Abstract
Introduction
N-trans-feruloyltyramine (NFT) is a bioactive compound present in many plant sources. The purpose of the studies was to investigate adverse effects, if any, of NFT produced through precision fermentation.
Methods
An in vitro Ames test was performed with NFT using bacterial strains at concentrations up to 1580 µg/plate with and without S9. The in vitro micronucleus assay was performed in human peripheral blood cells in culture, with and without, metabolic activation at three different doses. In the subchronic toxicity study, adult Sprague Dawley rats (10/sex/group) were fed diets prepared with target doses of 0, 5000, 10,000 or 20,000 ppm of NFT for 90 days.
Results
In the Ames assay, there were no NFT-related or concentration dependent increases in revertant colony numbers in any of the tester strains. In the in vitro micronucleus assay, there was no statistically significant increase in the number of binucleated cells with micronuclei compared to the vehicle control. NFT was found to be non-genotoxic when evaluated in the in vitro Ames and micronucleus assays. In the 90-day rodent study, NFT was well tolerated, with no related adverse findings observed at any of the dose levels tested. There were no NFT related adverse histopathological changes observed in the high dose group of both the sexes.
Conclusion
The No observed adverse effect level of NFT was determined as 1474 mg/kg body weight/day in males and 1958 mg/kg body weight/day in females based on the actual intake at the dose levels tested and under the experimental conditions employed.
Introduction
N-trans-feruloyltyramine (NFT) is an active phenylpropanoid compound that is present in several bacterial, mammalian, and plant species. 1 NFT was first isolated as a natural component of Solanum melongena L. (i.e., eggplant) roots 2 and has since importantly been identified in a range of edible plants including garlic,3,4 New Zealand spinach, 5 and black pepper.6,7 In plants, hydroxycinnamic acid amides (HCAAs) like NFT are associated with immune response due to their antioxidant activity, but they are thought to also function in a range of growth and development processes, such as flower formation and tuberization. 8 In mammals, NFT plays an important role in many metabolic functions, has been shown to assist in recovery of lost functions and is known to have antioxidant properties. 9 An example of NFT’s impact on metabolic functions can be seen in its modulation of hepatic nuclear factor 4α (HNF4α), where it serves as an agonist for HNF4α to initiate lipophagy to reduce excessive fat buildup in the liver, thus reducing the risk of fatty liver disease.10–12 Other examples of NFT’s part in providing a health benefit are its role in reducing cytotoxicity in human neuroblastoma cells,1,13 neurotoxicity in animal model brains, 14 and decreasing the risk of certain cancer types. 15 Recently, naturally occurring NFT, as a bioactive compound in hemp hulls, was demonstrated to have the potential to improve gut barrier function. 16 In a natural dietary context, NFT is often consumed as part of plant-based diets, contributing to the overall health-promoting effects of these foods. However, for the potential use of NFT in functional foods and nutritional supplements, its safety must first be established.
In a recent study, Chae et al., investigated the toxicological safety of a black pepper extract (BPE) in an in vitro Ames test and a 90-day rodent study that contained NFT. 17 The safety of BPE enriched with ∼50% NFT was demonstrated through lack of mutagenicity and no adverse histopathology findings. This present study aimed to investigate the safety of purified NFT derived from ferulic acid through precision fermentation of the strain Yarrowia lipolytica 3599_7. Y. lipolytica is a Generally Recognized as Safe (GRAS) substance that falls under the phylum of fungi and is also considered a budding yeast.18,19 Y. lipolytica has been commonly used in the production of other chemicals and organic acids 20 due to its rapid growth rate, flexible metabolic regulation, and its efficient use of low cost substrates.21,22 NFT is naturally present in several parts of the plant and has generally been studied using plant extracts.23–25 In this study, the choice to produce NFT through fermentation over plant extracts was driven by the advantages of employing Y. lipolytica, as it most importantly allows for the production of high purity NFT. Additionally, other factors such as potential scalability, sustainability, and adaptability to real-world applications were considered when comparing the two methods. 26
Although there is published literature on NFT administration in mouse and rat animal models to elucidate NFT’s effect on other cellular processes,1,4,27 this is the first study conducted to investigate the potential adverse effects, if any, of purified NFT. In this study, an in vitro GLP Ames assay and micronucleus assay were performed to test for genotoxicity while the long-term safety was inferred through a 90-day subchronic toxicology study in rats. Different doses of NFT were administered based on a 14-day toxicology study that was conducted in rats to determine the appropriate dose of NFT. The main objective of the 90-day toxicology study was to evaluate the potential oral subchronic toxicity in male and female rats continuously exposed to NFT in the diet.
Materials and methods
NFT preparation
Composition of N-trans-feruloyltryamine produced from Yarrowia lipolytica 3599_7.
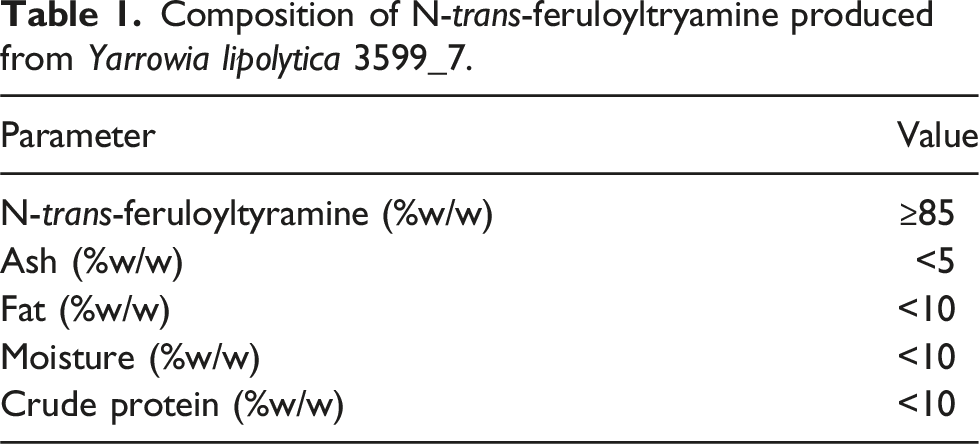
The construction of the production strain Y. lipolytica 3599_7 was conducted via the stable insertion of 6 genes of plant origin, synthesized de novo and codon-optimized prior to incorporation, into the host genome. Genetic stability and cell productivity were confirmed after culturing the strain in shake flasks for 72 generations prior to inoculating a 2L fermentation.
Ames test
The Ames test was performed to evaluate the potential mutagenic activity of NFT as per OECD 471 Guideline for the Testing of Chemicals: Bacterial Reverse Mutation Test. The test examined NFT’s ability to revert four histidine-dependent strains of Salmonella typhimurium - TA98, TA100, TA1535, TA1537 and one tryptophan dependent strain, Escherichia coli WP2 uvrA (pKM101) in the presence and absence of rat liver S9 activation system with preincubation and standard plate incorporation method. 28 NFT was tested at dose levels of 15.8, 50.0, 158.0, 500.0, 1580.0 μg/plate first using the preincubation method in the presence and absence of S9 followed by the plate incorporation method in the presence and absence of S9. The positive control substances used were sodium azide, acridine mutagen, ICR 191, 2-nitrofluorene, 2-aminoanthracene, and 4-nitroquinoline-n-oxide.
Bacterial revertant colonies were counted after incubation period for Salmonella typhimurium TA98, TA100, TA1535, TA1537 and Escherichia coli WP2 uvrA (pKM101). The strains were counted manually, and the results were expressed as numbers of revertant colonies per plate (CFU/plate). All plates from a single experiment, including control item, positive control plates, and vehicle control plates, were counted using the same method at the same time to reduce errors and avoid false positives. Plates that were too numerous to count were counted by dividing into 2 to 4 equal parts.
The following criteria were used to interpret results. The mean revertant colony counts for spontaneous control and vehicle control were to be within the laboratory’s historical control range to consider the experiment valid. The test item was considered a mutagen if a reproducible, dose-related increase in the number of revertant colonies in one or more strains was observed. The test item was considered a weak mutagen if a reproducible, dose-related increase in the number of revertant colonies in one or more strains but the number of revertants is not double the number of revertant colonies. The test item was considered a non-mutagen if no dose-related increase in the number of revertant colonies were observed in at least two independent experiments. If the test item could not be identified clearly as a mutagen or a non-mutagen, the results were classified as inconclusive.
In vitro mammalian cell micronucleus test
The in vitro mammalian cell micronucleus (IVMN) test was conducted in accordance with following guideline: OECD test guideline 487, in vitro mammalian cell micronucleus test, adopted on 19 July 2016, Corrected: 4 July 2023. 29 The protocol was approved by the ethical committee (Protocol No. PDCEC/01/M34/2021-22 valid up to 9th February 2025) for in vitro genotoxicity tests by chromosomal aberration and/ or micronucleus tests using human peripheral blood lymphocytes. The study was also conducted in compliance with the OECD Principles of Good Laboratory Practice No. 1 as revised in 1997 and adopted on November 26th, 1997 ENV/MC/CHEM (98)17 concerning mutual acceptance of data in the assessment of chemicals, [C(81)30(FINAL)] and [C(89)87(FINAL)] and as per Standard Operating Procedures prevailed during the study. 30
The IVMN test was conducted to assess the potential of NFT to induce clastogenicity or aneugenicity in human peripheral blood lymphocytes in culture. Peripheral blood was collected from healthy human volunteers and blood lymphocyte cultures were prepared for testing by mitogenic stimulation with 2% Phytohemagglutinin for about 48 h. For dose range finding, six dimethyl sulfoxide stocks of NFT were prepared to obtain the exposure concentrations from 4.12 to 1000 μg/mL in three experimental conditions: absence of S9 mix for short and long exposure (4 h and 24 h) and presence of S9 mix for short exposure (4 h). For micronuclei evaluation, three dimethyl sulfoxide stocks (100X) of NFT were prepared for 3 experimental conditions: absence of S9 mix in short and long exposure (4 h and 24 h), and presence of S9 mix for short exposure (4 h). Concurrent vehicle control (1% v/v DMSO) and positive controls (vinblastine sulfate salt, cyclophosphamide monohydrate, and mitomycin C) were included in the study.
Three NFT stocks were spiked at 1% v/v duplicate (N = 2) to lymphocyte cultures to achieve exposure concentrations from 27.78 , 83.33, and 250 μg/mL in absence (4 h) and presence of S9 mix (4 h) for a short term exposure. For long term exposure three NFT stocks were spiked at 1% v/v to lymphocyte cultures in duplicates (N = 2) to obtain the exposure concentrations 9.26, 27.78 and 83.33 μg/mL in the absence (24 h) of S9 mix. The exposed cultures were collected by centrifugation at 1000×g for 10 min at 22°C and subjected to hypotonic treatment (1% w/v Trisodium citrate dihydrate). Cells were then fixed by adding an equal volume of cold fixative solution. The fixed cells were collected by centrifugation and then suspended with fixative solution. The fixed cell suspension was stored in a refrigerator overnight prior to slide preparation.
Collected cells were mounted on slides (two slides per test concentration). Five hundred cells were assessed for cytotoxicity via the cytokinesis-block proliferative index (CBPI), and frequency of micronuclei was assessed in 2000 binucleated cells.
CBPI was determined as follows:
1. Cytokinesis Block Proliferative Index (CBPI) was calculated as follows, by scoring at least 500 cells per concentration.
2. % Cytotoxicity or Cytostasis, which indicated the inhibition of cell growth of treated cultures in comparison to control cultures, was calculated using formula:
Micronuclei frequency was calculated as follows:
Statistical analysis was performed on the results by Chi-square test for pair wise comparisons between each treated and vehicle control groups using GraphPad Prism Software, version 5.03. The data point was considered significant at p < 0.05 for all statistical analysis in comparison to the respective vehicle control.
90-day toxicology study
The study was conducted to determine the systemic toxic potential of NFT in Sprague Dawley rats when fed daily, orally through powdered feed at graded dose levels for 90 consecutive days and to determine the no-observed-adverse-effect level (NOAEL). This study was conducted in accordance with the following guideline: the OECD test guideline 408 (adopted 25th June, 2018), approved IAEC protocol ((ABD/IAEC/PR/251-21-24 and Approval date: 06 August, 2021). 31 The study was also conducted in compliance with the OECD Principles of Good Laboratory Practice No. 1 as revised in 1997 and adopted on November 26th, 1997 ENV/MC/CHEM(98)17 concerning mutual acceptance of data in the assessment of chemicals, 30 [C(81)30(FINAL)] and [C(89)87(FINAL)] and as per standard operating procedures that prevailed during the study. 32 The study also adhered to the US FDA Toxicological Principles for the Safety Assessment of Food Ingredients, Redbook 2000, IV.C. 4. a.22. 33
Powder feed-based dose formulations of NFT were prepared at low dose (1 mg/g) and high dose (100 mg/g) strength. Dimethyl sulfoxide (DMSO) based dose formulations of NFT were prepared at low test concentration (0.100 mg/mL) and high test concentration (200 mg/mL). High performance liquid chromatography (HPLC-PDA) method was then performed to validate NFT in dose formulation through quantification of NFT in each dose (Figure 1). N-trans-feruloyltryamine chromatogram. Chromatogram of N-Trans-feruloyltyramine in high dose formulation sample.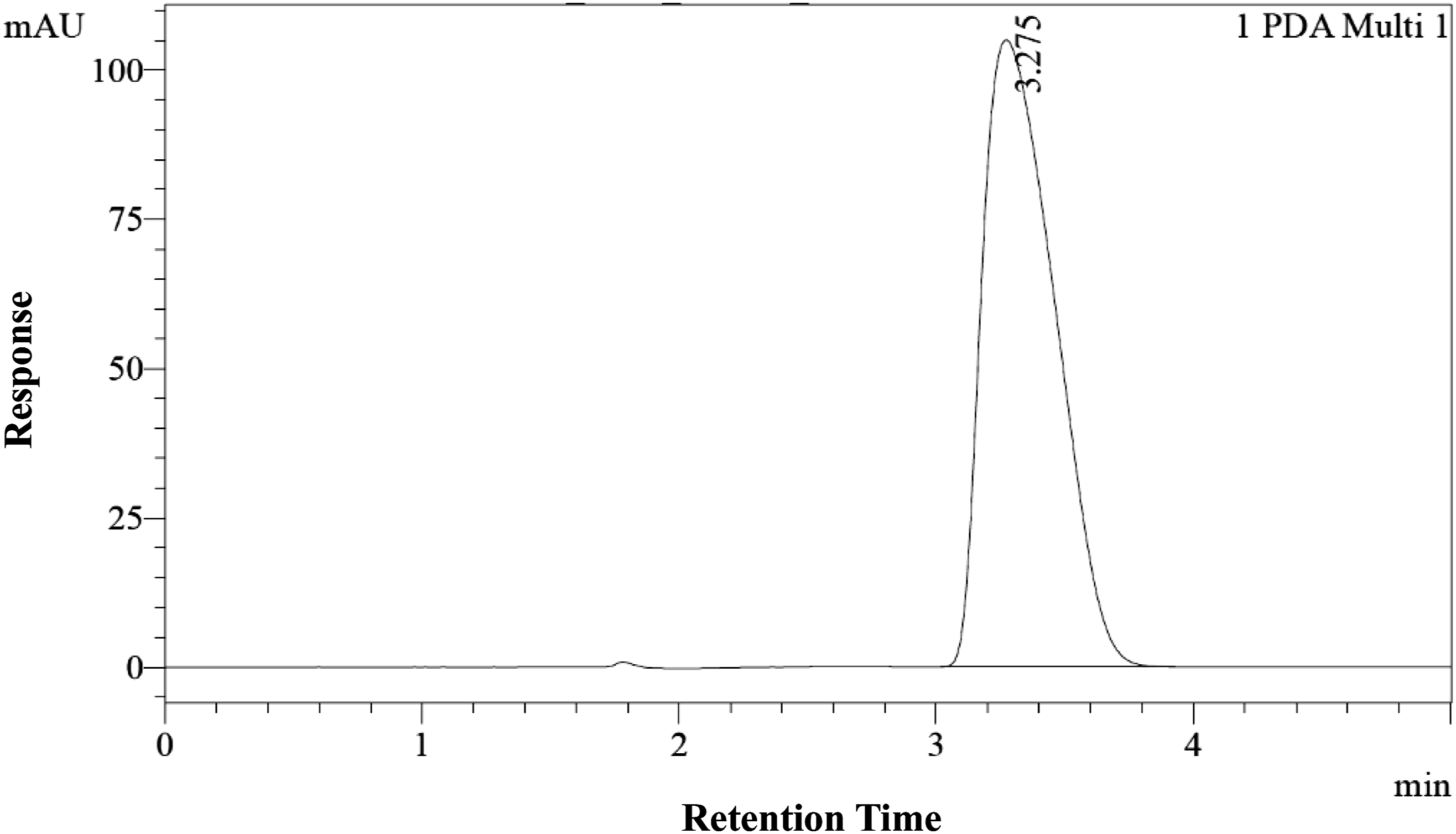
Animals
Animal care complied with the regulations of the Committee for the Control and Supervision of Experiments on Animals (CCSEA) guidelines for laboratory animal facility published in the Gazette of India, 1998 34 and Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC). 35 The protocol (animal research and reporting of experiments) for the in vivo study prepared in compliance with the CCSEA guideline was reviewed and approved by the Institutional Animal Ethics Committee (IAEC) prior to the initiation of the study. A total of 80 Sprague Dawley rats were used (40 males; 40 females). Animals were acclimated to experimental room conditions for 5 and 6 days for males and females respectively, during which they were observed once daily for clinical signs, twice daily for mortality/morbidity, and subjected once weekly to detailed clinical examination prior to randomization. For treatment, animals were selected based on veterinary assessments that determined no clinical signs of disease or injury.
Treatment
The dose levels for the 90-day study were selected based on a 14-day toxicity study. The selected target intake (dose) levels and treatment groups were: vehicle control (G1; NFT, 0 ppm), low dose (G2; NFT, 5000 ppm), mid dose (G3; NFT, 10,000 ppm) and high dose (G4; NFT, 20,000 ppm). NFT mixed with powdered feed was fed orally in graduated doses to several groups of experimental animals, one dose level per group for a period of 90 days in Sprague Dawley rats. The animals were fed ad libitum throughout the acclimatization and experimental period except for a period of fasting. RO + UV treated and autoclaved water was available ad libitum throughout the acclimation and experimental periods. On the final day of the acclimation period, animals were stratified according to weight after which they were distributed into four experimental groups as mentioned above, such that body weight variation of animals selected for the study did not exceed ±20% of the mean body weight of each sex.
Assessments
Clinical observations
Animals were observed for mortality and morbidity twice a day throughout the study period and once on the day of necropsy. All the animals were observed once daily for visible clinical signs of disease or injury by cage side observation. Detailed clinical examinations were performed during acclimatization period on day of randomization (day before treatment) and at weekly intervals (Day 8, 15, 22, 29, 36, 43, 50, 57, 64, 71, 78, 85 & 90) during the treatment period.
Body weight and feed consumption
Individual animal body weight (g) was recorded on the day of randomization, on day 1 of treatment before dosing and once in a week (±1 day) thereafter throughout the experimental period (Day 8, 15, 22, 29, 36, 43, 50, 57, 64, 71, 78, 85 & 90). Individual animal fasting body weight was recorded on the day of scheduled necropsy (Day 91) before blood collection. The body weight change (%) was calculated compared to the day 1 body weight. Feed input and feed leftover was measured at least once in a week from day 1 of treatment. Feed consumption (g/animal/day) was calculated weekly once (±1 day).
Ophthalmological examination
Ophthalmological examination was performed using an indirect ophthalmoscope for all the animals before initiation of treatment (during acclimatization period). Examination of vehicle control (G1) and high dose groups (G4; NFT, 20,000 ppm) was performed in the 13th week of the study.
Neurobehavioral assessment
Functional observation battery of tests (FOB) was carried out during 12th week for vehicle control (G1) and high dose (G4; NFT, 20,000 ppm) group animals. The following functional observation battery of tests were performed: home cage measurements (Posture, respiratory pattern, vocalization, palpebral closure), hand-held measurements (Ease of removal, ease of handling, lacrimation, salivation, chromodacryorrhea, hair coat, muscle tone, exophthalmos), open field measurements (clonic/tonic involuntary movement, gait, mobility, piloerection, arousal, stereotype, number of defecations, number of urinations, number of rearing), reflex measurements (visual response, touch response, auditory response, pupil reflex, air righting reflex, pinna reflex, tail pinch, palpebral reflex, propioception), neuromuscular measurements (grip strength test, hind limb foot splay), physiological measurement (rectal temperature measurement).
Clinical pathology (hematology, clinical chemistry, coagulation, urinalysis, and hormone analysis)
Clinical pathology was performed for all animals, including, hematology, coagulation, clinical chemistry, urinalysis, and hormone analysis. Blood samples from overnight fasted animals were collected from all the surviving animals on day 91 (G1, NFT 0 ppm; G2, NFT 5000 ppm; G3, NFT 10,000 ppm; and G4, NFT 20,000 ppm groups). Water was provided ad libitum during the fasting period. Blood was collected from retro-orbital plexus with a fine capillary tube under mild isoflurane anesthesia.
The following were assessed for hematological analysis using the ADVIA 2120 (Siemens Limited) Hematology Analyzer: hematocrit, hemoglobin concentration, red blood cell (RBC) count, white blood cell (WBC) count, platelet count, erythrocyte indices (mean cell volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration), and differential leukocyte count (neutrophils, lymphocytes, monocytes, eosinophils, basophils, reticulocytes). Coagulation was also assessed using KC4 Delta coagulation analyzer for prothrombin time and activated partial thromboplastin time.
Clinical chemistry parameters were estimated using a Dimension XPAND Plus clinical chemistry analyzer (Siemens Limited). Electrolytes like sodium, potassium, and chloride were analyzed by electrolyte analyzer (Siemens RC 744). The parameters assessed were: alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP) gamma glutamyl transferase (GGT), glucose (GLUC), total cholesterol (CHOL), high density lipoprotein (HDL), low density lipoprotein (LDL), triglycerides (TGL), blood urea nitrogen (BUN), creatinine (CRE), total protein (TP), total bilirubin (TBI), albumin (ALB), globulin (calculated) (GLOB), sodium (Na+), potassium (K+), chloride (Cl-), phosphorus (PHOS), and Calcium (Ca).
For urinalysis, the animals were fasted overnight with free access to water ad libitum. Urine samples were collected from all animals in the vehicle control and main study groups during the last week of dosing (week 13). Urine specimens were analyzed for the following parameters using CLINITEK Status + urine analyzer: physical examination (volume, color, appearance/clarity), chemical examination (pH, specific gravity, occult blood, urobilinogen, ketone bodies, protein, bilirubin, glucose, nitrite, leukocytes), microscopic examination (epithelial cells, RBC, casts, crystals, pus cells/WBC, bacteria, yeast).
For hormone analysis, Thyroxine (T4), Triiodothyronine (T3) and thyroid stimulating hormone (TSH) concentrations were estimated from serum with ELISA kits by the competitive inhibition enzyme immunoassay technique. On day 90, the stages of estrus cycle of all females were determined through vaginal cytology.
Organ/tissue collection and histopathology
On Day 91 of the study, necropsy was performed on all animals after euthanasia. Necropsy also included examination of the external surface of the body, all orifices, cranial, thoracic and abdominal cavities and their contents. The following tissues and organs were collected, weighed, and preserved: adrenals, brain (including cerebrum, cerebellum, mid brain, and pons), epididymides, heart, kidneys, liver, ovaries, prostate, seminal vesicles with coagulating glands, spleen, testes, thymus, thyroid and parathyroid glands, pituitary gland, uterus with cervix. The following organs and tissues were collected and preserved (not weighed): aorta, bone marrow smear, caecum, colon and rectum, duodenum, jejunum and ileum, Peyer’s patches, esophagus, eyes with optic nerve, lungs, lymph nodes (mesenteric & submandibular), pancreas, salivary glands, sciatic nerve, skeletal muscle, skin with mammary gland, spinal cord (cervical, mid-thoracic, and lumbar), sternum, stomach, and trachea.
Histopathological examination was performed on all the preserved organs and tissues from G1, NFT 0 ppm animals and G4; NFT, 20,000 ppm animals. Organs were sectioned and stained with hematoxylin and eosin.
Statistical analysis
For all parameters assessed, statistical analysis using GraphPad Prism Software, version 5.03, was performed. Data were analyzed by One way ANOVA. When One-way ANOVA was significant (p < 0.05), Dunnett’s post-test was performed for comparing treatment groups (G2, NFT 5000 ppm; G3, NFT 10,000 ppm; and G4, NFT 20,000 ppm) with the vehicle control group (G1, NFT 0 ppm). Student’s t test was considered for analysis relevant parameters, and male and female data were considered separately for analysis. All analysis and comparisons were evaluated at the 95% level of confidence (p < 0.05).
Results
Mutagenicity study
The potential mutagenic activity of NFT was investigated in a bacterial reverse mutation test (Ames assay). This study was conducted with histidine-dependent strains of Salmonella typhimurium (TA98, TA100, TA1535, and TA1537) and Escherichia coli WP2 uvrA (pKM101) in the presence or absence of rat liver S9 metabolic activation using pre-incubation and standard plate incorporation methods. Ultrapure water (as a component of the S9 mix or phosphate buffer solution) served as the negative control, and dimethylsulfoxide (DMSO) served as the vehicle control.
A preliminary cytotoxicity determination was conducted with NFT at concentrations up to 5000 µg/plate in S. typhimurium TA100 and E. coli WP2 uvrA (pKM101) (with and without S9) to identify an appropriate dose range for the mutagenicity assay. This test was performed using pre-incubation and standard plate incorporation methods, each in duplicate. Cytotoxicity was observed in S. typhimurium TA100 at 1580 and 5000 µg/plate and in E. coli WP2 uvrA (pKM101) at 5000 µg/plate.
Bacterial reverse mutation test conducted with N-trans-feruoyltyramine (pre-incubation and plate incorporation methods).

NA: not applicable.
amean of replicate (3) plates.
bMutagenicity Factor = mean revertants (NFT)/mean revertants (Negative Control).
cNegative: DMSO.
dPositive control agents: 2NF: 2-nitrofluorene; SA: Sodium Azide; ICR 191: Acridine mutagen ICR 191; 4NQ: 4-Nitroquinoline N-oxide; 2AA: 2 - aminoanthracine.
Therefore, under the conditions of this assay, it was concluded that NFT is not mutagenic at concentrations up to 500 μg/plate.
In vitro mammalian micronucleus assay in human peripheral blood lymphocytes
The potential for clastogenic and aneugenic activity of NFT was evaluated in an in vitro mammalian cell micronucleus test conducted with human peripheral blood lymphocytes. A preliminary solubility test of the NFT test article was performed in autoclaved ultrapure water and DMSO. The NFT test article was observed to be insoluble in autoclaved ultrapure water at 20 mg/mL and 10 mg/mL but was observed to be soluble in DMSO at 200 mg/mL. This 200 mg/mL DMSO stock was serially diluted to obtain 100, 50, 25, 12.5, and 6.25 mg/mL intermediate stocks, which were each spiked into cell culture media and observed for solubility and precipitation. The 1000 and 2000 μg/mL test concentrations resulted in high precipitation followed by moderate-to-high precipitation at 500 μg/mL and slight precipitation at 250 μg/mL. No precipitation was observed at 125 μg/mL in the culture medium; hence, the 250 μg/mL concentration with slight precipitation was selected as the highest test concentration for the in vitro mammalian cell micronucleus test. The pH and osmolality of the NFT treatments at the tested concentrations of 250 μg/mL and below were found to be suitable for testing in the lymphocyte cultures.
A dose range–finding cytotoxicity test was first conducted in blood lymphocyte cultures to select the appropriate dose levels for the in vitro micronucleus test. Cytotoxicity of >60% was observed at test concentrations of 250 µg/mL in the 24 h treatment. Therefore, results from this assessment indicated that NFT levels up to 250 µg/mL in the short-term exposures (4 h in the absence and presence of S9) and up to 83.33 µg/mL in the long-term exposure (24 h in the absence of S9) were appropriate for use in the micronuclei evaluation test.
In vitro Mammalian cell micronucleus test in human peripheral blood lymphocytes treated with NFT.

Note. Human Peripheral Blood Lymphocyte cultures exposed to N-trans-feruloyltyramine (NFT) in absence and presence of rat metabolic activation system (S9), were added with cytokinesis blocking agent Cytochalasin B (
***_ p < 0.0001, CBPI = (No. of mono nucleated cells) + (2 × No. of binucleated cells) + (3 × No. of multinucleated cells)/Total No. of cells, % Cytotoxicity = 100 - [100{(CBPIT - 1)/(CBPIC - 1)}].
aLowest insoluble concentration.
The frequency of binucleated cells with micronuclei (%MNBN) following exposure to NFT in the 4-h short-term exposures in the absence and presence of S9 and 24-h longer-term exposure in the absence of S9 were not significantly different from those in the vehicle control (4-h short-term exposure without S9, 0.15, 0.15, 0.15%; 4-h short-term exposure with S9, 0.05, 0.15, 0.10%; 24-h longer-term exposure without S9, 0.10, 0.20, 0.25%) at the three NFT concentrations respectively (Table 3). The micronuclei observed in the blood cultures treated with vehicle and positive controls were within the 95% control limits of the historical control data, and the positive controls induced significant increases in micronuclei frequency relative to the vehicle control.
Therefore, it was concluded that the NFT test article did not induce a significant increase in micronuclei frequency in cultured human peripheral blood lymphocytes. Hence, NFT is considered non-clastogenic and non-aneugenic at the tested concentrations in the absence and presence of metabolic activation.
90-day dietary toxicology study in rats
The 90-day oral toxicity study in rats was conducted in accordance with OECD Guidelines for Testing of Chemicals, No. 408 under GLP. 31 Sprague Dawley rats (n = 10/sex/group) were provided powdered feed containing 0 (G1, control), 5000 (G2, low dose), 10,000 (G3, mid dose), or 20,000 (G4, high dose) ppm of NFT for 90 consecutive days, corresponding to an intake of 0, 357, 728, or 1474 mg NFT/kg body weight/day, respectively, for males and 0, 490, 987, or 1958 mg NFT/kg body weight/day, respectively, for females. Doses were selected based on the non-adverse changes to body weight and body weight gain observed at mid and high doses in the 14-day repeated dose toxicity study.
All animals were observed once daily for clinical signs of toxicity. Mortality and morbidity were not observed in vehicle control and NFT treatment groups throughout the experimental period in both the sexes. There were also no clinical signs of disease or injury throughout the study attributed to NFT treatment. No treatment related adverse effects were observed in weekly body weight and body weight change in males when compared with the concurrent vehicle control group. Significant decreases in body weight gain of females (G3, NFT 10,000 ppm, G4, NFT 20,000 ppm) were observed in week 12 and week 13 compared to the vehicle control group (G1, NFT 0 ppm) (Figure 2). This decrease was found to be non-dose dependent, less than 15%, and body weights were found to be comparable to the vehicle control group with no correlated change in feed consumption (Figure 2). Summary of body weight (g) in males and females.
Feed consumption was not affected by NFT administration in groups of both sexes during the experimental period and was comparable to the respective vehicle control group (Figure 3(a) and (b)). Significant increases in feed consumption were observed in week 5 in G2, G3, G4 and decreases in week 7, 11, and 12 of G4 male group. Significant decreases in feed consumption were observed in week 10 (G3, NFT 10,000 ppm; G4, NFT 20,000 ppm), 11 (G3, NFT 10,000 ppm), & 12 (G3, NFT 10,000 ppm) females compared to their respective vehicle control groups. The changes in feed consumption were found to be either inconsistent or non-dose dependent and were considered incidental. (a) Summary of feed consumption (g/animal/day) in males. (b) Summary of feed consumption (g/animal/day) in females.
No NFT-related ophthalmological abnormalities were observed in the animals of high dose group of both the sexes (G4, NFT 20,000 ppm) examined compared to the respective vehicle control group.
Functional observation battery of tests performed showed no treatment related adverse effects in the high dose group of both the sexes and were comparable with the concurrent vehicle control group (G1, NFT 0 ppm).
Clinical pathology
Hematology and coagulation
Hematology data from rats treated with NFT.

ANEU: absolute neutrophil; ALYM: absolute lymphocyte; AMON: absolute monocyte; AEOS: absolute eosinophil; ABASO: absolute basophil; ARETIC: absolute reticulocyte; RBC: red blood cell count; HGB: hemoglobin; HCT: hematocrit; MCV: mean corpuscular (cell) volume; MCH: mean corpuscular (cell) hemoglobin; MCHC: mean corpuscular (cell) hemoglobin concentration; PLT: platelet count; WBC: white blood cell count.
*– p < 0.05.
Clinical chemistry
Clinical chemistry data of rats treated with NFT.

CRE: creatinine; CHOL: total cholesterol; TGL: triglycerides; HDL: high density lipoprotein; LDL: low density lipoprotein; GLUC: glucose; Cl−: chloride; TP: total protein; ALB: albumin; GLOB (calc): globulin (calculated); PHOS: phosphorus; Ca: calcium; Na+: sodium; K+: Potassium; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; GGT: Gamma-glutamyl transferase; TBI: total bilirubin; BUN: blood urea nitrogen.
*– p < 0.05.
Significant increases in HDL and LDL in high dose (G4, NFT 20,000 ppm) females were observed compared to respective vehicle control (Table 5). The changes were observed in single sex and no correlated histopathological changes were observed (Supplemental Figure S1). The changes in HDL and LDL of rats were found to be not toxicologically relevant to humans considering the differences in the lipid profile.
Hormone analysis
Thyroid hormone measurements in male and female rats treated with NFT.

Urinalysis
Urinalysis of rats administered with NFT.

UVOL: urine volume; SG: specific gravity; URO: urobilinogen.
Necropsy, absolute organ weights, and histopathology
Post necropsy absolute organ weights of rats treated with NFT.

Histopathology examinations of male rats administered with NFT.

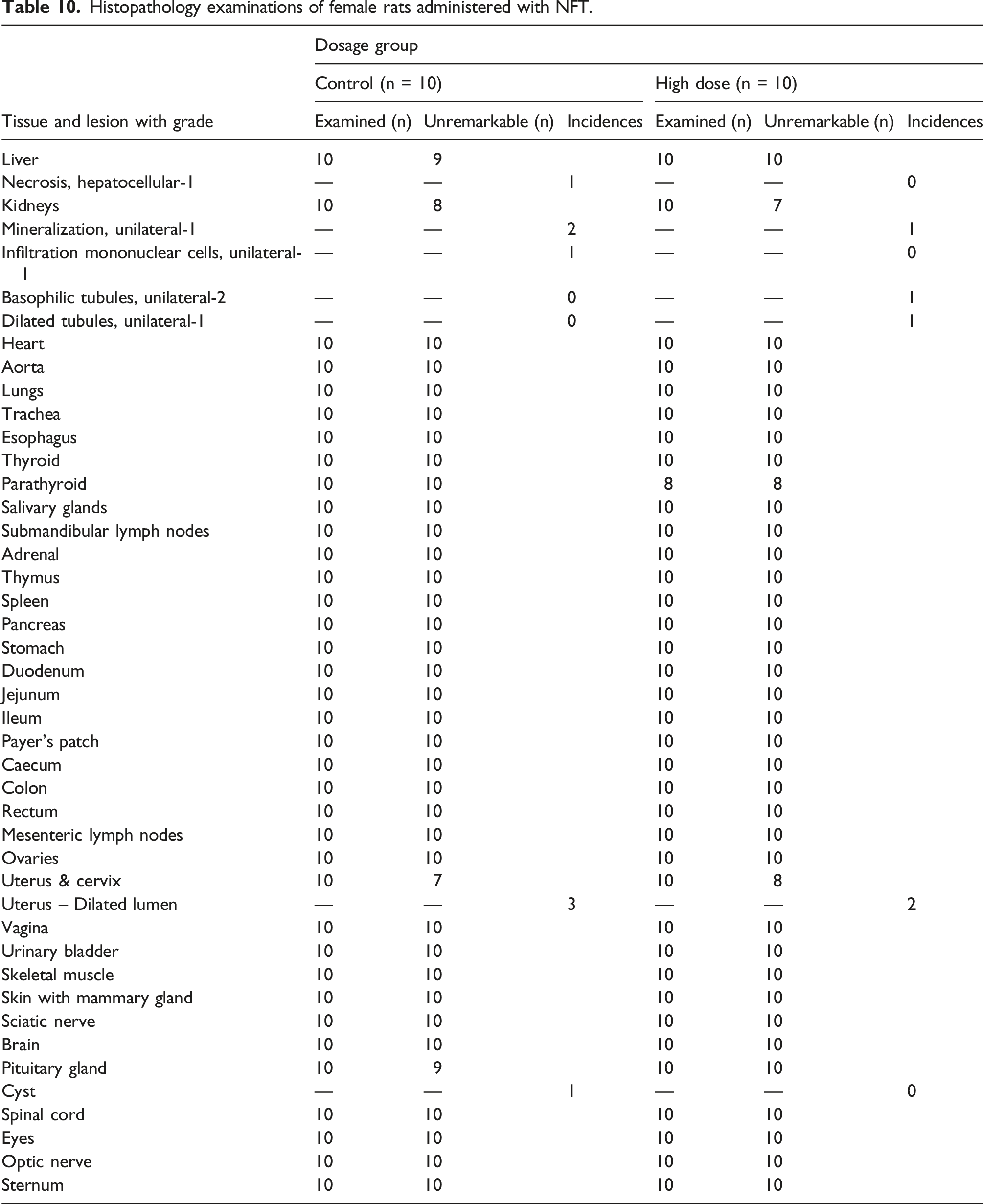
Histopathology examinations of female rats administered with NFT.
Discussion
Naturally occurring bioactive compounds are well recognized for their health promoting properties, 36 and NFT has gained recent attention for its involvement in cellular signaling modulation in several different organs. NFT was originally isolated from eggplant roots and commonly found in a wide range of plant crops. This bioactive compound therefore has an extensive history of safe consumption as a component of plants regularly consumed in the background diet of humans. NFT has been identified and studied using various plant extracts, such as Stephania hispidula, 23 Tinospora tuberculata, 24 and Smilax aristolochifolia. 25 However, large scale extraction of NFT from these plants can be resource-intensive and dependent on the seasonal availability of plants with potential variability in yield and purity. To address these challenges, a high purity NFT production method was developed using precision fermentation of Y. lipolytica followed by purification via downstream processing. Although NFT is generally safe when consumed as part of a natural diet, further research is needed to establish its safety profile in concentrated forms for its use in functional foods or dietary supplements.
Therefore, this present study was directed towards understanding the safety of NFT derived via fermentation with a genetically engineered strain Y. lipolytica 3599_7. The safety of Y. lipolytica has been well-established from its natural occurrence in foods such as cheese, other dairy products, and meats. 37 The use of Y. lipolytica in the production of food enzymes, high-intensity sweeteners, and an eicosapentaenoic acid–rich triglyceride oil has also been documented in a series of GRAS notices (GRNs) notified to the U.S. FDA (GRNs 355, 382, 632, 759, 882, and 940).38–43 In the European Union (EU), Y. lipolytica has Qualified Presumption of Safety (QPS) status for the production of food additives and enzymes on the basis that it is a commonly occurring species in many habitats and environments, with a widely reported presence in dairy and meat products. 44 Its QPS status has been renewed annually. 45 EFSA has published several scientific opinions related to novel food ingredients produced from Y. lipolytica, each indicating an absence of safety concerns.46–48 Furthermore, Y. lipolytica is included in the International Dairy Federation (IDF) inventory of microbial food cultures with demonstrable safety in fermented food products. 49
A series of toxicological studies were conducted to evaluate the potential genotoxicity and systemic toxicity of NFT produced from fermentation with Y. lipolytica (strain 3599_7). The findings from these studies provide significant insights into the safety of NFT obtained through precision fermentation for use in a variety of conventional food and beverage products.
The Ames test, a standard assay for evaluating mutagenic potential, revealed that NFT did not induce mutagenic responses in Salmonella typhimurium or Escherichia coli strains across a range of concentrations, both in the presence and absence of metabolic activation. The absence of a dose-related increase in revertant colony counts and the alignment with historical control values affirm the non-mutagenic nature of NFT. These results were consistent with a recently published study that utilized NFT enriched black pepper extract as a test substance, which was also non-mutagenic in the Ames test. 17 The in vitro mammalian cell micronucleus test demonstrated that NFT does not exhibit clastogenic or aneugenic effects under the tested conditions. The frequencies of binucleated cells with micronuclei were consistent with vehicle controls and within the historical control range, further supporting the genotoxic safety of NFT.
The in vivo (subchronic) repeated dose 90-day oral dietary toxicity study was conducted to determine if NFT administration causes systemic toxicity and other adverse effects in adult Sprague-Dawley rats. NFT doses of 0 (G1, control), 5000 (G2, low dose), 10,000 (G3, mid dose),and 20,000 (G4, high dose) ppm were selected based on the non-adverse changes to body weight and body weight gain observed at mid and high doses in the repeated dose 14-day oral dietary toxicity study. Furthermore, the choice of the doses in the experiment was designed to support the safety of an intended daily human intake of 120 mg/day NFT (∼1.7 mg/kg/day) for a 70 kg individual. 50
The 90-day oral toxicity study in Sprague Dawley rats revealed no treatment-related mortality or significant clinical signs of toxicity. Minor variations in body weight and feed consumption observed in the high-dose groups were less than 10% and did not translate into adverse clinical outcomes, suggesting that these effects are not of toxicological significance.
Hematological and clinical chemistry evaluations identified statistically significant changes in specific parameters; however, these findings were isolated, lacked dose-dependency, and were not supported by histopathological correlates. For instance, alterations in parameters such as hemoglobin, and glucose levels were observed, but their sporadic nature and absence of corroborative findings minimize their toxicological relevance.
Specifically, a significant increase in ALT (Alanine amino transferase) was observed in the high-dose males (33%) compared to the vehicle control group. However, the changes observed were found to be marginal, observed in only males, with no changes in the correlated hepatocellular marker like AST (Aspartate amino transferase) and no correlated microscopic changes in the liver (Table 5). Hence the observed changes were considered to be non-treatment related. Similar significant increases were observed in HDL (High density lipoprotein) and LDL (Low density lipoprotein) levels in the high dose (G4, NFT 20,000 ppm) group compared to the respective vehicle control. The changes were observed in female rats only and no correlated histopathological changes were observed. The changes in HDL and LDL of rats were found not to be toxicologically relevant to humans considering the differences in the lipid profile. It is understood that rats have lower levels of cholesteryl ester transfer protein than humans, an enzyme crucial for transferring cholesterol between lipoproteins, which further impacts the distribution of cholesterol between LDL and HDL. 51 A significant decrease in glucose levels (Table 5) was observed in the high-dose males (11.5%) compared to the vehicle control group. The changes observed were found to be marginal, did not correlate with any change in parameters such as triglycerides or changes on microscopic examination of the pancreas and liver. Hence the changes were considered as not due to treatment related.
Necropsy and histopathological examinations further confirmed the absence of treatment-related adverse effects. While occasional changes in organ weights (e.g., spleen, brain) were noted, these lacked a consistent dose-response relationship and were not accompanied by histopathological evidence of toxicity. The overall absence of pathological lesions in major organs supports the conclusion that NFT does not induce significant systemic toxicity at the tested dose levels. These results were also consistent with the 90-day subchronic toxicity study conducted with NFT containing black pepper extract, 17 which showed that administration of a test substance containing NFT caused no adverse effects in rats after similar examinations.
The findings of this study align with previous research and most importantly, this study extends the understanding of NFT’s safety profile, addressing a critical gap in the literature by focusing on its toxicological evaluation. In the absence of treatment-related adverse effects, the No Observed Adverse Effect Level (NOAEL) of NFT was determined to be 1474 mg/kg body weight/day for males and 1958 mg/kg body weight/day for females based on the feed consumption and subsequent test article intake (corresponding to the highest dietary concentration evaluated: 20,000 ppm).
In conclusion, the results of this study indicate that NFT obtained through precision fermentation is safe and suggest its tolerability up to 1474 and 1958 mg/kg body weight for daily administration of 90 days in male and female rats, respectively. The findings from present investigations also revealed that NFT is unlikely to cause any genotoxic effects.
Supplemental Material
Supplemental Material - The safety assessment of N-trans-feruloyltyramine using In vitro genotoxicity studies and 90-day toxicity study in rats
Supplemental Material for The safety assessment of N-trans-feruloyltyramine using In vitro genotoxicity studies and 90-day toxicity study in rats by Sungwon Lee, Srinivas Seekallu, Suresh Babu Venkataramaiah, Chandrashekar Mataguru Doreswamy, Mohan Cheluru Umesh, Sandeep Malleshappa, Sajeev Justin Dev, Ganadhal Puttaramaiah Chethankumara, Nagaraju Lohith, Gajanan Rajpal Deshmukh, Brian Premkumar and Brinda Mahadevan in Human & Experimental Toxicology
Footnotes
Acknowledgments
All aspects of cell engineering, along with the initial fermentation and downstream processes, at bench and pilot scale, were developed by Cellibre, Inc., based in San Diego, California.
Statements and declarations
Author contributions
All the authors contributed to the conceptualization, methodology, investigation and review of the manuscript, while the majority of the writing, review and editing was performed by SL, BP and BM.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Brightseed sponsored the study. SL is an employee of Brightseed. BM has worked as a consultant with Brightseed Inc., under a cost reimbursable contract for this project.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
