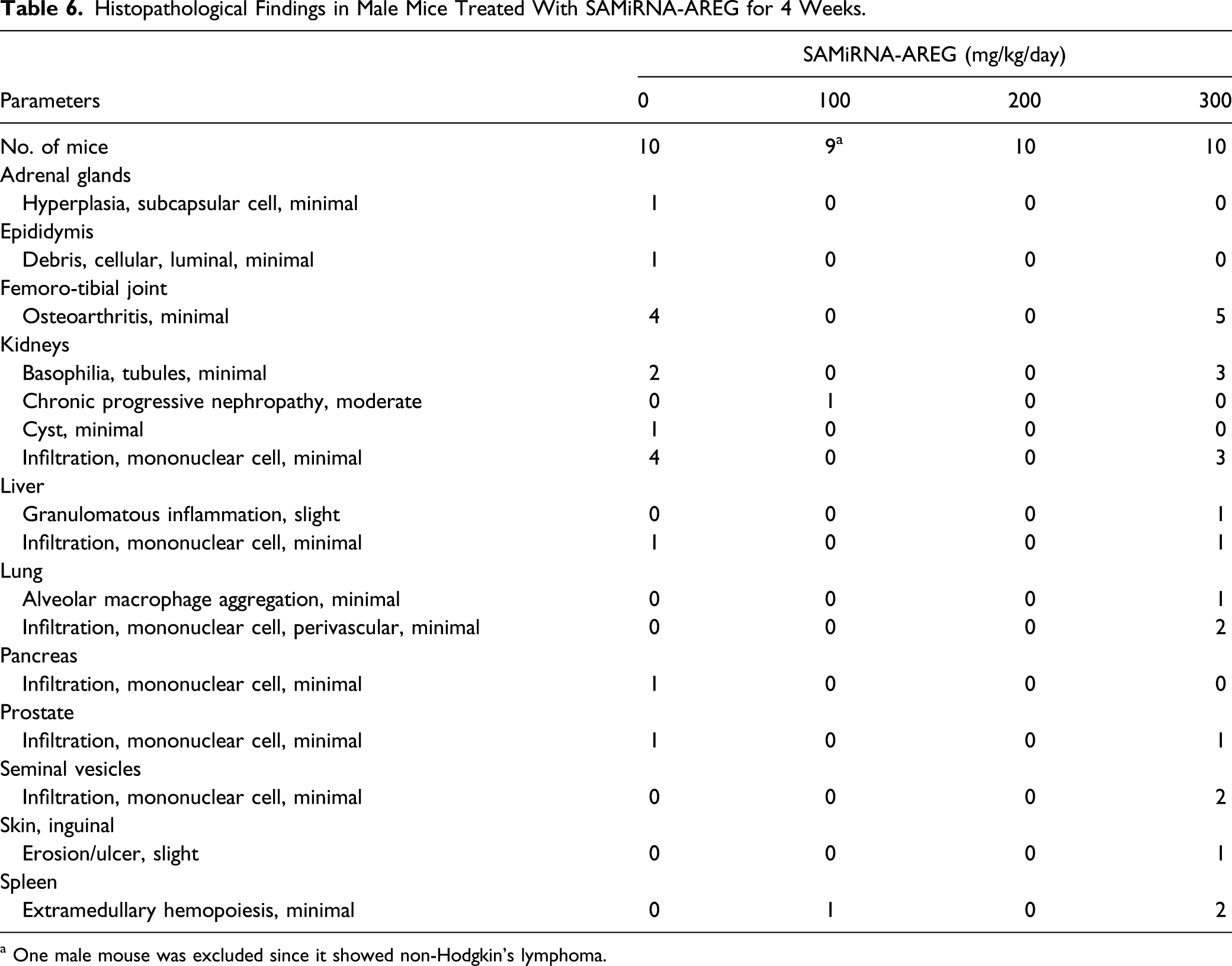
a One male mouse was excluded since it showed non-Hodgkin’s lymphoma.
Histopathological Findings in Female Mice Treated With SAMiRNA-AREG for 4 Weeks.
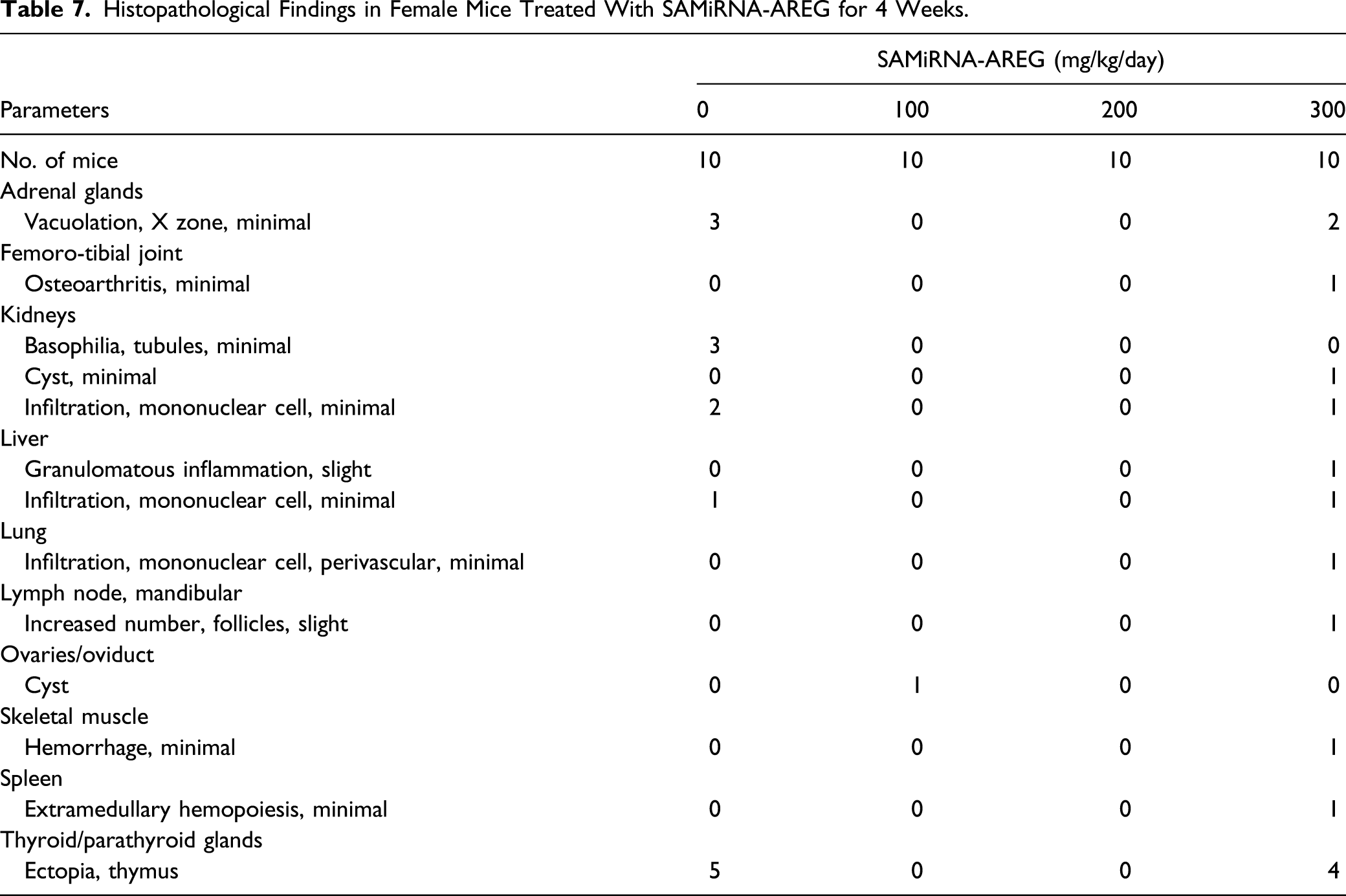
Discussion
The present study aimed to evaluate the potential toxicity of SAMiRNA-AREG by a 4-week repeated intravenous injection to ICR mice at dose levels of 0, 100, 200, or 300 mg/kg/day. The recovery groups from the vehicle control and 300 mg/kg/day groups were observed for a 2-week recovery period. The results of this study showed that SAMiRNA-AREG did not cause treatment-related adverse effects at dose levels up to 300 mg/kg/day in mice after 4-week repeated intravenous doses.
There were no treatment-related clinical signs of toxicity and mortality with any of the doses tested for both sexes. Likewise, no treatment-related body changes were found with any of the doses tested during the treatment and recovery period. The significant decrease in food consumption on test day 15 observed in the female treatment groups was not considered to be caused by the test reagent since this finding was observed only in the female treatment groups temporarily and the body weight in the treatment groups was not affected during the study period. Furthermore, the food consumption in the vehicle control group was increased on day 15 relative to any other time period. Thus, we concluded that the decreased food consumption observed in the female treatment groups was probably an incidental finding and was not of toxicological significance. In addition, based on the urinalysis and ophthalmological examination results, SAMiRNA-AREG, at doses up to 300 mg/kg/day, did not cause any abnormal changes, in the SAMiRNA-AREG–treated groups for both sexes.
In the dose range–finding study, a significant decrease in the HGB and HCT of the male 300 mg/kg/day group was considered an accidental change because these changes were not observed in the present study. The percentage of LUC is a differential count parameter measured by certain routine hematology analyzers and reflects peroxidase-negative cells and activated LYMs. It was reported that LUC is a potentially important parameter in the assessment of immune activation levels in HIV infection. 24 In the present study, the significant increases in the percentages of BAS and LUC observed in the 200 and 300 mg/kg/day dose groups were considered as treatment-related effects because these findings showed a dose–response relationship and were observed in the both sexes. However, they were of no toxicological significance because the changes were only marginal25-27 and were not accompanied by other relevant results. Moreover, the increased percentages of BAS and LUC were not observed at the end of the 2-week recovery period. Similarly, in a previous study, 3 times intratracheal instillation of SAMiRNA-AREG 3 mg/kg at 48 hours interval did not cause any changes in hematological and serum biochemical parameters in mice. 9 In a monkey study, no treatment-related changes were observed in the percentages of BAS and LUC when SAMiRNA-AREG was administered intravenously once a week for 4 weeks at dose levels of 0, 25, 50, or 100 mg/kg/day (unpublished data). The significant increase of MON percentage observed in the male 200 mg/kg/day group and the significant decrease of LYM percentage observed in the female 100 and 300 mg/kg/day groups were of no toxicological significance because these changes were observed only in the one sex and did not exhibit a dose–response relationship, and the values were within the limits of normal biological variation for mice.25,28
The significant decrease of serum ALP levels observed in the male 300 mg/kg/day group and the female 100 and 200 mg/kg/day groups were not considered as treatment-related effects because they did not show a dose–response relationship and were not accompanied by correlative histopathological changes. The significant increase of Na level in the female 200 and 300 mg/kg/day groups was also of doubtful toxicological significance because these findings were within the limits of normal mice biological variations,25,26,29 and there were no changes in other related parameters.
The abnormal gross findings, including pale discoloration of the kidneys and enlargement of the liver and spleen observed in the male 100 and 300 mg/kg/day groups and cyst of the ovaries observed in the female 100 mg/kg/day group, were considered incidental findings, since these findings occurred infrequently, were not accompanied by correlated pathological alterations, and did not exhibit a dose–response relationship.
There are several potential causes of increased spleen weight, including hematologic malignancies, inflammatory diseases, liver diseases, venous thrombosis, splenic congestion, cytopenias, splenic sequestration, connective tissue diseases, infiltrative diseases, and focal lesions.30,31 In the present study, the significant increases of absolute and relative weights of spleen observed in the male 300 mg/kg/day group were closely related to treatment of SAMiRNA-AREG because these findings were accompanied by a correlated histopathological findings, such as an increased incidence of extramedullary hemopoiesis in the spleen. The increased spleen weight observed in this study was consistent with the result of a dose range–finding study of SAMiRNA-AREG conducted in this laboratory. However, the histopathological changes in the spleen extramedullary hemopoiesis were minimal and occurred at a low incidence. Moreover, this finding was no longer observed after the 2-week recovery period. Furthermore, the weight change in the spleen was observed only in the male mice and did not exhibit a dose–response relationship. Therefore, the weight change in the spleen was not interpreted to be of toxicological significance.
The major histopathological changes observed in this study included slight osteoarthritis in femoro-tibial joint, basophilic tubule, and mononuclear cell infiltration in kidneys in the male 300 mg/kg/day group and ectopic thymus in thyroid glands in the female 300 mg/kg/day group. In a previous study, normal mice had mild osteoarthritic changes of up to 45% referred as aging-related osteoarthritis. 32 It has been reported that tubular basophilia and glomerular nephritis spontaneously occurred in ICR mice about 6% 33 and 14%, 34 respectively. In another study, ectopic thymus in thyroid/parathyroid glands occurred in more than 21% of mice. 35 However, in this study, since these changes were not accompanied by corresponding changes in other parameters and were also observed in the vehicle control group at a high incidence, they were considered to be of no toxicological significance. The mononuclear cell infiltration in the kidneys, liver, pancreas, or prostate was observed in the SAMiRNA-AREG–treated group. However, these histopathological changes were considered incidental findings because they occurred at a low incidence, were observed in the vehicle control group, and did not show a dose–response relationship. The other histological changes observed in the SAMiRNA-AREG–treated groups were not considered treatment-related effects because they occurred at a low incidence, did not exhibit a dose–response relationship, and were no longer observed after the 2-week recovery period.
In conclusion, the 4-week repeated intravenous dose of the SAMiRNA-AREG in ICR mice caused an increase in the percentages of BAS and LUC at ≥ 200 mg/kg/day of both sexes and an increase in the absolute and relative spleen weights at 300 mg/kg/day of male mice. However, these findings were of no toxicological significance because they were within the normal limits of the reference data and were not accompanied by other relevant changes. Moreover, they were no longer observed at the end of the 2-week recovery period. Under the present experimental conditions, the NOAEL of SAMiRNA-AREG was ≥300 mg/kg/day for both sexes and the target organs were not identified. The NOAEL obtained in this study corresponds to about 750 times of the anticipated human clinical dose of SAMiRNA-AREG, that is, .4 mg/kg/day. 9 The findings from these non-clinical studies suggest a favorable safety profile of the SAMiRNA-AREG fibrotic therapeutics and support its further clinical development.
Footnotes
Author Contributions
Hyeon-Young Kim and Tae Rim Kim contributed equally to this work as co-first authors. Hyeon-Young Kim and Tae Rim Kim: writing—original draft; Sung-Hwan Kim: conceptualization; In-Hyeon Kim and Je-Oh Lim: investigation and data analysis; Jun-Hong Park and Sungil Yun: experimental planning; In-Chul Lee: conceptualization; Han-Oh Park: project administration and funding acquisition; and Jong-Choon Kim: funding acquisition, supervision, and writing—review and editing. All authors gave final approval and agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Civil and Military Dual-use Technology Program (14-CM-EB-09; NTIS 1695004819) and Korea Drug Development Fund (KDDF) funded by Ministry of Science, ICT, and Future Planning (MSIP); the Ministry of Trade, Industry, and Energy (MOTIE); and the Ministry of Health and Welfare (MOHW) (Grant No KDDF-201706-03, Republic of Korea). It was also supported by Bioneer Corporation and siRNAgen Therapeutics Research Fund.
