Abstract
This study consisted of a 28-day oral repeat dose (repeat dose toxicity [RDT]) phase and a developmental and reproductive (developmental and reproductive toxicity [DART]) phase with rats. Rats were treated with Dechlorane Plus at doses of 0, 750, 1500, or 5000 mg/kg by gavage. For the RDT phase, no effects were observed on in-life parameters or clinical or anatomic pathology. In the DART phase, no effects were observed on reproductive or fertility indices, or fetal development through lactation day (LD) 4. No effects were noted on gestation day (GD) 20 implantation data, fetal indices, or external and visceral examinations. Mortalities occurred across all dose groups, although these were gavage-related errors and not compound related. Microscopic evidence of gavage-related errors included adhesions, inflammation, and fibrosis in the thoracic and pleural cavities. These findings were not test article related as they were observed only in animals with evidence of gavage injury. The no-observable-effect level (NOEL) in both phases of study was 5000 mg/kg.
Introduction
During the last 20 years, the use of flame retardants, particularly in children’s clothing, received a great deal of regulatory scrutiny resulting in a greater level of testing for human health effects, and also received environmental attention. The brominated flame retardants (BFRs) have use in a variety of consumer applications, including structural foams for furniture and in electronic products, for example, televisions. 1 Much of the testing of the BFR that was published concentrated on specific BFRs although there remains limited data on this class of chemicals. 2 Dechlorane Plus (CAS# 13560-89-9) was introduced as a flame retardant several years ago, but the published data for this compound also was limited in spite of the large volume of production. Because of the production volume of this compound, the US Environmental Protection Agency (EPA) listed Dechlorane Plus (DP) as a high production volume (HPV) chemical. None of the previous studies have been published in the peer-reviewed literature. (The results of these unpublished studies can be found at the EPA HPV Web site (http://www.epa.gov/chemrtk/pubs/hpvrstp.htm.)
The acute oral toxicity of DP was examined in male rats. In that study, rats were treated with a single oral dose of 10 to 3160 mg/kg as 1% or 10% suspensions in corn oil. 3 No mortality occurred at any doses, and no toxicologically significant gross pathology findings were noted. In a separate study conducted by Industrial Bio-Test Laboratories (IBT) in the mid-1970s, rats were treated with a single oral dose of DP at 1500, 3000, 6000, 12 500, or 25 000 mg/kg. In a 90-day toxicity study, male and female rats were administered DP in the diet at concentrations of 10 000, 30 000, or 100 000 ppm; the equivalent oral doses for males were approximately 522, 1567, and 5200 mg/kg, respectively, and for females 654, 1960, and 6500 mg/kg, respectively. Other than increased absolute and relative liver weights of rats treated with the high dietary concentration of DP, no other toxicologically significant finding was observed. 4 The acute and repeat dose toxicity studies suggested that little of the orally administered dose of DP was absorbed.
In subsequent metabolism studies, rats were administered a single oral dose of either 1 or 113 mg/kg radiolabeled DP by gavage to examine the absorption, tissue distribution, and elimination of the test article. 5 About 93% to 98% of the dose was excreted in feces unchanged within 4 days with less than 0.1% of the radiolabel excreted in urine and only 0.004% was excreted in expired air. Plasma levels peaked at 10 hours after dose administration and the radiolabel was at least 65% DP with the remaining suspected to be metabolites although these were not identified. Tissue residues did not increase proportionally with dose; a 113-fold increase in dose resulted in an 8-fold decrease in the percentage of the administered dose excreted in urine and remaining in the carcass. Within 4 days of dosing, only 26% of the radiolabel remaining in the carcass was DP. The ovaries and liver contained the highest levels of DP. These data further suggest that little DP is absorbed into systemic circulation.
The 90-day IBT study was criticized by the EPA, since it had been conducted prior to implementation of Good Laboratory Practice (GLP) regulations. 6 Hence, a 28-day repeat dose toxicity study (RDT) and a reproductive toxicity study were undertaken, consistent with Organization for Economic Cooperation and Development (OECD) Guideline 7 422. The 2 objectives of this study were (1) to evaluate the test article for possible adverse effects following repeated oral dosing over a 28-day period that included an evaluation of neurologic potential (repeat dose toxicity phase; RDT) and (2) to evaluate male and female reproductive performance (gonadal function, mating behavior, conception, development of the conceptus, parturition, and early neonatal development; developmental and reproductive toxicity phase; DART). The study was conducted according to GLP regulations (EPA and OECD), and the use of animals in this study was consistent with the guidelines and practices established by AAALAC International (Association for Assessment and Accreditation of Laboratory Animal Care).
Materials and Methods
Test Substance and Vehicle
Dechlorane Plus (99% purity) was supplied by Occidental Chemical Corporation (Niagara Falls, New York). Prior to study initiation, test batches of DP were prepared using multiple vehicles to determine the most appropriate for oral dosing. Based on this assessment, corn oil was used as the vehicle in this study with formulations of the test article prepared weekly at nominal concentrations of 75, 150, and 500 mg/mL. The suspensions (white in color) were stored refrigerated when not being administered. Samples of the suspensions were collected periodically during the study to verify concentration levels. Homogeneity and stability analyses of the dosing formulations were also assessed. Based on those analyses, the measured concentrations were within about 12% of the target concentration, indicating that the prepared suspensions were acceptable and stable (data not shown).
Animals and Dose Administration
A total of 135 male and 135 female experimentally naive CD (Crl:CD[SD]) rats (approximately 6-8 weeks of age) were obtained from Charles River Laboratories (Portage, Michigan, and Raleigh, North Carolina). During the 14-day acclimation period, the animals were observed daily with respect to general health and any signs of disease, and body weights were taken. Based on the pretest body weights, the animals were randomized to study groups as shown in Table 1 .
Group Assignment of Rats Treated With Dechlorane Plus
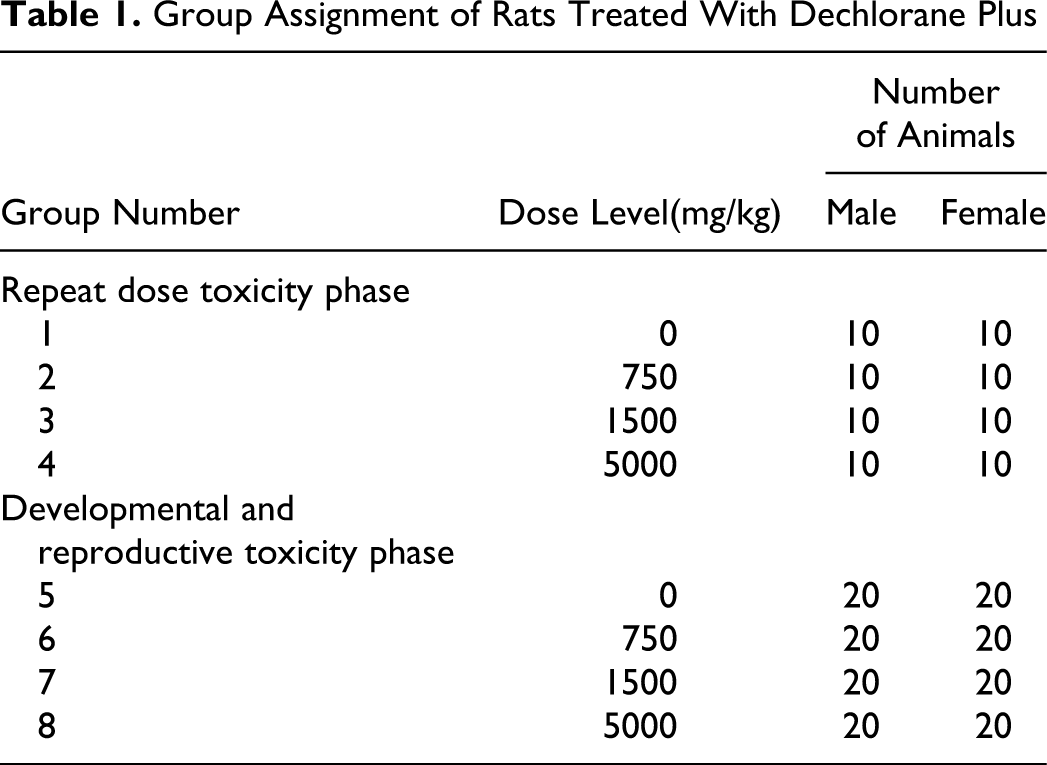
For the animals in the DART phase, 10 females/group were used for evaluation of developmental toxicity with surviving animals used for the reproductive toxicity phase. The animals were individually housed in suspended, stainless steel, wire mesh type cages, except during pairing, near parturition, and during lactation in an environmentally controlled room. During pairing, each female was housed in the cage of a male at a ratio of 1:1. Gestation day (GD) 0 is the day when evidence of mating (vaginal plug and/or sperm in the vaginal rinse) was confirmed. On approximately GD 20, females selected for parturition were individually housed in plastic solid bottom cages containing wood chip bedding. Females were housed in these solid bottom cages with the pups until euthanasia (lactation day [LD] 4).
All doses were administered via oral gavage at a dose volume of 10 mL/kg. In the RDT phase, the vehicle and test article were administered once daily for 28 consecutive days. In the DART phase, the vehicle and test article were administered for at least 63 consecutive days to the males (21 days premating, 14 day mating period, and 28 days after completion of the mating period). Females were treated for up to 64 days dependent on reproductive performance and gestation length (21 days premating, 14 days of mating, and up to 25 days after mating [GD 0 through LD 3]). A magnetic stir bar on a stir plate was used for mixing the formulations throughout test article administration. The dosing formulation at 5000 mg/kg (500 mg/mL), which was particularly viscous, was inverted several times periodically during dosing to ensure complete mixing of the suspension. Individual doses were based on the most recent body weights.
Fluorescent lighting was provided for approximately 12 hours/d. Temperature and humidity were continuously monitored and recorded and ranged from 64 to 79°F and 30% to 70%, respectively. Meal Lab Diet (Certified Rodent diet #5002 PMI Nutrition International, Inc and tap water were available ad libitum, except when animals were fasted for clinical pathology evaluations.
In-life Examinations
All animals were observed for morbidity, mortality, injury, and the availability of food and water twice daily throughout the duration of the study. Food consumption was measured and recorded weekly during the study. Body weights for all animals in the RDT phase were measured periodically with food consumption recorded weekly. For rats in the DART phase, body weights were recorded on day 1 and at 3- and 4-day intervals during the premating period. Males and unmated females were weighed at 3- to 4-day intervals thereafter and mated females were weighed on GDs 0, 3, 7, 10, 14, 17, and 20. Females allowed to deliver and retain a litter to LD 4 were also weighed on LDs 0 and 4. Food consumption was recorded weekly during the premating period and mated females had food consumption recorded at corresponding intervals with body weights.
A detailed clinical examination of each animal designated to the RDT phase was performed daily, approximately 60 minutes post dose and included evaluation of the skin, fur, eyes, ears, nose, oral cavity, thorax, abdomen, external genitalia, limbs and feet, respiratory and circulatory effects, autonomic effects such as salivation, and nervous system effects including tremors, convulsions, reactivity to handling, and abnormal behavior. In addition, functional observational battery (FOB) evaluations were conducted without knowledge on the part of the testers of the treatment groups. Functional observational battery evaluations, including those conducted in the home cage, during handling, in the open field, and others, were conducted on 5 randomly selected animals/gender/group at pretest and during week 4. The animals were given at least 20 minutes to acclimate to the testing room. During open-field evaluations, each animal was observed for a minimum of 3 minutes in an observation box measuring 20 × 20 × 8 inches. The parameters evaluated in the FOB were based on those outlined in Moser et al. 8,9 The observations included evaluation of activity and arousal, posture, rearing, abnormal behavior, clonic and tonic movements, gait, mobility, stereotypy, righting reflex, response to stimulus (approach, click, tail pinch, and touch), palpebral closure, pupil response, piloerection, exophthalmos, lacrimation, salivation, and respiration. Qualitative and/or quantitative measures of defecation and urination were also recorded. Forelimb and hind limb grip strength was measured using the procedure described by Meyer et al 10 and hind limb splay was quantitatively measured as described by Edwards and Parker. 11 Pain perception was assessed by measuring the latency of response to a nociceptive (thermal) stimulus when each animal was placed on a hot plate apparatus set to 52°C as described by Ankier. 12
Clinical Pathology
For rats in the RDT phase, clinical pathology evaluations (hematology, coagulation, and clinical chemistry) were conducted on the first 5 animals/gender/group euthanized and necropsied at termination of the 28-day phase. The animals had access to drinking water but were fasted overnight prior to sample collection.
Hematology measures consisted of white blood cell count (total and differential), red blood cell count, hemoglobin, hematocrit, mean corpuscular hemoglobin (MCH), mean corpuscular volume (MCV), MCH concentration (MCHC; calculated), reticulocyte count (absolute and relative), and platelet count. Prothrombin and activated partial thromboplastin time also were measured. Clinical chemistry measures consisted of alkaline phosphatase (ALP), total bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyl transferase (GGT), sorbitol dehydrogenase (SDH), urea nitrogen, creatinine, total protein, albumin, globulin, and A/G (albumin/globulin) ratio (calculated), glucose, total cholesterol, sodium, potassium, chloride, calcium, and phosphorus.
Developmental and Reproductive Toxicity Phase Evaluations
At initiation of test article administration and until evidence of copulation was observed or the cohabitation period ended, females were examined daily by vaginal lavage to establish estrous cyclicity. After 3 weeks of treatment, each female was housed in the cage of a male from the same treatment group. Positive evidence of copulation was established by daily inspection for a copulatory plug in the vagina and/or a vaginal lavage for the presence of sperm. The day on which positive evidence of copulation was observed was considered GD 0. After evidence of mating was observed, the female was returned to an individual cage for the remainder of the study. The maximum pairing period was 14 days, at the end of which any females with no confirmed evidence of mating were returned to individual cages until study termination.
Toward the end of the gestation period, selected mated females were examined twice daily for signs of parturition. The mated females were allowed to give birth (F1). The duration of gestation was calculated, and any abnormalities occurring at parturition were recorded. The day on which all pups were delivered was designated as LD 0. The litters were examined as soon as possible after delivery. Litter size, number of stillborn and live born pups, number of males and females, individual body weights, and gross abnormalities of the pups were recorded for each litter. Individual pup body weights and pup external examinations were recorded on LD 0 and LD 4. Any abnormal behavior observed in the pups was recorded daily. On LD 4, the dams and pups were euthanized and necropsied.
Pathological Evaluations
Repeat dose toxicity phase
Necropsy examinations were performed on animals found dead and on all surviving animals at the scheduled necropsy. The animals were euthanized by carbon dioxide inhalation. The animals were examined carefully for external abnormalities. Selected organs were weighed. The abdominal, thoracic, and cranial cavities were examined for abnormalities and the organs removed, examined, and placed in fixative. The pituitary was fixed in situ. Tissues were fixed in neutral buffered formalin, except for the testes, which were fixed using a modified Davidson fixative. Formalin was infused into the lung via the trachea and into the urinary bladder. Tissues were stained with hematoxylin and eosin and examined microscopically from 5 animals/gender in the control and high-dose groups
Developmental and reproductive toxicity phase
On GD 20, randomly selected animals/group at 0, 750, 1500, and 5000 mg/kg, respectively, were euthanized by carbon dioxide inhalation, followed by exsanguination via the abdominal vena cava, and immediately subject to a cesarean section. The skin was reflected from a ventral midline incision to examine mammary tissue and locate any subcutaneous masses. The abdominal cavity was opened and the uterus was exposed. Beginning at the distal end of the left uterine horn, the location of viable and nonviable fetuses, early and late resorptions for each uterine horn, position of the cervix, and the number of corpora lutea on each ovary were recorded. Viable fetuses responded to touch. In contrast, nonviable fetuses did not respond to touch and did not show signs of autolysis. Late resorptions had recognizable fetal form but were undergoing autolysis, whereas early resorptions were characterized as implantation sites with no recognizable fetal characteristics. The uterus was excised and a gravid uterine weight was recorded. The fetuses were removed by making a dorsal incision longitudinally along both uterine horns. The embryonic membrane of each fetus was gently removed, and each fetus was pulled away from the placenta, fully extending the umbilical cord. The placentae were grossly examined. Uteri from females that appeared nongravid were opened and placed in 10% ammonium sulfide solution for detection of implantation sites. 13 If no foci were seen, the female was considered nonpregnant. A necropsy was conducted for each female
Each fetus was individually weighed, sexed, tagged, and examined for external malformations and developmental variations. Each fetus was euthanized by intraperitoneal injection of sodium pentobarbital solution. Approximately one half of the fetuses in each litter were decapitated and the head section placed in Bouin solution. All fetuses were processed for visceral examination using a microdissection procedure. Heads were processed for evaluation using a razor-blade sectioning technique. Following the microdissection evaluations, the viscera from each fetus was removed and the carcasses preserved in 95% ethanol for possible further evaluation of the skeletal structures. Fetal findings were classified as malformations or developmental variations under criteria approved by a developmental toxicologist, unless otherwise specified.
At the termination of the study, all surviving DART animals (males and unmated females) were euthanized by carbon dioxide inhalation followed by exsanguination. The animals were examined carefully for external abnormalities, including palpable masses. The abdominal, thoracic, and cranial cavities were examined for abnormalities. Special emphasis was placed on organs of the reproductive system. Implantation sites were counted and recorded for females in the parturition phase of study. In males, the epididymides and testes were weighed.
On LD 4, pups were euthanized and externally examined, and any abnormalities were recorded and the carcasses were then discarded. Dead pups found at birth or during LD 0 to LD 4 and pups euthanized in extremis were examined internally for irregularities, and only pups with gross lesions or malformations were saved (10% neutral buffered formalin) for possible further evaluation.
Statistics
Data for each sex within a set were analyzed separately. The raw data were tabulated within each time interval, and the mean and standard deviation were calculated for each end point by sex and group. Data for some end points, as indicated, were transformed by a log transformation prior to conducting the specified analysis. Body weights, food consumption, clinical pathology, FOB parameters, and organ weight data were statistically analyzed by pairwise comparisons or Levene/analysis of variance (ANOVA)−Dunnett/Welch test. For these end points, the treated animal was the experimental unit.
For DART end points, data were analyzed by pairwise comparison tests. Fetal sex ratios, preimplantation and postimplantation losses were transformed by arcsin square root transformations and then statistically compared by a pairwise comparison test. Fertility data were statistically analyzed by pairwise comparisons, covariate analysis, or Fisher exact test. Litter size was used as a covariate. Fetal malformation and developmental variation data were analyzed by Fisher exact test with the litter as the experimental unit.
For pairwise comparisons, if sample sizes for all groups were 3 or more, Levene test was used to assess homogeneity of group variances for each specified end point and for all collection intervals. 14 If Levene test was not significant (P > 0.01), a pooled estimate of the variance (mean square error) was computed from a 1-way ANOVA and used by a Dunnett comparison of each treatment group with the control group. If Levene test was significant (P < 0.01), comparisons with the control group were made using Welch t test with a Bonferroni correction. In the case that sample size was less than 3 for at least 1 treatment group, Levene method could not be implemented. Groups with sample sizes less than 3 were excluded from the analysis and control−treatment pairwise comparisons that satisfied the sample size assumption (n > 3) were conducted using Welch t test with a Bonferroni correction. If there were only 2 groups involved, the above methodology applied and the Dunnett’s test reduced to a Student t test. Results of all pairwise comparisons are reported at the 0.05 and 0.01 significance levels. All end points were analyzed using 2-tailed tests.
Results
During the study, 15 mortalities occurred across all groups for both the RDT and DART phases (Table 2 ).
Total Mortality of Rats Treated With Dechlorane Plus
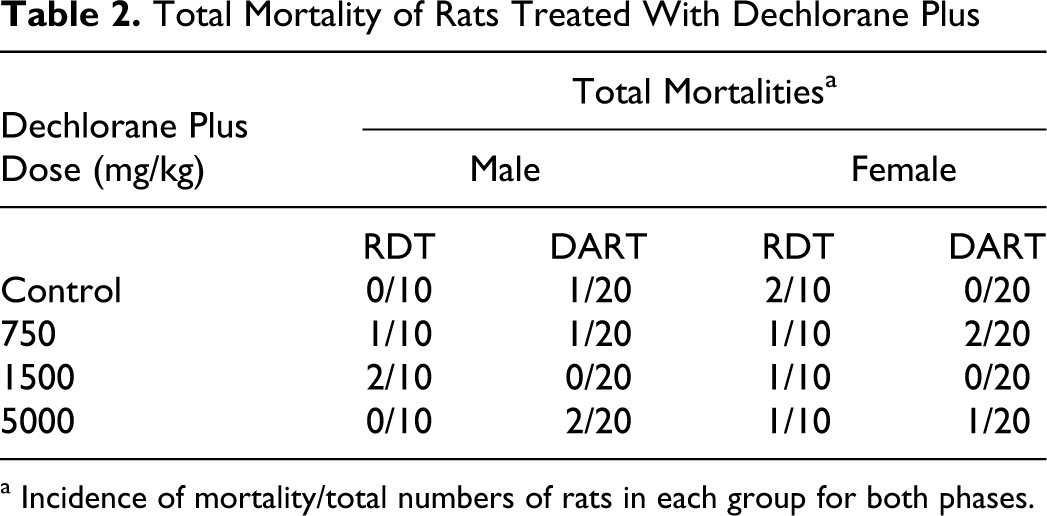
a Incidence of mortality/total numbers of rats in each group for both phases.
One control female designated to the RDT phase and 2 rats designated to the DART phase (1 control male and one 750 mg/kg male) had no macroscopic lesions to indicate a possible cause of death. However, all other animals that died on study had findings indicative of gavage injury with deposition of the test article in the thoracic and/or pericardial cavity. This may have been directly related to a mis-dose into the lungs/thoracic cavity due to the viscosity of the test article or from aspiration into the lungs of the viscous test article from the buccal cavity. Clinical signs of toxicity were evident consistent with the probable mis-dosing and included breathing irregularities (ie, breathing audible, difficult, rapid) and/or decreased activity. In most instances, these same animals also had macroscopic lesions involving adhesions of the tissues in the thoracic cavity as described below.
Macroscopic changes were observed in the thoracic cavity, heart, mediastinum, lung, mediastinal lymph node, and/or thymus of all those animals considered to have been mis-dosed, that died during the study or survived to termination (Table 3 ). Changes indicative of suspected gavage injury included adhesions and white fluid in the thoracic cavity; inflammation and fibrosis and white foreign material in the pericardial sac and mediastinum; and inflammatory cells and fibrosis in the mediastinal lymph node. The lungs contained multiple white foci and/or a white mass, consistent microscopically with adhesions, inflammation, and fibrosis, or lung lobes had black discoloration. The thymus gland contained white foci or had white discoloration, consistent microscopically with mixed inflammation and fibrosis. Occasionally the thymus gland was not identified due to incorporation into a thickened mediastinum. Macroscopic changes attributed to suspected gavage injury in animals dying prior to scheduled study termination, but not observed in animals at terminal necropsy, included an abscess in the parietal pericardium; red fluid in the thoracic cavity, consistent with a blood clot; and foreign material consistent with the test article in the subcutis of the left axillary region. Three animals also had laceration or perforation in the trachea or esophagus.
Incidence of Gross Necropsy Findings in Rats Treated With Dechlorane Plus a
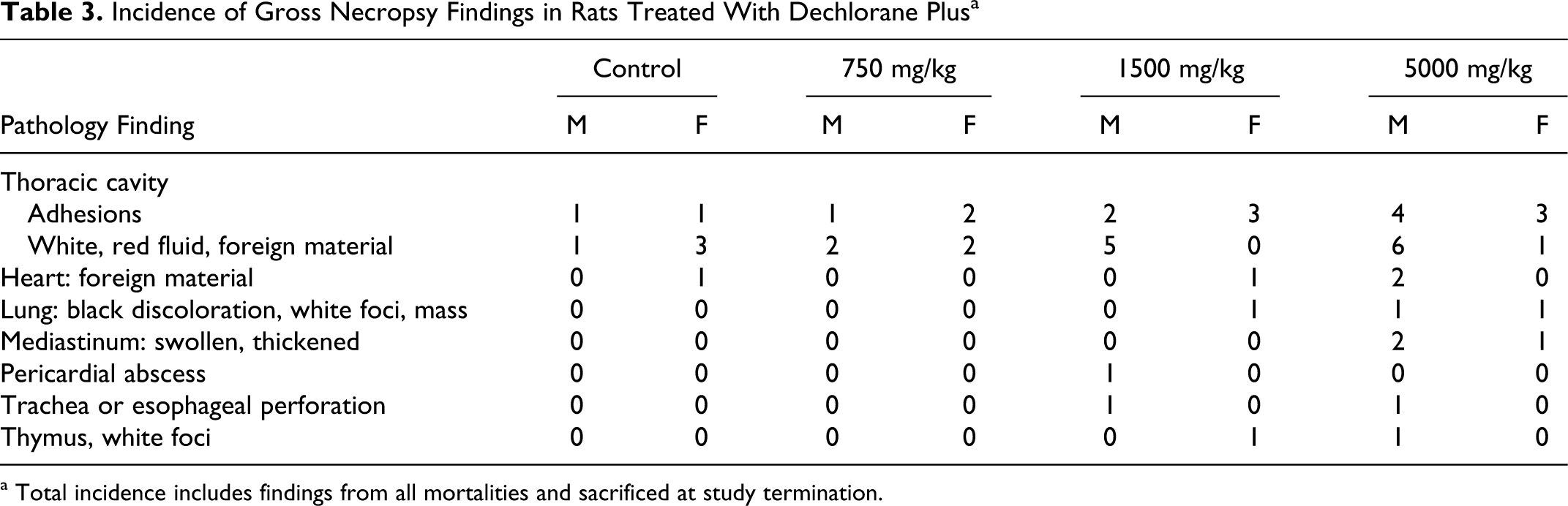
a Total incidence includes findings from all mortalities and sacrificed at study termination.
In animals that died prior to study termination, there was microscopic evidence of suspected gavage injury observed that were not observed in animals at terminal necropsy (Tables 4 and 5 ). In those animals that died prior to study termination, foreign material was observed in the lung suggestive of aspiration, which included plant material derived from diet and rare blue granular to globular material, which may have represented test article material. In addition, other findings observed consisted of acute to subacute inflammation of the lung or lung abscess, hypertrophy or hyperplasia of the mesothelial lining of the lung and heart, and subacute to chronic inflammation of the mediastinum with mediastinal fibrosis.
Microscopic Findings in Rats That Died Prior to Scheduled Necropsy: Repeat Dose Toxicity Phase a
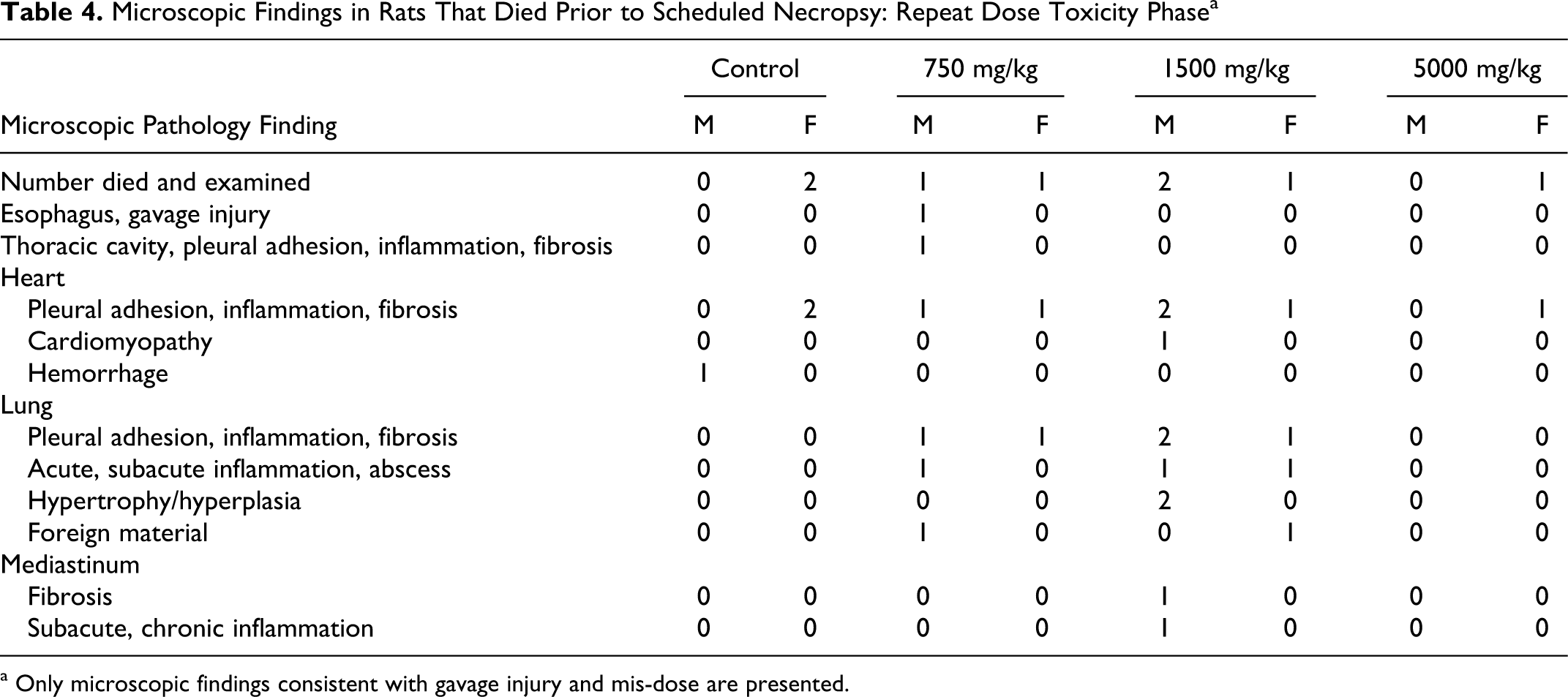
a Only microscopic findings consistent with gavage injury and mis-dose are presented.
Microscopic Findings in Rats That Survived to Scheduled Necropsy: Repeat Dose Toxicity Phase a
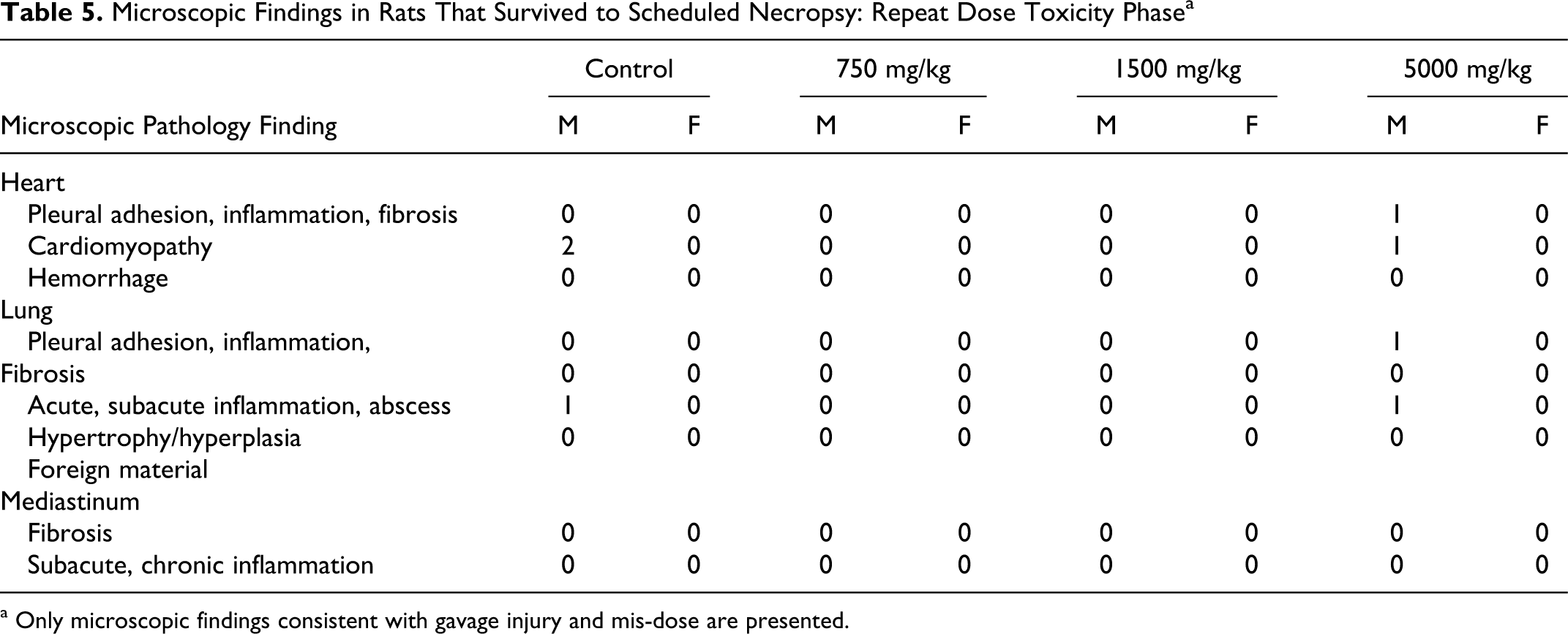
a Only microscopic findings consistent with gavage injury and mis-dose are presented.
There were additional microscopic findings suspected to be associated with an antigenic stimulus, immune response, or a physiological stress response secondary to the presence of the test article material in the thoracic cavity that were found in animals that died prior to scheduled study termination, but not in animals that survived to study termination. Those findings included Kupffer cell hypertrophy in the liver; increased mitotic figures in the liver; generalized and paracortical mandibular, mediastinal, and mesenteric lymph node depletion; minimal cortical lymphoid necrosis in the mandibular lymph node; sinus erythrocytosis and erythrophagocytosis in the mesenteric lymph node; generalized Peyer patch lymphoid depletion; and generalized lymphoid depletion in the spleen. These findings were consistent with an expected physiological response to trauma and foreign material. They were not considered test article related as they were not dose dependent and were observed in animals with evidence of suspected gavage injury.
The test article as formulated in corn oil was quite viscous, which resulted in difficulty of administration. Viscosity measurements were conducted after study initiation for the concentrations of DP in corn oil used in this study. The results of those analyses revealed that corn oil had a viscosity of 60 cps. For the 75, 150, and 500 mg/mL formulations, the viscosity measures were 70, 90 cps, and 5000 to 9000 cps, respectively. Clearly, particularly at the high concentration, the viscosity of the test article suspensions most likely contributed to the mis-dosing and mortalities observed in this study. In addition, it was retrospectively recognized that because of the high viscosity, the rate at which the technical staff administered the suspensions also may have contributed to the mortality.
Repeat Dose Toxicity Phase
No effect of treatment with DP was evident from FOB evaluations that included observations in the various categories of assessment (activity/arousal, autonomic, neuromuscular, physiological, and sensimotor). The only statistically significant difference seen at the week 4 evaluation was a lower frequency of urination in comparison to controls in the 750 and 5000 mg/kg females. In both groups, the mean frequency in urination was 0.2 in comparison to a mean of 2.0 in controls. The mean incidence in the 1500 mg/kg females was 0.4. In the absence of a dose response, this difference in urination count for females, while statistically significant, was not considered toxicologically meaningful. In males at week 4, mean urinations over the observation period in the treated groups ranged from 0.4 to 2.2 and did not differ statistically from the zero frequency in controls.
No effects were evident in the treated animals on motor activity, which distinguished basic movements, fine movements, rearing counts, and total distance. The one exception was a statistically significant increase in rearing counts for the 1500 mg/kg males during the 20- to 30-minute trial period. In the absence of other changes on motor activity as well as other behavioral observations, and in the absence of a similar response in the 5000 mg/kg males, this change was considered spurious and unrelated to treatment.
No effect of treatment with DP was evident from the weekly neurobehavioral observations. No tremors, convulsions, abnormal behavior, or change in gait were seen among the control or treated animals over the 4-week period. Some variability was seen among the groups week-to-week in occurrences of stereotypy, movement, grip response, and motor activity grid results, but no effect of treatment was evident (data not shown).
Mean body weights of male rats during the treatment period in the 750 and 1500 mg/kg groups were comparable to controls (Figure 1 ). At 5000 mg/kg, mean body weights were slightly lower but not statistically significantly different from controls through much of the treatment period. Considerable variability in body weights was seen among the 5000 mg/kg males, and this was largely attributed to data for 3 animals with compromised health status due to gavage errors and/or aspiration of the test article based on macroscopic lesions within the thoracic cavity. Exclusive of data for these 3 males, the mean male weights at days 1 and 25 in the 5000 mg/kg group were 329.6 and 427.3 g, respectively, and similar to the 327.6 and 438.4 g, respectively, in controls. In females, mean body weights for the treated groups were comparable to controls. No effects on food consumption were observed in any dose groups (data not shown).
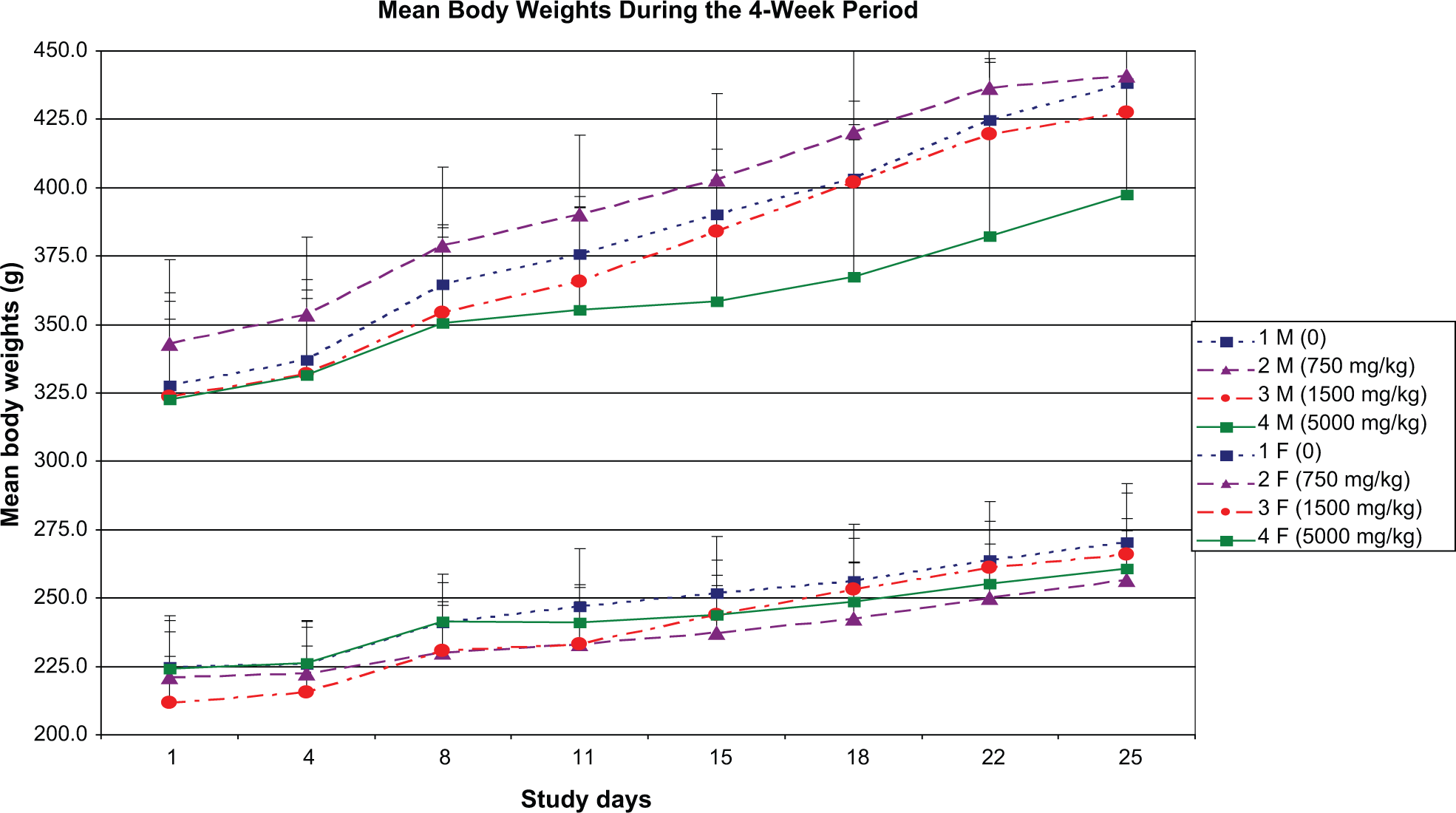
Mean (± standard deviation) body weights of male and female rats (n = 10/sex/group) designated to the repeat dose toxicity phase treated orally for 28 days with Dechlorane Plus
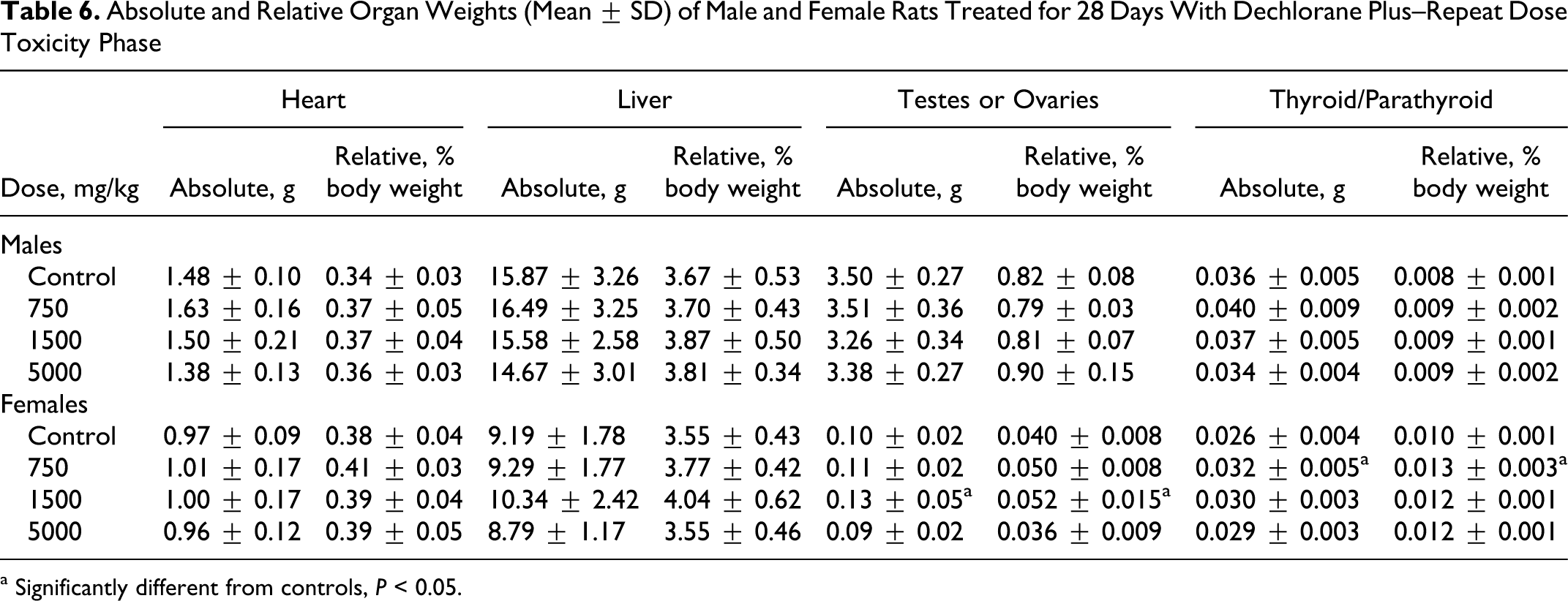
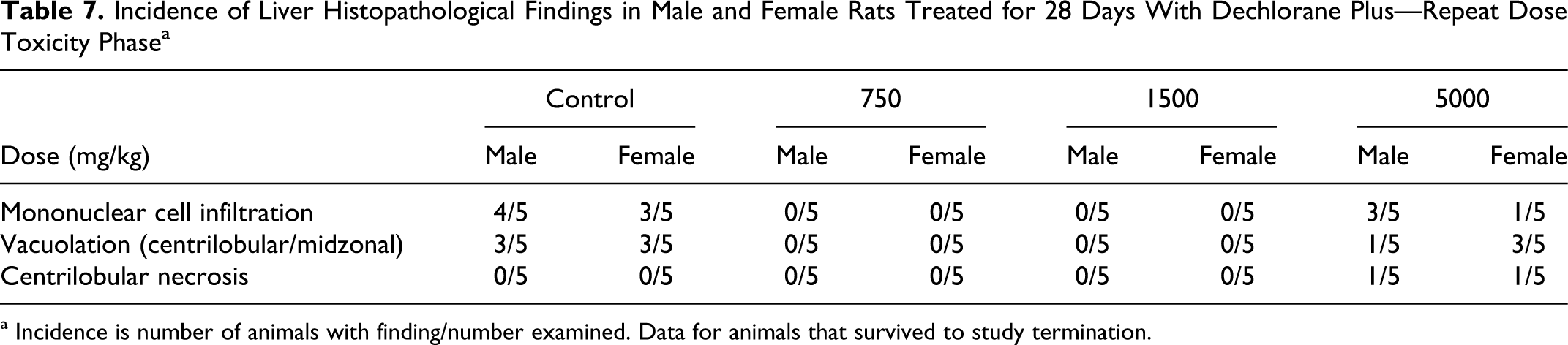
No test article-related effects on hematology, coagulation, or clinical chemistry parameters were observed in this study. No statistically significant organ weight changes were observed in males at any dose level (Table 6 ). In females, the absolute and relative (percentage body weight) ovarian weights were significantly increased at 1500 mg/kg (mean absolute increased 38.9%, mean relative to body weight increased 38.4%, and mean relative to brain weight increased 37.5%) but not at 5000 mg/kg. Mean absolute and relative (to body and brain weight) thyroid/parathyroid gland weights were statistically significantly increased in females at 750 mg/kg (mean absolute increased 21.8%, mean relative to body weight increased 30.7%, and mean relative to brain weight increased 24.6%) but not at 1500 mg/kg or 5000 mg/kg. Ovary and thyroid/parathyroid weight changes were not considered test article related as they were not dose related, and there were no correlating microscopic changes. Liver and heart weights were not different compared to controls but are included here based on macroscopic and microscopic changes noted in the heart, and the liver was considered a target organ in the previous study with DP. 4 However, no toxicologically significant liver histopathological changes were noted in the treated rats (Table 7 ). Microscopic findings in the heart (Table 5) were considered related to the gavage errors. No other organ weight changes that were considered of toxicological significance were observed in this study.
Absolute and Relative Organ Weights (Mean ± SD) of Male and Female Rats Treated for 28 Days With Dechlorane Plus–Repeat Dose Toxicity Phase
a Significantly different from controls, P < 0.05.
Incidence of Liver Histopathological Findings in Male and Female Rats Treated for 28 Days With Dechlorane Plus—Repeat Dose Toxicity Phase a
a Incidence is number of animals with finding/number examined. Data for animals that survived to study termination.
There were no microscopic test article-related toxicity effects in either sex at any dose level. Any changes in heart weights on an individual animal basis were considered related to the adhesions and other effects noted macroscopically and microscopically secondary to the gavage errors described above (Tables 3 and 4). Similarly, in the liver, vacuolation was noted in the control and high-dose animals, suggesting that this finding was related to the corn oil vehicle. Most microscopic findings observed in animals at terminal necropsy and animals dying prior to study termination were associated with suspected gavage injury. Other microscopic findings were considered incidental changes observed in animals of this age and breed. Microscopic changes suspected to be associated with an antigenic stimulus, immune response, and/or a physiological stress response secondary to the presence of suspected test article in the thoracic cavity included lymphocyte/plasmacyte hyperplasia in the mediastinal and mandibular lymph nodes, lymphoid depletion in marginal zones of the spleen, and generalized lymphoid depletion of the thymus gland. Findings were consistent with an expected physiological response to trauma and foreign material and not considered test article related.
Developmental and Reproductive Toxicity Phase
In rats designated to the DART phase, a yellow discoloration of the feces was seen among the treated animals but the highest prevalence was in the 5000 mg/kg group. This was also seen in a few animals in the RDT phase but apparently became more prevalent in these animals with the longer duration of treatment. The yellow discoloration of the feces was attributed to the presence of white test article in the presence of brownish gastrointestinal contents and food, and suggested fecal elimination of the test article with minimal absorption. Other findings seen with slightly higher incidence in the 1500 and 5000 mg/kg males in comparison to controls included salivation, feces few/absent, and red material around the nose or mouth, but in the absence of similar increases among the females, these were considered of little toxicological significance. Similar to rats in the RDT phase of study, there were a few animals in the control and treated groups with breathing irregularities (ie, breathing audible, difficult, rapid) and/or decreased activity. In most instances, these same animals also had macroscopic lesions involving adhesions of the tissues in the thoracic cavity, which were attributable to dosing or aspiration of the vehicle or test article. No effect of treatment with DP was evident on body weights or body weight gains in the males or females. No effect of treatment with DP was evident on food consumption during the premating, gestation, or postmating periods.
No effect of treatment with DP was evident from estrous cyclicity data over the premating period (Table 8 ). Mean cycle length and the mean number of estrous cycles over the 3-week recording interval for the treated groups were comparable to controls.
Estrous Cyclicity Data for Rats Treated With Dechlorane Plus for the Premating Period—DART Phase

No effect of treatment with DP was evident from reproductive and fertility indices (Table 9 ). Mating indices in the treated groups ranged from 95% to 100% and were comparable to the 95% in controls. Fertility indices in the treated groups ranged from 90.0% to 94.7% and were higher than controls and, similarly, fecundity indices in the treated group (94.4%-94.7%) were comparable to control (84.2%). Mean copulatory interval, that is days to mating, in the treated groups ranged from 2.5 to 3.8 days and was comparable to the 3.1 days in controls.
Reproductive and Fertility Measures (Mean ± SD) in Rats Treated With Dechlorane Plus
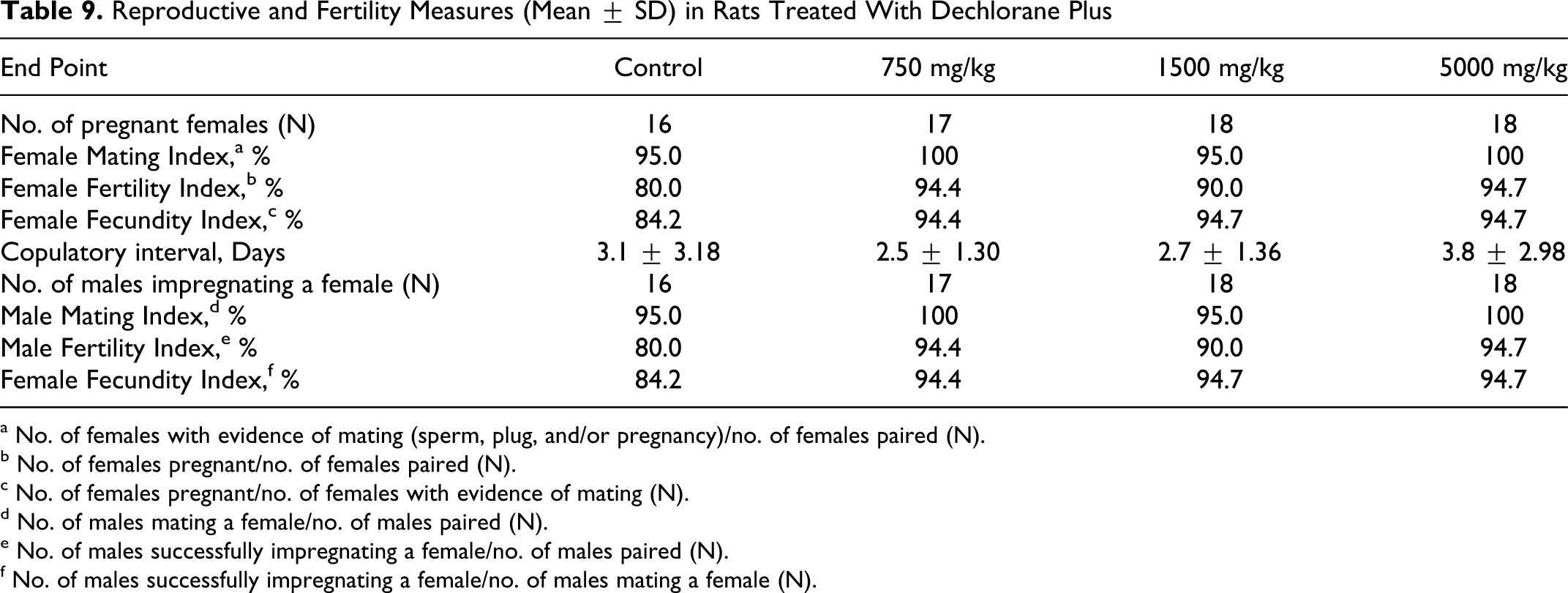
a No. of females with evidence of mating (sperm, plug, and/or pregnancy)/no. of females paired (N).
b No. of females pregnant/no. of females paired (N).
c No. of females pregnant/no. of females with evidence of mating (N).
d No. of males mating a female/no. of males paired (N).
e No. of males successfully impregnating a female/no. of males paired (N).
f No. of males successfully impregnating a female/no. of males mating a female (N).
A total of 10 females in each of the control, 1500, and 5000 mg/kg groups and 9 in the 750 mg/kg group were designated for GD 20 maternal cesarean-section evaluations. Pregnancy rates were 80%, 100%, 100% and 90% in the control, 750, 1500, and 5000 mg/kg groups, respectively, providing 8, 9, 10, and 9 GD 20 in utero litters, respectively, with viable fetuses for evaluation (Table 10 ). No effect of treatment with DP was evident from uterine implantation data. The mean numbers of corpora lutea, uterine implantations, viable fetuses, and resorption (early, late, and total) in the treated groups were comparable to controls. Mean pre- and postimplantation loss indices in the treated groups were also comparable to controls. Slight increases in the mean number of early resorption sites (1.8) and postimplantation loss index (13.90%) in the 1500 mg/kg group in comparison to controls (0.8% and 5.39%, respectively) were not statistically significant, and in the absence of a similar increase at 5000 mg/kg (0.8% and 5.62%, respectively) was not considered related to treatment with DP. Gravid uterine weights, adjusted GD 20 body weights, and adjusted body weight gain GDs 0 to 20 for the treated groups also were comparable to controls. No effect of treatment with DP was evident on fetal gender ratios (data not reported).
Maternal and Developmental Observations (Mean ± SD)—GD 20 Uterine Examination in Rats Treated With Dechlorane Plus
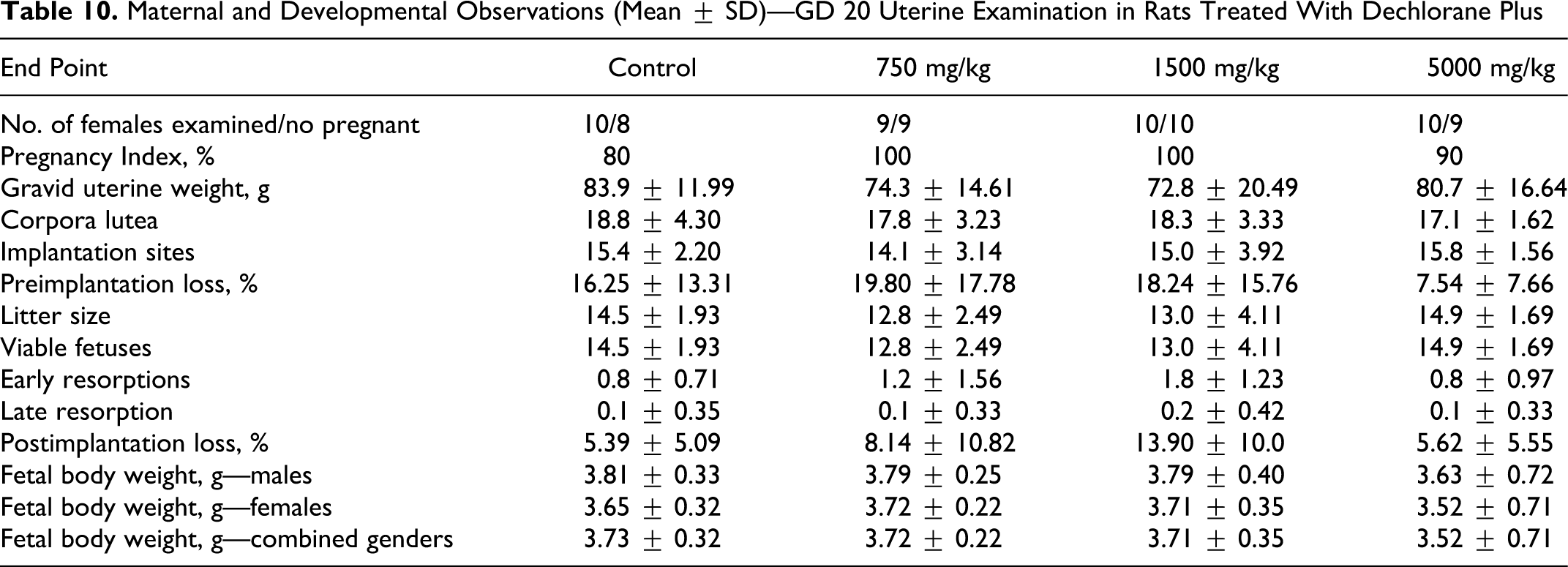
Mean fetal weights, distinguished by gender and for both genders combined, in the treated groups (Table 10) did not differ statistically from controls. In the 5000 mg/kg group, mean fetal weights were about 5% lower than controls, and this was attributed to extremely small fetuses within the litter of 1 female at 5000 mg/kg (average fetal litter weight 1.72 g). This female had macroscopic lesions involving adhesions of the thoracic cavity viscera and pockets of abscess, and the compromised health status of this animal was considered to have adversely affected the growth of the fetuses. Exclusive of fetal weights for this 1 litter, mean fetal weight for the 5000 mg/kg group was 3.74 g (combined genders) and was similar to the 3.73 g in controls.
No effect of treatment was evident from fetal external evaluations. The only malformation seen at external examination was an omphalocele in 1 fetus from the 750 mg/kg group. This fetus also had a bent tail; this was considered a variation in the laboratory. In the absence of this malformation among fetuses at the higher dose levels, its occurrence was considered spontaneous and unrelated to treatment. In the control, 750, 1500, and 5000 mg/kg groups, there were 116, 115, 130, and 134 fetuses available for evaluation.
No visceral malformations were seen among the control and treated fetuses and the few visceral variations seen among fetuses in the treated groups occurred at low incidence and in the absence of a dose-related effect, these findings were considered unrelated to treatment (data not shown). Thus, no effect of treatment with DP was evident from fetal external or visceral examinations.
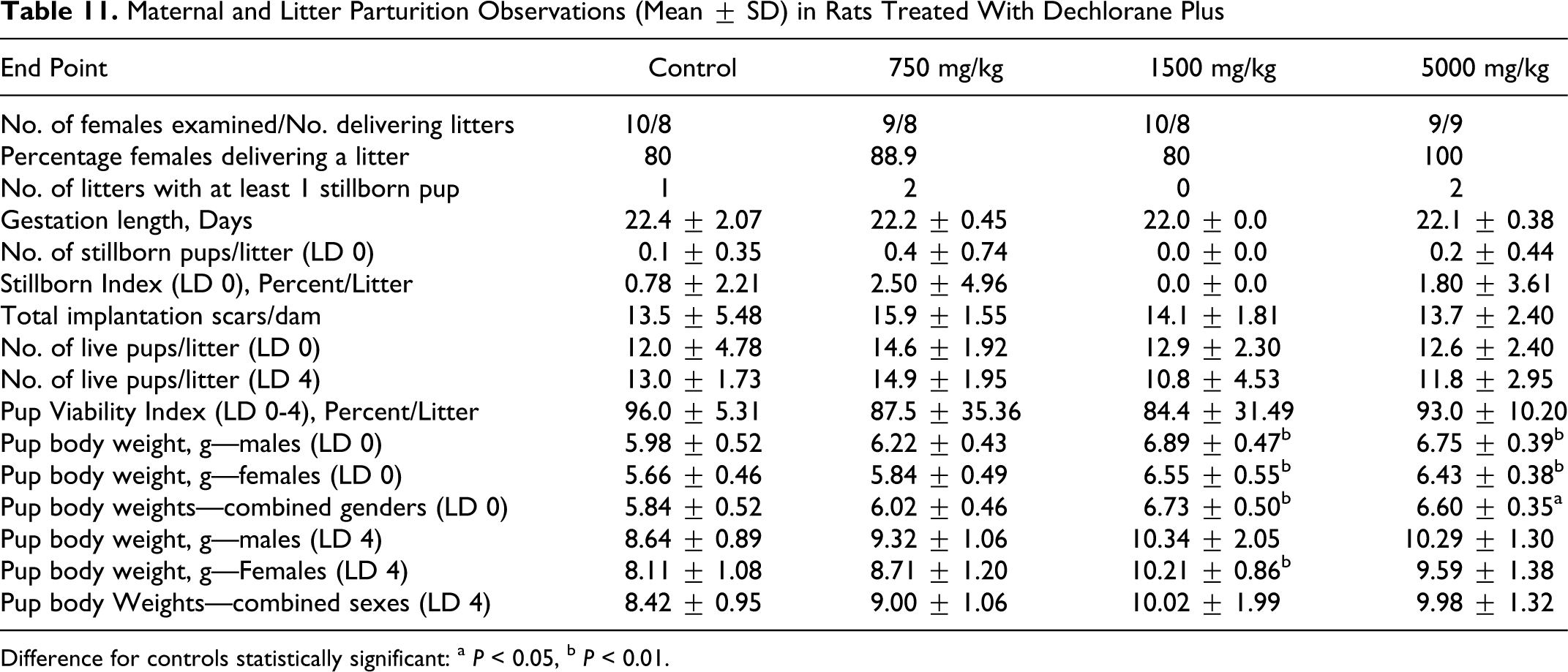
No effect of treatment with DP was evident from parturition data (Table 11 ). From the 10, 9, 10, and 9 females retained from the control, 750, 1500, and 5000 mg/kg groups, respectively, for the parturition phase of study, 8 females in each of the control, 750, and 1500 mg/kg groups and 9 in the 5000 mg/kg group delivered litters.
Maternal and Litter Parturition Observations (Mean ± SD) in Rats Treated With Dechlorane Plus
Difference for controls statistically significant:
a P < 0.05.
b P < 0.01.
Mean gestation length, the incidence of litters with stillborn pups, mean number of pups (live, stillborn, and total) at birth, gestation index, and mean percentage stillborn pups per litter for the treated groups were comparable to controls. The mean number of uterine implantation scars at necropsy in the treated groups also was comparable to the controls and in all groups, the mean number of scars correlated closely with the mean total number of pups (live plus dead) at birth. One control female had a protracted gestation (26 days), and delivered a single live pup that died shortly after birth. No difficult or protracted deliveries were seen among the treated females. No effect of treatment was evident from pup gender ratios (percentage males/litter) at birth or LD 4. Pup gender ratios at birth ranged from 47.7% to 54.3% in the treated groups and were comparable to the 59.1% gender ratio in controls. Gender ratios at LD 4 were similar to those seen at birth, and ranged from 49.0% to 60.2% in the treated groups and were comparable to the 61.5% in controls. Pup survival over the LD 0 to LD 4 (ie, Viability Index) was comparable between the control and treated groups, and no effect of treatment was evident.
No effect of treatment was evident from clinical examination of the pups at birth or LD 4. The few clinical findings seen among the pups from the treated groups occurred at low incidence and were considered incidental and unrelated to treatment (data not presented). Mean pup body weights (Table 11) at birth in the treated groups were higher than controls and in the 1500 and 5000 mg/kg groups, the increased weights by sex and for the sexes combined were statistically significant from controls. Mean pup weights at birth in the 750, 1500, and 5000 mg/kg groups were about 3%, 15%, and 13% heavier than controls, respectively. This increase in pup weights at birth was not considered toxicologically meaningful or indicative of a treatment-related response. Pup' body weights for the treated groups continued to be heavier than controls at LD 4, but only in the 1500 mg/kg female pups was this difference from controls statistically significant. No macroscopic findings were evident in stillborn pups, pups found dead during LD 0 to LD 4, or at terminal euthanasia on LD 4.
No macroscopic findings were seen in parental males designated to the DART phase. Exclusive of the lesions in the viscera of the thoracic cavity that were attributable to gavage errors or aspiration of the test article into the lungs, other macroscopic findings occurred at low incidence and were considered unrelated to treatment. Terminal body weights in the treated males did not differ statistically from controls. Absolute weights of the testes and epididymides and epididymal weights relative to terminal body weight for the treated groups were comparable to controls (data not presented). The only statistically significant difference from controls seen in these organ weight data was an increase in testes weights relative to body weight in the 5000 mg/kg group. In the absence of a dose-related effect, the increase in relative testis weight for this group was not considered toxicologically meaningful or indicative of a treatment-related response.
Discussion
This study was conducted to evaluate for possible adverse effects of the test article, DP, following repeated oral dosing of rats over a 28-day period that included an evaluation of potential neurologic effects (RDT). In addition, this study was conducted to evaluate male and female reproductive performance that included an evaluation of gonadal function, mating behavior, conception, development of the conceptus, parturition, and early neonatal development (DART). Oral dose levels in both phases of study were 750, 1500, or 5000 mg/kg. Control animals received the vehicle (corn oil). The dose levels used in this study were based on an unpublished 90-day dietary study conducted in the mid-1970s. 4 In that study, rats were treated with DP at dietary concentrations of 10 000, 30 000, or 100 000 ppm, equivalent to oral doses in males of approximately 522, 1567, and 5200 mg/kg, respectively, and in females of approximately 654, 1960, and 6500 mg/kg, respectively.
In both the RDT and DART phases, several animals across all groups died during the study, but there was no correlation to dose or time-on-study. Based on gross necropsy or histopathological examination, most of these deaths in the treated groups could be related to gavage injury or aspiration of the test article and not directly related to treatment. Irrespective of these deaths, macroscopic or microscopic lesions observed in the thoracic cavity (ie, adhesions, inflammation, and fibrosis) of some animals that survived to scheduled termination also showed evidence of dosing error or aspiration of the test article. In those animals that died during the study and in survivors, clinical signs of toxicity were evident consistent with the probable mis-dosing. During the course of the study as deaths were occurring, it became readily apparent that the test article as formulated in corn oil may have been too viscous, which resulted in difficulty of administration. The results of viscosity measurement revealed that corn oil had a viscosity of 60 cps whereas for the 75, 150, and 500 mg/mL formulations, the viscosity measures were 70, 90 cps, and 5000 to 9000 cps, respectively. Clearly, particularly at the high concentration, the viscosity of the test article suspensions most likely contributed to the mis-dosing and mortalities observed. Moreover, it was apparent that the rapidity by which the technical staff administered the suspensions also may have contributed to the mortality. Finally, although the doses were selected to achieve the high doses used in the dietary study, recognizing the difference in dosing regimen, in hindsight the high and intermediate dose levels were thought to be too high. Regardless, in spite of the gavage-related effects, no systemic toxicity was observed in this study that could be directly correlated with DP.
Corn oil was found to be the vehicle of choice to suspend DP after investigating several alternative vehicles. In addition to adding to the caloric intake of animals, there are data to suggest that this vehicle may affect the toxicity of some substances. In the 1980s and 1990s, several investigators examined the possible enhanced effect of corn oil on liver and kidney toxicity with oral administration of chloroform, carbon tetrachloride, and trihalomethanes. Lilly and colleagues 15 did not find an effect of corn oil on the hepatotoxicity of bromodichloromethane, whereas Raymond and Plaa 16 showed that corn oil enhanced the kidney toxicity of chloroform with a minimal effect on the liver toxicity of carbon tetrachloride following acute administration. These latter authors suggested that the effect of corn oil on carbon tetrachloride toxicity may be related to altered absorption of compounds. Koporec et al, 17 however, showed that subchronic oral administration of carbon tetrachloride in corn oil did not affect the hepatotoxicity of the compound. In the current study, no hepatic effects were observed although, as suggested above, this may be related more to the low absorption of the compound rather than the use of corn oil as a vehicle.
In a 2-year study of corn oil administration to rats, there were dose-related increases in pathology findings in the pancreas, kidney, and adrenal. 18 No evidence of systemic toxicity in the current study was evident that could be related to the findings with corn oil reported by the National Toxicology Program (NTP). 18
In the early dietary administration study of DP conducted by IBT, other than increased absolute and relative liver weights of rats, no other toxicologically significant finding was observed. 4 This study was criticized by the EPA since it had been conducted prior to implementation of GLPs. 6 In addition, an increased liver weight with microscopic evidence of hepatomegaly was observed in rats exposed by inhalation for 28 days (6 h/d, 5 d/week) to DP at concentrations of 0.64 and 1.524 mg/L. 6 In the current study, however, no increase in liver weights was observed and no toxicologically significant microscopic pathology changes were observed. The liver appears to be a target organ for the BFRs 2 and is certainly a target organ for certain highly halogenated-substituted compounds, for example, carbon tetrachloride, trichloroethylene, and so on. Although DP is a highly chlorinated compound, the lack of target organ toxicity in this study is considered due to the limited absorption of the compound as seen in the metabolism study with radiolabeled DP; blood levels of DP were not determined in the current study.5 In the metabolism study, about 0.01% or less of the administered dose was found in the blood, urine, liver, and kidney (only tissues examined) with approximately 95% of the radiolabeled compound appearing in the feces. These data certainly suggest that DP is not well absorbed into systemic circulation.
Parameters evaluated in the RDT phase and unaffected by treatment included clinical signs of toxicity, body weights, food consumption, neurobehavioral, and FOB evaluations to include motor activity, clinical pathology, organ weights, and macroscopic and microscopic evaluations. A slight increase in ovary weights was observed in rats treated with 1500 mg/kg DP but not at the higher dose. In the absence of a dose-related change and with no corresponding microscopic evidence, the change in ovarian weight was not considered biologically significant. Furthermore, in the absence of a reproductive effect during the DART phase, the change in ovarian weight was considered normal biological variation.
Parameters evaluated and unaffected by treatment in the DART phase included clinical signs of toxicity, body weights (3-week premating period, gestation, and lactation), food consumption (premating and gestation), estrous cyclicity, reproduction/fertility indices, GD 20 uterine implantation data, fetal body weights, fetal gender ratios, and fetal external and visceral examinations, parturition (gestation length, litter size), and pup body weights, gender ratios, survival, and clinical findings through LD 4. On the basis of these data, the no-observable-effect level (NOEL) with DP in both the RDTand DART phases of study was 5000 mg/kg, the highest dose level evaluated.
Footnotes
Acknowledgment
This study was sponsored by Occidental Chemical Corp.
The authors declared a potential conflict of interest (e.g. a financial relationship with the commercial organizations or products discussed in this article) as follows: Author Janice M. Nyberg is employed by Occidental Chemical Corp.
The authors received no financial support for the research and/or authorship of this article.
