Abstract
Background:
Eltrombopag is an oral thrombopoietin receptor agonist that stimulates the production of normally functioning platelets. The aim of this meta-analysis is to synthesize evidence about the safety and efficacy of eltrombopag for both adult and children with primary immune thrombocytopenia (ITP).
Methods:
A computer literature search of PubMed, Scopus, Web of Science, and Cochrane Central was conducted. Records were screened for eligible studies, and data were extracted and synthesized using Review Manager for Windows. Subgroup analysis and sensitivity analysis were conducted to investigate whether treatment effect varies significantly between adults and children.
Results:
Six randomized controlled trials (N = 611 patients) were included in the final analysis. The overall effect estimates favored eltrombopag group in terms of overall platelet response (relative risk [RR]: 3.42; 95% confidence interval [CI]: 2.51-4.65; P < .0001), incidence of significant bleeding (RR: 0.56; 95% CI: 0.41-0.77; P = .0004), and number of cases needed to rescue treatment (RR: 0.45; 95% CI: 0.32-0.65; P < .0001). The efficacy of eltrombopag did not differ significantly between children and adults except for incidence of any bleeding (RR: 0.83 vs 0.51; P = .008).
Conclusion:
Eltrombopag is a tolerable and effective drug for the management of chronic ITP in children and adults.
Introduction
Primary immune thrombocytopenia (ITP) is defined as isolated thrombocytopenia without any associated significant clinical conditions or present etiology of thrombocytopenia, and its diagnosis depends in the first place on excluding any other possible cause of secondary thrombocytopenia. 1 –3 The ITP is a chronic autoimmune condition in which humoral and cell-mediated immune responses participate in the development of thrombocytopenia with increased platelet destruction and defective platelet production characterized by low platelet counts (<100 × 109/L). The pathogenesis of ITP depends on the presence of autoantibodies mediating the destruction of blood platelets. 4
Treatment is usually indicated in patients with platelet counts <30 × 109/L and those with counts <50 × 109/L who are at risk of bleeding. There are variable treatment modalities that have been involved in the management of ITP. Corticosteroids and intravenous immunoglobulins hinder platelet destruction by autoantibodies, which augment platelet production. However, the platelet production proved to remain suboptimal in patients receiving these medications with many relapses or require a long-term therapy. 4 –6 Also subcutaneous injection of Amgen (AMG 531), a thrombopoiesis-stimulating protein, was proved to have a positive effect on a number of patients having chronic ITP who received either a single injection or multiple ones. 7,8
Eltrombopag (SB-497115) is an orally administered thrombopoietin receptor agonist (TPOra), which binds to the transmembrane domain of the receptor and initiates thrombopoietin signaling. This interaction stimulates proliferation and differentiation of cells in megakaryocytic lineage. Previous studies showed that eltrombopag was able to maintain increased platelet counts and reduce bleeding events and the need for concomitant therapies; eltrombopag administration increased platelet production in normal volunteers and in patients with thrombocytopenia secondary to hepatitis C virus. 4,9
The aim of this meta-analysis is to synthesize evidence from published randomized controlled trials (RCTs) about the safety and efficacy of eltrombopag for both adult and children with ITP.
Methods
We followed the PRISMA statement guidelines during the preparation of this review and meta-analysis. 10
Inclusion and Exclusion Criteria
We included RCTs with the following criteria: (1) studies which examined the efficacy of eltrombopag in chronic ITP; (2) studies in which population was patients (adults or children) with a clinical diagnosis of chronic ITP and a platelet count less than 30 × 109/L; (3) studies providing sufficient data reliable for pooling in the meta-analysis; and (4) studies written in English language. In the case of multiple reports for the same study population, we analyzed the data of the most complete data set. Studies were excluded for the following reasons: (1) studies in which patients were not randomized and (2) thesis and conference papers.
Literature Search Strategy
We searched for all published RCTs in the following electronic databases: PubMed, Scopus, Web of Science, and Cochrane Central. We used Mesh database and the following search queries: (((“Thrombocytopenia”[Mesh]) AND “eltrombopag” [Supplementary Concept]) OR (“Purpura, Thrombocytopenic, Idiopathic”[Mesh]) AND “eltrombopag” [Supplementary Concept]). Three authors screened the title and abstract of retrieved records for eligibility. Full texts of potentially eligible studies were reviewed, for selecting the eligible studies for meta-analysis.
Data Extraction
Three authors extracted the data independently using an online data extraction form. The extracted data included the following: (1) study design; (2) study population; (3) risk of bias domains; and (4) study outcomes: overall platelet response, incidence of significant bleeding (World Health Organization [WHO] grades II-IV), incidence of any bleeding, number of cases needed to rescue treatment, and safety outcomes. Disagreements were resolved by the census.
Quality Assessment
The quality of the retrieved RCTs was assessed according to Cochrane Handbook of Systematic Reviews of Interventions 5.1.0 (updated March 2011). Risk of bias assessment included the following domains: sequence generation (selection bias), allocation sequence concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective outcome reporting (reporting bias), and other potential sources of bias; the authors’ judgment is categorized as “low risk,” “high risk,” or “unclear risk” of bias. We used the quality assessment table provided in (part 2, chapter 8.5) the same book. 11
Measures of Treatment Effect
The primary outcome measurement, in studies assessing efficacy of eltrombopag ITP, was overall platelet response defined as platelet counts of at least 50 × 109/L in the absence of rescue therapy, incidence of significant bleeding (WHO grades II-IV) according to WHO bleeding scale, incidence of any bleeding (WHO grades I-IV), number of cases needed to rescue treatment, and incidence of adverse events.
Dealing With Missing Data
In the case of missing standard deviation of mean change from baseline, it was calculated from standard error or 95% confidence interval (CI) according to Altman. 12
Data Synthesis
Dichotomous data were pooled as relative risk (RR) in a fixed-effect model using Mantel-Haenszel (M-H) method 13 ; fixed-effect model was used under the assumption that included studies are comparable in terms of study design, assessed quality, and measures of treatment effect. We used Review Manager 5.3 for Windows.
Sensitivity Analysis
To make sure that no single study is affecting the results, and to test whether the overall effect size is statistically robust, we performed sensitivity analysis excluding 1 study in each scenario.
Assessment of Heterogeneity
Heterogeneity was assessed by visual inspection of the forest plots and measured by I2 and χ2 tests. The χ2 was used to test the existence of significant heterogeneity while I2 quantifies the variability in effect estimates that is due to heterogeneity, if present. I2 test was interpreted according to the recommendations of Cochrane Handbook of Systematic Reviews and meta-analysis (0%-40%: might not be important; 30%-60%: may represent moderate heterogeneity; 50%-90%: may represent substantial heterogeneity; and 75%-100%: considerable heterogeneity). In the case of a significant heterogeneity (χ2; P < .1), a random effect model was used. Otherwise, a fixed-effect model was used.
Publication Bias
According to Egger and colleagues, 14,15 publication bias assessment is not reliable for less than 10 pooled studies. Therefore, in the present study, we could not assess the existence of publication bias by Egger test for funnel plot asymmetry.
Results
Search Results
We retrieved 821 unique articles. A total of 27 full texts were retrieved and screened for eligibility. In which, 21 articles were excluded and 6 RCTs (N = 611 patients) were included in this study (Figure 1). Reasons for study exclusion are shown in Supplementary File 1.
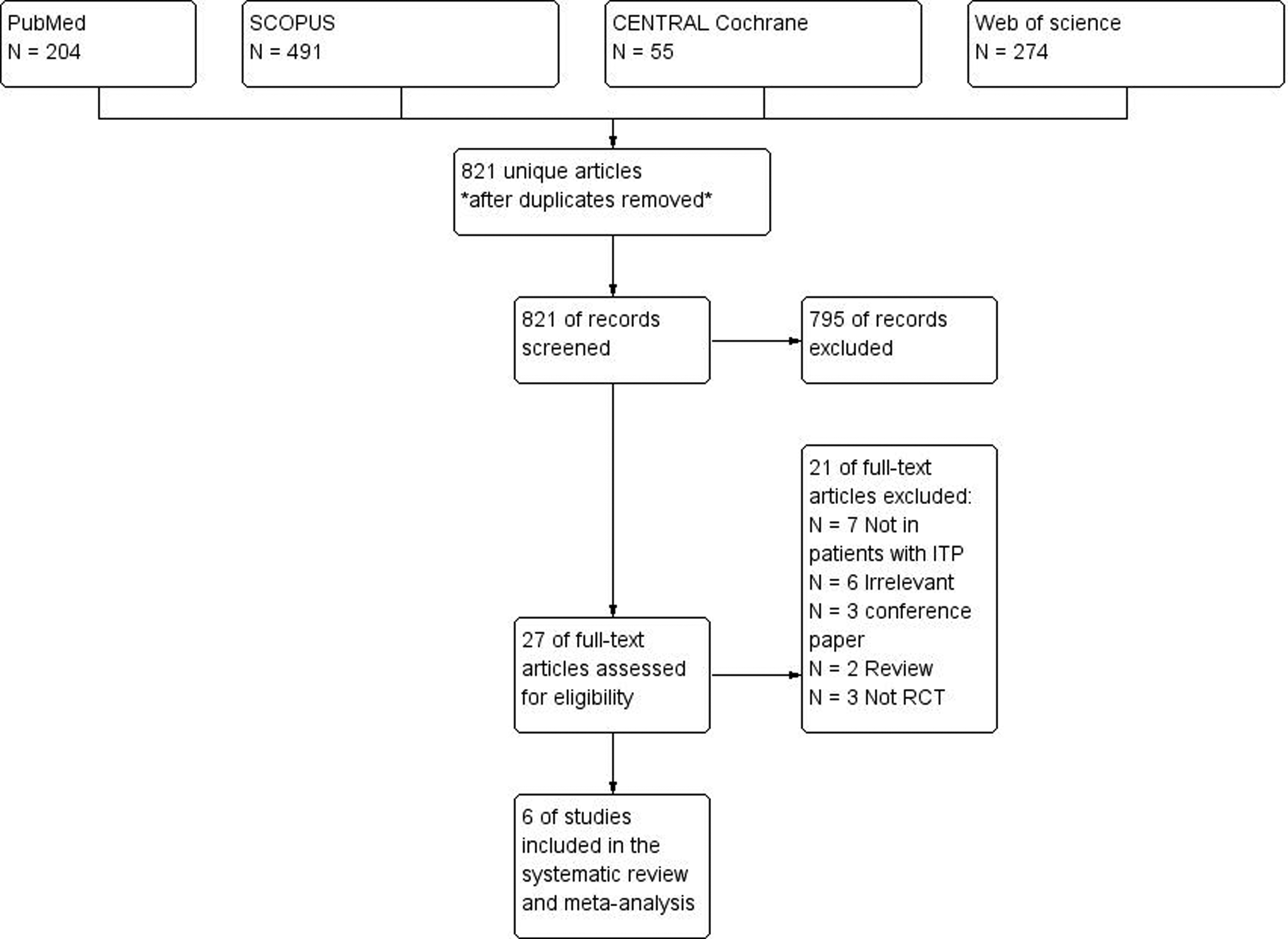
The PRISMA flow diagram of studies’ screening and selection.
Five studies assessed eltrombopag for 6 weeks and only 1 study assessed it for 13 weeks. Eltrombopag was administrated at 30, 50, or 75 mg daily in 1 study, 4 at 50 mg in 2 studies, 16,17 and at the dose-ranging schedule in the remaining 3 studies. 18 –20 Summary of included studies and their main results is shown in Table 1, and the baseline characteristics of their populations are shown in Table 2.
Summary of Included Studies.
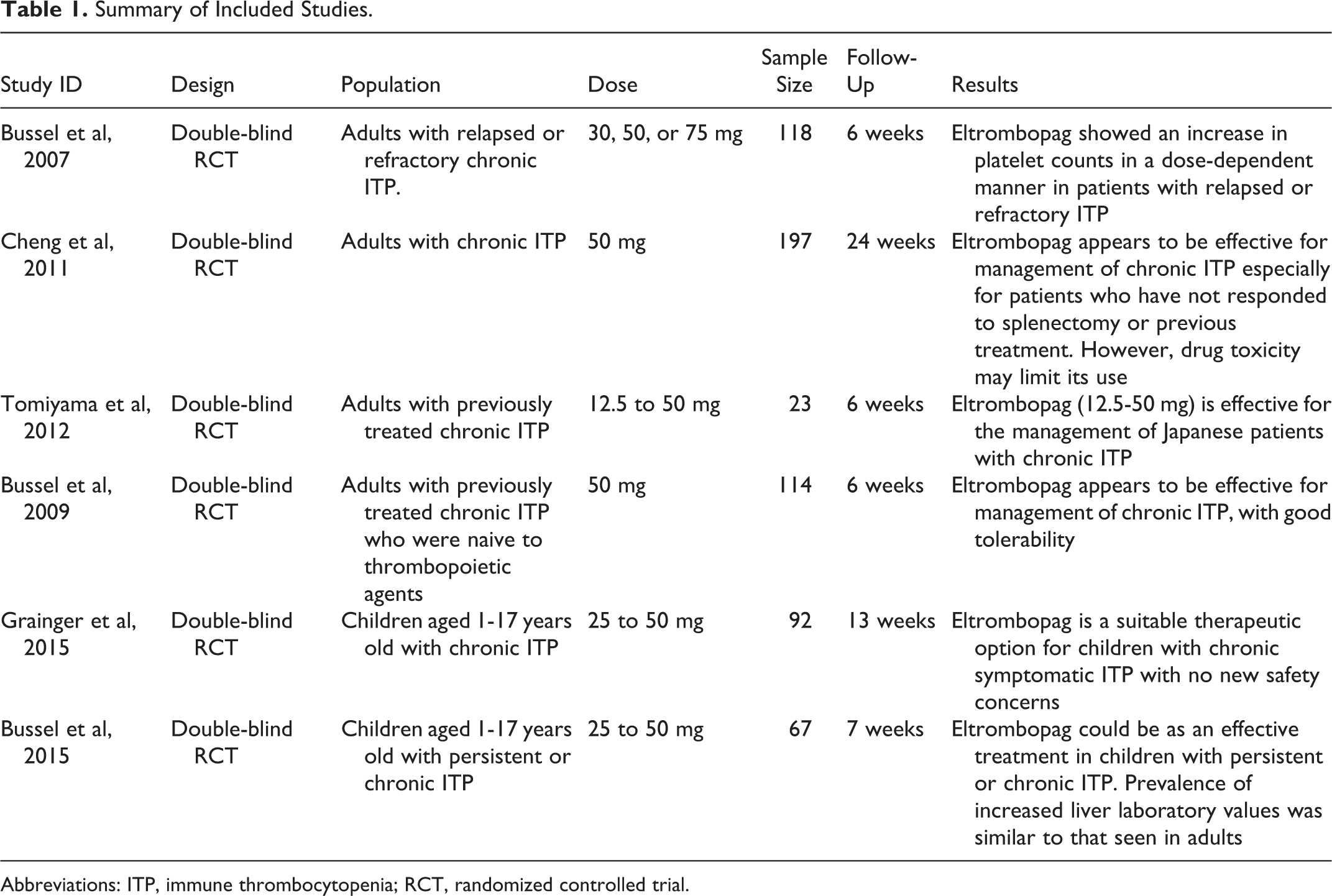
Abbreviations: ITP, immune thrombocytopenia; RCT, randomized controlled trial.
Baseline Characters of Included Studies.
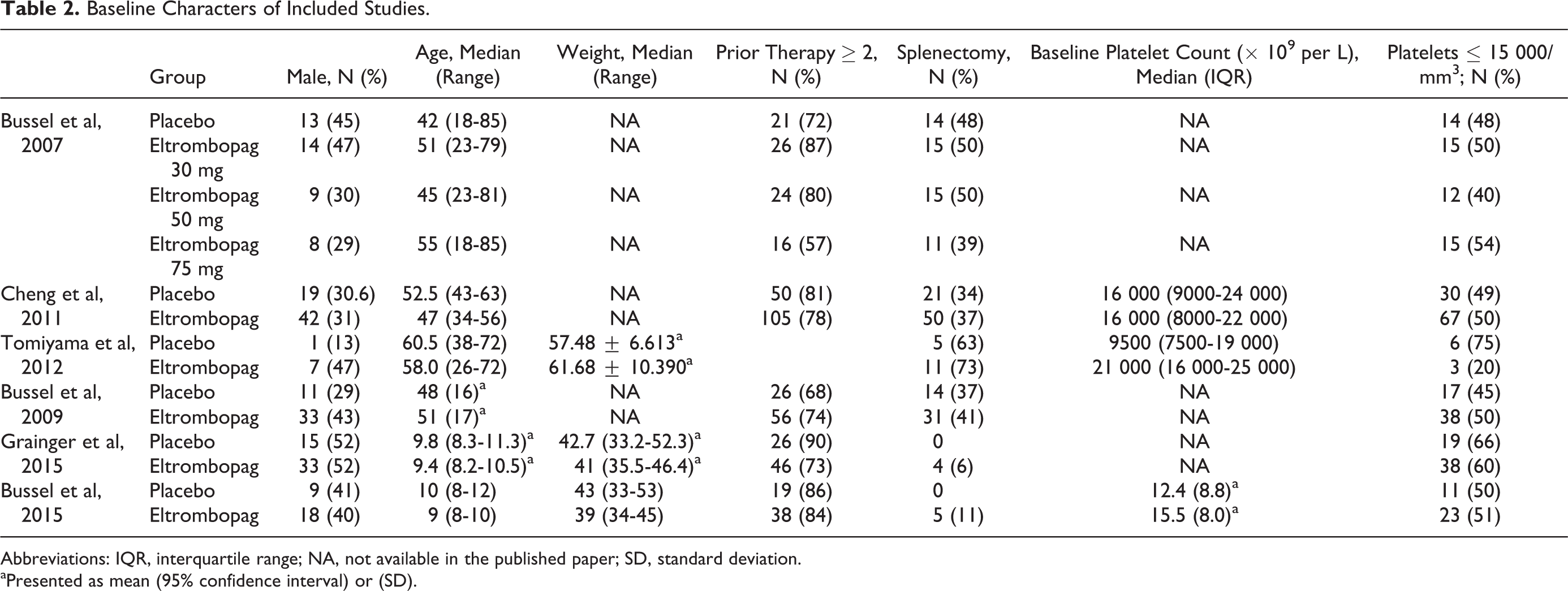
Abbreviations: IQR, interquartile range; NA, not available in the published paper; SD, standard deviation.
aPresented as mean (95% confidence interval) or (SD).
Quality of Included Studies
The quality of the included studies was from moderate to high quality according to the Cochrane risk of bias assessment tool. Summary of quality assessment domains of included studies is shown in Figure 2. Authors’ judgments with justifications are shown in Supplementary File 2.
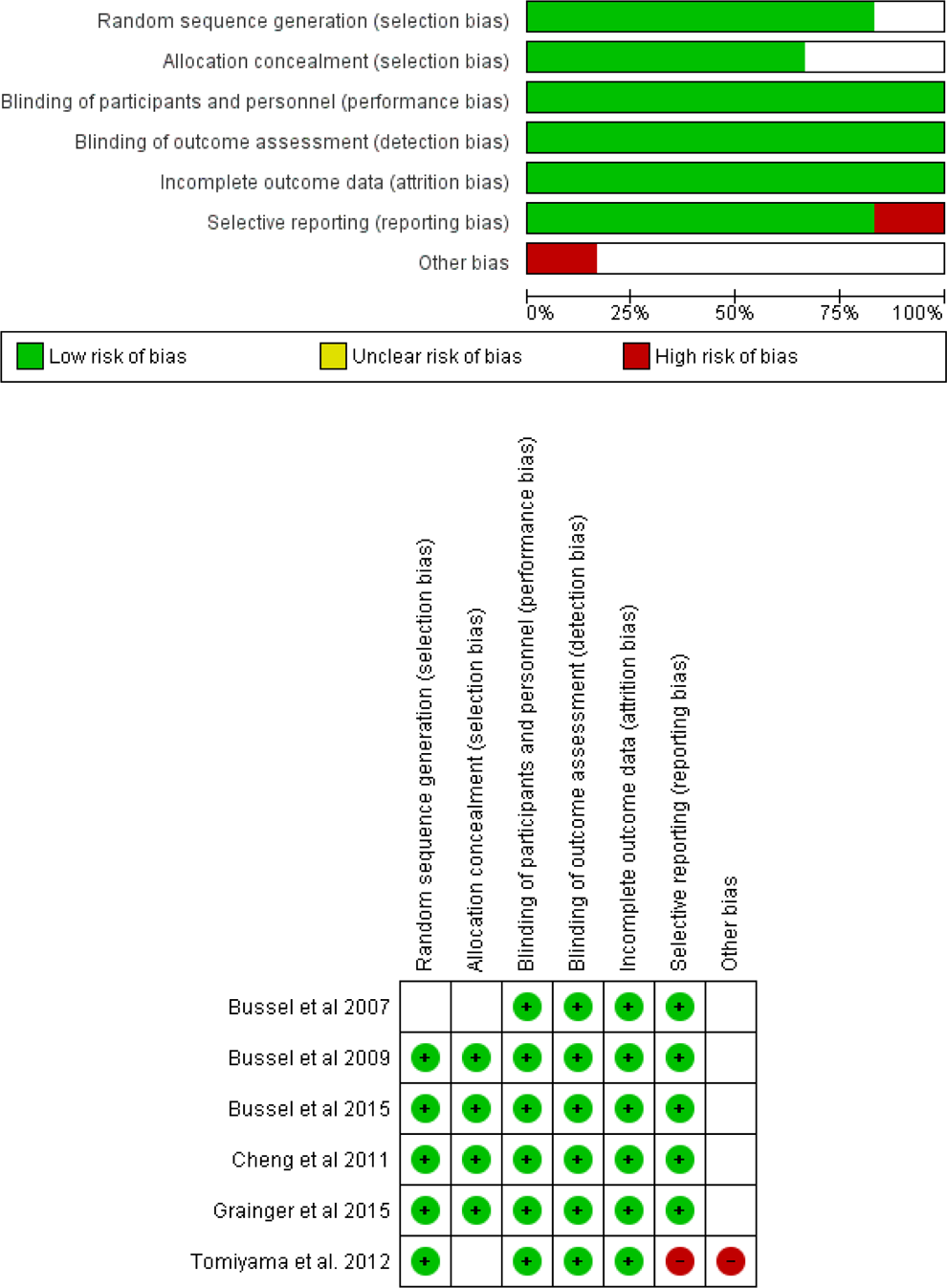
The risk of bias summary and risk of bias graph according to Cochrane Risk of Bias assessment tool.
Efficacy analysis
Overall effect estimate favored eltrombopag group in terms of the overall platelet response (RR = 3.42; 95% CI [2.51-4.65]; P < .0001; Figure 3A), pooled studies were homogenous (I2 = 22%; P = .27); incidence of significant bleeding (WHO grades II-IV; RR = 0.56; 95% CI: 0.41-0.77; P = .0004; Figure 3B), pooled studies were homogenous (I2 = 0%; P = .40); number of cases needed to rescue treatment (RR = 0.45; 95% CI: 0.32-0.65; P < .0001; Figure 3C), pooled studies were homogenous (I2 = 38%; P = .20); and incidence of any bleeding (RR = 0.74; 95% CI: 0.66-0.83; P < .00001; Figure 3D), pooled studies were not homogenous, and heterogeneity was best resolved by excluding Bussel et al study, 18 which recruited children with chronic ITP (I2 = 0%; P = .48).
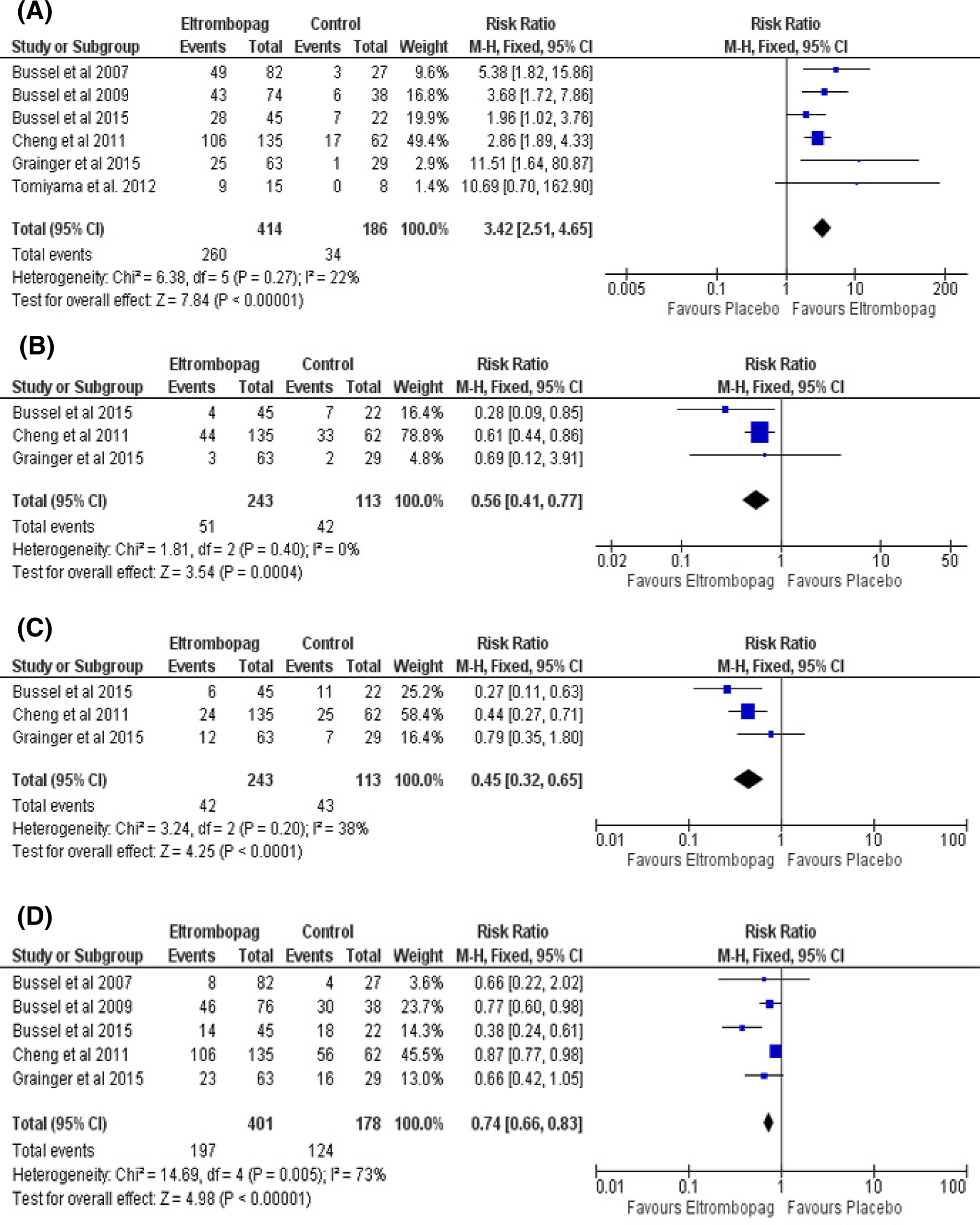
Forest plots of relative risk in efficacy measures. A, Overall platelet response. B, Incidence of significant bleeding. C, Number of cases needed rescue treatment. D, Incidence of any bleeding. CI indicates confidence interval; M-H, Mantel-Haenszel; RR, relative risk.
Sensitivity analysis
For all efficacy outcomes, the superiority of eltrombopag remains significant after excluding 1 study at the time (data not shown).
Subgroup analysis according to age
Among the 6 included studies, 4 studies assessed eltrombopag in adult population while the remaining 2 studies assessed the drug among children with chronic ITP (Table 2). For subgroup analysis, the efficacy of eltrombopag did not differ significantly between children and adults except for incidence of any bleeding (WHO grades I-IV; RR: 0.83 vs 0.51; P = .008; Figure 4).
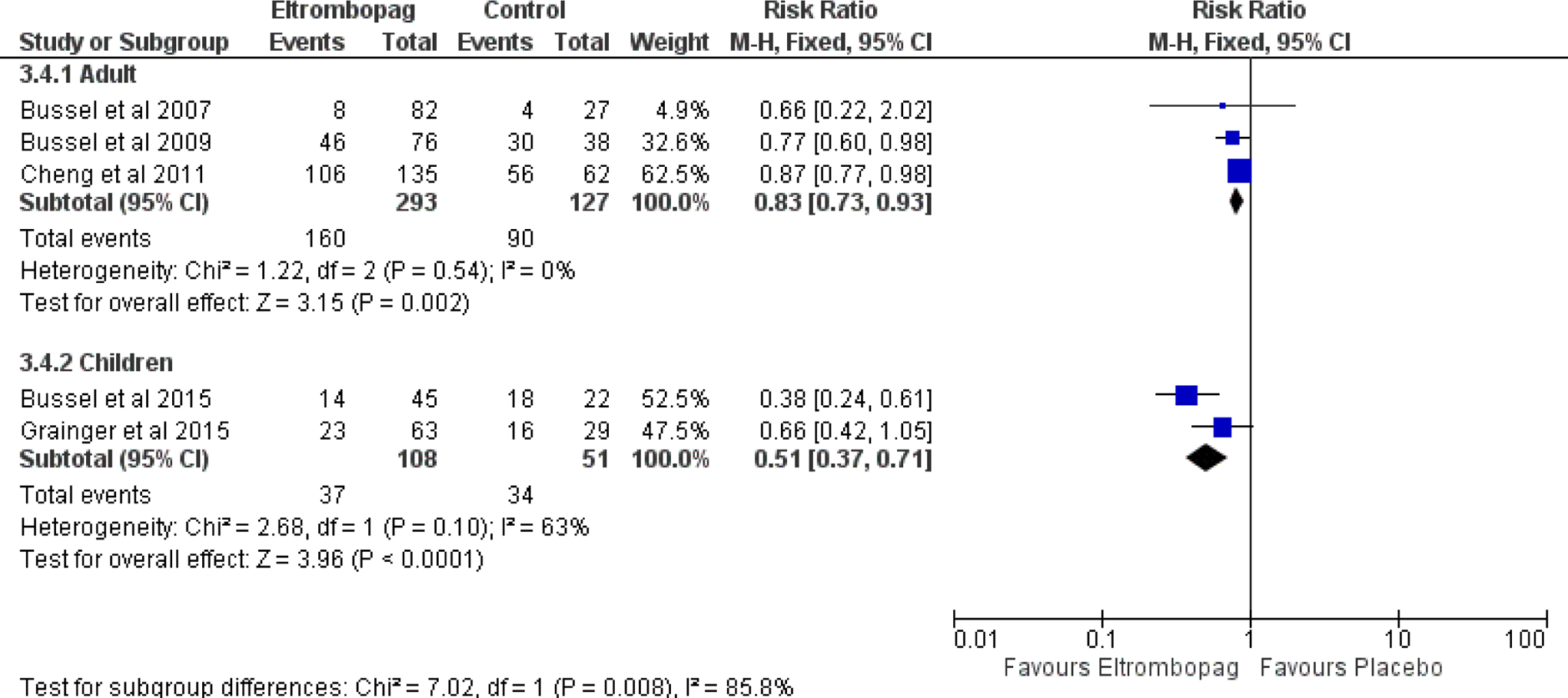
Forest plot of subgroup analysis of the incidence of any bleeding with 95% confidence intervals. CI indicates confidence interval; M-H, Mantel-Haenszel; RR, relative risk.
Thromboembolic events
Among the 6 included studies, only RAISE and Tomiyama et al studies reported the occurrence of thromboembolic events in eltrombopag group, 2% and 7% (1 patient), respectively. 17,20 All remaining studies stated that no thromboembolic events were recorded during the course of the study.
Other adverse events
The total number of adverse events reported in both groups did not differ significantly; the frequency of adverse events was not higher in the eltrombopag group when compared to placebo (RR: 0.95; 95% CI [0.87-1.05]; P = .32).
The pooled RR for adverse events was as follows: severe adverse events (RR = 0.73; 95% CI [0.35-1.51]; P = .39); headache (RR = 0.85; 95% CI [0.62-1.17]; P = .33), diarrhea (RR = 1.33; 95% CI [0.67-2.64]; P = .42); and upper respiratory tract infection (RR = 1.84; 95% CI [0.94-3.63]; P = .08). For all adverse events, the pooled effects were not heterogeneous (χ2; P > .1).
Discussion
Summary of Main Results
The present meta-analysis study provides level 1 evidence that eltrombopag is an effective agent in the treatment of chronic ITP. Eltrombopag significantly improves overall platelet response, reduces the incidence of significant bleeding or any bleeding events, and decreases the number of patients requiring a rescue treatment. Eltrombopag also appears to be well tolerated by patients with ITP. We found no statistically significant difference in the risk of developing any adverse events, severe adverse events, headache, diarrhea, and upper respiratory tract infection.
Eltrombopag was approved by the US Food and Drug Administration and European Medicines Agency for the treatment of adults with chronic ITP; our results run in consistency with all the 6 included RCTs (Table 1) as well published nonrandomized trials, in EXTEND study, 21 a long-term study which enrolled patients who have participated in previous eltrombopag trials and have decided to continue with the treatment. eltrombopag obtained platelet counts ≥50 × 109/L without any ITP medications in 85% of the patients, and it clarified comprehensive data on usage eltrombopag in treatment of chronic ITP for a long time.
Eltrombopag and Risk of Bleeding
The ITP is characterized by antiplatelet antibodies that accelerate the destruction of platelets; an increased risk of serious bleeding has been linked with platelet counts <20 × 109/L. 22,23 Eltrombopag initiates thrombopoietin signaling and stimulates proliferation and differentiation of cells in megakaryocytic lineage, which may result in increased platelet production with subsequent reduction in the risk of bleeding. 24 Our results show that eltrombopag significantly reduces the incidence of significant bleeding or any bleeding events, and these results run in consistency with all included RCTs (Table 1) as well nonrandomized studies which reported a significant reduction in the risk of bleeding in eltrombopag group. 25,21 In contrary, a previous Cochrane review showed that TPOra did not improve significant bleeding events in chronic ITP 26 ; however, this discrepancy can be explained by the fact that their pooled analysis for significant bleeding included only 1 RCT that compared romiplostim with placebo, whereas our pooled analysis included 3 RCTs that compared eltrombopag, the drug of interest, with placebo which gives our analysis more statistical power.
Our subgroup analysis showed that the efficacy of eltrombopag differs significantly between children and adults for incidence of any bleeding (WHO grades I-IV) (Figure 4); however, eltrombopag significantly reduces the incidence of any bleeding among children with ITP as well adult (P = .002 and P < .001, respectively).
Eltrombopag for Adult and Children Patients With ITP
For newly diagnosed ITP patients, corticosteroids for 2 to 4 weeks remain the first-line therapy, and eltrombopag was approved for the treatment of ITP patients who have failed initial treatments with corticosteroids, immunoglobulins, or splenectomy. In contrast, available evidence was not sufficient for its approval in children. Treatment options for children with the persistent disease are immunosuppressive drugs, rituximab, or splenectomy. 27 Associated significant complications caused by long-term use of corticosteroids or immunoglobulin often offset their benefits and may influence physical activities and quality of life of children. 28,29 Our subgroup analysis showed that eltrombopag is an effective agent in children as well in adults; International Working Group guidelines reported that if the safety and efficacy data reported for adults were reproduced in children, eltrombopag could be used in children with persistent highly symptomatic ITP who are resistant to the first-line treatment. 30
Risk of Thromboembolic Events With TPOra
Recent reports reveal growing evidence of an association between autoimmune diseases, including ITP, and occurrence of venous thrombosis and pulmonary embolism 31,32 ; in addition, the risk of thromboembolism is one of the adverse events of interest in patients managed with TPOra. 33 A recent meta-analysis showed a higher risk of thromboembolic events in patients treated with TPOra; however, subgroup analysis showed no statistically significant increase among patients with ITP. 34 Moreover, a pooled analysis of 13 clinical trials assessing romiplostim in patients with ITP showed a higher incidence of thromboembolic events among romiplostim group (7.5 and 5.5 per 100 patient-years in the romiplostim and placebo groups, respectively). 35 In the presented review, current data demonstrated that thromboembolic events of eltrombopag were similar to that of placebo. Only RAISE study, 17 which was the only long-term study, and Tomiyama et al 20 reported the occurrence of thromboembolic events in eltrombopag group (2% and 7%, respectively). Further long-term studies are needed to assess the long-term safety of eltrombopag.
Eltrombopag or Romiplostim
Eltrombopag and romiplostim are the currently marketed TPOra for ITP. We could not identify a published clinical trial that directly compares the safety and efficacy of eltrombopag and romiplostim. Also, published literature shows controversy regarding the superiority of either drug in terms of safety; Nguyen and colleagues 36 suggested an increased risk of thrombosis with eltrombopag compared with romiplostim based on the global individual case safety report database (VigiBase). In contrary, a recent report shows comparable efficacy and safety profile between both drugs. 37
Agreements and Disagreements With Other Reviews
We could not identify a published meta-analysis study about the efficacy of eltrombopag alone; however, results of this study are consistent with the Cochrane review of 2011 which analyzed the efficacy of TPOra for chronic ITP, except for the incidence of significant bleeding. 26
Overall Completeness
Of the total of 588 patients in the 5 RCTs, there were 112 discontinuations (88 of 407 in eltrombopag group [22%] and 24 of 180 in placebo group [13%]). Reasons included adverse events, consent withdrawn, the need for alternative treatment, and administrative decisions. However, we think that this is not likely to affect the study outcomes because authors of all studies analyzed their data in an intention to treat approach; they considered the total number of patients allocated to the treatment groups irrespective of any discontinuation or withdrawal after randomization.
Quality of the Evidence
We performed all steps in strict accordance to Cochrane Handbook of Systematic Reviews of Interventions and followed PRISMA checklist. This evidence is based on RCTs; search methods and eligibility criteria were well defined.
Conclusion
In conclusion, this study provides class 1 evidence that eltrombopag is a safe and effective drug for the management of both adults and children with chronic ITP.
Footnotes
Authors’ Note
Ahmed Saber Elgebaly has full access to all data in the study and takes responsibility for the integrity of presented information and accuracy of the data analysis. Ahmed Saber Elgebaly contributed to the study concept and design, data analysis, revision and appraisal of the manuscript, proofreading the manuscript, and study monitoring and supervision. Ahmed Saber Elgebaly, Ahmed Menshawy, and Gehad El Ashal contributed to Internet searching. Gehad El Ashal and Ahmed Menshawy contributed to selection of studies. Ahmed Saber Elgebaly, Gehad El Ashal, and Mohamed Elfil contributed to data extraction. Ahmed Saber Elgebaly and Gehad El Ashal contributed to quality assessment. Ahmed Saber Elgebaly, Gehad El Ashal, Mohamed Elfil, and Ahmed Menshawy contributed to drafting the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
