Abstract
The highly conserved human and mouse SLC39A8 gene encodes the divalent cation/bicarbonate symporter ZIP8 expressed ubiquitously in most cell types. Our bacterial artificial chromosome-transgenic BTZIP8-3 line has 3 additional copies of the Slc39a8 gene in addition to its constitutive diploid pair found in wild-type (WT) mice. In liver, kidney, lung, testis, gastrointestinal tract, and brain, BTZIP8-3 mice are known to express ∼2.5 times greater amounts of ZIP8, compared with WT mice. Herein we administered cadmium chloride (CdCl2) in drinking water (100 mg/L through week 2, 200 mg/L through week 4, 400 mg/L through week 8, 800 mg/L through week 12, and 1600 mg/L through week 20, when the experiment was concluded). We postulated that Cd uptake and distribution––and, therefore, toxicity in certain tissues––would be enhanced in BTZIP8-3, compared with WT mice. BTZIP8-3 and WT groups ingested comparable amounts of Cd. Compared with WT, BTZIP8-3 mice showed tissue specific: increases in Cd, zinc, and manganese content and decreases in calcium content. Both Cd-exposed BTZIP8-3 and WT were similar in lower urinary pH; increased plasma alanine and aspartate aminotransferase activities; elevated iron and copper content in liver, kidney, lung, and testis; and higher blood urea nitrogen and kidney weight. Histological changes in liver, kidney, lung, and testis were minimal. In summary, at the daily oral Cd exposures chosen for this study, 5 versus 2 Slc39a8 gene copies result in no differences in Cd toxicity but do cause differences in tissue-specific content of Cd, zinc, manganese, calcium, iron, and copper.
Keywords
Introduction
Cadmium (Cd, Cd2+) is a nonessential heavy metal, whose environmental levels have greatly increased since the industrial revolution and have made a significant contribution to environmental contamination. 1 In 2007, the Agency for Toxic Substances Disease Registry (ATSDR) posted a Comprehensive Environmental Response, Compensation, and Liability Act List of Hazardous Substances in the Environment 2 in which Cd was ranked #7, with regard to its abundance in toxic waste dump sites. Chemicals in these toxic waste dump sites are well known to leach into drinking water, and humans are major recipients at the end of the food chain.
Cadmium is both toxic and carcinogenic. Acute Cd doses can be toxic to virtually any tissue lung, bone, central nervous system, gastrointestinal (GI) tract, liver, ovary, testis, placenta, and developing embryo. 3 Chronic low Cd exposure principally causes renal proximal tubular metabolic acidosis and osteomalacia (renal Fanconi syndrome). Cd accumulates with age, predominantly in kidney. 4 Cd is classified by International Agency for Research on Cancer (IARC) as a “category I” human lung carcinogen. Populations at highest risk for accidental Cd consumption and therefore Cd-induced lung cancer and renal disease include cigarette smokers, those ingesting large amounts of contaminated shellfish, and those with anemia, malnutrition, and/or with infections. 3,5 Dietary Cd has also been recently implicated in osteoporosis and increased risk of bone fractures. 6
This laboratory has identified and characterized the mouse Slc39a8 gene that encodes the ZIP8 transporter. 7 Although the endogenous function of ZIP8 was first identified as a zinc-bicarbonate symporter, 8 the essential metals iron and manganese can also be substrates, 4,9,10,11 and ZIP8 exhibits a strikingly high affinity for the nonessential metal Cd. 4,9,10,11 The ZIP8 is ubiquitous, located in virtually all cell types of the body but is highest in apical membranes of the renal proximal tubule epithelial cells, pulmonary alveolar epithelial cells, and testis vascular endothelial cells. 4,7,12
Using transgenic techniques with a bacterial artificial chromosome (BAC) clone, this laboratory created and characterized the BAC-transgenic BTZIP8-3 mouse line that carries 3 additional Slc39a8 genes in addition to its normal diploid 2 copies. Interestingly, the amounts of ZIP8 messenger RNA (mRNA) and protein in liver, kidney, lung, testis, GI tract, and brain of BTZIP8-3 mice are ∼2.5-fold greater than that of wild-type (WT) mice. 12 Subcutaneous Cd treatment results in acute renal failure and signs of proximal tubular damage in the BTZIP8-3 but not nontransgenic littermates. 12
In human populations, Cd uptake occurs primarily by way of the GI tract and lung. 3 ,4,5,13 We therefore wished to determine whether Cd in the drinking water might lead to greater risk of Cd-induced toxicity in BTZIP8-3 (due to 2.5 times more ZIP8 in all tissues), compared with WT mice. The present study explores this hypothesis.
Materials and Methods
Chemicals
Cadmium chloride (CdCl2) and sucrose were purchased from Fisher Scientific (Pittsburgh, Pennsylvania).
Animals
All mouse experiments were conducted in accordance with the National Institutes of Health standards for the care and use of experimental animals and the University of Cincinnati Medical Center Institutional Animal Care and Use Committee. Both adult male and female BTZIP8-3 mice and their nontransgenic littermates (referred throughout this report as “WT”) were used in all experiments. The BTZIP8-3 mouse line and nontransgenic littermates were generated on a C57BL/6J background. 12 Animals had free access to standard rodent chow and drinking water. Groups of 3 to 4 mice (male and female for both genotypes) were given progressively increasing concentrations of oral CdCl2 in their drinking water. Matching numbers of control animals of the same genotype and sex were provided with tap water without any CdCl2.
Treatment
We wanted to mimic a long-term, chronic Cd exposure situation typically observed for humans exposed to high levels of environmental Cd. We thus designed a 20-week experiment in mice and exposed them to escalating Cd levels in drinking water. Therefore, the following oral CdCl2 regimen was carried out: 100 mg/L of drinking water (weeks 1 and 2), 200 mg/L (weeks 3 and 4), 400 mg/L (weeks 5 and 6), 800 mg/L (weeks 7-16), and 1600 mg/L (weeks 17-20). Water consumption was measured twice weekly. Upon increasing the CdCl2 concentration to 800 mg CdCl2/L, to the drinking water we added 2% sucrose (wt/vol), in order to mask the metallic taste of Cd and be assured the mice would continue drinking Cd-containing water ad libitum. Control groups also received the same amounts of sucrose. Once a week the quantities of Cd ingested and body weights were measured, and urine was collected by placing animals in metabolic cages for 16 h (10 pm until 2 pm) and given access to 2% sucrose to promote water consumption and diuresis. Urine was collected on ice at 8 and 16 hours and combined from each entire group (N = 4-8) for measurements. Ice was used to preserve enzymatic activities that would be measured later and was changed at the 8-hour collection time-point.
Weekly Enzyme and Small-Molecule Metabolite Measurements to Assess Kidney Function
Plasma alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were measured, following the manufacturer’s suggested protocols and using Hitachi AST (SGOT) and ALT (SGPT) reagent kits, respectively, from Pointe Scientific (Canton, Michigan). Blood urea nitrogen was determined using the Stanbio enzymatic procedure 2050 (Stanbio, Boerne, Texas). Urinary and plasma creatinine were measured by the Stanbio direct creatinine Liquicolor procedure (no. 0420). Urinary N-acetyl-β-
Urinary γ-glutamyltranspeptidase (GGT) was measured using glutamyl derivatives of aromatic amines as substrate;
Sample Preparation for Determining Tissue Metal Concentrations
At completion of the 20-week experiment, tissues (∼50-100 mg of kidney, liver, lung, or testis) were placed in septum-sealed glass tubes and treated with 0.5 mL of 50% (v/v) HNO3 and subjected to microwave digestion (150 W; 1.00-minute ramp time, 2.00-minute hold time; 250 psi; 170°C), using the CEM Explorer system equipped with the Discover autosampler (Matthews, North Carolina). 15 Digested samples were then diluted to 10.0 mL with doubly distilled water containing a yttrium internal standard (100 µg/L).
Inductively Coupled Plasma Mass Spectrometry
An Agilent 7500ce (Agilent Technologies, Tokyo, Japan) inductively coupled plasma mass spectrometer, equipped with shielded-torch and collision/reaction-cell technology was used for the element-specific detection of 111Cd plus 28 other elements comprising a total of 44 other isotopes. The collision/reaction cell consisted of an octopole-ion guide, operated in “rf-only” mode, which also served for the removal of polyatomic interferences. Forward power was 1500 W (with shielded torch); plasma gas-flow rate was 15.6 L/min; auxiliary gas-flow rate was 1.0 L/min; carrier gas-flow rate was 1.20 L/min; the nebulizer was glass-expansion microcentric; spray chamber was ∼2°C (Scott double channel); sampling depth was 6 mm; sampling and skimmer cones comprised nickel; and dwell time was 0.1 s. Isotopes monitored included 6,7 Li, 9 Be, 23 Na, 24,26 Mg, 27Al, 39K, 43,44Ca, 51V, 52,53Cr, 55Mn, 56,57Fe, 59Co, 60,62Ni, 63,65Cu, 66,68Zn, 69,71Ga, 75As, 77,78,80Se, 85Rb, 86,88Sr, 107,109Ag, 111Cd, 115In, 133Cs, 137,138Ba, 203,205Tl, 207,208Pb, 209Bi, 238238U, and 89Y (internal standard). The octopole-reaction system used helium (flow optimized before each experiment).
Statistical Analysis
Statistical significance between groups was determined by 2-way analysis of variance among groups, using the Tukey test for post hoc analysis. All assays were performed in duplicate or triplicate, and repeated at least twice. Statistical analyses were performed using SAS statistical software (SAS Institute Inc, Cary, North Carolina) and Sigma Plot (Systat Software, Inc, Point Richmond, California). P values of <.05 were regarded as statistically significant.
Results
Amount of Cd Ingested
We first wished to establish that, when provided with Cd-laced drinking water ad libitum, both WT and BTZIP8-3 males and females consumed similar amounts of oral Cd; this was the case for the first 15 weeks––with more variability seen thereafter (Figure S1). Total water intake for all groups decreased in week 6 (data not shown), which we surmised might be caused by the mice refusing to drink due to tasting the metal; starting with week 7, we therefore decided to add 2% sucrose (wt/vol) to the Cd-containing (as well as control) drinking water, and this appeared to mask the metallic taste and total water intake returned to normal.
For reasons not clear, in week 16 both male and female BTZIP8-3 groups took in substantially less Cd, but total weekly Cd consumption was again no different among the groups for weeks 18 to 20 (Figure S1). In weeks 19 and 20 for WT females and week 20 for BTZIP8-3 females, total weekly Cd consumption declined somewhat; however, these animals did not appear overtly ill. No statistically significant differences in Cd ingestion were found: the total amount of Cd ingested over the entire 20-week experiment was ∼237 mg Cd per mouse for WT and ∼219 mg for BTZIP8-3 mice.
Effects of Oral Cd on Urinary pH
No significant differences were found in urinary glucose, ketones, protein, or blood (data not shown). From week 8 onward (Figure 1), urinary pH in both Cd-exposed WT and BTZIP8-3 mice was significantly decreased, compared with that in the control groups. These data are consistent with the well known effects of chronic Cd causing renal proximal tubule metabolic acidosis. 4,16
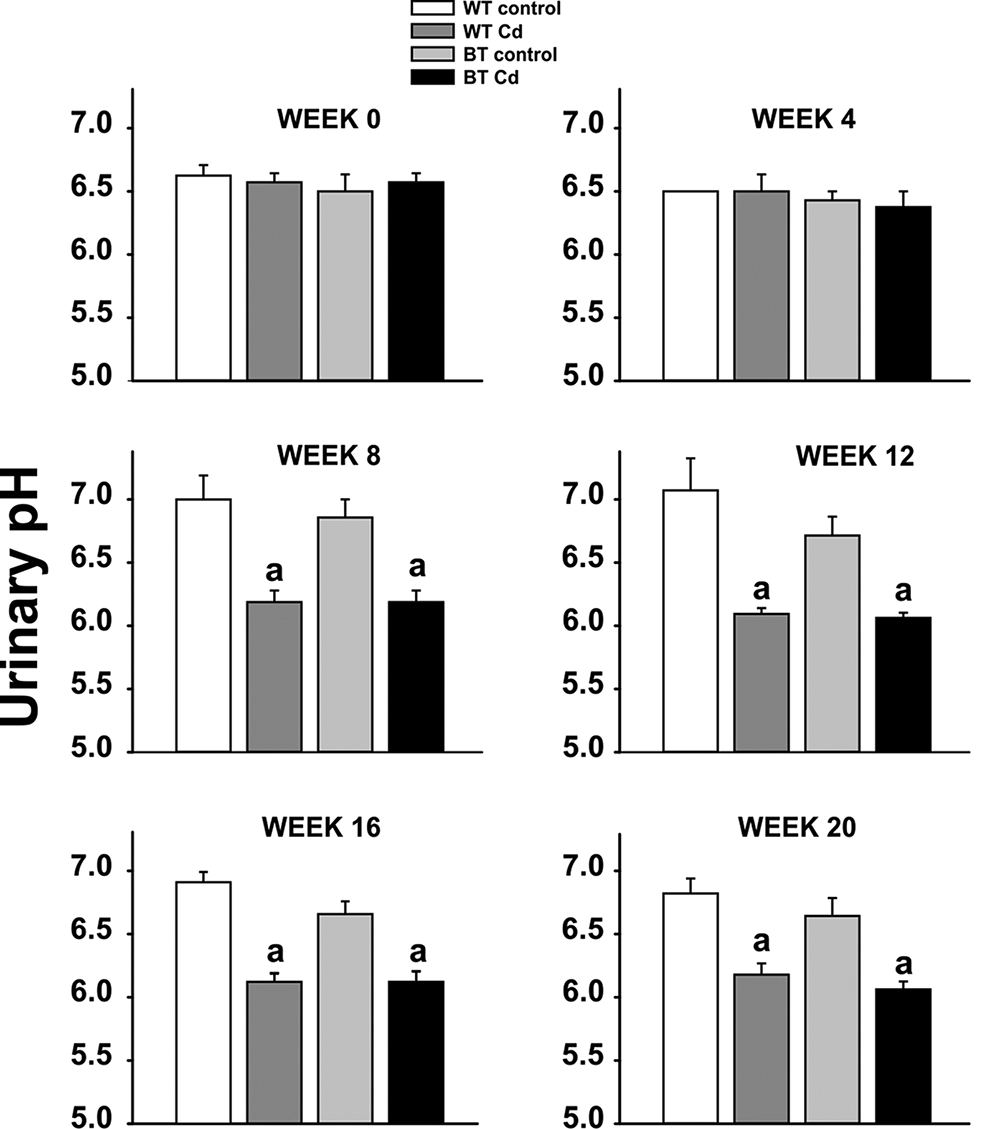
Urinary pH (mean ± standard error of the mean [SEM], brackets) shown at representative 4-week intervals for the 20-week experiment. The 4 groups (N = 7 or 8) included wild-type (WT), no Cd; WT, exposed to Cd at increasing doses; BTZIP8-3 (BT), no Cd; BT, exposed to Cd at increasing doses. aSignificantly different (P < .05) from control with same genotype. bSignificantly different (P < .05) from the other genotype’s matched experimental group.
Effects of Oral Cd on Plasma and Urinary Enzyme Levels
Plasma ALT and AST levels are additional biomarker indicators of early toxicity. From week 8 onward (Figure 2A), plasma ALT values in both Cd-exposed WT and BTZIP8-3 mice were significantly elevated, compared with that in the control groups. From week 4 onward for Cd-exposed BTZIP8-3 and from week 8 onward for Cd-exposed WT (Figure 2B), plasma AST levels were significantly elevated, compared with that in the control groups. Although not statistically significant, a trend was seen in urinary GGT activity being always higher in both Cd-exposed WT and BTZIP8-3 than in non-Cd-exposed mice (Figure S2).
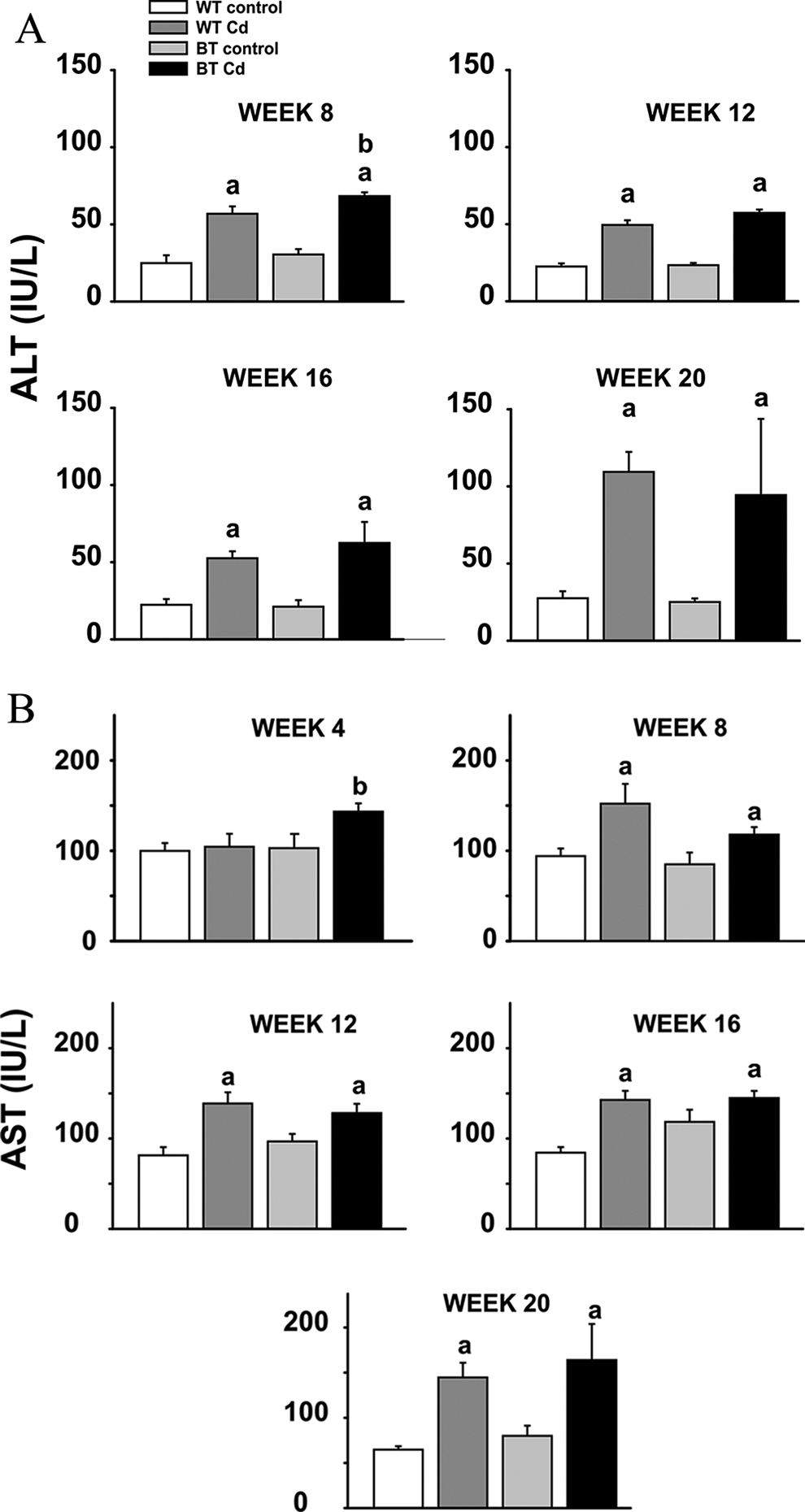
Plasma ALT and AST activities (IU/L) shown at representative 4-week intervals for the 4 groups over the 20-week experiment. A, Plasma alanine aminotransferase (ALT) from week 8 through week 20. B, Plasma aspartate aminotransferase (AST) from week 4 through week 20. The 4 groups, group size, symbols, and P values are identical to those described in Figure 1. ALT indicates alanine aminotransferase.
Serum and urinary creatinine and urinary NAG activities were also determined weekly throughout the 20-week experiment, and––although a trend was seen at some time points, that is, Cd-exposed groups higher than non-Cd-exposed controls––no statistically significant differences among the groups were found (data not shown).
Histology
Liver, kidney, lung, testis, GI tract, and brain were sectioned and stained with hematoxylin and eosin. Using a grid and performing a double-blind analysis, we made a semiquantitative (1+ to 5+) assessment of lipid and glycogen content. Lipid-filled intracellular droplets were significantly decreased (P = .004) in both Cd-exposed WT and BTZIP8-3, compared with non-Cd-exposed control groups; glycogen content trended downward (P = .07) in Cd-exposed BTZIP8-3 compared with Cd-exposed WT and both non-Cd-exposed control groups (data not illustrated). No other differences in liver, or in any of the other 5 tissues among the 4 groups, were seen.
Tissue Element Analysis
We measured a total of 29 elements and 45 isotopes including Cd, in liver, kidney, lung, and testis. Twenty elements were below the level of detection and were not studied further; potassium, sodium, and aluminum were detected but showed no remarkable differences among the control and Cd-exposed WT and BTZIP8-3 mice (Table S1). Tissue content of the remaining 6 elements listed in Table S1 is illustrated in Figure 3. Cd levels (Figure 3, upper left) were 10- to >20-fold higher in liver and kidney than in lung or testis. Consistent with the fact that BTZIP8-3 has three more copies of the Slc39a8 gene than WT mice, BTZIP8-3 contained significantly more Cd than WT in kidney, liver and lung but not testis.
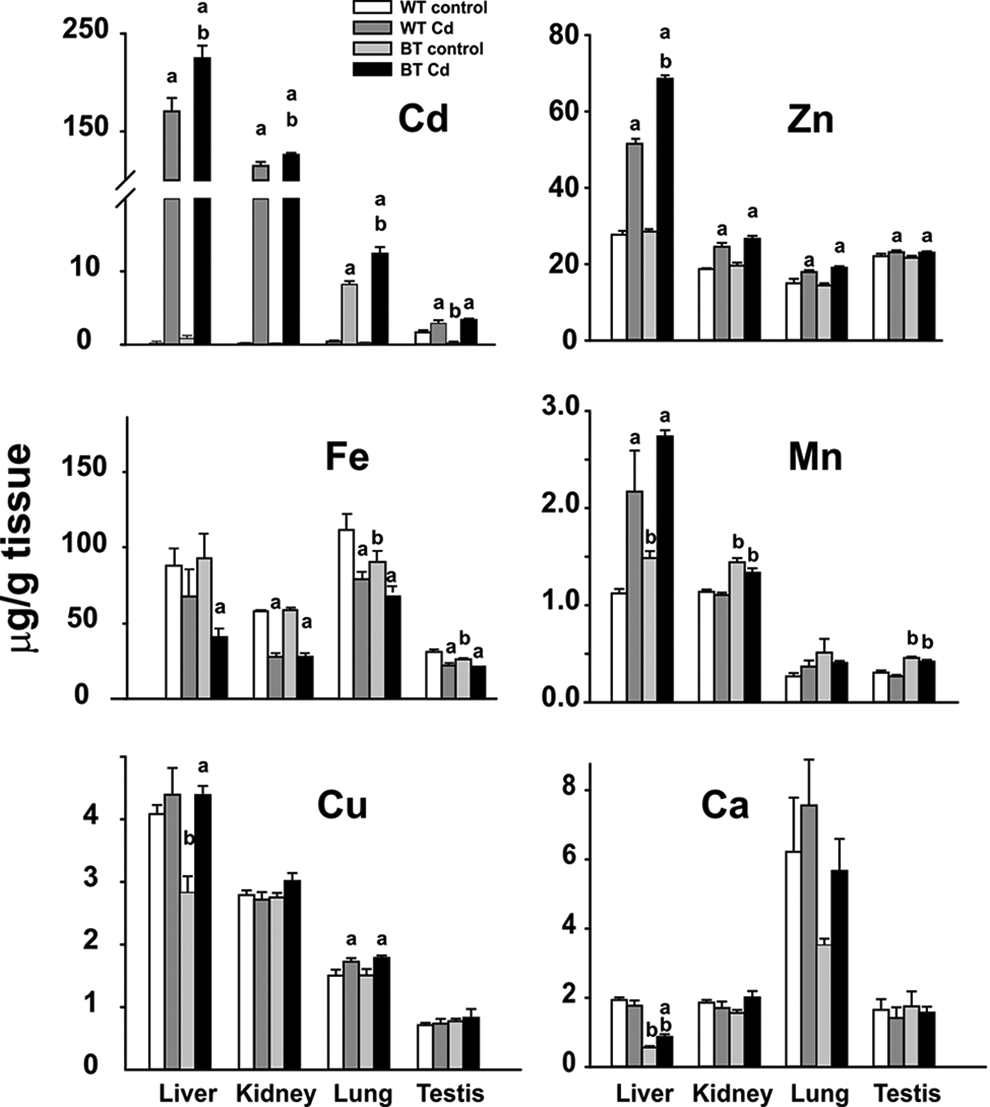
Tissue content (μg/g wet weight) of 6 metals, at end of the 20-week experiment of mice receiving oral Cd. Liver, kidney, lung, and testis metal contents were measured in control and Cd-exposed wild-type (WT) mice and control and Cd-treated BTZIP8-3 mice. Note that values on ordinate sometimes vary by 3 orders of magnitude. Bars represent means (N = 7 or 8) and brackets represent standard error of the mean (SEM). aSignificantly different (P < .05) from control with same genotype. bSignificantly different (P < .05) from other genotype's matched experimental group.
Zinc (Zn) levels (Figure 3, upper middle) were 2- to 3-times higher in liver than in kidney, lung or testis in Cd-exposed mice. Except in liver where BTZIP8-3 contained significantly more Zn than WT in mice treated with Cd, this effect was not seen in the other 3 tissues examined.
Interestingly, iron (Fe) levels (Figure 3, upper right) were lower in both Cd-exposed WT and BTZIP8-3 mice in all 4 tissues examined. On the other hand, no important differences in Fe concentrations were seen in any of the 4 tissues between Cd-exposed WT and BTZIP8-3, whereas subtle differences were found in control WT versus BTZIP8-3 lung and testis.
Manganese (Mn) levels (Figure 3, lower left) were 3- to 5-fold higher in liver and kidney than in lung or testis. BTZIP8-3 contained significantly more Mn than WT in control and Cd-exposed liver, kidney and testis but not lung. These data are consistent with our previous finding that ZIP8 has an extremely high affinity for Mn 1 and therefore might be a preferred transporter of Mn in specific tissues or cell types. 4,8,9,10
Copper (Cu) levels (Figure 3, lower middle) were 2- to ∼6-times greater in liver and kidney than in lung or testis. Less Cu was found in control BTZIP8-3 than in control WT liver. Compared with control mice, significantly more Cu was found in Cd-exposed BTZIP8-3 liver and lung, as well as in Cd-exposed BTZIP8-3 lung.
Calcium (Ca) content (Figure 3, lower right) was significantly decreased in both control and Cd-exposed BTZIP8-3 liver but not in control liver, or control or Cd-exposed kidney, lung or testis. Ca levels were 2- to 4-fold greater in lung than in liver, kidney or testis.
Blood Urea Nitrogen and Kidney Weights
At the conclusion of 20 weeks, BUN values were significantly elevated in Cd-exposed WT and BTIP8-3 mice, compared with controls (Figure 4A). No significant difference was found between the elevated BUN in Cd-exposed BTZIP8-3 mice and that in Cd-exposed WT mice. Kidney weights were also significantly greater in Cd-exposed WT and BTIP8-3 mice, compared with controls (Figure 4B). No significant differences were seen, however, between the Cd-exposed WT and the CD-exposed BTZIP8-3 mice.
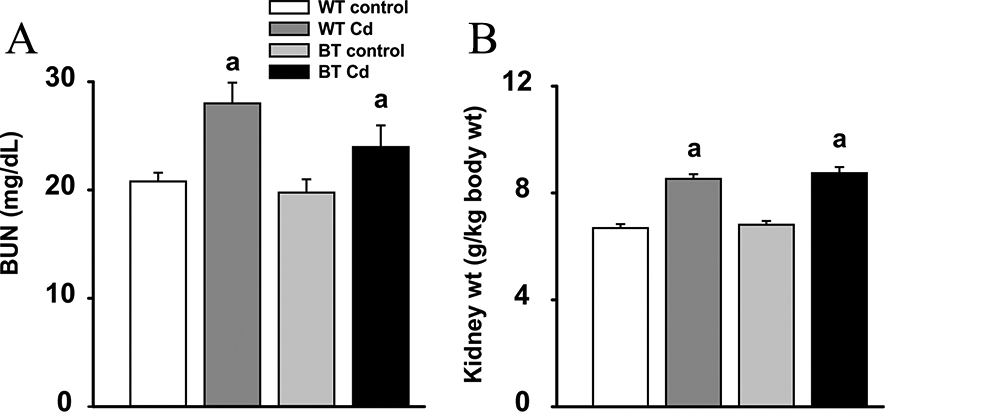
Blood urea nitrogen (BUN) and kidney weights at end of the 20-week experiment of mice exposed to oral Cd. A, BUN (mg/dL) was measured weekly; no significant differences were seen until the last several weeks of the experiment. aSignificantly different (P = .03) from control within same genotype. B, Kidney weights (g/kg body weight). aSignificantly different (P < .001) from control within same genotype.
Discussion
We began the present study by postulating that 3 additional copies of the Slc39a8 gene––and therefore ∼2.5-times more ZIP8 present in all tissues of the BTZIP8-3 mouse––would enhance oral Cd uptake and, hence, would increase Cd-induced toxicity in BTZIP8-3 considerably more than WT mice. We wished to mimic a long term, chronic Cd exposure situation typically observed for humans exposed to high levels of environmental Cd. We thus designed a 20-week experiment in mice and exposed them to escalating Cd levels in drinking water. CdCl2 in drinking water was thus increased incrementally from 100 mg/L to a maximum of 1600 mg/L; this resulted in Cd ingested (week 1) beginning at ∼50 mg/kg body weight and ending between 425 and 625 mg/kg (week 20).
To put this into perspective, the amount of Cd absorbed from smoking 1 pack of cigarettes per day is ∼3 µg/day 17 ; a 70-kg person smoking 2 packs a day for 50 years would therefore amount to taking up ∼1.6 mg of Cd per kg, which is sufficient in some patients to cause renal failure. 18 Throughout our 20-week experiment, similar amounts of ingested Cd were seen among BTZIP8-3 and WT males and females (Figure S1).
In an earlier study from this laboratory, 30 μmol/kg of Cd administered subcutaneously as a single dose (∼0.165 mg/mouse) caused severe renal toxicity in 12 h and death in less than 48 h in BTZP8-3 but not WT mice. 12 In contrast, the mice in the present study ingested ∼230 mg Cd over 140 days (1.64 mg/mouse/day), that is, about 10 times more oral Cd each day for 20 weeks than the 1 subcutaneous dose; interestingly, not only was the amount of renal damage in BTZIP8-3 not lethal, but also the degree of kidney toxicity was similar between BTZIP-3 and WT mice. Decreases in urinary pH, consistent with metabolic acidosis, were found by week 8 and were similar between Cd-exposed BTZIP8-3 and WT (Figure 1).
Significantly elevated plasma ALT and AST activities (Figure 2), and a trend in elevated urinary GGT activities (Figure S2), were also detected from week 8 onward and were similar between Cd-exposed BTZIP8-3 and WT, as compared with non-Cd-exposed control mice. Although abnormal increases in ALT and AST levels are more predictive of liver 19 and muscle 20 damage, respectively, general tissue toxicity in Cd-exposed mice may also contribute to increases in plasma ALT and AST levels. Abnormally elevated urinary GGT activity is a more specific indicator of renal insult. Elevated BUN levels and kidney weights at week 20 were also similar between Cd-exposed BTZIP8-3 and WT mice; these parameters further suggest Cd-induced early kidney damage, although histologically no significant alterations other than intracellular lipid globule content were seen (data not shown). Chronic Cd exposure can damage various organs and tissues––including kidney, liver, lung, pancreas, testis, placenta, and bone––with kidney and liver being the 2 primary target organs. 21
Curiously, both iron and copper content in liver, kidney, lung, and testis were similar between Cd-exposed BTZIP8-3 and WT (Figure 3). Fe2+ is an endogenous cation known to be transported by ZIP8 9,11 ; perhaps Fe transport by ZIP8 is more important in hematopoietic 10 and placental 9,10 cell types than in liver, kidney, lung or testis. In contrast, Cu2+ has been shown not to be effectively transported by ZIP8. 9,11
Elevated BUN values and increased kidney weights were similar in Cd-exposed WT and BTZIP8-3 mice (Figure 4). These data are further evidence that 2 copies of the Slc39a8 gene appear to work as well as 5 copies in ZIP8-mediated toxicity of oral Cd in this study.
Compared with Cd-exposed WT, Cd-exposed BTZIP8-3 mice exhibited statistically significant increases in Cd content in liver, kidney, lung, and testis; Zn content in liver; and Mn content in kidney and testis (Figure 3). Although these differences were not as dramatic as the 2.5-fold differences in ZIP8 mRNA and protein seen when comparing BTZIP8-3 and WT, 12 these results indicate that increased levels of ZIP8 in these BTZIP8-3 tissues do function to enhance transport of Cd, Zn, and Mn––3 well-established substrates for ZIP8. 4,8,9 On the other hand, Cu content did not follow the pattern of Cd, Zn, or Mn (Figure 3); and Cu is known not to be a strong substrate for ZIP8. 4,8,9
Unexpectedly, decreases in calcium content were seen in liver of both Cd-exposed BTZIP8-3 and WT mice compared with controls (Figure 3). Calcium channels have long been proposed as a major means of Cd uptake (reviewed in 22 ). However, ZIP8 is a poor transporter of Ca, and Ca is a weak inhibitor of Cd or Zn uptake. 11
Whatever changes seen in liver, kidney, lung, and testis were histologically unremarkable between Cd-exposed BTZIP8-3 and WT mice; the only difference of note was a decrease in lipid-filled intracellular droplets in both Cd-exposed WT and BTZIP8-3, compared with non-Cd-exposed control groups. These results underscore the well-known fact that numerous plasma and urinary enzyme biomarkers are far more sensitive than gross microscopic alterations in detecting early toxicity of chronic oral Cd exposure.
Are there other Cd transporters of importance, especially in the GI tract during oral exposure of this environmental heavy metal? SLC11A2 (also called NRAMP2) is a proton-coupled divalent metal transporter (DMT1) with a preference for Fe uptake and has been implicated in Cd uptake––especially from the GI tract––and subsequent toxicity in mammals. 23 In Xenopus oocytes the Km and Vmax values of ZIP8-mediated Cd uptake 11 were found to have >2-fold greater affinity that that for SLC11A2-mediated Cd uptake. 24
When this project was completed, we had not yet appreciated the tissue distribution of ZIP8 compared with that of the ZIP14 transporter. 4 The gene evolutionarily closest to Slc39a8 is Slc39a14 which codes for ZIP14; ZIP14 is also ubiquitous in many cell types but highest in GI tract and liver. 4,25 ZIP8 and ZIP14 display very similar cation substrate specificities 4,9,11,25,26 yet show distinctly different cell-type distribution; ZIP8 is predominant in kidney, testis, and lung, whereas ZIP14 is most highly expressed in GI tract and liver––suggesting nonredundant functions. 4,9,25 This project was principally a study of oral Cd uptake, however, and ZIP14 (and perhaps DMT1 as well) are likely to be more influential than ZIP8 in Cd absorption from the GI tract. 4,8,9,25
Future plans are to compare BTZIP8-3 versus WT mice at a single dose of oral Cd over 1 year of time. The more than 2 dozen divalent cation transporters, metallothionein, reduced glutathione levels, and other parameters of oxidative stress, plus assessment of apoptosis in kidney and liver––will also be carried out in this prospective study.
In conclusion, chronic oral Cd exposure at relatively high doses chosen for this 20-week study, in mice carrying 5 versus 2 Slc39a8 gene copies (or 2.5-times more ZIP8 in liver, kidney, lung, testis, GI tract, and brain), appears not to affect the early signs of Cd toxicity, as measured by several biochemical markers but not yet detectable histologically. However, Cd-exposed BTZIP8-3 mice carrying 2.5-fold more ZIP8 in all tissues does indeed exhibit more Cd content in liver, kidney, lung, and testis; Zn content in liver; and Mn content in kidney and testis.
Footnotes
Acknowledgment
We thank our colleagues, especially Lei He, for valuable discussions and careful reading of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded, in part, by NIH R01 ES010416 (DWN), R01 DK062809 (MS), and P30 ES006096 (DWN).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
