Abstract
Background
Sepsis-associated thrombocytopenia (SAT) is a common complication in critically ill patients and is associated with increased disease severity and adverse outcomes. Platelet transfusion is the main supportive therapy but provides limited and transient benefit. Evidence regarding thrombopoietin-receptor agonists in SAT remains scarce.
Objectives
To evaluate the association between avatrombopag administration and platelet recovery, clinical outcomes, and safety in critically ill patients with SAT.
Methods
We conducted a single-center, retrospective observational cohort study of adult ICU patients with SAT between May 2022 and May 2024. Patients were categorized according to avatrombopag exposure during the ICU stay. The primary outcome was platelet recovery at day 14. Secondary outcomes included platelet recovery at days 7 and 10, 28-day mortality, and ICU length of stay. Confounding was addressed using propensity score–based inverse probability of treatment weighting (IPTW). Associations were assessed using weighted regression models, with prespecified sensitivity and subgroup analyses.
Results
Among 256 included patients, 119 received avatrombopag and 137 did not. After IPTW adjustment, avatrombopag use was independently associated with higher odds of platelet recovery at day 14 (odds ratio 3.58, 95% confidence interval 1.31-9.79). No consistent benefit was observed for early platelet recovery at day 7 or day 10. Avatrombopag use was not associated with reduced 28-day mortality. Rates of bleeding and thrombotic events were similar between groups. However, avatrombopag exposure was consistently associated with a longer ICU length of stay. Subgroup analyses suggested greater platelet recovery benefit in patients with higher platelet transfusion requirements and greater illness severity.
Conclusions
In critically ill patients with SAT, avatrombopag administration was associated with improved delayed platelet recovery without increased bleeding or thrombotic risk. This hematologic benefit did not translate into improved short-term survival and was accompanied by prolonged ICU stay.
Keywords
Background
Sepsis-associated thrombocytopenia (SAT) is a common hematologic complication in critically ill patients and has consistently been associated with increased disease severity, organ dysfunction, and adverse clinical outcomes. 1 Platelets play a central role in the pathophysiology of sepsis through their involvement in hemostasis, inflammation, endothelial interaction, and immunothrombosis. 2 Accordingly, a decline in platelet count during sepsis is often interpreted as a marker of systemic dysregulation and disease progression rather than an isolated laboratory abnormality.
The mechanisms underlying SAT are heterogeneous and multifactorial. In addition to impaired platelet production, accelerated platelet consumption driven by inflammatory activation, immune-mediated clearance, and disseminated intravascular coagulation–like processes contribute substantially to thrombocytopenia in sepsis.2,3 This pathophysiologic complexity complicates therapeutic decision-making and raises uncertainty regarding whether pharmacologic correction of platelet counts can meaningfully improve clinical outcomes in critically ill patients.
Current management of SAT primarily relies on supportive strategies, including treatment of the underlying infection and platelet transfusion in selected cases. However, platelet transfusion is associated with several limitations, such as short-lived efficacy, transfusion-related complications, and uncertain impact on clinically relevant outcomes. 1 These challenges have prompted interest in alternative approaches that stimulate endogenous platelet production rather than transiently replacing circulating platelets.
Thrombopoietin-receptor agonists (TPO-RAs) represent a pharmacologic strategy designed to enhance megakaryopoiesis and platelet production. Avatrombopag, an orally administered TPO-RA, has demonstrated efficacy in increasing platelet counts in immune thrombocytopenia and chronic liver disease–associated thrombocytopenia, with a generally favorable safety profile.4,5 Nevertheless, evidence supporting the use of avatrombopag in the setting of sepsis remains extremely limited. The pathophysiology of SAT differs substantially from that of non-infectious thrombocytopenia, and the timing, magnitude, and clinical relevance of platelet responses to TPO-RAs in critically ill septic patients are poorly defined.
Importantly, existing studies of TPO-RAs have largely focused on platelet count responses, with limited evaluation of time-dependent platelet recovery, survival, safety outcomes, or ICU resource utilization. In the context of sepsis, where thrombocytopenia may reflect disease severity rather than a direct therapeutic target, it remains unclear whether pharmacologic stimulation of thrombopoiesis confers meaningful clinical benefit or introduces unintended trade-offs.1,6
Given these knowledge gaps, we conducted a retrospective observational cohort study in critically ill patients with sepsis-associated thrombocytopenia to evaluate the association between avatrombopag administration and platelet recovery over time. Using propensity score–based methods to account for confounding, we aimed to assess the effects of avatrombopag on platelet recovery at predefined time points, survival, safety outcomes, and ICU length of stay. By integrating hematologic, clinical, and resource-related endpoints, this study sought to provide real-world evidence to inform the potential role of avatrombopag in the management of SAT.
Methods
Study Design and Setting
This was a single-center, retrospective, observational cohort study conducted in the Department of Critical Care Medicine at Southern Medical University Nanfang Hospital, a tertiary academic medical center in China. Consecutive adult patients admitted to the intensive care unit (ICU) between May 2022 and May 2024 with a diagnosis of sepsis complicated by thrombocytopenia were screened for eligibility.
Study Population
Patients were classified into an avatrombopag group or a non-avatrombopag group according to whether avatrombopag was administered at any time during the ICU stay. Sepsis was diagnosed in accordance with the Sepsis-3 definition, namely suspected or documented infection accompanied by an acute increase in the Sequential Organ Failure Assessment (SOFA) score of at least 2 points.
Sepsis-associated thrombocytopenia (SAT) was defined as a platelet count below 100 × 109/L or a relative decrease of 30% or more from baseline platelet levels following the onset of sepsis. Alternative causes of thrombocytopenia, including hematologic malignancies, inherited platelet disorders, and definite drug-induced thrombocytopenia, were carefully excluded based on clinical records.
Avatrombopag was administered at an initial dose of 40 mg once daily. The treatment was initiated when patients developed sepsis-associated thrombocytopenia, defined as a platelet count below 100 × 109/L or a relative decrease in platelet count of ≥30%. The treatment continued until platelet recovery or ICU discharge, whichever occurred first. The dosage of avatrombopag was adjusted based on clinical response and patient tolerance, in line with the recommended guidelines for treatment of thrombocytopenia.
Eligibility Criteria
Adult patients aged 18 years or older who were admitted to the ICU, fulfilled the Sepsis-3 criteria, and developed thrombocytopenia during their ICU stay were eligible for inclusion. Patients were excluded if thrombocytopenia was primarily attributable to hematologic diseases, active major bleeding, or confirmed drug-induced causes such as heparin-induced thrombocytopenia. Additional exclusion criteria included a history of hematopoietic stem cell or solid organ transplantation, receipt of chemotherapy or radiotherapy within the preceding two weeks, advanced liver cirrhosis with hypersplenism, end-stage liver disease, end-stage renal disease requiring long-term dialysis prior to ICU admission, or severe immunosuppression due to active malignancy treatment or immunosuppressive therapy other than low- to moderate-dose corticosteroids (≤2 mg/kg/day prednisone or equivalent). Patients with an ICU length of stay shorter than 24 h, those who were pregnant or lactating, and those concurrently enrolled in another interventional clinical trial were also excluded.
Use of antiplatelet agents or anticoagulant therapy was not considered an exclusion criterion; instead, such treatments were recorded in detail and adjusted for in the subsequent statistical analyses.
Data Collection and Intervention
Baseline demographic characteristics, including age and sex, were extracted from the electronic medical records within the first 24 h of ICU admission. Information on comorbidities, sites of infection, and disease severity was collected, with illness severity quantified using the Acute Physiology and Chronic Health Evaluation II (APACHE II) score and the SOFA score. Laboratory data included complete blood counts, coagulation parameters, renal and hepatic function tests, and inflammatory biomarkers, as detailed in supplemental Table 1.
Data regarding concomitant medications, blood product transfusions, and life-support interventions, including mechanical ventilation and continuous renal replacement therapy (CRRT), were also collected. For laboratory variables measured more than once on the same day, the most extreme value was retained for analysis in order to reflect the worst physiological status. Serial platelet counts were recorded daily for up to 14 days following cohort entry or until ICU discharge or death, whichever occurred first.
Outcomes
The primary outcome of interest was platelet recovery at day 14 after cohort entry. Platelet recovery was evaluated using two complementary measures: the platelet recovery value, defined as the absolute change in platelet count from baseline, and the platelet recovery rate, defined as attainment of a platelet count of at least 100 × 109/L or an increase of at least 50 × 109/L from baseline.
Secondary outcomes included platelet recovery at days 7 and 10, 28-day all-cause mortality, and ICU length of stay. In addition, a composite endpoint was defined as platelet recovery among patients who survived during the treatment period.
Statistical Analysis
Continuous variables are presented as means with standard deviations or medians with interquartile ranges, depending on their distribution, and were compared using Student's t tests or Mann–Whitney U tests, as appropriate. Categorical variables are summarized as counts with percentages and were compared using chi-square tests or Fisher's exact tests. All statistical tests were two-sided, and a P value of less than 0.05 was considered statistically significant.
Given the observational nature of the study, propensity score weighting was applied to reduce confounding by indication. Propensity scores were estimated using logistic regression models incorporating clinically relevant baseline variables, including demographic characteristics, comorbidities, disease severity scores, laboratory parameters, transfusion exposure, and concomitant treatments. The primary weighting approach was inverse probability of treatment weighting (IPTW). Covariate balance before and after weighting was assessed using standardized mean differences, with values below 0.10 indicating acceptable balance.
Associations between avatrombopag exposure and clinical outcomes were evaluated using logistic regression models for binary outcomes, linear regression models for continuous outcomes, and Cox proportional hazards models for time-to-event outcomes. Effect estimates are reported as odds ratios, regression coefficients, or hazard ratios with corresponding 95% confidence intervals.
Handling of Missing Data
Missing baseline covariates were addressed using multiple imputation with chained equations. For longitudinal platelet measurements, last observation carried forward was applied when follow-up values were unavailable due to ICU discharge or death (Figure 1).
Pattern and proportion of missing data across baseline variables. Visualization of the proportion and patterns of missing data across baseline variables included in the propensity score model. The left panel displays the proportion of missing values for each variable, while the right panel illustrates the combinations of missingness across variables, with red cells indicating missing values and blue cells indicating observed data. Variables with higher rates of missingness included procalcitonin (PCT), interleukin-6 (IL-6), and N-terminal pro-B-type natriuretic peptide (Pro-BNP). The observed missing data patterns support the use of multiple imputation prior to propensity score estimation and subsequent inverse probability of treatment weighting (IPTW) analyses.
Sensitivity and Subgroup Analyses
Several prespecified sensitivity analyses were performed to assess the robustness of the primary findings. These included alternative propensity score weighting strategies, namely standardized mortality ratio weighting and standardized morbidity prevalence weighting, using the same covariate set. In addition, two multivariable regression models were fitted on the unweighted cohort. The first model adjusted for age, diabetes mellitus, APACHE II score, baseline platelet count, and platelet transfusion, while the second model further adjusted for sex, comorbidities, mechanical ventilation, and CRRT.
Prespecified subgroup analyses were conducted according to baseline platelet count (≤50 × 109/L vs >50 × 109/L), total platelet transfusion dose (≥2 units vs <2 units), and baseline APACHE II score (≥25 vs <25).
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Southern Medical University Nanfang Hospital. Given the retrospective design and the use of anonymized data, the requirement for informed consent was waived.
Results
Study Population
Between May 2022 and May 2024, a total of 537 patients with sepsis-associated thrombocytopenia were screened in the Department of Critical Care Medicine at Nanfang Hospital, Southern Medical University. After applying predefined exclusion criteria, including age <18 years (n = 53), hematologic malignancies (n = 110), advanced liver cirrhosis (n = 49), and bleeding disorders (n = 68), 256 patients were included in the final analysis. Among them, 119 patients received avatrombopag during ICU treatment, whereas 137 did not (Figure 2).
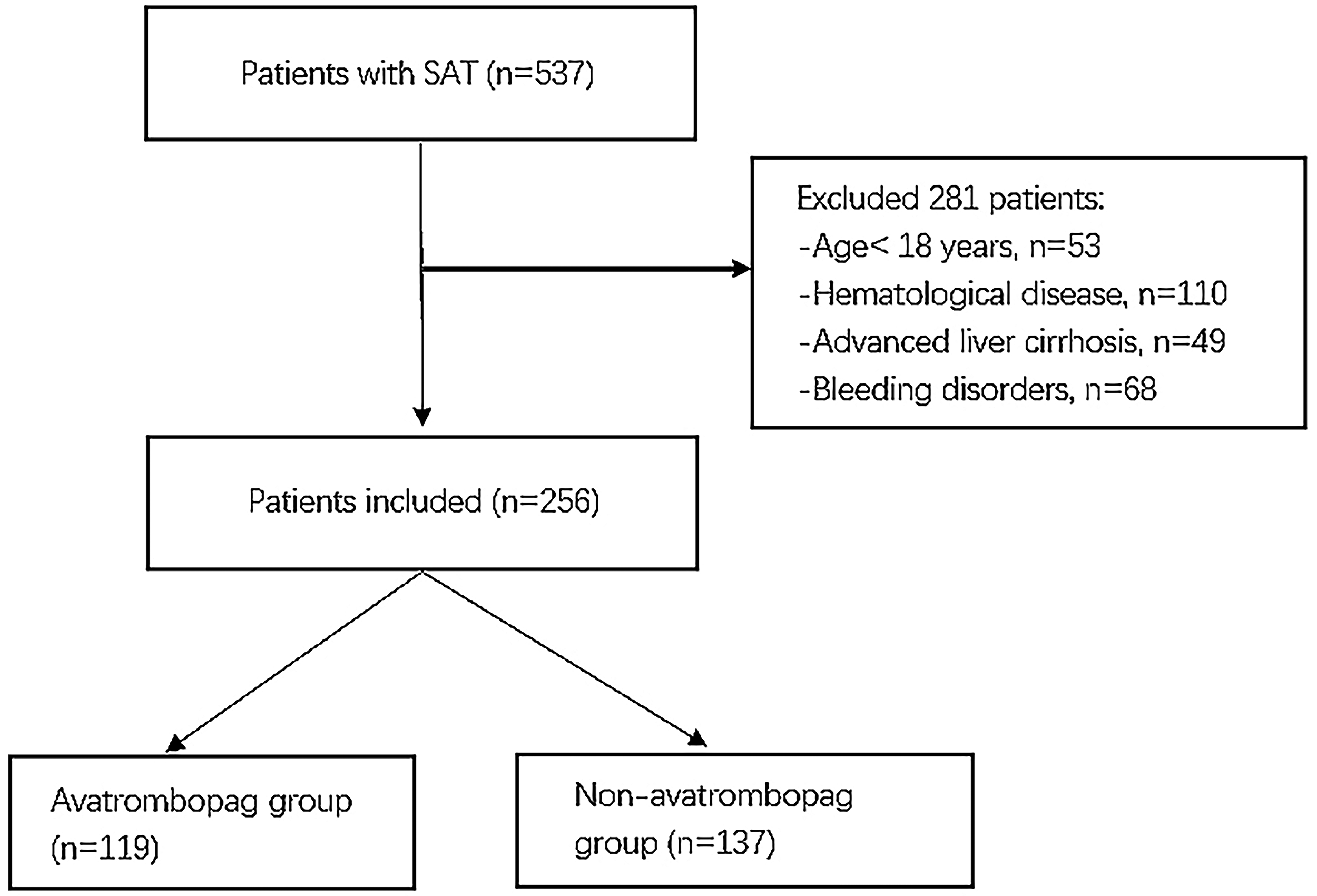
Study flow diagram of patient selection and treatment allocation.
Baseline Characteristics and Covariate Balance
Baseline demographic characteristics, comorbidities, infection sites, illness severity, laboratory parameters, and ICU interventions are summarized in Table 1.
Baseline Characteristics of the Study Population Before and After IPTW.

Continuous variables are presented as median (interquartile range), and categorical variables as number (percentage). Standardized mean differences (SMDs) < 0.10 indicate adequate covariate balance after weighting.
Abbreviations: IPTW, inverse probability of treatment weighting; SMD, standardized mean difference; ICU, intensive care unit; CRRT, continuous renal replacement therapy; APACHE II, Acute Physiology and Chronic Health Evaluation II; SOFA, Sequential Organ Failure Assessment.
*The result which P-value < 0.05 markede in the table.
Before weighting, patients in the avatrombopag group exhibited higher disease severity, as reflected by higher APACHE II scores (median 26 vs 22, P = 0.004), lower baseline platelet counts, and greater exposure to platelet transfusions and thrombopoietic agents (P < 0.001). Differences were also observed in inflammatory markers and coagulation parameters. Moreover, the avatrombopag group had a higher proportion of patients who received platelet transfusions and median platelet transfusion dose compared to the non-avatrombopag group (80.67% vs 59.12%, P < 0.001; 2.0 units vs 1.0 units, P < 0.001).
After inverse probability of treatment weighting (IPTW), visual inspection of the propensity score distribution confirmed sufficient overlap between groups (Figure 3), baseline covariates were well balanced between the two groups, with most standardized mean differences (SMDs) below 10%. Although several variables showed SMDs exceeding 15%, key prognostic factors—including platelet counts, transfusion exposure, and thrombopoietin-receptor agonist use—remained adequately balanced (Table 1, Figure 4).

Distribution of propensity scores for patients treated with and without avatrombopag. The histogram illustrates the overlap in propensity score distributions between the avatrombopag group (group = 1) and the non-avatrombopag group (group = 0) based on the estimated probability of receiving avatrombopag. Visual inspection demonstrates adequate common support between groups, indicating sufficient overlap for inverse probability of treatment weighting (IPTW) analyses. This overlap supports the validity of subsequent weighted comparisons of platelet recovery and clinical outcomes.

Standardized mean differences of baseline covariates across analytic methods. Standardized mean differences (SMDs) of baseline covariates between the avatrombopag and non-avatrombopag groups before adjustment and after weighting. Covariate balance is shown for the unadjusted cohort, the stabilized inverse probability of treatment weighting (IPTW) cohort, and the unstabilized standardized mortality ratio weighting (SMRW) cohort.
Primary Outcome: Platelet Recovery at Day 14
In the unadjusted analysis, avatrombopag administration was associated with a higher platelet recovery rate by day 14 compared with the non-avatrombopag group (44.5% vs 36.5%; OR 1.40, 95% CI 1.33-1.47; P < 0.001) (Table 2).
Primary and Secondary Study Outcomes.

Outcome data are derived from the unweighted cohort. Platelet recovery rate was defined as the proportion of patients whose platelet count reached ≥100 × 109/L or increased by ≥50 × 109/L from baseline. Platelet recovery rate among survivors refers to platelet recovery among patients who survived during the observation period.
aEffect size for 28-day mortality is expressed as hazard ratio (HR). bEffect sizes for platelet recovery rate, platelet recovery rate among survivors, and adverse bleeding and thrombotic events are expressed as odds ratios (ORs). cEffect sizes for platelet count, platelet recovery value, and ICU length of stay are expressed as β coefficients.
*The result which P-value < 0.05 markede in the table.
Abbreviations: ICU, intensive care unit; IQR, interquartile range.
This association persisted after adjustment. In the IPTW-weighted analysis, avatrombopag use was associated with significantly increased odds of platelet recovery at day 14 (OR 3.58, 95% CI 1.31-9.79; P = 0.013). Similar results were observed using SMRW and multivariable regression models (Table 3).
Association Between Avatrombopag Administration and Clinical Outcomes After Adjustment.

Associations between avatrombopag use and clinical outcomes were assessed using propensity score–based weighting methods and multivariable regression models. IPTW and SMRW analyses were performed using the same covariate set. Model 1 was adjusted for age, diabetes mellitus, APACHE II score, baseline platelet count, and platelet transfusion. Model 2 was additionally adjusted for sex, comorbidities, mechanical ventilation, and CRRT.
*The result which P-value < 0.05 markede in the table.
Abbreviations: IPTW, inverse probability of treatment weighting; SMRW, standardized mortality ratio weighting; ICU, intensive care unit; CRRT, continuous renal replacement therapy; APACHE II, Acute Physiology and Chronic Health Evaluation II.
Survival-Adjusted Platelet Recovery
When restricting the analysis to patients surviving to each assessment point, avatrombopag use was associated with a higher probability of survival-adjusted platelet recovery at day 14. The IPTW-adjusted odds ratio was 3.14 (95% CI 1.12-8.74; P = 0.029). This finding remained consistent across multivariable models (Table 3).
Secondary Outcomes
Early Platelet Response
At day 7, avatrombopag use was associated with lower platelet counts (β −29.9 × 109/L, 95% CI −49.7 to −10.1; P = 0.003) and a lower platelet recovery rate (OR 0.94, 95% CI 0.89-0.99; P = 0.03) in unadjusted analyses (Table 2). These early differences were not consistently observed after adjustment.
No statistically significant differences in platelet counts or recovery rates were observed at day 10 across most analytical approaches.
Mortality
There was no significant difference in 28-day mortality between groups in the primary analyses. The IPTW-adjusted hazard ratio for mortality was 0.84 (95% CI 0.56-1.26; P = 0.511) (Table 3). Kaplan–Meier survival curves were similar between groups (Figure 5).

Kaplan–Meier survival curves by analytic approach. Kaplan–Meier survival curves comparing 28-day survival between the avatrombopag and non-avatrombopag groups. Panel A shows the unweighted analysis, panel B shows survival curves estimated using unstabilized weighting, and panel C shows survival curves estimated using stabilized inverse probability of treatment weighting (IPTW). Shaded areas represent 95% confidence intervals.
A sensitivity analysis using SMRW suggested a lower mortality risk associated with avatrombopag (HR 0.63, 95% CI 0.42-0.95; P = 0.046); however, this association was not observed in the prespecified primary analyses.
ICU Length of Stay and Safety Outcomes
Avatrombopag use was consistently associated with a
The incidence of adverse bleeding events and thrombotic events did not differ significantly between groups in any analysis (Tables 2–3).
Subgroup Analyses
Subgroup analyses stratified by platelet transfusion dose and illness severity are presented in Figure 6 and Table 4.

Subgroup analyses of platelet recovery and ICU length of stay. Subgroup analyses of the association between avatrombopag use and clinical outcomes. Forest plots show odds ratios (ORs) for platelet recovery at day 14 (A) and survival-adjusted platelet recovery at day 14 (B), and mean differences (MDs) for ICU length of stay (C), across prespecified subgroups stratified by illness severity and platelet transfusion exposure. Points indicate effect estimates and horizontal lines represent 95% confidence intervals.
Primary/Secondary Study Outcomes in Patients with Platelet Transfusion Dose Over 2 U and APACHE II Over Than 25.

This table presents outcomes in a prespecified high-risk subgroup defined by higher platelet transfusion requirements and greater illness severity. Platelet recovery rate was defined as the proportion of patients whose platelet count reached ≥100 × 109/L or increased by ≥50 × 109/L from baseline.
aEffect size for 28-day mortality is expressed as hazard ratio (HR). bEffect sizes for platelet recovery rate, platelet recovery rate among survivors, and adverse bleeding and thrombotic events are expressed as odds ratios (ORs). cEffect sizes for platelet count, platelet recovery value, and ICU length of stay are expressed as β coefficients.
*The result which P-value < 0.05 markede in the table.
Abbreviations: ICU, intensive care unit; APACHE II, Acute Physiology and Chronic Health Evaluation II; IQR, interquartile range.
Among patients receiving ≥2 units of platelet transfusion, avatrombopag use was associated with a higher platelet recovery rate at day 14 (adjusted OR 3.15, 95% CI 1.25-7.96; P = 0.015). In this high-risk subgroup, avatrombopag was also associated with a longer ICU stay after adjustment (β 6.3 days, 95% CI 0.3-12.4; P = 0.042).
Discussion
In this retrospective cohort of critically ill patients with sepsis-associated thrombocytopenia (SAT), avatrombopag administration was independently associated with a higher rate of platelet recovery at day 14. This association was consistent across multiple adjustment strategies, including inverse probability of treatment weighting and multivariable regression models. In contrast, avatrombopag use was not associated with a robust reduction in 28-day mortality. Importantly, no increase in bleeding or thrombotic events was observed, whereas avatrombopag exposure was consistently associated with a prolonged length of stay in the intensive care unit (ICU). Together, these findings extend the limited real-world evidence regarding thrombopoietin-receptor agonists in critically ill patients with SAT and highlight a complex balance between hematologic benefit and resource utilization. 1
Thrombocytopenia is a frequent complication of sepsis and has been repeatedly associated with greater illness severity, organ dysfunction, and adverse outcomes. Rather than representing a single pathological entity, SAT encompasses a heterogeneous spectrum of mechanisms, including inflammation-driven platelet consumption, immune-mediated platelet clearance, endothelial activation, and impaired platelet production.7,8 Consequently, platelet count is widely regarded as both a prognostic marker and a potential therapeutic target in sepsis, although the causal relationship between platelet correction and clinical outcomes remains uncertain.
From a pathophysiological perspective, the delayed platelet recovery observed with avatrombopag in our study is biologically plausible. During the early phase of sepsis, immunothrombosis and disseminated coagulation activation may result in rapid platelet consumption that outweighs compensatory thrombopoiesis. In this setting, pharmacological stimulation of platelet production is unlikely to produce an immediate increase in circulating platelet counts. Thrombopoietin-receptor agonists primarily act by promoting megakaryocyte maturation and platelet production, a process that requires several days to translate into measurable hematologic effects. As systemic inflammation and consumptive processes subside, the impact of enhanced thrombopoiesis may become more apparent, providing a mechanistic explanation for the late platelet recovery observed at day 14 rather than during the early treatment period.5,8
Evidence supporting the use of thrombopoietin-receptor agonists in SAT remains limited. Most clinical data regarding these agents originate from immune thrombocytopenia, chronic liver disease, or perioperative settings, where the underlying mechanisms of thrombocytopenia differ substantially from those in sepsis. Recent studies in critically ill populations have primarily focused on recombinant human thrombopoietin, often with modest sample sizes and heterogeneous endpoints. Our findings complement emerging evidence by specifically evaluating avatrombopag in a real-world sepsis cohort, while underscoring differences in the timing and clinical implications of platelet recovery compared with non-infectious thrombocytopenia.9,10
Safety is a major concern when considering thrombopoietin-receptor agonists, particularly regarding thrombotic risk. In the present study, avatrombopag use was not associated with an increased incidence of either bleeding or thrombotic events across all analytical approaches. This observation is consistent with recent meta-analyses and pharmacovigilance studies suggesting that, although thrombotic events have been reported with thrombopoietin-receptor agonists, the absolute risk appears modest and highly dependent on patient characteristics and clinical context.6,11 Nonetheless, given the prothrombotic milieu of sepsis, careful patient selection and close monitoring remain essential when these agents are used in critically ill populations.
Despite its association with improved platelet recovery, avatrombopag use was consistently associated with prolonged ICU length of stay. This finding should not be interpreted as a direct adverse effect of the drug itself but rather as a reflection of treatment selection and disease complexity. Patients receiving avatrombopag in our cohort were more severely ill and required greater transfusion support, factors that may have influenced clinicians’ decisions regarding ICU discharge timing. 2 The prolonged ICU stay observed in these patients may also reflect the more intensive supportive care required due to the underlying severity of their sepsis and multi-organ dysfunction. 3 In critically ill patients, prolonged ICU stays are often a result of factors such as inadequate recovery of organ function, the need for extended life support, and ongoing management of severe infections, rather than the direct effect of pharmacologic treatments. 3 Similar dissociations between improvements in intermediate hematologic endpoints and increased ICU resource utilization have been reported for other supportive interventions in critical care, highlighting the importance of interpreting surrogate outcomes within the broader clinical context. 12
In contrast to its hematologic effects, avatrombopag was not associated with a significant reduction in 28-day mortality in the primary analyses. This finding aligns with the concept that thrombocytopenia in sepsis primarily reflects underlying disease severity rather than representing a direct causal pathway to death. Although a mortality signal was observed in one sensitivity analysis, this result should be interpreted cautiously given the potential for residual confounding. Improvements in surrogate hematologic endpoints do not necessarily translate into survival benefits in heterogeneous ICU populations.1,13
Subgroup analyses suggested that patients with higher platelet transfusion requirements and greater illness severity may derive greater hematologic benefit from avatrombopag. These findings should be considered hypothesis generating, as subgroup analyses are inherently susceptible to multiplicity and residual confounding. Nevertheless, they support the notion that pharmacological stimulation of thrombopoiesis may be most relevant in patients with preserved bone marrow responsiveness despite severe consumptive stress. 9
Several limitations of this study should be acknowledged. Its retrospective, single-center design limits causal inference and generalizability. As an observational analysis, our findings should be interpreted as associations rather than evidence of causality, and residual confounding cannot be fully excluded. Variability in the timing and dosing of avatrombopag, as well as potential unmeasured confounders (eg, clinician prescribing preferences, dynamic changes in illness severity, and time-varying co-interventions), may have influenced the observed associations despite robust adjustment strategies. Although propensity score-based methods improved covariate balance, they cannot account for factors that were not measured or incompletely captured in the dataset. In addition, longer-term outcomes beyond 28 days were not assessed. However, strengths of this study include a well-characterized ICU cohort, comprehensive adjustment using propensity score–based methods, and simultaneous evaluation of hematologic, safety, and resource-related outcomes.
In conclusion, avatrombopag use in patients with sepsis-associated thrombocytopenia was associated with improved delayed platelet recovery without an apparent increase in bleeding or thrombotic risk. However, this hematologic benefit did not translate into a survival advantage and was accompanied by increased ICU length of stay. These findings suggest that avatrombopag may serve as a supportive strategy to facilitate platelet recovery in carefully selected patients, while emphasizing the need for prospective studies to define optimal patient selection, timing of initiation, and integration with transfusion strategies in critically ill populations.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261431920 - Supplemental material for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study
Supplemental material, sj-docx-1-cat-10.1177_10760296261431920 for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study by Nengxian Shi, Jin He, Qian Xu, Xin Zhao, Yunying Bao, Dan He, Yucheng Li, Hongbin Hu, Zhenhua Zeng and Ranghui Yu, Qiang Wei in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296261431920 - Supplemental material for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study
Supplemental material, sj-docx-2-cat-10.1177_10760296261431920 for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study by Nengxian Shi, Jin He, Qian Xu, Xin Zhao, Yunying Bao, Dan He, Yucheng Li, Hongbin Hu, Zhenhua Zeng and Ranghui Yu, Qiang Wei in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-3-cat-10.1177_10760296261431920 - Supplemental material for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study
Supplemental material, sj-docx-3-cat-10.1177_10760296261431920 for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study by Nengxian Shi, Jin He, Qian Xu, Xin Zhao, Yunying Bao, Dan He, Yucheng Li, Hongbin Hu, Zhenhua Zeng and Ranghui Yu, Qiang Wei in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-4-cat-10.1177_10760296261431920 - Supplemental material for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study
Supplemental material, sj-docx-4-cat-10.1177_10760296261431920 for Avatrombopag and Platelet Recovery in Sepsis-Associated Thrombocytopenia: A Retrospective Observational Cohort Study by Nengxian Shi, Jin He, Qian Xu, Xin Zhao, Yunying Bao, Dan He, Yucheng Li, Hongbin Hu, Zhenhua Zeng and Ranghui Yu, Qiang Wei in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
