Abstract
Higher olefins (HO) are used primarily as intermediates in the production of other chemicals, such as polymers, fatty acids, plasticizer alcohols, surfactants, lubricants, amine oxides, and detergent alcohols. The potential toxicity of five HO (i.e., 1-Octene, Nonene, Decene, Hexadecene, and 1-Octadecene) with carbon ranging from C8 to C18 was examined in a combined repeated dose and reproduction/developmental toxicity screening study (OECD TG 422). These five HO were administered to Han Wistar rats by gavage at 0 (controls), 100, 300, and 1000 mg/kg bw/day. As a group of substances, adaptive changes in the liver (liver weight increase without pathological evidence), as well as increased kidney weight in male rats, were observed in HO with carbon numbers from C8 to C10. The overall systemic no observed adverse effect level (NOAEL) for all HO was determined at 1000 mg/kg bw/day. In the reproductive/developmental toxicity assessment, offspring viability, size, and weights were reduced in litters from females treated with Nonene at 1000 mg/kg bw/day. The overall no observed effects level (NOEL) for reproductive toxicity was considered to be 300 mg/kg bw/day for Nonene and 1000 mg/kg bw/day for the other four HO, respectively. These data significantly enrich the database on the toxicity of linear and branched HO, allowing comparison with similar data published on a range of linear and branched HO. Comparisons between structural class and study outcome provide further supportive data in order to validate the read-across hypothesis as part of an overall holistic testing strategy.
Keywords
Introduction
Higher olefins (HO) are versatile hydrocarbons and used primarily as intermediates for a number of important industrial and consumer products, such as polymers, fatty acids, plasticizer alcohols, surfactants, lubricants, amine oxides, and detergent alcohols. 1 Generally, they are alkenes with six to more than 30 carbons and a single double bond between two of the carbons (sum formula of CnH2n). HO demonstrate low acute toxicity by the oral, inhalation, and dermal routes of exposure, with no potential in genotoxicity and carcinogenicity. 2 Although HO are not eye irritants or skin sensitizer, prolonged exposure of the skin may cause skin irritation. 2
Depending on the manufacturing processes, there are 4 general types of hydrocarbons that may be present in HO (Figure 1): 1. linear alpha olefins (i.e., vinyl—straight chain with a single double bond in the alpha position), 2. linear internal olefins (i.e., cis/trans disubstituted—straight chain with a single double bond in an internal position), 3. branched alpha olefins (i.e., vinylidene—isomerized olefins with a single double bond in the alpha position), and 4. branched internal olefins (i.e., trisubstituted and tetrasubstituted—isomerized olefins with a single double bond in an internal position). Various olefins and branching units referred to in the following as (A) Cis/trans disubstituted (linear internal); (B) Trisubstituted (branched internal); (C) Tetrasubstituted (branched internal); (D) Vinyl (linear alpha); and (E) Vinylidene (branched alpha).
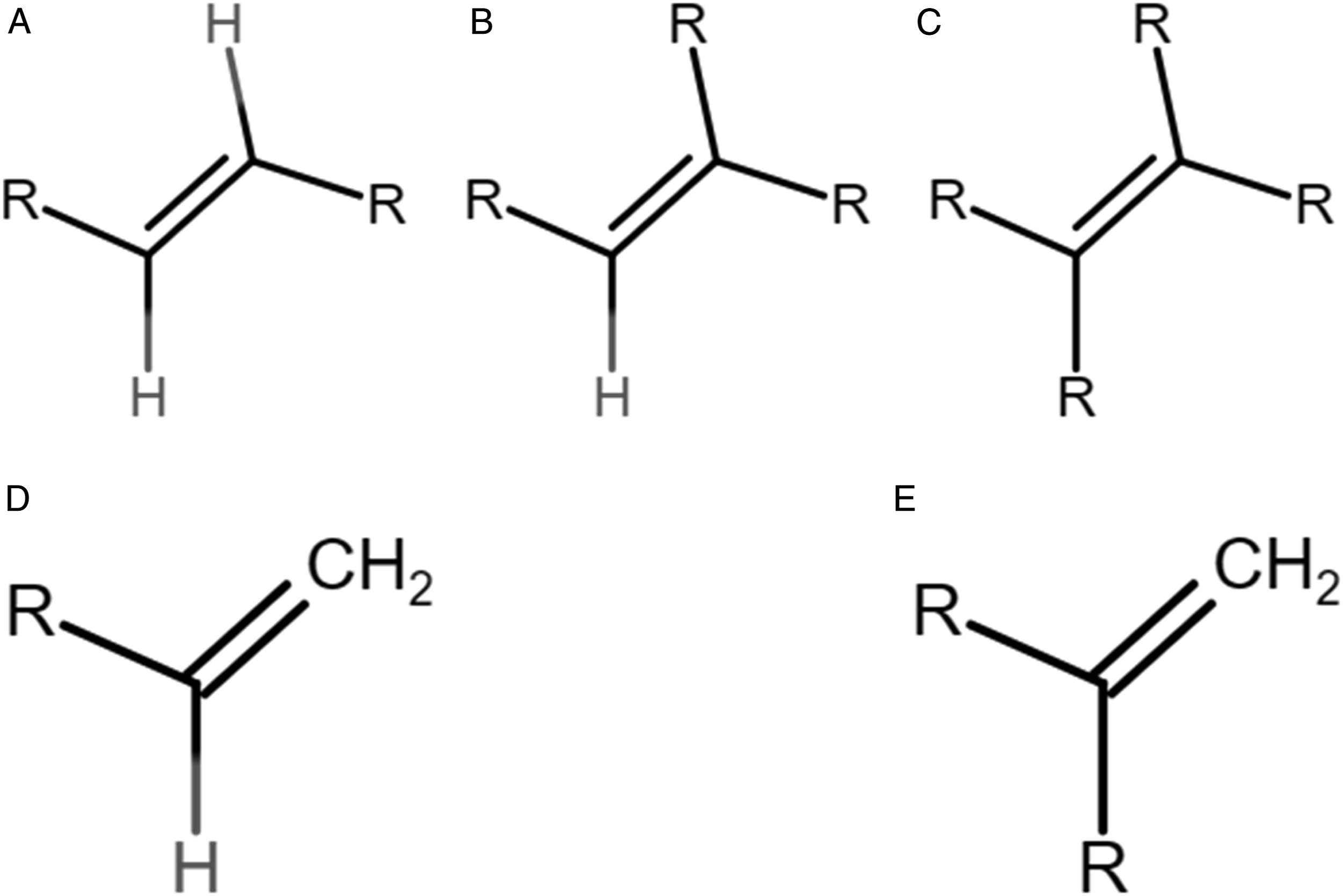
The different positions of the double bond influence the metabolism of HO, which, in turn, may lead to differences in toxicity. Experimental studies in vitro demonstrated that olefins are oxidized by hepatic microsomal enzymes to glycols (diols) via an epoxide intermediate. 3 While hydrolysis of the oxirane ring by epoxides hydrolase is rapid for alpha-olefins, glycol formation for internal- and branched-chain olefins is less efficient due to steric hindrance in the region of the oxirane ring by alkyl groups. This was illustrated for a series of C8-olefins, where the rate of hydrolysis of the epoxide intermediate showed the following trend 4 : 1,2 epoxy-n-C8 > 4,5 epoxy-n-C8 > 2,3 epoxy-iso-C8. In addition, other in vitro investigations, using hepatic microsomal fractions from phenobarbital-induced rats, have demonstrated loss of cytochrome P-450 due to alkylation of the haem function, with monooxygenase inactivation occurring to a greater extent in linear and branched alpha olefins relative to that found with internal olefins (with or without branching) where no or minimal loss of cytochrome P-450 was detected. 5 Altogether, these results suggest that alpha olefins might be biologically more reactive than internal and/or branched olefins. Furthermore, in our previous HO bioavailability study, we applied in silico simulation which showed the predicted reactivity of HO decreased with the following order: linear alpha (vinyl) > branched alpha (vinylidene) > linear internal (cis/trans disubstituted) > branched internal (trisubstituted) > branched internal (tetrasubstituted). 6 However, these results were based in vitro or in silico; meanwhile, we recognize and do not exclude the possibility that internal and branched molecules may induce systemic effects through mechanisms independent of epoxide formation in vivo. On the other hand, we also assessed the bioavailability of a series of HO via rat everted gut testing and concluded that the absorption of intestine is carbon number dependent with poor or barely no absorption if the carbon number is larger than 14 carbon. 6 Based on this data, our hypothesis is that HO with low carbon number and linear alpha configuration may generate more (adverse) effects than the rest of the HO.
Higher Olefins Used in the OECD 422 Studies.
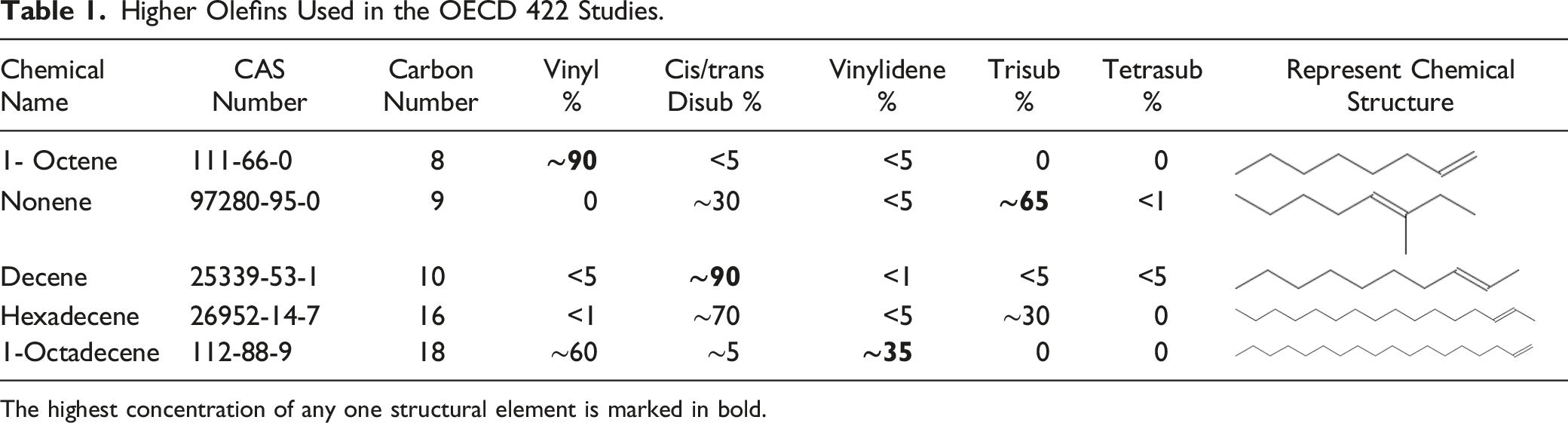
The highest concentration of any one structural element is marked in bold.
Materials and Methods
Test Materials and Dosing
Five test materials 1-octene (CAS No. 111-66-0), Nonene (CAS No. 97280-95-0), Decene (CAS No. 25339-53-1), Hexadecene (CAS No. 26952-14-7), and 1-Octadecene (CAS No. 112-88-9) were either obtained from a commercial source or lab-synthesized. A full list of test materials and their details are provided in Table 1. The selection of the test materials is based on three factors: commercial availability, carbon number, and the composition of the substances.
The test materials were prepared at appropriate concentrations by dissolving in Arachis oil BP (Evans Ltd., Liverpool, UK). The stability and homogeneity of the test material formulations were determined and confirmed to be stable for at least 21 days when stored at approximately 4°C in the dark. Formulations were therefore prepared fortnightly and stored at approximately 4°C in the dark under nitrogen. The analyzed concentrations of test material in the formulations were within the acceptable range (100% ± 10%) for the purposes of the studies.
The dose levels were selected based on available toxicity data including the results of 14-day dose range-finding (DRF) studies. In the DRF studies, no adverse effect of treatment had been apparent at doses up to 1000 mg/kg bw/day, which is the highest dose that is recommended in studies based on OECD TG 422 guideline. A high dose of 1000 mg/kg bw/day was therefore selected for these studies with 300 and 100 mg/kg bw/day as intermediate and low dose, respectively.
Animals and Animal Husbandry
All experiments were performed in compliance with UK Good Laboratory Practice (GLP) standards (Schedule 1, Good Laboratory Practice Regulations (SI 1999 No. 3106 as amended by SI 2004 No. 994)), which, in turn, are in accordance with GLP standards published as OECD Principles on Good Laboratory Practice (revised 1997, ENV/MC/CHEM(98)17) and are also in accordance with the requirements of Directives 2004/9/EC and 2004/10/EC and the United Kingdom’s Animals (Scientific Procedure) Act 1986.8,9
Male and female Wistar Han strain rats were obtained from Harlan Laboratories U.K. Ltd., Blackthorn, Bicester, Oxon, UK. The animals were acclimatized for eight days during which time their health status was assessed. At the start of treatment, the males weighed 311 to 358 g, the females weighed 192 to 234 g, and were approximately 12 weeks old. The animals were housed in a single air-conditioned room within the Harlan Laboratories Ltd., Shardlow, UK Barrier Maintained Rodent Facility. The rate of air exchange was at least fifteen air changes per hour, and the low-intensity fluorescent lighting was controlled to give 12 hours continuous light and 12 hours darkness. The temperature and relative humidity controls were set to achieve target values of 22 ± 3°C and 50 ± 20%, respectively.
Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test
The studies presented were performed in compliance with OECD Test Guideline No. 422: “Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test” (adopted 22 March 1996) (see Appendix Figure A1) at Harlan Laboratories Ltd., Shardlow, Derby, Derbyshire, UK. 7
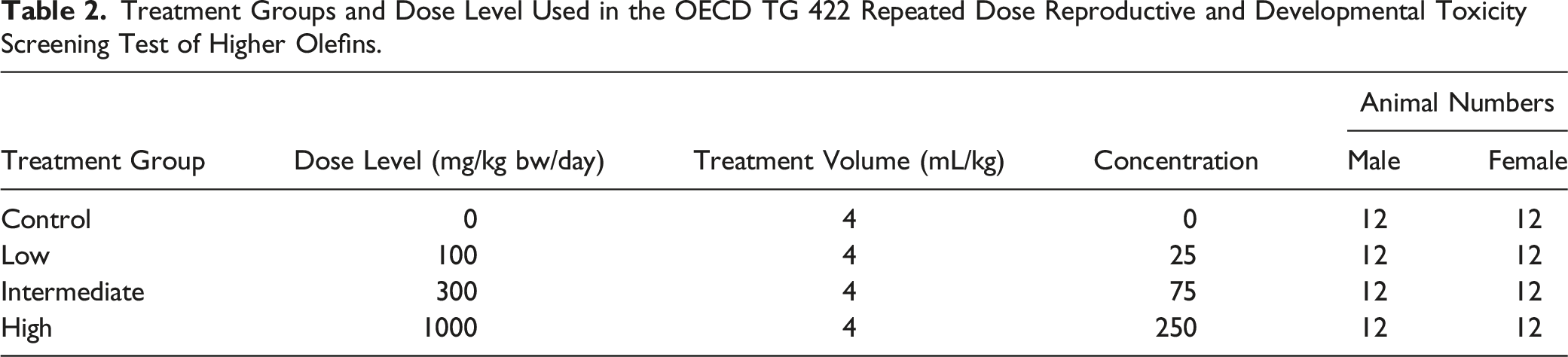
Treatment Groups and Dose Level Used in the OECD TG 422 Repeated Dose Reproductive and Developmental Toxicity Screening Test of Higher Olefins.
Hematological and clinical chemistry investigations were performed on five males and five females randomly selected from each test and control group prior to termination. Animals were not fasted prior to sampling. The following parameters were measured: hemoglobin (Hb), erythrocyte count (RBC), hematocrit (Hct), erythrocyte indices (i.e., mean corpuscular hemoglobin (MCH), mean corpuscular volume (MCV), and mean corpuscular hemoglobin concentration (MCHC)), total leucocyte count (WBC), differential leucocyte count (i.e., neutrophils (Neut), lymphocytes (Lymph), monocytes (Mono), eosinophils (Eos), and basophils (Bas)), platelet count (PLT), reticulocyte count (Retic), prothrombin time (CT) was assessed by ‘Innovin’ and Activated partial thromboplastin time (APTT) was assessed by ‘Actin FS’ using samples collected into sodium citrate solution (.11 mol/L). The following parameters were measured on plasma: urea, calcium (Ca++), glucose inorganic phosphorus (P), total protein (Tot.Prot.), aspartate aminotransferase (ASAT), albumin alanine aminotransferase (ALAT), albumin/globulin (A/G) ratio (by calculation), alkaline phosphatase (AP), Sodium (Na+), creatinine (Creat.), potassium (K+), total cholesterol (Chol), chloride (Cl-), total bilirubin (Bili), and bile acids.
Histopathological examinations were conducted on the preserved organs and tissues of five males and five females in the control and 1000 mg/kg bw/day dose groups. Samples of the following tissues were prepared: adrenals, ovaries, aorta (thoracic), pancreas, bone & bone marrow (femur including stifle joint), pituitary, bone & bone marrow (sternum), prostate, brain (including cerebrum, cerebellum, and pons), oesophagus, caecum, rectum, coagulating gland, salivary glands (submaxillary), colon, sciatic nerve, duodenum, seminal vesicles, epididymides (preserved in Modified Davidson’s fluid), Skin (hind limb), eyes (fixed in Davidson’s fluid), spinal cord (cervical, mid-thoracic and Gross lesions lumbar), heart, spleen, ileum (including Peyer’s patches), stomach, jejunum, thyroid/parathyroid, kidneys, trachea, liver, testes (preserved in Modified Davidson’s fluid), lungs (with bronchi) (were inflated to approximately normal inspiratory volume with buffered 10% formalin before immersion in fixative), thymus, lymph nodes (mandibular and mesenteric), urinary bladder, mammary gland, uterus/cervix, muscle (skeletal), and vagina. In addition, sections of testes from all control and 1000 mg/kg bw/day males were also stained with Periodic Acid-Schiff (PAS) stain and examined. Kidney sections from three males from each treatment group and control group were stained immunohistochemically for α-2u globulin (Rat α-2u globulin (AUG) Affinity Purified Polyclonal Ab, Batch No. AF586, R&D Systems Europe Ltd., 19 Barton Lane, Abingdon Science Park, Abingdon, OX14 3NB, UK) and examined.
Reproductive and Developmental Toxicity
The following reproduction and developmental parameters were recorded for each female and litter: date of pairing, date of mating, date and time of observed start of parturition, date and time of observed completion of parturition, number of offspring born, number of offspring alive, sex of offspring, clinical condition of offspring, and individual offspring weights. In addition, all live offspring were assessed for the physical development via surface righting reflex.
Statistical Analysis
The GraphPad prism software version 8.0 was used for graphical representation. Where appropriate, the homogeneity of variance from mean values was analyzed using Bartlett’s test. Intergroup variance was assessed using suitable ANOVA. Any transformed data were analyzed to find the lowest treatment level that showed a significant effect using the Williams Test for parametric data or the Shirley Test for nonparametric data. If no dose-response was found but the data shows non-homogeneity of means, the data were analyzed by a stepwise Dunnett’s (parametric) or Steel (non-parametric) test to determine the significant difference from the control group. Where the data were unsuitable for these analyses, pair-wise tests were performed using the Student t-test (parametric) or the Mann–Whitney U test (non-parametric). Due to the preponderance of non-normally distributed data, reproductive parameters (implantation losses, offspring sex ratio, and offspring surface righting) were analyzed using nonparametric analyses. To detect the significance of intergroup differences from control, statistical significance was achieved at a level of P < .05.
Results and Discussion
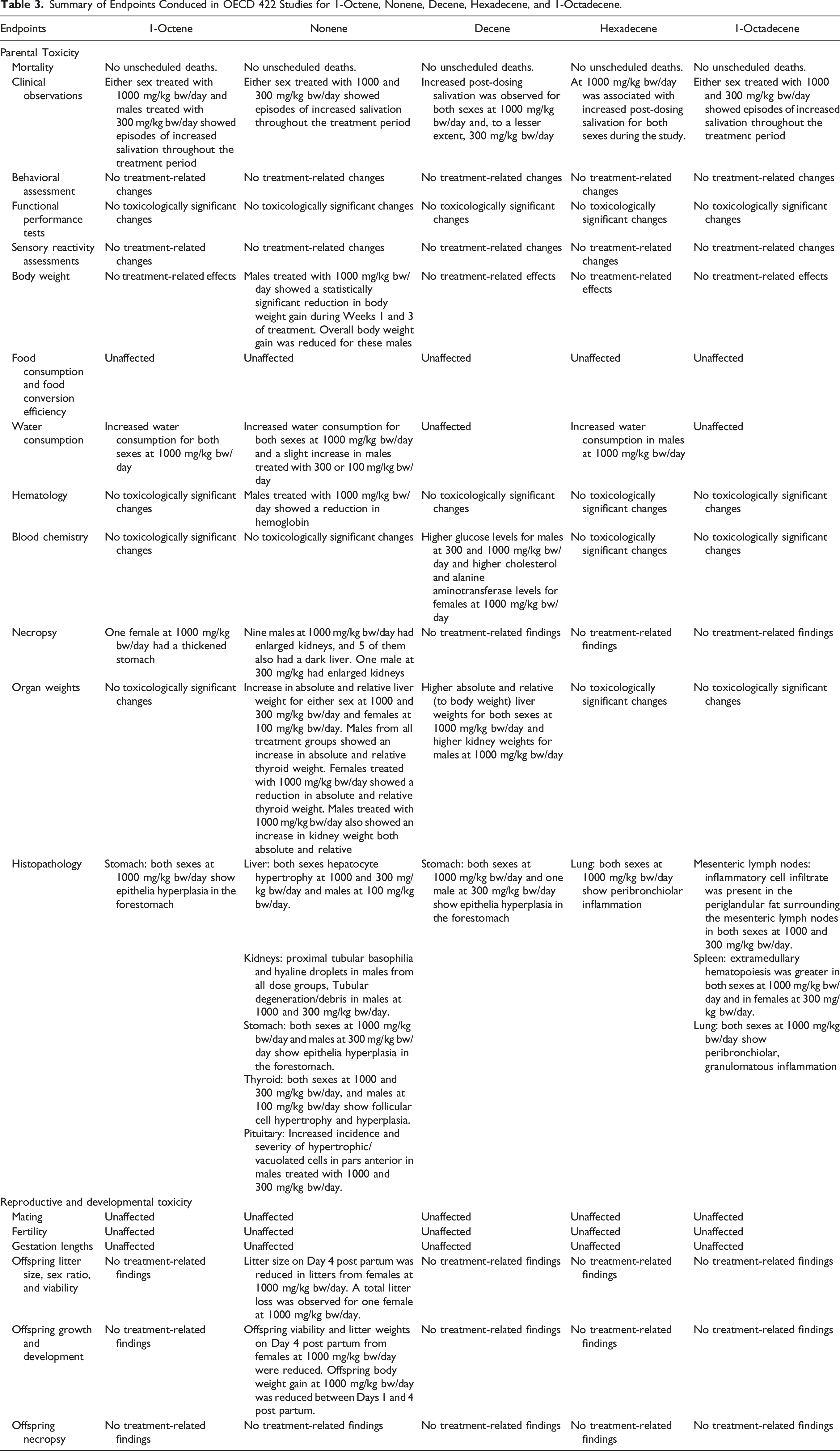
Summary of Endpoints Conduced in OECD 422 Studies for 1-Octene, Nonene, Decene, Hexadecene, and 1-Octadecene.
Parental Toxicity
Generally, for all five substances (i.e., 1-Octene, Nonene, Decene, Hexadecene, and 1-Octadecene), no deaths related to treatment were observed in any of the dose groups (i.e. 0, 100, 300, and 1000 mg/kg bw/day). In detailed clinical observations, all substances induced episodes of increased salivation throughout the treatment period at the high dose. But in both males and females, no such effect was observed at 100 mg/kg bw/day. Increased salivation is considered to reflect distaste of the dosing formulations rather than any adverse systemic effect of treatment. The salivation incidence observed in these studies accompanied by increased water consumption is frequently observed when a slightly unpalatable or irritant test material is administered via the oral gavage route. 14 Besides, no other treatment-related effects on behavioral assessment, functional performance tests (i.e., motor activity and forelimb/hindlimb grip strength), or sensory reactivity were observed in either sex for any substance at any dose level (Table 3).
Body Weight and Body Weight Gain
No statistical significance has been achieved for any of the HO when body weight gains were compared to controls for the various dose levels tested. Except for Nonene, none of the HO showed treatment-related effects on body weight gain in any dose group for either sex throughout the study, including gestation and lactation phases for females. Males treated with Nonene at 1000 mg/kg bw/day showed a statistically significant reduction in body weight gain during Weeks 1 (62%) and 3 (82%) of treatment, and overall body weight and body weight gain were reduced 6.7% (not statistically significant) and 38% (not statistically significant), respectively, when compared to control for these males (Figure 2). However, this effect is considered a result of local irritation of the digestive tract of the test material rather than a true effect of systemic toxicity. This assertion is supported by histopathological findings discussed further on. No reduced body weight gain effects were detected in treated females or males with Nonene at 300 or 100 mg/kg bw/day. In addition, females treated with 300 mg/kg bw/day Nonene showed a statistically significant increase in cumulative body weight gain during the final week of gestation. An increase in body weight gain is considered not to represent an adverse effect of treatment; therefore, the intergroup difference was considered not to be of toxicological importance. Body weight of animals orally administrated with Nonene (A) males, (B) females Pre-mating days, (C) females Gestation days, and (D) females Lactation days. Body weight gain of (E) males and (F) females. * compared to control, P < .05.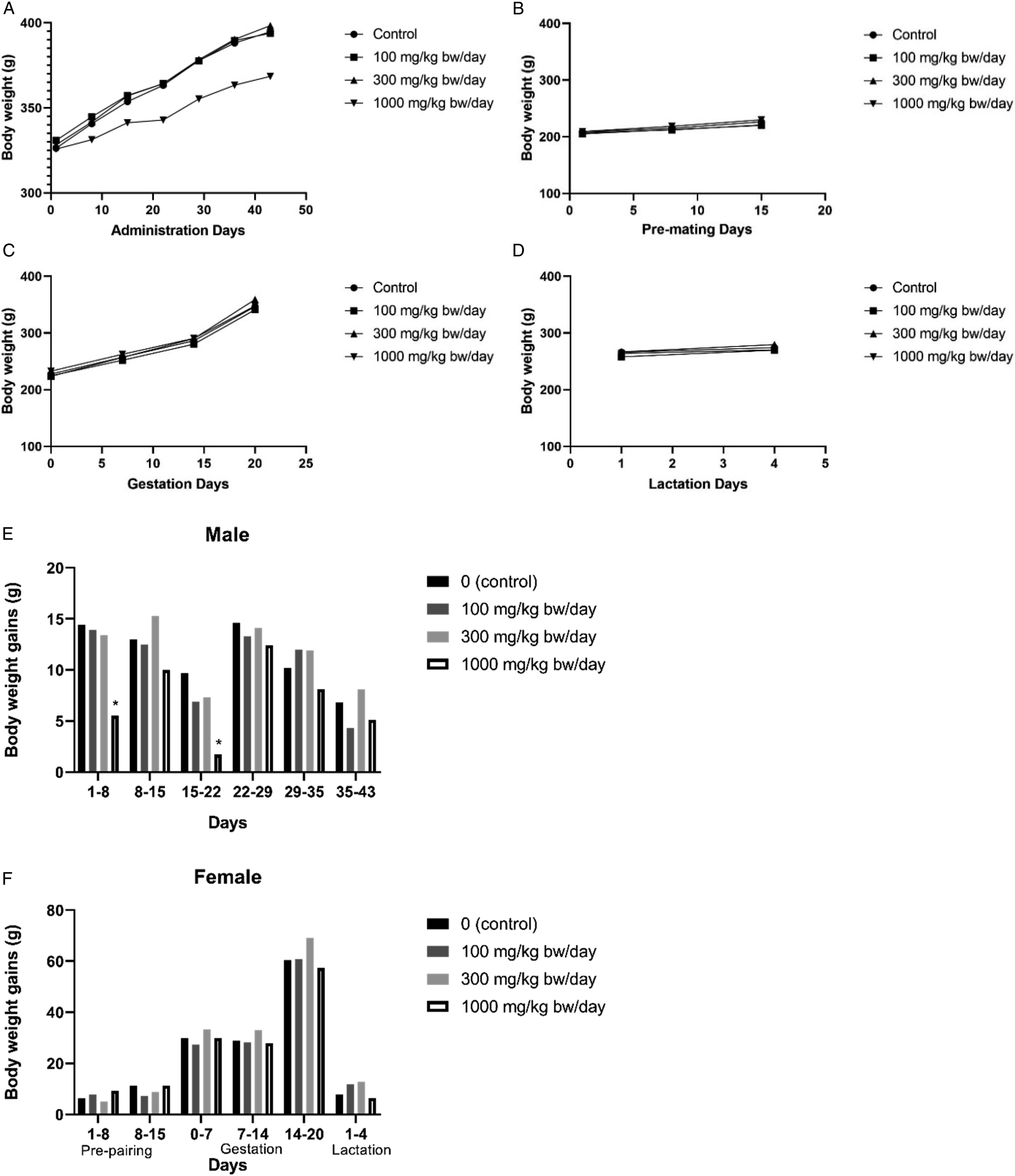
Food and Water Consumption
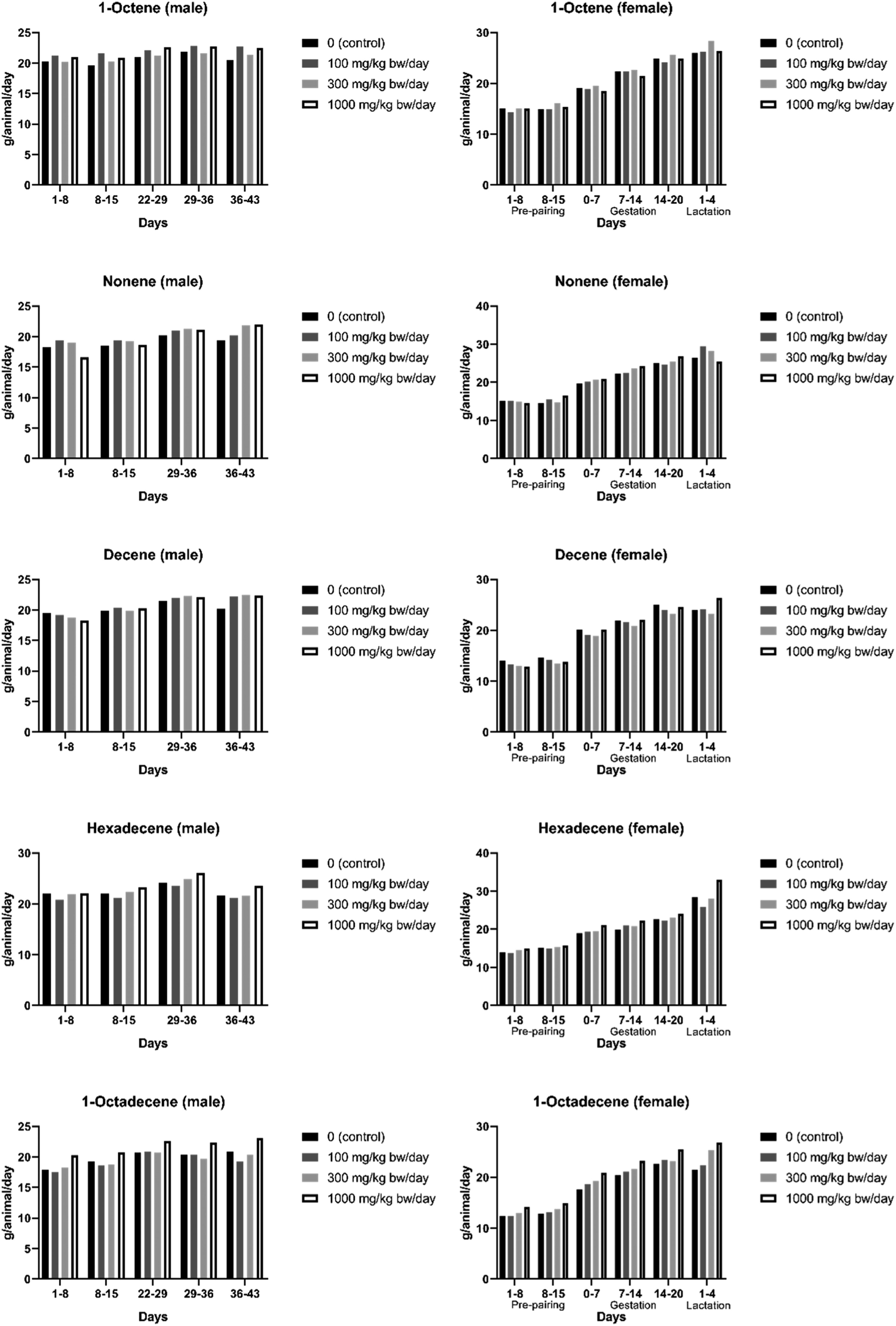
For all test materials, food consumption and food conversion efficiency were unaffected by treatment throughout the study, which included gestation and lactation phases for females at 100, 300, or 1000 mg/kg bw/day (Table 3). However, for water consumption during the pre-pairing phase, only the Decene and 1-Octadecene treatment groups did not reveal any significant difference in overall water consumption. Exposed to 1-Octene caused an increase in overall water consumption during the pre-pairing phase in animals of either sex treated with 1000 mg/kg bw/day; Nonene exposure also caused an increase in water intake at 1000 mg/kg bw/day in both sexes (approximately 53% for male and 111% for females) and a slight increase (approximately 13%) in males treated with 300 or 100 mg/kg bw/day throughout the treatment period; Hexadecene caused an increase in male rats only at 1000 mg/kg bw/day throughout the pre-pairing and post pairing phases of the study (Figure 3). As mentioned in section 3.1, increased water consumption is frequently observed when a test material which is slightly unpalatable or irritant is orally dosed.
14
Pre-mating daily water consumption of rat (both sex) orally administrated with 1-Octene, Nonene, Decene, Hexadecene, or 1-Octadecene.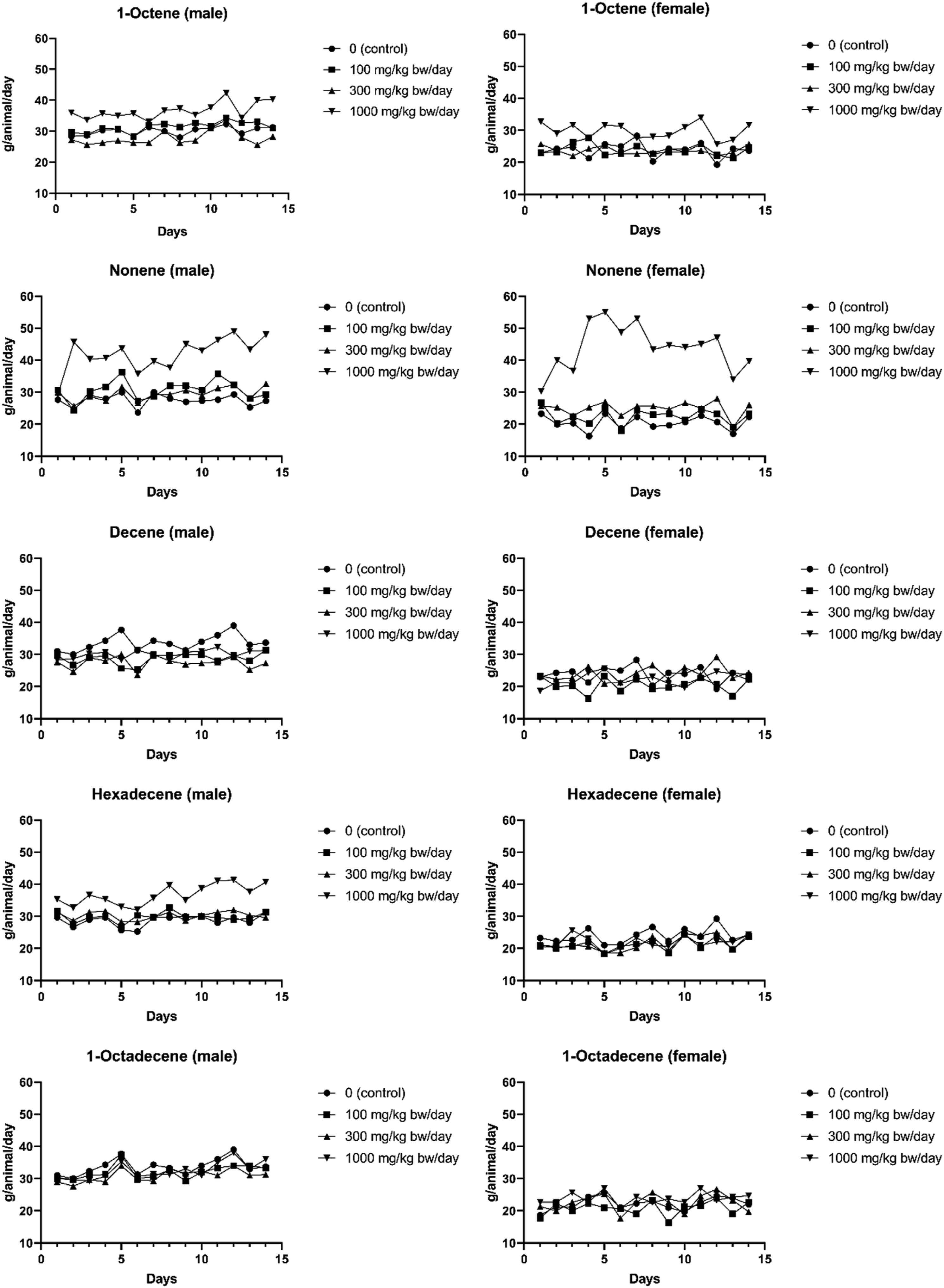
Hematological and Blood Chemistry
Hematological Parameters of Parental Rats Receiving Nonene by Gavage.
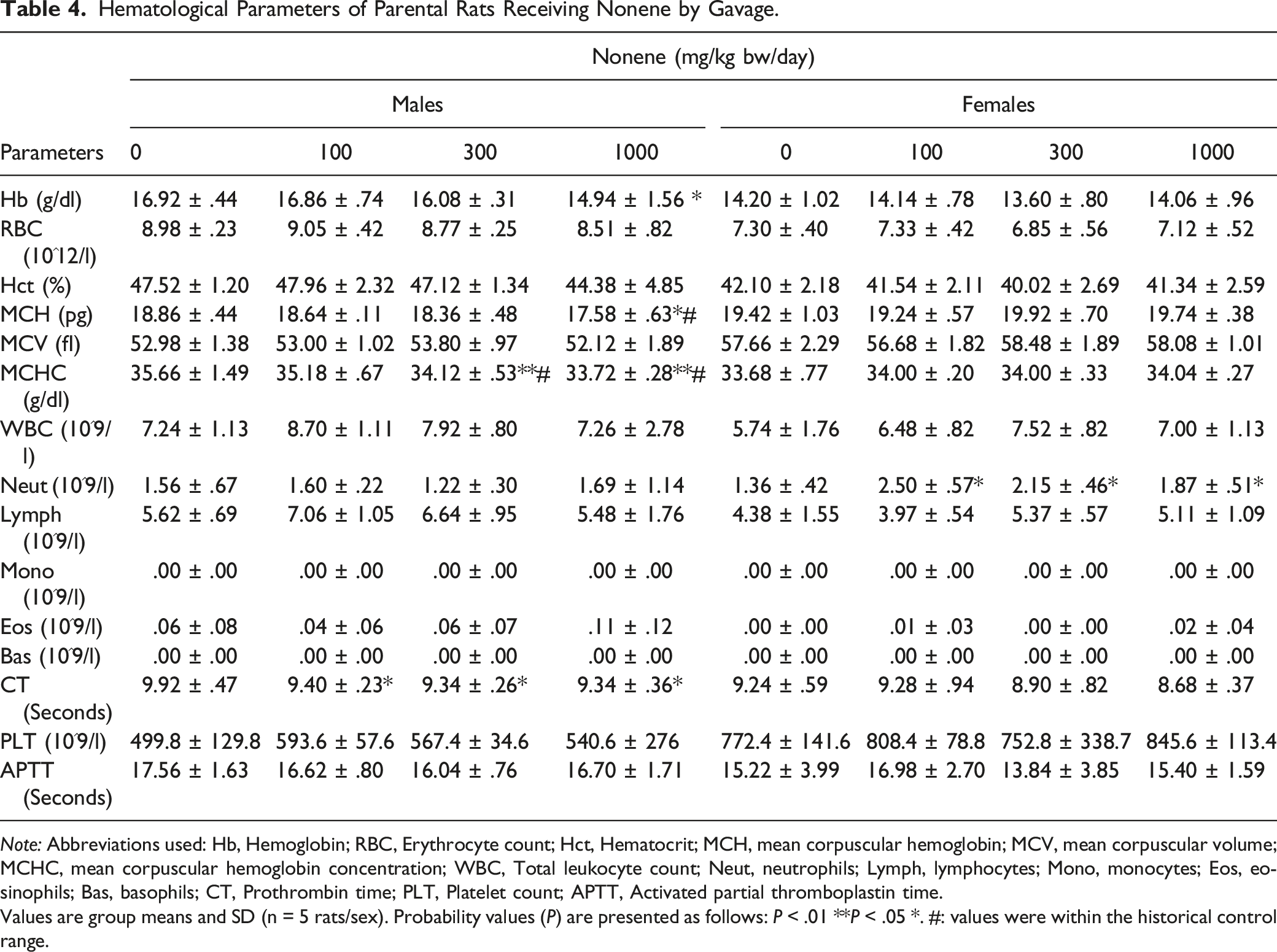
Note: Abbreviations used: Hb, Hemoglobin; RBC, Erythrocyte count; Hct, Hematocrit; MCH, mean corpuscular hemoglobin; MCV, mean corpuscular volume; MCHC, mean corpuscular hemoglobin concentration; WBC, Total leukocyte count; Neut, neutrophils; Lymph, lymphocytes; Mono, monocytes; Eos, eosinophils; Bas, basophils; CT, Prothrombin time; PLT, Platelet count; APTT, Activated partial thromboplastin time.
Values are group means and SD (n = 5 rats/sex). Probability values (P) are presented as follows: P < .01 **P < .05 *. #: values were within the historical control range.
Biochemical Parameters of Parental Rats Receiving Decene by Gavage.
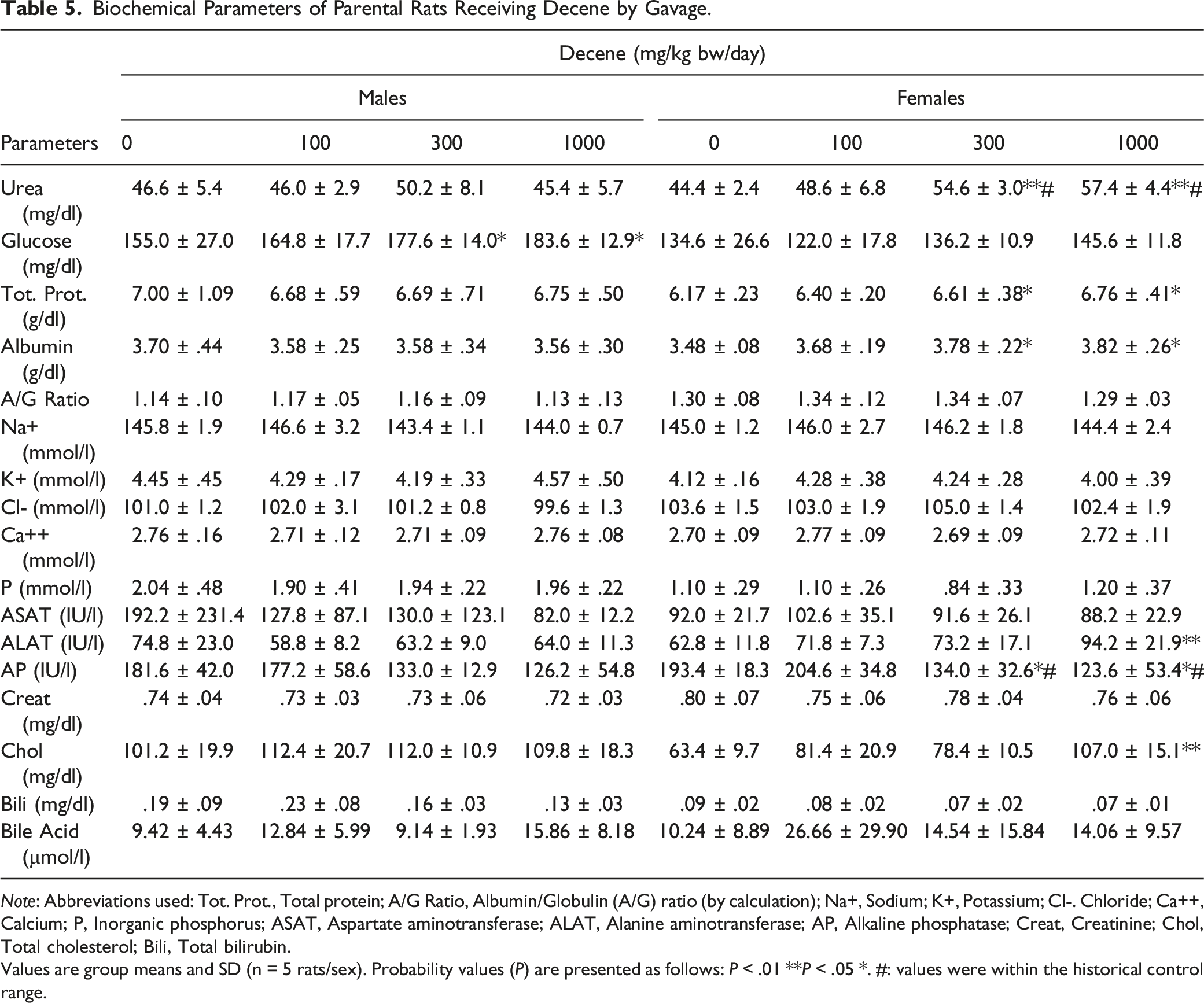
Note: Abbreviations used: Tot. Prot., Total protein; A/G Ratio, Albumin/Globulin (A/G) ratio (by calculation); Na+, Sodium; K+, Potassium; Cl-. Chloride; Ca++, Calcium; P, Inorganic phosphorus; ASAT, Aspartate aminotransferase; ALAT, Alanine aminotransferase; AP, Alkaline phosphatase; Creat, Creatinine; Chol, Total cholesterol; Bili, Total bilirubin.
Values are group means and SD (n = 5 rats/sex). Probability values (P) are presented as follows: P < .01 **P < .05 *. #: values were within the historical control range.
Necropsy, Organ Weights, and Histopathological Findings
It is well recognized that the changes in organ weights together with findings in the necropsy and histopathological examinations are sensitive indicators of the detection of potentially toxic chemicals. 18 Indeed, for all substances, variations as well as common effects were identified during necropsy, organ weights, and histopathological examinations in various treatment groups (Table 3). Thickened stomach was identified in one female rat after treatment with 1000 mg/kg bw/day 1-Octene during the necropsy. Together with epithelial hyperplasia in the forestomach identified at microscopic examination (Appendix Table A1), the stomach changes are considered to be an effect of treatment. While these microscopic findings probably represent an adverse effect of treatment, they are considered to reflect local irritation rather than any adverse systemic toxicity of the test material. In addition, humans do not have a physiological counterpart to the rat forestomach where substances may be present for a prolonged period of time; therefore, the macroscopic stomach changes detected have limited relevance to human toxicity. There were also changes identified in kidneys for one male rat at dose level of 1000 mg/kg bw/day and in lungs for one female rat treated with 300 mg/kg bw/day; however, the intergroup differences were considered not to be of toxicological importance in the absence of any histology correlates. In addition, other findings for 1-Octene exposure were not reported here since the values were well within the normal background range or had no associated histological findings. However, it is worth mentioning those results here so that we can have a better comparison of the observed effects in 1-Octene among other HO which will be discussed later. For instance, males treated with 1000 mg/kg bw/day showed an increase in liver and kidney weight both absolute and relative, males from all treatment groups also showed an increase in absolute and relative adrenal weight, and females treated with 1000 mg/kg bw/day showed an increase in absolute and relative thyroid weight. In terms of risk assessment, the findings observed on 1-Octene exposure would suggest that a NOAEL can be established at 1000 mg/kg bw/day because these findings were not evidence of true systemic toxicity based on the absence of any histology correlates.
Absolute Organ Weights in Rats Receiving Nonene by Gavage.
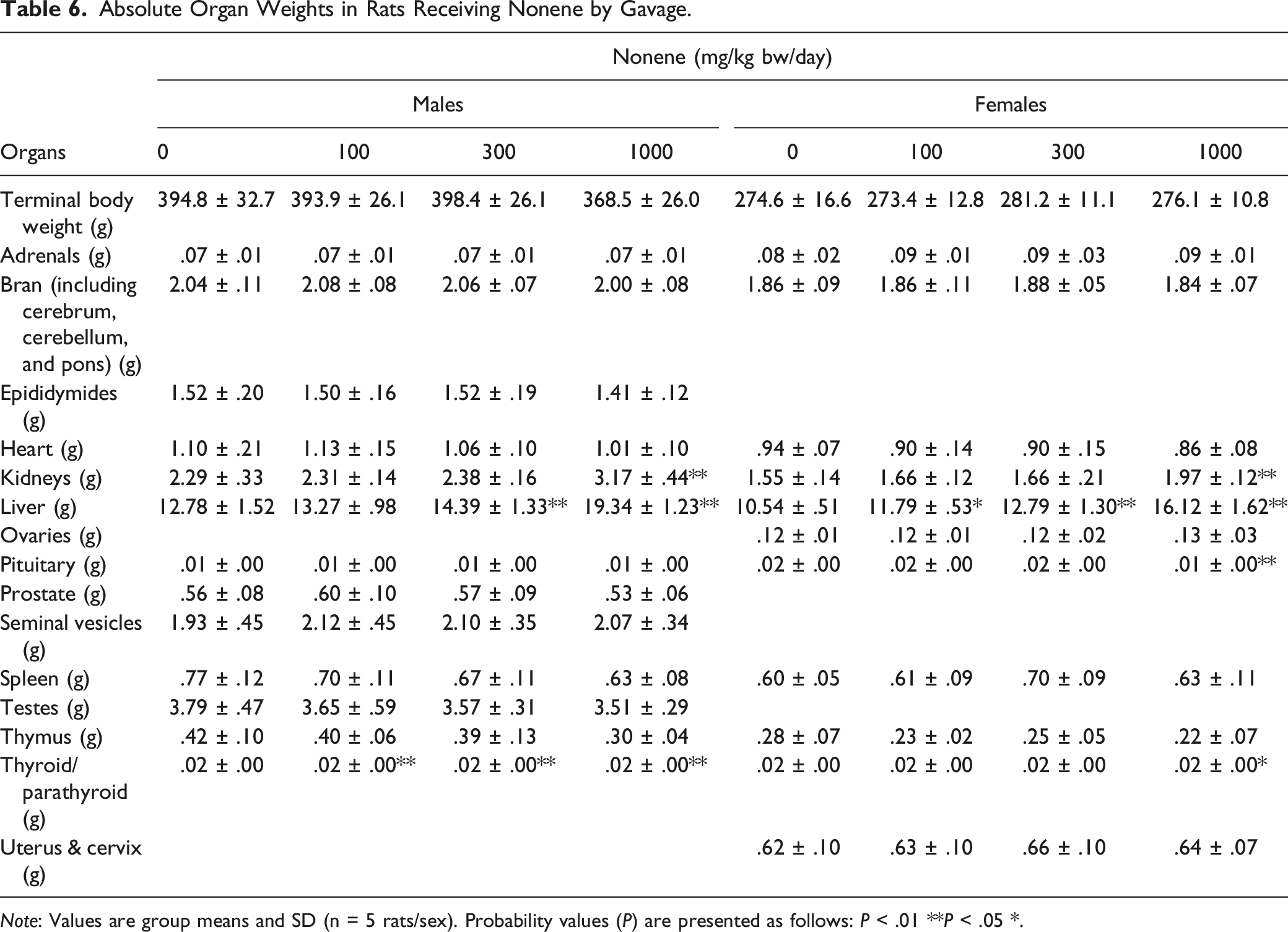
Note: Values are group means and SD (n = 5 rats/sex). Probability values (P) are presented as follows: P < .01 **P < .05 *.
Absolute Organ Weights in Rats Receiving Decene by Gavage.
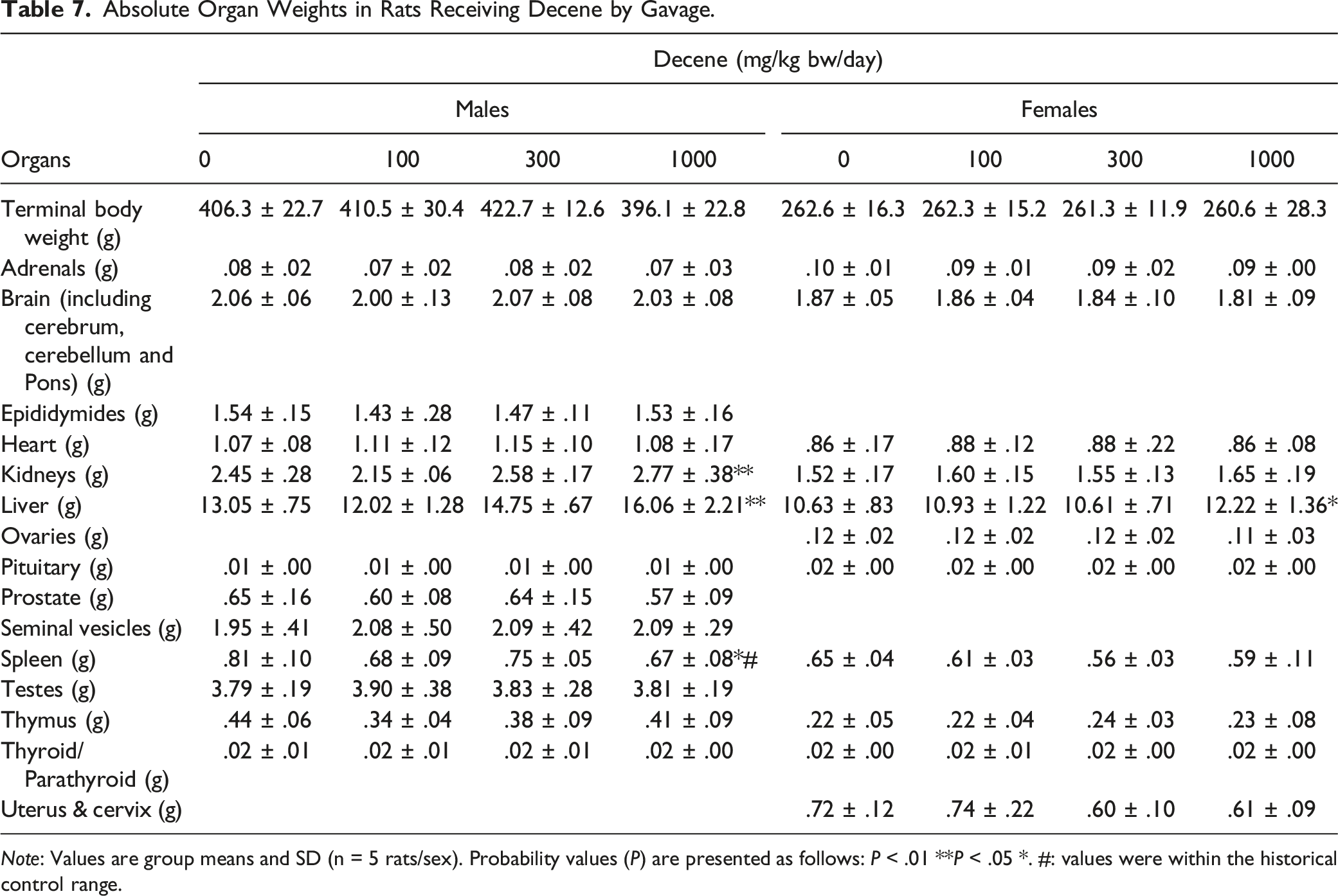
Note: Values are group means and SD (n = 5 rats/sex). Probability values (P) are presented as follows: P < .01 **P < .05 *. #: values were within the historical control range.
Macroscopic necropsy findings, organ weights, or histopathological examinations did not reveal any Hexadecene treatment-related effects identified in the stomach, liver, or kidney, but minimal or moderate peribronchiolar inflammation was observed in the lungs for both sexes at 1000 mg/kg bw/day (Appendix Table A4). The most likely explanation for this finding is that it arose from accidental aspiration of small amounts of the test material during the gavage procedure, which also been shown in other hydrocarbon oral gavage studies, 30 and it is considered very unlikely to have arisen as a consequence of systemic toxicity. As such, this dosage (i.e., 1000 mg/kg bw/day) represents a NOEL for adult toxicity and certainly is a clear NOAEL.
Unlike Hexadecene, the mesenteric lymph nodes and spleen showed significant changes after oral gavage with 1-Octadecene (Appendix Table A5). A minimal or mild inflammatory cell infiltrate was present in the periglandular fat surrounding the mesenteric lymph nodes (MLNs) in animals of either sex treated with 1000 and 300 mg/kg bw/day. The infiltrate was mixed in character, principally composed of lymphocytes and neutrophils, and tended to have a perivascular or perilymphatic orientation. The incidence and severity of extramedullary hematopoiesis was increased in the spleen in animals of either sex treated with 1000 mg/kg bw/day and in females treated with 300 mg/kg bw/day. The inter-group differences in females were not clearly dosage-related, and taking into account the low number of animals examined and the absence of supportive intergroup differences in hematological values or bone marrow findings, these differences were considered more likely to have arisen as a result of individual variation. Similar effects (i.e., histiocytosis in MLNs and spleen changes) were also observed with other similar molecular weight hydrocarbon molecules (i.e., mineral oil saturated hydrocarbons) after oral exposure of rats, and the joint FAO/WHO Expert Committee on Food Additives (JECFA 2012) considered that these effects observed in MLNs do not represent an adverse effect but rather should be considered a nonspecific, adaptive change of low toxicological concern. 31 In addition, there were no statistically significant differences in adult organ weights that, in the absence of any evidence of histopathological change, were considered to be of any toxicological significance. In conclusion, a NOAEL can be established at 1000 mg/kg bw/day for both sexes because the findings were of low toxicological concern.
Reproductive and Developmental Findings
Reproductive/Developmental Parameters in Rats Treated Orally With Nonene.
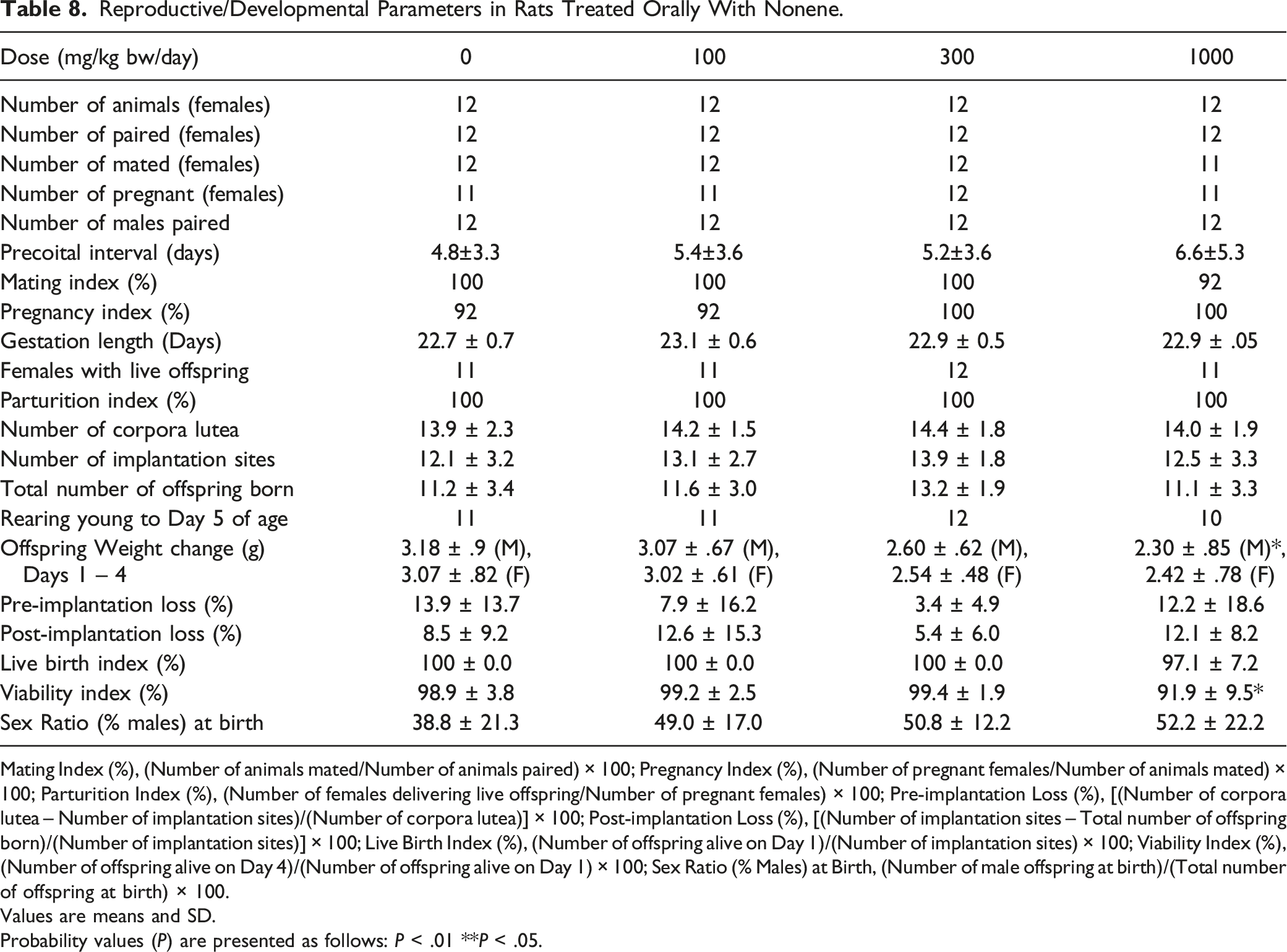
Mating Index (%), (Number of animals mated/Number of animals paired) × 100; Pregnancy Index (%), (Number of pregnant females/Number of animals mated) × 100; Parturition Index (%), (Number of females delivering live offspring/Number of pregnant females) × 100; Pre-implantation Loss (%), [(Number of corpora lutea – Number of implantation sites)/(Number of corpora lutea)] × 100; Post-implantation Loss (%), [(Number of implantation sites – Total number of offspring born)/(Number of implantation sites)] × 100; Live Birth Index (%), (Number of offspring alive on Day 1)/(Number of implantation sites) × 100; Viability Index (%), (Number of offspring alive on Day 4)/(Number of offspring alive on Day 1) × 100; Sex Ratio (% Males) at Birth, (Number of male offspring at birth)/(Total number of offspring at birth) × 100.
Values are means and SD.
Probability values (P) are presented as follows: P < .01 **P < .05.
Trend of Current Data and Compare with Other Publicly Available HO Data
The overall results show that all five tested HO have comparable levels of low toxicity in rats. Specifically, in 28 days repeated dose toxicity studies, 1-Octene, Nonene, Decene, Hexadecene, and 1-Octadecene show a NOAEL for systemic toxicity at 1000 mg/kg bw/day. Although 1-Octadecene showed changes in the mesenteric lymph nodes and spleen (with a NOEL at 100 mg/kg bw/day), the observed differences were not considered to be of any toxicological significance in the absence of supportive evidence, for instance, hematological values or bone marrow finding correlations. In addition, there were typical effects observed after administration of the test materials due to the chemical properties. For instance, increased post-dosing salivation and water consumption due to distaste of the dosing formulations was seen in several instances. Also, common effects, such as microscopic changes in the forestomach and increased liver and kidneys weight, were only observed after administration of low carbon number alkenes (i.e., C8, C9, and C10) in contrast to high carbon number alkenes (i.e., C16 and C18). Besides the well-known irritative properties of low molecular weight alkenes, most of observed effects suggest that low carbon number alkenes are more readily metabolized than high carbon number alkenes. One of the possible explanations for the observed phenomenon is the bioavailability of the test material. Our previous in vitro gut absorption study showed that the carbon number, rather than other aspects of olefin structure (alpha or internal olefin; presence of branching; odd or even number chain length), determine the bioavailability after ingestion, suggesting that above a certain carbon number (∼C14) uptake from the gut is likely to be very low. 6 Another aspect linked to bioavailability is the viscosity of the HO. According to the database of ECHA, a trend of increasing viscosity with increasing carbon number is seen in these types of substances. As carbon number increases so does the molecular weight so that viscosity acts as an indirect measure of molecular weight. Consequently and according to Kramers’ theory, increased chemical viscosity is associated with a decrease in the maximum rate of catalysis by enzymes. 32 This might explain why some effects were only observed in low carbon number alkenes simply because high carbon number alkenes were metabolized slower than low carbon number alkenes or not at all. However, from the molecular weight point of view, comparing Nonene and Hexadecane (both Nonene and Hexadecane have similar composition (i.e., mainly Disub and Trisub)), the dose level of Nonene on a molar base is close to twice that of Hexadecane. The observed effects of Nonene treatment at 1000 mg/kg bw, which are absent with Hexadecane treatment, may be simply due to the dose level which is, on a molar basis, almost twice as high for Nonene than for Hexadecane. Overall, based on the NOAELs, all five HO appear not to be different with regard to repeated dose toxicity, while based on NOEL, low carbon number HO showed some effects due to higher bioavailability and irritation. For the reproductive/developmental toxicity, four HO (i.e., 1-Octene, Decene, Hexadecene, and 1-Octadecene) showed a NOEL at 1000 mg/kg bw/day, and only Nonene demonstrated reproductive toxicity effects at 1000 mg/kg bw/day based on offspring viability. However, according to the composition information (Table 1), a similar effect was expected to be observed with Hexadecane, whereas it has a NOEL for the reproductive/developmental toxicity at 1000 mg/kg bw/day. Therefore, bioavailability is not the only factor contributing to the reproductive toxicity effects, and other factors, such as the trisubstituted (branched internal) composition, may play a role.
Other Sub-acute And/Or Reproduction/Developmental Toxicity Studies Tested on Higher Olefins Which Not Reported in Current Report.
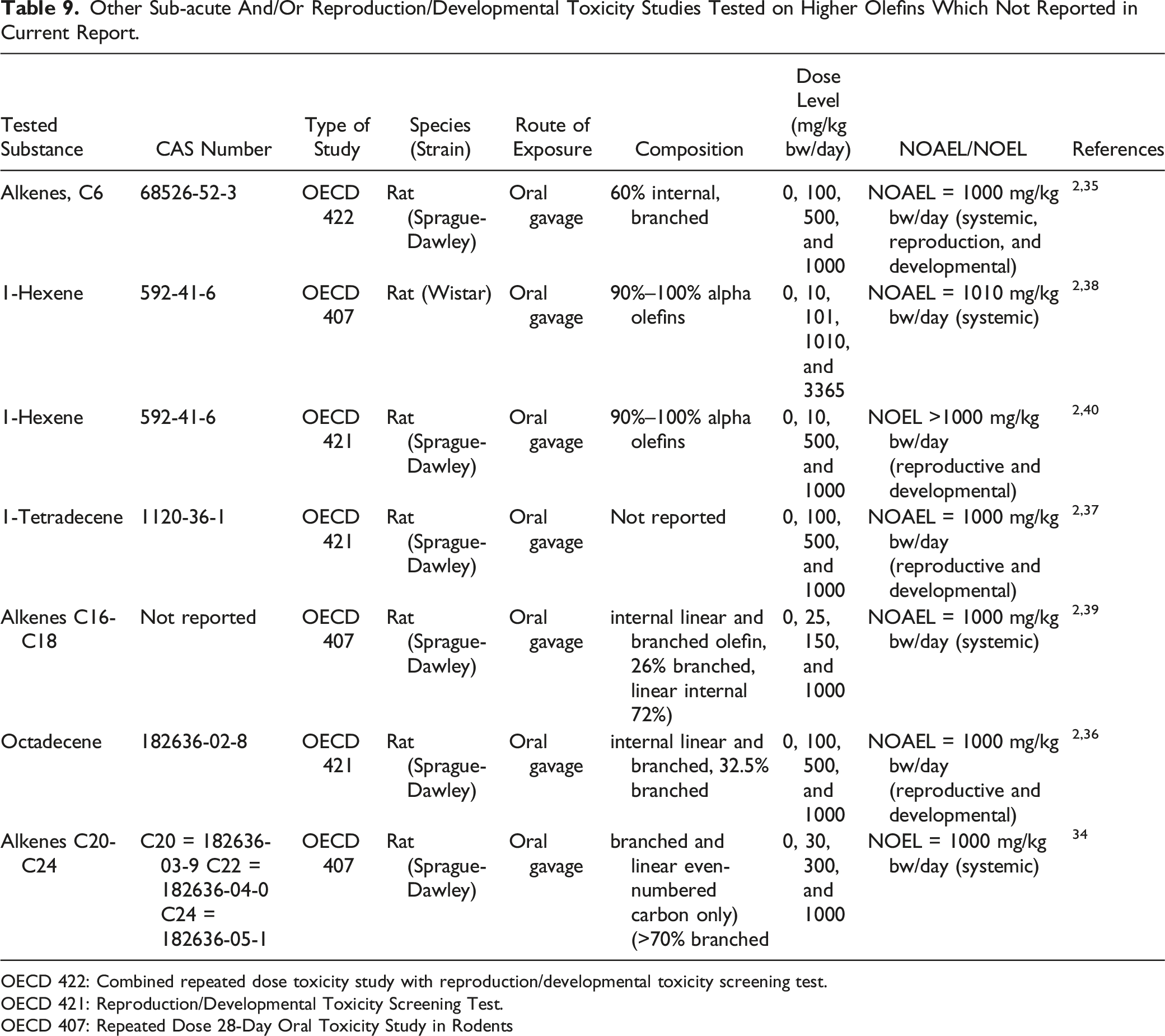
OECD 422: Combined repeated dose toxicity study with reproduction/developmental toxicity screening test.
OECD 421: Reproduction/Developmental Toxicity Screening Test.
OECD 407: Repeated Dose 28-Day Oral Toxicity Study in Rodents
By comparing the results reported in the current report with previously published data, the irritative effects, as well as the increased liver and kidney weight observed with C6 and C14, were also confirmed in the present report for C8, C9, and C10. However, these observed effects are considered either adaptive (i.e., liver effects) or unrelated to humans (i.e., male rat kidney effects because of alpha-2-urinary globulin deposition). In addition, HO with carbon numbers > C16 showed nearly no adverse effect that are related to the test material in the sub-acute toxicity studies. Furthermore, the observed effects were rather dependent on the carbon numbers than the double-bond position (i.e., type of the HO). Altogether, based on the data reported in the current study, the trend of HO (i.e., from C6 to C24) for systemic toxicity, which has NOAEL values of 1000 mg/kg bw/day, is consistent.
For reproductive and developmental toxicity-related endpoints, no toxicologically meaningful differences were noted in F0 and F1 for all HO reported in Table 9 (i.e., 1-hexene, alkenes C6 1-tetradecene, and octadecene). Therefore, the NOAEL for HO reported in Table 9 regarding the reproductive and developmental toxicity was determined at 1000 mg/kg bw/day. By pooling all available HO reproductive and developmental toxicity data together (from C6 to C24), only C9 (i.e., Nonene) showed effects on developmental toxicity based on litter effects, including reduced size, weight, and viability at 1000 mg/kg bw. In addition, no trend was observed for reproductive and developmental toxicity either based on carbon number or type of double-bond. In order to further explore the observed effects of Nonene related to fetal development, a prenatal developmental toxicity study according to the OECD TG 414 guidelines and an extended one-generation reproductive toxicity study (EOGRTS) according to the OECD TG 443 guidelines are suggested to be conducted.
Conclusions
Our initial hypothesis was that HO with low carbon number and linear alpha configuration may generate more adverse effects than the rest of the HO. Based on evidence from the combined oral repeated dose and reproductive/development toxicity screening studies performed according to OECD TG 422, in rats, except for nonane, all other four tested HO were not considered to cause adverse systemic, reproductive, and developmental toxicity at dose levels up to 1000 mg/kg bw/day as no toxicologically significant effects on males or females were observed for any toxicologically relevant endpoint, even for the low carbon number and linear alpha configurations. Nonene consisting of highly branched molecules showed a NOEL of 300 mg/kg bw/day for reproductive toxicity. All observed effects in current study were consistent with previously published data on these substances or similar substances.
Pooling all available information, the generic effects of HO are (a) increased liver weight (adaptive liver response) observed from C6 to C14; (b) increased kidneys weight observed in males (hyaline droplets formation or alpha-2-urinary globulin nephropathy) from C6 to C14; (c) gavage induced forestomach local irritation observed from C6 to C10; (d) decreased gut absorption with increasing carbon chain length; (e) decreased severity of systemic effect by increasing carbon chain; and (f) Nonene (C9) seemingly an outlier showed effects on both systemic (i.e., stomach changes, organ weight, and microscopic findings in stomach, liver, kidney, thyroid, and pituitary) and reproductive toxicity with a NOEL of 300 mg/kg bw/day, indicating that at equivalent molecular weight basis, highly branched HO may cause more severe effects, apparently independent of the double bond position.
Footnotes
Acknowledgments
The authors would like to thank all members of the HOPA REACH Consortium (Higher Olefins and Poly Alpha Olefins REACH Consortium) and its members for helpful discussions and input during development of the manuscript and for assistance in preparation of the manuscript.
Author Contributions
The submitting author affirms that all individuals listed as authors agree that they have met the criteria of authorship and agree to the conclusions of the study. In order to meet the requirements of authorship, each author must have contributed to at least one aspect of each of the four criteria, as listed below. Please note that for Criteria 1 and 2, authors only to meet one of the two items listed. These criteria are not to be used as a means to disqualify colleagues from authorship who otherwise meet authorship criteria by denying them the opportunity to meet criteria 2 or 3. Therefore, all individuals who meet the first criterion should have the opportunity to participate in the drafting, review, and final approval of the manuscript. Any individuals not meeting the criteria may be mentioned in the Acknowledgements section of the manuscript.
Per the criteria defined by the International Committee for Medical Journal Editors (ICJME), please note the contribution made by each author listed in the manuscript. Please select items from the drop down menu.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors of this article are either employed by companies that manufacture petroleum products or consultants.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The HOPA REACH Consortium (Higher Olefins and Poly Alpha Olefins REACH Consortium, website: ![]() ) and its members provided financial support for Penman Consulting staff and consultant participation, and the employers of the other authors provided salary and travel support in the normal course of their work.
) and its members provided financial support for Penman Consulting staff and consultant participation, and the employers of the other authors provided salary and travel support in the normal course of their work.
Correction (November 2023):
In Table 1, there was a typo in the CAS no. of Nonene. The correct CAS no. is CAS 97280-95-0 and it has been corrected in the article.
Appendix
Summaries of Key Histopathological Findings in Rats Administered 1-Octene. -, Not examined; +, Minimal; ++, Slight; +++, Moderate.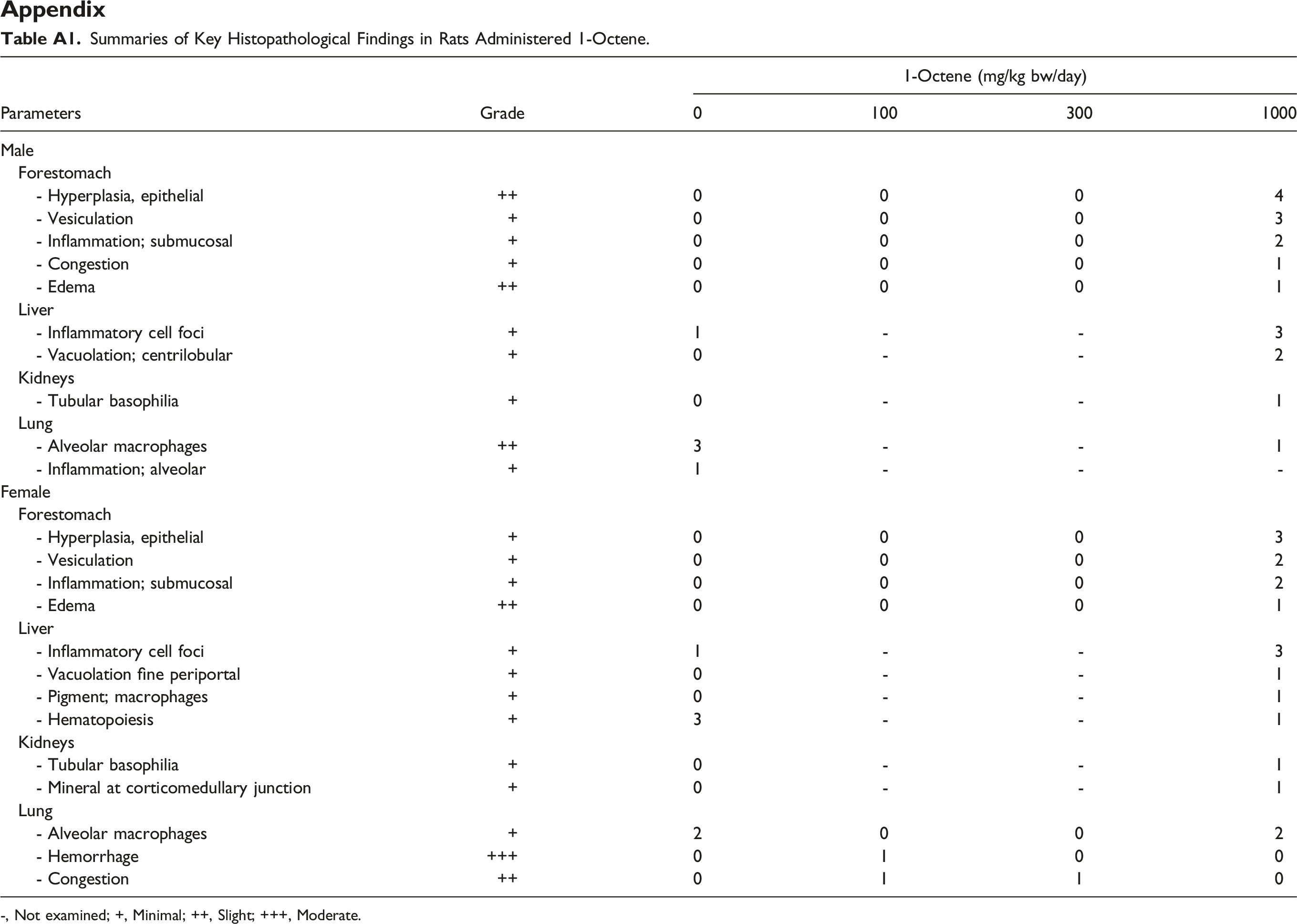
Parameters
Grade
1-Octene (mg/kg bw/day)
0
100
300
1000
Male
Forestomach
- Hyperplasia, epithelial
++
0
0
0
4
- Vesiculation
+
0
0
0
3
- Inflammation; submucosal
+
0
0
0
2
- Congestion
+
0
0
0
1
- Edema
++
0
0
0
1
Liver
- Inflammatory cell foci
+
1
-
-
3
- Vacuolation; centrilobular
+
0
-
-
2
Kidneys
- Tubular basophilia
+
0
-
-
1
Lung
- Alveolar macrophages
++
3
-
-
1
- Inflammation; alveolar
+
1
-
-
-
Female
Forestomach
- Hyperplasia, epithelial
+
0
0
0
3
- Vesiculation
+
0
0
0
2
- Inflammation; submucosal
+
0
0
0
2
- Edema
++
0
0
0
1
Liver
- Inflammatory cell foci
+
1
-
-
3
- Vacuolation fine periportal
+
0
-
-
1
- Pigment; macrophages
+
0
-
-
1
- Hematopoiesis
+
3
-
-
1
Kidneys
- Tubular basophilia
+
0
-
-
1
- Mineral at corticomedullary junction
+
0
-
-
1
Lung
- Alveolar macrophages
+
2
0
0
2
- Hemorrhage
+++
0
1
0
0
- Congestion
++
0
1
1
0
Summaries of Key Histopathological Findings in Rats Administered Nonene. -, Not examined; +, Minimal; ++, Slight; +++, Moderate.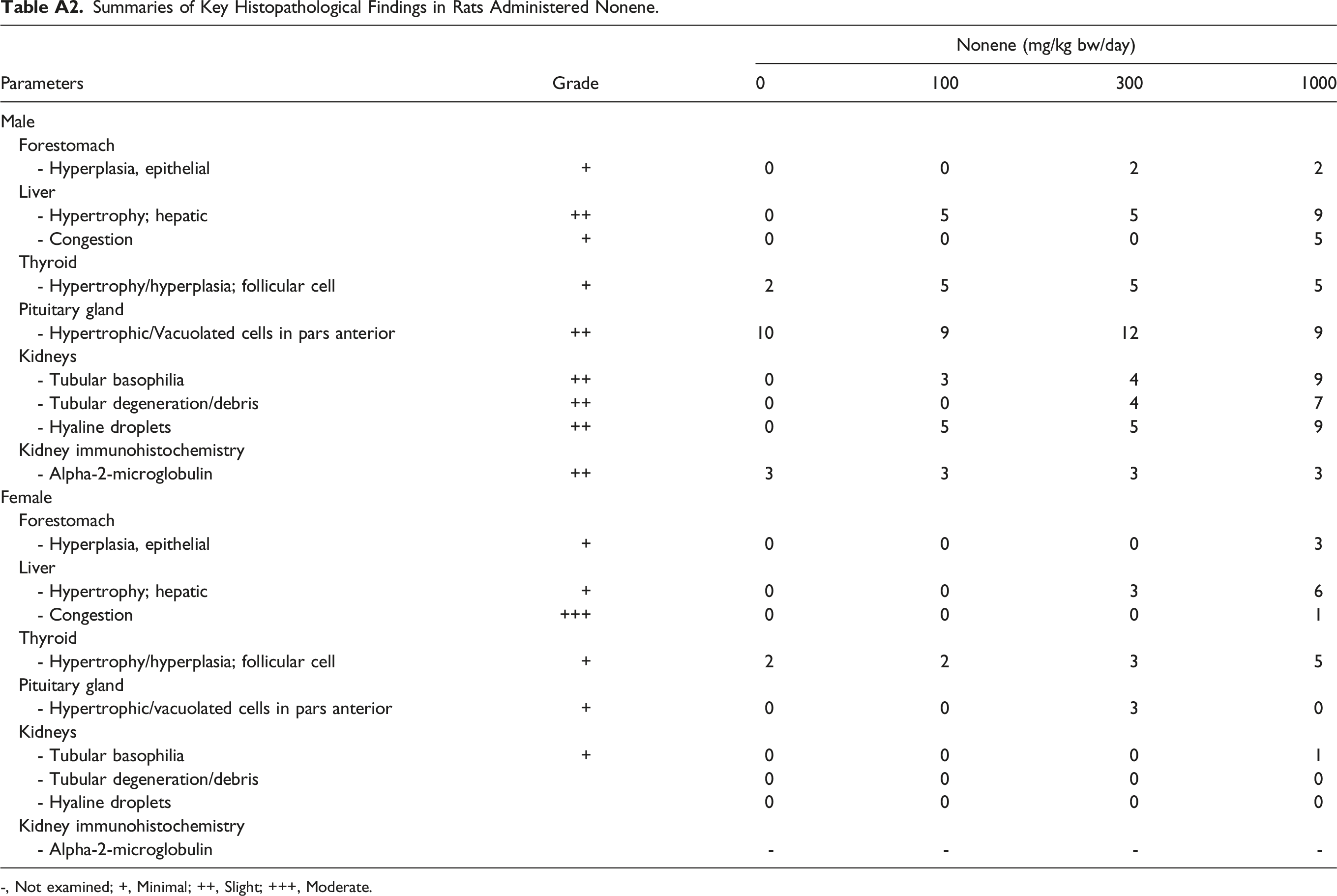
Parameters
Grade
Nonene (mg/kg bw/day)
0
100
300
1000
Male
Forestomach
- Hyperplasia, epithelial
+
0
0
2
2
Liver
- Hypertrophy; hepatic
++
0
5
5
9
- Congestion
+
0
0
0
5
Thyroid
- Hypertrophy/hyperplasia; follicular cell
+
2
5
5
5
Pituitary gland
- Hypertrophic/Vacuolated cells in pars anterior
++
10
9
12
9
Kidneys
- Tubular basophilia
++
0
3
4
9
- Tubular degeneration/debris
++
0
0
4
7
- Hyaline droplets
++
0
5
5
9
Kidney immunohistochemistry
- Alpha-2-microglobulin
++
3
3
3
3
Female
Forestomach
- Hyperplasia, epithelial
+
0
0
0
3
Liver
- Hypertrophy; hepatic
+
0
0
3
6
- Congestion
+++
0
0
0
1
Thyroid
- Hypertrophy/hyperplasia; follicular cell
+
2
2
3
5
Pituitary gland
- Hypertrophic/vacuolated cells in pars anterior
+
0
0
3
0
Kidneys
- Tubular basophilia
+
0
0
0
1
- Tubular degeneration/debris
0
0
0
0
- Hyaline droplets
0
0
0
0
Kidney immunohistochemistry
- Alpha-2-microglobulin
-
-
-
-
Summaries of Key Histopathological Findings in Rats Administered Decene. -, Not examined; +, Minimal; ++, Slight; +++, Moderate.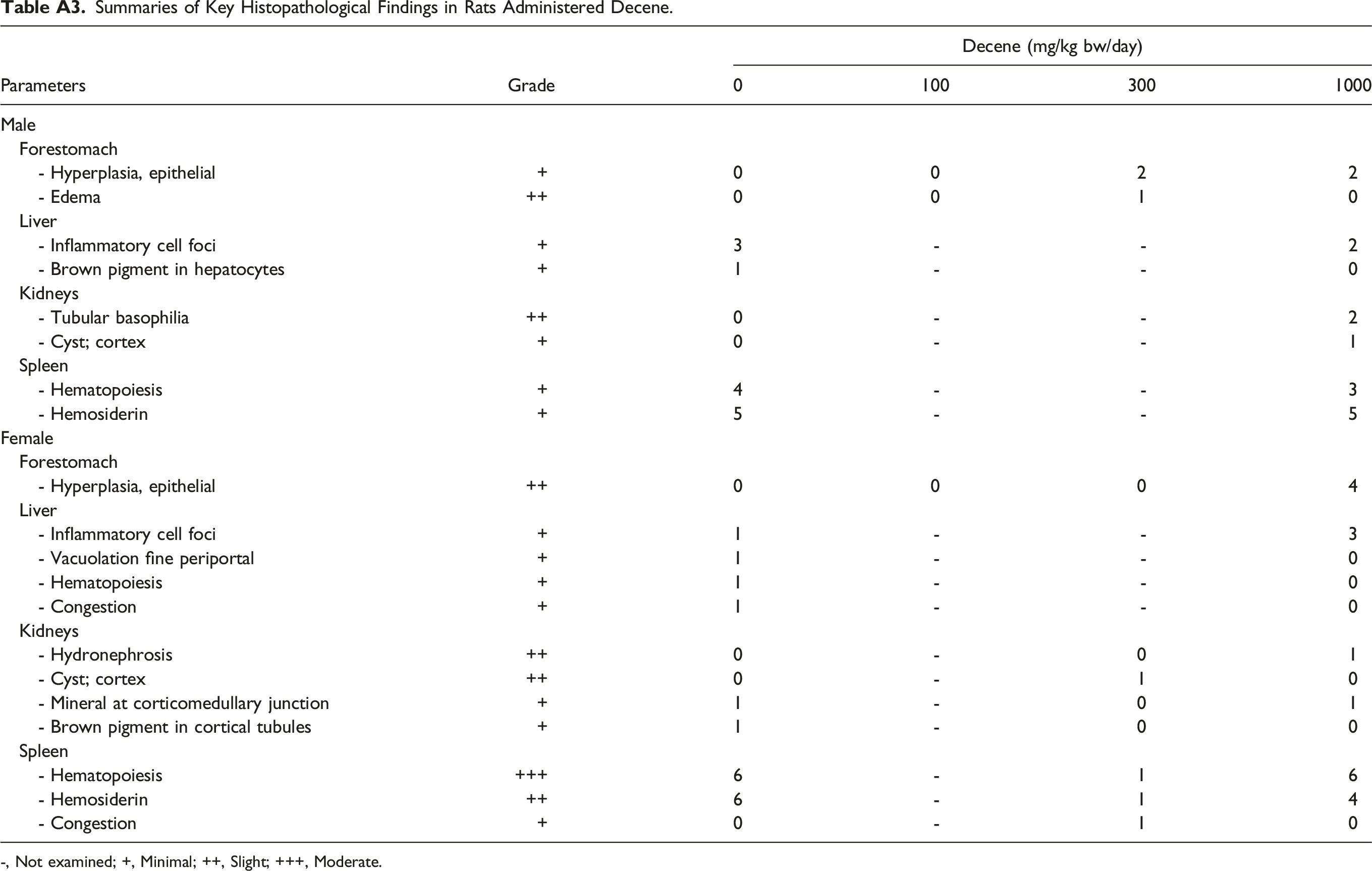
Parameters
Grade
Decene (mg/kg bw/day)
0
100
300
1000
Male
Forestomach
- Hyperplasia, epithelial
+
0
0
2
2
- Edema
++
0
0
1
0
Liver
- Inflammatory cell foci
+
3
-
-
2
- Brown pigment in hepatocytes
+
1
-
-
0
Kidneys
- Tubular basophilia
++
0
-
-
2
- Cyst; cortex
+
0
-
-
1
Spleen
- Hematopoiesis
+
4
-
-
3
- Hemosiderin
+
5
-
-
5
Female
Forestomach
- Hyperplasia, epithelial
++
0
0
0
4
Liver
- Inflammatory cell foci
+
1
-
-
3
- Vacuolation fine periportal
+
1
-
-
0
- Hematopoiesis
+
1
-
-
0
- Congestion
+
1
-
-
0
Kidneys
- Hydronephrosis
++
0
-
0
1
- Cyst; cortex
++
0
-
1
0
- Mineral at corticomedullary junction
+
1
-
0
1
- Brown pigment in cortical tubules
+
1
-
0
0
Spleen
- Hematopoiesis
+++
6
-
1
6
- Hemosiderin
++
6
-
1
4
- Congestion
+
0
-
1
0
Summaries of Key Histopathological Findings in Rats Administered Hexadecene. -, Not examined; +, Minimal; ++, Slight; +++, Moderate.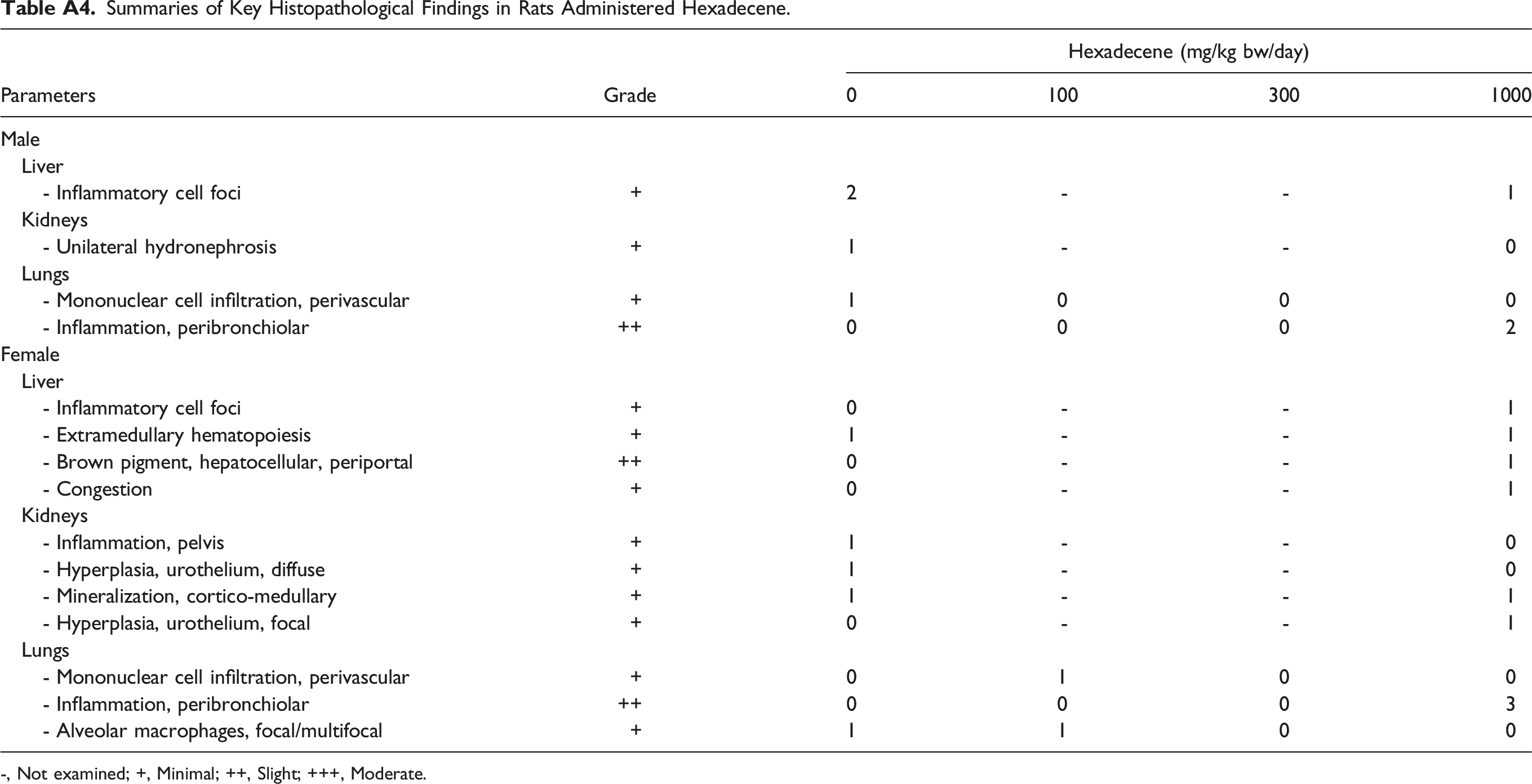
Parameters
Grade
Hexadecene (mg/kg bw/day)
0
100
300
1000
Male
Liver
- Inflammatory cell foci
+
2
-
-
1
Kidneys
- Unilateral hydronephrosis
+
1
-
-
0
Lungs
- Mononuclear cell infiltration, perivascular
+
1
0
0
0
- Inflammation, peribronchiolar
++
0
0
0
2
Female
Liver
- Inflammatory cell foci
+
0
-
-
1
- Extramedullary hematopoiesis
+
1
-
-
1
- Brown pigment, hepatocellular, periportal
++
0
-
-
1
- Congestion
+
0
-
-
1
Kidneys
- Inflammation, pelvis
+
1
-
-
0
- Hyperplasia, urothelium, diffuse
+
1
-
-
0
- Mineralization, cortico-medullary
+
1
-
-
1
- Hyperplasia, urothelium, focal
+
0
-
-
1
Lungs
- Mononuclear cell infiltration, perivascular
+
0
1
0
0
- Inflammation, peribronchiolar
++
0
0
0
3
- Alveolar macrophages, focal/multifocal
+
1
1
0
0
Summaries of Key Histopathological Findings in Rats Administered 1-Octadecene. -, Not examined; +, Minimal; ++, Slight; +++, Moderate.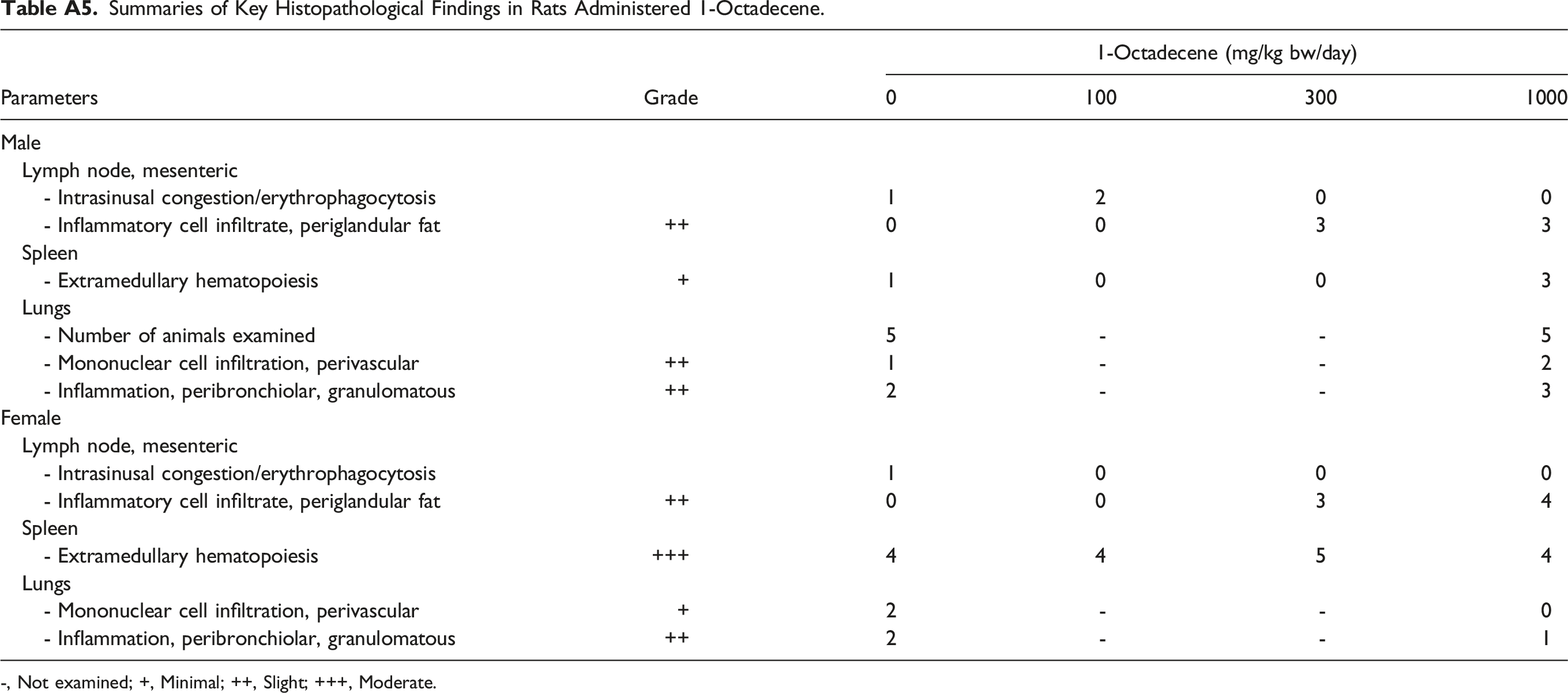
Parameters
Grade
1-Octadecene (mg/kg bw/day)
0
100
300
1000
Male
Lymph node, mesenteric
- Intrasinusal congestion/erythrophagocytosis
1
2
0
0
- Inflammatory cell infiltrate, periglandular fat
++
0
0
3
3
Spleen
- Extramedullary hematopoiesis
+
1
0
0
3
Lungs
- Number of animals examined
5
-
-
5
- Mononuclear cell infiltration, perivascular
++
1
-
-
2
- Inflammation, peribronchiolar, granulomatous
++
2
-
-
3
Female
Lymph node, mesenteric
- Intrasinusal congestion/erythrophagocytosis
1
0
0
0
- Inflammatory cell infiltrate, periglandular fat
++
0
0
3
4
Spleen
- Extramedullary hematopoiesis
+++
4
4
5
4
Lungs
- Mononuclear cell infiltration, perivascular
+
2
-
-
0
- Inflammation, peribronchiolar, granulomatous
++
2
-
-
1
Individual and Group Mean Litter Size, Litter Weights, and Viability Index at 1000 mg/kg bw/day Nonene. SD: standard deviation; N: number.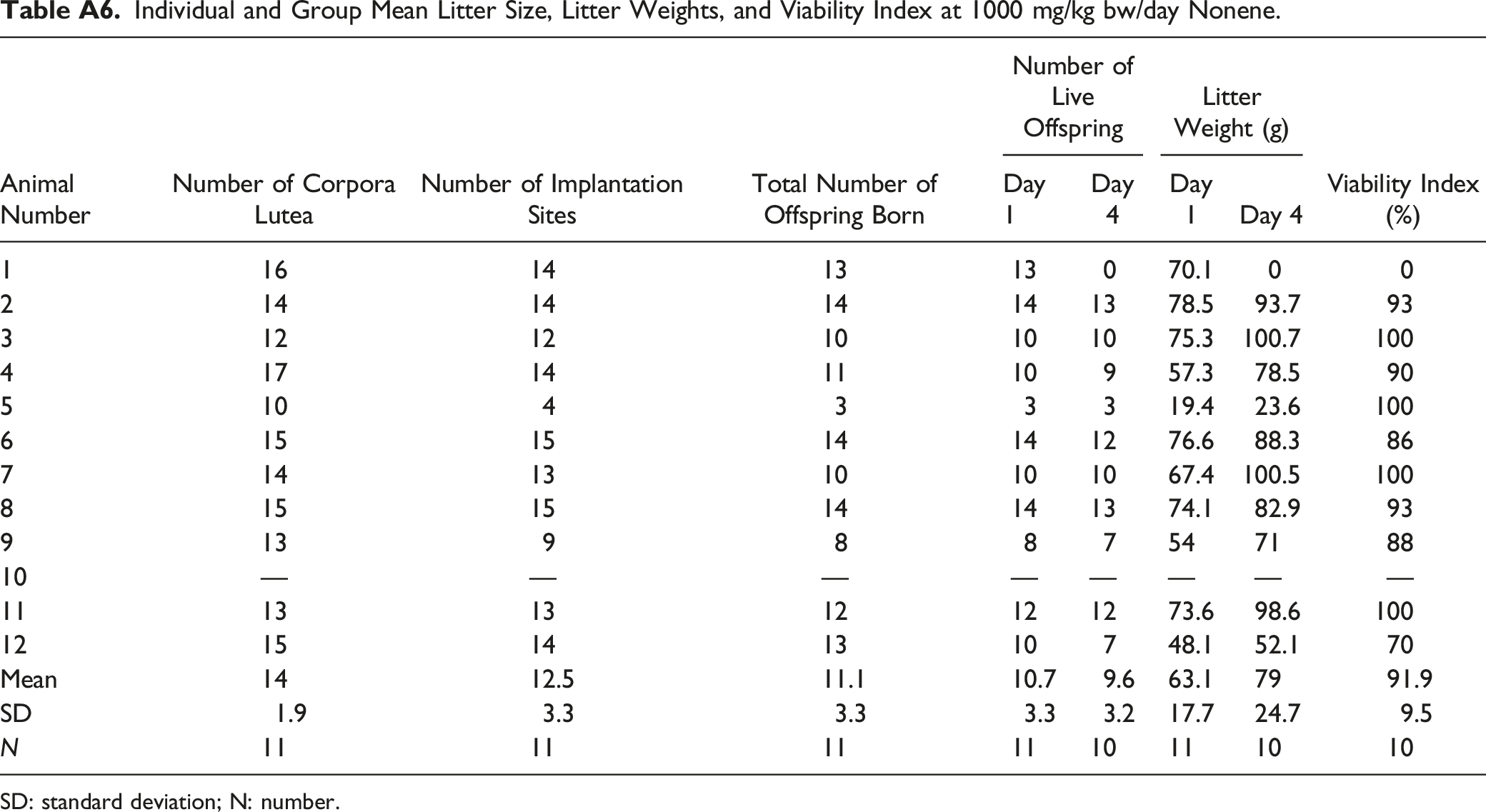
Animal Number
Number of Corpora Lutea
Number of Implantation Sites
Total Number of Offspring Born
Number of Live Offspring
Litter Weight (g)
Viability Index (%)
Day 1
Day 4
Day 1
Day 4
1
16
14
13
13
0
70.1
0
0
2
14
14
14
14
13
78.5
93.7
93
3
12
12
10
10
10
75.3
100.7
100
4
17
14
11
10
9
57.3
78.5
90
5
10
4
3
3
3
19.4
23.6
100
6
15
15
14
14
12
76.6
88.3
86
7
14
13
10
10
10
67.4
100.5
100
8
15
15
14
14
13
74.1
82.9
93
9
13
9
8
8
7
54
71
88
10
—
—
—
—
—
—
—
—
11
13
13
12
12
12
73.6
98.6
100
12
15
14
13
10
7
48.1
52.1
70
Mean
14
12.5
11.1
10.7
9.6
63.1
79
91.9
SD
1.9
3.3
3.3
3.3
3.2
17.7
24.7
9.5
N
11
11
11
11
10
11
10
10
Diagram of the experimental schedule, indicating the maximum study duration, based on a full 14-day mating period. Adopted from OECD Guideline for Testing of Chemicals No. 422: Combined Repeated Dose Toxicity Study with the Reproduction/Developmental Toxicity Screening Test 22.03.96. Group mean food consumption on both sex for 1-Octene, Nonene, Decene, Hexadecene, and 1-Octadecene.