Abstract
RWJ-800088 is a novel, potent polyethylene glycol (PEG)-conjugated thrombopoietin (TPO) mimetic that increases platelet levels and protects against thrombocytopenia. A nonclinical safety program was customized for this peptide that takes into account its protein-like structure, synthetic chemical nature, agonist pharmacologic activity, and mode of administration. In repeat-dose toxicity studies, the salient findings were dose-related increases in circulating platelet counts, mean platelet volume, and megakaryocytes in the bone marrow with no antibody formation. Reversible myelofibrosis and hyperostosis were observed in rats, but not dogs, when the circulating platelet levels exceeded 3× those of vehicle controls. The bone effects were due to the exaggerated pharmacologic effect and excessive stimulation and elevation of megakaryocytes by TPO, which results in intramedullary proliferation of fibroblasts and mesenchymal cells followed by osseous metaplasia. These findings support the use of platelet elevations of >3× as a stopping criterion to prevent potential adverse bone-related effects in humans.
Keywords
Introduction
Thrombopoietin (TPO) is an endogenous glycoprotein that is the primary regulator of megakaryocyte and platelet production during megakaryocytopoiesis, which is a continuous developmental process of platelet production in which hematopoietic stem cells undergo proliferation, differentiation, and maturation. 1 Thrombopoietin functions in part by activating the cytokine receptor c-mpl, which is found on the surface of megakaryocytes, platelets, and hematopoetic stem cells. 2 –4 In adult mice, elimination of the TPO gene or its receptor results in a greater than 80% decrease in megakaryocytes and platelets but normal levels of other hematopoietic cell types. 5 Several studies have identified the liver as the major site for TPO production. 6 Additional sites include the kidneys, which express a significant amount of TPO messenger RNA (mRNA) and are also responsible for erythropoietin production; bone marrow, where it can be directly delivered to the resident megakaryocytes; as well as spleen, testis, muscle, and brain. 3,7 Several studies have shown that TPO is cleared from circulation, at least in part, through binding to the c-mpl receptors on the platelet surface, internalization, and subsequent degradation, which serves as a negative feedback mechanism for platelet production. 2,8 Similar to other hematopoietic growth factors, several lines of evidence indicate that TPO has a broader range of activities that go beyond its role in megakaryocytopoiesis, which include a role in stimulating erythroid production and other progenitor cell types as well as enhancement of in vivo megakaryocyte and platelet production and erythroid recovery following radiation or other perturbations of homeostasis in the hematolymphoid compartment. 1,4
Clinically, elevated or reduced TPO levels and/or mutations in the c-mpl receptor are associated with a number of different disease states. For example, endogenous TPO levels are elevated in many disease states including aplastic anemia, iatrogenic thrombocytopenia, congenital amegakaryocytic thrombocytopenia, and lymphoid malignancies, such as lymphoma, myeloma, and chronic lymphocytic leukemia (associated with endogenous TPO levels of 2-12× normal). 3,9,10 Disease states in which endogenous TPO levels are reduced include acute myeloid leukemia and immune thrombocytopenic purpura (ITP). Immune thrombocytopenic purpura is common in patients with chronic liver disease and aquired immunodeficiency syndrome, and patients undergoing chemotherapy. 3 Possible mechanisms for reduced levels of endogenous TPO include elevated expression of the c-mpl receptor on platelets in these patients or reduced TPO production. In the past, the main treatment for these conditions has involved platelet transfusions. 11 However, transfusions are associated with the risk of pathogen transfer, are available in limited quantities due to the increasing use of platelets and a declining donor population, 12 and can delay the rate of recovery from chemotherapy-related thrombocytopenia. 13,14
Early recombinant forms of TPO showed promising results 15 for treatment of thrombocytopenia. However, subsequent studies with first-generation TPO agonists failed because the recombinant proteins generated antibodies that cross-reacted with c-mpl ligands and resulted in paradoxical thrombocytopenia. 16,17 Two second-generation TPO agonists were approved in 2008 for the treatment of immune-mediated thrombocytopenia purpura: romiplostim (Nplate®, Amgen Inc.), which is a subcutaneously (sc) administered TPO peptide mimetic, and eltrombopag (Promacta®, GlaxoSmithKline, Inc.), which is an orally administered TPO nonpeptide mimetic. In clinical studies that included no more than 600 patients treated for limited durations (156 weeks for romiplostim and 151 days for eltrombopag), both agents increased the platelet count in >80% of patients and showed a trend toward lower bleeding events. Issues that require further investigation in clinical studies include rebound thrombocytopenia, thromboembolic complications, increased bone marrow reticulin, and increased myeloblast counts. 11,18
RWJ-800088 is a novel next generation polyethylene glycol-conjugated (PEGylated) TPO mimetic peptide (Figure 1) with potential for treatment of thrombocytopenia purpura and treatment of thrombocytopenia and anemia resulting from exposure to chemotherapy or radiation. 12,19 The compound is comprised of a 29-amino acid synthetic peptide that has no homology with endogenous TPO, thus reducing the potential for generation of antibodies to endogenous TPO. PEGylation of the peptide leads to a significantly reduced clearance and increased half-life of the compound 20 –22 without loss of potency and may reduce the likelihood of antibody formation due to steric hindrance. 19 RWJ-800088 has an estimated EC50 (Effective Concentration at which 50% of the maximal activity has been reached) of approximately 5 pmol/L (0.2 ng/mL) in a human TPO (huTPO) receptor assay in vitro and stimulates megakaryocyte lineage-specific growth and differentiation in vivo (internal results). The PEGylated TPO mimetic peptide was synthesized on solid resin support.
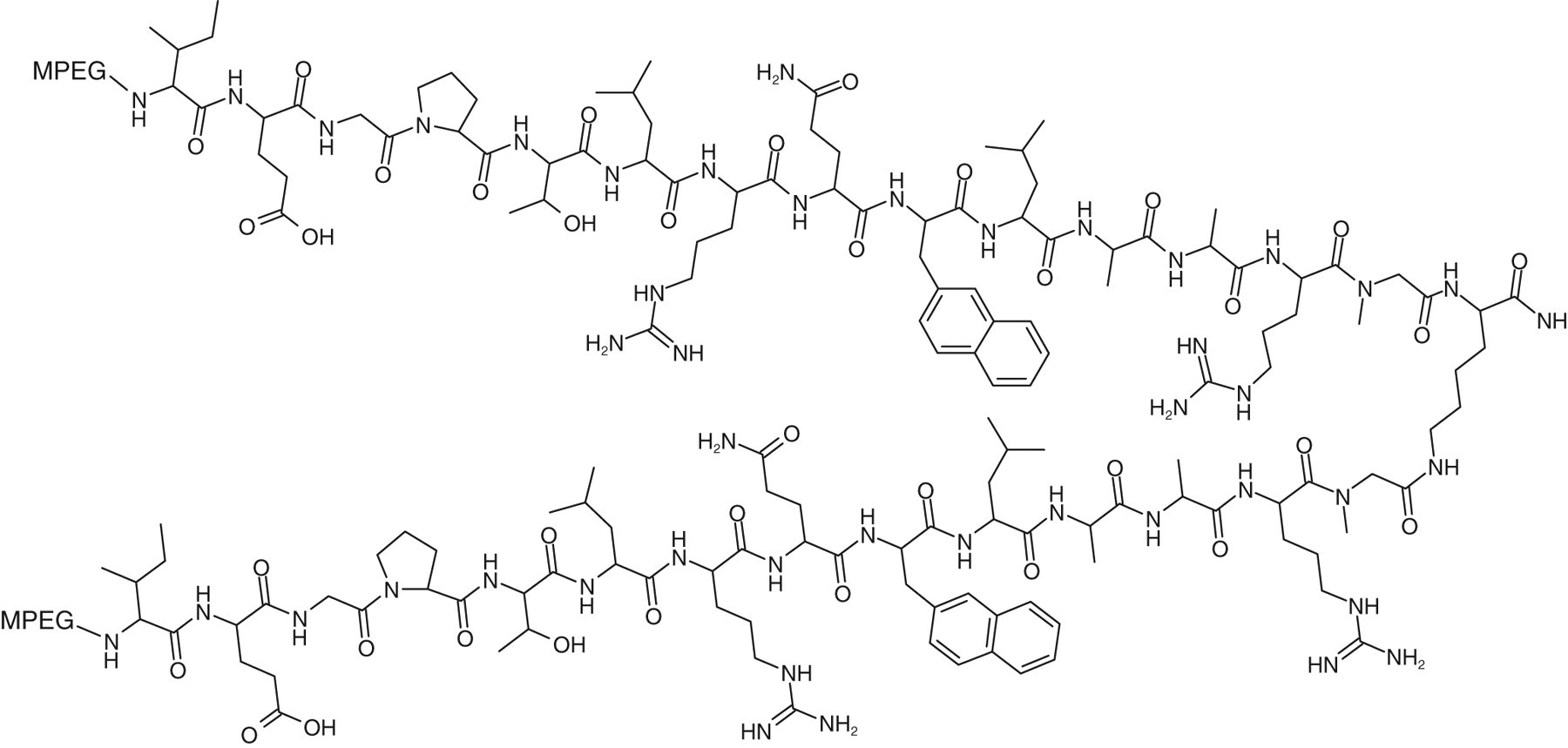
Molecular structure of RWJ-800088 (MPEG = ∼20,000 g/Mole linear PEG).
Due to the dual nature of the molecule, i.e. its protein-like structure and function coupled with its synthetic chemical nature arising from a fully synthetic process, a nonclinical development plan was customized for this compound that was an amalgamation of the most appropriate studies from available internationally accepted regulatory guidance documents (ICH S6 and ICH M3). Accordingly, it falls under the scope of the ICH Topic S6 guidance on the Preclinical Safety Evaluation of Biotechnology-Derived Pharmaceuticals. Section 4.2.3 of the guidance states “the expected consequence of metabolism of biotechnology-derived pharmaceuticals is the degradation to small peptides and individual amino acids. Therefore, the metabolic pathways are generally understood. Classical biotransformation studies as performed for pharmaceuticals are not needed.” Therefore, metabolism studies were not conducted. In addition, although genetic toxicity studies may not be required for molecules of this type under ICH S6, the genetic toxicology battery of studies was conducted because of the synthetic nature of this compound and how it is synthesized.
This article describes the nonclinical safety results of RWJ-800088 and how these data and understanding of the mechanism for the toxicological effects were used to select circulating platelet levels as a marker for monitoring clinical safety. Four areas that are discussed in detail are the approach that was used for evaluating immunogenic potential (based on the concern regarding immunogenicity of other TPO agonists), the mechanism and relevance of the myelofibrosis and hyperostosis observed in rats (due to the exaggerated pharmacology of the compound), the implications of the pharmacodynamic differences across species, and the integrated assessment of how the nonclinical results impact the monitoring of clinical safety.
Materials and Methods
Test Article and Vehicle
RWJ-800088 is a fully synthetic peptide (Figure 1), with a molecular weight of 3295 Da for the parent compound without polyethylene glycol (PEG), and approximately 43,295 Da with 2 linear PEG chains (MW ~20 000 Da each). RWJ-800088 is composed of 2 identical 14-amino acid peptide chains linked by a lysyl residue and linked on each N-terminal to a PEG chain. RWJ-800088 differs from the TPO mimetic analog GW395058
20,21
based on the substitution of a 2β napthyl alanine in place of the 1β napthyl alanine, resulting in an approximately 10-fold lower in vitro potency in Baf/3 cells (TPO responsive immortalized murine bone marrow cell line
23
) with a luciferase reporter gene that were transfected with the human TPO receptor (internal result). The full chemical name is: Methoxypolyethyleneglycol20000-propionyl-
The PEGylated TPO mimetic peptide was synthesized via fmoc-protected amino acid coupling on solid resin support. The bulk peptide was purified by chromatographic techniques. The PEG groups were added as the terminal step in the synthetic process, through conventional PEGylation chemistry. The test material used in these studies was formulated in sterile and nonpyrogenic phosphate-buffered saline or 0.9% sodium chloride.
Nonclinical Studies
All studies were conducted in accordance with good laboratory practices or best scientific principles and in an Association for Assessment and Accreditation of Laboratory Animal Care approved laboratory in accordance with Institutional Animal Care and Use Committee guidelines. For intravenous (iv) administration, RWJ-800088 was injected via the lateral tail vein in rats and via the cephalic or saphenous veins in dogs. For sc administration, RWJ-800088 was injected into a single site in the dorsal region in both rats and dogs.
Pharmacokinetics, Toxicokinetics, and Bioanalysis
Table 1 lists nonclinical studies in which the pharmacokinetics and toxicokinetics of RWJ-800088 were assessed. Single-dose pharmacokinetics of RWJ-800088 were evaluated in rats, rabbits, and dogs after a single iv dose. To support toxicity studies, toxicokinetics were evaluated in rats and dogs after multiple dose (daily and once or twice weekly) iv or sc administration. The toxicokinetics of RWJ-800088 were also evaluated in reproductive and developmental toxicity studies (segment II) in pregnant rats and rabbits administered RWJ-800088 iv (twice weekly) during organogenesis.
Nonclinical Pharmacokinetics and Toxicokinetics for RWJ-800088
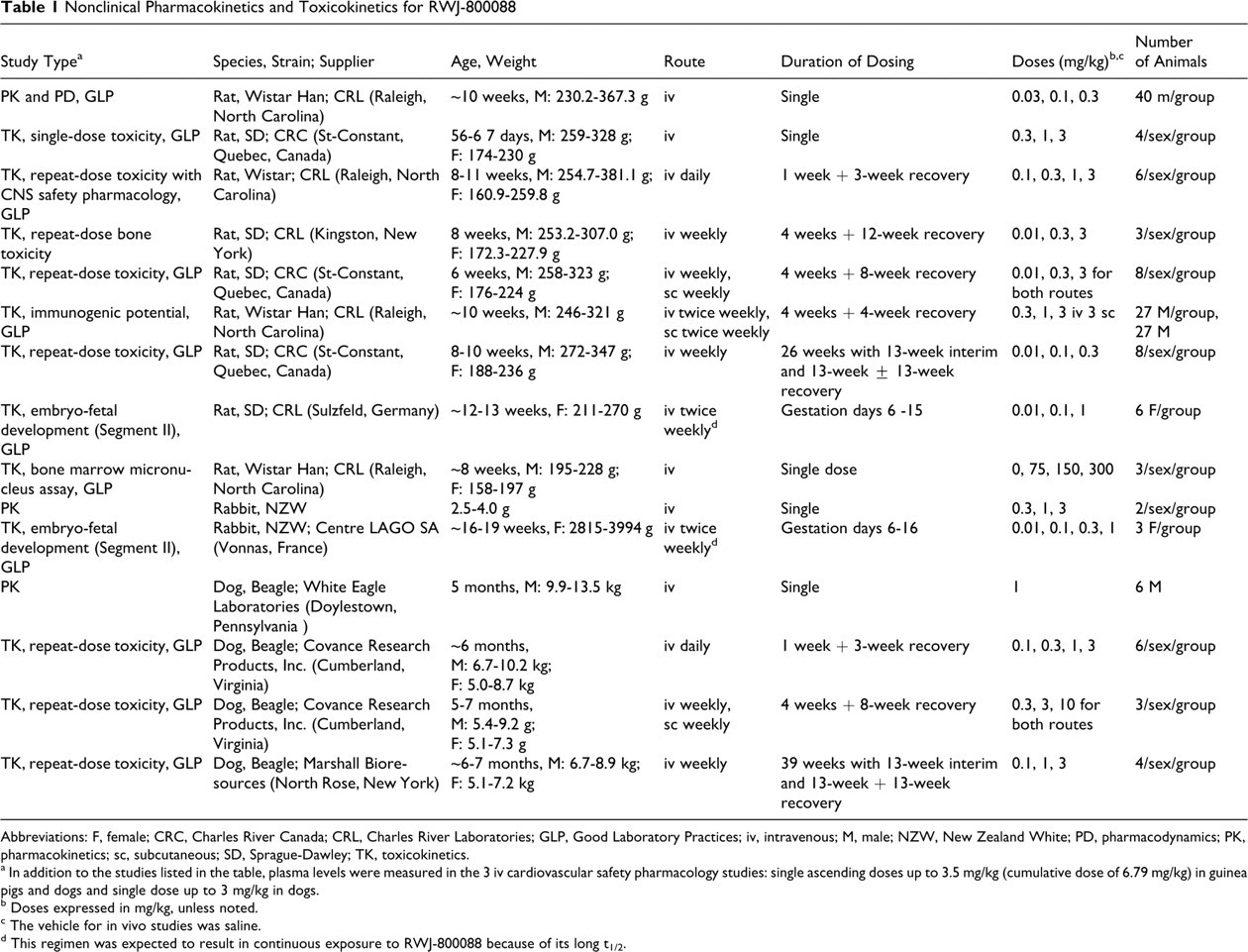
Abbreviations: F, female; CRC, Charles River Canada; CRL, Charles River Laboratories; GLP, Good Laboratory Practices; iv, intravenous; M, male; NZW, New Zealand White; PD, pharmacodynamics; PK, pharmacokinetics; sc, subcutaneous; SD, Sprague-Dawley; TK, toxicokinetics.
a In addition to the studies listed in the table, plasma levels were measured in the 3 iv cardiovascular safety pharmacology studies: single ascending doses up to 3.5 mg/kg (cumulative dose of 6.79 mg/kg) in guinea pigs and dogs and single dose up to 3 mg/kg in dogs.
b Doses expressed in mg/kg, unless noted.
c The vehicle for in vivo studies was saline.
d This regimen was expected to result in continuous exposure to RWJ-800088 because of its long t1/2.
RWJ-800088 plasma levels were quantified using a sandwich enzyme-linked immunosorbent assay (ELISA) with a capture polyclonal antibody specific for non-PEGylated RWJ-800088 and a second indicator monoclonal antibody specific for PEG, with a lower limit of quantification (LLOQ) of ≤10 ng/mL in rat, rabbit, and dog plasma (early studies had an LLOQ of 98 ng/mL), providing sufficient limits to support individual study objectives. There were no oral dosing studies conducted with RWJ-800088 since this is not considered to be a viable route of administration.
Pharmacokinetic analysis was conducted using the WinNonlin (Version 4.0.1)-validated computer program (Pharsight, Sunnyvale, California). Mean plasma concentrations of RWJ-800088 were calculated at each sampling time at each dose level in all dose groups. Mean plasma concentration–time profiles were evaluated by noncompartmental pharmacokinetic analysis. Peak plasma concentrations (Cmax) were determined, and the area under the plasma concentration–time curve from time 0 to 24 hours after dosing (AUC0-24h) was calculated using the linear up/log down trapezoidal rule.
Toxicity, Immunogenicity, Antigenicity, and Blood Compatibility Studies
Tables 2 and 3 provide an overview of studies with RWJ-800088 to assess toxicity, immunogenicity, antigenicity, and blood compatibility and present study design information on species, strain, age, route of administration, duration of dosing, doses, and number of animals. All of the changes in hematology and clinical chemistry parameters are reported as mean values across genders since no significant gender differences were observed.
Nonclinical Toxicology Program for RWJ-800088: Single- and Repeat-Dose Toxicity, Investigative Toxicity
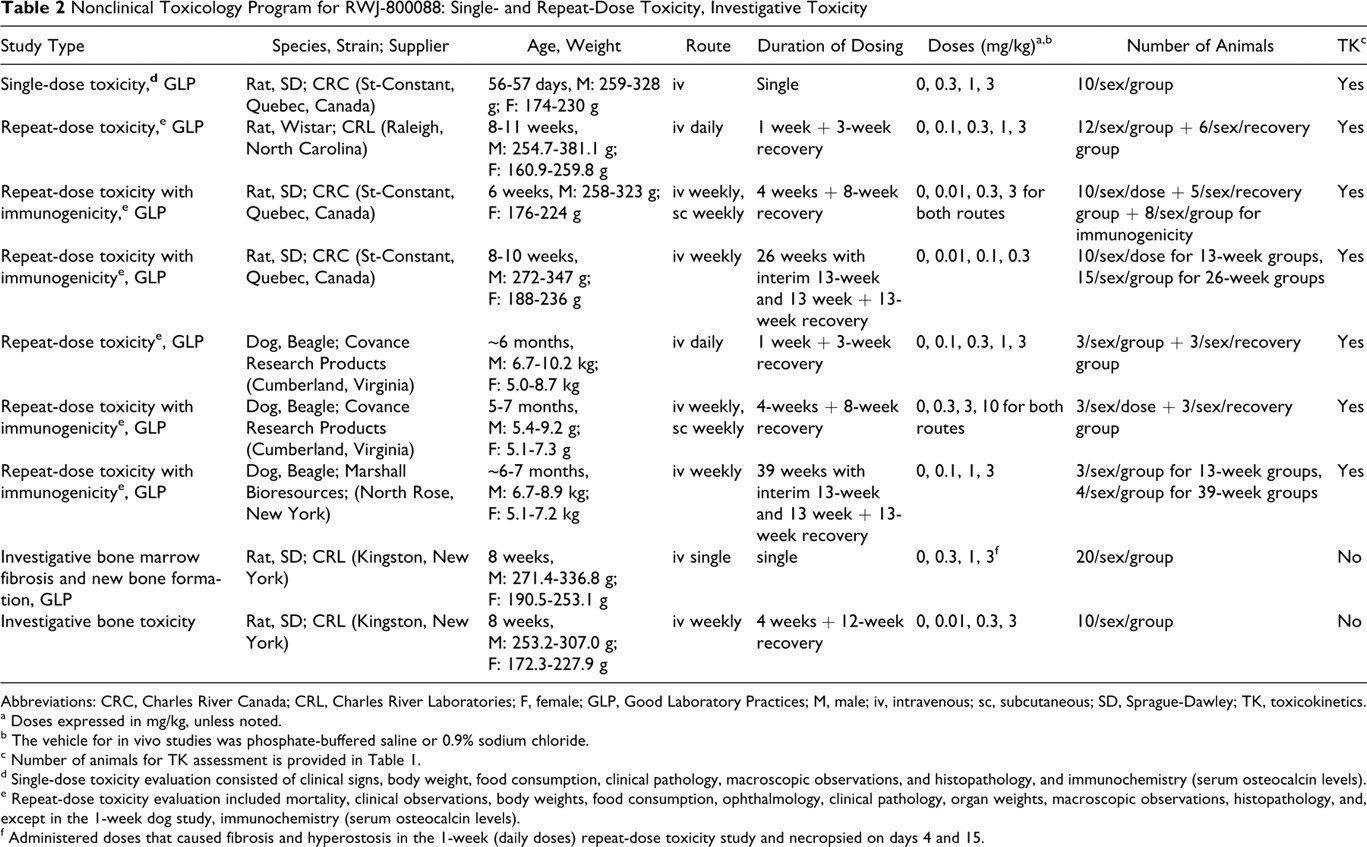
Abbreviations: CRC, Charles River Canada; CRL, Charles River Laboratories; F, female; GLP, Good Laboratory Practices; M, male; iv, intravenous; sc, subcutaneous; SD, Sprague-Dawley; TK, toxicokinetics.
a Doses expressed in mg/kg, unless noted.
b The vehicle for in vivo studies was phosphate-buffered saline or 0.9% sodium chloride.
c Number of animals for TK assessment is provided in Table 1.
d Single-dose toxicity evaluation consisted of clinical signs, body weight, food consumption, clinical pathology, macroscopic observations, and histopathology, and immunochemistry (serum osteocalcin levels).
e Repeat-dose toxicity evaluation included mortality, clinical observations, body weights, food consumption, ophthalmology, clinical pathology, organ weights, macroscopic observations, histopathology, and, except in the 1-week dog study, immunochemistry (serum osteocalcin levels).
f Administered doses that caused fibrosis and hyperostosis in the 1-week (daily doses) repeat-dose toxicity study and necropsied on days 4 and 15.
Nonclinical Toxicology Program for RWJ-800088: Immunogenicity, Antigenicity, and Blood Compatibility Studies
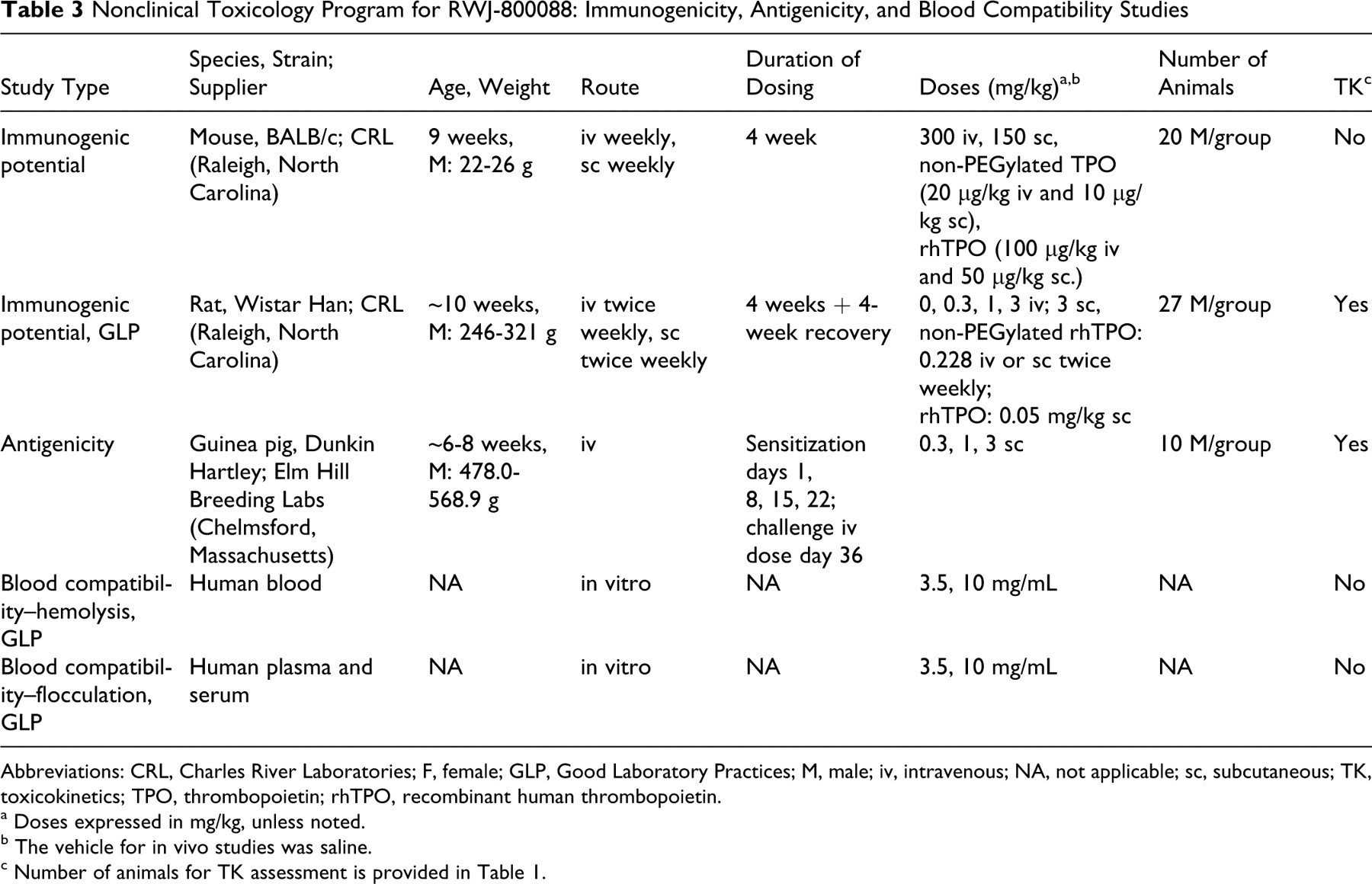
Abbreviations: CRL, Charles River Laboratories; F, female; GLP, Good Laboratory Practices; M, male; iv, intravenous; NA, not applicable; sc, subcutaneous; TK, toxicokinetics; TPO, thrombopoietin; rhTPO, recombinant human thrombopoietin.
a Doses expressed in mg/kg, unless noted.
b The vehicle for in vivo studies was saline.
c Number of animals for TK assessment is provided in Table 1.
Single- and repeat-dose and investigative toxicity
The toxicity of RWJ-800088 was evaluated after a single iv dose in rats, daily iv doses for 1 week in rats and dogs, weekly iv or sc doses for 4 weeks in rats and dogs, and weekly iv doses for 13 and 26 weeks in rats and 13 and 39 weeks in dogs. Both the iv and sc routes were evaluated in the 4-week studies to support potential administration by both routes in the clinic. Overt toxicity was assessed based on routine study parameters including body weight and food consumption, in-life observations, hematology, activated partial thromboplastin time (APTT), partial thromboplastin time (PTT), clinical chemistry, urinalysis, and macroscopic and microscopic observations. Additional toxicity assessments, except in the 1-week studies, included immunochemistry (serum oseteocalcin levels; Biomedical Technologies Osteocalcin RIA kit, Stoughton, Massachusetts) and analysis of blood serum using a validated Biacore method for the presence of antibodies to RWJ-800088, non-PEGylated RWJ-800088 (JNJ-27382667), and recombinant human TPO (rhTPO) with a confirmatory immunodepletion assay to evaluate immunogenic potential (see below for information on Biacore method). An investigative study in rats evaluated whether doses that caused fibrosis and hyperostosis in the 1-week (daily doses) study occurred after a single iv injection.
Antigenicity and immunogenicity assessment
In a guinea pig antigenicity study, male guinea pigs were exposed to sensitizing doses of RWJ-800088 (0.3, 1, and 3 mg/kg), prepared with complete Freund's adjuvant, that were given subcutaneously (sc) on days 1, 8, 15, and 22 followed by an iv challenge dose of RWJ-800088 (1 mg/kg), vehicle (buffered saline), or the positive control antigen (ovalbumin, 0.5 mg/kg) on day 36. Immunogenic potential was assessed in a pilot study mice (weekly doses) and a definitive study in (twice weekly doses) treated iv or sc for 4 weeks. In mice, RWJ-800088 (150 mg/kg sc and 300 mg/kg iv), positive control rhTPO (50 mg/kg sc and 100 mg/kg iv), negative control non-PEGylated RWJ-800088 (10 mg/kg sc and 20 mg/kg iv), or vehicle (0.9% sodium chloride) were administered once daily on days 0, 7, 14, 21, and 28 with additional sc dosing on days 1, 8, 15, 22, and 29. Mice were euthanized on day 33 and day 39, respectively, for the measurement of platelets and antibodies against non-PEGylated test compound or rhTPO (Biacore surface plasmon resonance). Some mice that received test articles on days 0 and 1 were euthanized on days 5 and 11 to evaluate platelets and antibodies, respectively. In rats, RWJ-800088 was administered by the iv (0.3, 1, or 3 mg/kg) and sc (3 mg/kg) routes twice weekly for 4 weeks. The negative control was non-PEGylated RWJ-800088 (0.228 mg/kg, iv or sc), and the positive control was rhTPO (0.05 mg/kg sc on days 1, 3, 7, and 21). Blood samples were obtained for antibody determination (Biacore assay) at predose (day 1), during dosing (days 8, 15, and 22), and postdose (days 29, 37, 43, and 50).
Blood compatibility
Blood compatibility of RWJ-800088 (3.5 and 10 mg/mL) was assessed to evaluate its hemolytic potential in human blood and flocculation formation in human serum and human plasma. In the hemolysis study, samples were incubated for 45 minutes at 37°C, centrifuged for 5 minutes at 1000 g, and then the amount of hemoglobin in the supernatant was measured by assessing optical density in a spectrophotometer at 540 nm. Hemolysis ≤10% was considered insignificant. In the flocculation study, samples were incubated for 30 minutes at room temperature. The tubes were then examined macroscopically and microscopically for precipitation or coagulation, and an aliquot from each tube was centrifuged at 14 000 rpm for 10 minutes and examined for the presence or absence of a pellet. The presence of precipitation, a pellet, and/or coagulation in this assay indicates that the mixture is incompatible with blood.
Bioanalytical Methods for Detection of Serum Antibodies
Serum antibodies to RWJ-800088 or non-PEGylated RWJ-800088 were assayed by surface plasmon resonance using a Biacore 3000 instrument system (GE Healthcare, Umea, Sweden) method that was developed for a TPO-mimetic analog 20 and modified for RWJ-800088. Presence of antibodies was confirmed by immunodepletion, which consisted of independently adding non-PEGylated RWJ-800088 and RWJ-800088 to control samples prior to analysis. A Biacore method was also used to detect serum antibodies to rhTPO in the mouse immunogenicity study and validated for use in detecting anti-rhTPO antibodies in the rat immunogenicity study. The Biacore analytical method uses an antibody directed against the protein portion of RWJ-800088 as a positive control because it was not possible to initiate an immune response using the PEGylated molecule. Therefore, a surrogate rabbit polyclonal anti-non-PEGylated RWJ-800088 antibody was used as the assay positive control. The assay validation of the immunodepletion portion of the assay clearly showed that approximately the same mass of non-PEGylated RWJ-800088 and RWJ-800088 inhibits equivalent masses of rabbit polyclonal anti-non-PEGylated RWJ-800088 antibody. These data suggest that the antibody reacts with both the peptide and the PEGylated peptide to the same extent and further support that antibodies formed against RWJ-800088 in species tested would react against the non-PEGylated RWJ-800088 sensor and undergo inhibition when immunodepletion reagents are added.
The Biacore method was also validated for detecting anti-RWJ-800088 antibodies to support the 4-week and chronic rat and dog toxicity studies (unpublished method). The sensitivity for anti-RWJ-800088 antibodies was 1000 ng/mL in rat serum and 150 ng/mL in dog serum, which are within the recommended range of sensitivities. 22 In a mini-validation of the Biacore method for rat serum, the sensitivity was 100 ng/mL. The criteria for a positive response in rat and dog serum in the confirmatory immunodepletion assay were ≥50% and ≥35% inhibition of the signal after preincubation with both non-PEGylated RWJ-800088 and RWJ-800088, respectively.
Safety Pharmacology, Genetic and Reproductive Toxicity, and Local Tolerance Studies
Table 4 provides an overview of studies with RWJ-800088 to assess safety pharmacology, genetic and reproductive toxicity, and local tolerance and presents study design information on species, strain, age, route of administration, duration of dosing, doses, and number of animals.
Nonclinical Toxicology Program for RWJ-800088: Safety Pharmacology, Genotoxicity, Developmental and Reproductive Toxicity, and Local Tolerance
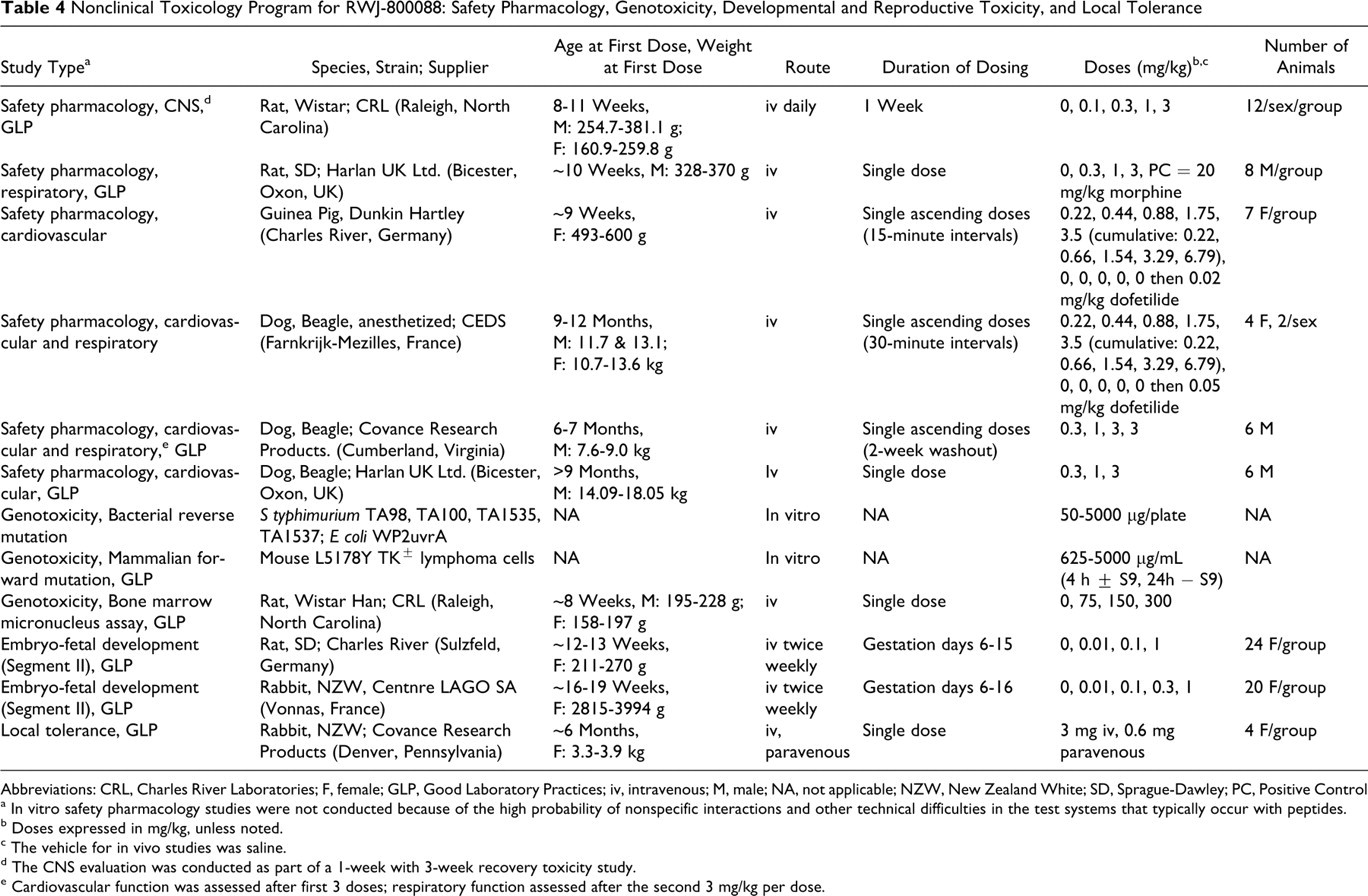
Abbreviations: CRL, Charles River Laboratories; F, female; GLP, Good Laboratory Practices; iv, intravenous; M, male; NA, not applicable; NZW, New Zealand White; SD, Sprague-Dawley; PC, Positive Control
a In vitro safety pharmacology studies were not conducted because of the high probability of nonspecific interactions and other technical difficulties in the test systems that typically occur with peptides.
b Doses expressed in mg/kg, unless noted.
c The vehicle for in vivo studies was saline.
d The CNS evaluation was conducted as part of a 1-week with 3-week recovery toxicity study.
e Cardiovascular function was assessed after first 3 doses; respiratory function assessed after the second 3 mg/kg per dose.
Safety pharmacology
In vitro safety pharmacology studies were not conducted with RWJ-800088 because of the high probability of nonspecific interactions and other technical difficulties in the test systems that typically occur with peptides. In vivo test systems were considered appropriate for RWJ-800088 safety pharmacology testing (as described in section 3.4.3 of ICH S7B [Step 2] guideline for QTc interval prolongation [in vivo nonclinical assessments]). These studies evaluated the effects of a single iv dose of RWJ-800088 on the central nervous system (through a functional observational battery) 24 and the respiratory system of rats as well as the effects of single ascending doses on the respiratory 25 and cardiovascular system of dogs 26,27 and cardiovascular system of guinea pigs. 28 The methods used in these studies were similar to those described in the citations provided above and are therefore not presented here.
Genetic toxicity
Due to its synthetic chemical nature, RWJ-800088 was evaluated for genetic toxicity in a standard battery of studies 29 : the bacterial reverse mutation assay (with and without microsomal activation), the thymidine kinase+/− locus-forward mutation assay in L5178Y mouse lymphoma cells, and an in vivo iv rat bone marrow micronucleus assay conducted as part of the 1-week toxicity study. All tests included appropriate negative and positive controls.
Reproductive toxicity
The effects of RWJ-800088 on embryo-fetal development were evaluated in rats and rabbits administered RWJ-800088 twice weekly iv during the period of organogenesis using standard nonclinical teratology study designs. 30 This iv regimen was expected to result in continued exposure to RWJ-800088 because of its long t1/2 (approximately 8-31 hours in rats and approximately 26-36 hours in rabbits for iv administration) and expected or exaggerated pharmacologic effects (eg, increased megakaryocytes, platelet counts, and mean platelet volume; bone marrow fibrosis; and hyperostosis) observed with all dosing regimens (single dose, daily dose, and once or twice weekly doses) in toxicity studies. Evaluation of maternal and embryo-fetal toxicity was based on clinical signs, body weight, food consumption, and macroscopic abnormalities in dams; pregnancy parameters (number of pregnancies, corpora lutea, implantations, early and late resorptions, and live and dead fetuses; litter size); and fetal weight, sex ratio, and external, visceral, and skeletal anomalies.
Local tolerance
A local tolerance study in rabbits investigated the potential of venous and tissue irritancy following a single iv or paravenous injection in the left or right ear based on clinical observations, Draize evaluation, and day 5 macroscopic observations and histopathology.
Statistical analyses
Study data were analyzed by classical statistical analyses used in toxicology studies (eg, Dunnett t test for parametric data and Dunn test for nonparametric data in repeat-dose studies; Fisher Exact probability test; and Mann-Whitney U test in reproductive toxicity studies) at a significance level of 5% or less.
Results
Pharmacokinetics and Toxicokinetics
The toxicokinetics of RWJ-800088 was evaluated in rats, rabbits, and dogs as part of the single- and repeat-dose toxicity and reproductive and developmental toxicity studies. A summary of the key exposures in these studies is provided in Table 5.
Summary of Key RWJ-800088 Exposures After IV Administration
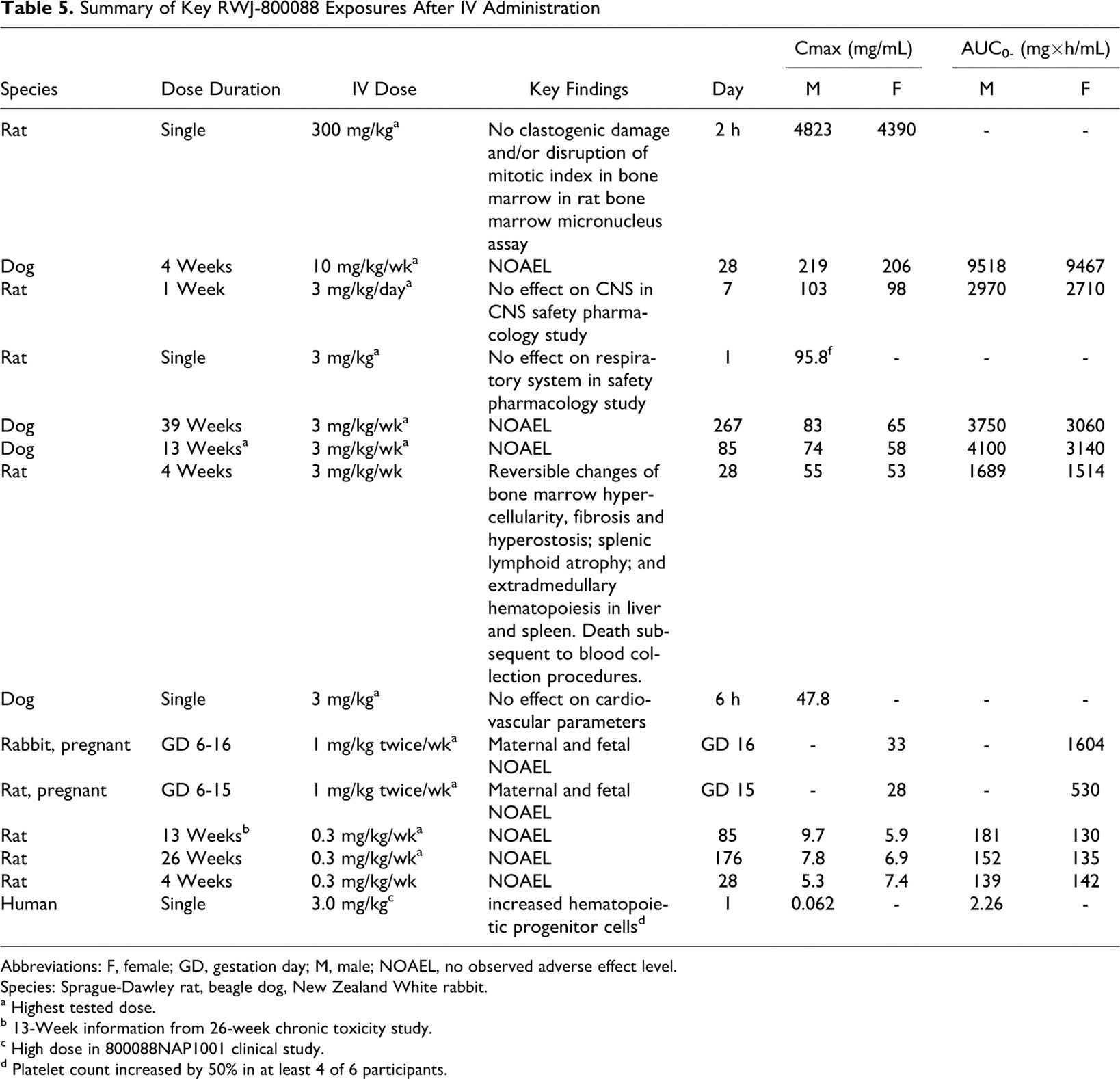
Abbreviations: F, female; GD, gestation day; M, male; NOAEL, no observed adverse effect level.
Species: Sprague-Dawley rat, beagle dog, New Zealand White rabbit.
a Highest tested dose.
b 13-Week information from 26-week chronic toxicity study.
c High dose in 800088NAP1001 clinical study.
d Platelet count increased by 50% in at least 4 of 6 participants.
RWJ-800088 has a long t1/2 (8-31 hours in rats, ~26-36 hours in rabbits, and 18-56 hours in dogs); a low clearance (CL < 10 mL/h per kg << liver blood flow [3312, 1854, and 1848 mL/h per kg in rats, dogs, and rabbits, respectively 31 ] and glomerular filtration rate [314, 368, and 432 mL/h per kg in rats, dogs, and rabbits, respectively 31 ]); and a low volume of distribution (<100 mL/kg), indicating that RWJ-800088 resides primarily in the vascular compartment. In contrast, the non-PEGylated form of RWJ-800088 is rapidly cleared (t1/2 is ~1 hour in mice 20,21 ). The limited tissue distribution and peptidic and PEGylated properties of the compound suggest that there is minimal potential for metabolic interactions. The compound did not accumulate in plasma, and none of the typical liver or pharmacokinetic changes often associated with liver enzyme induction were observed following once per week dosing in rat (6-month) and dog (9-month) chronic toxicity studies, at exposure levels >10× the lowest dose at which platelet elevations were observed. 32,33 There were no oral dosing studies conducted with RWJ-800088.
In single- and repeat-dose iv and sc studies, exposure (Cmax and AUC) increased in a dose-related manner in rats, rabbits, and dogs. At equivalent doses, exposure was 2 to 3 times higher based on AUC and 3 to 10 times higher based on Cmax after iv than sc administration. No significant gender-related differences were evident. The t1/2 values in rabbits (approximately 26-36 hours) and dogs (approximately 18-56 hours) were approximately 2 times higher than in rats (approximately 8-31 hours, with an average of approximately hours).
Single-Dose Toxicity Studies
In rats given a single iv dose of RWJ-800088 at 0.3 or 3 mg/kg, effects were limited to the expected platelet elevations (1.75× and 2.98× respectively), and megakaryocyte hyperplasia in bone marrow and spleen at 3 mg/kg. Mortality at the high dose occurred in association with blood collection on day 8. Thrombus formation (3 mg/kg) indicative of hemorrhage was observed in the ventral cervical and axillary regions, suggesting possible treatment-related causes of death.
Repeat-Dose and Investigative Toxicity Studies in Rats
1-Week IV (daily dosing) toxicity rat study
In a 1-week iv toxicity study with 3-week recovery period (daily dosing), rats were administered RWJ-800088 at 0.1, 0.3, 1, or 3 mg/kg per d. RWJ-800088 had no effect on mortality, clinical observations, body weight, body weight gain, food consumption, or ophthalmoscopic, coagulation, or urinalysis parameters. There was no functional effect on the central nervous system (functional observational battery and locomotor activity). On day 10, increases in platelets (2.2×, 3.0×, 3.2×, 3.2× vs control at 0.1, 0.3, 1, and 3 mg/kg per d, respectively), mean platelet volume (1.08-1.10× vs control at ≥1 mg/kg per d), and total white blood cell counts (1.93-3.97× vs control in neutrophil, lymphocyte, and monocyte at ≥1 mg/kg per d) were observed. There was also a modest (≤1.6× control) increase in serum aspartate transaminase (AST) at ≥1 mg/kg per d with no histological correlates in the liver. There were also no effects on coagulation parameters (APTT and PTT). Histologically, increased numbers of megakaryocytes within multiple tissues (spleen, liver, lung, adrenal gland, and bone marrow of the femur and sternum) were observed at the end of the treatment phase at all doses. Dose-related increases in bone marrow fibrosis (myelofibrosis) were observed in males at ≥0.3 mg/kg per d and in females at ≥1 mg/kg per d, with occasional hyperostosis at 3 mg/kg per d. At the end of the 3-week recovery period, fibrosis was no longer present, while hyperostosis was still present in males at ≥0.3 mg/kg per d and in females at ≥0.1 mg/kg per d. These hematology and clinical biochemistry parameters were also reversible within the 3-week recovery period, and the hyperostosis showed a trend toward recovery (the fibrotic response in the marrow appeared to be resolving by conversion to bone). Based on adverse, exaggerated pharmacologic responses of bone marrow fibrosis and hyperostosis, the no observed adverse effect level (NOAEL) for 1-week daily iv administration was 0.1 mg/kg per d in male rats and 0.3 mg/kg per d in female rats. At the NOAEL in males and females, Cmax values were 3.78 and 10.5 μg/mL, AUC0-24h values were 54.0 and 150 μg×h/mL, and AUC0- ∞ values were 96.2 and 258 μg·h/mL, respectively.
A subsequent investigative study evaluated whether a single iv dose followed by a 15-day observation period was associated with fibrosis or hyperostosis in rats administered 0.3, 1, or 3 mg/kg, the doses shown to cause these effects in the 1-week study with daily dosing. There was megakaryocytosis of the bone marrow with a dose-related increase in megakaryocyte numbers (expected pharmacologic effect) in the femur and sternum at all doses on day 4, but megakaryocytosis was not present on day 15. In a single male rat administered 3 mg/kg, moderate hyperostosis was present on day 15 in the femur but not the sternum. There were no bone lesions in males at lower doses, and females were unaffected following a single dose of 0.3, 1, or 3 mg/kg. The hyperostosis involved formation of new trabecular bone limited to the diaphyseal region of marrow cavity and was identical to the bone changes seen in the toxicology studies of longer duration. The single incidence of hyperostosis and the absence of myelofibrosis, especially on day 4, suggested that a single iv dose of 3 mg/kg of RWJ-800088 is at the threshold of the dose response required for proliferation of connective tissue in rat bone marrow.
4-Week iv and sc (weekly dosing) toxicity rat studies
In a 4-week iv (weekly dosing) mechanistic bone toxicity study with a 12-week recovery period, rats were administered once weekly iv injections of RWJ-800088 at 0, 0.1, 0.3, and 3 mg/kg per wk. RWJ-800088 had no effect on body weight, food consumption, clinical observations, and serum osteocalcin, and only a slight elevation in total alkaline phosphatase ([ALP] 1.6× control) at 3 mg/kg per wk. Adverse effects were limited to 3 mg/kg per wk and included 4 deaths subsequent to the blood collection procedure, fibrosis of the bone marrow leading to hyperostosis (Figure 2), and lymphoid depletion of the marginal zone of the spleen. The deaths were considered associated with the test compound because large numbers of platelets were noted microscopically in the vessels of the lungs (particularly the distending pulmonary capillaries). This would be expected to result in impaired circulatory flow in the lungs, which coupled with expected hypotension during blood collection may have caused circulatory collapse. New bone formation in the marrow was a consequence of early fibrosis, based on the distinct histological transition. The bone was most prominent as trabecular growth within the diaphyseal area (Figure 2). There was considerable interindividual variation in the extent of bone formation at 3 mg/kg per wk, with no fibrosis or hyperostosis being noted at lower doses. The significance of lymphoid depletion in the marginal zone of the spleen of most of the high-dose rats is unclear.
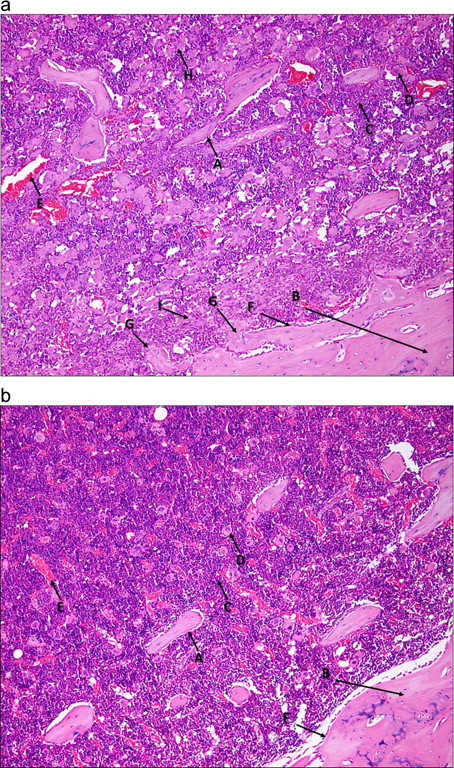
A, A diaphyseal bone marrow section treated with hematoxylin and eosin (H&E) stain at ×100 magnification from a rat treated once weekly with 3 mg/kg of RWJ-800088 for 4 weeks compared (B) a diaphyseal bone marrow section treated with H&E stain at ×100 magnification section from a control rat. A, Bony spicules. B, Cortical bone. C, Bone marrow. D, Megakaryocyte. E, Blood vessel. F, Osteoblast (increased number in treated vs control). G, Hyperostosis. H, Megakaryocytosis. Myelofibrosis (cells with spindle-shaped nuclei). Demonstrates that myelofibrosis and hyperostosis occurs in RWJ-800088-treated rats at this dose level. The are in the diaphysis, and (3) is taken at some specific high-power magnification.
Other compound-related findings were considered pharmacologic effects of the compound or related nonadverse findings. These included hematologic findings of minimally to markedly higher platelet counts (maximum mean elevations of 2.3× at 0.3 mg/kg per wk and 6.22× relative to controls at 3 mg/kg per wk); minimally higher mean platelet volume (maximum mean increase of 1.15× vs control at 3 mg/kg per wk only); minimally to mildly lower red blood cell, hemoglobin, and hematocrit values (maximum mean decreases to ~0.9× control at 3 mg/kg per wk only); and minimally to moderately higher leukocyte (neutrophils, monocytes, and lymphocytes) counts (~2.0× at 3 mg/kg per wk). The hematologic changes that were present on day 8 were generally comparable or slightly greater on days 15, 20, and 29 post dose. Corresponding histologic findings included increased megakaryocytes in the marrow, spleen, and to a lesser extent in the liver, lungs, and adrenals; decreased marrow adipose tissue; and increased erythropoiesis in the spleen and liver. These microscopic effects were generally limited to the 3 mg/kg per wk dose, although several rats at 0.3 mg/kg per wk had minimally increased megakaryocytes in the spleen. The extent of megakaryocytosis with each dose (minimal at the low and mid dose and mild to marked at the high dose) correlated with the magnitude of platelet elevation. All clinical pathology changes were normalized by day 57 (5 weeks after the final dose), and all pathology changes were reversed by day 113 (12 weeks after the final dose). Based on the pathology findings of hyperostosis and splenic marginal zone depletion at 3 mg/kg per wk, the NOAEL for weekly iv administration for 4 weeks to male and female rats was 0.3 mg/kg per wk, which corresponded to Cmax values of 7.16 and 6.80 μg/mL, and AUC0- ∞ values of 170 and 125 μg×h/mL, respectively.
In a subsequent definitive 4-week iv and sc (weekly) toxicity study with an 8-week recovery period, rats were administered once weekly iv or sc injections of RWJ-800088 at 0, 0.01, 0.3, or 3 mg/kg per wk (5 doses). RWJ-800088 administered iv or sc had no effects on body weight, body weight gain, clinical observations, food consumption, ophthalmology, or serum osteocalcin levels. There were 4 deaths after iv administration and 3 deaths after sc administration. There were no treatment-related clinical signs in these animals. The bleeding procedure was a major contributing factor for 3 deaths (1 each at 0, 0.3, and 3 mg/kg per wk iv). Pathological evaluation did not elucidate the cause of the other 4 deaths (1 at 3 mg/kg per wk iv and 3 at 3 mg/kg per wk sc); however, 3 of the deaths occurred after blood collection. In addition, 2 immunogenicity phase animals at 3 mg/kg per wk iv died after blood collection (cause unknown based on pathological evaluation), and 1 toxicokinetic animal was euthanized due to poor condition.
For both routes of administration, expected or exaggerated pharmacologic changes in hematology and serum chemistry parameters were observed that were similar in magnitude to the changes described in the 4-week study described above. Based on histological examination, animals treated at ≥0.3 mg/kg per wk showed dose-related hypercellularity (bone marrow) and lymphoid atrophy (spleen). Animals treated at ≥3.0 mg/kg per wk showed hyperostosis and fibrosis in the femur, tibia, and sternum, manifesting as a thickening of the trabeculae and extramedullary hematopoiesis (liver and spleen). All findings were reversible. There were no antibodies to RWJ-800088. The NOAEL in rats was 0.3 mg/kg per wk for both iv and sc administration for 4 weeks. At 0.3 mg/kg per wk iv in male and female rats, the Cmax was 5.32 and 7.35 μg/mL, and AUC0- ∞ was 139 and 142 μg·h/mL, respectively. At 0.3 mg/kg per wk sc in male and female rats, the Cmax was 0.39 and 0.73 μg/mL, and AUC0- ∞ was 35.3 and 55.7 μg·h/mL, respectively.
6-Month iv (weekly dosing) rat toxicity study with recovery
In a chronic iv (weekly) toxicity study, rats were administered once weekly iv injections of RWJ-800088 at 0, 0.01, 0.1, or 0.3 mg/kg per wk for 26 weeks, with an interim necropsy (13 weeks) and a 13-week recovery period. The interim evaluation was conducted with the 0.3 mg/kg per wk group, and all changes were reversible. There were no compound-related deaths. There were also no effects on body weight, food consumption, clinical observations, ophthalmology, or serum osteocalcin levels and no effects on coagulation parameters (APTT and PTT).
RWJ-800088 resulted in nonadverse, minimal-to-mild changes in hematology and serum chemistry parameters. There were increases in mean platelet counts (maximum mean elevations at 0.3 mg/kg per wk of 1.92× at 0.1 mg/kg per wk and 2.3× vs control) and mean platelet volume (~1.2× vs control at 0.1 and 0.3 mg/kg per wk), which were expected pharmacologic effects. Platelet elevations remained fairly constant throughout the course of the 6-month study. Serum chemistry changes were slight increases (< ∼2x control) in alanine aminotransferase (ALT) in week 13, AST in weeks 13 and 26, and Alkaline Phosphatase (ALP) (in weeks 1 and 13 at 0.3 mg/kg per wk), with no corresponding findings in liver pathology; increases in potassium at 0.3 mg/kg per wk in week 1 and at ≥0.1 mg/kg per wk in weeks 13 and 26 (~1.11-1.26× vs control); and increases in phosphorus at ≥0.1 mg/kg per wk in week 13, and at ≥0.01 mg/kg per wk in week 26 (~1.11-1.26× vs control). In the 13-week recovery group treated at 0.3 mg/kg per wk, the effect on mean platelet counts and serum chemistry parameters was reversible compared to controls, while mean platelet volume continued to be increased compared to controls, although it was lower compared to the treatment period.
Pathology findings were minimally to slightly increased megakaryocytes in the spleen at ≥0.1 mg/kg per wk in male rats in the 13-week interim group, increased spleen weights (absolute and relative to body weight) at ≥0.1 mg/kg per wk in both the 13-week interim and 26-week main groups (more pronounced in males), enlarged spleens (1.12× vs control) at 0.3 mg/kg per wk in males in the 13-week interim group, and minimal megakaryocytic hypercellularity of the bone marrow at ≥0.1 mg/kg per wk in both males and females in the 13-week interim and 26-week main groups. These findings were considered a pharmacologic response and, except for increased spleen weight in the 13-week interim group of males, were reversible. There were no antibodies to RWJ-800088.
The NOAEL for once weekly iv administration to male and female rats for 13 or 26 weeks was 0.3 mg/kg per wk. The NOAEL in males and females corresponded at week 13 to Cmax values of 9.70 and 5.85 μg/mL and AUC0- ∞ values of 181 and 130 μg·h/mL, and at week 26 to Cmax values of 7.76 and 6.92 μg/mL and AUC0- ∞ values of 152 and 135 μg·h/mL, respectively.
Repeat-Dose and Investigative Toxicity Studies in Dogs
1-Week iv (daily dosing) dog toxicity study
In a 1-week iv toxicity study with 3-week recovery period, dogs were administered daily injections of 0, 0.1, 0.3, 1, or 3 mg/kg per d. There were no treatment-related clinical observations or effects on body weight, body weight gain, food consumption, ophthalmology, urinalysis, organ weights, or gross pathology and no effects on coagulation parameters (APTT and PTT). On day 10, significantly higher platelet counts were noted in males and females administered 1 or 3 mg/kg per d compared with controls (1.8× and 2.1× at 1 and 3 mg/kg per d, respectively). This effect reversed by day 28, except in females administered 3 mg/kg per d. The major histopathological change noted on day 10 was the expected pharmacologic response of dose-related megakaryocytosis in the spleen and bone marrow. Megakaryocytosis was evident in the splenic red pulp region, with minimal-to-mild severity in 20 of 24 of the dogs treated with 0.1, 0.3, 1, or 3 mg/kg per d. Additionally, minimal megakaryocytosis was evident in the sternal bone marrow of 10 of 12 dogs treated with 1 or 3 mg/kg per d and in the femoral bone marrow of 9 of 24 dogs treated with 0.1, 0.3, 1, or 3 mg/kg per d. In the 3-week recovery animals, a minimal residual effect of increased megakaryocytes was evident in the splenic red pulp. Based on these findings, the NOAEL for 1-week daily iv administration in male and female dogs was 3 mg/kg per d. At the NOAEL for male and female dogs, the Cmax values were 115 and 122 μg/mL, AUC0-24h values were 1870 and 2090 μg·h/mL, and AUC0- ∞ values were 5910 and 6310 μg·h/mL, respectively.
4-Week iv and sc (weekly dosing) dog toxicity study
In a 4-week toxicity study with 8-week recovery period, dogs were administered weekly iv or sc injections of RWJ-800088 at 0, 0.3, 3, or 10 mg/kg per wk (5 doses). The 10 mg/kg per wk dose was selected to maximize identification and characterization of potential toxicity due to exaggerated pharmacology. There were no effects on body weight, body weight gain, food consumption, clinical observations, ophthalmology, serum osteocalcin levels, urinalysis, or organ weights by either route. The doses of 3 and 10 mg/kg per wk were associated with a dose-related but slight prolongation of the APTT on day 31. Similar APTT elevations (~2.6× vs control) and increases in mean platelet volume (1.2-15× vs control) were observed at 3 and 10 mg/kg per wk. Histologically, the expected pharmacologic responses of minimal-to-moderate megakaryocytosis of the splenic red pulp and bone marrow with the anticipated increased platelet counts were observed at 3 and 10 mg/kg per wk. All findings were reversible and not considered adverse. There were no differences in the incidence or severity of these findings between the 2 routes of administration. There were no antibodies to RWJ-800088 (see immunogenicity section below). Based on these findings, the NOAEL for weekly administration of RWJ-800088 for 4 weeks was 10 mg/kg per wk for both iv and sc administration. At 10 mg/kg per wk iv in male and female dogs, the Cmax was 219 and 206 μg/mL, and AUC0- ∞ was 9518 and 9467 μg·h/mL, respectively. At 10 μg/kg per wk sc in male and female dogs, the Cmax was 93.6 and 90.0 μg/mL, and AUC0-24h was 5244 and 5405 μg·h/mL, respectively.
9-Month iv (weekly dosing) dog toxicity study with recovery
In a chronic toxicity study, dogs were administered once weekly iv injections of RWJ-800088 at 0, 0.1, 1, or 3 mg/kg per wk for 13 weeks; 0 or 3 mg/kg per wk for 13 weeks followed by a 13-week recovery period; or 0, 0.1, 1, or 3 mg/kg per wk for 39 weeks. At 13 and 39 weeks, the expected findings of increased platelet count and increased mean platelet volume were observed during all stages of the dosing period at all doses. The changes were similar in magnitude to the changes observed in the 4-week dog study. The hematology changes were consistent with the microscopic findings of increased megakaryocytosis in the splenic red pulp and bone marrow at >1 mg/kg per wk at 13 weeks and at ≥0.1 mg/kg per wk at 39 weeks. Additionally, dose-related increases in intravascular megakaryocytes were observed in the lungs at ≥0.1 mg/kg per wk at both 13 and 39 weeks, and these findings were consistent with the pharmacologic action of RWJ-800088. Based on these findings, the NOAEL for once weekly iv administration for 13 or 39 weeks in dogs was 3 mg/kg per wk for both sexes. The NOAEL in male and female dogs corresponded at week 13 to Cmax values of 73.7 and 58.2 mg/mL and AUC0- ∞ values of 4100 and 3140 mg·h/mL, and at week 39 to Cmax values of 82.9 and 64.8 mg/mL and AUC0- ∞ values of 3750 and 3060 μg·h/mL, respectively.
Antigenicity and Immunogenicity
In the antigenicity study in male guinea pigs, minimal anaphylaxis was noted in 1 of 10 animals sensitized with 1 mg/kg RWJ-800088, but the level was below the criteria for induction of a positive anaphylactic response. Minimal-to-severe anaphylaxis was observed in animals treated with the positive control, ovalbumin (0.5 mg/kg). No anaphylaxis was noted in animals from the other treatment groups. Mortality (no anaphylaxis) in 2 animals treated with RWJ-800088, 1 each at 0.3 mg/kg (day 9) and 1 mg/kg (day 3), appeared to be unrelated to treatment because of the absence of a dose response. In the group receiving ovalbumin, 4 of 10 animals died after administration of the challenge dose, which was preceded by their display of severe anaphylactic signs. Based on these findings, RWJ-800088 did not induce anaphylactic responses in guinea pigs.
In a 4-week pilot study to determine whether an immunogenic response could be elicited against RWJ-800088 in male mice, no antibodies were detected against non-PEGylated RWJ-800088 in any plasma sample. Plasma samples from mice treated with rhTPO contained high levels of rhTPO-specific antibodies (>90 μg/mL by day 33 after sc and 20 to 70 μg/mL after iv administration). None of the other treatment groups exhibited detectable levels of rhTPO-specific antibodies. Markedly elevated platelet levels (7× over vehicle) were measured in mice treated sc or iv with RWJ-800088. No change in platelet count was seen after treatment with the non-PEGylated test compound. On day 33, mice given sc rhTPO had about a 70% decrease in platelets, but no change in platelets was found with iv dosing.
In a definitive immunogenicity in rats, at 3 mg/kg exposure after a sc dose was about half that after an iv dose. There were no clinical observations during the study. The deaths of 3 rats were considered a consequence of blood sampling. Serum tested negative for antibodies to RWJ-800088 or non-PEGylated RWJ-800088 in all samples from all respective dose groups. Antibodies to rhTPO were detected in all serum samples from the rhTPO-treated (positive control) groups (days 15, 22, 29, 37, 43, and 50), except at predose and day 8. Cross-reactivity of serum samples positive for rhTPO antibody with non-PEGylated RWJ-800088 on the Biacore sensor was negative in 16 of 18 samples. Two samples, one taken on day 22 and one taken on day 50, showed minimal inhibition above the arbitrarily set assay cutoff. These results, although reported as positive, are not likely cross-reacting with non-PEGylated RWJ-800088 surface. Assay validation data support a much higher assay cutoff for percentage inhibition after immunodepletion. Dose-dependent increases in platelets by iv RWJ-800088 started as early as day 4 and reached a plateau by days 11 to 12, achieving a maximum of 4 to 5 times over control (Table 6). The platelet counts began to decline at day 31 and reached control levels by day 43. The platelet effects of the RWJ-800088 3 mg/kg sc dose were comparable with those elicited by the 3 mg/kg iv dose. RWJ-800088 also produced dose-dependent increases in mean platelet volume, lymphocytes, neutrophils, monocytes, and eosinophils throughout most of the iv dosing period. Declines in these values started at day 36 and returned to normal by the end of study. Similar increases in hematology parameters and reversal were seen with the 3 mg/kg sc dose. There were no increases in platelets from rhTPO treatment. Starting on day 22, platelet levels began to decline to below control levels, suggesting that the development of rh TPO antibodies led to the development of thrombocytopenia.
Hematology Findings in a Rat 4-Week Intravenous and Subcutaneous (Twice Weekly) Study on Immunogenic Potential
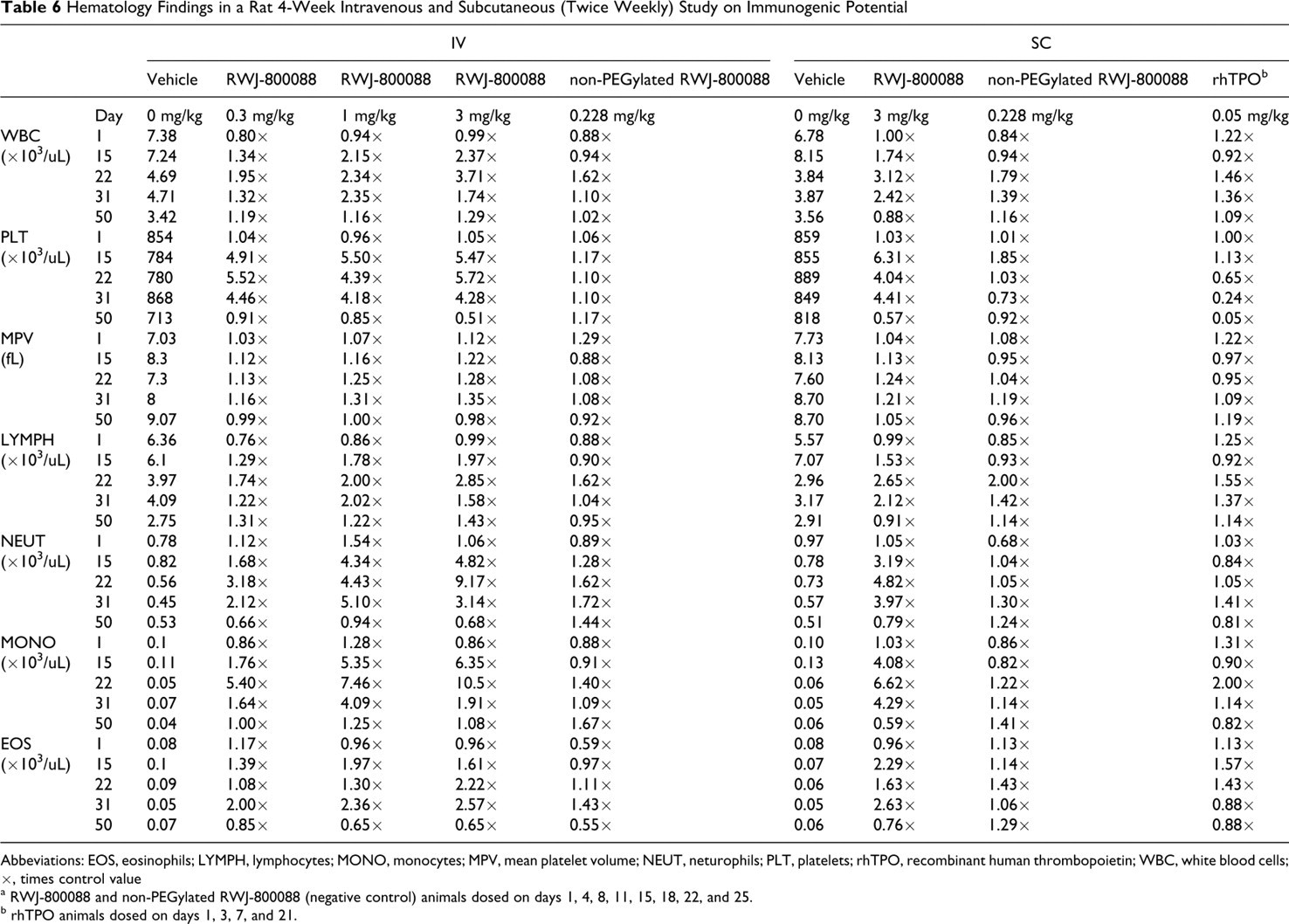
Abbeviations: EOS, eosinophils; LYMPH, lymphocytes; MONO, monocytes; MPV, mean platelet volume; NEUT, neturophils; PLT, platelets; rhTPO, recombinant human thrombopoietin; WBC, white blood cells; ×, times control value
a RWJ-800088 and non-PEGylated RWJ-800088 (negative control) animals dosed on days 1, 4, 8, 11, 15, 18, 22, and 25.
b rhTPO animals dosed on days 1, 3, 7, and 21.
Blood Compatibility
RWJ-800088 (3.5 and 10 mg/mL) caused a slight increase in optical density (15.5%), which upon visual inspection appeared related to the slightly cloudy appearance of the plasma at 10 mg/mL, but no significant hemolysis at either concentration. It did not cause flocculation at either concentration, and macroscopic or microscopic evidence of precipitation or coagulation was not detected. Based on these findings, RWJ-800088 at concentrations up to 10 mg/mL is considered compatible with human blood.
Safety Pharmacology
RWJ-800088 at doses up to 3 mg/kg iv had no effect on the CNS function of rats given single or repeat (daily for 1 week) doses (Cmax values ≥46.8 μg/mL), the respiratory system of rats given a single dose (estimated Cmax of 95.8 μg/mL based on Cmax in male rats at this dose in a single-dose toxicity study), or the respiratory and cardiovascular system of dogs given a single dose (mean plasma concentration at 6 hours postdose of 47.8 μg/mL).
In anesthetized guinea pigs at cumulative doses up to 6.79 μg/kg iv (mean plasma concentration of 184.5 μg/mL), RWJ-800088 had no consistent or notable cardiohemodynamic effects under conditions of sinus rhythm or cardiac pacing.
In anesthetized dogs, RWJ-800088 had no effect on the respiratory system at cumulative doses up to 6.79 mg/kg iv (plasma concentration ≥0.27 μg/mL) and no consistent or notable cardiovascular or cardioelectrophysiological effects at cumulative doses up to 1.54 mg/kg iv (plasma concentration ≤28.0 μg/mL at 5 minutes postdose). There were modest increases in carotid artery blood flow at cumulative doses of 3.29 mg/kg (plasma concentration ≥54.6 μg/mL) and 6.79 mg/kg (plasma concentration ≥127 μg/mL) and a slightly increased heart rate at a cumulative dose of 6.79 mg/kg. Modest enhancement of cardiac contractile performance (isovolumic left ventricular pressure [LVdp/dt max] increased up to +15% at a cumulative dose of 3.29 mg/kg vs +3% with solvent, P > 0.05), a slight increase in systemic vascular resistance (up to +11% at a cumulative dose of 3.29 mg/kg vs −2% with solvent, P > 0.05) and a trend for augmented aortic diastolic blood pressure (up to +12% at a cumulative dose of 3.29mg/kg vs −1% with solvent, P > 0.05).
Genetic Toxicity Studies
RWJ-800088 tested negative in the bacterial reverse mutation assay at concentrations up to 5000 μg/plate in 4 Salmonella typhimurium strains (TA98, TA100, TA1535, and TA1537) or in Escherichia coli strain WP2uvrA. In L5178Y mouse lymphoma cells, RWJ-800088 concentrations up to 5000 μg/mL with S9 (4-hour treatment) and without S9 (4- and 24-hour treatments) did not induce dose-dependent or ≥2-fold increases in the average forward mutation frequency at the thymidine kinase+/− (TK+/−) gene locus, compared to vehicle controls. In an in vivo rat bone marrow micronucleus test, single iv doses up to 300 mg/kg did not induce clastogenic damage or disrupt the mitotic apparatus, as there was not a statistically significant increase in the frequency of micronucleated polychromatic erythrocytes (PCEs) or decreases in the ratio of PCEs-to-normochromatic erythrocytes in the bone marrow, compared to vehicle controls. RWJ-800088 plasma concentrations in satellite toxicokinetics groups increased in a dose-related manner, were similar in males and females, and decreased between the 1- and 8-hour sampling times. At the high dose of 300 mg/kg, plasma concentrations in males and females were 4823 and 4390 μg/mL at 2 hours and 3070 and 2800 at 8 hours, respectively.
Reproductive Toxicity
There was no maternal toxicity in pregnant rats or rabbits administered 1 mg iv twice weekly during organogenesis. At fetal examination, there was no effect on body weight or sex ratio, and no external, skeletal, or visceral abnormalities were observed. Based on the absence of toxicity, the maternal and fetal NOAEL for both the rat and rabbit was >1 mg/kg for twice weekly iv administration.
Local Tolerance
In anesthetized rabbits injected iv (3 mg) or paravenous (0.6 mg) with RWJ-800088 in the left ear, there was no injection site irritation (assessed Draize scoring) and no gross or histopathological findings indicative of tissue irritancy, compared to vehicle controls (saline by either route in the right ear). In repeat-dose toxicity studies, there was no evidence of local tissue irritancy when RWJ-800088 was administered iv (bolus) or sc weekly for 4 weeks (5 doses) to rats and dogs at doses up to 3 and 10 mg/kg per wk, respectively, or iv weekly for 26 weeks to rats at doses up to 0.3 mg/kg per wk, or for 39 weeks to dogs at doses up to 3 mg/kg per wk. There were no noteworthy clinical observations at the injection sites or histopathological findings in tissue collected from the injection sites.
Discussion
Evaluation of Immunogenic Potential
No antibodies have been detected in any of the nonclinical studies indicating that the compound is not highly immunogenic in animals. 34 In a single-dose clinical study with RWJ-800088, no antibody formation against RWJ-800088 or endogenous TPO was observed. In addition, the compound bears no sequence homology to endogenous TPO and showed minimal risk of cross-reactivity to endogenous TPO suggesting that even if it did become immunogenic, an immune-mediated thrombocytopenia is unlikely. Taken together, these data suggest a low risk of antibody formation in humans.
In contrast, the development of the several previous TPO agonists, including rhTPO and PEGylated recombinant megakaryocyte growth and development factor, was hampered by the generation of neutralizing antibodies that cross-react with native TPO, resulting in paradoxical thrombocytopenia. 16,17,35,36 Amgen megakaryopoiesis protein 2 (AMG 531; Nplate® [Romiplostim]), which has no amino acid sequence homology to endogenous TPO (eTPO), was immunogenic in mice, rats, and monkeys. 37 Antibodies binding to eTPO were also observed. However, no eTPO neutralizing antibodies were detected in any of the species tested.
Underlying Mechanism for Myelofibrosis and Hyperostosis in Rats
Myelofibrosis is characterized by fibroblast proliferation with collagen deposition in the marrow cavity. Abnormal megakaryocyte proliferation and myelofibrosis are common features in chronic myeloproliferative disorders (CMPDs), which comprise a heterogeneous group of stem cell disorders. Thrombopoietin and its receptor (c-mpl) are responsible for hematopoietic stem cell survival, differentiation of hematopoietic progenitor cells, and regulation of megakaryocyte and platelet formation. 13 Problems with the regulation of expression of c-mpl have also been implicated in CMPDs. In CMPDs, there is excessive deposition of extracellular matrix proteins and collagen, which are products of fibroblasts. 38
The histological findings of myelofibrosis followed by hyperostosis in the bone marrow at 3 mg/kg in the rat 1- and 4-week studies but not in the dog toxicity studies and lower dose levels in the rat toxicity studies with RWJ-800088 suggest that there is a threshold for elevation of megakaryocyte numbers and corresponding platelet elevations. Above this threshold, intramedullary proliferation of fibroblasts and mesenchymal cells followed by osseous metaplasia occurs. Published literature supports the association between higher TPO levels and myelofibrosis and hyperostosis via elevated levels of megakaryocytes and platelets as well as activated fibrogenic growth factors and osteoprotegerin. 39 –44 It has been shown that megakaryocytes and platelets play an important role in the pathogenesis of myelofibrosis through localized release of fibrogenic cytokines in an autocrine fashion. 38 This localized stimulation of fibroblasts/mesenchymal cells is induced by growth factors such as platelet-derived growth factor (PDGF) and transforming growth factor-β (TGF-β), both of which are produced abundantly by megakaryocytes and platelets. 45 Platelet-derived growth factor is a potent mitogen for fibroblasts, and TGF-β has broader effects on many cell types including fibroblasts and osteoblasts, the primary cells responsible for the development of myelofibrosis and hyperosteosis.
Despite the evidence for a relationship between the elevated growth factor levels and myelofibrosis, 39,46 because the growth factor levels were not measured in this study, it cannot be ruled out that megakaryocyte and platelet elevations may act to produce myelofibrosis/hyperostosis through a different or additional pathway. However, in a mouse model of myelofibrosis and osteosclerosis induced by overexpession of TPO (c-mpl Ligand), development of myelofibrosis correlated well with increases in megakaryocytes, platelets, PDGF, and TFG-β. 38,47 When normal bone marrow cells were transplanted into the transgenic mice carrying the overexpressed c-mpl ligand, myelofibrosis and osteosclerosis were reversed and megakaryocytes, platelets, PDGF, and TFG-β returned to normal levels. 44 Consistent with the reversibility that was demonstrated in these mice, in the rat studies with RWJ-800088, there was reversibility of myelofibrosis during the off treatment recovery period. In addition, the dose-response and pharmacology data suggest that there is a threshold level for development of myelofibrosis and hyperostosis. Future studies could measure plasma PDGF and TFG-β along with platelet counts and megakaryocytes to better define the cytokine and hematopoietic threshold necessary for the development of myelofibrosis and hyperostosis.
In conclusion, the data suggest that the fibrosis and hyperostosis in the marrow occurs as a result of RWJ-800088 induced increases in the levels of megakaryocytes and platelets. Those increases likely act by elevating fibrogenic growth factors resulting in intramedullary proliferation of fibroblasts and mesenchymal cells followed by osseous metaplasia.
Pharmacodynamic Differences Across Species
A plot of the platelet levels versus study day in the rat and dog toxicity studies following once weekly dosing is shown in Figure 3. In both rats and dogs, platelet elevations above control levels were observed within 3 to 7 days of dosing. At doses of 0.3 and 3 mg/kg, platelet elevations were higher in rats (2.5× and 4.7× control, respectively, and reaching as high as 6.1× in the 4-week study—not shown in Figure 3) compared to dogs (1.3× and 3.1× control, respectively). In addition, the extent of megakaryocytosis in the bone marrow was moderate to marked at 3 mg/kg per wk in rats, whereas it was only minimal to slight in dogs at 10 mg/kg. There was complete recovery of platelet levels to baseline at all dose levels in both species, except at 10 mg/kg in dogs where the levels were approximately 0.7× the control values. These data demonstrate that RWJ-800088 is more potent in rats than dogs.
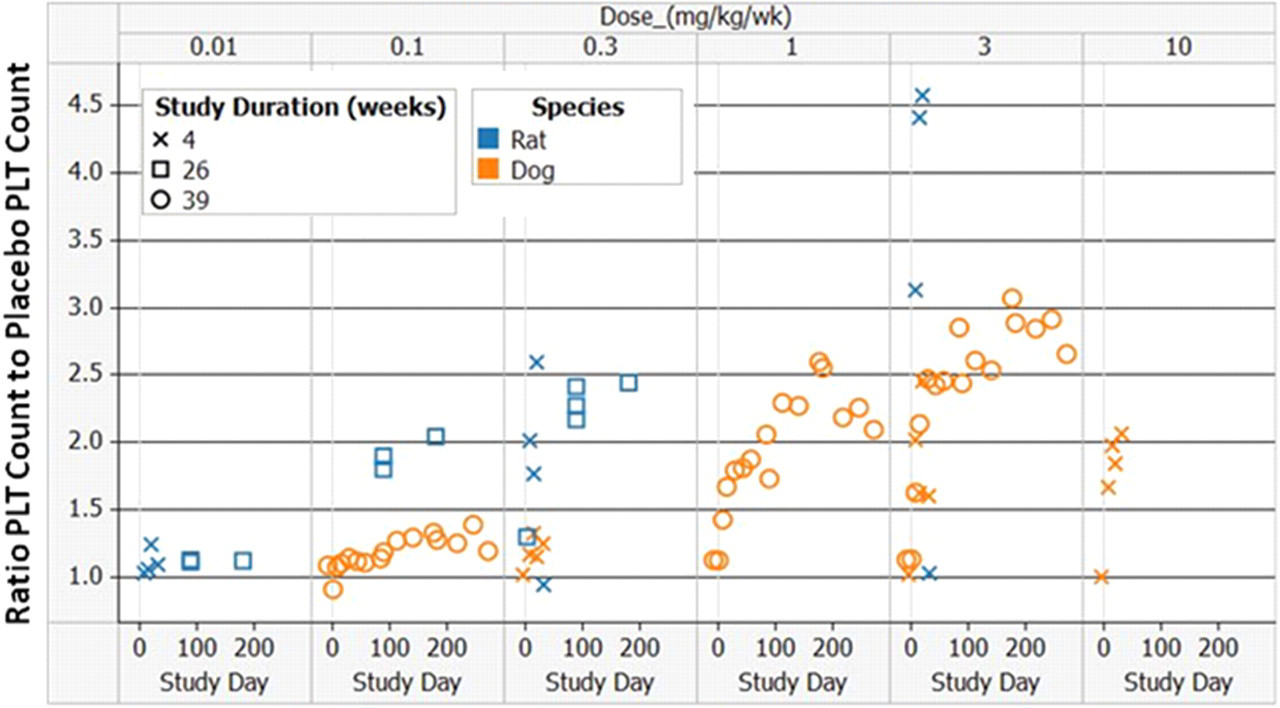
Platelet (PLT) levels versus study day in the definitive 4-week rat and dog, 26-week rat, and 39-week dog toxicity studies following once weekly dosing. Note: platelet levels returned to baseline in all recovery groups (not shown).
A plot of the platelet elevation versus dose from the human single ascending dose study in healthy volunteers 13,20 and the 4-week rat and dog toxicity studies is shown in Figure 4. The dose that results in a 2 to 3.5× elevation in humans (0.003 mg/kg) is 1000 times lower than the doses that result in a 2 to 3× elevation in rats and dogs (3 mg/kg). Consistent with the greater in vivo potency of RWJ-800088 in humans based on dose, the AUC ratio between rats and healthy human volunteers that resulted in a 3-fold increase in platelet counts in each species was 700.
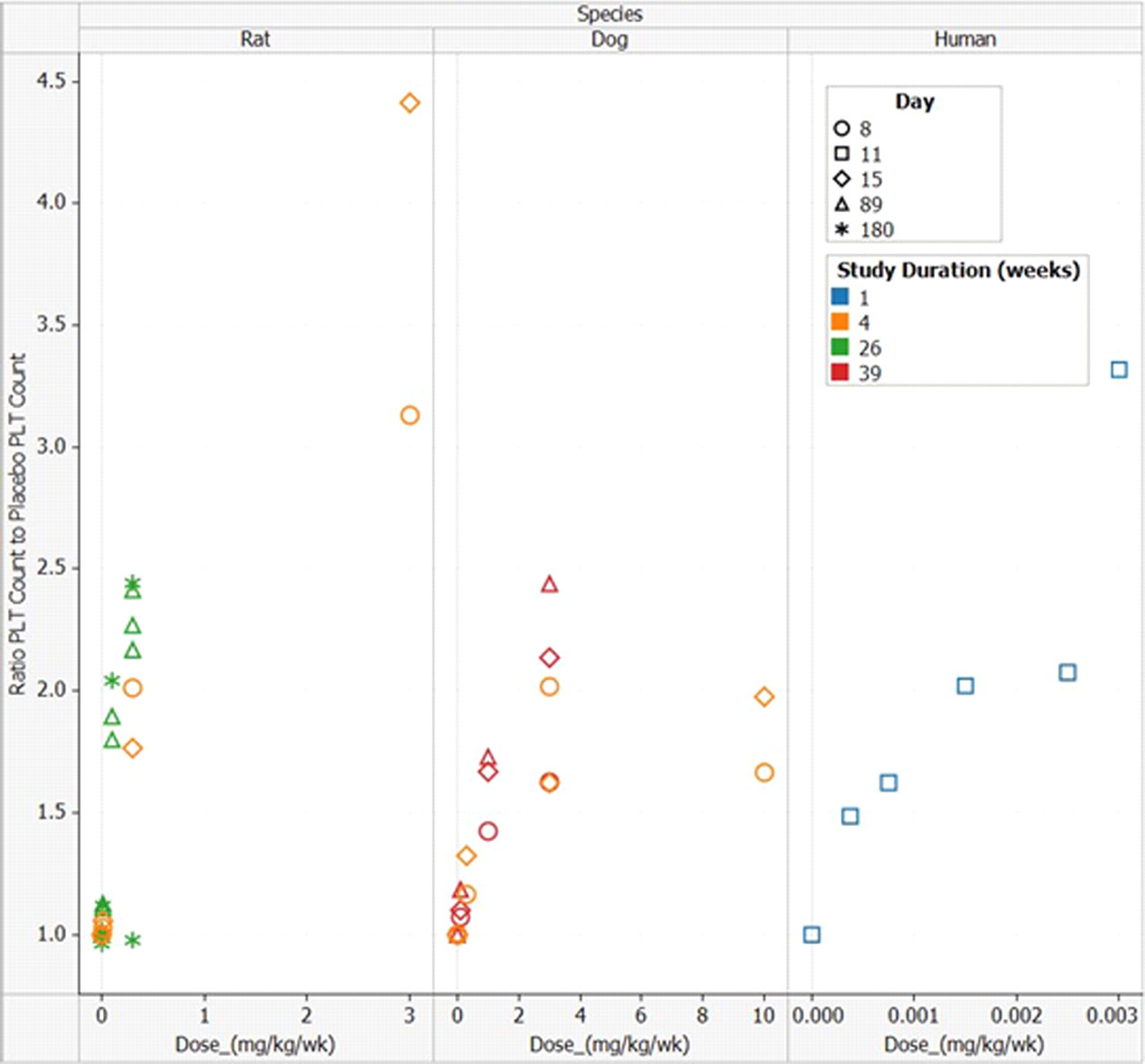
Comparison of the platelet (PLT) levels versus dose following once weekly IV administration of RWJ-800088 to rats, dogs, and humans. 19 (The shape of the symbols indicates the day of the study on which the platelet levels were measured, and the color of the symbol indicates the overall duration of the study from which the data were taken.)
A possible explanation for the occurrence of myelofibrosis in rats at 3 mg/kg per wk but not dogs at any dose is the difference in the in vivo potency with respect to megakaryocyte and platelet elevation effects. The extent of megakaryocytic hypercellularity in the bone marrow of dogs at 10 mg/kg (and in rats at <3 mg/kg) was only minimal to slight compared with moderate to marked in rats at 3 mg/kg. The platelet elevations in rats reached as high as 4.5-fold at 3 mg/kg, whereas the maximum elevation in dogs was 3-fold at 3 mg/kg and lower at 10 mg/kg. Myelofibrosis in the rats was only observed at 3× elevations and higher and was correlated with more extensive megakaryocytosis.
Potential Toxicities Related to the PEG and Peptide Structure of the Compound
Administration of PEGylated molecules may result in vacuole formation in tissues (especially choroid plexus; macrophages of liver, lymph nodes, and spleen; and renal tubular epithelium), regardless of whether the native molecule is metabolized. 33 Vacuole formation is dependent on the dose, molecular weight of the PEG, and the properties of the compound. 32,33 The tissues above were examined in the toxicity studies with RWJ-800088, and there was no evidence of any vacuole formation. The absence of vacuole formation is consistent with data showing that the kidneys have sufficient capacity to clear hydrophilic proteins, polymers, and polymerprotein conjugates such as 50 kDa PEG by renal ultrafiltration, with a t½ of 16 hours in mice. 32,33
Potential Toxicities Related to the Pharmacodynamic Effect on Platelet Levels
Based on its effects on platelet levels, potential effects on blood coagulation were assessed as part of the in vitro and in vivo toxicity studies. No toxicologically relevant effects were observed on clotting times in any of the toxicity studies or following administration to humans. 19
Safety Margins and Implications for Monitoring of Clinical Safety
Safety pharmacology
Safety pharmacology studies demonstrate that RWJ-800088 has low potential for inducing central nervous system, respiratory, or cardiovascular functional effects, with large margins relative to the clinical exposure (Table 5). Although not seen in conscious dogs, the modest increase in common carotid arterial blood flow in the anesthetized dog is likely a hemodynamic consequence of the modest enhancement of cardiac contractile performance (LVdp/dt max). This effect, together with a slight increase in systemic vascular resistance and, consequently, a trend for augmented aortic diastolic blood pressure, is likely to have resulted in the increase in the pressure wave in the common carotid artery. This observation is unlikely to have any relevant effect on cerebral capillary blood flow because in anesthetized dogs only 2% to 3% of the carotid blood flow is distributed to the brain, whereas 40% to 50% is shunted to the venous side via venous anastomoses localized in extracranial structures, such as head, skin, ears, tongue, and nasal mucosa. 48 At doses with no notable effect in the anesthetized dog, exposure values were 452 times the Cmax value (0.062 μg/mL) in healthy male participants, given the high dose of 3 μg/kg in a single ascending dose clinical study. 19
Toxicity studies
RWJ-800088 concentrations that resulted in increased platelet levels following exposure to RWJ-800088 in 4-week and chronic toxicity studies in rats and dogs and exposure to RWJ-800088 after iv administration to healthy human male participants (0.375 to 3 μg/kg) are summarized in Figure 4. Safety margins were calculated by comparing exposure at the NOAEL iv doses in the repeat-dose rat and dog studies (once per week) and developmental toxicity rat and rabbit studies (twice per week) with exposure at 3 mg/kg in the single ascending dose clinical study, which is the highest anticipated human clinical dose (Table 5). For repeat-dose studies in rats, the most sensitive species, the exposure margins based on Cmax and AUC were ≥102 and ≥62, respectively, for a clinical dose of 3 μg/kg. For developmental toxicity studies, the safety margins based on AUC were 235 and 710, respectively, for a clinical dose of 3 μg/kg.
While these large exposure margins provide some assurance of safety, because of the difference in the in vivo potency between the nonclinical species and humans, exposure margins alone are not sufficient to protect against the potential for TPO-induced myelofibrosis/hyperostosis in humans. However, based on the correlation between platelet elevation and myelofibrosis/hyperostosis in the nonclinical studies, platelet counts represent a safety marker for use in clinical studies. The data in the rat and dog studies demonstrate that below a platelet elevation of 3×, the extent of megakaryocyte elevation is limited and there is no evidence of myelofibrosis/hyperostosis. In addition, despite in vivo potency differences between rats and humans (1000 times lower in humans to achieve a 2-3× elevation), the rat offers a sensitive model for setting a conservative cutoff for platelet elevation that is expected to correlate with the risk of myelofibrosis/hyperostosis in humans. The peak levels of platelets are reached between days 4 and 6 in rats 49 compared to 12 and 14 days in humans. 50 Consequently, higher platelet turnover may result in a higher level of megakaryocytosis in rats compared to humans and, hence, an earlier onset of megakaryocyte-related toxicities like myelofibrosis and associated disorders (bone formation) after TPO dosing.
Conclusion
The nonclinical safety studies presented herein as well as the results from safety pharmacology and genetic and reproductive toxicology studies demonstrate that RWJ-800088 is safe and well tolerated with large safety margins for a clinical dose of 3 μg/kg. The immunogenicity results suggest that RWJ-800088 has a low risk for antibody formation in humans. The main findings in the toxicity studies were dose-related increases in megakaryocytes in spleen and bone marrow, platelet counts and mean platelet volume, and bone marrow fibrosis and hyperostosis in rats (only in the 1-week and 4-week studies) but not dogs. Other findings included increased spleen weights in rats and increased intravascular platelets in the lung vasculature of dogs (13- and 39-week studies). These findings were expected pharmacologic responses and were reversible. The mechanism for the myelofibrosis and hyperostosis is likely due to the exaggerated pharmacologic effect of excessive stimulation of megakaryocytes by TPO. When circulating platelets are elevated by more than 3×, there are increased megakaryocytes in the bone marrow and fibrogenic growth factors, which result in intramedullary proliferation of fibroblasts and mesenchymal cells followed by osseous metaplasia. The large exposure ratios and mechanistic understanding of the exaggerated pharmacological effects support the use of platelet counts as a parameter to monitor and prevent potential adverse bone-related effects in humans.
Footnotes
Acknowledgments
The authors would like to thank our colleagues who contributed to the studies presented in the manuscript and in particular Tara Huth for her help in the preparation of the manuscript and Michael F. Kelley, LeRoy Hall, and Jim Proctor for helpful input.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) declared the following financial support for the research and/or authorship of this article: funding for this work was provided by Johnson & Johnson Research & Development, LLC.
