Abstract
Wheat bran extract (WBE) is a food-grade preparation that is highly enriched in arabinoxylan-oligosaccharides. As part of the safety evaluation of WBE, its genotoxic potential was assessed in a bacterial reverse mutagenicity assay (Ames test) and a chromosome aberration assay on Chinese hamster lung fibroblast cells. These in vitro genotoxicity assays showed no evidence of mutagenic or clastogenic activity with WBE. The safety of WBE was furthermore evaluated in a subchronic toxicity study on rats that were fed a semisynthetic diet (AIN 93G) containing 0.3%, 1.5%, or 7.5% WBE for 13 weeks, corresponding to an average intake of 0.2, 0.9, and 4.4 g/kg body weight (bw) per day, with control groups receiving the unsupplemented AIN 93G, AIN 93G with 7.5% inulin, or AIN 93G with 7.5% wheat bran. Based on this rat-feeding study, the no-observed-adverse-effect level (NOAEL) for WBE was determined as 4.4 g/kg (bw)/d, the highest dose tested.
Introduction
Wheat bran extract (WBE) is a food-grade preparation that is highly enriched in arabinoxylan-oligosaccharides (AXOS) and that is produced by enzymatic extraction from wheat bran. The procedure for manufacturing WBE is based on the procedure described in Swennen et al. 1 Briefly, the bran is suspended in water containing an amylase to remove the starch from the bran. Thereafter, the destarched bran comprising the water-unextractable hemicelluloses is separated from the solubles, washed, and resuspended in water in the presence of a hemicellulase enzyme, which releases and solubilises AXOS and β-glucans. Subsequently, the liquid fraction containing the solubilized hemicelluloses is isolated, purified by ion exchange resin treatment, and dried.
As shown in Figure 1, the AXOS in WBE consist of a backbone of β-1,4-linked
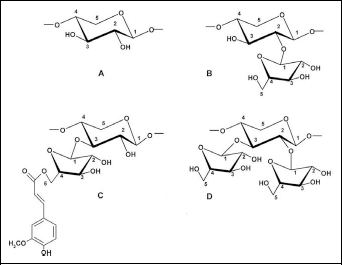
Key structural elements of AXOS. A, Unsubstituted xylose. B, Xylose residue substituted at the O-2 position with arabinose. C, Xylose residue substituted at the O-3 position with arabinose. D, Xylose residue substituted at the O-2 and O-3 position with arabinose. Structure C shows the link of ferulic acid to the O-5 position of an arabinose residue. AXOS indicates arabinoxylan-oligosaccharides.
Arabinoxylan-oligosaccharides were shown in previous studies to have prebiotic properties, 6 –10 as they selectively stimulate the growth of bifidobacteria in the colon (for a review, see Broekaert et al 11 ). Bifidobacteria are generally recognized as beneficial colonic bacteria and are used as probiotic supplements in a wide variety of food products. The prebiotic activity of WBE is associated with promotion of colonic butyrate production and decrease in colonic protein fermentation. 9,10,12 Butyrate is the main energy source for colon cells 13 and as such a crucial parameter in colon function and health. Reduced protein fermentation in the colon is desirable, because bacterial amino acid degradation results in the production of toxic catabolites, some of which have been implicated in bowel diseases and bowel cancer. 14 –16 In addition to its nutritional and prebiotic properties, the physicochemical characteristics of WBE allow a practical and stable incorporation in a wide variety of food products without substantially affecting their taste or texture. In particular, the thermal stability and resistance to acid degradation make WBE suitable for use as an ingredient in processed foods. 17
Although no food ingredients similar to WBE are presently available on the market, consumers have been exposed over centuries to AXOS similar to those provided in WBE. Arabinoxylan-oligosaccharides are mainly ingested through consumption of cereal-based food products such as bread and beer. 10,18 During brewing and bread making, cereal arabinoxylans are exposed to either or both endogenous and exogenous xylanases, 19 thus resulting in the generation of AXOS, reaching levels of up to about 2 g/L in beer 18 and up to about 1.5 g/100 g in bread. 20 This indicates that AXOS have a long history of safe consumption in humans. In addition, trials have previously been conducted in Wistar rats whereby AXOS, purified from wheat bran in a similar way as WBE, were administered at doses up to 4% 9 and 4.8 % 5 of the diet. In none of these trials, adverse effects or effects on body weight or food intake were observed, again pointing to the high safety profile of AXOS-containing extracts from wheat bran.
Trials designed to assess the safety of AXOS or AXOS-containing WBE have not been performed to date. It is important to subject industrially produced WBE to a safety assessment because the average chain length and the nature of branching of the AXOS polymers is specific to the manufacturing method. Food safety authorities in most countries require evidence to support a reasonable certainty of no harm 21 for the particular article of commerce. Accordingly, the purpose of the current study was to assess the genotoxic potential of WBE using mutagenicity and clastogenicity assays and the potential of WBE for general toxicity in a 90-day repeat dose dietary feeding study on rats.
Materials and Methods
Composition of WBE
Wheat bran extract was produced from wheat bran by Fugeia NV (Leuven, Belgium). Wheat bran extract was analyzed for the content of AXOS, average degree of polymerization (DP) of AXOS, arabinose−xylose ratio (A/X ratio) of AXOS, bound ferulic acid, bound glucuronic acid, glucose as part of poly/oligosaccharides, mannose as part of poly/oligosaccharides, galactose as part of poly/oligosaccharides, free monosaccharides, moisture, protein, and ash by the analytical procedures outlined in Femia et al. 5 Fat was measured by the Soxhlet method using petroleum ether extraction according to method ISO 1443. The presence of mesophilic bacteria, yeasts, fungi and Salmonella was determined according to method ISO 4833, ISO 7954, ISO7 954, and ISO 6579/cor1, respectively. The presence of the heavy metals As, Cd, Pb, and Hg was determined according to a Eurofins method based on ISO 17294-1/2.
Mutagenicity Assays
Wheat bran extract was studied for potential mutagenic activity by means of the Ames test in compliance with GLP (Good Laboratory Practice) requirements as specified in the Organization for Economic Co-operation and Development (OECD) GLP 21 and in accordance with a study plan outlined prior to execution of the test, the ICH (International Conference on Harmonization) Guidelines (S2A, 1995; S2B, 1997), EPA (Environmental Protection Agency) Health Effects Test Guidelines (OPPTS 870.5100, EPA 712-C-98-247 [1998], and EPA 712-C-96-247 [1996]), and the OECD Guidelines 471 (1997). The mutagenicity study was conducted at LAB Research Ltd (Veszprém, Szabadságpuszta, Hungary), where the raw data set of the study was archived under GLP conditions.
The tester strains used in this study were the histidine-requiring auxotrophic strains of Salmonella typhimurium (TA98, TA100, TA1535, and TA1537, obtained from Invitrogen Services/BioReliance Corporation, Rockville, Maryland), and a tryptophan-requiring auxotrophic strain of Escherichia coli (WP2uvrA, obtained from MOLTOX, Boone, North Carolina). Metabolic activation was performed using a cofactor-supplemented post-mitochondrial fraction (S9 fraction), prepared from livers of phenobarbital/β-naphthoflavone-induced rats according to Ames et al 22 and Maron and Ames. 23 Wheat bran extract was dissolved at 100 mg/mL in distilled water and was tested in 2 independent experiments at 5, 15.8, 50, 158, 500, 1581, and 5000 µg/plate, the latter being the recommended maximum concentration. The strain-specific positive control chemicals were 4-nitro-1,2-phenylenediamine at 4 µg/plate for S typhimurium strain TA98, sodium azide at 2 µg/plate for strains TA100 and TA1535, 9-aminoacridine at 50 µg/plate for strain TA1537, methyl methanesulfonate at 2 µL/plate for E coli strain WP2uvrA. For conditions of metabolic activation, 2-aminoanthracene was used as a positive control at 2 µg/plate for all S typhimurium strains and at 50 µg/plate for E coli strain WP2uvrA. Distilled water (solvent vehicle for WBE, sodium azide, and methyl methanesulfonate) and dimethyl sulfoxide (DMSO, solvent vehicle for 4-nitro-1,2-phenylenediamine, 9-aminoacridine, and 2-aminoanthracene) were used as the negative controls. Mutagenicity was evaluated in 2 independent experiments. In the first experiment, appropriately diluted WBE, negative control, or positive control chemicals were mixed with S9 mix or 100 mmol/L sodium phosphate buffer (pH 7.4), plus an overnight culture of the test strain in Nutrient Broth No 2 (Oxoid, Basingstoke, England), and 2 mL of molten top agar, followed by plating on minimal glucose agar (Merck, Darmstadt, Germany) and incubating the plates for 48 hours at 37°C. In the second experiment, WBE was pre-incubated for 20 minutes at 37°C in a shaking incubator with an overnight culture of the tester strain and S9 mix or sodium phosphate buffer. Wheat bran extract at different concentrations and the controls (untreated, negative controls, and positive control chemicals) were tested in triplicate.
Clastogenicity Assays
Wheat bran extract was studied for potential clastogenic activity on Chinese hamster lung cells in compliance with GLP requirements as specified in the OECD Principles of Good Laboratory Practices 21 and in accordance with a study plan outlined prior to execution of the test, the ICH Guidelines (S2A, 1995; S2B, 1997), EPA Health Effects Test Guidelines (OPPTS 870.5100, EPA 712-C-98-247 [1998], and EPA 712-C-96-247 [1996]), and the OECD Guidelines 471 (1997). The clastogenicity study was conducted at LAB Research Ltd, where the raw data set of the study was archived under GLP conditions.
Chinese hamster lung fibroblast cells (V79 cell line: European Collection of Cell Cultures) were used as indicator cells for detecting structural chromosome aberrations. Metabolic activation was performed using the above-described S9 fraction.
Cytotoxic/Cytostatic Activity Assay
Wheat bran extract was dissolved at 25 mg/mL in Dulbecco modified Eagle medium (DMEM). A preliminary dose selection of WBE was performed to determine WBE doses that induce >50% growth inhibition (cytotoxic/cytostatic activity). Evaluation of cell numbers was performed after a 3-hour exposure and 20 hours from the beginning of the treatment. This treatment/evaluation schedule will be referred to with 3+/20 in case of presence of CA S9 mix and 3−/20 in case of absence of CA S9 mix. Evaluation of cell numbers was also performed after 28 hours from the beginning of the treatment with 20-hour exposure without S9 and 3-hour exposure with S9 and these treatment/evaluation schedules will be referred to as 20−/28 and 3+/28, respectively. The cell numbers of the treatment groups were noted as percentage of viable cells in relation to the solvent control. The results of these experiments were used to select the concentrations of WBE to be used in the subsequent chromosome aberration assays.
Chromosome Aberration Assay
In 2 independent experiments, both run in duplicate, chromosome aberration assays were conducted in a similar way as the preliminary dose selection assay using the treatment schedules explained above and at WBE concentrations given below. The first experiment was performed using the 3+/20 and 3−/20 treatment/evaluation schedules at 1250, 2500, and 5000 μg/mL WBE. The second experiment was performed using the 20−/28 treatment/evaluation schedule at 56.25, 312.5, 625, 1250, and 2500 μg/mL WBE and using the 3+/28 treatment/evaluation schedule at 625, 1250, 2500, and 5000 μg/mL WBE. Dulbecco modified Eagle medium was used as a negative control. Two hundred metaphase cells containing 22 ± 2 centromeres were evaluated for structural aberrations from each experimental group. Chromatid and chromosome type aberrations (gaps, deletions [breaks], and exchanges) were recorded separately. Gaps were not considered chromosome aberrations, as recommended by Mace et al. 24 Additionally, the number of polyploid and endo-reduplicated cells was scored. The nomenclature and classification of chromosome aberrations were given based on Savage 25,26 and International System for Human Cytogenetic Nomenclature (ISCN). 27
14-Day Pilot Rat-Feeding Study
Prior to performing a 90-day dietary toxicity study with WBE in rats, a 14-day non-GLP pilot study was conducted in rats to establish palatability, tolerance, and suitable dose levels for the full GLP 90-day study. The test involved 3 groups of 5 male and 5 female Wistar rats/group. The controls were fed basal semisynthetic diet AIN 93G, 28 whereas the low- and high-dose groups received the AIN93G semisynthetic diet fully balanced to contain 5% and 10% WBE, respectively. The WBE-supplemented diets were prepared by replacing a corresponding part of the pregelatinized corn starch, the main component of the AIN93G semisynthetic diet, with WBE.
Composition of the Diets for 90-Day Rat-Feeding Study
Six different diets were prepared on the basis of the semisynthetic diet AIN 93G. 28 The composition of the diets is shown in Table 1. Three diets contained WBE at incorporation levels of 0.3%, 1.5%, and 7.5% (w/w). Three control diets were prepared. The first control diet was the basic AIN 93G semisynthetic diet on its own, the second control diet was the AIN 93G diet supplemented with 7.5% inulin (widely used as non-digestible fiber), and the third control diet was the AIN 93G diet supplemented with 7.5% wheat bran (the source of WBE). The WBE, inulin, and wheat bran were added to the appropriate diets at the expense of corn starch, with the aim to maintain an equivalent nutritional balance with exception of starch content.
Composition of the Diets Used in the 90-Day Feeding Trial a
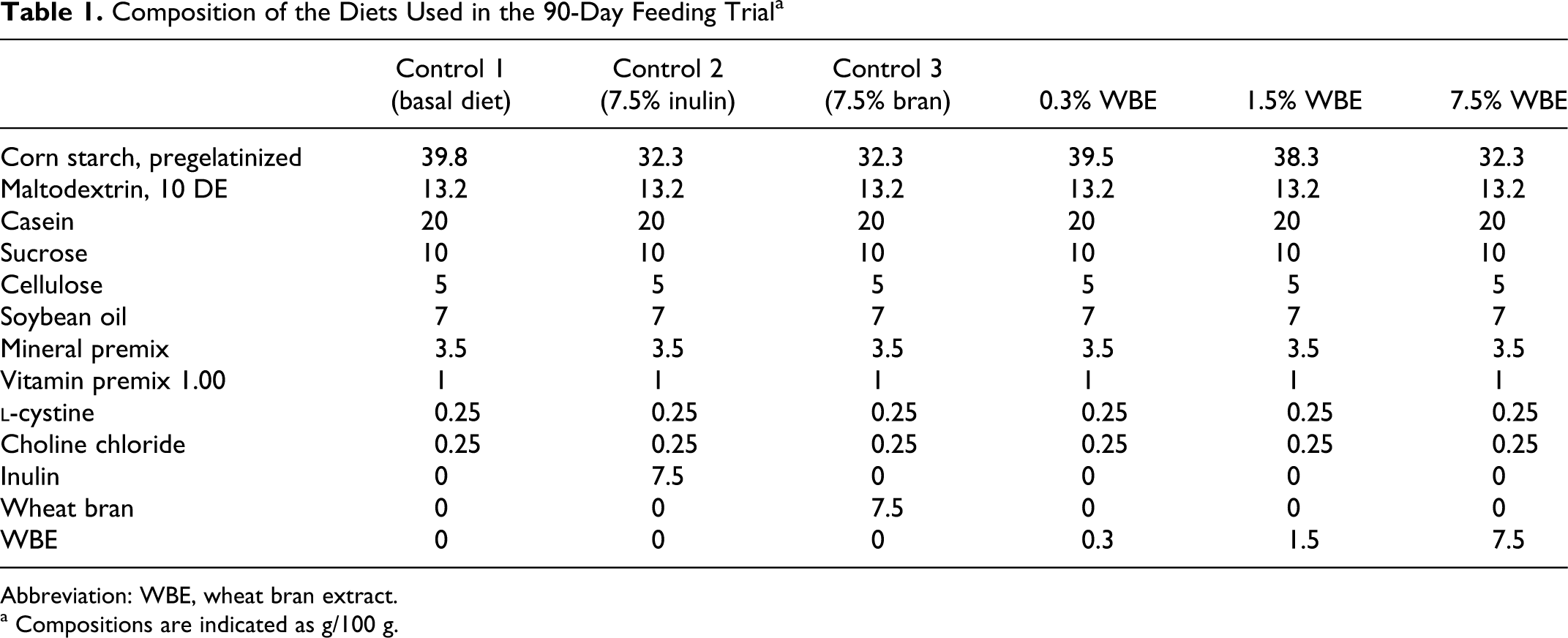
Abbreviation: WBE, wheat bran extract.
a Compositions are indicated as g/100 g.
All diets were prepared and pelletized (10 mm diameter) in parallel by Ssniff Spezialdiäten GmbH (Soest, Germany) using the same batches of raw materials. Diets were stored in sealed bags in a cold room at 5°C and were allowed to equilibrate to room temperature prior to administration. The diets were analyzed for presence of potential microbial and chemicalcontaminants by SGS Germany GmbH (Hamburg, Germany). The results of microbial analyses showed that no contamination occurred during the feed preparations. The results of chemical contaminant analysis showed that aflatoxins, deoxynivalenol, pesticides (165 tested), polychlorinated biphenyls, arsenic, lead, cadmium, and mercury were all below or around analytical detection limits in the diets, with the exception of the AIN 93G diet supplemented with 7.5% wheat bran which contained the pesticide pirimiphos-methyl at 0.17 mg/kg. The stability, homogeneity, and identity of the WBE-containing diets was verified by measuring the AXOS content by the methods described in Femia et al 5 in compliance with GLP requirements as specified in the OECD “Principles of Good Laboratory Practices” 21 and was executed by Intertek ASG (Manchester, UK). The diet samples of the high-, mid- and low-dose WBE diets proved to be homogeneous. The determinations performed at the start, mid, and end of the study show that the product was stable during the study. The variation between the theoretical concentrations and the achieved concentrations was less than 15%, so they were assumed nominally identical.
Animals and Animal Husbandry
A total of 120 Wistar rats (strain Crl:(WI)BR), 60 males and 60 females, were purchased from Toxi Coop Ltd. (Budapest, Hungary) and acclimated for 7 days before the start of the experiment. The animals were specific pathogen free (SPF), were in good health, and had 9 weeks of age at onset of treatment. The weight range of the animals at onset of treatment was 206 to 250 g for the males and 182 to 241 g for females. Animals were housed at 5 per cage in a controlled environment (temperature 20.6-24.8°C; humidity 30%-60%; 15-20 air exchanges per hour; 12-h/d of artificial light) and allowed to eat and drink ad libitum.
The 90-Day Rat Dietary Subchronic Toxicity Assay
The protocol was conducted in accordance with a study plan setup prior to execution of the tests, the OECD Guidelines for Testing of Chemicals (No: 408, 21 September 1998), the US Food and Drug Administration (FDA) Food Additive Safety Assessment (Redbook) Guidelines (1982), and in compliance with GLP requirements as specified in the OECD “Principles of Good Laboratory Practices” (OECD, 1999). The 90-day rat-feeding study was conducted at LAB Research Ltd, and the histologic and clinical pathology work reported herein was undertaken at LAB Research Ltd, where the raw data set of the study was archived under GLP conditions.
In all, 20 animals (10 males and 10 females) were assigned to 1 of the 3 control groups (basal diet control group, 7.5% inulin control group, and 7.5% bran control group) and to 1 of the 3 WBE dose groups (low-dose group at 0.3% WBE, mid-dose group at 1.5% WBE, and high-dose group at 7.5% WBE). Animals were randomized and allocated to treatment groups which were from then onward fed the respective study diets every day for at least 90 consecutive days.
Animals were individually observed for mortality and clinical signs, weighed, and their food and water consumption measured throughout the study in line with the OECD guideline. Tests for general physical condition, neurobehavioral observations, reactivity tests to different type of stimuli (auditory, visual, and proprioceptive), assessment of grip strength and motor activity 29 were conducted on all animals during week 13. Ophthalmological observations were performed with an ophthalmoscope on all animals before treatment and during week 13 on the animals of the basal diet control group 1 and the high-dose group 6.
The day before euthanasia, the animals were starved overnight with free access to water. Before the necropsy, urine samples were collected for urinalysis. Animals were anesthetised with sodium pentobarbital (Euthasol 40%, AST Beheer B.V., Oudewater, The Netherlands) and blood samples were collected by cardiac puncture under deep anesthesia. Blood samples were taken from each animal for standard hematology, blood coagulation tests, and clinical chemistry analysis per the OEDC guidelines. Necropsy with macroscopic postmortem examination was performed on all animals. After exsanguination, the external appearance and the appearance of tissues and organs were observed macroscopically. Organs were weighed (liver, kidneys, testes, epididymides, uterus, thymus, spleen, heart, brain, full cecum, adrenals, and ovaries). The full set of tissues were preserved by fixation in 10% buffered formalin solution in line with regulatory guidelines. Full histopathology was performed in the basal diet control group, the 7.5% bran control group, and the mid- and high-dose WBE groups by light microscopy on hematoxylin-eosin/phloxine stained slides.
Statistics
Statistical analysis of the chromosome aberration data was performed using the Fisher Exact test on the number of cells with chromosome aberrations (with and without gaps) and on the total number of aberrations per test condition. Evaluation of the rat-feeding trial was made by comparing the data for each of the groups against the basal diet control group, for each of the WBE groups against the 7.5% inulin control group, and for each of the WBE groups against the 7.5% bran control group. The heterogeneity of variance between groups was checked by Bartlett homogeneity of variance test. Where no significant heterogeneity was detected, a 1-way analysis of variance (ANOVA) test was carried out. If the ANOVA test indicated a significant difference, Duncan Multiple Range test was used to assess the significance of intergroup differences. Where significant heterogeneity was found, the normal distribution of data was examined by the Kolmogorov-Smirnov test. In case of nonnormal distribution, the nonparametric method of Kruskal-Wallis 1-way ANOVA was applied. If the Kruskal-Wallis test indicated a significant difference, the intergroup comparisons were performed using the Mann-Whitney U test.
Results
Composition of the Test Substance
The composition of WBE is provided in Table 2. Wheat bran extract contains about 80% of AXOS with an average DP of 5. Ferulic acid, which is ester linked to AXOS, constitutes about 1.5% of WBE. Glucuronic acid, which is linked by glucosidic linkages to AXOS, represents about 1% of WBE. The remaining part of the product consists of glucans (mainly β-D-(1,3)(1,4)-linked glucan oligomers), with traces (<1%) of protein, minerals, and monosaccharides. The content of the heavy metals As, Cd, Pb, and Hg is below 0.1 ppm each. Mesophilic bacteria, yeasts, and fungi are below 100/g, and Salmonella is absent in 25 g.
Composition of the WBE Preparation Used for the Toxicological Studies

Abbreviations: AXOS, arabinoxylan-oligosaccharides; A/X ratio, arabinose−xylose ratio.
Mutagenic Activity
Two independent experiments were carried out to assess whether WBE exerts mutagenic activity on any of the 5 different S typhimurium or E coli tester strains. The first experiment was performed by the plate incorporation method without pre-incubation of the tester strains with WBE. This experiment demonstrated that WBE at up to 5000 µg/plate did not cause a significant increase in the mean number of revertant colonies in any of the 5 S typhimurium or E coli strains tested, either with or without metabolic activation (data not shown). The second experiment was performed with a 20-minutes pre-incubation of tester strain, WBE, and phosphate buffer or S9 mix at 37°C. Results of this experiment are presented in Table 3. Significant increases in the number of revertant colonies were observed in the presence of each of the tested positive control chemicals, and these increases were within the historical control range, demonstrating the sensitivity of the tester strains used and the efficiency of the metabolic activation system. The number of revertant colonies of the negative control plates was within the historical range for all strains, and no cytotoxicity was observed. This experiment confirmed that WBE at up to 5000 µg/plate did not cause a significant increase in the mean number of revertant colonies in any of the 5 S typhimurium or E coli tester strains, either with or without metabolic activation.
Revertant Colony Numbers for 5 Different Bacterial Tester Strains Treated With Different Doses of WBE With Preincubation for 20 minutes Without or With S9 Metabolic Activation a
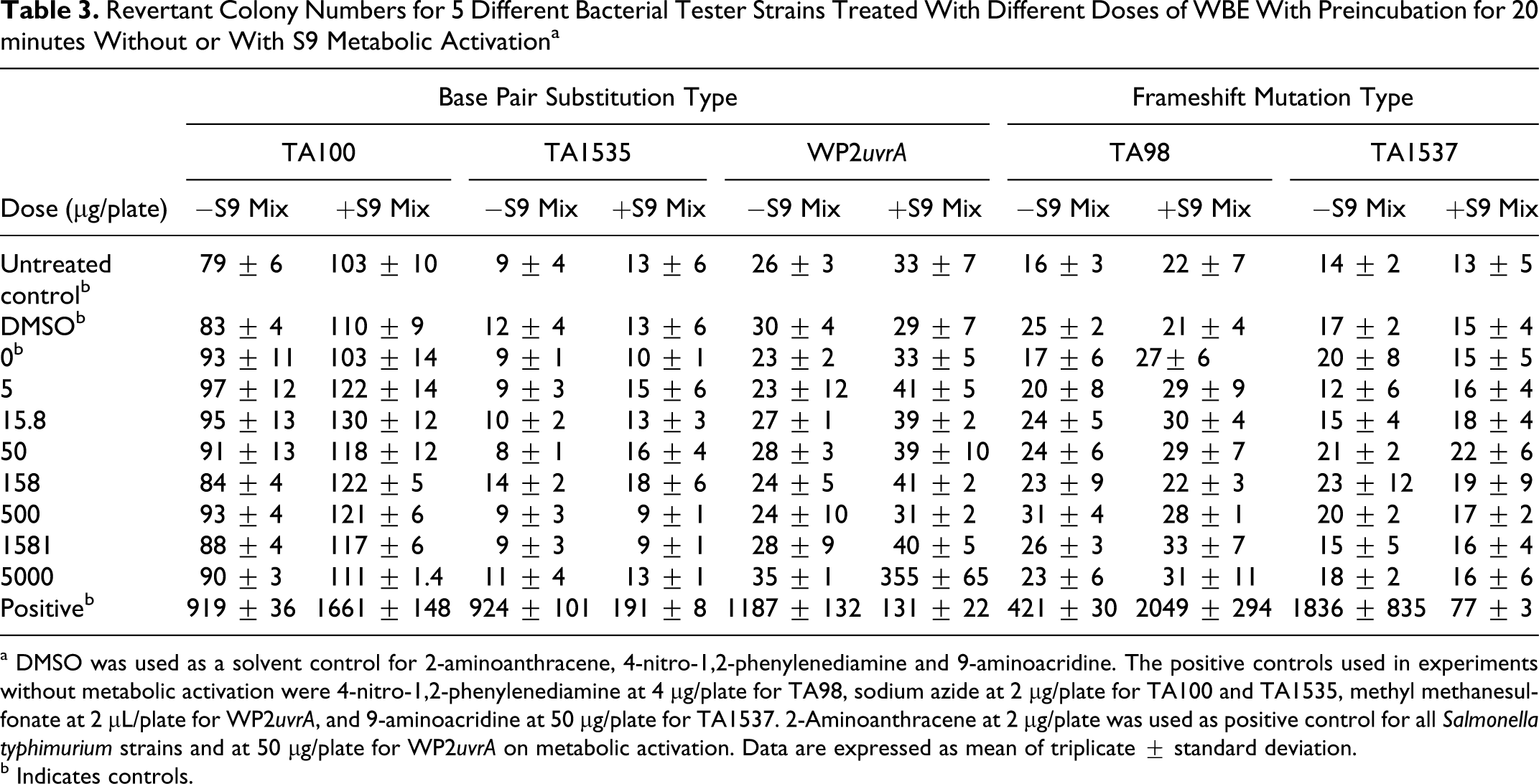
a DMSO was used as a solvent control for 2-aminoanthracene, 4-nitro-1,2-phenylenediamine and 9-aminoacridine. The positive controls used in experiments without metabolic activation were 4-nitro-1,2-phenylenediamine at 4 µg/plate for TA98, sodium azide at 2 µg/plate for TA100 and TA1535, methyl methanesulfonate at 2 µL/plate for WP2uvrA, and 9-aminoacridine at 50 µg/plate for TA1537. 2-Aminoanthracene at 2 µg/plate was used as positive control for all Salmonella typhimurium strains and at 50 µg/plate for WP2uvrA on metabolic activation. Data are expressed as mean of triplicate ± standard deviation.
b Indicates controls.
Chromosome Aberration Activity
In a preliminary assay, it was assessed whether WBE has a cytotoxic/cytostatic effect on Chinese hamster lung fibroblast cells growing in DMEM. After 3-hour treatment and 20-hour evaluation, with (3+/20) or without metabolic activation (3−/20), WBE caused a reduction in the number of cells by <20% at 5000 µg/mL. After 20-hour treatment without metabolic activation and 28-hour evaluation (20−/28), WBE reduced the number of cells gradually in a dose-dependent way by 54% at 2500 µg/mL and 58% at 5000 µg/mL. After 3-hour treatment with metabolic activation and 28-hour evaluation (3+/28), WBE reduced the number of cells gradually in a dose-dependent way by 23% at 2500 µg/mL and 32% at 5000 µg/mL. Based on these results, it was decided to use 2500 µg/mL and 5000 µg/mL as the highest concentration for experiments with a 20-hour treatment and with a 3-hour treatment, respectively.
Subsequently, 2 independent experiments were performed to determine the effect of WBE on chromosome aberrations. The results of the experiments, using 3−/20 or 3+/20 and 20−/28 or 3+/28 treatment/evaluation schedules, are shown in Table 4. The first independent experiment, using the 20−/28 and 3+/28 treatment/evaluation schedules, demonstrated that there were no biologically significant increases in the number of cells showing structural chromosome aberrations, either with or without metabolic activation, up to and including the concentrations that clearly showed growth inhibition of the cells. Additionally, there were no statistically significant differences between treatment and control groups and no dose−response relationships in the number of chromosome aberrations. The validity of the test was shown using ethylmethane sulphonate (0.4 and 1.0 L/mL) and N-nitrosodimethylamine (1.0 L/mL) as positive controls, which caused chromosome aberrations within the historical range. In addition, in the second independent experiment, which used the 3+/20 and 3−/20 treatment/evaluation schedules, no statistically significant differences between treatment and control groups and no dose−response relationships in the number of chromosome aberrations were observed (Table 4). Moreover, there were no increases in the rate of polyploid or endo-reduplicated metaphases in either experiment, neither with nor without metabolic activation.
Chromosome Aberration Analysis of Chinese Hamster Lung Fibroblast Cells Treated With Different Doses of WBE
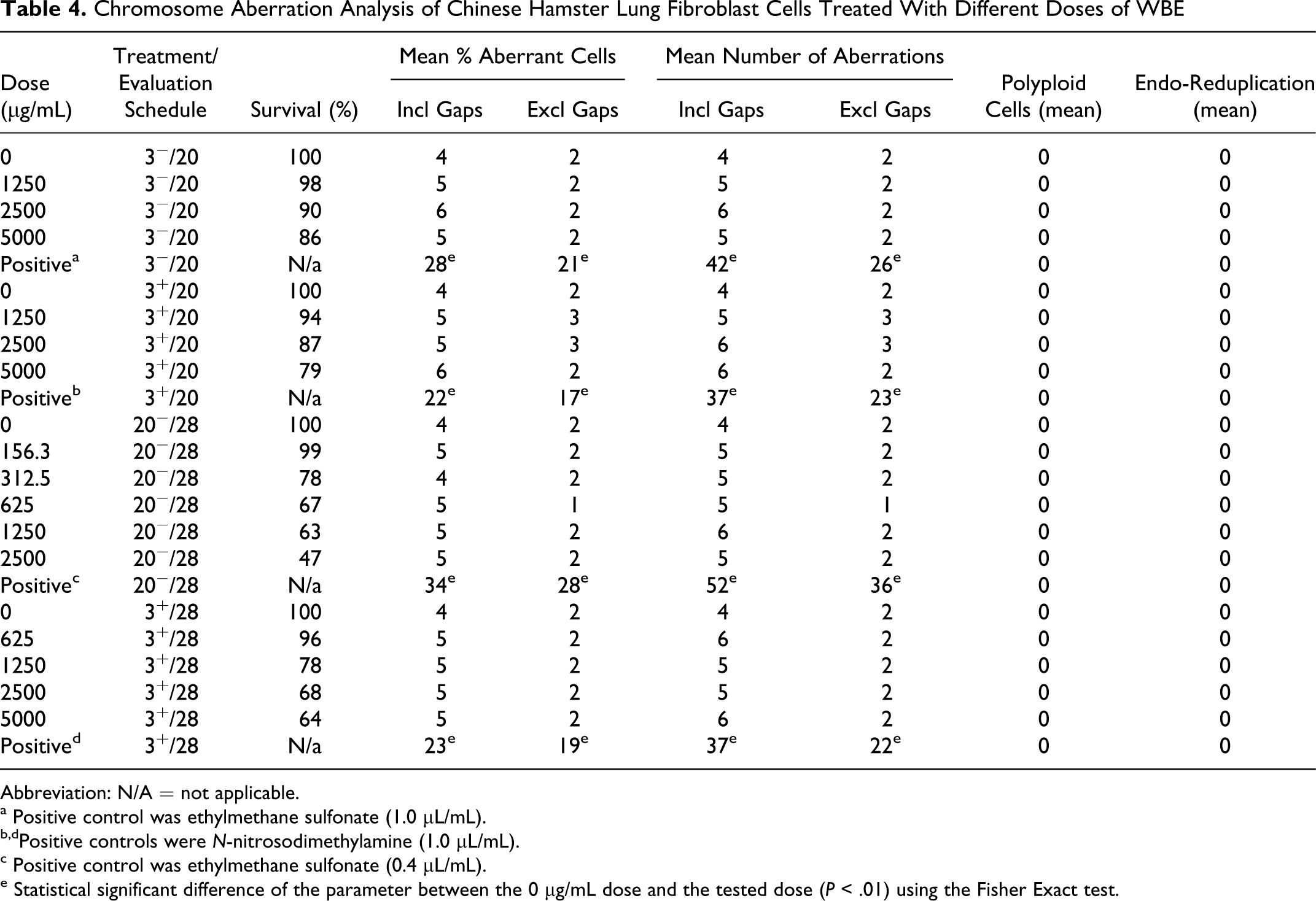
Abbreviation: N/A = not applicable.
a Positive control was ethylmethane sulfonate (1.0 µL/mL).
b,dPositive controls were N-nitrosodimethylamine (1.0 µL/mL).
c Positive control was ethylmethane sulfonate (0.4 µL/mL).
e Statistical significant difference of the parameter between the 0 µg/mL dose and the tested dose (P < .01) using the Fisher Exact test.
14-Day Pilot Rat-Feeding Study
The pilot study showed that WBE administered to male and female rats for 14 consecutive days was palatable, well tolerated, and devoid of adverse effects up to the maximum practicable dietary inclusion level of 10%, corresponding to a mean intake of 6919 mg/kg body weight/day for the 2 genders. The only WBE-related finding was an increase of the cecal weight. In the absence of an adverse effect at the 10% inclusion level, a high-dose level of 7.5% WBE was selected as appropriate for the subsequent 90-day subchronic toxicity test, taking into account known rodent-specific adaptations to high amounts of fermentable fiber in the diet, such as the cecal enlargement, and recognizing that even at 7.5% the absolute dosage of WBE would amount to approximately 5000 mg/kg body weight/day, that is approximately 50-fold the mean WBE intake anticipated for a 60-kg person.
The 90-Day Rat-Feeding Trial: Mortality, Clinical Signs, General Condition, Behavior, and Ophthalmoscopy
All rats fed either control diets or WBE-containing diets survived to the end of the study. No WBE treatment-related clinical signs or changes in animal behavior, general physical condition, reactions to stimuli, grip strength, or motor activity were noted during the course of the study. Light brown feces were observed in all groups including the controls from day 3 of the treatment. This finding was attributed to the basal AIN 93G diet. No diarrhea or soft feces were observed during the study, an indication that WBE was gastrointestinally tolerated up to the highest dose. No ocular changes were observed by ophthalmology.
The 90-Day Rat-Feeding Trial: Animal Body Weights, Feed and Water Consumption
The cumulative body weight curves and weight gain curves of the rats fed the six different diets are presented in Figure 2. Overall, the mean body weights and body weight gains of both genders were comparable in the WBE-treated and control animals, with no significant changes noted that could be ascribed to WBE administration. The low weight gain values observed for all groups in the last week (week 13) reflect the preterminal procedures such as food deprivation for urine collection.
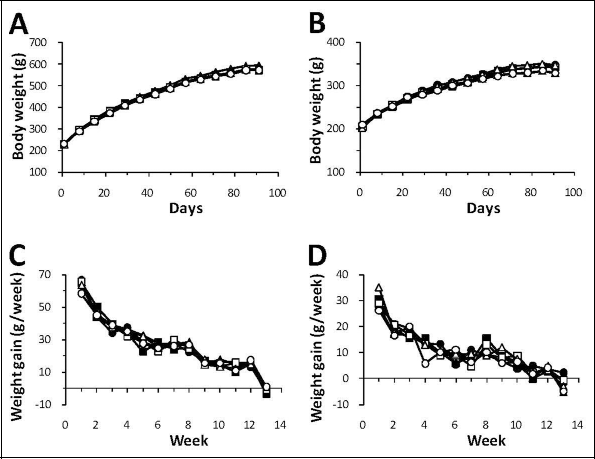
Cumulative mean body weight curves for male (A) and female rats (B) and mean weight gain for male (C) and female rats (D) during the 90-day study. ● indicates control group 1 (basal diet); ■, control group 2 (7.5% inulin); ▲, control group 3 (7.5% wheat bran); □, low-dose group 4 (0.3% WBE); Δ, mid-dose group 5 (1.5% WBE); ˆ, high-dose group 6 (7.5% WBE); WBE, wheat bran extract.
There were no significant test item-related differences in the mean daily food consumption, whether for male or female animals (Figures 3A and 3B). When compared to the basal diet control group, higher water consumption, attaining statistical significance during some weeks, was observed in the male animals fed WBE at all tested dose levels without a consistent dose response (Figure 3C). There was no statistically significant difference in water consumption by male rats compared to the 7.5% inulin control group, whereas compared to the 7.5% bran control group water consumption was lower in all WBE-treated groups. In the WBE-treated female animals, there was a minor statistically significant higher water consumption relative to the basal diet control group and to the 7.5% bran control group at the end of the study, whereas all WBE-treated female groups tended to have a lower water consumption compared to the 7.5% inulin group (Figure 3D).
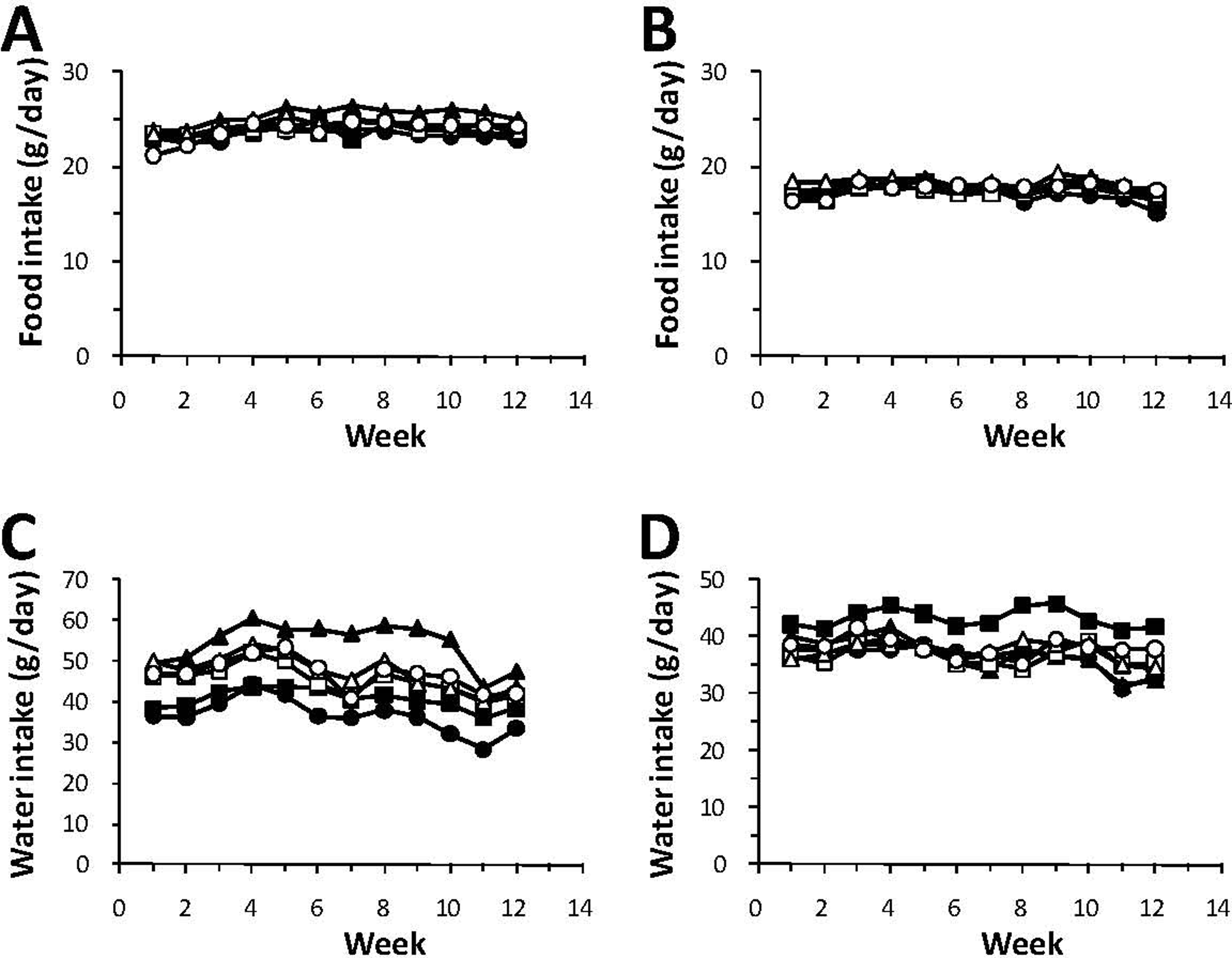
Mean food intake for male (A) and female rats (B) and mean water intake for male (C), and female rats (D) during the 90-day study. ● indicates control group 1 (basal diet); ■, control group 2 (7.5% inulin); ▲, control group 3 (7.5% wheat bran); □, low-dose group 4 (0.3% WBE); Δ, mid-dose group 5 (1.5% WBE); ˆ, high-dose group 6 (7.5% WBE); WBE, wheat bran extract.
The mean intake of WBE over the 13 week treatment period for the males and females, respectively, were 166 and 178 mg/kg bw/d (0.3% WBE group), 836 and 917 mg/kg bw/d (1.5% WBE group) and 4138 and 4571 mg/kg bw/d (7.5% WBE group).
The 90-Day Rat-Feeding Trial: Hematology and Blood Coagulation
There were no effects on coagulation parameters in WBE-treated animals compared to the basal diet control group. Relative to the basal diet control group, minor variations were noted throughout all WBE dose groups in both genders for the mean values of certain hematology parameters such as mean corpuscular (erythrocyte) volume (MCV) and mean corpuscular (erythrocyte) hemoglobin (MCH), red blood cell (erythrocyte) count (RBC), mean platelet (thrombocyte) volume (MPV), neutrophil, or lymphocyte (NE/LY %) values. Although on occasion statistically significant, these variations were generally within the normal ranges for the population of Wistar rats of this age and did not show any dose- or gender-related pattern, thus they were considered incidental and not related to WBE administration. Because there were no toxicologically significant changes, the hematology and coagulation data tables have been omitted for brevity.
The 90-Day Rat-Feeding Trial: Blood Chemistry
In the WBE-treated males, the variations in clinical chemistry parameters consisted of slightly yet statistically significant higher calcium and phosphorus and lower aspartate amino transferase (AST) mean values at all the dose levels, as well as lower bile acids (statistically significant at 0.3% and 7.5% WBE; Table 5). In addition, possible WBE-related changes were noted for other clinical chemistry parameters, without attaining statistical significance: decreased cholesterol at all the dose levels, lower alanine amino transferase (ALT) values at all the dose levels, and lower total bilirubin (T-BIL) in the 1.5% and 7.5% WBE groups. The values of the clinical chemistry parameters, which were significantly different when compared to the basal diet control group (calcium, phosphorus, and AST), were similar or differed to a lesser degree when compared to either the 7.5% inulin or the 7.5% bran control groups.
Blood chemistry data for male rats at week 13.
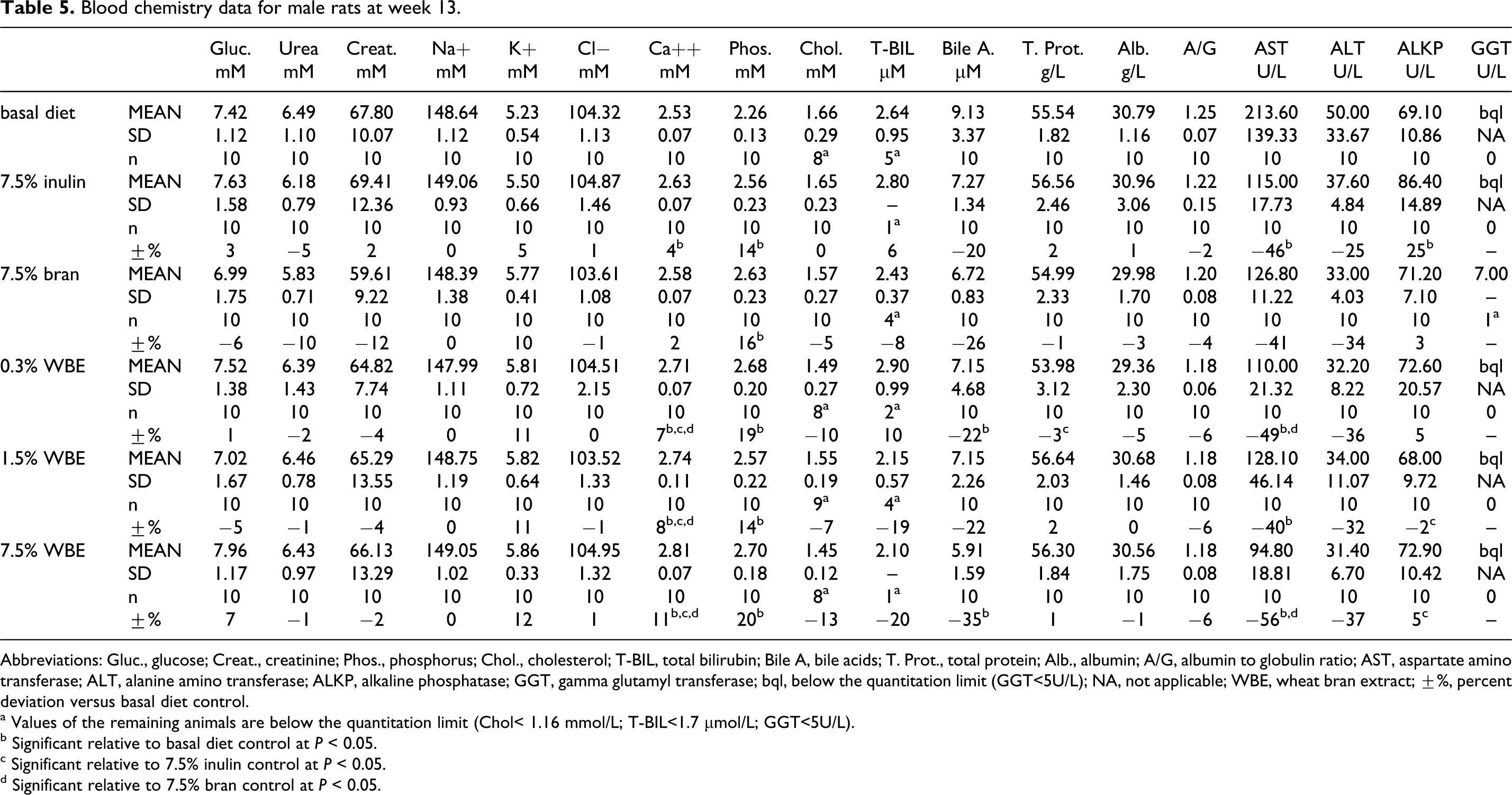
Abbreviations: Gluc., glucose; Creat., creatinine; Phos., phosphorus; Chol., cholesterol; T-BIL, total bilirubin; Bile A, bile acids; T. Prot., total protein; Alb., albumin; A/G, albumin to globulin ratio; AST, aspartate amino transferase; ALT, alanine amino transferase; ALKP, alkaline phosphatase; GGT, gamma glutamyl transferase; bql, below the quantitation limit (GGT<5U/L); NA, not applicable; WBE, wheat bran extract; ±%, percent deviation versus basal diet control.
a Values of the remaining animals are below the quantitation limit (Chol< 1.16 mmol/L; T-BIL<1.7 µmol/L; GGT<5U/L).
b Significant relative to basal diet control at P < 0.05.
c Significant relative to 7.5% inulin control at P < 0.05.
d Significant relative to 7.5% bran control at P < 0.05.
In the WBE-treated females, statistically significant variations in several clinical chemistry parameters in comparison with the basal diet control group occurred at all the WBE dose levels tested (Table 6). Potassium was statistically significantly increased at 1.5% and 7.5% WBE. AST, ALT, and alkaline phosphatase (ALKP) values were reduced compared to the basal diet control group at all the WBE dose levels tested, attaining statistical significance at 1.5% and/or 7.5 % WBE (AST, ALT, and/or ALKP, respectively). Urea was slightly higher and albumin-to-globulin ratio (A/G) ratio was slightly lower at 7.5% WBE. Calcium and phosphorus were slightly higher than in the basal diet control group at all WBE doses; however, no clear dose response was noted. Creatinine was higher in all female WBE groups and chloride was slightly decreased at 0.3% WBE relative to the basal diet control group; however, as no dose response was noted and no significant effects occurred in the male animals, these variations were not considered correlated with test item administration. A tendency to a slight reduction was noted in the T-BIL, bile acids, and cholesterol values in the 1.5% and 7.5% WBE groups yet not attaining statistical significance. Similar statistically significant differences for the female animals were observed relative to the 7.5% inulin control group for urea, creatinine, A/G ratio, ALT, and ALKP; but for potassium, calcium, and phosphorus, there were no or much smaller differences. Compared with the 7.5% bran control group, differences in urea, A/G ratio, and calcium were similar to those relative to the basal diet control group; but for creatinine, potassium, phosphorus, ALT, and ALKP, there were no or much smaller differences. Overall, the small changes observed in the WBE-treated groups were of a similar nature as compared to the 7.5% inulin or 7.5% bran control groups.
Blood chemistry data for female rats at week 13.
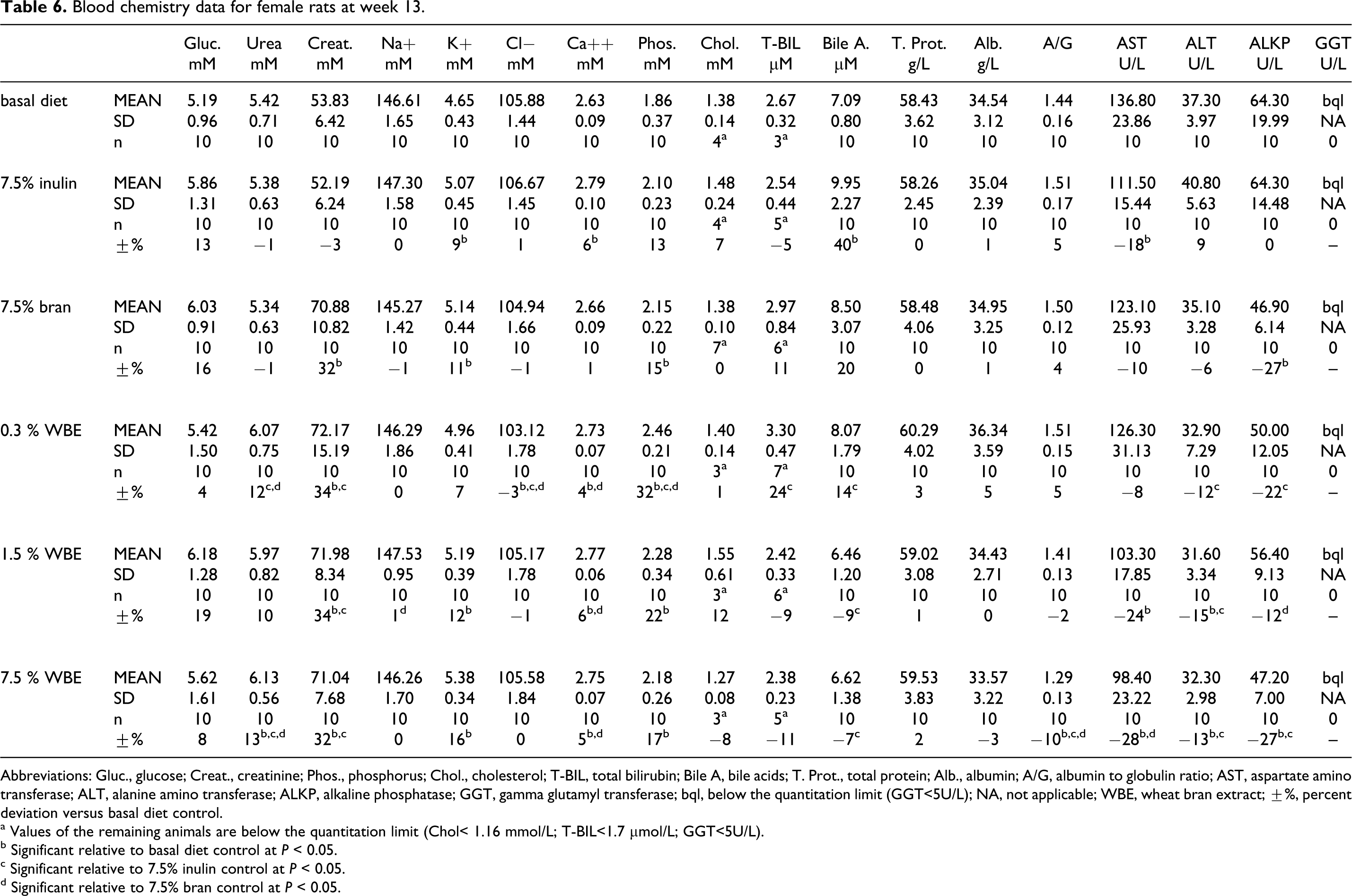
Abbreviations: Gluc., glucose; Creat., creatinine; Phos., phosphorus; Chol., cholesterol; T-BIL, total bilirubin; Bile A, bile acids; T. Prot., total protein; Alb., albumin; A/G, albumin to globulin ratio; AST, aspartate amino transferase; ALT, alanine amino transferase; ALKP, alkaline phosphatase; GGT, gamma glutamyl transferase; bql, below the quantitation limit (GGT<5U/L); NA, not applicable; WBE, wheat bran extract; ±%, percent deviation versus basal diet control.
a Values of the remaining animals are below the quantitation limit (Chol< 1.16 mmol/L; T-BIL<1.7 µmol/L; GGT<5U/L).
b Significant relative to basal diet control at P < 0.05.
c Significant relative to 7.5% inulin control at P < 0.05.
d Significant relative to 7.5% bran control at P < 0.05.
The 90-Day Rat-Feeding Trial: Urine Chemistry
There were no abnormal findings in the urinalysis parameters evaluated (pH, protein, glucose, nitrite, urobilinogen, bilirubin, ketones, leukocytes, erythrocytes, specific gravity, volume, and sediment).
Minor variations, on occasion attaining statistical significance (i.e. specific gravity, 7.5% WBE in female rats), were noted compared to basal control group; however, in the absence of a consistent dose- or gender-related consistent response, these variations were neither considered toxicologically significant nor an adverse effect of WBE administration. For brevity, data tables have been omitted.
The 90-Day Rat-Feeding Trial: Absolute and Relative Organ Weights
A statistically significant increase relative to the basal diet control group was noted in the absolute weights and relative-to-body-mass weights of the full cecum in all the male WBE-treated groups and the 7.5% WBE female group (Figure 4). Large increases in cecal weights relative to the basal diet control group were also observed for the 7.5% inulin control group, which were very similar to those of the 7.5% WBE group in both genders. The 7.5% bran control cecal weights were close to the basal control values.
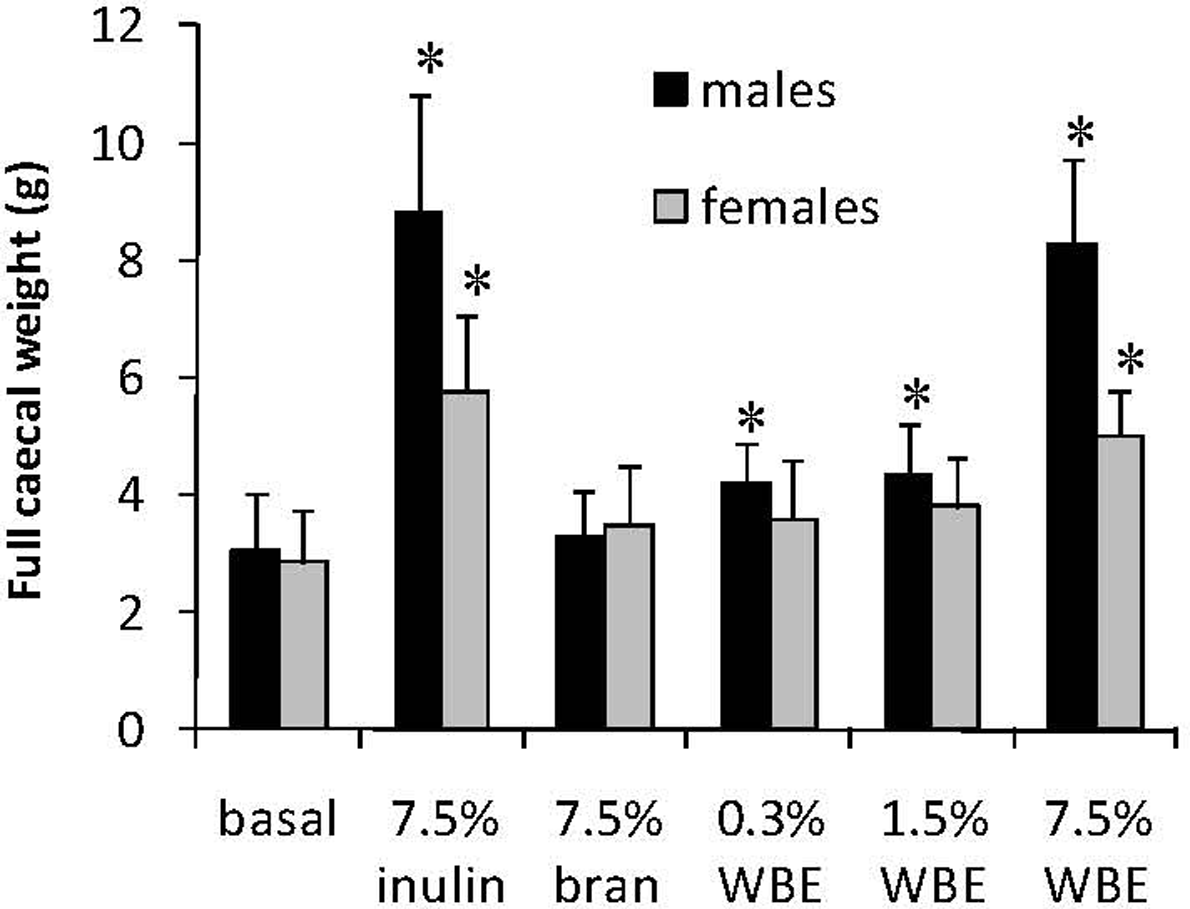
Full absolute cecal weight of male and female rats fed basal diet, 7.5% inulin, 7.5% wheat bran, or WBE at 0.3%, 1.5%, and 7.5%. Data are expressed as means and error bars represent the standard deviation. Asterisks indicate statistical significance (P < .05) relative to the corresponding value of the rats fed basal diet. WBE indicates wheat bran etxract.
There was no statistically significant effect of WBE on either absolute or relative-to-body-mass weights of the other organs of rats of either sex compared to the basal diet control group (data not shown). These organs included brain, liver, heart, spleen, kidneys, thymus, kidney, adrenals, testes (males), epididymides (males), uterus (females), and ovaries (females).
The 90-Day Rat-Feeding Trial: Pathological and Histological Findings
There was no evidence of treatment-related adverse macroscopic findings in any of the animals at postmortem examination. The main histopathological findings of the different organs examined are presented in Table 7. In the 7.5% WBE group, bilateral hypertrophy of the renal cortical tubules was recorded in 9 of 10 males and 7 of 10 females. The detected lesions were all considered minimal on a scale of minimal, mild, and marked. In the 1.5% WBE group, this finding was marginal, as it was observed in only 1 of 10 male and 2 of 10 females. Corticomedullary congestion and/or hemorrhage, noted in 3 of 10 7.5% WBE group females and 3 of 10 males and 4 of 10 females from the 1.5% WBE group, was considered related to the terminal procedures and unrelated to treatment. In the liver, minimal-to-mild hepatocellular vacuolation, mostly centrilobular, was noted in most of the animals examined of all analyzed groups. Based on the similar distribution throughout the groups, this finding was not ascribed to WBE administration but considered associated with the basal diet (AIN 93G diet). Other changes were considered associated with incidental, procedural, or dietary factors (Table 7).
Incidence of Histopathological Findings in Male and Female Rats
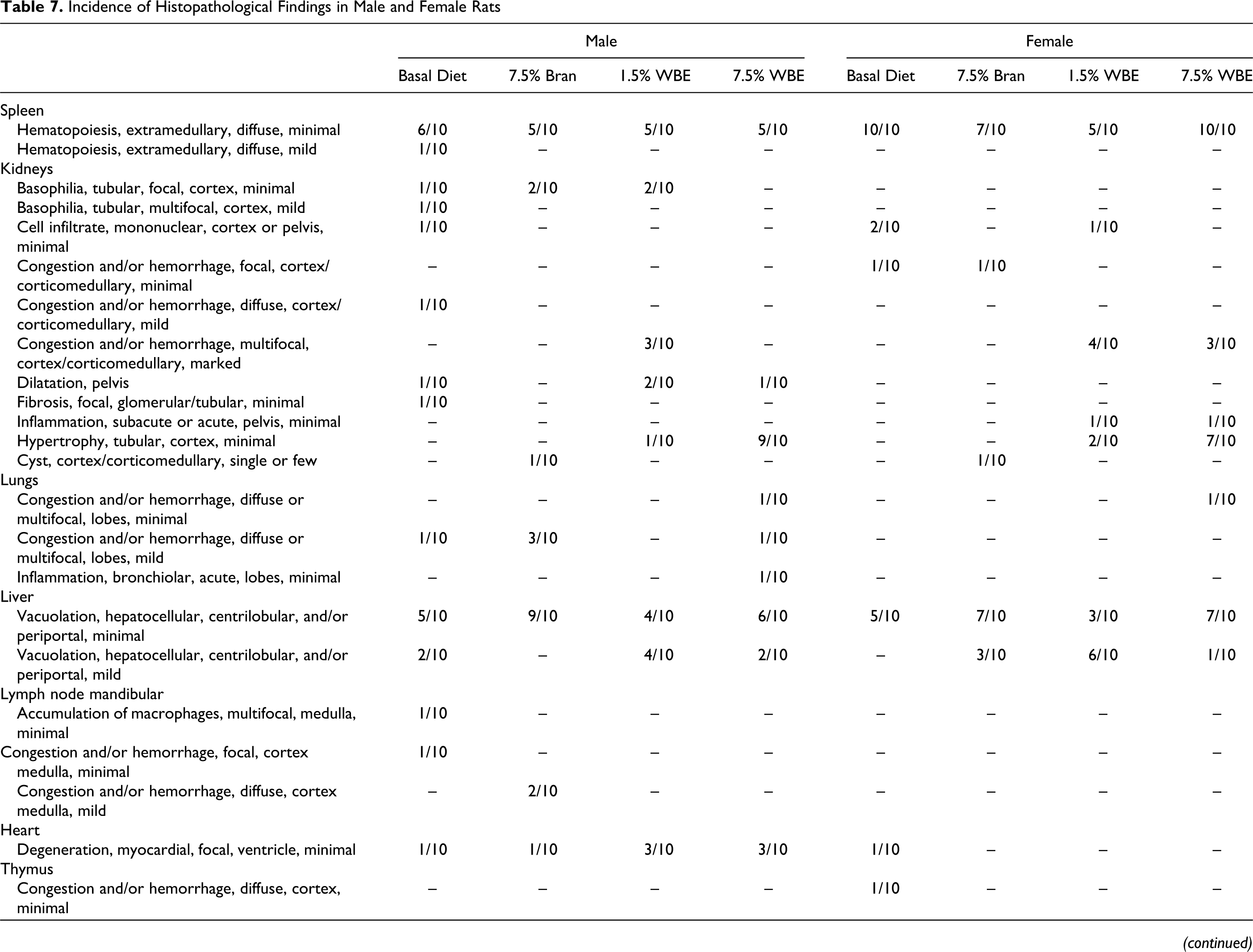
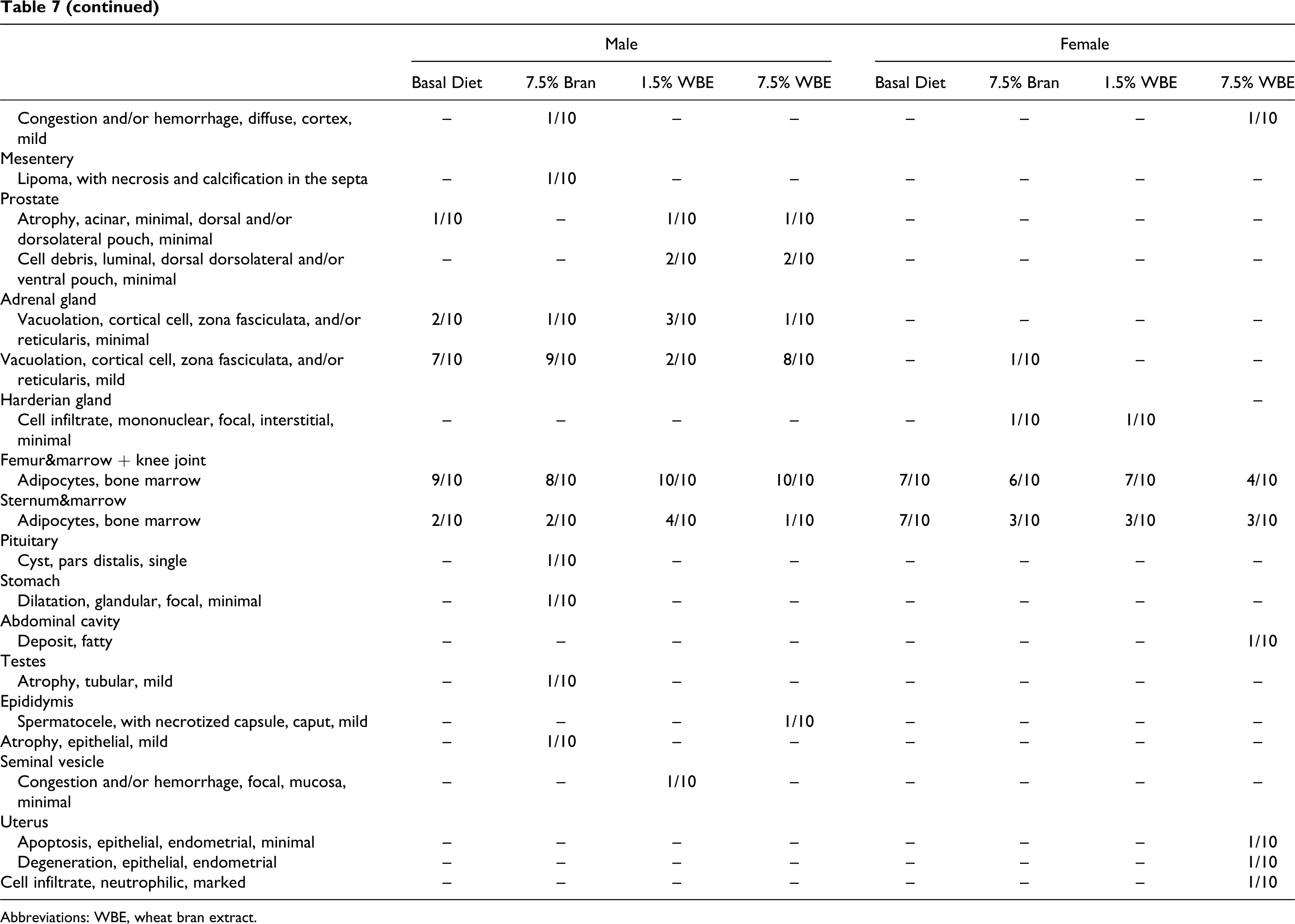
Abbreviations: WBE, wheat bran extract.
Discussion
Wheat bran extract has been investigated in vitro genotoxicity and in vivo subchronic toxicity assays. All studies followed accepted testing guidelines including GLP, as recommended by international regulatory agencies (OECD, EFSA, and US FDA).
The results of the Ames test demonstrate that WBE has no mutagenic activity, neither without nor with metabolic activation, on any of the 5 S typhimurium and E coli tester strains under the test conditions used. Moreover, in 2 independent experiments using 3- and 20-hour treatment periods with or without metabolic activation on V79 mammalian cells, WBE did not induce significant increases in the number of chromosomal aberrations, up to and including the concentrations that clearly showed growth inhibition of the cells.
The results reported here with WBE are consistent with genotoxicity information found in the literature for other oligosaccharide preparations with prebiotic properties. Fructo-oligosaccharides (FOS), which consisted of oligomers of β-1,2-linked
The main components of WBE, AXOS, and β-glucan are not digested in the upper gastrointestinal tract of animals but are readily fermented in the large intestine. 11,32,33 This fermentation typically results in the formation of hydrogen and carbon dioxide and of the short-chain fatty acids (SCFA) acetate, propionate, butyrate, and lactate. 34,35 Further, the fermentation of AXOS is associated with the release and absorption of ferulic acid. 36 Short-chain fatty acids acetate and ferulic acid are both benign well-metabolized digestion products 13,37,38 that are also produced upon digestion of natural plant-derived fibers present in a regular healthy diet rich in cereals, fruits, and vegetables. Therefore, as supported by the current study, no systemic toxicity in animals is expected as a result of the ingestion and metabolism of these main WBE constituents.
Wheat bran extract had no effect on behavior, body weight, weight gain, and food consumption of rats. However, the addition of dietary fiber to the diet was associated with a tendency toward increased water consumption. This effect was most clearly present in the male animals fed the wheat bran control diets but was also seen, to a lesser extent, in the inulin and WBE-treated animals. Increased water intake was previously observed in rats fed 7.5% of psyllium bran in the diet. 39
Wheat bran extract intake did not affect organ weight, with the exception of full cecal weight, nor was it associated with macroscopic effects in any of the organs. The observed effect of WBE on cecal weight is considered a physiological adaptation to consumption of fermentable nondigestible carbohydrates, with closely matching results for 7.5% inulin and 7.5% WBE. It is well documented that dietary inclusion at high doses of fermentable nondigestible carbohydrates, such as xylo-oligosaccharides (structurally related to AXOS, the main component of WBE), inulin and FOS, results in increased cecal weights. 40 –43 As the main fraction of WBE is not digested in the upper gastrointestinal tract, the addition of WBE to the diet results in an increased presence of fermentable material in the colon and cecum. 44 This typically results in an increase of the weight of the cecum and colon. 45
Histopathologically, minimal bilateral hypertrophy of the renal cortical tubules was observed in males and females, in particular in the high-dose WBE group. These findings were not accompanied with any degenerative changes or changes in kidney weight. This suggests that they reflect an adaptive response rather than a toxic effect. The renal effect is not inconsistent with an adaptive response to the excretion of increased levels of certain serum electrolytes as observed in the plasma clinical chemistry data.
There were no toxicologically relevant differences between the control and WBE-treated groups on hematology parameters in the male and female animals. When compared to the basal diet control group, certain blood chemistry parameters in the WBE-treated groups showed minor differences that are likely related to physiological effects resulting from WBE administration. Variations in serum levels of creatinine, potassium, calcium and/or phosphorus, AST, ALT, and/or ALKP, and/or bile acids were noted in the WBE-treated animals. However, a similar pattern of changes was seen in animals treated with control diets containing inulin or wheat bran, suggesting that this profile reflects physiological changes associated with the presence of increased levels of nondigestible carbohydrates in the diet. The increased levels of serum calcium, potassium, and phosphorus are consistent with published observations that the intake of fermentable nondigestible carbohydrates, such as soluble fibers, promotes the bioavailability of these electrolytes from the gut. 46 –48 Furthermore, the observed reduction in blood serum activity of the main liver marker enzymes taken together with unchanged serum total protein levels and reduced serum bile acid and cholesterol levels, without any liver necrosis, may be considered a positive effect. These findings may be consistent with increased dietary fiber intake, as similar changes were seen in the 7.5% inulin control group and the 7.5% bran control group. The reason for decreased activity of the serum liver enzymes could not be determined, although it may be associated with fiber content of the diet and decreased levels of cholesterol. The hypocholesterolemic effect of plant-derived dietary fiber has been ascribed to fiber-induced alteration of intestinal absorption, intestinal or pancreatic hormone secretion, lipoprotein metabolism, bile acid metabolism, or fermentation by-products, and their effects on hepatic cholesterol synthesis. 39 Furthermore, certain dietary fibers were reported to increase fecal bile acid excretion. 49
In conclusion, WBE does not have mutagenic or clastogenic activity under the conditions of the genotoxicity assays used, and the subchronic toxicity study revealed no treatment-related adverse affects due to the feeding of WBE to rats during 13 weeks at levels up to 7.5% in the diet. The no-observed-adverse-effect levels (NOAELs) were established by the highest tested doses: 4138 mg/kg bw/d in male rats and 4571 mg/kg bw/d in female rats or the combined daily average intake of 4.4 g/kg bw. The possible daily intake in the United States of WBE based on the intended use of WBE as an ingredient in a broad range of food products at levels up to 3.2 g/serving was estimated using the CSFII 2003-04 (USDA, 2005) database of food products. The consumption by the US population of food categories intended for addition of WBE resulted in estimated mean all-user intakes of WBE of 5.0 g/person per d (100 mg/kg bw/d). The 90th percentile all-user intakes of WBE from all intended food uses by the total population was 10.1 g/person per d (220 mg/kg bw/d). The FDA commonly uses the estimated daily intake for the 90th percentile consumer of a food additive as a measure of high chronic dietary intake. Considering that this potential high chronic intake is still about 20-fold lower than the NOAELs found in the current study, these results demonstrate the safety of WBE for oral consumption.
Footnotes
Acknowledgments
We thank the scientific, technical, and quality assurance staff of LAB Research Ltd at the test facility of Veszprém employees (Hungary) for skillful conduct of these studies. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Notes
Isabelle François, Olivier Lescroart, Wim S. Veraverbeke, and Willem Broekaert are presently employed by Fugeia NV, which manufactures the Wheat Bran Extract product and is the sole sponsor source of funding for the studies described herein. Raluca Kubaszky, Judit Hargitai and David Esdaile are presently employed by LAB Research Ltd., and Erzsébet Beres is a retired ex-employee of LAB Research Ltd. Andrew Cockburn is presently employed by Toxico-Logical Consulting Ltd. Madhu Soni works as a Consulting Toxicologist.
