Abstract
Objectives
We investigated the effects of repeated intra-articular injections of liquid sevoflurane to articular structures, as this inhalational anesthetic is being repurposed as an antimicrobial agent, which would make sevoflurane a novel alternative for the treatment of septic arthritis.
Methods
The left knees of nine Sprague-Dawley rats were injected with 150 μL of liquid sevoflurane for five consecutive days, whereas the right knees were injected with the same volume of saline to serve as controls. Animals were examined daily for clinical signs of local and systemic toxic effects attributable to sevoflurane. Rats were euthanized in groups of three at days 7, 14, and 35 after the first injection, and left and right knees were sent for histological assessment.
Results
Local signs on knees consisted of transient bilateral scabs, and an unexpected subcutaneous emphysema affecting only left knees, which was attributed to sevoflurane. No rat presented with limp, and animal welfare was good during the study period. Two out of the three left knees from rats sacrificed at day 7 showed mild histological changes, specifically a mild infiltration of lymphocytes. All other seven left knees as well as all nine right knees were completely normal at histological examination.
Conclusion
We concluded that repeated intra-articular injections of sevoflurane seemed safe for articular structures in noninfected knees. Further studies focused on the safety of intra-articular sevoflurane in infected knees, as well as on its effectiveness for the treatment of septic arthritis are warranted.
Introduction
The incidence of septic arthritis is growing worldwide. Initially, treatment includes surgery and empirical antibiotics, and treatment failure could have deleterious consequences, such as complete damage and destruction of the joint, or even patient death.1,2 However, the incidence of septic arthritis caused by resistant organisms is also growing; thus, increasing the risk of empirical antibiotic treatment failure. 3 Therefore, new therapeutic alternatives need to be developed.
Haloethers are a family of well-known inhalational anesthetic drugs, which are halogenated derivatives obtained from diethyl ether. These agents are available as volatile liquids, allowing administration via inhalation using anesthetic machines. Furthermore, haloethers have been classified as nonconventional antibiotics, owing to a certain degree of
Currently, sevoflurane is employed worldwide for inhalational anesthesia, and after more than 20 years of application, its efficacy and safety are well established in this setting. 13 Over the last 10 years, sevoflurane has been repurposed as a topical analgesic for the treatment of painful chronic wounds.14-17 As a topical analgesic, the simple method of administration involves the irrigation of the wound bed with liquid sevoflurane, which provides a rapid, intense, and long-lasting analgesic effect. 18 This new technique seems to be safe to the skin, as no histological local adverse effects associated with topical use have been reported.
In addition to the analgesic effect, an antimicrobial effect has been reported in infected wounds.19-27 Interestingly, some of the infections successfully treated with topical sevoflurane were caused by bacteria resistant to conventional antibiotics, such as methicillin resistant
In addition to open wounds applications, the liquid formulation allows sevoflurane to be injected into open infected cavities through the fistulous tract which connects the cavity with the skin.19,20 Thus, a further step in clinical application as an antimicrobial agent would be the injection of sevoflurane through a needle into closed infected cavities. This eventual treatment modality could be useful for treating infectious arthritis, similar to the previous administration of ether.10,12 However, there is currently no information available regarding the safety of intra-articular liquid sevoflurane injections.
The general objective of the present study was to determine the safety of injecting liquid sevoflurane into the knee of rodents. The main objective was to assess histological changes caused by repeated injections of liquid sevoflurane into the knee of Sprague-Dawley rats. In terms of secondary objectives, we assessed the clinical effects of sevoflurane, both local effects on the knees and the systemic effects in experimental animals.
Materials and methods
Experimental animals and husbandry
Adult male and female Sprague-Dawley rats were obtained from colonies established at the animal facility. Two pairs of founders were purchased from a commercial breeder (Charles River Laboratories, Barcelona, Spain).
As only one dose of sevoflurane was tested, the sample size was intended to be of 10 animals, five of each gender, which followed the recommendation stated in OECD Test Guidelines for Chemicals. The final sample consisted of five males, aged 35–41 weeks, weighing 674–873 g, and four females, aged 41–45 weeks and weighing 270–406 g. No inclusion or exclusion criteria were applied. These nine animals were randomly housed divided by gender in groups of two or three animals per cage in climate-controlled, individually ventilated, type 1500U cages (Tecniplast, Italy), containing aspen chip bedding material (Tapvei, 2HK10 Kg, Estonia), with a 12 h light/dark schedule. Animals were maintained under controlled ambient conditions (temperature: 22 ± 2°C; relative humidity: 30%−70%) and had unlimited access to water and standard rat food (Envigo Laboratories, Barcelona, Spain).
Each animal served as its own control. Histological evaluation of the left and right joints under study was performed after euthanizing the nine rats in groups of three, at three different time points; the day of the first injection was deemed day 0. On day 7, three randomly selected animals were euthanized (Group #1) to assess for any eventual histological damage caused by sevoflurane. The same selection procedure was repeated on day 14 (Group #2) to assess for reversibility of changes eventually found at day 7, and the remaining three animals were euthanized on day 35 (Group #3) to assess for the presence of any damage, which should be considered as permanent.
Intra-articular injection
The animals were anesthetized as described below and placed in a dorsal recumbent position on a warming blanket. The hind legs were shaved, and the knees were sterilized with chlorhexidine, as all injections were performed under sterile conditions.
The puncture point was localized between the upper third of the patella and the lateral part of the knee. The needle (30 G ½” BD Microlance TM) was inserted perpendicular to the leg axis and parallel to the floor until contact with the joint, which was identified by loss of resistance after breaching the articular capsule. 28
The left knees (cases) were injected with 150 μL of sevoflurane (Sevorane©, 250 mL bottles containing pure sevoflurane; AbbVie, Camproverde, Italy), with the quantity selected to prevent rupture of the capsule owing to an excess of liquid. 28 The right knees (controls) were injected with 150 μL of saline.
The knees were injected daily for five consecutive days. All injections were performed in the morning by the same experienced researcher to avoid possible bias owing to personal differences while performing the technique.
Clinical assessment
Clinical variables were assessed on days 0, 1, 2, 3, 4, and 7 for the three groups, on day 14 for the remaining Groups #2 and #3, and on days 21, 28, and 35 for the remaining Group #3.
Animals were weighed and examined for any discernible systemic signs suggesting adverse effects induced by sevoflurane, namely, drowsiness; both knees were examined for any local signs suggestive of arthritis, such as limp or itching. The mediolateral diameter of 40º-flexed knees (in millimeters) was measured using a calibrated caliper.
Histological evaluation
The animals were euthanized as described below, and both knee joints were fixed with 4% formaldehyde and stored. Later, the samples were decalcified for 10 days in a solution containing 15% formic acid, with a longitudinal section performed on day 7 to hasten decalcification. Subsequently, the samples were embedded in paraffin and 3-μm thick histological sections were cut and stained with hematoxylin-eosin.
Two pathologists who were blinded to the injections performed examined histological samples, assessing any possible damage in the synovium, cartilage, and bone.
According to the literature,29,30 a 5-point scale was used to grade synovial acute inflammatory changes (0 = no inflammation; 1 = minimal inflammation, mild congestion, and edema; 2 = mild inflammation, erosion of the joint surface, congestion, and edema, and a small number of neutrophils; 3 = moderate inflammation, presence of neutrophils, and macrophages, and synoviocyte hyperplasia; 4 = severe inflammation, presence of neutrophils and macrophages, synoviocyte hyperplasia, and fibrin exudation) and hemorrhage (0 = no hemorrhage; 1 = minimal; 2 = moderate; 3 = severe; 4 = very severe).
Animal welfare, anesthesia, and euthanasia
All experiments were performed in accordance with the European Directive (2010/63/UE) and Spanish law (Real Decreto 53/2013) for the protection of animals used for scientific purposes. The experimental protocols were approved by the Animal Experimentation Ethics Committee of Castilla-La Mancha University (PR-2016-11-17). As recommended by the Federation for Laboratory Animal Science Associations, rats at our animal facility were tested periodically to ensure that the colony remained free of pathogens.
A veterinary who was aware of all experimental phases also supervised the daily assessment of animal welfare. The clinical assessment of welfare was based on the general appearance of the animal, body posture, mobility (limp), body weight, coat and grooming, ocular appearance, indications of self-harm (joint knee), and behavior and sociability. The humane endpoints considered were weight loss > 20%, severe lethargy, markedly rough coat/piloerection, or excessive chromodacryorrhea.
All intra-articular injections were performed under general anesthesia, which was induced using a mixture of 5% vol of sevoflurane in 2 L/min oxygen for 4–5 min, followed by maintenance using 2–3% vol of sevoflurane in 1 L/min oxygen.
After completion of the sequential study periods, the animals were euthanized in a CO2 chamber filled with CO2 at a low rate, specifically a 30% chamber volume per minute.
Statistical analysis
As the number of animals was small, the data were considered to be non-normally distributed. Thus, quantitative variables are described as median and absolute range, and nonparametric tests were used for comparisons. The Wilcoxon test was used to compare the baseline weight with the weight for each of the study days. The Mann–Whitney U test was used to compare the percentage of increment in the diameter between the left and the right knees.
Categorical clinical and histological variables are shown as absolute frequency and percentage, and Fisher’s exact test was used to assess associations between them and knees.
SPSS Statistic v20 package was used for statistical analysis. A standard bilateral
All relevant data are presented in the manuscript and supplementary material. The ARRIVE guidelines 2.0 author checklist is also presented in supplementary material.
Results
Baseline characteristics of the nine animals.

R: rat. M: male. F: female.
Systemic signs and animal welfare
In general, the welfare status of animals was good. In addition, the pre-established humane endpoint criteria were not met by any animal.
Weight during the experimental period.
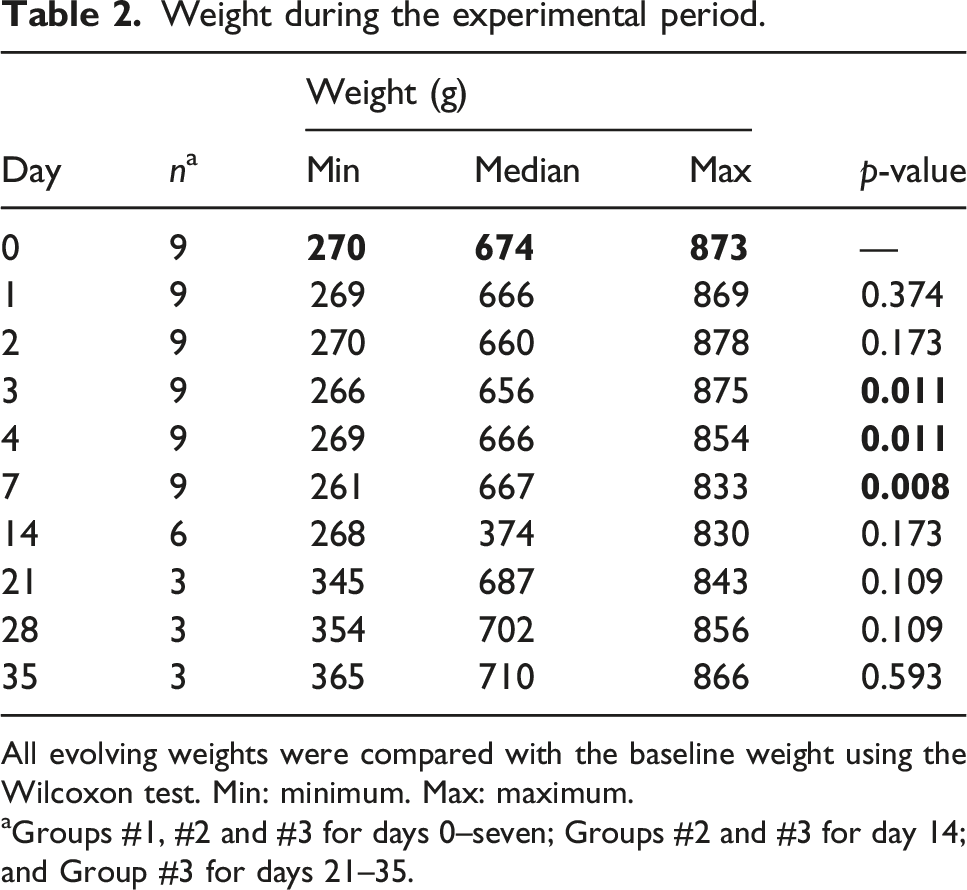
All evolving weights were compared with the baseline weight using the Wilcoxon test. Min: minimum. Max: maximum.
aGroups #1, #2 and #3 for days 0–seven; Groups #2 and #3 for day 14; and Group #3 for days 21–35.
No animal developed a limp owing to the intra-articular injections, and drowsiness caused by sevoflurane was not suspected.
Local signs on injected knees
Diameter of the left (cases) and right (controls) knees during the experimental period.

Mann–Whitney U test for all
aGroups #1, #2 and #3 for days 0–seven; Groups #2 and #3 for day 14; and Group #3 for days 21–35.
Absolute frequency of scabs in left (cases) and right (controls) knees during the experimental period.

Fisher’s exact test for all
aGroups #1, #2 and #3 for days 0–7; Groups #2 and #3 for day 14; and Group #3 for days 21–35.
Absolute frequency of subcutaneous emphysema in left (cases) and right (controls) knees during the experimental period.
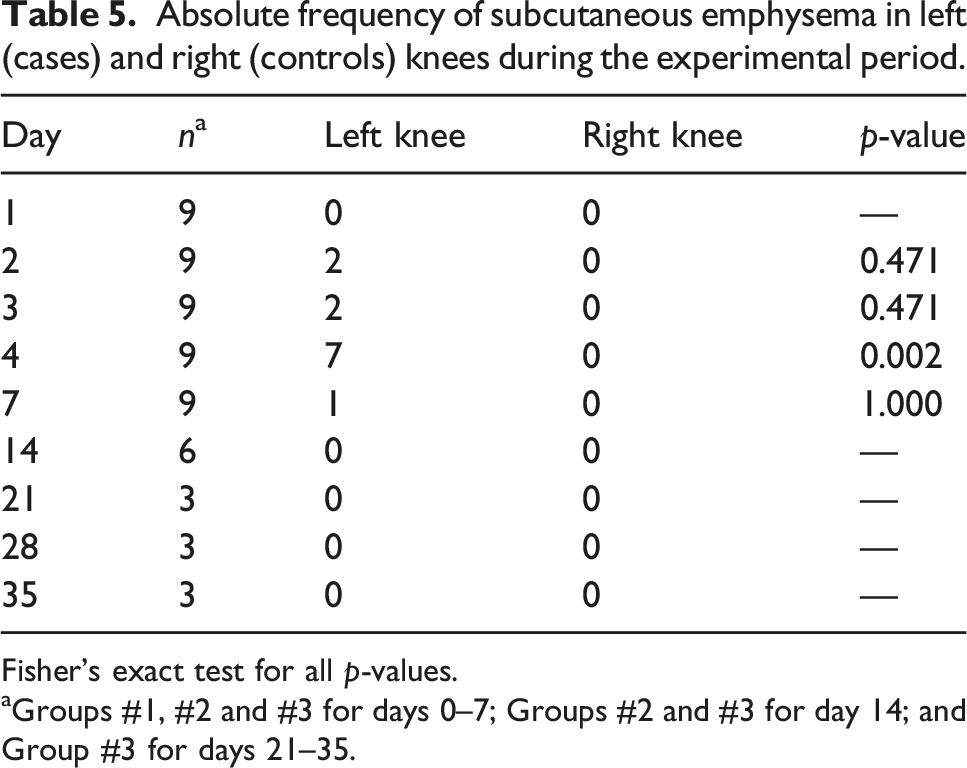
Fisher’s exact test for all
aGroups #1, #2 and #3 for days 0–7; Groups #2 and #3 for day 14; and Group #3 for days 21–35.
Histological findings
Histological findings were extremely scarce as most joints appeared strictly normal at examination, specifically all right knees, all left knees from Groups #2 and #3, and one left knee from Group #1 ( Microphotographs showing histological findings in left and right knees after five consecutive daily injections of 150 μL of sevoflurane or saline. 
These mild changes were limited to mild hyperplasia of the synovium, which was also marginally infiltrated by few lymphocytes, while neutrophils were not observed (Figure 1 upper and left panel); thus, acute inflammatory changes were graded as one on the 5-point scale. Articular cartilage and the subjacent bone showed no changes suggestive of erosion, calcification, hypertrophy, or hemorrhage.
Discussion
In the present study, we observed that intra-articular injections of 150 μL of sevoflurane for five consecutive days caused no permanent damage to the joint structures, as confirmed by the absence of permanent histological changes.
A few histological changes were observed in two rats which were euthanized 3 days after the completion of sevoflurane injections. Specifically, the synovial membrane showed mild hyperplasia, yet with an absence of neutrophils; therefore, the potential for activating an acute inflammatory response and damage to the synovial membrane was markedly low. A marginal infiltration of lymphocytes was observed. These cells possess a lower potential to damage the synovium; moreover, this is a normal nonspecific synovial response to external insults. 31
Our findings cannot be directly compared with other reports in literature as this is the first report on the effects on intra-articular sevoflurane administration. However, some available information indirectly supports the lack of sevoflurane toxicity on articular structures.
Previously, injections of diethyl ether, the parent compound of sevoflurane, into the joint of rabbits caused a mild transient effusion, in which endothelial cells predominated at early stages and were then substituted by neutrophils. However, this type of effusion did not last beyond 15 min, and the joints appeared normal after a week. 32 It seems reasonable to assume that neutrophils were removed from the joint and that the inflammatory cascade was not activated.
In other preclinical study where rats received an intraperitoneal injection of isoflurane, methoxyflurane, and sevoflurane, the latter drug was the only agent that did not induce severe damage to the peritoneum and intraperitoneal organs, even when lethal doses were employed. 33 These differences in toxicity for drugs from the same family could be explained by differences in their chemical structures. Early compounds such as methoxyflurane and isoflurane (as well as enflurane) are fluorinated and chlorinated, whereas more recent compounds such as sevoflurane (and desflurane) lack chlorine in their chemical structure, with ether lacking both chlorine and fluorine. 34
Over the last 10 years, sevoflurane has been repurposed as a topical analgesic for painful wounds, with accumulating clinical data supporting this new indication. To date, there is no evidence regarding the possible harmful effects of sevoflurane on the skin, 18 even after daily administration for 12 months. 35 Furthermore, sevoflurane has recently been reported as an analgesic for treating painful conditions affecting rectal and urethral mucosae, specifically a postoperative urethritis 36 and an anal fissure. 37 The patient with urethritis received only one dose of sevoflurane, which was insufficient to conclusively establish safety. However, the patient suffering from a painful anal fissure was successfully treated with repeated doses of 3 mL sevoflurane, presenting no indication of mucosal damage; importantly, the fissure reportedly healed.
Concerning the eventual effects of sevoflurane specifically on joints, three clinical cases describing infections affecting the elbow, 38 ankle, 23 and knee 25 need to be addressed. Osteosynthesis material was involved in all three infections, but all of them were successfully resolved with sevoflurane in addition to systemic antibiotics. No patient presented any toxic effects attributable to sevoflurane on the joints. Despite experiencing an infectious process, all three patients remained asymptomatic after the wounds healing process was completed, suggesting a lack of synovial damage. Furthermore, the two patients with elbow and ankle infections presented no clinical signs suggestive of arthrosis, suggesting a lack of toxic effects on the cartilage.
Collectively, these findings suggest that the topical application of liquid sevoflurane appears to be safe to body tissues, including intra-articular structures.
Contrary to the absence of definitive histological changes in the joints, two transient extra-articular local effects were observed, namely, scabs and subcutaneous emphysema.
In the present study, scabs as a consequence of scratching appeared from day 2 onward and were more frequent in right knees. Moreover, considering that the irrigation of sevoflurane on wounds carries the potential to cause itching, 18 scabs could be expected on left knees owing to the induced itching, but not on right knees. However, the reasons for scratching may differ considerably. The volume for injection (150 μL) was selected to avoid mechanical damage to the articular capsule owing to overdistension. 28 However, the distension produced by this volume could be sufficiently intense to cause a painful sensation, which, in turn, could result in scratching. In this case, the intense analgesic effect of sevoflurane could alleviate this sensation, which, in turn, could prevent scratching of the left knees. In other studies, volumes as high as 250 μL of either saline or local anesthetics, such as levobupivacaine 29 or procaine, 30 have been injected into the knees of Sprague-Dawley rats, and scabs were not observed. However, the rats received a single injection,29,30 and this difference in experimental design could explain the absence of scabs.
All the animals developed some degree of subcutaneous emphysema, which exclusively affected the left knees injected with sevoflurane. Thus, sevoflurane may be then the reasonable causative agent. At 37°C, each milliliter of sevoflurane volatilizes into 195 mL of gas. 39 Thus, 29 mL of gas would be obtained after injecting of 150 μL of liquid sevoflurane. Part of this volume of gas, escaping the joint, may have transitorily been trapped into subcutaneous structures, creating small cavities that later disappeared. In line with this hypothesis, intra-abdominal bubbling has been observed following irrigation of the abdominal cavity with ether (also a volatile liquid) in patients suffering from intra-abdominal infections. 11 Moreover, ether was reported to quickly volatilize and then remain in its gaseous state for several days in the cavity of tuberculous abscesses injecting a solution of iodoform in ether into abscesses. 40 In terms of the current study, the presence of mild and transient subcutaneous emphysema was considered irrelevant.
Concerning animal welfare, no animal showed signs suggestive of systemic effects that could be attributed to the anesthetic drug following recovery from anesthesia. This is consistent with the lack of systemic effects observed after topical sevoflurane application to chronic wounds 18 and when injected into body cavities. 19,20,36,37 It has been proposed that, as for any other drug applied topically, sevoflurane will enter the venous system, where it can subsequently reach alveolar capillaries. Owing to its volatility, it diffuses into the alveoli and is exhaled through respiration. Pulmonary elimination will prevent the drug from reaching the arterial system in significant amounts, which, in turn, will reduce the risk of systemic effects. 18
The main strength of this study was that the solutions were injected for several consecutive days instead of a singular injection. Previous reports evaluating the adverse effects of a single local anesthetic injection highlighted that the effects of repeated injections could considerably differ from those observed after a singular injection. 29,30 Additionally, repeated injections of any topical antimicrobial agent parallel a real therapeutic regimen for septic arthritis. Topical sevoflurane has been repeatedly applied for the successful treatment of infected ulcers and infected surgical wounds. 19-21,23-27
This study has several limitations
Sample size was not calculated and, considering the final small sample size, it could be argued that the histological results were underpowered. However, as stated below, our aim was not to detect any kind of histological damage. Instead, we aimed to rule out that intra-articular sevoflurane caused permanent and severe histological changes that could be evident even after treating few animals, in which case this drug would be excluded for further research. In our opinion, considering the absence of severe and permanent histological changes in the joint structures, this exploratory study fulfilled our objective.
In the present study, sevoflurane was additionally employed to anesthetize the experimental animals. Theoretically, it could be argued that sevoflurane administered by inhalation could have potentiated the eventual effects of injected sevoflurane on articular structures. However, considering the paucity of histological findings, it seems reasonable to discard this possibility.
Owing to the design of the present study, we were unable to assess whether sevoflurane could cause any type of transient acute toxicity to the joint. Previously, neutrophil infiltration has been observed the day after single intra-articular injections of conventional local anesthetics such as levobupivacaine,29,41 bupivacaine, or procaine 30 ; however, neutrophils were not observed beyond day 7. Nevertheless, we aimed to simulate the clinical conditions associated with the treatment of septic arthritis. Accordingly, sevoflurane injections were repeated for 5 days, as previously stated, and histological assessment was performed after treatment completion. This design was selected to reduce the number of animals to be sacrificed while simultaneously assessing the long-term changes, which are the important factors when treating septic arthritis.
This safety study lacks a dose-response curve. We selected an initial dose/volume after considering that the joints would not be damaged because of mechanical overdistension 28 while, at the same time, it was deemed to be high enough to be eventually useful for the treatment of arthritis. Based on the absence of definitive histological changes, we discarded to evaluate further lower doses to minimize the number of experimentation animals.
Although we investigated the safety of sevoflurane in normal knees, our primary goal remains to utilize sevoflurane for treating septic arthritis in the future. Reportedly, the effect of drugs could differ between normal and altered joints. 30 For example, local anesthetics were safe toward the cartilage of normal joints but demonstrated a toxic effect on osteoarthritic joints. 42 Hence, we cannot completely exclude the possibility of sevoflurane toxicity in infected joints.
Conclusion
We conclude that, under the described experimental conditions, repeated intra-articular injections of sevoflurane in rats appeared to be a safe procedure. Further experiments to investigate the efficacy of intra-articular sevoflurane as an antimicrobial agent for the treatment of septic arthritis are warranted.
Supplemental Material
sj-pdf-1-eji-10.1177_20587392211072639 – Supplemental Material for Clinical and histological safety assessment of repeated intra-articular sevoflurane injections into knee joints of Sprague-Dawley rats
Supplemental Material, sj-pdf-1-eji-10.1177_20587392211072639 for Clinical and histological safety assessment of repeated intra-articular sevoflurane injections into knee joints of Sprague-Dawley rats by Ainara Achaerandio-de Nova, María D Berenguer-Romero, Syonghyun Nam Cham, Mónica Gómez-Juárez Sango, Sergio Losa-Palacios, Angel Escudero-Jiménez, and Manuel Gerónimo-Pardo, in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the research, authorship, and/or publication of this article: the publication costs of this research were funded by Diputación de Albacete.
Ethics approval
Ethical approval for this study was obtained from the Animal Experimentation Ethics Committee of Castilla-La Mancha University (PR-2016-11-17).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
