Abstract
ALXN4100TPO, a thrombopoietin (TPO) receptor agonist, increases platelets, abrogates radiation-induced thrombocytopenia and affords significant survival benefit to lethally irradiated mice. This preliminary nonclinical safety study assessed effects of a single subcutaneous (sc) administration of ALXN4100TPO in CD2F1 mice randomized into naïve, control antibody (ALXN4200, 100 mg/kg), low (1 mg/kg), medium (10 mg/kg), or high (100 mg/kg) ALXN4100TPO doses. End points included clinical observations, body weight changes, hematology, histopathology, pharmacokinetics, pharmacodynamics by measuring platelet counts, and endogenous TPO (eTPO) levels. Salient findings were prominent increase in platelet counts and end cells of myeloid and lymphoid lineages; elevated megakaryopoiesis in bone marrow; and extramedullary hematopoiesis in spleen and liver. Serum ALXN4100TPO levels were maximum 24 hours after administration, with a half-life of 13 days. Endogenous TPO levels were elevated in 10 and 100 mg/kg ALXN4100TPO-treated groups. In conclusion, ALXN4100TPO (1-100 mg/kg, sc) treatment in CD2F1 mice resulted in profound pharmacological changes in the hematopoietic tissue; however, no life-threatening adverse events were observed.
Keywords
Introduction
Radiation therapy, as one of the most effective treatment modalities for neoplastic disorder, along with the threat of nuclear/radiological terrorism, creates imperatives for the development of agents in prophylaxis, mitigation, and treatment of radiation injury. Acute total body irradiation (TBI) induces extensive apoptotic/mitotic death of actively dividing progenitors, wherein the hematopoietic system is identified as among the most highly susceptible tissues to radiation injury. Profound and persistent hematological syndrome develops as a result of death and ablation of the lymphohematopoietic elements resulting in severe neutropenia and thrombocytopenia, thereby increasing the risk of death due to opportunistic infections and/or hemorrhage. Historically, administration of thrombopoietin (TPO) or megakaryocyte growth and development factor (MGDF) increased the survival of lethally irradiated animals (review 1 ).
Thrombopoietin is an endogenous glycoprotein that is the primary regulator of megakaryopoiesis and platelet production. Disease states wherein TPO administration is indicated include acute myeloid leukemia and immune thrombocytopenia purpura (ITP); ITP frequently presents in patients with chronic liver diseases and AIDS and in patients subjected to chemo- and radiation therapies. In addition to decreasing clinical thrombocytopenia resulting from chemotherapy or radiation-induced injury, TPO administration statistically increased the survival of supralethally irradiated mice2–5 and primates.6–8 Early recombinant forms of the native ligand were tested in preliminary clinical studies of chemotherapy-induced thrombocytopenia, platelet donors, and cyclic immune thrombocytopenia. Although the data were promising, there were associated adverse reactions in the form of antibody generation that cross-reacted with native ligand and subsequently suppressed endogenous TPO (eTPO) formation resulting in rebound thrombocytopenia. Hence, further clinical development of these first-generation molecules was discontinued. 9
Two second-generation TPO agonists currently approved for the treatment of ITP are romiplostim (Nplate; Amgen Thousand Oaks, CA.) and eltrombopag (Promacta; GlaxoSmithKline Middlesex, TW8 9GS, United Kingdom). Questions associated with these agonists that require investigation in clinical studies include rebound thrombocytopenia, thromboembolic complications, increased bone marrow reticulin, and increased myeloblast counts.10,11 Like other TPO mimetics, eltrombopag is species specific, demonstrating activity only in humans and chimpanzees, which poses considerable difficulties in conducting animal studies to obtain the Food and Drug Administration licensure for a radiation prophylaxis/mitigation indication. 1
ALXN4100TPO is a novel TPO receptor agonist that was synthesized in order to reduce the potential of antibody generation to eTPO. This was achieved using peptide sequences with no sequence homology to the native glycoprotein.12,13 The molecule comprises four 14-amino acid (aa) peptides inserted into the light- and heavy-chain regions of a previously described fully human antibody against anthrax (Bacillus anthracis)-protective antigen 83 (PA83), to reduce clearance and increase the half-life of the compound. The binding kinetics of ALXN4100TPO to myeloproliferative leukemia virus receptor (Mpl-r), the receptor for TPO, is equipotent to the native protein, as demonstrated in an in vitro receptor assay (human TPO), and the agonist stimulates megakaryopoiesis in 2 different mouse strains. 13 More significantly, ALXN4100TPO prevents radiation-induced lethality in mice by abrogating the ensuing thrombocytopenia and bone marrow atrophy. Given 24 hours before exposure, ALXN4100TPO demonstrates protection against radiation lethality with a dose reduction factor (DRF) of 1.32, while ALXN4100TPO administration 12 hours after radiation exposure increases survival with a DRF of 1.11. 13 These preliminary data indicate the potential of developing ALXN4100TPO as a promising candidate for radiation countermeasure efficacy.
Prior to advancing to a non-human primate model, it is necessary to provide evidence related to the safety of the drug. This article describes a nonclinical safety study of ALXN4100TPO in CD2F1 mice. The objectives of the preliminary single-dose toxicity study was to observe for antemortem and postmortem evidence of toxicity by evaluating changes in hematologic and serum chemistry parameters and to identify organ-specific histopathologic changes, if any, after dosing and during recovery.
Materials and Methods
Animal Care and Handling
Inbred male and female CD2F1 mice (Harlan Laboratories, Virginia), 8 to 9 weeks old were maintained in the Armed Forces Radiobiology Research Institute (AFRRI) vivarium. These animals were screened for microbiological and serological criteria as well as full histopathological examination by a board certified veterinary pathologist in any cases of unexpected morbidity and mortality. The veterinary staff determined all study animals to be disease and pathogen free during the quarantine period. Prior to initiation of experimental procedures, animals were acclimatized for a 2-week period. Healthy animals were housed 4 per box in conventional sterile polycarbonate boxes with filter covers (Microisolator; Lab Products Inc, Seaford, Delaware) and autoclaved hardwood chip bedding. Mice had access to Harlan Teklad Rodent diet 8604 (Purina Mills, St Louis, Missouri) and acidified water (pH 2.5-3.0) ad libitum. The animal rooms were maintained at 21°C ± 2°C and 50% ± 10% relative humidity with 10 to 15 cycles of fresh air hourly and a 12:12-hour light–dark cycle. All animal procedures were reviewed and approved by the AFRRI Institutional Animal Care and Use Committee using the principles outlined in the National Research Council’s Guide for the Care and Use of Laboratory Animals.
Test Article and Vehicle
Both control antibody and ALXN4100TPO were kindly supplied by Alexion Pharmaceuticals Inc (Connecticut). A previously described antibody to the anthrax PA83 protein designated as ALXN4200 was the carrier/control antibody. The control antibody used throughout these studies was derived by fusing the coding region of fully human Fab63L1D against anthrax toxin PA 63 14 with that of a human G2/G4 constant region. The peptide mimetic ALXN4100TPO was constructed by introducing sequences encoding the peptide mimetic into the light and heavy chains of the control antibody in Chinese hamster ovary cell lines using the GS gene expression system (Lonza Biologics, Portsmouth, New Hampshire). 13 ALXN4100TPO was purified from cell culture supernatants by Protein A chromatography. Endotoxin titers were determined using a QCL-1000 chromogenic Limulus Amebocyte Lysate end point assay kit (catalog no. 50-647U; Cambrex, New Jersey). The purity and molecular weight of ALXN4100TPO was assessed by capillary electrophoresis using Agilent Lab-on-a-chip (catalog no. 5067-1517; Agilent Technologies, Fort Worth, Texas).
Drug Administration
ALXN4200 and ALXN4100TPO were dissolved in phosphate-buffered saline (PBS) to appropriate concentrations just prior to drug administration. Both materials were administered as a single 0.15 mL/mouse subcutaneous (sc) injection in the dorsal area at the nape of the neck in all treatment animals.
Design for Safety Evaluation
The CD2F1 mice of average weight (males 26 ± 2 g and females 22 ± 1 g) were randomly divided into 5 groups of 10 mice/sex and treated as follows:
Naïve group (no injection) Control antibody (100 mg/kg ALXN4200 in PBS) 1 mg/kg ALXN4100TPO 10 mg/kg ALXN4100TPO 100 mg/kg ALXN4100TPO
Doses were calculated as mg/kg of body weight and administered as a single sc injection.
Single-Dose Toxicity Study
All animals were observed constantly for 1 hour subsequent to drug administration, then at 4 hours, 1 day, and daily for 14-day postinjection, at which point the study was terminated. Routine cage-side observations were made for signs of pharmacologic and toxicological effects (eg, tremors, convulsions, salivation, diarrhea, and lethargy; Supplementary Table 1) as well as for morbidity and mortality. In addition, clinical examination for injection site irritation and behavioral changes (ambulation, response to external stimulus) was assessed. Each animal was individually marked (ear punch), and their body weight was documented shortly before drug administration and then on days 2, 4, 6, 8, 10, and 14. Sequential killings were carried out on 1, 3, 7, 10, and 14 days post-drug administration to evaluate the onset and recovery of treatment-related changes as evaluated through hematology, serum chemistry, and examination of selected tissues for macroscopic and microscopic pathology by a board-certified veterinary pathologist.
Analysis of Blood
Blood (0.6-1.0 mL) was obtained by cardiac puncture using a nonheparinized syringe and a 23G needle from mice anesthetized with isoflurane (Hospira Inc, Lake Forest, Illinois). Samples were immediately transferred into precoded EDTA (Sigma-Aldrich, St Louis, Missouri) tubes and mixed gently on a rotary shaker until analysis. The coded tubes were analyzed for total red blood cell (RBC) counts, white blood cell (WBC) counts, absolute neutrophil counts, monocytes, lymphocytes, reticulocytes, hemoglobin (Hb), and platelets using ADVIA 2120 (Siemens Medical Solutions Diagnostics, Dublin, Ireland); and data were generated using MicroSoft software, version 5.9 (Arlington, Virginia). 15
Clinical Chemistry
To determine serum chemistry alterations, 0.5 mL of blood was obtained as described previously and collected in a serum separator tube (BD microtainer, catalog no. 365967). The tubes were incubated at room temperature for 30 minutes to allow for clotting, centrifuged at 7000 rpm for 10 minutes, and aliquoted into sterile tubes, and the serum was stored at −20°C until the time of analysis.
Pharmacokinetics
ALXN4100TPO serum concentrations were determined using a modified colorimetric enzyme-linked immunosorbent assay (ELISA). An ELISA plate was coated with sheep polyclonal anti-human kappa light-chain antibody (product code AU015, The Binding Site Company, Birmingham, UK). Serum samples of 50 µL volume (diluted 1:100) from ALXN4100TPO-treated mice were added to the plate. The plates were incubated at room temperature for 1 hour on a Thermo Scientific Titer Plate shaker (setting 6, Fullerton, California). After incubation and a wash, the captured ALXN4100TPO was detected with sheep antihuman γ chain horseradish peroxidase-conjugated antibody (The Binding Site Company, product code AP004), which recognizes the HC domain within ALXN4100TPO. The plates were developed with 3,3′,5,5′-tetramethylbenzidine substrate (catalog no. T0440, Sigma), and absorbance was read at 450 nm using a microplate reader (Model 680, BioRad, Hercules, California). The concentration of ALXN4100TPO in serum samples was determined by interpolation from an ALXN4100TPO standard curve using SoftmaxPro (part no. 0200-310, Molecular Devices, Sunnyvale, California).
Gross Pathology and Histopathology
A complete necropsy was performed on all animals in each group, and all collected tissues were examined microscopically from necropsies conducted on days 1 and 14 after drug administration. These included the adrenals, aorta, bone marrow (sternum and femur), brain, cecum, colon, duodenum, epididymides, esophagus, eyes, harderian glands, heart, ileum, jejunum, kidneys, lacrimal glands, larynx, liver, lungs, lymph nodes, mammary glands, pancreas, pharynx, pituitary, prostate, rectum, sciatic nerve, skeletal muscle, skin, spleen, stomach, thyroid, thymus, tongue, trachea, and urinary bladder. In males, the testes, seminal vesicles, and prostate were harvested, while in the females, the ovaries, oviduct, uterus, and vagina were harvested. Skin from the dorsal surface where ALXN4100TPO was administered was also examined histologically to monitor injection site changes. In conjunction with each terminal blood sampling on days 3, 7, and 10, all euthanized animals were necropsied and the sternebrae, spleen, and liver were collected for microscopic evaluation. Harvested tissue samples were fixed in 10% buffered formalin, and bone samples were decalcified in 12% to 18% sodium EDTA (pH 7.4-7.5). The specimens were dehydrated using graded ethanol concentrations and embedded in paraffin. Five-micron sections were stained with hematoxylin and eosin (H&E). A board-certified veterinary pathologist conducted blinded microscopic evaluation of these samples. Bone marrow was evaluated in situ within sternebrae and graded for total cellularity, myeloid-to-erythroid ratios, stromal changes, and megakaryocyte numbers and averaged per 10 high-power fields at 400× magnification using a BX41 Olympus microscope (Minneapolis, Minnesota). Images were captured with an Olympus DP70 camera and imported into Adobe Photoshop (version CS4) for analysis.
Detection of eTPO
Serum TPO levels were measured by means of an ELISA (catalog no. MTP00, Quantikine; R&D Systems, Minnesota) according to the manufacturer’s instructions.Sera were collected as described above, and both male and female samples were pooled into tubes (n = 10 each for male and n = 10 each for female) and stored at −80°C until the time of analysis. Briefly, standards, controls, and suitably diluted samples were added into the microplate wells precoated with a monoclonal antibody specific to mouse TPO where the mouse TPO was bound to the immobilized antibody. After washing, an enzyme-linked polyclonal antibody specific to mouse TPO was added to the wells, followed by a substrate that yielded a blue product and turned yellow when the stop solution was added. The intensity of the color was measured at 540 nm with reference to a standard curve prepared using known concentrations of TPO on a Spectra Max M5 plate reader (Molecular Devices). Each data point was measured in triplicate. Data presented are means ± standard error of the mean from 3 different samples.
Statistical Analysis
Means and standard errors of the means were calculated for pharmacokinetics, hematological, and clinical chemistry data. For all graphical presentations, data were pooled among males and females within the group as there were no significant differences in the outcome between the sexes. These data were analyzed using 2-way analysis of variance. For each parameter of interest, follow-up tests were performed for the simple main effect of the group at each time using Tukey method. Differences below the probability level of .05 were considered statistically significant. For pharmacokinetic data, a noncompartmental approach was used to compute area under the curve, maximum serum concentration (Cmax), time to reach maximum serum concentration (Tmax), biological half-life (t1/2), elimination rate constant (Ke), and mean retention time using PK Solutions 2.0 software (Summit Research Services, Montrose, Colorado).
Results
Clinical Signs and Body Weight Changes
All clinical observations were within normal limits. There was no mortality, and no indication of injection site injury, and the drug did not affect gait, ambulation, or general activity. No effects on body weight change were observed (Table 1).
Weight Changes in CD2F1 Mice Over 14-Day Post-ALXN4100TPO Administration.a
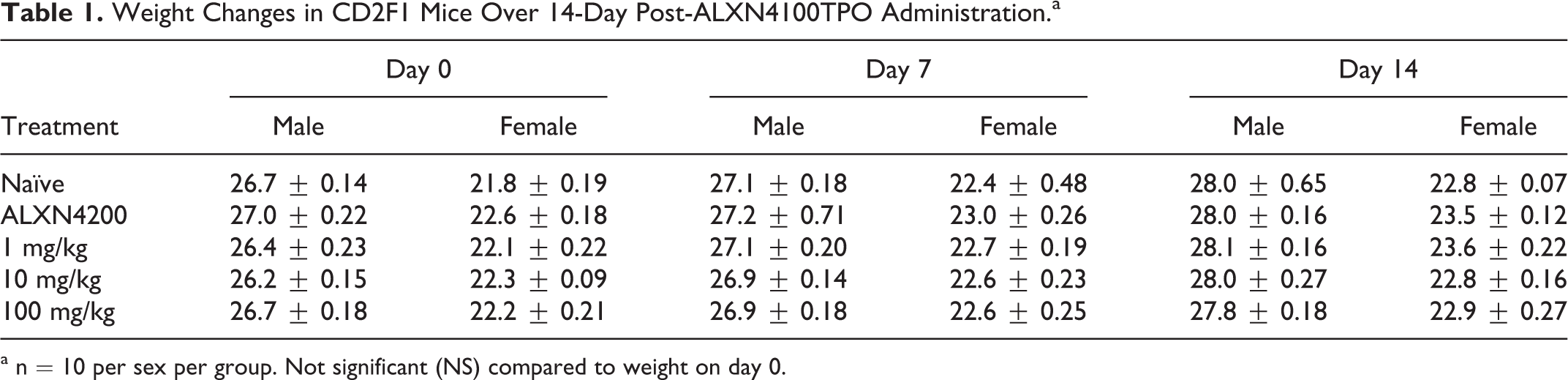
a n = 10 per sex per group. Not significant (NS) compared to weight on day 0.
ALXN4100TPO Dose-Dependent Multilineage Hematopoietic Stimulation
Drug responses for all parameters in both sexes were comparable, and hence compilations of data from both sexes are presented. Furthermore, all parameters for control groups (male and female) per time point were pooled to generate control/baseline or reference values (n = 100).
There were no changes observed on day 1 after drug administration in any of the blood indices, even at the 100 mg/kg dose, the highest dose level tested. However, on day 14 after drug administration, at all doses, ALXN4100TPO induced significant changes in several peripheral blood parameters (Table 2). Reductions were seen in all mean erythrocyte parameters (RBC, Hb, and hematocrit [HCT]). ALXN4100TPO reduced RBC counts to 96%, 95%, and 95% of the mean control/reference levels for 1, 10, and 100 mg/kg of the drug, respectively; the reduction was significant for 100 mg/kg at all time points of the studies (P < .05-.01). Similarly, Hb and HCT levels were lower than control values; although the reduction in Hb was not statistically significant, HCT counts were statistically significantly (P < .05-.001) lower for all doses of ALXN4100TPO tested.
Hematological Parameters in CD2F1 Mouse Blood Injected sc With ALXN4100TPO.a
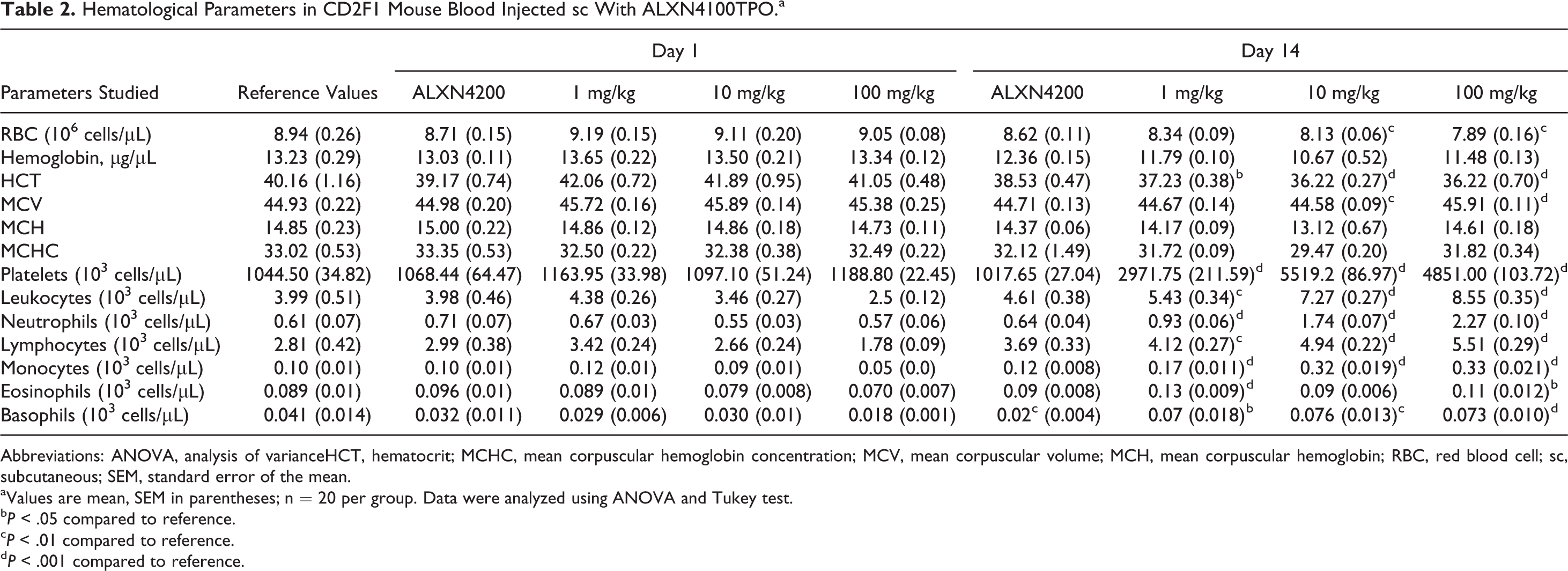
Abbreviations: ANOVA, analysis of varianceHCT, hematocrit; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MCH, mean corpuscular hemoglobin; RBC, red blood cell; sc, subcutaneous; SEM, standard error of the mean.
aValues are mean, SEM in parentheses; n = 20 per group. Data were analyzed using ANOVA and Tukey test.
bP < .05 compared to reference.
cP < .01 compared to reference.
dP < .001 compared to reference.
Conversely, leukocytes (WBC) and platelet counts were elevated on day 14 following ALXN4100TPO treatment, and the increase was dose dependent (Table 2). The WBC counts increased from 4.6 × 10 3 cells/µL in control group to 8.55 × 103 cells/µL in 100 mg/kg group; the increases ranged from 1.5- to 2.5-fold for different drug doses. These increases were in part due to increased neutrophil counts (1.5-, 3-, and 4-fold increase for 1, 10, and 100 mg/kg, respectively) and partly due to increased lymphocyte counts (1.2-, 1.6-, and 2-fold increase for 1, 10, and 100 mg/kg ALXN4100TPO, respectively). Apparent dosage-dependent increases were also observed for monocytes, eosinophils, and basophils (P < .05-.001). The most profound effect was observed in platelet counts. On day 14 after drug administration, platelet counts were highly elevated (P < .001) for all drug doses tested, with fold increases of ∼3-, 5.5-, and 4.8 for 1, 10, and 100 mg/kg of ALXN4100TPO administered, respectively.
Pharmocodynamics
Mean platelet counts following a single sc injection of 1, 10, or 100 mg/kg ALXN4100TPO in male and female mice are compiled and shown in Figure 1A. Single administration of ALXN4100TPO induced a dose-dependent increase in platelet counts on day 1; the counts remained significantly (P < .001) elevated from days 1 to 14 postdrug injection. Peak platelet counts were reached on day 10 for 1 mg/kg dose and day 14 for 10 and 100 mg/kg doses. The mean ratio of peak over baseline platelet counts (Pmax/P0) increased with dose from 4.18 at 1 mg/kg to 5.43 for 10 mg/kg group and 4.77 for the 100 mg/kg group. The platelet counts remained elevated on day 14. However, in a separate experiment, using 1 mg/kg ALXN4100TPO in age-matched CD2F1 mice, the study period was extended, and the platelet counts returned to baseline values on day 21 after drug administration (Figure 1; panel B).
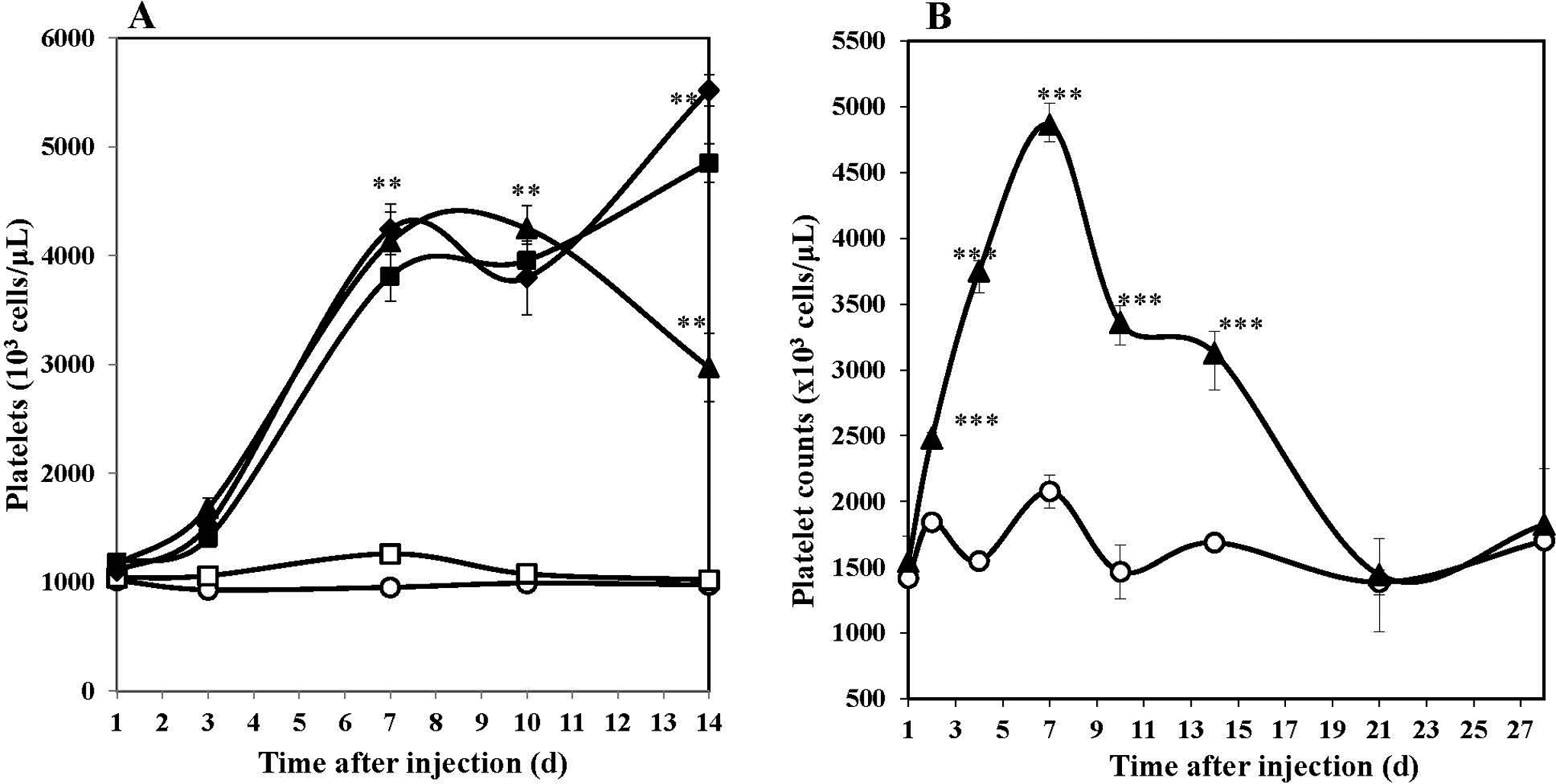
Mean platelet counts in CD2F1 mice receiving a single sc injection of ALXN4100TPO or PBS. A, Platelet counts for 14 days after ALXN4100TPO injection. B, Platelet counts over 28 days following a subcutaneous injection of 1 mg/kg ALXN4100TPO in 0.1 mL PBS. Each data point represents mean ± standard error of the mean (n = 20); data for male and female are compiled. **P < .01, and ***P < .001 compared to naïve control. Mice were treated as follows: naïve mice (open circle), control antibody (open square), 1 mg/kg ALXN4100TPO (closed triangle), 10 mg/kg ALXN4100TPO (closed diamond), and 100 mg/kg ALXN4100TPO (closed square). PBS indicates phosphate-buffered saline.
Due to the stimulation of multiple hematopoietic lineages by ALXN4100TPO, we also recorded the kinetics of changes in the myeloid, lymphoid, and erythroid lineages (Figure 2). The WBC (Figure 2A), neutrophil (Figure 2, panel B), and monocytes (Figure 2C) as well as lymphocytes (Figure 2D) demonstrated significant drug dose-dependent increases. Mean individual cell counts were significantly elevated on days 4 to 14 following drug administration; peak levels were observed on day 10 for all components at all drug doses studied. In contrast, the only parameter reduced by ALXN4100TPO treatment was of the erythroid lineage; higher doses of 10 and 100 mg/kg ALXN4100TPO reduced RBC counts significantly (P < .01-.001; Figure 3A). The nadir in RBC decrease was observed on day 3 after injection with 100 mg/kg ALXN4100TPO, and a second nadir was noted on day 10. In parallel, Hb (Figure 3; panel B) and HCT values were also reduced on days 3 to 14 following administration of 100 mg/kg ALXN4100TPO, but only HCT values were significantly reduced (P < .05-.001, not shown).
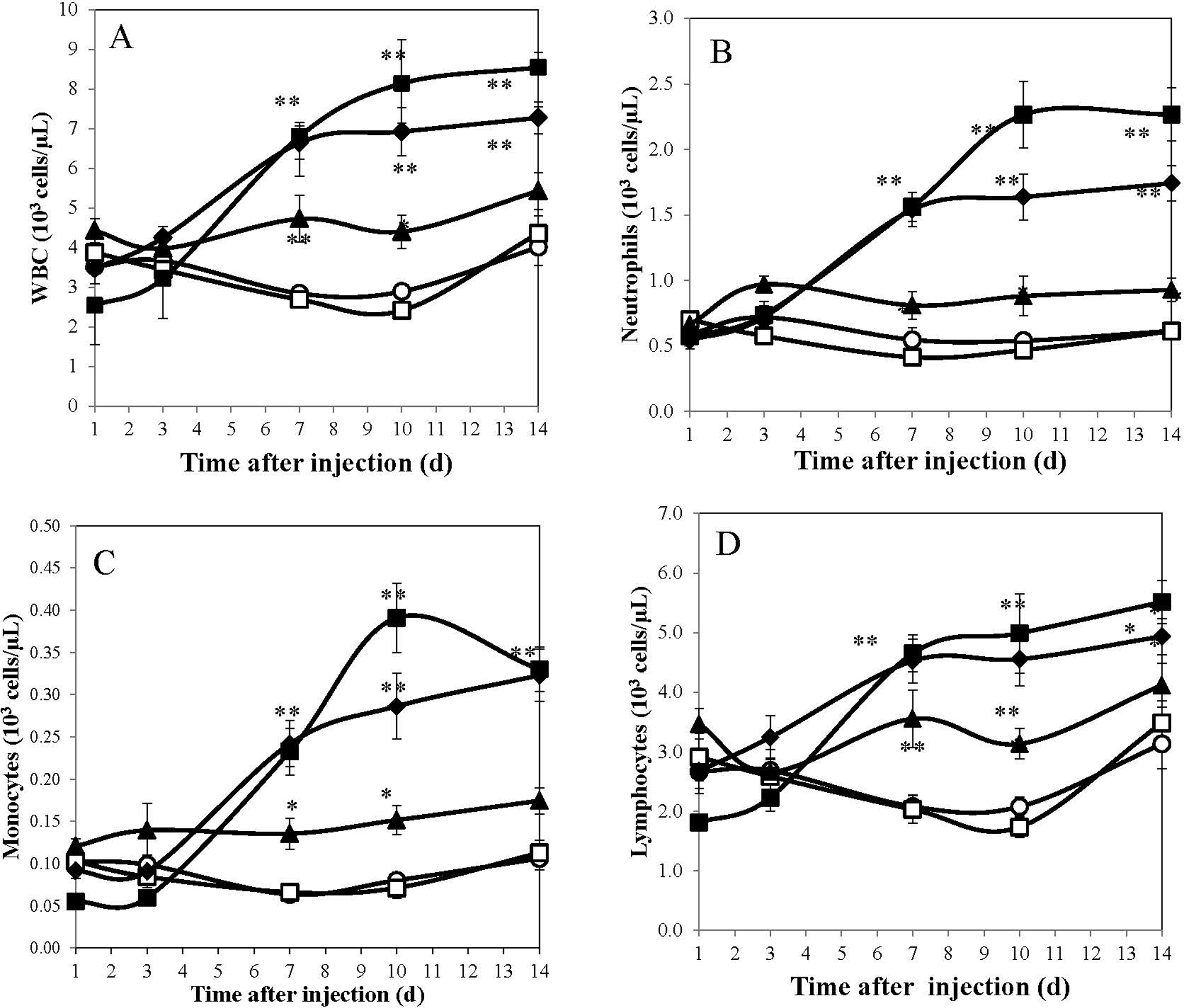
ALXN4100TPO treatment increased peripheral blood indices of several lineages. A, White blood cells (WBC), (B) neutrophils, (C) monocytes, and (D) lymphocytes in mouse peripheral blood days 1 to 14 following ALXN4100TPO treatment. Each data point represents mean ± standard error of the mean (n = 20); data for male and female are compiled. *P < .05, and **P < .01 compared to naïve control. Mice were treated as follows: naïve mice (open circle), control antibody (open square), 1 mg/kg ALXN4100TPO (closed triangle), 10 mg/kg ALXN4100TPO (closed diamond), and 100 mg/kg ALXN4100TPO (closed square).
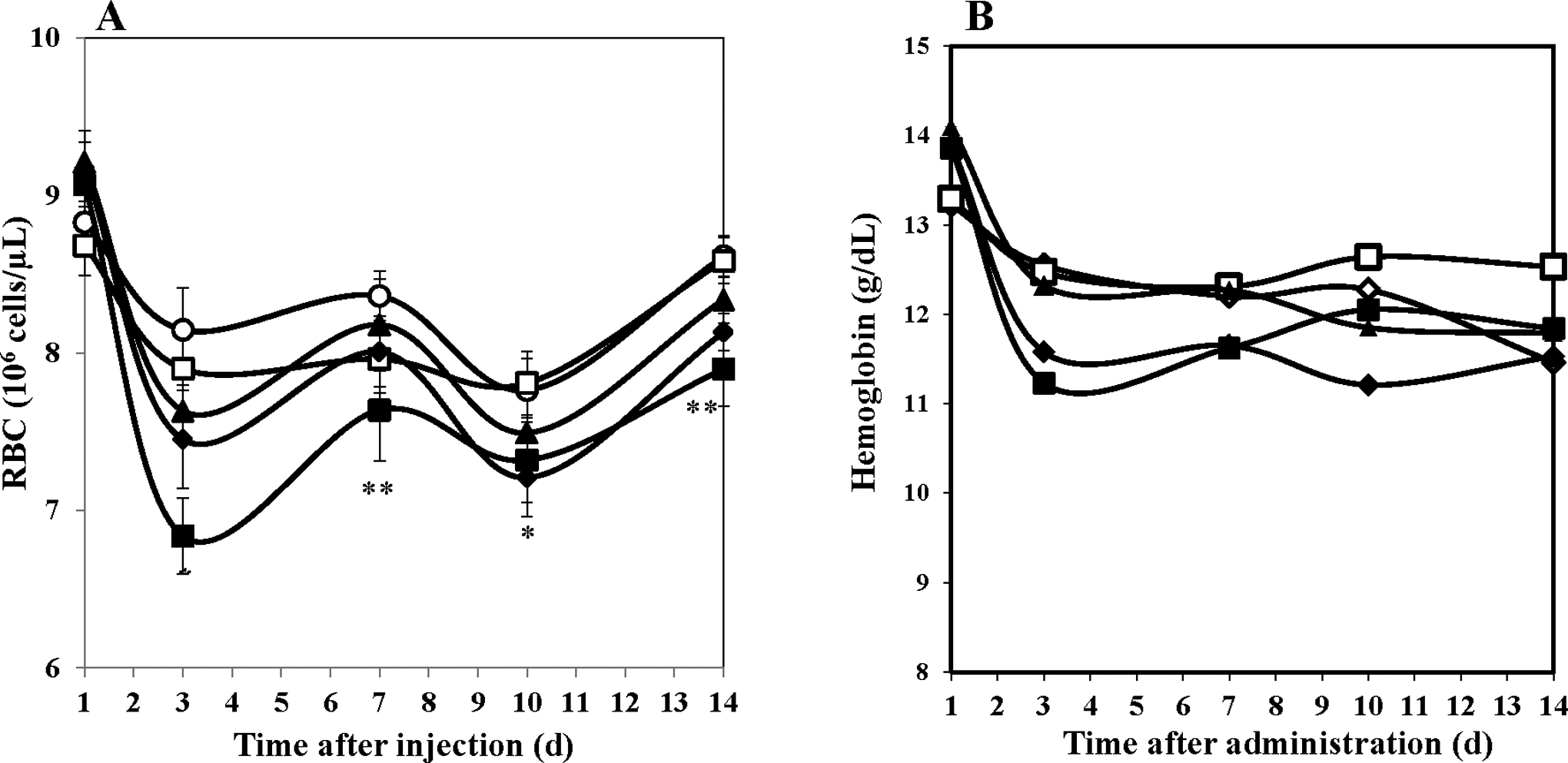
Decrease in erythroid components following ALXN4100TPO treatment. A, Red blood cells (RBC) and (B) hemoglobin in mouse peripheral blood. Each data point represents mean ± SEM (n = 20); data for male and female are compiled. *P < .05, and **P < .01 compared to naïve control Mice were treated as follows: naïve mice (open circle), control antibody (open square), 1 mg/kg ALXN4100TPO (closed triangle), 10 mg/kg ALXN4100TPO (closed diamond), and 100 mg/kg ALXN4100TPO (closed square). SEM for hemoglobin was narrow and masked by symbols. SEM indicates standard error of the mean.
Clinical Chemistry
All parameters outlined in Table 3 were recorded on days 1 and 14 after ALXN4100TPO administration. The highlights at 24 hours include elevated albumin and globulin levels for control antibody and all drug doses, but there was no apparent dose response. Increases in alkaline phosphatase (ALP) and alanine aminotransferase (ALT) were observed for control antibody, and ALT alone was increased at 24 hours after 1 mg/kg. However, at higher doses of 10 and 100 mg/kg, decreases in ALP and ALT were observed. There were no related histomorphological changes associated with these clinical chemistry changes. All other clinical parameters were within normal ranges. On day 14 after drug injection, all parameters were in the normal range except for reduced blood urea and increased CO2 in the 100 mg/kg group.
Compilation of Male and Female Clinical Chemistry Results.a
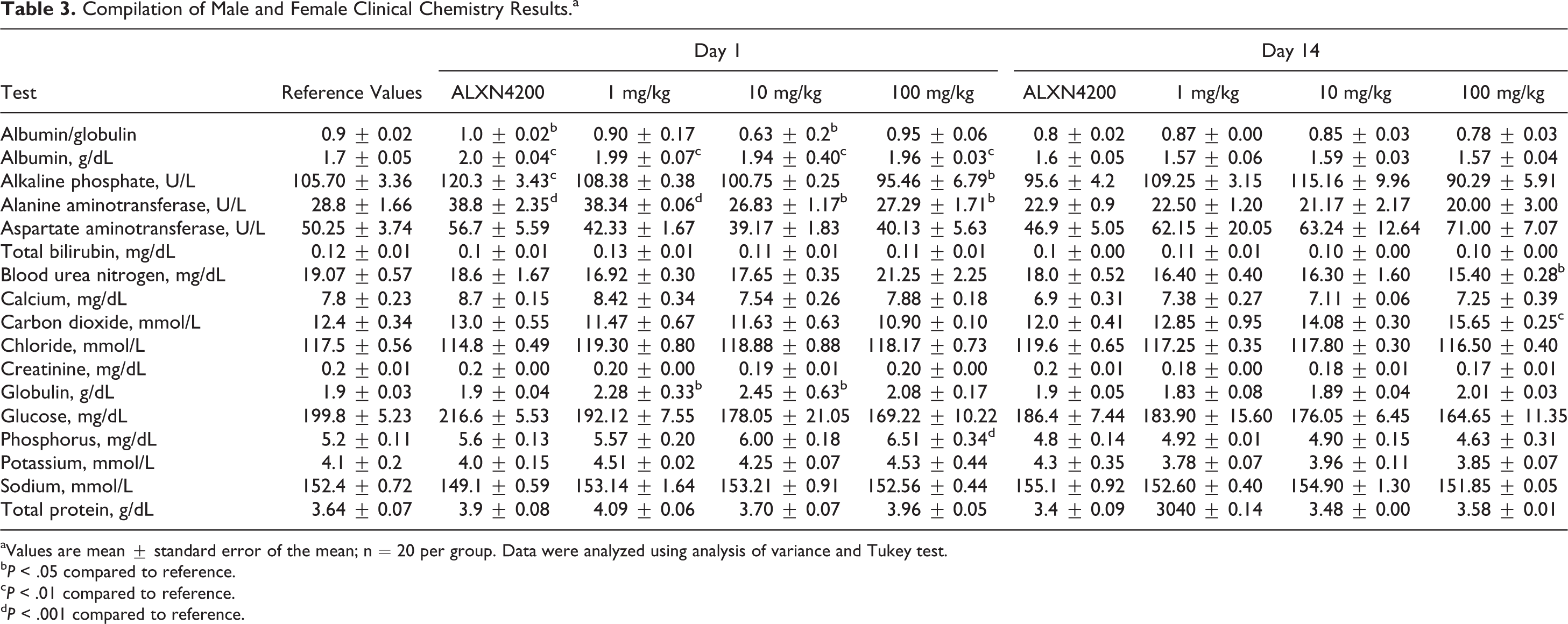
aValues are mean ± standard error of the mean; n = 20 per group. Data were analyzed using analysis of variance and Tukey test.
b P < .05 compared to reference.
c P < .01 compared to reference.
d P < .001 compared to reference.
Pharmacokinetics
Due to limited availability of serum, a separate experiment was conducted with multiple time points (0.5, 1, 2, 4, 8, 10, 12 hours, 1, 2, 3, 4, 7, 14, 21, 28, 35, and 42 days) following injection with 1 mg/kg ALXN4100TPO using 8 mice (male) per time point. Following a single sc administration of ALXN4100TPO, the absorption of ALXN4100TPO from the injection site appeared to be slow with a gradual increase in blood levels. Noncompartmental pharmacokinetic parameters for ALXN4100TPO are summarized in Table 4. The C max levels were reached at 24 hours postdose, and the highest serum concentration was 7972 ng/mL. Mean serum half-life was 13.4 days.
Summary of Noncompartmental Pharmacokinetic Parameters From Serum Concentration–Time Curve of ALXN4100TPO Following a Single Subcutaneous Administration in CD2F1 Mice.a

Abbreviations: AUC (0-∞), area under serum concentration to time curve from time 0 to infinity; CL, systemic clearance; C max, maximum serum concentration; MRT, mean retention time; T max, time required for drug to reach maximal serum concentration; t1/2, half-life in serum.
aData presented are mean (standard error of the mean), n = 8.
Histopathology and Bone Marrow Semi-quantitative Analysis
No significant macroscopic findings were noted during necropsy of all groups on both days 1 and 14 post-injection. No changes or reactions were observed at any of the drug or control antibody injection sites. Background lesions, which were noted in both experimental and control animals with relative frequency, included epicardial mineralization and an associated granulomatous epicarditis, and in many animals hepatocellular binucleation and centronuclear chromatin condensation were noted; however, these were deemed to be strain-specific background findings and were not associated with the treatment compound.
24-Hour Microscopic Evaluation
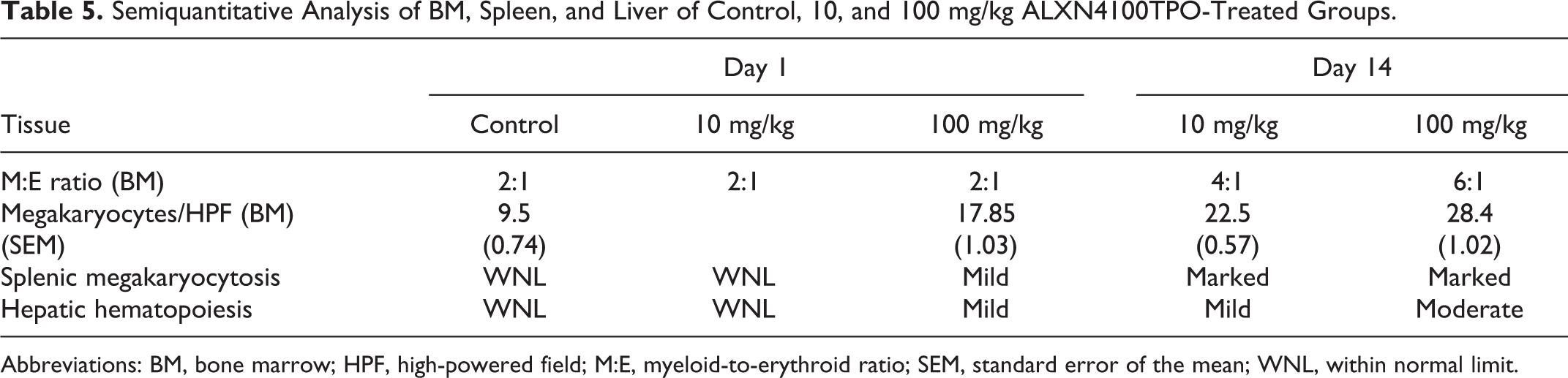
Low and high doses of ALXN4100TPO did not result in any lesions indicative of drug-related systemic toxicity in any organ systems (cardiovascular, respiratory, musculoskeletal, lymphoid, gastrointestinal, hepatic, neurological, endocrine, integumentary, reproductive, special senses, reproductive, and hematopoetic). The higher doses of 10 and 100 mg/kg ALXN4100TPO were associated with modest to significant increases in megakaryocytes in the sternum marrow, but no alterations in myeloid-to-erythroid ratio (M:E ratio; Table 5). At the highest dose of ALXN4100TPO tested, mice of both sexes were presented with mild degree of splenic and medullary megakaryocytic hyperplasia. Two cases of minimal hepatic intranuclear cytoplasmic inclusions, and 3 cases of megalocytosis (males, rare) were interpreted to be background lesions rather than associated with the experimental compound.
Semiquantitative Analysis of BM, Spleen, and Liver of Control, 10, and 100 mg/kg ALXN4100TPO-Treated Groups.
Abbreviations: BM, bone marrow; HPF, high-powered field; M:E, myeloid-to-erythroid ratio; SEM, standard error of the mean; WNL, within normal limit.
14-Day Microscopic Evaluation
Overall, there were no lesions indicative of drug-related systemic toxicity observed in drug-treated groups. In both sexes, the 10 and 100 mg/kg treated groups had increasing degrees of medullary (Figure 4; panels A-C), splenic (Figure 4; panels D-F), and hepatic extramedullary (Figure 4; panels G-I) megakaryocytic hyperplasia with megakaryocytes numbering upward of 30 per high-powered field in the 100 mg/kg group and with a concurrent granulocytic hyperplasia, skewing the myeloid to erythroid ratio toward the myeloid lineages (Table 5). Furthermore, within the hepatic sinusoids, megakaryocytic hyperplasia was also noted as a significant component of extramedullary hematopoeisis (EMH). The EMH is a regular finding in many organ systems in certain strains of mice; however, in the 10 and 100 mg/kg ALXN4100TPO groups, the high numbers of hepatic and splenic megakaryocytes correlates to the significant hyperplasia described in the bone marrow. Additional findings were deemed incidental and include renal lesions such as a single incidence of lymphoma as well as few inflammatory lesions and rare congenital dysplasias.
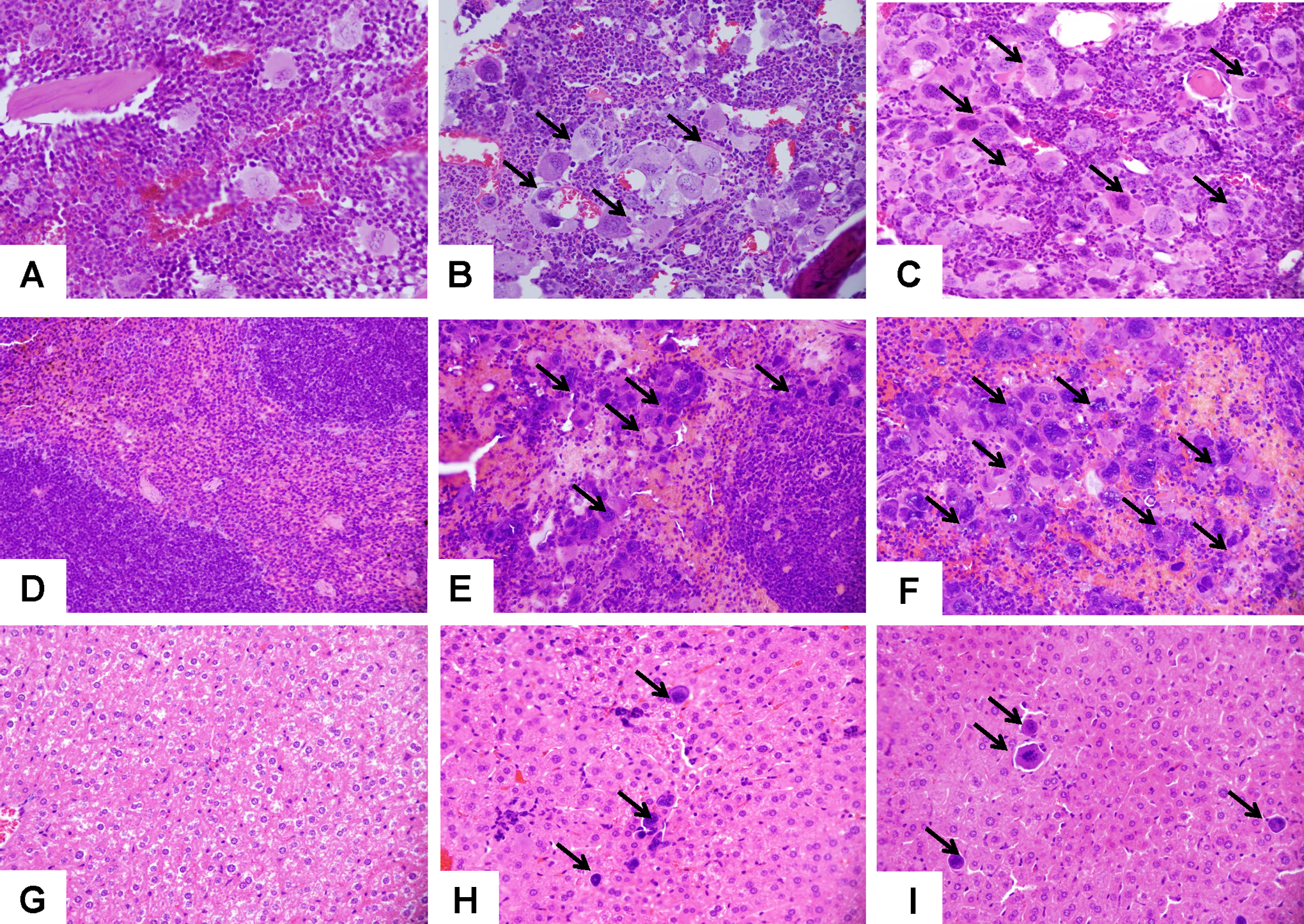
Representative images from histological sections on day 14 after ALXN4100TPO administration. A-C, Sternum marrow, (D-F) spleen (G-I), and liver sections taken on day 14 after ALXN4100TPO injection and shown at ×400 magnification. Treatment with 10 mg/kg (B, E, and H) or 100 mg/kg (C, F, and I) ALXN4100TPO increased total cellularity in bone marrow and megakaryocytosis (solid black arrows) in marrow, spleen, and liver, in comparison with naïve mice (A, D, and G).
Endogenous TPO
ALXN4200 (100 mg/kg) or 1 mg/kg ALXN4100TPO did not alter TPO levels from baseline over the course of the study (days 1-14 postdose). The higher drug doses of 10 and 100 mg/kg ALXN4100TPO increased TPO levels over baseline values, and the increases were dose dependent (Figure 5).
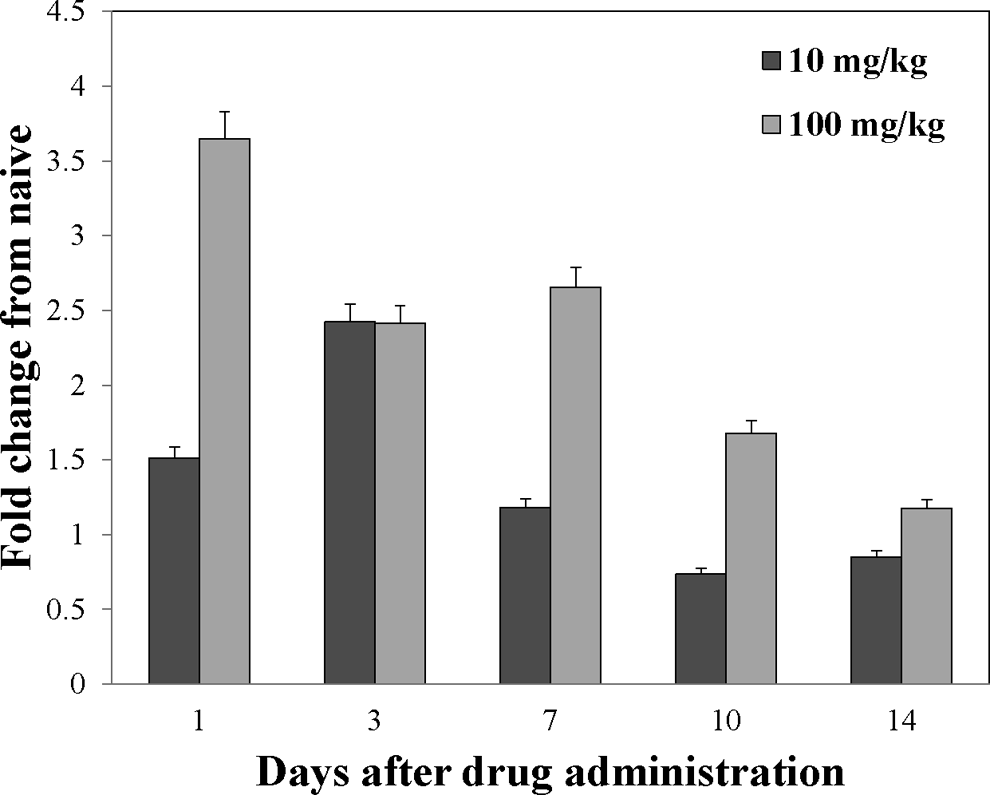
Effect of ALXN4100TPO on endogenous thrombopoietin (eTPO) expression in mouse serum. Fold increase in serum TPO concentration (pooled from 20 samples) for 10 and 100 mg/kg ALXN4100TPO-treated groups. ALXN4200 and 1 mg/kg ALXN4100TPO treatment did not alter endogenous TPO levels (not shown).
Discussion
ALXN4100TPO is a second-generation peptide agonist of TPO with a molecular weight of 59 kDa. It is a bioactive molecule that was proposed as a therapeutic agent for enhancing platelet production in patients having idiopathic thrombocytopenia. The peptide sequence in ALXN4100TPO (Ile-Glu-Gly-Pro-Thr-Leu-Arg-Gln-Trp-Leu-Ala-Ala-Arg-Ala) bears no sequence homology to eTPO and shows minimal risk of cross-reactivity, suggesting that immune-mediated thrombocytopenia is unlikely. Further, this is the first TPO agonist exhibiting potent radiation countermeasure efficacy in a rodent model. 13
The current nonclinical study was designed to evaluate tolerability, pharmacokinetics, and pharmacodynamics of ALXN4100TPO after a single sc injection in normal mice. The starting dose and single administration schedule utilized in the dose range finding study was patterned after preliminary radiation countermeasure studies with ALXN4100TPO, where a single dose of ALXN4100TPO administered prior to or after TBI afforded significant survival benefit. 13 Results from the current study indicate that a single injection of 1, 10, or 100 mg/kg ALXN4100TPO did not result in systemic toxicity, localized inflammatory changes, or adverse effects in either male or female CD2F1 mice.
Prominent hematological changes included increments in platelets, total WBC counts, neutrophils, monocytes, and lymphocytes, while a decrease in the total RBC count was observed for doses 1 to 100 mg/kg. Gross and microscopic histopathology observations noted an increased megakaryopoiesis as well as significant extramedullary hematopoiesis (EH) in both the spleen and the liver following ALXN4100TPO treatment. Multilineage stimulation by TPO and its agonists has been documented previously; Grossman et al 5 reported that TPO accelerates platelet, RBC, and neutrophil recovery in myelosuppressed mice. Eltrombopag stimulates trilineage hematopoiesis in patients with refractory severe aplastic anemia. 16 Native TPO and its second-generation agonist, eltrombopag, increase the platelets, neutrophils, and reticulocytes, while here we report increase in platelets, neutrophils as well as monocytes and lymphocytes.
Platelet counts increased following a 3- to 5-day delay after ALXN4100TPO administration; this may represent the marrow transit time that is required for megakaryocytes to complete cytoplasmic and nuclear maturation and emerge as platelets in circulation. This delay is consistent with observations for other Mpl ligands; in clinical trials, Nplate (AMG531) increased platelet counts on day 5 after drug administration, 17 while increase in platelet counts started 8 days after repeat dosing with eltrombopag. 18 Platelet increase was similar for 1 to 100 mg/kg ALXN4100TPO, with all groups achieving a peak on day 7 over control values, then platelets in the 1 mg/kg group began to decrease on day 14 and returned to baseline values by day 21. The higher dose groups demonstrated a second peak on day 14. However, even with the highest dose of ALXN4100TPO (100 mg/kg) tested, there was no evidence of thrombosis, although platelet levels were elevated 4 to 6 times higher than baseline. This phenomenon was confirmed microscopically by the absence of either emboli or thrombi in all tissues evaluated microscopically.
Notably, reductions in RBCs with the highest ALXN4100TPO dose were observed here. Decreases in erythrocyte numbers have been reported after treatment with other cytokines. Antierythrocyte antibodies, bone marrow suppression secondary to interferon γ induction, and hemodilution secondary to increased plasma volume were proposed as causes of anemia associated with interleukin (IL)-2, 19 IL-12, 20 and IL-621,22 administration. Erythropoietic and megakaryopoietic lineages share a common bipotential progenitor-granulocyte macrophage megakaryocyte colony-forming unit that also expresses cMpl-r, the receptor for TPO; we postulate that ALXN4100TPO increases megakaryopoiesis via a transient decrease in erythropoiesis. Indeed, in preliminary studies, we demonstrated that following ALXN4100TPO treatment, the number of CD71-positive erythropoietic progenitors decreased with a concomitant increase in CD45+ megakaryocyte progenitors. 23
Serum chemistry parameters were unchanged in naïve, control, and ALXN4100TPO-treated groups. ALXN4100TPO did not elevate ALP, AST, or ALT levels, which are markers of liver function. Similarly, the renal markers, serum creatinine, and urea were not altered by ALXN4100TPO.
ALXN4100TPO demonstrated a noncompartmental pharmacokinetics profile following sc dosing. Because native peptides generally have very short serum half-life, modifications are required to enhance half-life and thereby, therapeutic efficacy in vivo. ALXN4100TPO was designed by grafting a 14-aa agonist peptide into a human antibody framework to extend the half-life of the agonist. 12 The half-life of 13.2 days demonstrates the extended half-life of ALXN4100TPO in an in vivo model, conferring an advantage over the other reported TPO agonists. Romiplostim has a half-life of 1 to 3 days following sc administration in primates, 17 while eltrombopag has a half-life of 21 to 32 hours in healthy volunteers. 24
Microscopic evaluation of kidney, brain, stomach, lungs, thymus, and intestine, as well as testes and ovaries, showed normal architecture. In CD2F1 mice of both sexes injected with 10 and 100 mg/kg ALXN4100TPO, profound changes in bone marrow, spleen, and liver were noted. In the bone marrow, ALXN4100TPO induced megakaryopoietic hyperplasia as well as granulopoietic hyperplasia, while an increase in specific types of lymphoid cells was difficult to quantify in H&E stained sections. Histologically, there appeared to be an increase in the M:E ratio, with a preponderance of myeloid precursors. Increased EH by ALXN4100TPO in the spleen and liver provides useful insight into the potent stimulation of not only megakaryopoiesis but also myelopoiesis, which resulted in multilineage hypercellularity in peripheral blood. It has long been recognized that EH, or production of hematopoietic cells outside the adult bone marrow, typically occurs in sites of early blood formation, such as liver and spleen. 25 Similarly, administration of recombinant thrombopoietin (rhTPO) is known to stimulate megakaryopoiesis as well as myeloid and erythroid lineage proliferation. 26 Cessation of TPO administration led to reversal of megakaryocytic hyperplasia by an average of 34 days. 26 Since the present study was terminated on day 14 after ALXN4100TPO administration, extended observation times are indicated to determine whether changes in bone marrow, spleen, and liver at higher ALXN4100TPO doses are transient or permanent.
Clinical trials of the first-generation TPO molecules, including rhTPO and PEGylated recombinant MGDF, were hampered by the generation of neutralizing antibodies that cross-reacted with native TPO, resulting in paradoxical thrombocytopenia. 27 Romiplostim (AMG 531; Nplate), which has no aa sequence homology to eTPO, was immunogenic in mice, rats, and monkeys. 28 However, no eTPO neutralizing antibodies were detected in any of the species tested. Conversely, administration of recombinant human TPO (rHuTPO) to patients with thrombocytopenia led to the production of neutralizing antibody to TPO, which cross-reacted with eTPO, thereby reducing TPO concentration. 27 We evaluated TPO levels in mouse serum following ALXN4100TPO treatment to determine the effect of the agonist on eTPO. Unlike rHuTPO, ALXN4100TPO did not decrease circulating levels of TPO in mice in the 14-day observation period.
Conclusion
The nonclinical data presented here demonstrate that ALXN4100TPO is well tolerated and has a large margin of safety following single-dose administration. The most significant alterations observed were profound megakaryocytosis, EH in spleen and liver, and increase in platelets and other blood elements. These observations were expected pharmacological responses and related to stimulation of the hematopoietic system by ALXN4100TPO, especially at higher doses. The bioavailability of the drug after sc injection was relatively high and half-lives varied from 10 to 14 days. These preliminary findings indicate that further pivotal studies should include longer time points to study reversal of the exaggerated pharmacological effects and to monitor immunogenicity end points at later times following ALXN4100TPO treatment. Additional studies that include repeat dose toxicology in mice and definitive toxicology (single and repeat dose) studies in Rhesus macaque will be undertaken to confirm and broaden the safety profile of ALXN4100TPO.
Footnotes
Acknowledgments
The authors thank Dr Mark H. Whitnall for critical reviews, Drs Cara Olsen and Srinivas Mutalik for statistical support, and Mr Harley Clinton for hematological analysis of blood samples.
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and /or publication of this article: Tao Peng and Denise Devore are employed by Alexion Pharmaceuticals, Inc. However, the company did not direct the research nor fund any part of the study, with the exception of providing drug and support for estimating ALXN4100TPO in mouse serum.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the National Institute of Allergy and Infectious Diseases (NIAID).
