Abstract
To characterize the toxicological hazards of petroleum gases, 90-day inhalation toxicity (Organization for Economic Cooperation and Development [OECD] 413) and developmental toxicity (OECD 414) tests were conducted with liquefied propane gas (LPG) at concentrations of 1000, 5000, or 10 000 ppm. A micronucleus test (OECD 474) of LPG was also conducted. No systemic or developmental effects were observed; the overall no observed adverse effect concentration (NOAEC) was 10 000 ppm. Further, there was no effect of LPG exposure at levels up to 10 000 ppm on micronucleus induction and no evidence of bone marrow toxicity. Other alkane gases (ethane, propane, n-butane, and isobutane) were then evaluated in combined repeated exposure studies with reproduction/development toxicity screening tests (OECD 422). There were no toxicologically important changes in parameters relating to systemic toxicity or neurotoxicity for any of these gases at concentrations ranging from 9000 to 16 000 ppm. There was no evidence of effects on developmental or reproductive toxicity in the studies of ethane, propane, or n-butane at the highest concentrations tested. However, there was a reduction in mating in the high-exposure group (9000 ppm) of the isobutane study, which although not significantly different was outside the range previously observed in the testing laboratory. Assuming the reduction in mating to have been toxicologically significant, the NOAEC for the isobutane reproductive toxicity screening test was 3000 ppm (7125 mg/m3). A method is proposed by which the toxicity of any of the 106 complex petroleum gas streams can be estimated from its composition.
Keywords
Introduction
The United States Environmental Protection Agency (US EPA) announced a voluntary chemical data collection effort in 1998 called the High Production Volume (HPV) Challenge Program. 1 The HPV chemicals are those produced or imported into the United States in aggregate quantities of at least 1 million pounds per year. Approximately 400 petroleum substances were sponsored in the EPA’s Challenge Program by companies belonging to the Petroleum HPV Testing Group. The various substances were organized into 13 categories to facilitate data sharing and to avoid redundant testing. These categories included crude oil, gases, gasoline, kerosene/jet fuel, gas oils, heavy fuel oils, lubricating oils, waxes, aromatic extracts, asphalts, grease thickeners, petroleum coke, and hydrocarbon wastes. This article reports an investigation into the toxicological hazards of those petroleum-derived hydrocarbon substances that exist in the gaseous state under conditions of standard temperature and pressure. Based on a survey of the Chemical Abstract Services (CAS) registry numbers of substances identified through the voluntary HPV chemical evaluation process, there are 161 high-volume substances that have been identified by the petroleum industry as gases, making this the largest of the 13 petroleum substance categories. However, this category includes 2 very different types of gases, “petroleum hydrocarbon gases” that are complex substances primarily comprised of hydrocarbon constituents and are the subject of this report and “refinery gases” that contain primarily inorganic constituents. The petroleum hydrocarbon gas streams are identified by 106 CAS numbers, of which 92 are HPV substances and the other 14 are similar, related substances (Appendix A). There are also 55 “refinery gases” that are primarily composed of inorganic substances and either produced in the refinery for use as process gases (eg, H2) or generated as waste gases during refining processes (eg, H2S). The present report focuses on the toxicological properties of the petroleum gases and provides a method by which the toxicological hazards of these 106 complex substances can be estimated. The toxicological hazards of the inorganic refinery gases are outside the scope of this article, and inorganic constituents are only discussed to the extent that they are present at low levels in some petroleum hydrocarbon gas streams.
Petroleum hydrocarbon gases can either be produced in the refinery by distillation of crude oil or by separation in gas plants and are comprised primarily of C1 to C4 constituents (methane, ethane, propane, n-butane, and isobutane) in varying proportions, although there are higher molecular weight hydrocarbon constituents (primarily C5 to C6 alkanes, although depending on the process, benzene may also be present) that may be entrained in the gas streams. Gaseous hydrocarbon streams can also be produced by refining processes, particularly “cracking” processes by which large molecules are converted to smaller molecules either thermally or in the presence of a catalyst. These cracking processes, particularly catalytic cracking, create olefins, a type of hydrocarbon not normally present in crude oil or natural gas streams. Some of these hydrocarbon gas streams contain 1,3-butadiene at more than trace levels. The olefin-rich streams can be used as fuels and can also be used in the manufacture of petrochemicals.
Depending on the source or method of production, the petroleum hydrocarbon gases may also contain inorganic constituents such as hydrogen, nitrogen, and carbon dioxide. These low-molecular-weight hydrocarbon and/or inorganic constituents are believed to present few human health hazards other than asphyxiation. The more hazardous inorganic refinery gas constituents such as hydrogen sulfide and ammonia are not commonly found in petroleum gases at greater than trace levels. Some complex petroleum gases may also contain low levels of benzene and/or 1,3-butadiene. During production and refining, human contact with petroleum and refinery gases is limited, because the gaseous substances are maintained in closed systems to avoid loss and to minimize the likelihood of achieving explosive concentrations in air. Commonly, methane and ethane are burned in the refinery for energy recovery (ie, fuel gases). Propane and butanes (n-butane and isobutane) are typically used in the production of liquefied propane gas (LPG), which is used as a fuel by both domestic and industrial consumers. However, some gaseous substances have other uses, and some of the gases, particularly those that contain benzene and/or 1,3-butadiene, can be used as chemical feed stocks.
Information previously available suggested that the C1 to C4 hydrocarbons were asphyxiants at high concentrations but otherwise presented minimal toxicological hazards. Methane, ethane, propane, and butane are simple, low-molecular-weight molecules without functional groups. Data from inhalation studies of butane and isobutane indicate that when inhaled they are not well absorbed. 2 When these substances are absorbed, pharmacokinetic studies indicate that they are rapidly eliminated, primarily by inhalation. Filser et al 3 reported biological halftimes of 57 minutes for ethane and 8 minutes for pentane. Based on the levels in expired air, the elimination halftimes for propane and isobutane are in the range of 20 to 25 minutes. 4 In studies with volunteers, no symptoms were noted after 10-minute exposure to air containing 10 000 ppm propane, but 2-minute exposure to 100 000 ppm propane caused vertigo. Exposure to up to 10 000 ppm butane for 10 minutes produced no symptoms other than drowsiness. 5 Subsequent volunteer studies 4,6,7 showed that acute or repeated exposures to propane and isobutane at levels up to 1000 ppm for periods of up to 8 hours did not produce any untoward physiological effects. These data suggested that, in addition to being asphyxiants, low-molecular-weight alkanes might also cause acute central nervous system effects at high concentrations 8 but are unlikely to produce other effects at levels up to at least 1000 ppm.
Experimental studies of propane and isobutane in dogs,
9
mice,
10
rats,
11
and primates
12
demonstrated that certain gaseous hydrocarbons could produce cardiac sensitization if inhaled at very high levels (ie, >25 000 ppm).
13
Kirwin and Thomas
14
reported that low-molecular-weight alkanes were not mutagenic when tested in
The work described herein, which was undertaken for the purposes of satisfying the HPV obligations of the petroleum industry, included a review of the relevant CAS numbers to understand the types and ranges of constituents that might be present in these gases, a collection and review of hazard information on the constituents, and an assessment of the areas that required further study. This analysis led to the determination that further studies of the repeated dose and reproductive toxicity of representative substances were warranted, and, accordingly, studies of LPG were conducted following Organization for Economic Cooperation and Development (OECD) 413 (90-day inhalation toxicity study), OECD 414 (prenatal developmental toxicity), and OECD 474 (mammalian erythrocyte micronucleus test) guidelines to more precisely define the potential for toxicity for this substance as it is used as a fuel gas by the general population. In addition, 4 repeated dose/reproductive toxicity screening studies were conducted following the OECD 422 protocol to provide information on the potential of gas stream constituents to cause systemic toxicity and/or to affect fertility. Specific substances tested were ethane, n-propane, n-butane, and isobutane. The data from these studies were then used to develop a method by which the toxic properties of any of the 106 petroleum hydrocarbon gas streams could be calculated from compositional information.
Materials and Methods
Materials
Liquefied propane gas
Liquefied propane gas, CAS number 64741-79-3, was supplied by ChevronTexaco Energy Research and Technology Company (San Ramon, California). Based on the chromatographic evidence, the sample contained (by weight) approximately 93.5% propane/propylene. Other principal constituents included 3% butane isomers and 1.8% ethane with the remaining 1.7% being primarily other C1 to C5 alkanes and alkenes. The gas was used as supplied.
Other gases
Ethane (CAS number 74-84-0), propane (CAS number 74-98-6), n-butane (CAS number 106-97-8), and isobutane (CAS number 75-28-5) were purchased from MG Industries (Malvern, Pennsylvania). The purity of the gases as indicated by the supplier was given as 99.0% to 99.5% depending on the gas. Analytical confirmation at the testing facility by gas chromatography confirmed purities >99%. The gases were used as supplied.
Methods
Testing Guidelines
The repeated exposure study of LPG was in accordance with OECD 414 (repeated dose, inhalation), and the developmental toxicity test followed the recommendations of OECD 413 (prenatal developmental toxicity). In addition, femurs were taken from rats exposed in the repeated dose study and used to assess the potential for micronucleus formation following OECD 474. The studies of ethane, propane, butane, and isobutane were conducted using protocols that complied with OECD 422 (Combined Repeated Exposure Toxicity Study with the Reproduction/Development Toxicity Screening Test) and the US EPA OPPTS Health Effects Test Guideline 870.3650 (Combined Repeated Exposure Toxicity Study with the Reproduction/Development Toxicity Screening Test). The testing was conducted in accordance with EPA Good Laboratory Practices (40 CFR Part 792) and the Good Laboratory Guidelines from OECD (ENV/MC/CHEM (98)17). As there were similarities between the 90-day inhalation toxicity test of LPG and the repeated dose studies of the other gas constituents, the common elements of those tests are discussed together to the extent possible.
Inhalation Exposures
Rats were exposed 6 hours/d in Rochester design 1-m3 chambers (Wahmann, Baltimore, Maryland), which were operated at a minimum flow rate of 200 L/min. The final airflow was set to provide at least 1 air change (calculated by dividing the chamber volume by the airflow rate) in 5.0 minutes (12 air changes/h) and a T99 equilibrium time (calculated by multiplying the time required for a single air change by a constant, 4.6) of at most 23 minutes. The chamber size and airflow rates were considered adequate to maintain the animal loading factor below 5% and the oxygen level at 19% or higher. At the end of the exposure period, all animals remained in their chambers for a minimum of 30 minutes during which time the chamber was flushed with clean air at the same flow rate as was used for test material administration.
Each test gas was delivered from a single cylinder, through a regulator and 2 back pressure gauges, and branched, via 0.25-in tubing, to the 3 exposure chambers. For each chamber, 0.25-in tubing directed the test substance to a flow meter, regulated by a metering valve and into the inlet of the chamber. The desired test concentrations were achieved by diluting the gas streams with clean air.
The exposure levels were verified with a MIRAN Ambient Air Analyzer (Foxboro Wilks, Foxboro, Massachusetts) with a strip chart recorder. The test atmosphere was drawn from a sampling portal through the MIRAN, and the measurements were recorded at least 4 times during each exposure period. The exposure levels were determined by comparing the measured absorbance with a calibrated response curve constructed using the same instrument settings. Calibrations were done using a closed-loop system in which known volumes of gas were injected into a known 5.64 L volume of air in the Miran to create known concentrations of gas.
Animals
All the studies were conducted using Sprague-Dawley rats (Crl: CD (SD) IGS BR) obtained from Charles River Laboratories (Raleigh, North Carolina). The rats were approximately 6 weeks of age at receipt and were then held for an acclimation period of approximately 2 weeks, making them approximately 8 weeks old at study initiation. Those that were judged suitable for the study were randomly assigned by a computerized random assort program by sex and body weight, to control or treated groups.
Repeated Inhalation Exposure Studies
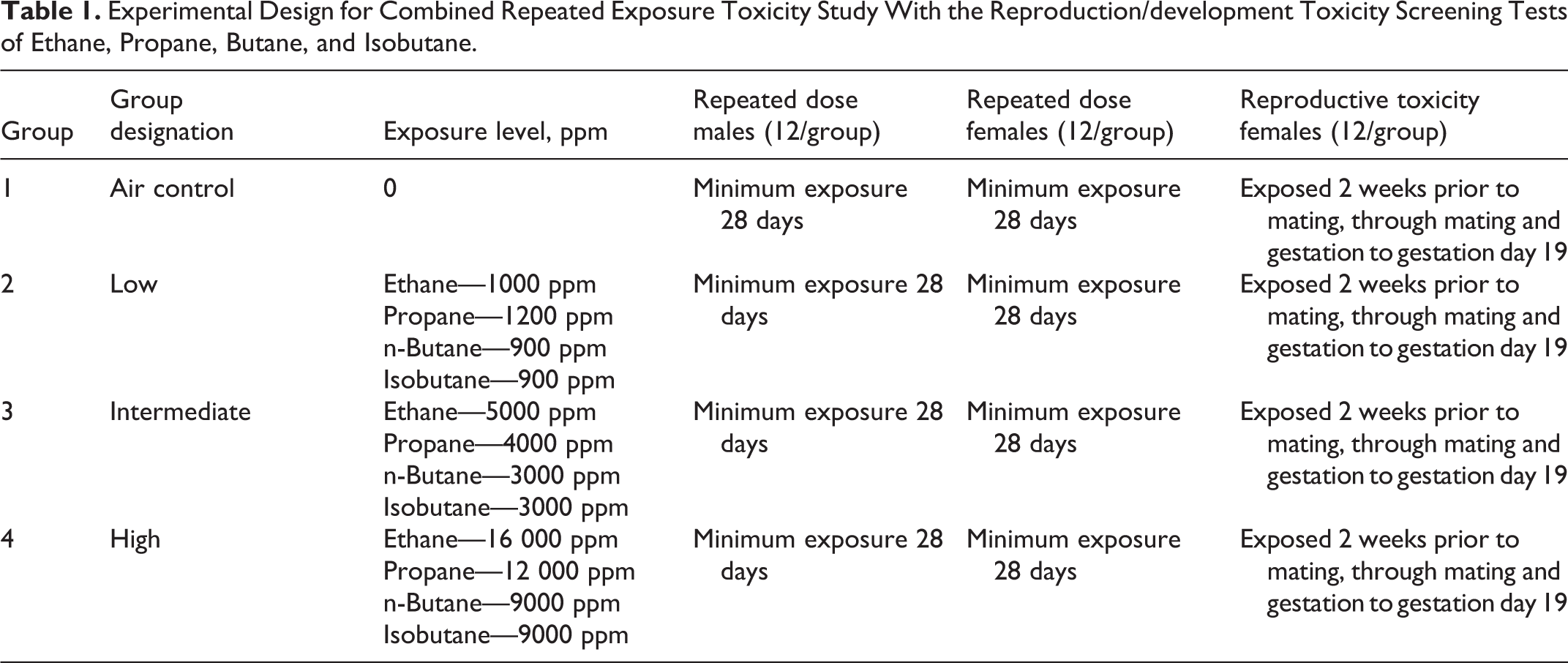
In the 90-day inhalation toxicity test of LPG, rats were exposed in groups of 15/sex/treatment group, 6 hours/d, 5 days/week for 13 weeks to LPG at target concentrations of 1000, 5000, or 10 000 ppm. The experimental outline for the repeated dose/reproductive toxicity screening tests of ethane, propane, butane, and isobutane is shown in Table 1. In the screening studies, rats were exposed in groups of 12 at target concentrations similar but somewhat different from those used in the LPG study, but in other respects, that is, rat strain, sacrifice, gross and pathological examinations, clinical examinations, neurological assessments, and statistical evaluation, the animals were treated as described subsequently. The highest exposure levels used in these studies were approximately half the lower explosive limits (18 000-30 000 ppm) for the gases 16 and were considered to be the highest levels that could be safely tested under laboratory conditions.
Experimental Design for Combined Repeated Exposure Toxicity Study With the Reproduction/development Toxicity Screening Tests of Ethane, Propane, Butane, and Isobutane.
Animal Husbandry
Practices were in accordance with
In Life Observations
All animals were checked at least once daily for mortality and/or signs of ill health. The animals were removed from the cages and given external examinations twice before initiation of exposures and on a weekly basis prior to exposure and during the exposure period. The examination included a physical examination for general condition, neurobehavioral observations, and functional observations. Body weights were recorded at the time of randomization into test groups, on the day that treatment was initiated, on a weekly basis during the study, and prior to scheduled sacrifice. Food consumption was monitored by weighing the feeders on a weekly basis, prior to refilling.
Neurobehavioral Assessments
Male and female rats that were not used in the reproductive toxicity assessment (described later) were tested for neurological effects. The testing was conducted during the last week of exposure and on days when the animals were not exposed. The testing consisted of a functional observation battery, which included sensory observations (startle response and tail pinch response), grip strength, and rectal temperature measurements. Motor activity was also tested in a Photobeam Activity System (San Diego Instruments, Inc, San Diego, California) device. Sessions were 60 minutes in length, divided into 12 five-minute intervals.
Terminal Sacrifice
Clinical pathology
Blood samples were collected from lightly anesthetized animals at study termination. Hematological measurements were made for hemoglobin concentration, hematocrit, erythrocyte count, platelet count, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, total leukocyte count, reticulocyte count, differential leukocyte count, and erythrocyte and platelet morphology. Additionally coagulation studies were conducted. Clinical chemistry evaluations included aspartate aminotransferase, alkaline aminotransferase, alkaline phosphatase, blood urea nitrogen, creatinine, glucose, cholesterol, total protein, triglycerides, albumin, total bilirubin, sodium, potassium, chloride, calcium, inorganic phosphorus, γ-glutamyl transpeptidase, globulin, and albumin–globulin ratio.
Postmortem evaluations
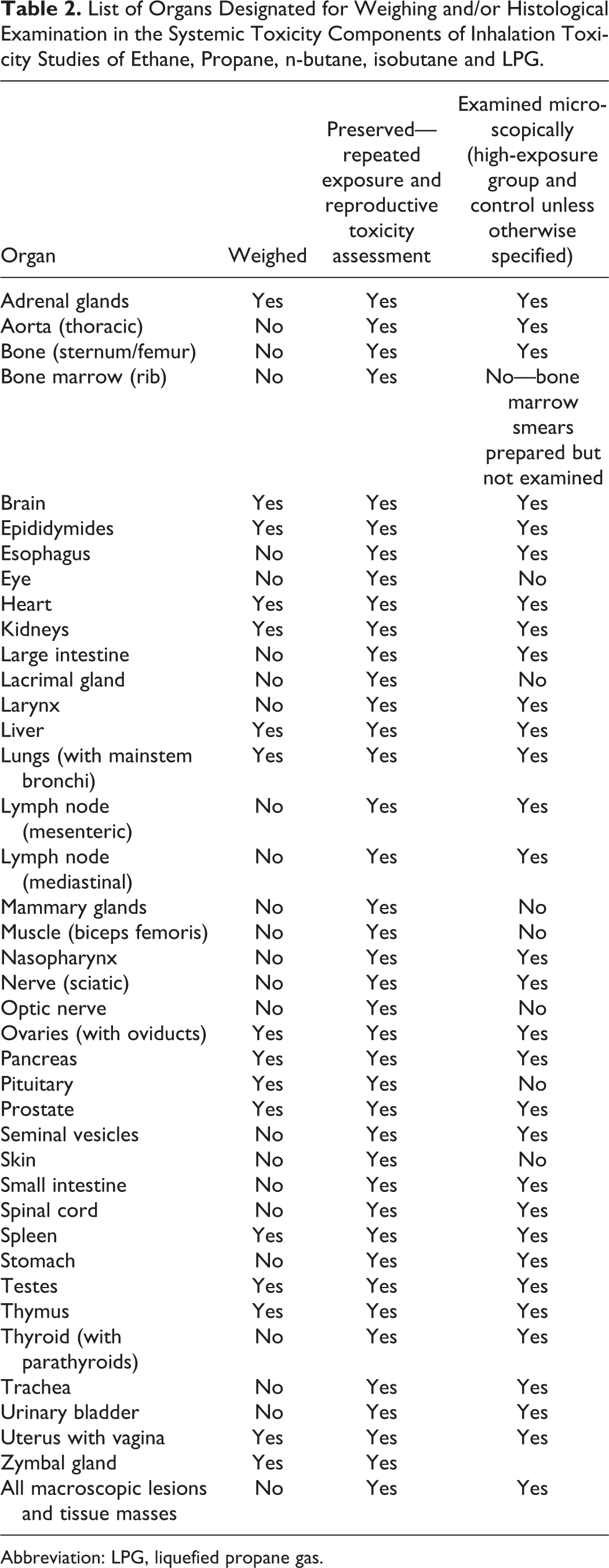
After sacrifice by carbon dioxide inhalation followed by exsanguination, each animal was given a postmortem macroscopic examination, and all observations were recorded. Organs that were taken for weighing and/or histologic examination are shown in Table 2. All tissues were preserved in 10% neutral-buffered formalin. Testes and epididymis were placed in Modified Davidson solution for 24 hours and then retained in 10% neutral-buffered formalin. Lungs and urinary bladder were infused with 10% neutral-buffered formalin for optimal preservation. After fixation, selected tissues as shown in Table 2 were routinely processed and embedded in paraffin. Sections were mounted on glass slides and stained with hematoxylin and eosin.
List of Organs Designated for Weighing and/or Histological Examination in the Systemic Toxicity Components of Inhalation Toxicity Studies of Ethane, Propane, n-butane, isobutane and LPG.
Abbreviation: LPG, liquefied propane gas.
Prenatal Developmental Toxicity (LPG)
This study was conducted with pregnant female Sprague-Dawley rats. A total of 100 timed pregnant rats were received on gestation days 0, 1, or 2, held for observation in the testing facility for 4 to 6 days, and then randomly assigned to study groups of 24. Exposures were initiated on gestational day (GD) 6 at levels of 0, 1000, 5000, or 10 000 ppm, 6 hours/d, 7 days/week to scheduled termination on GD 20. Body weights and food consumption were measured on GD 3, 6, 9, 12, 15, 18, and 20 (scheduled termination). The rats were euthanized by overexposure to carbon dioxide and then given a gross necropsy. The intact uteri were removed from all animals and weighed. Corpora lutea were counted, and the number per ovary was recorded. The number and location of live fetuses, late embryofetal deaths, and early embryonic deaths were recorded.
All live fetuses were weighed, identified, and given external examinations for defects. The fetuses were then euthanized by injection of sodium pentobarbital. Approximately half the fetuses were placed in Modified Davidson fixative for preservation and decalcification. These fetuses were then examined for soft tissue defects by a razor blade-sectioning technique based on Wilson and Warkany. 18 All malformations and variations were recorded. During the dissection process, the sex of each fetus was confirmed by visual inspection of the gonads. Following complete dissection of the fetuses, all carcasses and sections were preserved in 10% neutral-buffered formalin.
The remaining fetuses were eviscerated, placed in 70% isopropyl alcohol for preservation, and processed for staining of the skeleton using Alizarin Red S. Subsequently, these fetuses were evaluated for skeletal malformations and ossification variations. The skeletons were then stored in 100% glycerin. During the dissection process, the sex of each fetus was confirmed by internal inspection of the gonads.
Reproductive Toxicity Screening Tests (Ethane, Propane, n-Butane, and Isobutane)
Mating procedures
Following daily exposures for 14 consecutive days, male rats were cohoused with female rats designated for assessment of reproductive effects for 2 consecutive weeks or until mating was confirmed. Mating was confirmed by evidence of a vaginal plug or evidence of sperm in the vaginal smear. The day the mating was confirmed was designated GD 0, and the mated females were singly housed until termination. Females for which mating was not confirmed were cohoused with males for the entire 2-week mating period and then singly housed with daily observations until study termination.
Evaluations
Animals were examined daily for viability and clinical signs. Body weights were recorded at the time of randomization and weekly thereafter. Females scheduled for reproductive toxicity assessment were weighed on GDs 0, 7, 14, and 20 and on lactation days (LDs) 1 and 4. Feed consumption was recorded pretest and weekly during the treatment period. Feed consumption was not recorded during mating. During gestation feed consumption was recorded on GDs 0 to 7, 7 to 14, and 14 to 20 and on LDs 1 to 4.
Parturition and offspring
On day 18G, pregnant dams were transferred to bedding boxes and examined twice daily for signs of parturition. Litters were examined for number of live and dead pups; pups were sexed, and external malformations were recorded. Pups were examined on postnatal days (PND) 0 and 4, pup weights were taken on PNDs 1 and 4, and pup gender was verified on PND 4.
Examination of offspring
All offspring born dead or found dead during lactation were given macroscopic examinations. All offspring surviving to PND 4 were sacrificed and examined macroscopically. The offspring were not examined grossly.
Micronucleus Test (LPG)
Each LPG-exposed group contained 5 animals/sex that were used to assess the potential for micronucleus formation. These animals were also exposed 6 hours/d, 5 days/week for 13 weeks to LPG at levels of 1000, 5000, or 10 000 ppm. There was also a positive control group for the micronucleus studies, which contained 5 animals/sex. These positive control group rats were not exposed to LPG but, rather, were given intraperitoneal injections of 40 mg/kg of cyclophosphamide prior to sacrifice.
The rats scheduled for micronucleus assessment were sacrificed by overexposure to carbon dioxide, the right femur of each of the rats was removed, and the bone marrow was sampled. Unstained bone marrow slides (4/animal) were prepared. Two slides/animal were stained with acridine orange and evaluated using a fluorescent microscope for determination of micronucleus response. The other 2 slides were held in reserve.
Statistical Analysis
Repeated exposure and reproductive toxicity screening tests (including neurological evaluations)
Mean values for all exposed groups were compared to the mean value for the corresponding control group at each time interval.
Evaluation of equality of group means was made by the appropriate statistical test followed by a multiple comparison test if needed. Bartlett test
19,20
was performed to determine whether groups had equal variances. Organ weight data were analyzed by a parametric method, the standard 1-way analysis of variance (ANOVA) using the
Motor activity count data were analyzed using split-plot repeated measures ANOVA with model terms for group, animal within group, interval and group by interval interaction. If the group × interval interaction was significant (
Incidence data were analyzed using a Fisher exact test with Bonferonni correction to identify differences between the control and the treatment groups. 31 All statistical tests were conducted at the 5% and 1% risk levels.
Prenatal Developmental Toxicity Test
For analysis of continuous data including maternal body weight and body weight changes, maternal feed consumption, gravid uterine weights, implantation data, preimplantation loss, early embryonic deaths, live fetuses, late embryofetal deaths, total embryofetal deaths (and as percent implantation sites), mean percentage of female fetuses, and the mean values from all exposure groups were compared to the mean value of the control group at each time interval. Evaluation of equality of group means was made by the appropriate statistical method (either parametric or nonparametric), followed by a multiple comparison test if needed. Bartlett test 19,20,34 was performed to determine whether the groups had equal variances. For all parameters, if the variances were equal, parametric procedures were used; if not, nonparametric procedures were used.
The parametric method was the standard 1-way ANOVA using the
The incidence data including premature deliveries, total pregnancy loss (no live fetuses), maternal necropsy findings, external fetal defects, skeletal malformations and variations, and soft tissue malformations and variations were analyzed based on a generalized estimating equation application of the linearized model.
35
For litter end points, the model used the litter as the basis for analysis and considered correlation among littermates by incorporating an estimated constant correlation and the litter size as a covariate. If the dose group effect in the model was statistically significant, the dose group least squares means were tested pairwise versus the control group using
The fetal body weights (by sex and as a composite for both sexes) were analyzed by a mixed model ANOVA. The analysis used the litter as the basis for analysis and the litter size as a covariate. The model considered dose group, litter size, and fetal sex as explanatory variables. If the dose group effect in the model was statistically significant, the dose group least squares means were tested pairwise versus the control group using
Results
Liquefied Propane Gas
Exposure levels
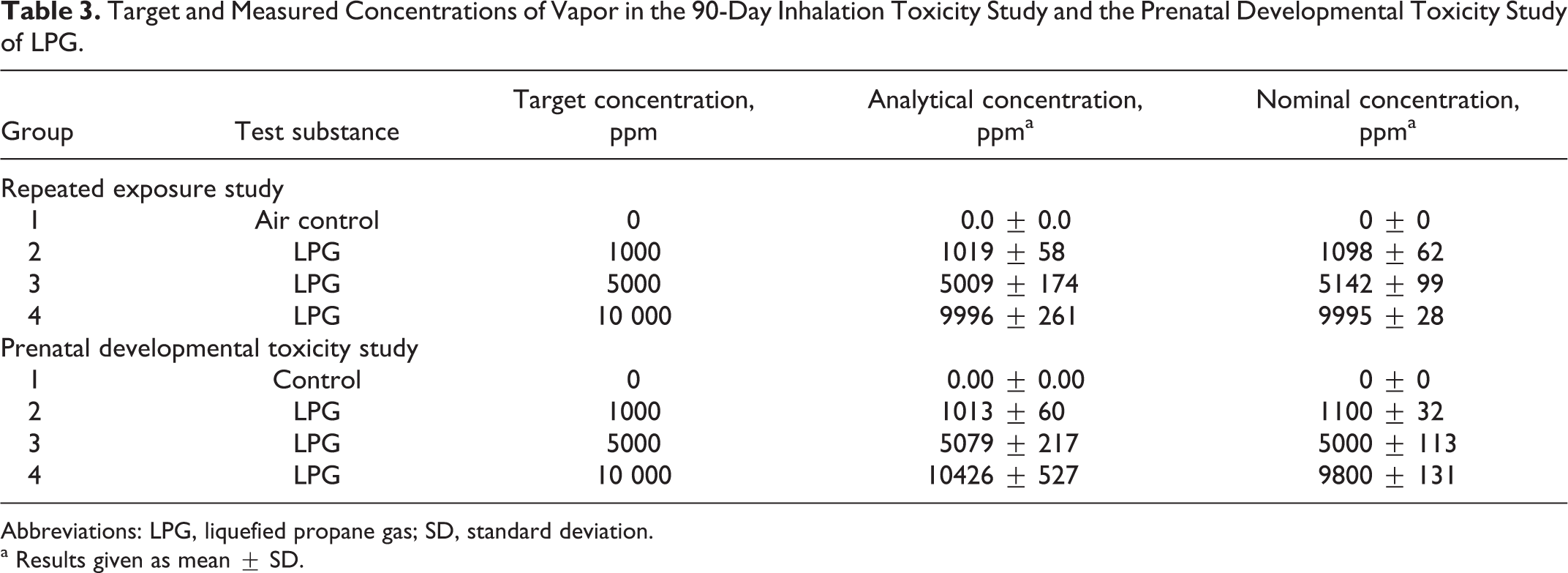
The mean (±standard deviation) analytically determined (infrared [IR]) and nominal (by volume of gas consumed) concentration values were close to the target concentrations of 1000, 5000, and 10 000 ppm as summarized in Table 3.
Target and Measured Concentrations of Vapor in the 90-Day Inhalation Toxicity Study and the Prenatal Developmental Toxicity Study of LPG.
Abbreviations: LPG, liquefied propane gas; SD, standard deviation.
a Results given as mean ± SD.
Assessment of systemic effects following repeated exposure
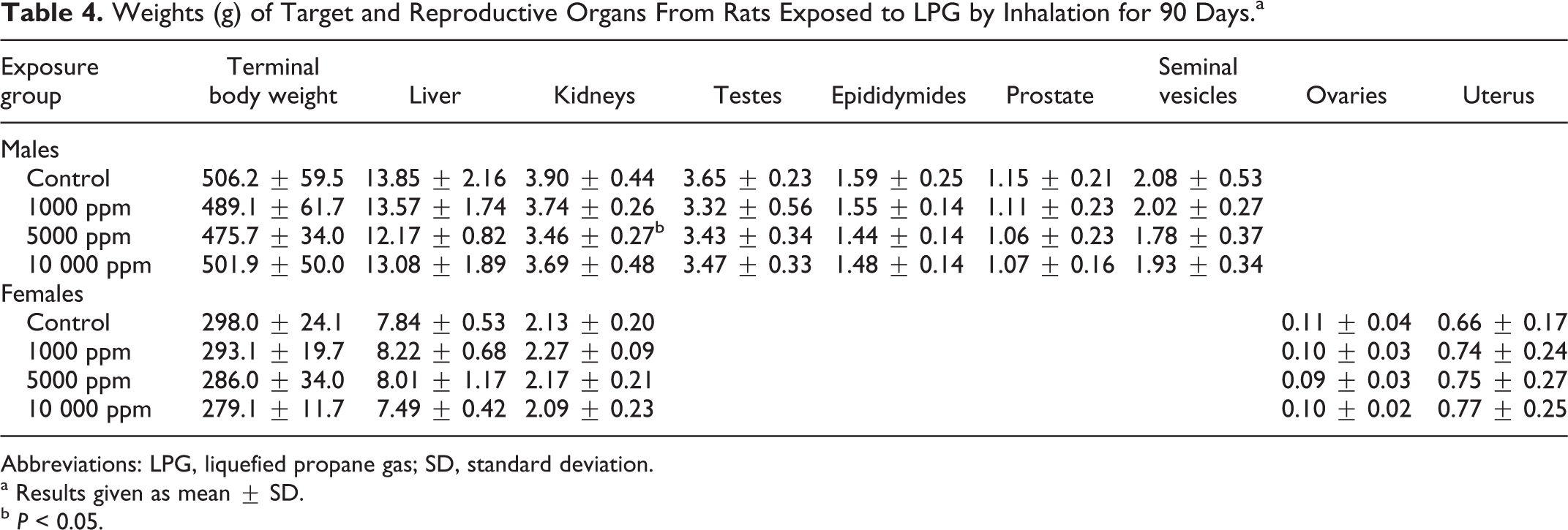
In the repeated inhalation toxicity study of LPG, 1 rat was sacrificed prior to scheduled termination, a female in the 1000 ppm group. As this rat was in the lowest exposure group, the death was considered to have been an incidental finding. There were no significant differences in body weight, body weight gain, or food consumption (data not shown). There were no statistically significant changes in hematological parameters or in clinical chemistry parameters (data not shown). The only statistically significant differences in organ weight data were decreased kidney and thymus weights; however, as the differences were not dose responsive, that is, statistical significance was achieved in the 5000 ppm but not the 10 000 ppm exposure group, they were judged to have been incidental findings. As a partial assessment of the potential for LPG to produce reproductive effects, the reproductive organs were evaluated for weight changes and also examined pathologically. There were no differences in weights of testes, epididymides, prostate, seminal vesicle, ovaries, or uterus (with vagina; Table 4), and no pathological changes were found in these organs during the microscopic examination. The number of normal sperm was significantly reduced in the high-exposure group (98.6% normal in control vs 95.3% in the 10 000 ppm group). This was judged to have been associated with a slight (but not statistically significant) increase in sperm with “mid-tail blobs,” a term used to describe a cytoplasmic droplet observed in the sperm tail that is lost during sperm maturation. To further assess the potential significance of these findings, a second set of slides were prepared from fixed sperm samples and were analyzed, and similar results were obtained. However, as there were similar numbers of headless sperm, sperm with abnormal heads, necks, or tails in the high-dose group and the controls, no differences in sperm counts, and no histological findings in the testes or accessory organs, this slight reduction in normal sperm was judged to have been incidental and not related to treatment. There were no effects in the neurological evaluation. The overall conclusion was that 10 000 ppm was the no observed adverse effect concentration (NOAEC) for the repeated exposure study of LPG.
Weights (g) of Target and Reproductive Organs From Rats Exposed to LPG by Inhalation for 90 Days.a
Abbreviations: LPG, liquefied propane gas; SD, standard deviation.
a Results given as mean ± SD.
b
Assessment of the potential for developmental toxicity
All animals survived to scheduled termination, weight gains were similar across groups, and all but 1 female (in the low-exposure group) had litters. There were no effects observed during gross necropsy or on pregnancy outcome in terms of corpora lutea numbers, pre- and postimplantation loss, early or late resorptions, or litter size and gravid uterine weights (Table 5). All values were within the ranges considered normal for this strain of rats. There were no differences in fetal weights. There were a few fetal abnormalities (Table 5) and variations (data not shown) in each of the treatment groups, but there was no consistency in response and no apparent relationship with exposure level. Similarly, there were no delays in ossification. As there were no treatment-related maternal or fetal effects found in this study, the NOAEC for both maternal and fetal effects was 10 000 ppm.
Results of Cesarean Section Data From the Prenatal Developmental Toxicity Study of LPG.
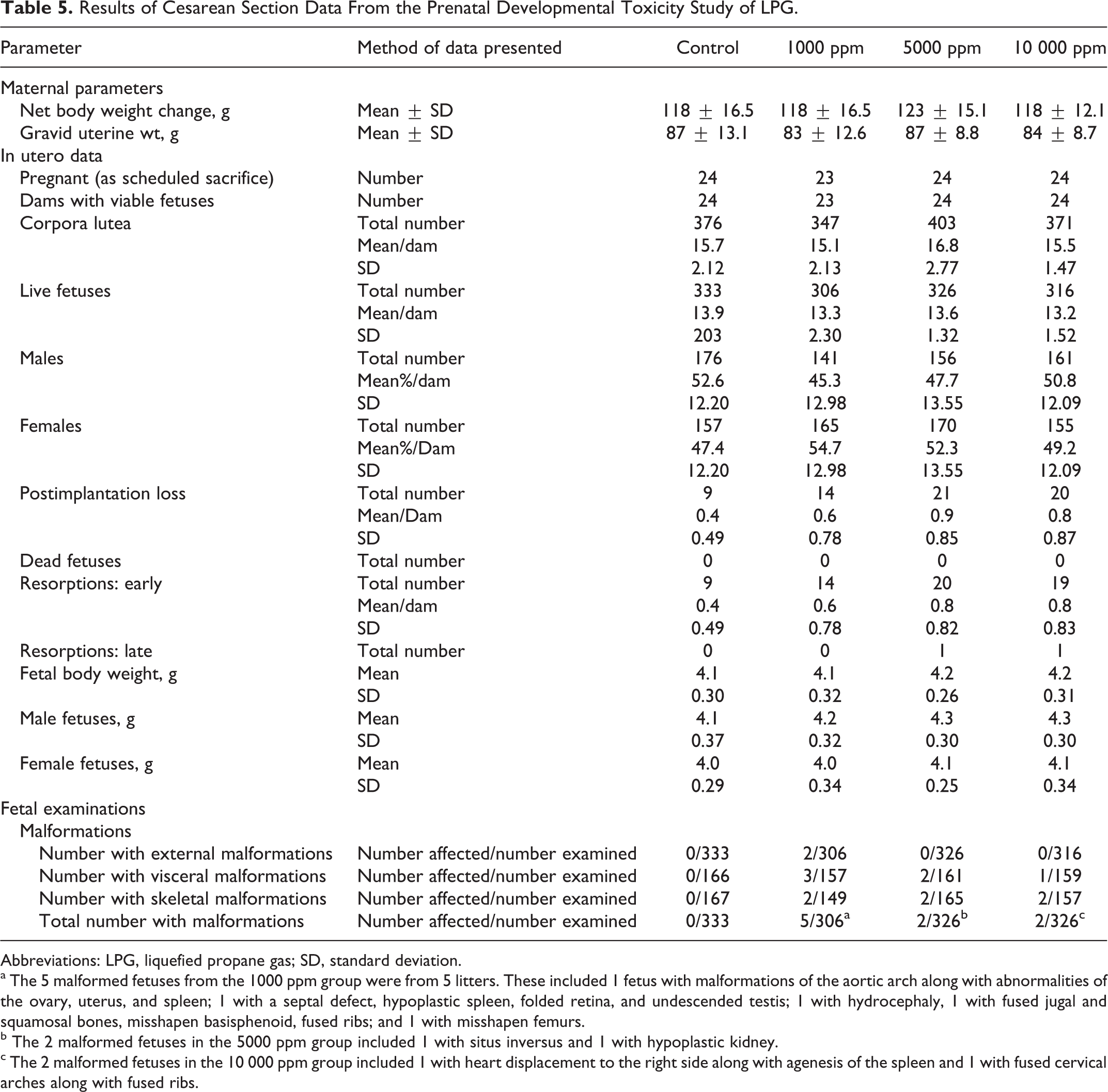
Abbreviations: LPG, liquefied propane gas; SD, standard deviation.
a The 5 malformed fetuses from the 1000 ppm group were from 5 litters. These included 1 fetus with malformations of the aortic arch along with abnormalities of the ovary, uterus, and spleen; 1 with a septal defect, hypoplastic spleen, folded retina, and undescended testis; 1 with hydrocephaly, 1 with fused jugal and squamosal bones, misshapen basisphenoid, fused ribs; and 1 with misshapen femurs.
b The 2 malformed fetuses in the 5000 ppm group included 1 with situs inversus and 1 with hypoplastic kidney.
c The 2 malformed fetuses in the 10 000 ppm group included 1 with heart displacement to the right side along with agenesis of the spleen and 1 with fused cervical arches along with fused ribs.
Assessment of the potential for micronucleus induction
The frequency of micronucleated erythrocytes was not significantly increased by LPG exposure, and there was not a significant decrease in the proportion of immature erythrocytes (Table 6). The positive control (cyclophosphamide, 40 mg/kg) significantly increased micronucleus frequency and significantly decreased the proportion of immature erythrocytes as expected. In summary, LPG exposure at levels up to 10 000 ppm did not cause chromosomal damage or induce bone marrow cell toxicity.
Summary of the Results of the Micronucleus Test of LPG.
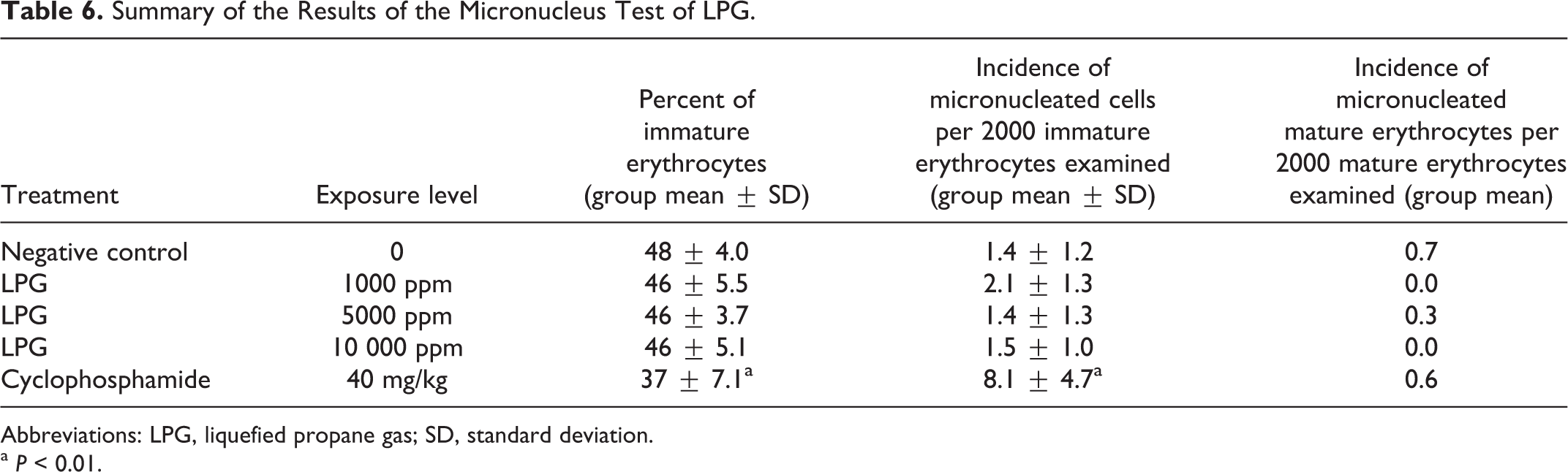
Abbreviations: LPG, liquefied propane gas; SD, standard deviation.
a
Ethane
Exposure levels
The mean (±standard deviation) analytically determined (IR) and nominal (by volume of gas consumed) concentration values were close to the target concentrations of 1600, 5000, and 16 000 ppm as summarized in Table 7.
Exposure Concentrations Maintained During the Repeated Dose Toxicity/ Reproductive Toxicity Screening Test of Ethane.
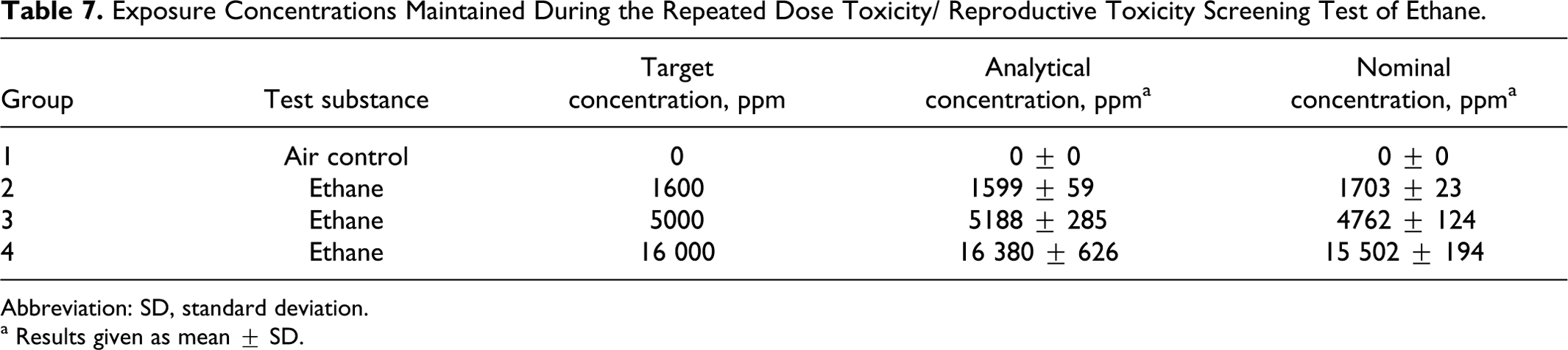
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
Assessment of systemic effects following repeated exposure
In the repeated inhalation toxicity portion of the ethane study, all rats survived to scheduled termination, and there were no consistent observations, other than transient red nasal discharge, during the exposure period. There were no treatment-related effects on body weight gain or food consumption (data not shown). The only statistically significant change in hematological parameters was a 15% increase in reticulocyte count in females from the 5000 and 16 000 ppm exposure groups. As there were no changes in any of the other hematological parameters in female rats and no changes in hematological parameters in exposed male rats, the increase in reticulocyte count in female rats was not considered to be toxicologically important. The only statistically significant finding in the clinical chemistry observations was a 2% increase in sodium concentration in high-dose males (data not shown). As there were no other changes and this difference was well within normal biological variability, it was not considered toxicologically important. The only statistically significant difference in organ weight was an increase in uterine weights in females from the 5000 ppm group. However, uterine weights in females from the 16 000 ppm group were not different from controls (organ weight data not shown). There were no postmortem observations, and there were no pathological observations suggestive of toxicological effects. There were no significant changes in functional observations or in motor activity tests. The overall NOAEC for the assessment of systemic toxicity by ethane was 16 000 ppm.
Reproductive toxicity assessment
In the assessment of the potential for developmental and/or reproductive effects, there were no mortalities, unusual clinical observations, or differences in body weights or body weight gain. The majority of the mated females became pregnant (Table 8). There were no significant differences in offspring born, percentage live born, survival to scheduled termination at PND 4, or offspring body weights. The overall NOAEC for the assessment of reproductive toxicity by ethane was 16 000 ppm.
Results of the Reproductive Toxicity Assessment of Ethane.
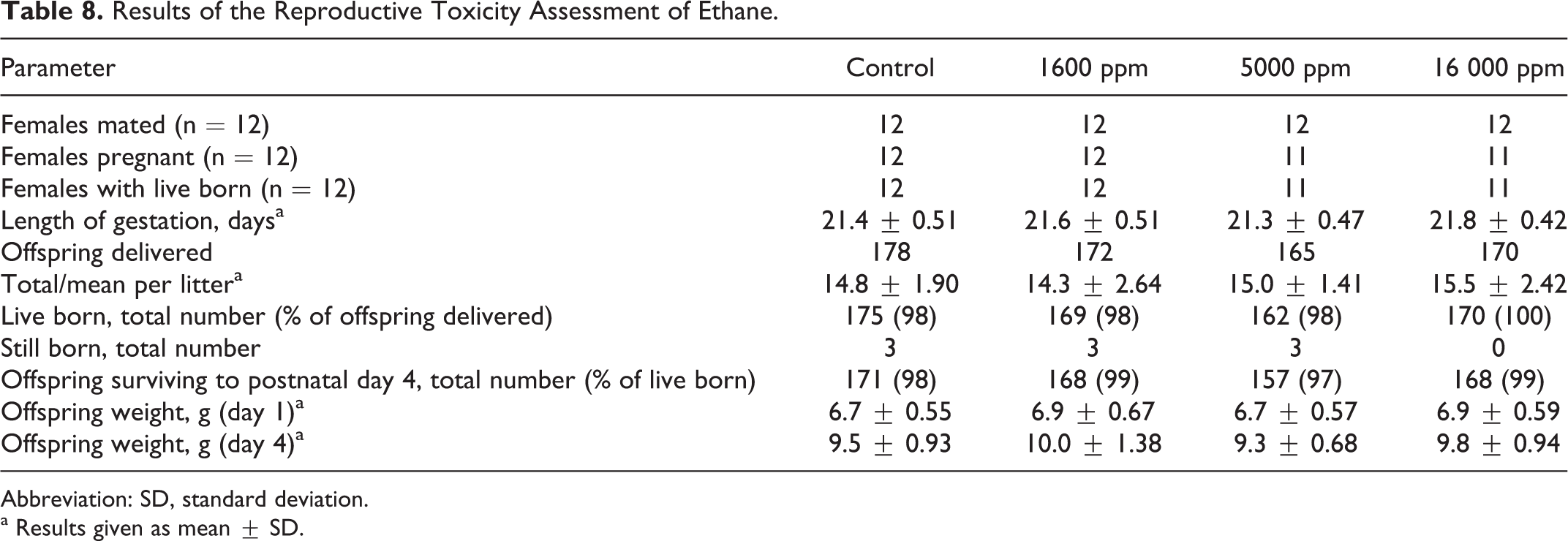
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
Propane
Exposure levels
The mean (±standard deviation) analytically determined (IR) and nominal (by volume of gas consumed) concentration values were close to the target concentrations of 1200, 4000, and 12 000 ppm as summarized in Table 9.
Exposure Concentrations Maintained During the Repeated dose Toxicity/Reproductive Toxicity Screening Test of Propane.
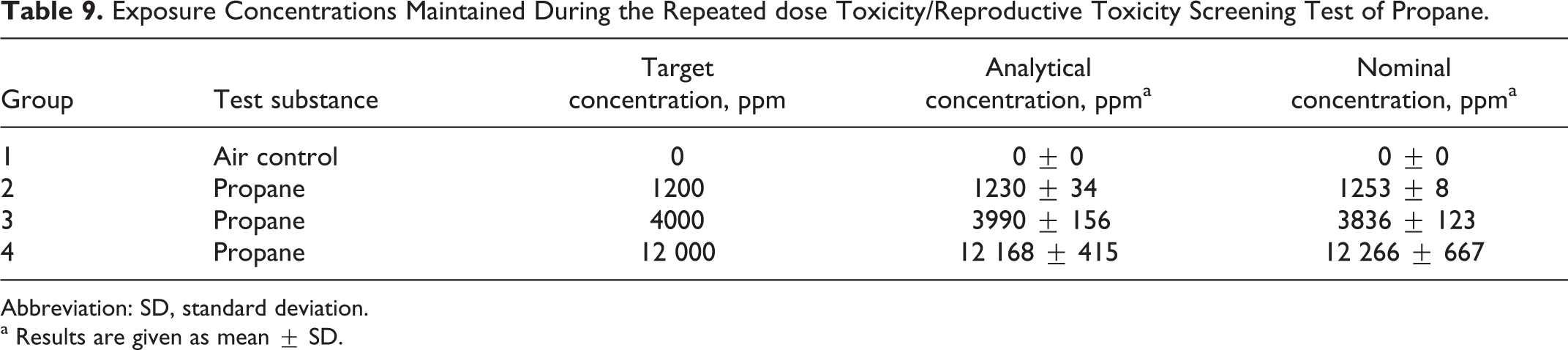
Abbreviation: SD, standard deviation.
a Results are given as mean ± SD.
Assessment of systemic effects following repeated exposure
In the repeated inhalation toxicity portion of the propane study, all rats survived to scheduled termination, and there were no remarkable observations, other than transient red nasal discharge, during the exposure period. The males in the 12 000 ppm exposure group had significantly lower body weights (control = 411 ± 27.8 g; 12 000 ppm group = 378 ± 24.3 g,
Weights (g) of Target and Reproductive Organs From Rats Exposed to Propane by Inhalation.a
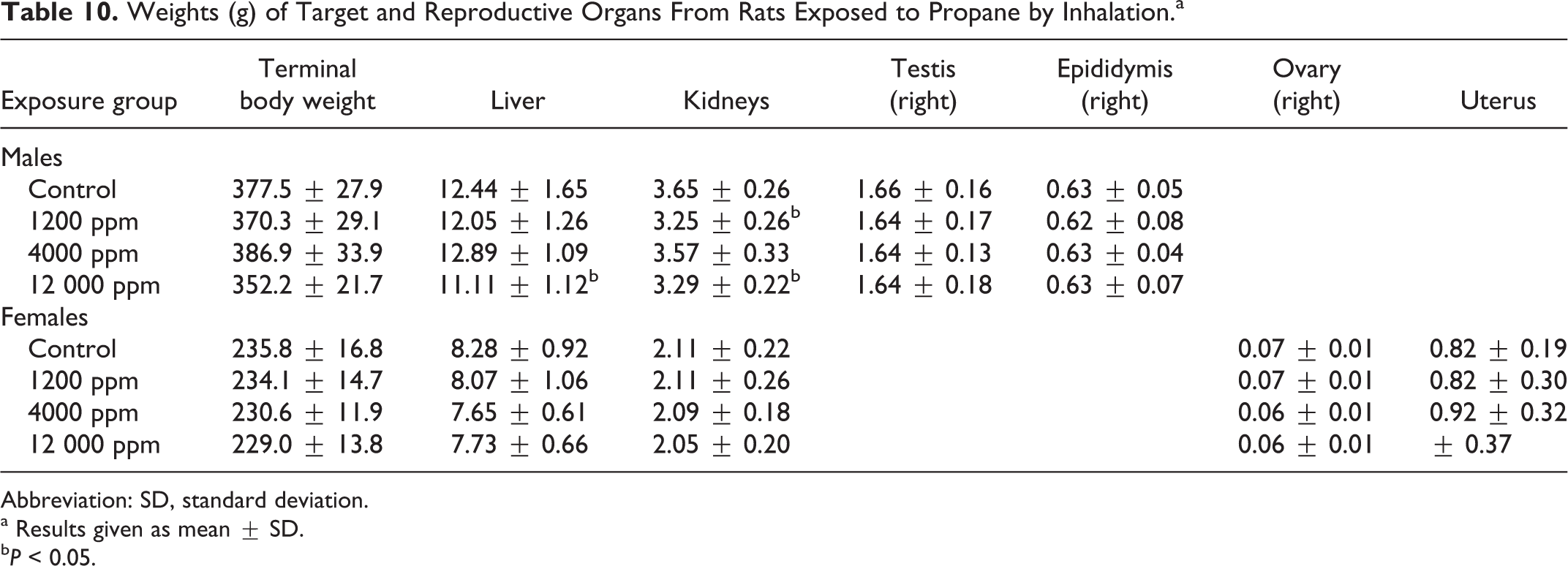
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
b
Reproductive toxicity assessment
In the assessment of the potential for developmental and/or reproductive effects, there were no mortalities, unusual clinical observations, or differences in body weights or body weight gain. The majority of the mated females became pregnant (Table 11). There were no exposure-related differences in any of the parturition parameters including preimplantation loss (as defined by the difference between the number of corpora lutea and the number of implantations detected in the uterus), postimplantation loss (as defined by the difference between the number of live pups born and the total number of implantations, thus including any stillborn pups), the total number of pups delivered, the number of pups dying, the viability (PND 4 survival) index, the pup sex ratio, and the number of live pups/litter when compared to the control group. Statistically significant decreases in the number of live-born pups and corresponding increases in the number of stillborn pups in the 4000 and 12 000 ppm groups were attributable to the complete loss of 1 litter in each group. These losses were preceded by severely reduced body weight gain in the last week of gestation for the respective dams. As there was no excess in mortality in any of the other litters in these groups, the losses of these 2 litters were considered incidental and not related to treatment. An overall NOAEC of 12 000 ppm for propane was determined for the fertility and reproductive toxicity end points in this study.
Results of the Reproductive Toxicity Assessment of Propane.

Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
b
c
n-butane
Exposure levels
The mean (±standard deviation) analytically determined (IR) and nominal (by volume of gas consumed) concentration values were close to the target concentrations of 900, 3000, and 9000 ppm as summarized in Table 12.
Exposure Concentrations Maintained During the Repeated Dose Toxicity/Reproductive Toxicity Screening Test of n-Butane.
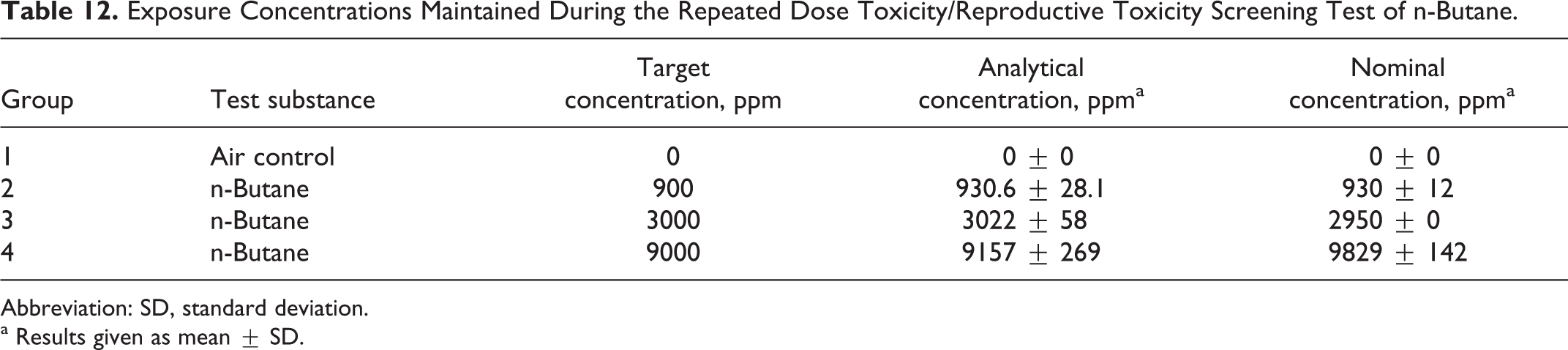
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
Assessment of systemic effects following repeated exposure
In the repeated dose segment of the butane study, there were no mortalities, no exposure-related differences in body weight, body weight gain, or food consumption (data not shown). There were no toxicologically important differences in hematology or clinical chemistry parameters (data not shown). There were no toxicologically significant differences in organ weight data; there were no notable observations during the postmortem examination; and no exposure-related differences were found during the histological evaluation (data not shown). In the neurological assessment, there were no differences in functional observations or motor activity (data not shown). The overall NOAEC for systemic effects of n-butane was 9000 ppm.
Reproductive toxicity assessment
In the assessment of the potential for developmental and/or reproductive effects, there were no mortalities, no unusual clinical observations, and no differences in body weights or body weight gain. There were no significant effects on mating (Table 13), no effects on offspring survival or body weights (Table 13), and no evidence of gross malformations (data not shown). The only statistically significant findings were on offspring delivered and live offspring per litter, but for these parameters, statistical significance was found only in the lowest exposure group, and, in both cases, the measurements in the exposed group were above the control values. As there is no obvious reason why exposure to n-butane would improve reproductive performance in rats, these differences were considered to be incidental. The overall NOAEC for fertility and reproductive effects of n-butane was 9000 ppm.
Results of the Reproductive Toxicity Assessment of n-Butane.
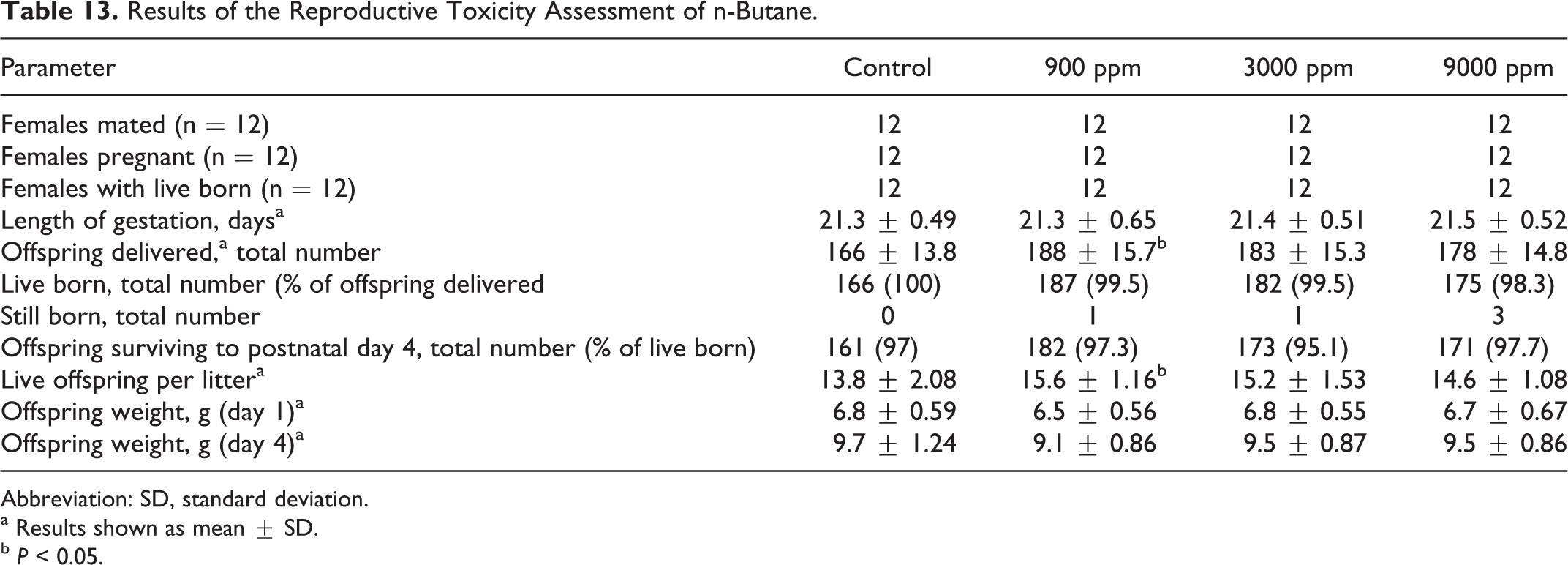
Abbreviation: SD, standard deviation.
a Results shown as mean ± SD.
b
Isobutane
Exposure levels
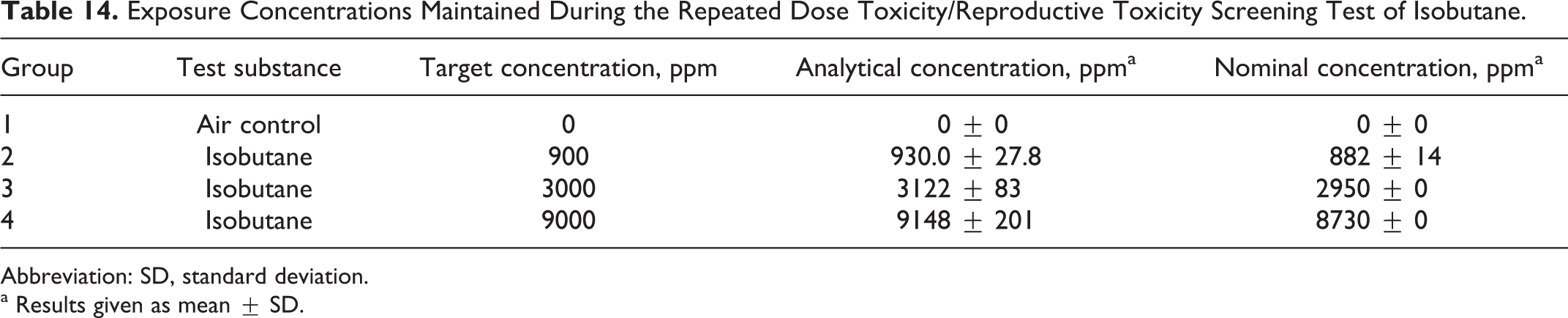
The mean (±standard deviation) analytically determined (IR) and nominal (by volume of gas consumed) concentration values were close to the target concentrations of 900, 3000, and 9000 ppm as summarized in Table 14.
Exposure Concentrations Maintained During the Repeated Dose Toxicity/Reproductive Toxicity Screening Test of Isobutane.
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
Assessment of systemic effects following repeated exposure
In the repeated dose segment of the isobutane study, there were no mortalities, no exposure-related differences in body weight, body weight gain, or food consumption (data not shown). There were some statistically significant changes in hematological parameters. There were significant increases in hemoglobin (control = 15.6 ± 0.6, 9000 ppm group = 16.1 ± 0.6) and mean corpuscular hemoglobin concentration (control =33.7 ± 0.5, 9000 ppm group = 34.2 ± 0.5) and reductions in platelet counts (control = 898 ± 214, 3000 ppm group = 721 ± 103, 9000 ppm group = 767 ± 161) in the males. But, among the females, the only significant differences were in frequency of absolute monocytes (control = 0.25 ± 0.12, 3000 ppm = 0.17 ± 0.07, 9000 ppm = 0.17 ± 0.17). As indicated, these differences were not consistent between the sexes, not associated with other changes in hematological parameters, and, for the most part, within normal physiological ranges. Accordingly these differences were not considered toxicologically important. The only statistically significant differences in clinical chemistry parameters were a slight increase in sodium levels in males (control = 147 ± 1.6, 9000 ppm = 149 ± 1.5) and a slight decrease in phosphorus levels in females (control = 9.0 ± 1.5, 9000 ppm = 8.0 ± 0.6). These differences were within the normal physiological ranges for these parameters and were not considered toxicologically important. There were no toxicologically significant differences in organ weight data; there were no notable observations during the postmortem examination; and no exposure-related differences were found during the histological evaluation (data not shown). In the neurological assessment, there were no differences in functional observations or motor activity (data not shown). The overall NOAEC for systemic effects of isobutane was 9000 ppm.
Reproductive toxicity assessment
In the assessment of the potential for developmental and/or reproductive effects, there were no mortalities, unusual clinical observations, or differences in body weights or body weight gain. However, only 9 of the 12 high-dose group females became pregnant. Although this difference was not statistically significant, it was low by comparison to the historical experience in the laboratory (Table 15). Accordingly, this outcome was considered to have been toxicologically important, and, for purposes of hazard assessment, 3000 ppm isobutane was judged to have been the NOAEC for fertility. However, as there were no differences in offspring/litter, survival of offspring to scheduled termination, or body weight gain (Table 15), and no evidence of gross malformations (data not shown), it was concluded that isobutane had no apparent effects on development at 9000 ppm.
Results of the Reproductive Toxicity Assessment of Isobutane.
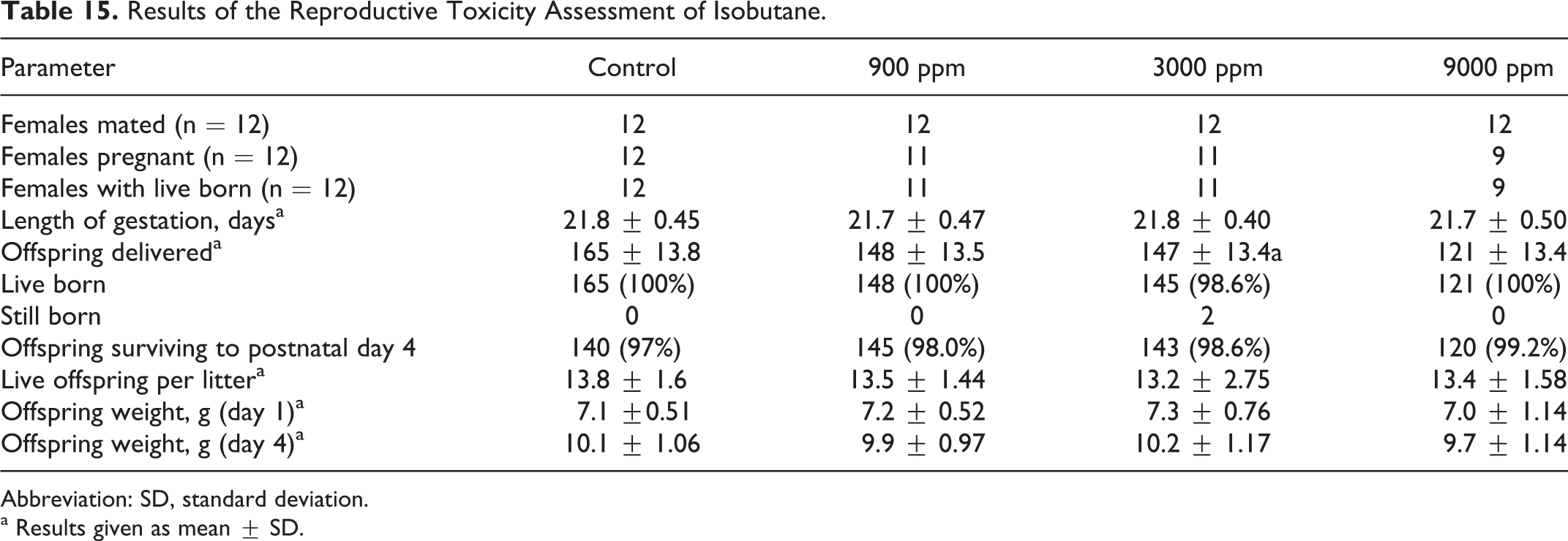
Abbreviation: SD, standard deviation.
a Results given as mean ± SD.
Discussion
Liquefied Propane Gas
As previously described, LPG was tested for systemic and developmental toxicity at levels up to 10 000 ppm following OECD 413 and 414 guidelines. There were no toxicologically important effects in either of these studies, and the highest dose tested in each study (10 000 ppm) was judged to have been the NOAEC for the respective end points. Additionally, the micronucleus test (OECD 474) provided evidence that LPG does not induce chromosomal effects under in vivo conditions.
Ethane, Propane, n-butane, and Isobutane
All 4 of these substances were tested for repeated dose and reproductive effects following OECD 422 guidelines for the repeated dose/reproductive toxicity screening test design. The potential for neurological effects was also assessed. As indicated in the results section, none of the measures of subchronic toxicity or neurological effects was significantly different from the corresponding control value. Similarly, there were no noteworthy findings identified during the pathological investigations. Accordingly, the overall NOAECs for repeated exposure and neurological effects studies were the highest concentrations tested, ranging from 9000 to 16 000 ppm. There were also no effects in the assessments of reproductive toxicity of ethane, propane, or butane, and the NOAECs for these substances were the highest concentrations tested. However, in the isobutane study, there was a reduction in fertility in the high-exposure group which, although not statistically significant, was outside the historical control range. Accordingly, 3000 ppm was taken as the NOAEC for reproductive effects of isobutane, and for the purposes of further analysis, this value was taken as the “worst case” NOAEC for the systemic and reproductive effects of C1 to C4 alkanes. It should be noted that a number of structurally related substances including ethane, propane, butane (present studies) as well as 2-methyl butane 37 did not produce reproductive effects. Further, there were no reproductive effects in a 2-generation reproductive toxicity test of gasoline vapor in which the principal constituents were butane and pentane isomers. 38 Nevertheless, the 3000 ppm NOAEC from the reproductive toxicity screening test was taken forward as an overall no effect level for all effects for C1 to C4 petroleum gas constituents as it represented a conservative basis for risk evaluation.
In summary, it was determined that the NOAECs for acute inhalation toxicity were >9000 ppm, as all animals survived repeated exposures at that level. The NOAECs for repeated inhalation toxicity were also judged to be >9000 ppm as repeated exposure at that level did not produce toxicological signs or symptoms, did not produce any toxicologically important histological changes, and did not affect neurological parameters. Based on the study of LPG and supporting data from the screening tests of the other low-molecular-weight alkanes, the NOAEC for developmental toxicity was judged to be >9000 ppm. The overall NOAEC for potential reproductive effects of C1 to C4 hydrocarbon gas constituents was judged to be 3000 ppm (7125 mg/m3) based on a small and not statistically significant reduction in fertility in the high-exposure group in the isobutane study. Finally, based on the micronucleus test of LPG in which no statistically significant differences were found along with published data indicating that low-molecular-weight alkanes are inactive in
Calculated Toxicological Effect Levels for Complex Petroleum Hydrocarbon Gases Based on Compositional Information
One of the challenges of the HPV program for the petroleum industry was to characterize the hazards of complex hydrocarbon substances. Within the category of hydrocarbon gases, there were 106 petroleum hydrocarbon gases as identified by CAS numbers, the majority of which were complex and comprised C1 to C4 alkane gases in various amounts, and, in some cases and depending on the specific methods of production, other constituents including C5 to C6 aliphatic hydrocarbons, benzene, and 1,3-butadiene. A method was developed by which the hazards of any complex petroleum hydrocarbon gas could be estimated from its composition using the results of animal tests of the individual constituents. For purposes of this calculation, the constituents of the streams were divided into 7 groups; the hydrocarbon constituents, C1 to C4 hydrocarbons (using predominantly data developed as part of this program), and the C5 to C6 aliphatic hydrocarbons (using predominantly literature data), the more hazardous constituents, benzene and 1,3-butadiene (for which predominantly literature data was used), and the inorganic gases; CO2, H2, N2 which, for purposes of this evaluation, were assumed to be simple asphyxiants and to have hazard properties similar to those of the C1 to C4 gases. It should be noted that the more hazardous inorganic gases such as hydrogen sulfide and ammonia are constituents of the refinery gases but as they are not normally present in petroleum gases at more than trace levels, they do not need to be accounted for in the calculation.
The expected no adverse effect concentrations of the toxicology tests related to the various HPV end points (acute toxicity, repeated dose toxicity, developmental toxicity, and reproductive toxicity) can then be estimated for any of the complex hydrocarbon gases using the following relationship:
The NOAEC values for the various end points are shown in Table 16.
No Observed Adverse Effect Concentrations for Petroleum Gas Constituents.

Abbreviation: LC50, lethal concentration 50.
a The values for C1 to C4 alkanes are based on the results of the present studies in which repeated exposures at 9000 ppm (21 375 mg/m3) had limited effects. The value for reproductive toxicity is based on the reproductive toxicity screening test of isobutane, using 3000 ppm (7125 mg/m3) as a no observed effect concentration for all effects.
b The values for C5 to C6 alkanes are based on the results of repeated exposure studies to n-pentane, 39,40 commercial hexane, 41–43 and isopentane. 37 Acute toxicity per se was not assessed but LC50 values could not be lower than the no effect levels in repeated exposure studies.
c Critical values for 1,3-butadiene are taken from Owen and Glaister (repeated dose toxicity), 44 Morissey et al (developmental toxicity), 45 and WIL Research (reproductive toxicity). 46
d Critical values for benzene are taken from Green et al. (repeated dose toxicity), 47,48 Kuna and Kapp 49 and Coate et al 50 (developmental toxicity), and Ward et al (reproductive toxicity). 51
For illustration purposes, nominal concentration ranges were assigned to the gases based on the information in the CAS descriptions, analytical information where available, and scientific judgment. Based on this analysis, benzene levels were expected to range from 0% to 1% and butadiene levels from 0% to 4%. As an example of a gas containing 1% benzene, consider “tail gas (petroleum), gas recovery plant deethanizer” (CAS number 68308-05-4) for which the nominal concentration ranges are C1 to C4 = 26% to 85%, C5 to C6 = 15% to 73%, and benzene = 0% to 1%. For the purposes of calculation, assume the maximum concentrations of the constituents with the lowest NOAEC values and assign the remainder to those with the highest, that is, set the C1 to C4 aliphatic constituents to 85% and benzene to 1% with the remainder (14%) being C5 to C6 constituents.
The calculated NOAEC for repeated dose toxicity for this gas would be: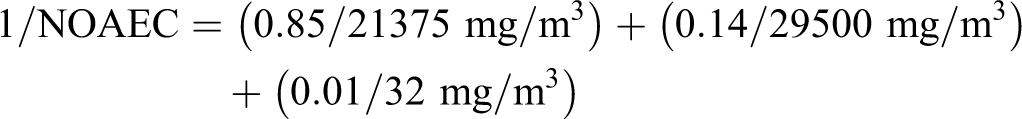
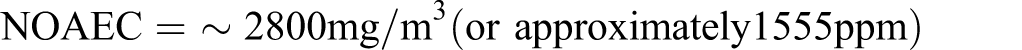
To evaluate the maximal impact of butadiene, consider “gases (petroleum), catalytic-cracked overheads” (CAS number 68409-99-4) for which the nominal concentration ranges are C1 to C4 = 65% to 93%, C5 to C6 = 7% to 31%, hydrogen = 0% to 3%, carbon dioxide = 0% to 1%, and butadiene = 0.5% to 4%. Assuming C1 to C4 = 65%, C5 to C6 = 31%, and butadiene = 4% (with hydrogen and carbon dioxide being subsumed in the C1 to C4 value), the calculated NOAEC for developmental toxicity would be: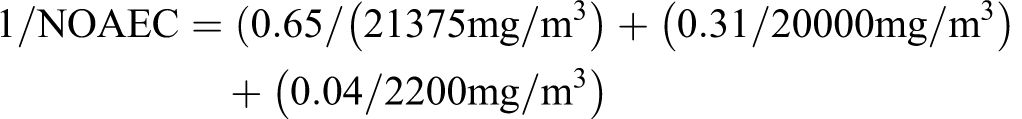
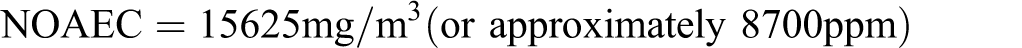
Based on the equations mentioned earlier, it is apparent that the petroleum hydrocarbon gases pose very limited acute toxic hazards. Rather, it seems more likely that in situations involving high exposures, the potential for fire probably represents a greater concern as for many of these constituents the lower flammability limits are below 20 000 ppm.
In situations involving repeated exposures, the hazard of any of the petroleum hydrocarbon gases is related to the potential for the streams to contain benzene and/or 1,3-butadiene. For petroleum hydrocarbon gases that do not contain appreciable amounts of benzene or 1,3-butadiene, the worst case situation is the potential for reproductive toxicity for which the NOAEC is 3000 ppm (7125 mg/m3). If benzene and/or 1,3-butadiene are present in these streams, the toxicity to animals can be predicted but may have little practical significance. The occupational control measures for streams containing benzene and 1,3-butadiene are related to the need to control exposures to levels below 1 ppm, the occupational exposure levels for these constituents. The occupational exposure levels for benzene and 1,3-butadiene are related to the potential for these substances to cause cancer in humans, not the results of toxicology studies in animals.
In conclusion, a method is described by which the potential for acute, repeated dose, developmental, and/or reproductive effects of complex petroleum hydrocarbon gases can be calculated based on the types and levels of constituents in complex gas streams. Two general conclusions were evident from these calculations:
First, it is evident from the data presented that the C1 to C4 alkane hydrocarbon gas constituents as well as the majority of C5 and C6 aliphatic constituents have limited potential to produce any of the assessed toxicological effects with worst case NOAEC values in the range of 3000 to 16 000 ppm. Benzene, on the other hand, has the potential to produce a number of toxicologically important effects at much lower levels. Accordingly, streams without benzene (and to a lesser extent 1,3-butadiene) will be relatively nonhazardous, but exposure to streams that contain benzene and/or 1,3-butadiene needs to be controlled to assure that exposures to these constituents do not exceed their regulatory values.
Second, the only other notable result in these studies was the selection of a NOAEC of 3000 ppm (7125 mg/m3) for isobutane based on the limited evidence of reduced fertility. As this NOAEC is lower than NOAEC values for other gas constituents and for other end points, the lowest calculated NOAEC values are likely to be those associated with reproductive toxicity as an end point and those values, ultimately, are likely to be the critical values for risk assessment purposes.
Footnotes
Appendix A
Acknowledgments
The authors would like to thank Chris Sexsmith for quality assurance support and Lynn Bennett for assistance in manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors of this article are employed by companies that manufacture petroleum products and contractors working on behalf of the petroleum industry HPV program.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was sponsored and funded by the Petroleum HPV Testing Group (PHPVTG), an unincorporated group of manufacturers affiliated by contractual obligation to fund a voluntary data disclosure and toxicity testing program on certain petroleum-related chemical substances in response to EPA’s HPV Challenge Program. The American Petroleum Institute (API) manages the Petroleum HPV Testing Group’s (PHPVTG’s) activities.
