Abstract
The high incidence of alcoholic poisoning in Indonesia is caused by the use of methanol as a surrogate ingredient of nonconforming alcohol. The product of methanol metabolism is toxic to the liver. Ranitidine has been studied as an antidote to reduce the effect of methanol toxicity. The present study aimed to assess the effect of ranitidine administration on the liver damage of Wistar rats with acute methanol intoxication. This research was an experimental study with randomized and posttest-only control group design. A total of 24 male Wistar rats divided into four groups, each consisting of six Rats. The control group (K0) served as a reference normal value. The control group (K1) was intoxicated with methanol 7 g/kg body weight (b.w.) through rat gavage tube. Treatment groups were intoxicated with methanol and 15 minutes after that, Ranitidine (30 mg/kg b.w. (P1) or 60 mg/kg b.w. (P2)) was given intraperitoneally. Ranitidine administration was proven to protect the liver tissue from damage due to methanol intoxication as was indicated from the histopathological examination. Ranitidine with dose 60 mg/kg b.w. is better in reducing the degree of liver tissue damage in acute methanol intoxication rats.
Introduction
Alcoholic poisoning often occurs in Indonesia. During the period 2008–2017 there were 840 reports of deaths due to illegal alcohol. 1 Meanwhile, from January to April 2018 the number of victims died due to the consumption of alcohol recorded 144 people. 2 It was caused by consuming nonconforming alcohol, which is a mixture of raw materials that do not go through the proper distillation process and alcohol substitution (surrogate), changes in ethanol-based ingredients to methanol. 1 Methanol (CH3OH) is a colorless, polar, volatile, flammable liquid with a mild alcoholic odor when pure. It is widely used as an industrial including paints, varnishes, antifreeze and gasoline deicers, cleansing solutions and laboratory solvent for extracting, washing, drying and recrystallization. 3
Methanol causes serious acute intoxication changes due to its metabolites, Formaldehyde, and Formic Acid. It can be absorbed by the body through the digestive system, skin, and lungs. Its absorption process takes 30 to 60 minutes. 4 Methanol metabolism takes place in the liver through three stages of oxidation. First, methanol into formaldehyde by Hepatic Alcohol Dehydrogenase (ADH), followed by oxidation of formaldehyde to formate or formic acid (depending on pH) with the help of the enzyme Aldehyde Dehydrogenase (ALDH). 5
The format elimination process through the folate-dependent pathway reaches 24 hours or more for methanol doses of more than 1 g/kg body weight. The slow process of elimination causes accumulation of formic acid in the body which triggers the emergence of toxic effects in the form of inflammatory processes in various organs and the occurrence of metabolic acidosis. 6
The hepatotoxic effects of alcohol metabolites have been reported by several studies. Acetaldehyde together with the gastrointestinal tract endotoxin may cause morphological and functional malformations, as well as the induction of the immune response. 7 –11 Formic acid, could acts as an inhibitor of cytochrome c oxidase, in which can disturb the oxygen utilization in tissue. This situation can increase the level of reactive oxygens species that further leads to inflammation, hepatocyte hypoxia, fatty liver, fibrosis, and cirrhosis. 12 In long-term consumption, alcohol may cause alcoholic liver disease (ALD). The onset of this illness is tightly related to the high doses of alcohol being consumed. 13 A study on Wistar rats showed that chronic ethanol consumption can cause significant histopathological liver changes, such as congestion, central vein dilation, hepatic portal vein branch dilation, Kupffer cells hyperplasia, fatty liver changes, and hepatocytes focal necrosis. 14
Comparative research on the effectiveness of Ranitidine on ethanol and fomepizole (as an antidote) and Dexamethasone (as an anti-inflammatory) in cases of acute methanol intoxication have been carried out previously, proving that Ranitidine is more significant in inhibiting methanol metabolism by decreasing formic acid levels. 6 Ranitidine is an inhibitor of the ADH and ALDH enzymes so it can be used as a therapeutic antidote in methanol poisoning. 15 Ranitidine is known to have more minimal side effects than ethanol and is economically cheaper and easier to obtain than fomepizole (4-MP). 16
Termination of the methanol metabolic chain by the administration of ADH and ALDH inhibitors before the format reaches toxic peak levels in the body is the reason for the authors to give Ranitidine in the 15th minute. The effect of graded dosage Ranitidine on the degree of liver cell damage is unknown. Based on this background, a study was conducted to determine the effect of graded dosage Ranitidine by earlier administration to determine the degree of liver cell damage in acute methanol intoxication.
Methods
Chemicals
Methanol (99.9%, Delta Trade Company, Egypt) were diluted in sterile water and administered as a 20% w/v solution. Ranitidine (50 mg/ 2 mL amp.) was purchased from Quantum Labs, diethyl ether (99.9%, Delta Trade Company, Egypt) were used in this study.
Study design and animal grouping
This study was conducted on 24 male healthy Wistar rats aged 2–3 months and weighing 150–200 g. All animals were acclimatized for 7 days in a care cage of 45 × 35 × 12 cm3, maintained on a 12-hour light/dark schedule, and supplied with food and water via ad libitum. Animals were handled with the Guide for Care and the approval from the Ethics Committee of the institution. Rats were randomly divided into four groups with six rats in each group: two control groups (K0 and K1) and two test groups (P1 and P2). In the control negative group (K0), animals were used as a reference for normal values. Control group (K1), animals were intoxicated with methanol (7 g/kg body weight) orally via rat gavage tube. Both test groups (P1 and P2) were intoxicated with methanol in the same procedure as control and 15 minutes after that, Ranitidine was given via intraperitoneally (30 mg/kg of body weight for P1 and 60 mg/kg of body weight for P2).
Specimen collection
Eight hours after the treatment, animals were anesthetized by ether inhalation and sacrificed through the cervical dislocation. Following the termination, the liver was taken out carefully and placed immediately in a container consisting of 10% formalin buffer and preservative.
Histological examination
Histological specimens of the liver were prepared by longitudinal cuts and stained with hematoxylin–eosin. Liver specimens were observed under a light microscope in five different fields of view, with magnifications of 400×. Each field of view was scored based on slightly modified Metavir, Knodell, 17 and AHHS scores. 18 The scoring criterion can be seen in Table 1.
Scoring of liver histopathological examination based on the Metavir, Knodell, and AHHS scoring.

Results
Methanol dosage for rats intoxication
A preliminary study was performed to determine the appropriate dose of methanol intoxication. Briefly, two Wistar rats were given methanol at a concentration of Lethal Dose-50 (LD-50) of 14 g/kg body weight via a gavage tube. After 8 hours following the methanol administration, one rat was found to be dead while the second one was found to be weak, unconscious, but the respiratory rate was still visible. The methanol dose was then cut into half and a quarter of LD-50, which is 7 g/kg and 3.5 g/kg of body weight and were given to two rats. After 8 hours of methanol administration, both rats were still alive and were active. Histopathological profile of both rats was evaluated (shown in Figure 1).

Photomicrographs of rat liver stained with hematoxylin–eosin (HE) with 400× magnification. (A) Rat given methanol of 3.5 g/kg body weight and (B) rat was given methanol of 7 g/kg body weight. Both liver specimens showed the presence of inflammatory cells, dead tissue (necrosis), cell degeneration, and fibrosis.
Rats treated with methanol at a concentration of 3.5 g/kg or 7 g/kg body weight developed an abnormal condition of the liver, while they were still alive and able to proceed with the experiment. Based on this data, methanol intoxication on Wistar rats was given at 7 g/kg of body weight as a dose that is able to give damage to the liver without causing death to the animals.
Scoring of the liver damage
Statistically, the average liver tissue damage between groups is different (p < 0.05). As predicted, animals in group K1 showed the highest liver damage due to methanol intoxication. Ranitidine treatment on methanol intoxicated rats (group P1 and P2) was proven to lower the score of liver damage (Table 2). Further analysis of intergroup liver damage scores showed each group were different significantly to each other (Table 3).
Scoring of liver examination and result of Kruskal Wallis test for all test groups.

* Significant if p < 0.05.
Intergroup Mann–Whitney difference test.
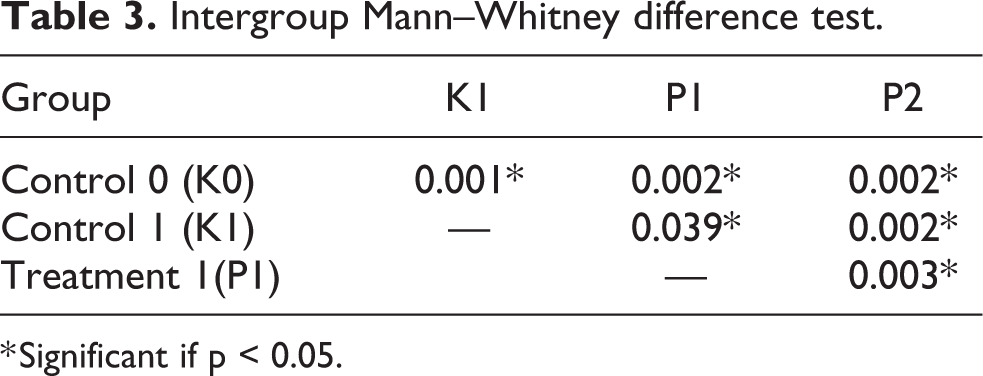
* Significant if p < 0.05.
Histological results
Histopathology examination of rats liver from the four groups was shown in Figure 2. As can be seen in Figure 2.A, control group 0 (K0) showed the condition of the normal liver tissue with no inflammatory cells, necrosis, fibrosis, bilirubinostatis were found. In control group 1 (K1), an increase of inflammatory cells was found on 1/3–2/3 portal which was a sign of moderate portal inflammation. Also, there is a grade 4 or severe degeneration that includes more than 2/3 lobules and the formation of grade 3 fibrosis is septal scarring without cirrhosis.
In treatment group 1 (P1), liver tissue degeneration rates were scored 3 in four of the rats indicating moderate degeneration (including more than 1/3–2/3 lobules), whereas in the other two rats the degree of degeneration was scored 1, indicating mild degeneration (acidophilic bodies, ballooning degeneration is less than 1/3 lobules). In addition to degeneration, portal inflammation was also found at grade 1 in five rats indicating mild inflammation occurs with distribution less than 1/3 portal channel, whereas in one rat portal inflammation was similar to group 1. In the case of fibrosis, we observed a decrease in the degree of fibrosis when compared to the control group 1 (K1), from grade 3 in the six rats, to grade 2 for five rats and grade 1 for one rat.
The degree of liver tissue degeneration in treatment group 2 (P2) decreased compare to treatment group P1, from score 3 to score 1 for four rats and score 0 for two rats. There was also an improvement in inflammation and fibrosis score observed in group P2 compare to P1. Mild portal inflammation (inflamed cell <1/3 portal areas) with score 1 was seen only in two rats while the remaining rats showed no inflammation. Portal fibrosis without septal fibrosis (score 1) was observed in three rats and the other three showed no fibrosis. All the histopathological scores for each rat in all groups were provided in Supplementary Table 1.

Photomicrographs of rat liver structure stained with hematoxylin–eosin (HE) and viewed with 400× magnification. (A) Control group K0 showed the normal structure of the liver with hexagonal shaped lobules with a central vein in the middle and trabecular arrangement of hepatocytes and portal triads; (B) control group K1 showed inflammation cells on 1/3–2/3 portal, degeneration more than 2/3 lobules and septal scarring without cirrhosis; (C) treatment group P1 showed degeneration on more than 1/3–2/3 lobules, portal inflammation less than 1/3 portal channel, and portal fibrosis with few septal fibrosis; (D) treatment group (P2) showed mild degeneration (acidophilic bodies, ballooning degeneration, without focus necrosis around <1/3 lobulus), portal fibrosis without septal fibrosis and absent of portal inflammation.
Discussion
Based on the results of this study, significant differences were obtained in the microscopic images of the liver between all groups. The group that was intoxicated with methanol clearly showed liver damage, which when was treated immediately with Ranitidine (within 15 minutes) showed improvement on the status of liver damage. The accumulation of alcohol metabolism such as formic acid could lead to metabolic acidosis. Formic acid acts as a cytochrome c oxidase inhibitor that causes impairment of oxygen utilization in tissue. Subsequently, this could lead to an increase in free radicals production and triggering liver damage. 19
The distribution of methanol metabolites takes place rapidly in the body fluids, including blood, intracellular fluid, and interstitial fluid. Peak levels of methanol metabolites in the blood are reached within 30–90 minutes after exposure. 20 Vasodilation increased vascular permeability, and activation of neutrophils and their migration are interdependent. Immediately after tissue damage, there is a constriction phase in the arterioles which is followed within a few seconds by dilatation of the arterioles and capillaries, thereby increasing blood flow to the damaged tissue. The dilated capillaries and venules trigger the collection of erythrocytes and the blood flow slows and then stops. Slowing blood flow causes neutrophils to come into direct contact with endothelial cells, resulting in adhesion of molecules to the membrane surface. Neutrophils phagocytose necrotic tissue debris. So that in the preparations, inflammation can be identified by the discovery of an inflammatory cell called neutrophils. 21 While in hydropic degeneration, there is a vacuole filled with water in the cytoplasm that does not contain fat or glycogen. This change occurs due to metabolic disorders such as hypoxia due to accumulation of formic acid and microcirculation disorders. 22
Ranitidine is known as one of the histamine (H2) receptor competitive antagonists. Ranitidine is an inhibitor of gastric alcohol dehydrogenase (ADH) and hepatic aldehyde dehydrogenase (ALDH), an enzyme that is responsible for metabolizing methanol into toxic products, namely formaldehyde and formic acid. Research that supports the finding in this study is a comparative study of the effects of Ranitidine, Dexamethasone, and their combination on blood formic acid levels and retinal cytochrome c release in acute methanol intoxication mouse models. It was concluded that Ranitidine treatment on methanol intoxicated mouse gives the best result in decreasing the concentration of formic acid in the blood and the release of retinal cytochrome c. 23 The results of this study are in line with what was observed on those previous study. Ranitidine can be used as a potent antidote in the case of methanol intoxication which protects the liver tissue from excessive damage due to metabolic acidosis.
Mann–Whitney different test between Rats in group P1 against P2 has a significant value of p = 0.003, proving that doubling the Ranitidine dose to 60 mg/kg body weight, which is twice the amount that was used for the treatment of peptic ulcer in rat, is better in reducing the liver damage. These results are in line with another study which observed that increasing the dose of Ranitidine by two times the initial dose proved to be more effective in inhibiting methanol metabolism into a toxic form thereby reducing the degree of degeneration of the optic nerve. 23
Conclusion
The administration of Ranitidine 30 mg/kg and 60 mg/kg after 15 minutes of methanol intoxication is significant in suppressing the level of liver tissue damage. The administration of Ranitidine 60 mg/kg is more effective in reducing liver tissue damage than Ranitidine 30 mg/kg.
Supplemental material
Supplementary_Table_1 - The effect of ranitidine administration in graded dosage to the degree of liver damage: A study on Wistar rats with acute methanol intoxication
Supplementary_Table_1 for The effect of ranitidine administration in graded dosage to the degree of liver damage: A study on Wistar rats with acute methanol intoxication by MP Nugrahanti, D Armalina, DL Partiningrum and F Fulyani in Human & Experimental Toxicology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
