Abstract
Naphthenic acids (NAs) are primarily cycloaliphatic carboxylic acids with 10 to 16 carbons. To characterize the potential of refined NAs (>70% purity) to cause reproductive and/or developmental effects, Sprague-Dawley rats (12/group) were given oral doses of 100, 300, or 900 mg/kg/d, beginning 14 days prior to mating, then an additional 14 days for males or through lactation day 3 for females (up to 53 days) in a repeated dose/reproductive toxicity test (Organization for Economic Cooperation and Development [OECD] 422). Potential mutagenic effects were assessed using Salmonella (OECD 471) and in in vivo micronucleus tests (OECD 474) using bone marrow taken from treated animals in the screening study described previously. Systemic effects included reduced terminal body weights, increased liver weights, and changes in a number of blood cell parameters. The overall no effect level for all target organ effects was 100 mg/kg/d. In the reproductive/developmental toxicity assessment, there were significant reductions in numbers of live born offspring in groups exposed to 300 and 900 mg/kg/d. The overall no effect level for developmental effects was 100 mg/kg/d. The data from the Salmonella and micronucleus tests provide evidence that refined NAs are not genotoxic.
Keywords
Introduction
The United States Environmental Protection Agency (USEPA) announced a voluntary chemical data collection effort in 1998 called the High Production Volume (HPV) Challenge Program. 1 The HPV chemicals are those produced or imported into the United States in aggregate quantities of at least 1 million pounds per year. Approximately 400 petroleum substances were sponsored in the EPA’s Challenge Program by companies belonging to the Petroleum HPV Testing Group (PHPVTG). These approximately 400 substances were organized into 13 categories to facilitate data sharing and to avoid redundant testing. The categories include crude oils, gases, gasoline, kerosene/jet fuel, gas oils, heavy fuel oils, lubricating oils, waxes, aromatic extracts, asphalts, grease thickeners, petroleum coke, and wastes. This article reports an investigation into the toxicological hazards of naphthenic acids (NAs), a specific type of waste.
“Naphthenic acids” (Chemical Abstracts Service [CAS] number 1338-24-5), as understood by the petroleum industry, is a collective term which describes all of the carboxylic acids present in crude oil. The NAs are described by a general formula RCOOH in which R represents the naphthene moieties that consist of cyclopentane and cyclohexane derivatives. The alkyl-substituted cycloaliphatic carboxylic acids are the most common constituents. The carboxyl group is usually attached to a side chain rather than directly to the ring. Aromatic, olefinic, hydroxyl, and dibasic acids may be present as minor components. 2 Other related substances are NAs (petroleum), crude NAs (CAS number 64754-89-8), and sodium salts (CAS number 61790-13-4).
The NAs are naturally occurring constituents of crude oil, particularly naphthenic and asphaltic crudes, and are removed from petroleum distillate streams by caustic extraction to reduce corrosion in the refinery and also to improve certain technical properties including burning qualities, storage stability, and odor of finished fuels, particularly kerosenes, jet fuels, and diesel fuels. The process of treating petroleum distillates by caustic extraction results in the formation of sodium salts of NAs (CAS number 61790-13-4). The NAs that can be recovered by acidulating the caustic solutions are referred to as crude NAs (CAS number 064754-89-8). The crude NAs can be further refined to produce the substances referred to by CAS number 1338-24-5 which are used for commercial applications. The substances described by the other CAS numbers are intermediates generated during the production of commercial NAs.
A review of the toxicological information on NAs, much of which was previously unpublished, indicated a low potential for acute toxicity; oral and dermal median lethal dose (LD50) values were ≥3000 mg/kg/d, and the inhalation LC50 was greater than the maximally attainable vapor concentration. However, NAs were irritating to both the skin and the eyes. 3 A sample of NAs extracted from oil sand tailings was tested in a 90-day gavage study at doses ranging from 0.6 to 60 mg/kg/d. 4,5 Effects on the high-dose group included reduced body weights, increased weights of brain, liver, and kidney, and changes in marker enzymes. The authors also reported seizure activity in the animals after 40 days of treatment. In a reproductive toxicity test reported only in abstract form, 5,6 the same test material was reported to have reduced fertility in rats but did not produce malformations. Studies of the mutagenic properties of NA salts indicated that these were not active in Salmonella or in vitro cytogenetics tests. 7,8
Based on a review of the toxicology data, the PHPVTG concluded that the potential acute toxicological effects of NAs had been sufficiently characterized but that further data were needed to assess the potential for repeated dose toxicity, reproductive/developmental toxicity, and genotoxic effects. Accordingly, a study was undertaken to evaluate refined NAs in a repeated dose/reproductive toxicity screening test and to embed a bone marrow micronucleus test in the study design. A bacterial mutagenicity test was also conducted. The results of these studies, along with other available data, were considered to provide sufficient evidence to characterize the toxicological hazards of NAs and also to satisfy the HPV obligations of the petroleum industry for this group of substances.
Materials and Methods
Materials
The test material was a sample of refined NAs (CAS number 1338-24-5), supplied by Merichem Chemicals and Refinery Services LLC and analyzed by published methods. 9 –11 The NAs are described by the general formula CnH2n+ZO2 in which n is the carbon number and Z is zero or a negative even number defining the hydrogen deficiency due to cyclization. The analysis showed that approximately 90% of the constituents had carbon numbers in the range of C10 to C16. 12 The ring distribution analysis indicated that the sample contained 39% 1-ring compounds, 31% 2-ring compounds, 5% 3-ring compounds, and 1% 4-ring compounds with the remaining 24% being comprised of constituents with no naphthenic rings. The dosing solutions were prepared by suspending the NAs in corn oil (Spectrum Chemical Manufacturing Corporation, New Brunswick, New Jersey) and diluting as appropriate. The positive control for the micronucleus test was cyclophosphamide (CAS number 6055-19-2), obtained from Sigma Aldrich, Inc (St Louis, Missouri).
Methods
In Vitro Studies
The bacterial mutagenesis test was consistent with published methods and followed Organization for Economic Cooperation and Development (OECD) 471 guidelines. 13 –15 Salmonella tester strains, TA98, TA100, TA1535, TA1537 and the Escherichia coli strain wp2uvrA were obtained from Molecular Toxicology (Boone, North Carolina). The test sample of refined NAs was dissolved in dimethyl sulfoxide to a final concentration of 100 mg/mL. This stock solution was then used to prepare a series of dosing solutions that were added to Petri plates in dosing volumes of 50 µL/plate in test concentrations ranging from 25 to 5000 µg/plate. Duplicate tests were conducted using triplicate plating. Tests were conducted with and without metabolic activation (Aroclor 1254-induced rat liver). Results are given as the mean (± standard deviation [SD]) revertant count/plate. The criterion for a positive response was either a 2-(TA100) or 3-fold (all other strains) increase in revertant counts by comparison to controls which was repeatable and/or dose responsive. Assay validity was assessed through the use of positive control substances including 2-nitrofluorene, sodium azide, 4-nitro-quinoline-n-oxide, and 2-aminoanthracene to test the responsiveness of each of the tester strains and the adequacy of the metabolic activation mix.
In Vivo Studies
A repeated dose/reproductive toxicity screening study was conducted in accordance with OECD 422 (Combined Repeated Dose Toxicity Study with Reproduction/Developmental Toxicity Screening). The study was also consistent with the USEPA Health Effects guideline, Combined Repeated Dose Toxicity Study with Reproduction/Developmental Toxicity Screening (OPPTS 870.3650). The micronucleus test design followed the USEPA guidelines for studies of this type (OPPTS 870.5395). The testing was in accordance with Good Laboratory Practice Guidelines of the OECD 16 and the USEPA. 17
Animal Husbandry
Sexually mature male and virgin female Sprague-Dawley rats (Crl:CD (SD); 66 males and 67 females) were obtained from Charles River Laboratories, Raleigh, North Carolina. Each rat was weighed, examined for good health, and assigned a unique identification number. The rats were housed individually in wire mesh cages and given ad libitum access to food (Certified Rodent LabDiet 5002; PMI Nutrition International, LLC St. Louis, Missouri) and water. The room temperature and humidity controls were set to maintain environmental conditions of 22°C ± 3°C and 50% ± 20% relative humidity. Animals were maintained in accordance with the recommendations of the National Research Council. 18
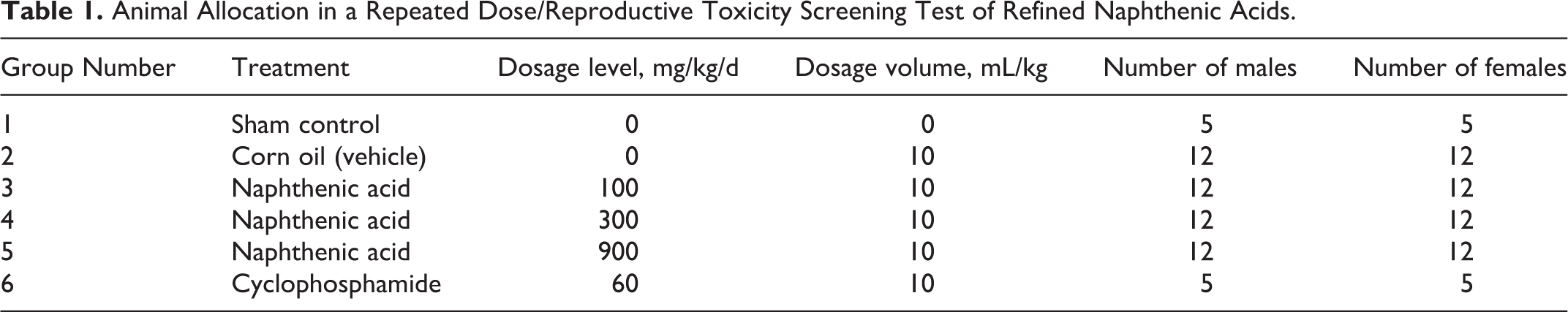
The rats were held for a 16-day acclimation period and assigned to dose groups by a computer program that assured homogeneity of group means and variances for body weight. The assignments are as shown in Table 1.
Animal Allocation in a Repeated Dose/Reproductive Toxicity Screening Test of Refined Naphthenic Acids.
Substance Administration
The doses were administered once daily by oral gavage using a Teflon-shafted, stainless steel, ball-tipped dosing cannula (Natume, Japan). All doses were given in 10 mL/kg volumes and were calculated based on the most recently recorded body weights. Dosing of males was initiated 14 days prior to pairing and throughout a 14-day mating period for a total of 28 to 29 doses. Dosing of females was also initiated 14 days prior to pairing and continued throughout the mating and gestational periods until postnatal day 3 (the day prior to scheduled termination). The total number of doses ranged from 39 to 53 depending on the time at which mating occurred. Rats used as the positive controls for the assessment of micronucleus induction were given cyclophosphamide (60 mg/kg) in single oral doses, approximately 24 hours prior to euthanasia for bone marrow collection.
Toxicological Evaluations
Clinical observations and survival
All rats were examined twice daily for mortality and general health. Detailed physical examinations of all animals were conducted weekly. Additionally, all animals were examined at the time of treatment, approximately 1 hour after test material administration, and all unusual observations were recorded. Pregnant females were also examined twice daily during the period of expected parturition and at parturition for dystocia or other difficulties.
Body weights and food consumption
Body weights of male rats were recorded 1 week prior to test substance administration, on the first day of dose administration, on a weekly basis during the study, and at termination. Body weights of female rats were recorded 1 week prior to test substance administration, on the first day of dose administration, and weekly until evidence of copulation was obtained. From that point, the body weights of female rats were recorded on gestation days 0, 4, 7, 11, 14, 17, and 20 and on lactation days 0, 1, and 4 (termination). For females for which there was no evidence of copulation, body weights were recorded weekly until termination. Body weights of offspring were recorded on postnatal day 1 and then prior to termination on postnatal day 4. Food consumption by adult animals was also recorded on the same schedule as the body weights.
Neurological evaluations
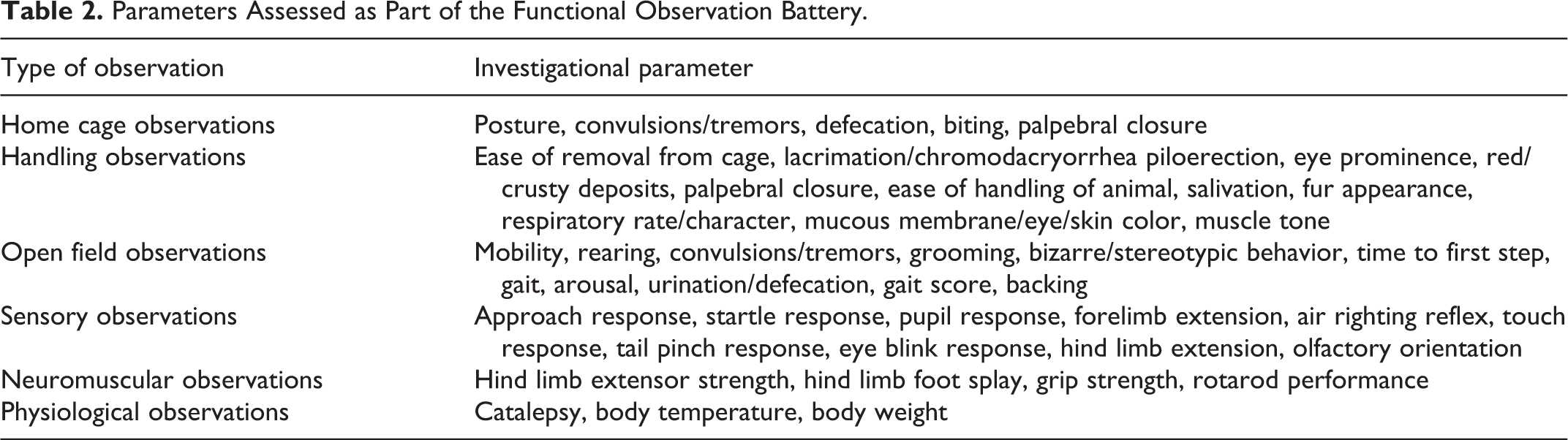
The potential for nervous system effects was assessed using a functional observation battery (FOB). Male rats in the vehicle (corn oil) and NA-treated groups were examined prior to initiation of dosing and after approximately 28 days of dosing, and female rats were examined prior to initiation of dosing and at termination. The FOB procedures were based on previously developed protocols. 19 –24 The testing was conducted in a sound attenuated room with a white noise generator set to operate at 70 ± 10 dB. The investigators who conducted the FOB were not aware of the treatment groups from which the respective animals were taken. The FOB consisted of the following: home cage observations, handling observations, open field observations, sensory observations, and neuromuscular observations (Table 2). In addition, there were physiological observations including body weight, body temperature, and examination for catalepsy. There was also an assessment of locomotor activity which was measured electronically using a computer-controlled system with a series of infrared photobeams in a clear plastic rectangular cage (Kinder Scientific Motor Monitor, Kinder Scientific, LLC, Poway, California). Animals were tested separately in 60-minute sessions divided into 5-minute intervals, tabulated to 10-minute intervals.
Parameters Assessed as Part of the Functional Observation Battery.
Clinical pathology
On the day of scheduled termination, blood samples were taken from all rats in the corn oil (vehicle) and NA-treated groups for assessment of hematological and serum chemistry parameters. The hematological investigation included measurements of total leukocyte count, erythrocyte count, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, platelet count, prothrombin time, activated partial prothrombin time, reticulocyte count, mean platelet volume, red cell distribution width, hemoglobin distribution width, differential leukocyte count, and red cell morphology. The serum chemistry investigation included measurements of concentrations of albumin, total protein, globulin, albumin/globulin ratio, total bilirubin, urea nitrogen, creatinine, alkaline phosphatase, alanine aminotransferase, aspartate aminotransferase, γ-glutamyltransferase, glucose, total cholesterol, calcium, chloride, phosphorus, potassium, sodium, triglycerides, and bile acids.
Gross and histological examinations
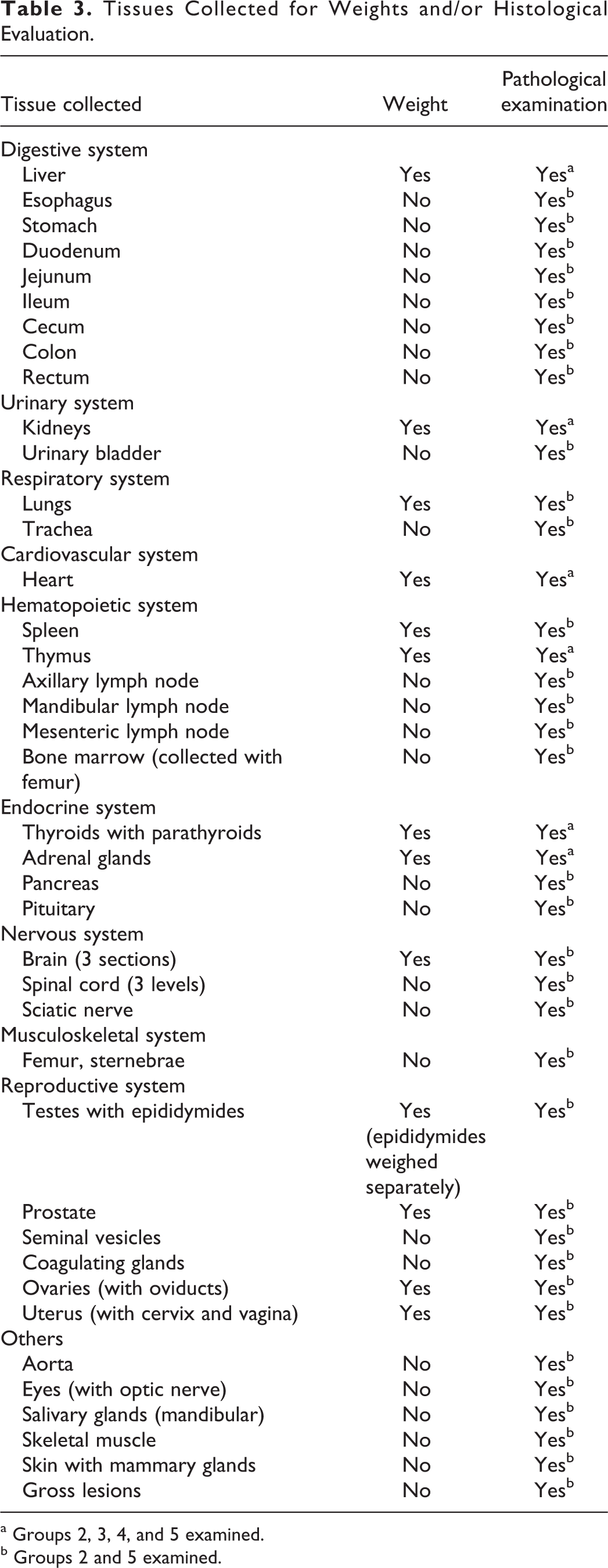
At termination, rats were euthanized by carbon dioxide inhalation. Necropsies were conducted on all animals sacrificed in extremis or at study termination. Organs were removed, weighed if this was planned, and placed in 10% neutral-buffered formalin for histologic examination. The disposition of organs and tissues was as listed in Table 3.
Tissues Collected for Weights and/or Histological Evaluation.
a Groups 2, 3, 4, and 5 examined.
b Groups 2 and 5 examined.
Micronucleus Test
Slide preparation
Bone marrow was collected from all animals at terminal sacrifice and flushed into a centrifuge tube using a syringe containing heat-inactivated fetal bovine serum (HIFBS). The bone marrow was centrifuged, the majority of the HIFBS was decanted, and the pellet was resuspended. Bone marrow smears were prepared by placing single drops of suspension on microscope slides (minimum of 2 per preparation). The slides were coded, air dried, fixed in methanol, and allowed to air dry a second time.
Slide evaluation
Coded slides were stained with either giemsa or acridine orange. 25 A total of 1000 erythrocytes/slide were evaluated (both polychromatic erythrocytes [PCEs] and normochromatic erythrocytes [NCEs] were counted), and the PCE/total erythrocytes (TEs) ratio was calculated. The number of micronucleated PCEs from a total of 2000 PCEs and the number of micronucleated NCEs from a total of 200 NCEs was then determined for each animal.
Reproductive Toxicity Evaluation
Breeding procedures
Mating was initiated after 14 days of dosing. Rats were mated on a 1:1 basis within each treatment group; females were cohoused with the males. Each mating pair was evaluated on a daily basis during the mating period. Successful mating was confirmed by the presence of a vaginal copulatory plug or the presence of sperm in a vaginal lavage. The day on which mating was confirmed was designated as gestational day (GD) 0.
Parturition
All females confirmed to have mated were placed in plastic maternity cages once mating was confirmed. Females for which copulation was not detected were placed in maternity cages at the end of the 14-day mating period. All females were allowed to deliver and to rear their young to postnatal day 4. On the day of parturition, all pups were examined for viability, for the presence of gross malformations, and to assess gender. The numbers of live and stillborn pups were recorded. Length of gestation was calculated as the time from confirmation of mating to the onset of delivery. Females for which there was no evidence of mating were sacrificed on postcohabitation day 25, those that showed evidence of mating but failed to deliver were euthanized on postmating day 25, and all others were euthanized on postnatal day 4. Uteri with no apparent microscopic evidence of implantation were opened and subsequently placed in 10% ammonium sulfide solution for detection of early implantation loss. 26
Evaluation of litters
All offspring were uniquely identified and examined daily for signs of mortality and ill health. All offspring were individually weighed on postnatal days 1 and 4. Gender was assessed on postnatal days 0 and 4. At scheduled termination (post-natal day 4), all surviving offspring were euthanized and discarded without further examination.
Statistical Analysis
Evaluation of toxicological and reproductive data
Parental mating, fertility, conception, and copulation indices were analyzed using the chi-square test with Yates correction. 27 Mean parental body weights (weekly, gestation, and lactation), body weight changes and food consumption, offspring body weights and body weight changes, gestation length, numbers of former implantation sites, numbers of corpora lutea, number of pups born, live litter size on post-natal day PND 0, unaccounted for sites, absolute and relative organ weights, clinical pathology values (except for γ-glutamyltransferase), precoital intervals, and continuous FOB values were evaluated by 1-way analysis of variance (ANOVA) 28 to determine intergroup differences between the vehicle control and test substance-treated groups. If the ANOVA revealed significant (P < 0.05) intergroup variance, Dunnett test 29 was used to compare the test substance-treated groups to the control group. Histopathological findings in the test substance-treated groups and FOB parameters yielding scalar or descriptive data were compared to the vehicle control group using Fisher exact test. 30 γ-Glutamyltransferase data, mean litter proportions (percent of litter) of males at birth, and postnatal survival were evaluated using the Kruskal-Wallis nonparametric ANOVA 31 to determine intergroup differences between the vehicle control and the test substance-treated groups. If the ANOVA revealed significant (P < 0.05) intergroup variance, Dunn test 32 was used to compare the test substance-treated groups to the vehicle control group.
Evaluation of motor activity data
Ambulatory and total counts measured during locomotor activity assessments were analyzed by sex and session by repeated measures ANOVA (RMANOVA). Factors in the model included treatment time (TRT), time interval (TIME), and the interaction of TIME and treatment group (TRT×TIME). The SAS procedure PROC MIXED was used for analysis with the random effect of animal included as the repeated measurement. The covariance structure across time was selected by comparing Akaike’s Information Criterion.
The monotonic dose–response relationship was evaluated using sequential linear trend tests based on ordinal spacing of dosage levels. The linear dose by time (LinTrt*Time) interaction was evaluated and, if significant at the 0.05 level, trend tests on treatment means were performed at the 0.05 significance level for each TIME. If the LinTrt*Time interaction was not significant, the trend test was conducted across the pooled TIMEs by session only.
Nonmonotonic dose responses were evaluated whenever no significant linear trends were detected, but TRT and/or TRT*TIME interaction was significant at the 0.01 level. Within the framework of the RMANOVA, pairwise comparisons were made for each individual treated group with the control group through linear contrasts. If TRT*TIME was significant, the comparisons were conducted for each TIME. If only the TRT effect was significant, the comparisons were conducted across the pooled TIMEs of the entire session.
Evaluation of micronucleus test data
The percentages of PCEs, micronucleated cells in NCEs, and the ratios of PCEs to TEs in the test substance- and vehicle-treated groups were compared using ANOVA. 28 If the ANOVA revealed significant (P < 0.05) intergroup variance, Dunnett test 29 was used to compare each test substance-treated group to the vehicle control group. In addition, the positive control and vehicle control groups were compared using a separate parametric 1-way ANOVA. 28
Results
Bacterial Mutagenesis Test
The test sample of refined NAs was not mutagenic in bacteria either in the presence or in the absence of metabolic activation. The data shown (Table 4) are the results of the initial mutational assays. Data from conformational studies provided equivalent results. Toxicity as evidenced by reductions in spontaneous mutation frequency was observed at levels of 350 to 500 µg/plate. The positive controls responded as expected confirming both the sensitivity and the selectivity of the test system.
Results of the Initial Set of Mutational Tests in Salmonella and Escherichia coli Utilizing Plate Incorporation Assays, With and Without Metabolic Activation.
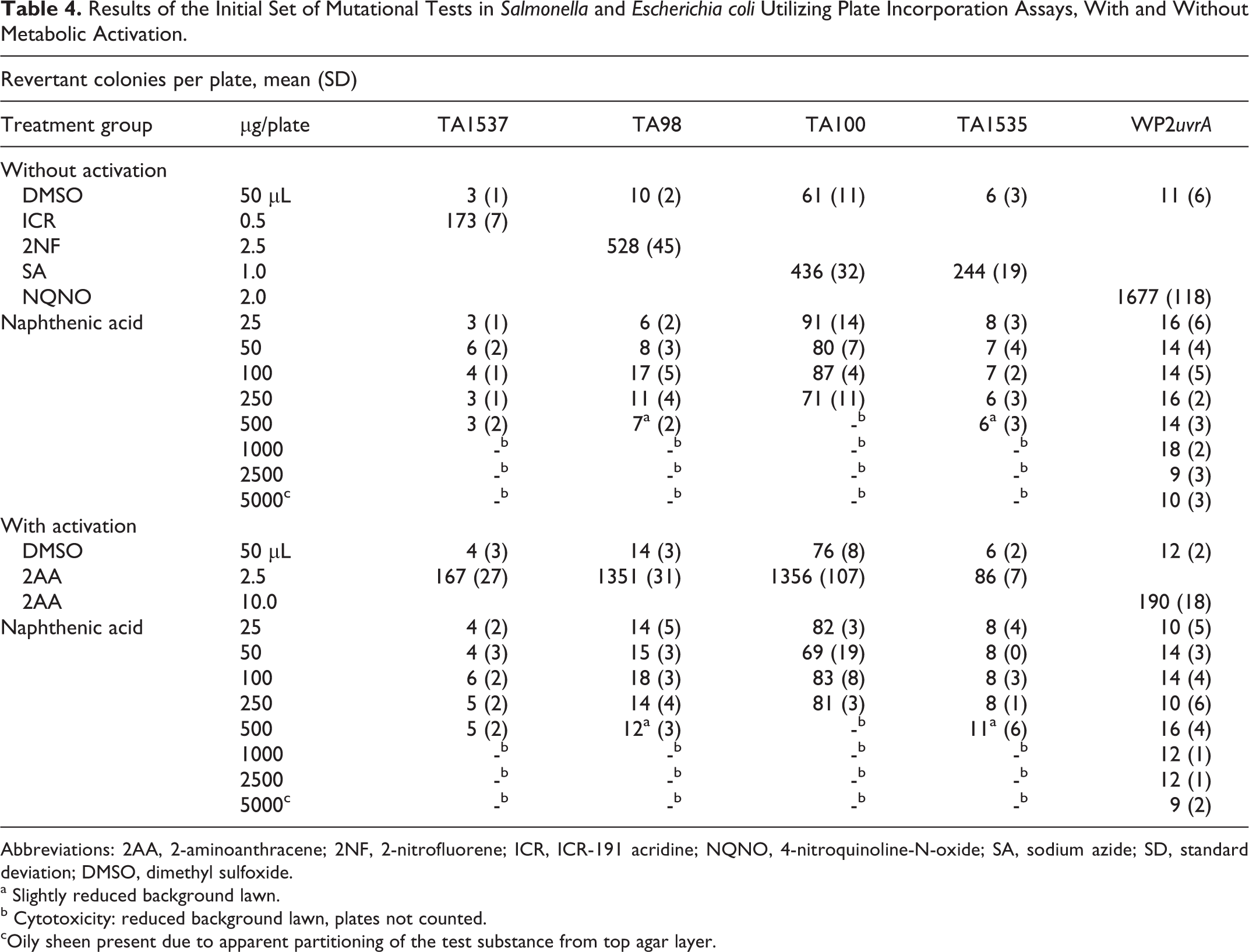
Abbreviations: 2AA, 2-aminoanthracene; 2NF, 2-nitrofluorene; ICR, ICR-191 acridine; NQNO, 4-nitroquinoline-N-oxide; SA, sodium azide; SD, standard deviation; DMSO, dimethyl sulfoxide.
a Slightly reduced background lawn.
b Cytotoxicity: reduced background lawn, plates not counted.
cOily sheen present due to apparent partitioning of the test substance from top agar layer.
In Vivo Tests—Survival and Clinical Observations
Two high-dose females were terminated on lactation day 2; 1 was sacrificed in extremis due to poor clinical conditions resulting from acute inflammation of the uterus; the other was sacrificed due to total litter loss. All other rats survived to scheduled termination. Clinical observations, which were noted only in high-dose group females and approximately an hour of dosing included hunched posture; rocking, lurching, and/or swaying while ambulating; walking on tiptoes; hypoactivity; and shallow respiration. Some of the high-dose group males also exhibited hunched posture.
Body Weight, Weight Gain, and Food Consumption
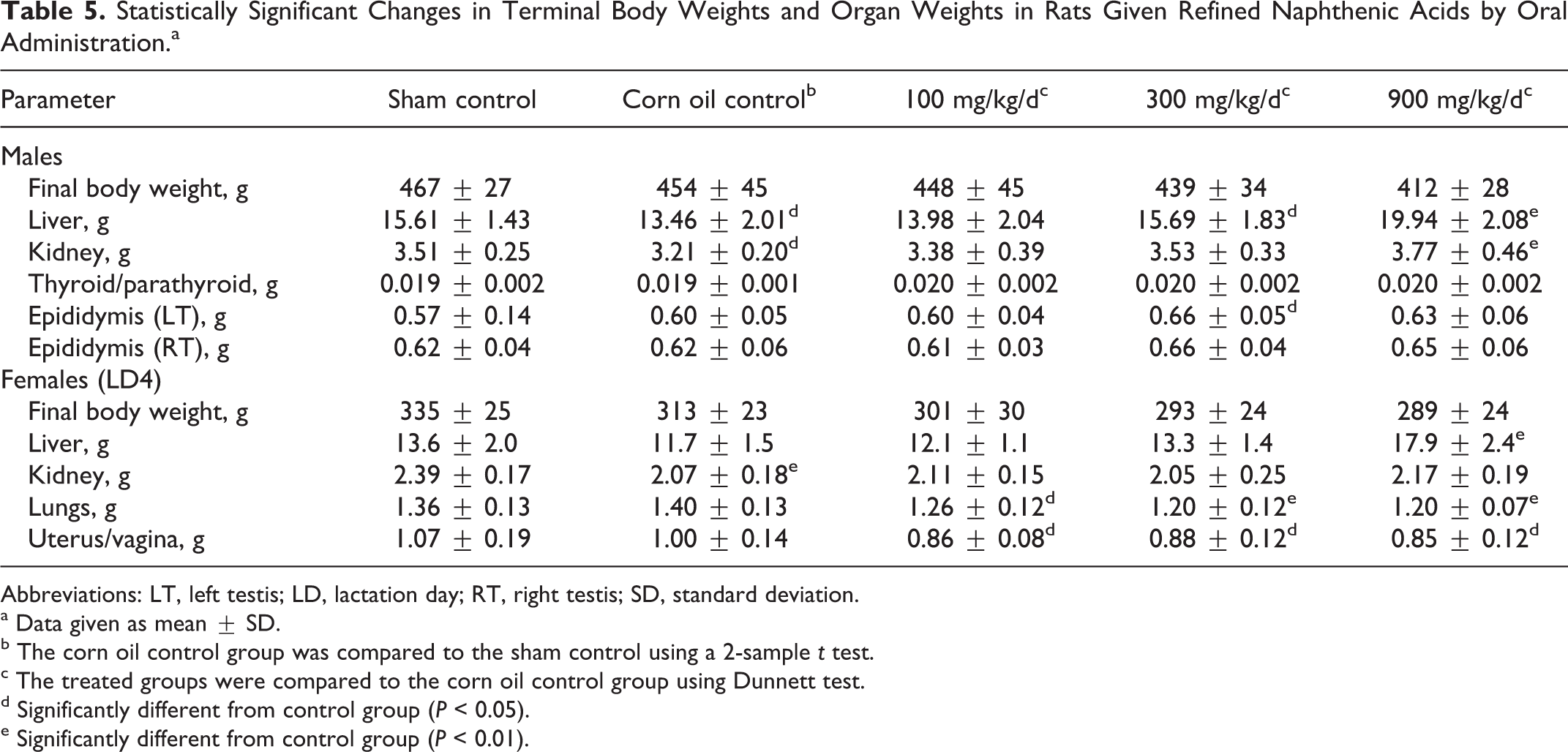
Body weights in high-dose group males were lower than control values, but the overall difference was less than 10%, and the differences were not statistically significant (terminal body weight data are shown in Table 5). Among the females, the body weights in the high-dose group were lower than control values but not significantly different at the end of the mating period. During gestation, the body weights were similar through day 14, but differences were greater in late gestation, and by GD 20 the body weights in the high-dose group were 8% below control values; however, these differences were not statistically significant (Table 6). Differences in body weight gain were associated with significantly reduced food consumption in the high-dose group animals during the mating period.
Statistically Significant Changes in Terminal Body Weights and Organ Weights in Rats Given Refined Naphthenic Acids by Oral Administration.a
Abbreviations: LT, left testis; LD, lactation day; RT, right testis; SD, standard deviation.
a Data given as mean ± SD.
b The corn oil control group was compared to the sham control using a 2-sample t test.
c The treated groups were compared to the corn oil control group using Dunnett test.
d Significantly different from control group (P < 0.05).
e Significantly different from control group (P < 0.01).
Summary of Body Weight Gain in Female Rats in the Premating, Mating, Gestational, and Lactational Phases.

Neurological Assessment
There were no statistically significant differences in parameters assessed as part of the FOB including home cage observations, handling parameters, open field observations, sensory observations, or neuromuscular observations. There were some small differences in body weight gain as indicated previously, but none of the other physiological parameters (catalepsy and body temperature) were affected by treatment. There were also no differences in locomotor activity patterns (data not shown).
Clinical Pathology
There were some differences in hematological values, primarily reductions in parameters related to hemoglobin content which were considered to have been treatment related. However, the differences were small and there was no consistency between males and females. (Note that the data summary [Table 7] reports only the parameters in which statistically significant differences were recorded).
Results of Assessments of Hematology Parameters Which were Statistically Different From Control Values.a,b

Abbreviations: APTT, activated partial prothrombin time; HDW, hemoglobin distribution width; RDW, red blood cell distribution width; SD, standard deviation.
a Parameters not affected by treatment included:
Males—white blood cell count, mean corpuscular volume (fL), mean corpuscular hemoglobin (pg), mean corpuscular hemoglobin content (g/dL), prothrombin time (seconds), APTT (seconds), reticulocytes (%), reticulocytes, absolute (103/µL), MPV (fL), neutrophils (%), lymphocytes (%), monocytes (%), eosinophils (%), basophils (%), leukocytes(%), neutrophils, absolute (103/µL), lymphocytes, absolute (103/µL), monocytes, absolute (103/µL), eosinophils, absolute (103/µL), basophils, absolute (103/µL).
Females—red blood cell count (106/µL), hemoglobin content (g/dL), hematocrit (%), mean corpuscular volume (fL), mean corpuscular hemoglobin (pg), mean corpuscular hemoglobin content (g/dL), platelet count (103/µL), prothrombin time (seconds), reticulocytes (%), reticulocytes, absolute (103/µL), MPV (fL), neutrophils (%), lymphocytes (%), monocytes (%), eosinophils (%), basophils (%), leucocytes (%), neutrophils, absolute (103/µL), eosinophils, absolute (103/µL), basophils, absolute (103/µL), leukocytes absolute (103/µL), RDW (%), HDW (%).
b Results given as mean ± SD. N = 12 (males), N = 9-12 (females).
c The treated groups were compared to the corn oil group using Dunnett test.
d Significantly different from corn oil control (P < 0.05).
e Significantly different from corn oil control (P < 0.01).
The clinical chemistry values showed a similar pattern. Among males, the only statistically significant differences between control were for creatinine (control value = 0.3 ± 0.1 mg/dL vs a value of 0.4 ± 0.0 mg/dL in the high-dose group, P < .01) and chloride (control value = 104 ± 1.1 mEq/L vs a value of 102 ± 1.3 mEq/L in the high-dose group, P < 0.01). Among the female rats, statistically significant differences were found for albumin (control = 4.3 ± 0.2 g/dL vs 4.7 ± 0.3 g/dL in the high-dose group, P < 0.05), total protein (control = 6.3 ± 0.3 g/dL vs 6.7 ± 0.4 g/dL in the high-dose group, P < 0.05), glucose (control = 115 ± 11 mg/dL vs 130 ± 8.0 mg/dL in the high-dose group, P < 0.05), cholesterol (control = 69 ± 14 mg/dL in the control vs 89 ± 19 mg/dL in the high-dose group, P < 0.05), calcium (control = 10.6 ± 0.4 mg/dL in the control vs 11.5 ± 0.6 mg/dL in the high-dose group, P < 0.01), and phosphorus (control = 3.9 ± 0.6 mg/dL vs 5.5 ± 1.2 mg/dL in the high-dose group, P < 0.05). All of the differences were small and most were within the historical range of the laboratory. Additionally, when differences were found they were mostly significant at only the 0.05 level, and there was no consistency of response between the sexes. In the absence of any corresponding pathological findings, these differences were most likely incidental.
Results of the Gross and Pathologic Examinations
The only notable gross observations were those of pale kidneys in the high-dose males and a reduction in the number of corpora lutea in the high-dose group females. The effects in females are discussed in more detail subsequently, in connection with the reproductive toxicity findings. Otherwise, the results of the gross examination were not remarkable. Organ weight determinations in males revealed increases in weights of liver, kidney, thyroid/parathyroid, and epididymis although the differences in thyroid/parathyroid and epididymal weights were only statistically different when compared on a relative to body weight basis. In females, there was a significant increase in liver weights and significant reductions in lung weights and absolute uterine weights (Table 5). The lung weights were within the historical range for the laboratory and were not associated with any pathological changes. The uterine weights were not significantly different when compared relative to body weights. All gravid females were in lactational anestrus and undergoing involution. All uterine weight values fell within the historical range for the laboratory and were not associated with any gross, histopathological, or clinical pathology changes.
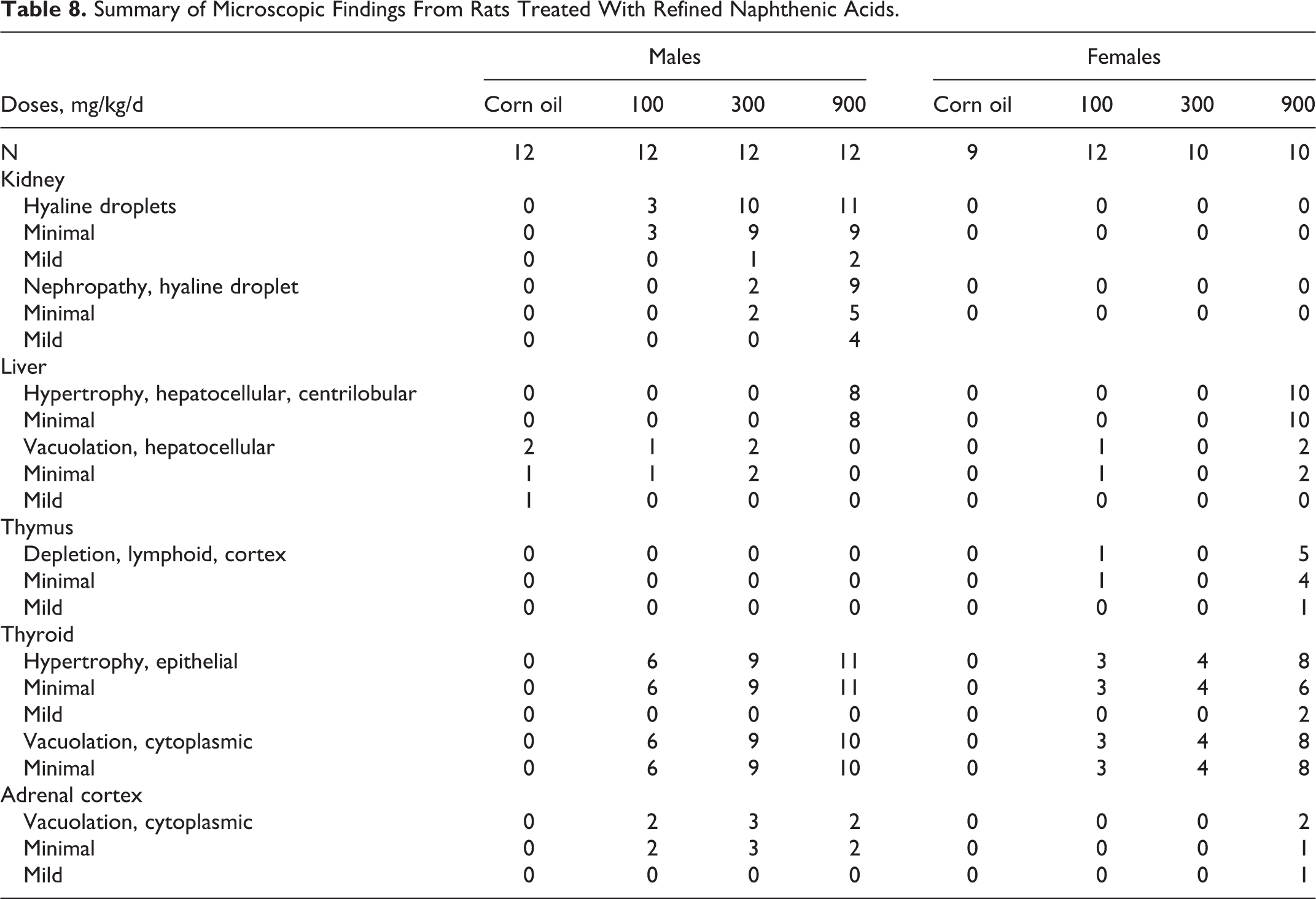
The results of the pathological investigation are summarized in Table 8. Kidney changes, reported in male rats only, were consistent with hyaline-droplet nephropathy (α2u-globulin-mediated nephropathy). The liver changes, found in organs from both male and female rats from the high-dose group, were described as hepatocellular hypertrophy. Other changes included cortical lymphoid depletion of the thymus in females, primarily in rats from the high-dose group. Epithelial hypertrophy and cytoplasmic vacuolation of the thyroid gland were noted in all treatment groups, and cytoplasmic vacuolation of the zona fasciculate in the adrenal cortex was reported in males from all treatment groups and in high-dose group females.
Summary of Microscopic Findings From Rats Treated With Refined Naphthenic Acids.
Assessment of Reproductive and Developmental Toxicity
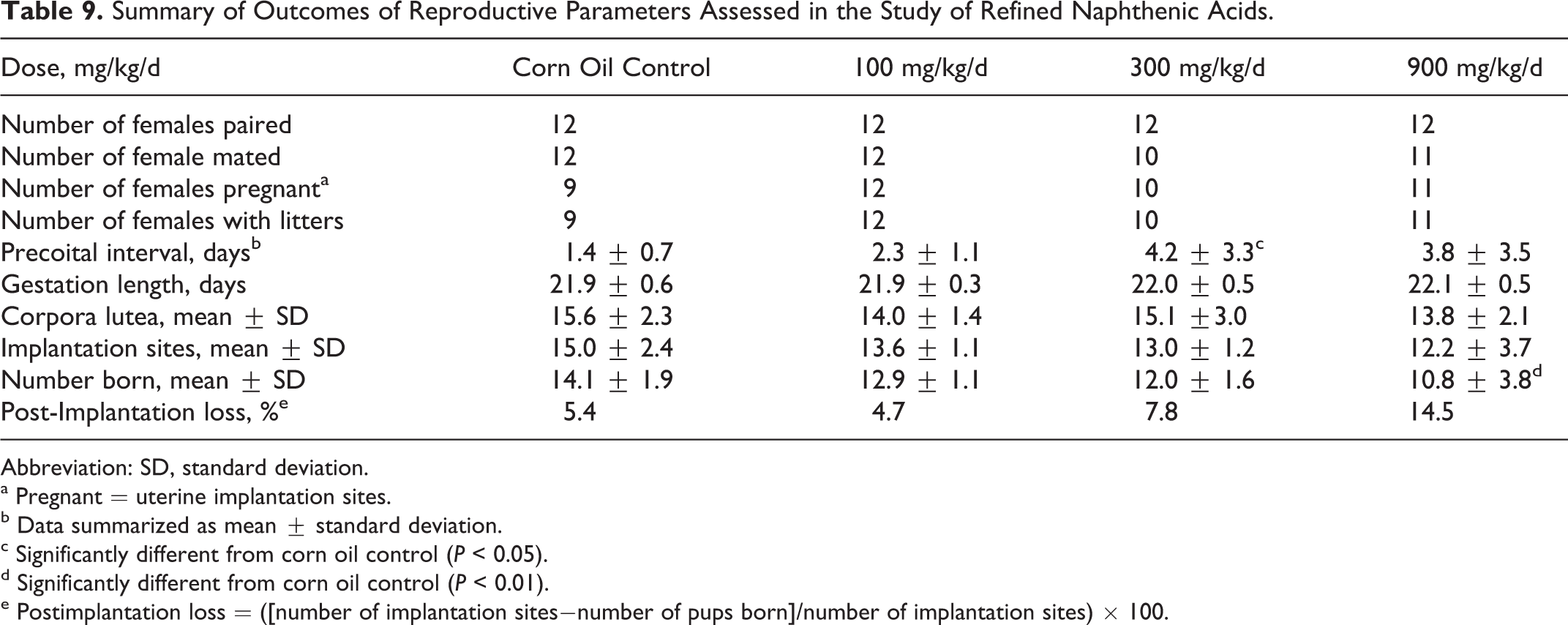
Reproductive performance
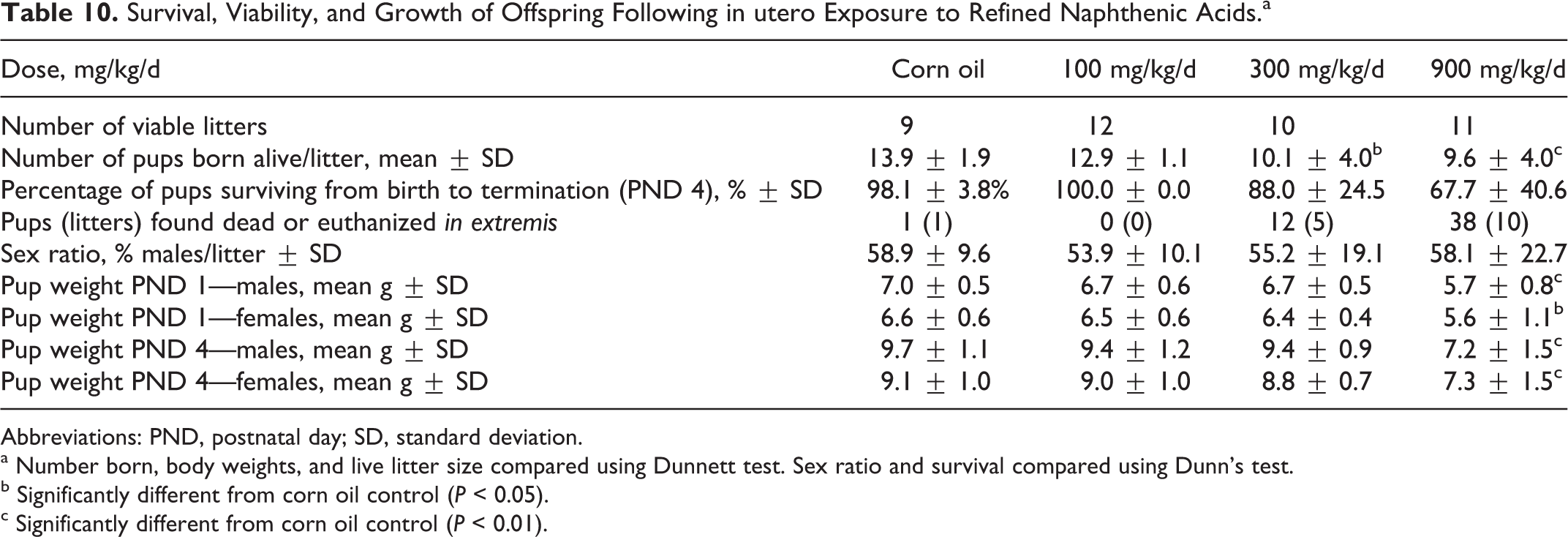
As shown in Table 9, there were no apparent treatment-related effects on mating. A single female in the 300 mg/kg/d group had a precoital interval of 13 days, resulting in a statistically significant increase in precoital incidence in this group. Otherwise, all of the pairs productively mated and pre-coital intervals were generally within the historical control range for the laboratory. Further, the length of the gestational period was similar across the groups. There were reductions in the numbers of corpora lutea and implantation sites in the high-dose group, but the differences were not statistically significant (Table 9). However, there was a significant reduction in the number of offspring born/litter in the high-dose group (Table 10). There was also a significant reduction in survival in offspring in the high-dose group, and those offspring that did survive had significantly lower body weights than those in the control groups. The numbers of pups found dead or euthanized in extremis during the period postnatal day 0 to 4 were control = 1 (1), 100 mg/kg/d = 0 (0), 300 mg/kg/d = 12 (6), and 900 mg/kg/d = 38 (10). The numbers of litters involved are given in parentheses.
Summary of Outcomes of Reproductive Parameters Assessed in the Study of Refined Naphthenic Acids.
Abbreviation: SD, standard deviation.
a Pregnant = uterine implantation sites.
b Data summarized as mean ± standard deviation.
c Significantly different from corn oil control (P < 0.05).
d Significantly different from corn oil control (P < 0.01).
e Postimplantation loss = ([number of implantation sites−number of pups born]/number of implantation sites) × 100.
Survival, Viability, and Growth of Offspring Following in utero Exposure to Refined Naphthenic Acids.a
Abbreviations: PND, postnatal day; SD, standard deviation.
a Number born, body weights, and live litter size compared using Dunnett test. Sex ratio and survival compared using Dunn’s test.
b Significantly different from corn oil control (P < 0.05).
c Significantly different from corn oil control (P < 0.01).
Results of the micronucleus test
As shown in Table 11, the frequencies of micronuclei in bone marrow from rats treated with refined NAs did not differ statistically from those in the sham and vehicle control groups. A significant increase in micronucleus frequency was found in material harvested from rats treated with the positive control, cyclophosphamide.
Summary of Results of Micronucleus Data from Rats Treated With Refined Naphthenic Acids.
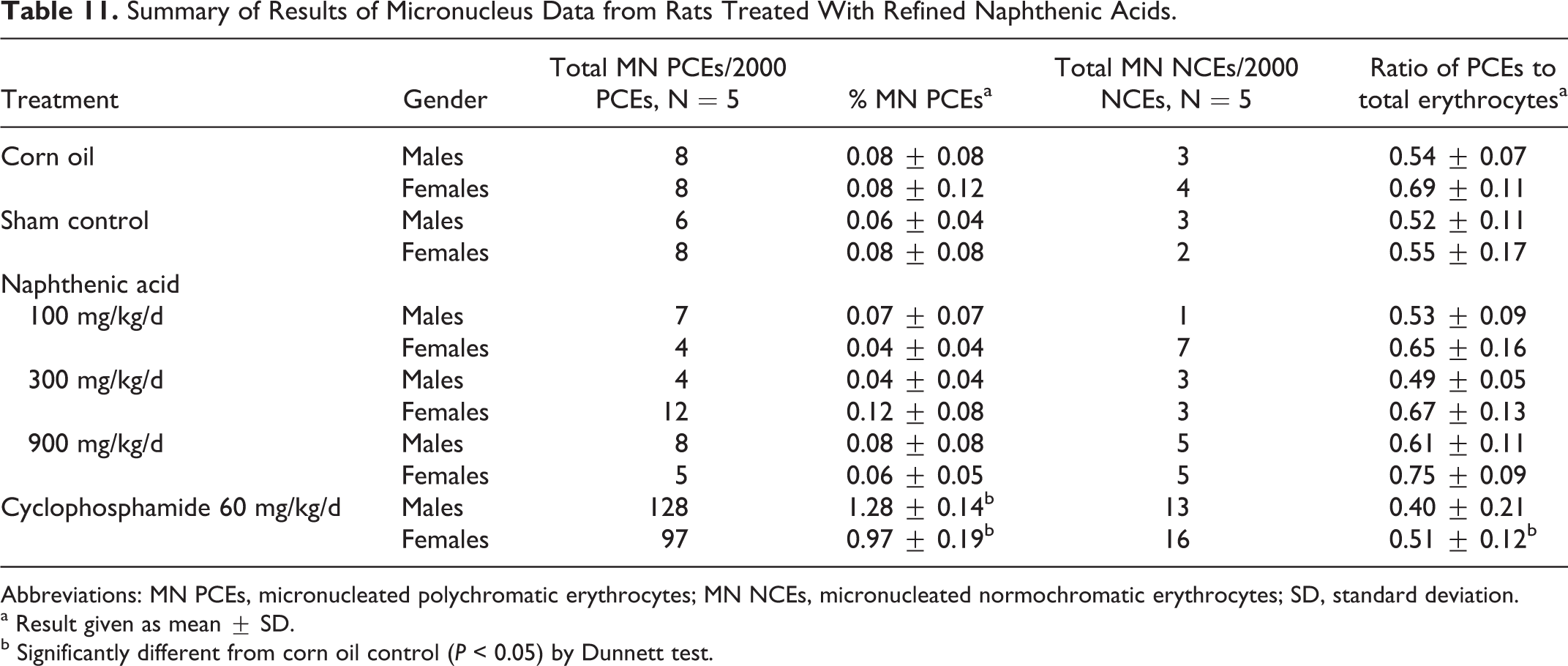
Abbreviations: MN PCEs, micronucleated polychromatic erythrocytes; MN NCEs, micronucleated normochromatic erythrocytes; SD, standard deviation.
a Result given as mean ± SD.
b Significantly different from corn oil control (P < 0.05) by Dunnett test.
Discussion
The objective of this study was to assess the potential for orally administered, refined NAs to cause systemic toxicity and/or affect development or reproductive performance in rats; the potential for genotoxic effects was also assessed. The in vivo testing was consistent with the guidelines for OECD guideline 422, Combined Repeated Dose Toxicity Study with the Reproductive/Developmental Toxicity test. The reproductive toxicity screening study design also included an assessment for the potential of refined NAs to induce micronucleus formation in the bone marrow of male and female rats.
The doses for the reproductive toxicity screening study were based on preliminary screening tests, which indicated that repeated treatment with 1000 mg/kg/d was not lethal but produced acute effects and also affected organ weights and fetal survival and development. Based in part on these data as well as a previous study of the effects of NAs isolated from tailings from an oil sand operation, the doses chosen for the present study were 100, 300, and 900 mg/kg/d.
Within the first hour of dosing, female rats exhibited a number of clinical findings including hunched posture; rocking, lurching, or swaying while ambulating; walking on tiptoes; hypoactivity; and shallow respiration. The male rats exhibited hunched posture but none of the other findings. Similar findings were documented in previous acute toxicity studies. 3 However, aside from these acute observations, most of the rats survived. There were 2 rats that were sacrificed prior to scheduled termination. One of these was a female in the high-dose group, for which the necropsy findings were consistent with acute inflammation of the uterus. The other unscheduled death was also a high-dose group female, sacrificed on lactation day 2 due to complete litter loss. Neither of these early terminations was considered to have been directly due to treatment. There was also some evidence of reduced weight gain in high-dose animals, but the overall differences were less than 10% and not significantly different from control.
The systemic findings were not particularly remarkable. There were no significant findings in the neurological assessments. There were a number of statistically significant findings in the clinical chemistry and hematology evaluations, but the differences were small, not consistent between the genders, and, for the most part, unrelated to microscopic changes. Further, virtually all of these differences were within the range of historical control data at the testing facility and were not considered to have been adverse outcomes.
The gross and pathological assessments did reveal some differences that were treatment related but were unlikely to have been toxicologically important. Liver weights were significantly increased in high-dose groups of both male and female rats, and there was also a statistically significant increase in liver weight of males from the 300 mg/kg/d dose group. The histological findings were essentially limited to minimal evidence of hepatocellular hypertrophy in the high-dose group animals. As none of the liver enzyme markers were increased, this was most likely evidence of enhanced metabolic capacity and adaptive rather than adverse. 33 Kidney weights were significantly elevated in the male rats from the high-dose group but not in the female rats. The histological evidence revealed the presence of hyaline droplets, mostly judged to have been of minimal severity, which increased in frequency in the male rats in a dose-dependent manner. As these were not found in female rats, the histological findings and gender specificity suggested that the kidney changes were the consequence of an α-2u-globulin-related process which is male rat specific and not relevant to humans. 34 –36
Other changes included higher mean thyroid/parathyroid weights with corresponding epithelial hypertrophy and cytoplasmic vacuolation. The histologic changes were mostly judged as minimal. It is plausible that these changes reflected a compensatory response related to the increased metabolic capacity of the liver and rapid turnover of thyroid hormones in rats by comparison to humans. 37,38 Lymphoid depletion of the thymus was observed in the high-dose females, and microscopic findings of cytoplasmic vacuolation of the adrenal cortex were noted in the males and high-dose group females. The lymphoid cortical depletion of the thymus and adrenal cortex vacuolation were considered to have been responses to stress, 39 although cytoplasmic vacuolation of the adrenal cortex can also occur spontaneously 40 or as the result of pharmacological effects. 41 The overall no effect level for all target organ effects was 100 mg/kg/d.
There were no apparent treatment-related effects on mating or on frequency of mating, time to mate, mating success, or length of the gestational period. However, there were significant differences in number of offspring born, number live born as well as growth and survival during early lactation. The most profound effects were seen in the high-dose group, but there were also significant effects in the 300 mg/kg/d group. The overall no effect level of developmental effects was 100 mg/kg/d.
The assessment of genotoxic effects included a mutagenicity test in bacteria and a micronucleus test. The bacterial assay for mutagenic effects produced negative results. The micronucleus test data provided evidence that repeated oral treatment of rats with refined NAs at levels up to 900 mg/kg/d did not significantly increase the incidence of PCEs. As the positive and negative controls responded appropriately, it was concluded that refined NAs did not induce micronuclei in rat bone marrow under the conditions of this assay.
To the extent they could be compared, the data from the present study were qualitatively similar to those previously published, but there were substantial quantitative differences. In an oral toxicity study, repeated administration of NAs derived from Athabasca oil sands (AOSs) to female rats at daily doses of 60 mg/kg/d resulted in significant increases in liver weights and brain weights. There were some small but statistically significant changes in serum chemistry and hematology values, but in the main these were not considered to have been toxicologically important. The pathological changes were principally in the liver, and the most unusual observation was that of seizures in the rats which were observed at the time of dosing but only after 40 days of dosing. 5,42 In the present study, there was a significant increase in liver weights in high-dose females with corresponding liver pathology but at a dose (900 mg/kg/d) much higher than that reported in the previous study. There were no effects on brain weight, and, although there were some acute effects in rats immediately after dosing, there were no observations of seizures. The basis for the qualitative differences between the present and previous studies is not obvious. It is possible that the toxicological differences were related to compositional differences in the test materials or the method of dosing. More specifically, with respect to compositional differences, the NA samples used in the present study was refined and contained NAs with carbon numbers ranging from C10 to C16, whereas the NA samples tested by Rogers had carbon numbers ranging from C14 to C18, and, depending on the method of preparation, may have contained more impurities. Rogers et al 42 commented on some of these issues in a article describing the methods used to isolate, purify, and characterize the AOS-derived test material in their previous study. Their preparation was approximately 85% NAs (as compared to a 70% preparation used in the present study) with low levels of metals and polycyclic aromatic hydrocarbons. However, Rogers also noted that his test material contained approximately equal amounts of 1, 2, 3 and 4-ringed structures, and, in this regard, differed from a commercial preparation containing 70% 1- and 2-ringed structures. He commented that these compositional differences could result in toxicological differences and, for that reason, studies of commercial preparations might not accurately reflect the properties of the AOS-derived material. By comparison, the NA samples used in the present study had 70% 1- and 2-ringed structures.
It is also possible that the toxicological differences were related to methodological differences. If so, 1 factor which may have contributed to the difference is the choice of vehicle for dose administration. In the present study, the test sample was suspended in corn oil whereas in the previous study the test material was administered in aqueous solutions. The choice of vehicle may have influenced uptake of NAs from the gastrointestinal tract, leading to differences in systemic dose.
The 2 most important differences between the present and previous studies were the observations of seizures in the repeated dose study and the quantitative differences in fetal death and resorption in the developmental toxicity studies. More specifically, in the previous repeated dose study, the seizures were not observed until after the 40th day of dosing, whereas in the present study most of the animals were sacrificed before the 40th dosing day. However, because the administered doses used in the present study were so much higher, the total administered dose after 90 days in the previous study (5400 mg/kg) had been achieved within the first 6 dosing days of the present study. So it is difficult to explain the absence of seizures in the present study simply on the basis of the fewer total days of dosing but could be accounted for by differences in absorbed doses.
The comparison of reproductive studies also indicates qualitative similarities but quantitative differences. The most profound effect in the previous study was that the majority of the treated females did not have offspring. More specifically, 14 dams were mated but in the high dose (60 mg/kg/d), only 1 had a litter containing 7 live offspring as compared to an average litter size of 14.3 in the controls. 5,6 By comparison, in the present study there was no reduction in the number of dams with litters although there was a reduction in number of pups born live and in weights of offspring in the high-dose group (900 mg/kg/d). There was also evidence of reduced offspring survival during the postnatal period in the present study; however, in the previous study, all offspring were euthanized at birth, so comparative information on postnatal survival is not available. In summary, there were similarities in outcome between the 2 studies, but the previously reported effects of treatment with 60 mg/kg/d NAs from oil sands were more profound than those associated with treatment with 900 mg/kg/d refined NAs in the present study. As mentioned previously, it seems more likely that the quantitative differences were related to compositional differences in the samples tested but may have been related to differences in internal doses resulting from the use of different dosing vehicles.
In conclusion, based primarily on the evidence of reduced frequencies of live born offspring and reduced offspring survival during the first 4 days of lactation, 100 mg/kg/d was determined to be the no observed adverse effect level for the present study. Further, there was no evidence that the refined NAs were mutagenic under either in vitro or in vivo conditions.
Footnotes
Acknowledgments
The authors would also like to thank Chris Sexsmith for quality assurance support and Lynn Bennett for assistance in manuscript preparation.
Authors’ Note
Some of the statistical analysis was conducted by BioSTAT Consultants, Inc, and the slide evaluation and statistical analysis of data from the micronucleus test were conducted by Midwest Bioresearch, Inc. Compositional information on the test sample was provided by Dr Phillip Fedorak, University of Alberta, Edmonton, Alberta.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors of this article are either employed by companies that manufacture petroleum products or consultants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored and funded by the PHPVTG, an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program. The American Petroleum Institute (API) manages the PHPVTG’s activities.
