Abstract
Connexins form the gap junctional channels that mediate cell-to-cell communication, and also form hemichannels present at the plasma membrane. Hemichannels are permeable to small hydrophilic compounds, including molecules involved in autocrine and paracrine signaling. An abnormal hemichannel opening causes or contributes to cell damage in common human disorders (e.g., cardiac infarct, cerebrovascular accidents, deafness, skin diseases, and cataracts) and is therefore a potential pharmacological target. The discovery of useful hemichannels inhibitors has been hampered in part by the lack of suitable high-throughput functional assays. Here, we developed and characterized an assay useful to assess the function of hemichannels formed by human connexins expressed in a genetically modified Escherichia coli strain. The LB2003 cells, devoid of three key K+ uptake transport mechanisms, cannot grow in low-[K+] medium, but expression of Cx26, Cx43, or Cx46 rescues their growth defect (growth complementation). We developed a protocol for a simple, inexpensive, easily scalable, reproducible, and sensitive assay that should be useful for the discovery of new and better hemichannel inhibitors based on the analysis of small-compound libraries.
Introduction
There are 21 human connexin isoforms that belong to the family of gap junction–forming channels. 1 Six connexin subunits oligomerize to form a hemichannel (HC), and two HCs, one from each of two adjacent cells, dock head to head to form a gap junction channel (GJC) 1 . HCs and GJCs are permeable to small hydrophilic molecules. 1 HCs are involved in physiological processes such as autocrine and paracrine signaling, providing a pathway for efflux from cells of molecules such as ATP, NAD+, glutamate, and prostaglandins.2-4 HCs are also important in the pathophysiology of important disorders (e.g., a sustained opening of HCs has been implicated in the cell damage that results from hypoxia),5-7 and are therefore important pharmacological targets.
Most available inhibitors of connexin HCs are neither specific nor isoform selective.8,9 This is in part due to the absence of a simple functional assay that can be scaled up for the discovery of novel HC inhibitors by high-throughput screening (HTS). A few assays have been developed for HTS of GJC function.10-12 These assays, however, were designed to assess GJCs, and lack the ability to screen for HC function.10-12 A number of HC assays are available, including well-established dye uptake and electrophysiology assays.1,13 These assays are very useful to study HC properties, but are not readily amenable to screen large chemical libraries due to their complexity and cost.
We recently reported that human connexin 26 (Cx26) HCs can be functionally expressed in Escherichia coli and made a preliminary report of a novel growth complementation assay using the E. coli strain LB2003 as host. 14 LB2003 cells are devoid of three K+ uptake systems, Kdp, Kup, and Trk, and as a result cannot grow in low-[K+] medium.14-16 The inability of LB2003 to grow in low-[K+] medium can be rescued by supplementing the medium with [K+] or by transforming the cells with K+-selective ion channels (see Fig. 1A ).15,17,18 In our brief report, we showed that expression of human Cx26 can also complement the lost-growth phenotype in low-[K+] medium. 14 Here, we evaluated the HC E. coli–based functional assay in detail and show that it can be applied to three different connexin isoforms (Cx26, Cx43, and Cx46) and has the potential for HTS.
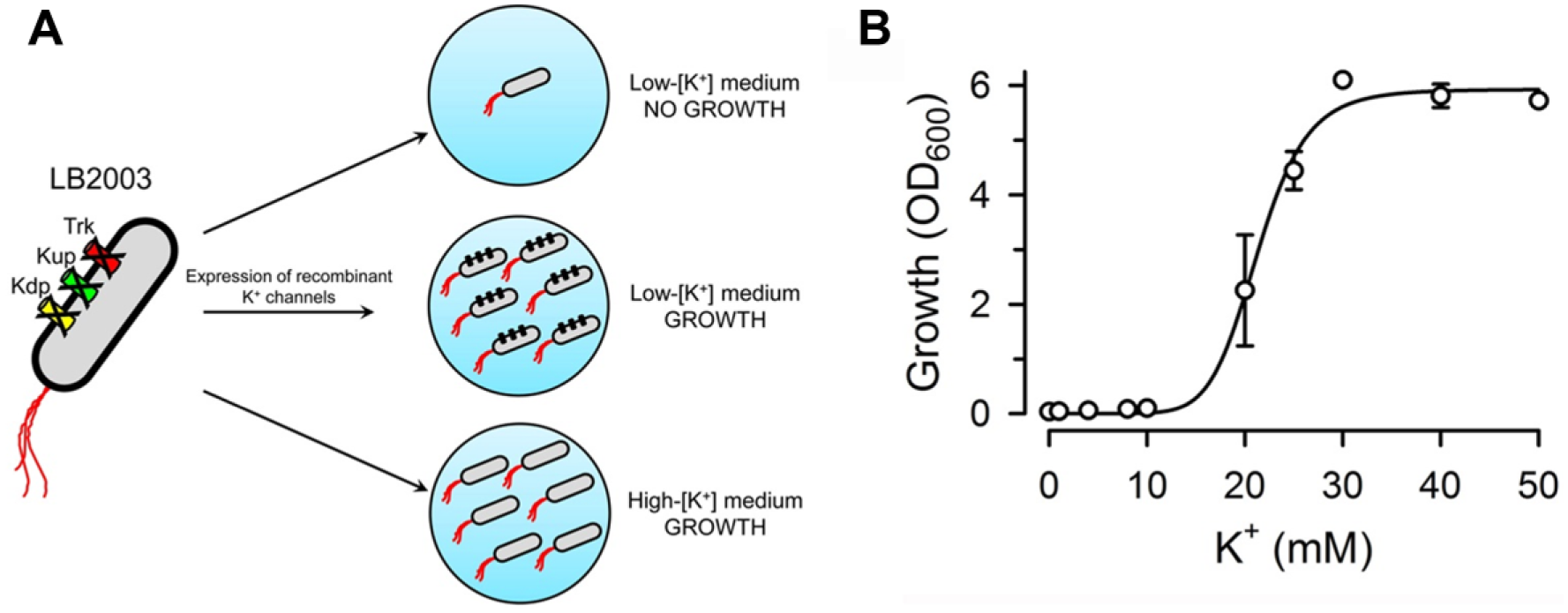
Properties of the host cells used for the growth complementation assay. (
Materials and Methods
Molecular Biology
The human Cx26, Cx43, and Cx46 DNAs were synthesized by GenScript (Piscataway, NJ). The experiments were carried out in accordance with the Texas Tech University Health Sciences Center (TTUHSC)–approved guidelines, and the experimental protocols were approved by the TTUHSC Recombinant DNA Biosafety Committee. The plasmid for expression of Cx26 was described recently. 14 Basically, we used a partially optimized DNA coding for wild-type human Cx26 fused to a poly-His tag (six His) at the C-terminal end. The tag was preceded by a TEV protease cleavage sequence. This DNA, subcloned into the NcoI/HindIII sites of pQE60 (Qiagen), was used for all the experiments shown here, and is referred to as Cx26 (pQE-Cx26 plasmid; see Supplementary Information). The pQE-Cx26 plasmid was used to drive expression of Cx26, from which we purified active HCs. 14 The G45E and D50N mutants were generated from pQE-Cx26 by site-directed mutagenesis. The human Cx43 and Cx46 DNAs with a C-terminal poly-His tag were also cloned into the NcoI/HindIII sites of pQE60. The human Cx43 and Cx46 DNAs used were optimized for E. coli expression (see Supplementary Information), but pilot results also showed complementation with the original Cx43 human sequence. As positive control for the growth complementation assay, we used the plasmid pJ404 containing the DNA of a Methanocaldococcus janaschii voltage-gated potassium channel (MVP) with a poly-His tag (Mj0139-PJ404). 19
Bacterial Strain
The strain of E. coli LB2003, used as host for the growth complementation studies, was generously provided by Dr. E. P. Bakker (Osnabrük University, Germany). LB2003 cells are devoid of three key K+ transporters (Δtrk, Δkup, andΔkdp strain).15,17 They were transformed with pREP4, a plasmid that confers kanamycin resistance, and were stored as a 25% glycerol stock at −80 °C.
Growth Complementation Assay
Freshly prepared competent LB2003 containing the pREP4 plasmid were transformed with Mj0139-PJ404 (plasmid carrying the coding sequence of the hyperpolarization-activated K+ channel MVP), pQE60 (empty plasmid [EP]), pQE-Cx26 (human Cx26 DNA into pQE60), or pQE-Cx43 (human Cx43 into pQE60). These cells were grown overnight in Luria-Bertani (LB) medium (Becton Dickinson, Franklin Lakes, NJ) supplemented with 100 mM KCl and 0.4 mg/mL ampicillin (pQE-60 confers resistance to ampicillin). The cells were grown overnight at 37 °C with shaking at 250 rpm, and were harvested at ~16 h. Cell pellets were washed four times in Na+ liquid medium (NLM) to remove any residual potassium from the LB medium, and diluted with the desired growth medium to an optical density measured at 600 nm (OD600) of 0.2. For the growth assays, we used two defined media, NLM and K+ liquid medium (KLM). NLM contained 46 mM Na2HPO4, 23 mM NaH2PO4, 8 mM (NH4)2SO4, 0.4 mM MgSO4, 0.012 mM FeSO4, 1 mM sodium citrate, 44 mM glucose, and 0.006 mM thiamine hydrochloride, with 0.4 mg/mL ampicillin, pH 7.0. KLM was identical to NLM except for the equimolar replacement of Na+ with K+. For growth complementation by Cx26 and Cx43 in low-[K+] medium, we used NLM + 4 mM KCl and NLM + 8 mM KCl, respectively. These [K+] values gave the best assay signal-to-noise ratios. LB2003 cells transformed with EP and MVP were used as negative and positive controls, respectively. Expression of Cx26 and MVP was induced with 0.5 mM isopropyl-β-D-thiogalactopyranoside (IPTG) at time zero (the time of dilution to OD600 = 0.2). We found that growth complementation by Cx43 expression worked better after induction with 10 µM IPTG. Transformed cells in the appropriate growth medium were seeded in 24-well plates (931565-G-1X, Thompson Scientific Co., Oceanside, CA) with 3 mL of sample per well, and the plates were incubated at 30 °C, with shaking at 250 rpm. The initial OD600 = 0.2 was subtracted for growth calculations. For the experiments with 96-well plates (Falcon 353072, Corning Inc., Corning, NY), the conditions were similar, except that a volume of 200 µL/well was used, and OD600 was measured on a plate reader (Synergy 4, BioTek, Winooski, VT). In all cases, we used 84 wells for experiments, with the remaining wells filled with culture medium alone as an internal control of path length variability. KLM was used to assess the toxicity of HC inhibitors in the host cells transformed with pREP4.
Assessment of Bacterial K+ Content
LB2003 cells transformed with PQE60 or pQE-Cx26 were grown under the conditions of the complementation assay described above (medium with 4 mM [K+]) for 18 h. Then, the cells were pelleted, resuspended to an OD600 of 1.3 in ice-cold NLM, and pelleted by centrifugation at 4 °C in preweighted tubes. After determination of the wet weight, the pellets were resuspended in 20 mL of a buffer containing 0.5 mg/mL lysozyme in 150 mM NaCl and 20 mM Tris/HCl, pH 7.5. After 1 h incubation at room temperature with gentle rotation, lysis was completed on a homogenizer (EmulsiFlex-C3, Avestin, Ottawa, ON). The lysate was centrifuged at 4 °C for 1 h at 100,000g, and [K+] in the supernatant was measured with a K+-selective electrode.
Western Blots
Recombinant protein expression was assessed from Western blots of cell lysates (MVP) or membranes (Cx26 and Cx43) using antibodies against the His tag (anti-Hexa-His, GenScript). Cx26 purified from insect cells 14 was used as a control to assess levels of expression of Cx26 and Cx43 in bacteria. An Odyssey Infrared Imager (Li-Cor Biosciences, Lincoln, NE) was used for the detection of the secondary antibody goat-anti-mouse Alexa Fluor 680 (Life Technologies, Grand Island, NY).
Statistics
Data shown are means ± SEM or SD, as indicated. Statistically significant differences were calculated by the Student t test for unpaired data or one-way analysis of variance (ANOVA), as appropriate. The Z′ factor was calculated to assess the potential for scalability of the Cx26 complementation assay for high throughput. The Z′ factor can take into account the day-to-day and well-to-well variability of the sample (e.g., Cx26) and control (e.g., EP) and is frequently used to test the quality of the assay.
20
Z′ was calculated as Z′ = 1 − (3σs + 3σc)/|µs – µc|, where σs and σc are the standard deviations and µ
Results
Growth Complementation by Cx26 and Cx43
It is well established that LB2003 cells do not grow in media with low [K+],15-17,21 but before assessing growth complementation we needed to determine the [K+] growth dependence in NLM supplemented with K+. NLM is a nominally K+-free defined medium. Figure 1B illustrates such dependence, and shows that there is essentially no growth at [K+] values of 10 mM or lower, with 50% of the maximal growth at 21 mM [K+].
We recently demonstrated growth complementation in LB2003 cells by expression of Cx26. 14 Here, we aimed at optimizing the protocol and determining whether the growth complementation assay also works for other connexin isoforms. For the Cx26 studies, we found that NLM supplemented with 4 mM K+ and 0.5 mM IPTG worked very well because under these conditions, LB2003 cells that were not transformed (growth relative to Cx26 = 0.06 ± 0.02) or were transformed with the EP did not grow ( Fig. 2A , open circles). Figure 2A shows the time dependence of the growth measured at 30 °C in cells expressing Cx26 (filled circles). Cx26 and Cx43 are among the most dissimilar connexins, 1 and accordingly, we needed to optimize the Cx43 assay. Early studies showed erratic results when the IPTG concentration used for induction was the same as that used for the Cx26 growth complementation (0.5 mM). Analyses of experiments at different IPTG concentrations indicated that the assay worked better using 10 µM IPTG. Although we did not study the reason for the reduced growth at higher IPTG concentrations, it is conceivable that high expression of Cx43 HCs (nonselective large channels) with higher IPTG concentrations can have toxic effects that result in reduced cell growth. Figure 2B shows the time course of the growth complementation by Cx43 in NLM supplemented with 8 mM K+ and 10 µM IPTG. As was the case for Cx26, growth reached a plateau at ~18 h ( Fig. 2B , closed circles). Since one of our aims was to simplify the assay for eventual scaling up to an HTS format, based on the data in Figure 2A , B , we decided on 18 h incubation for the single-point assay. The 18 h data in Figure 2C show growth complementation by Cx26 and the hyperpolarization-activated K+ channel MVP (the positive control). The growth in cells transformed with pQE60 (EP; negative control) was minimal, if any. The Western blots showing anti-His antibody reactive bands ( Fig. 2D ) confirmed the expression of MVP, Cx26, and Cx43, as expected, and consistent with our previous report on human Cx26 expressed in bacteria. 14 The major Cx26 and Cx43 bands ran at the expected sizes for the monomers (oligomers are also evident, especially for Cx26; pointed at by arrows), whereas the positive control MVP ran as a monomer and tetramer. 19 Expression of Cx43 was higher than that of Cx26, which is consistent with the increased growth complementation ( Fig. 2B vs. Fig. 2A ). There were no immunoreactive MVP, Cx26, and Cx43 bands in the samples of LB2003 cells transformed with EP ( Fig. 2D ).
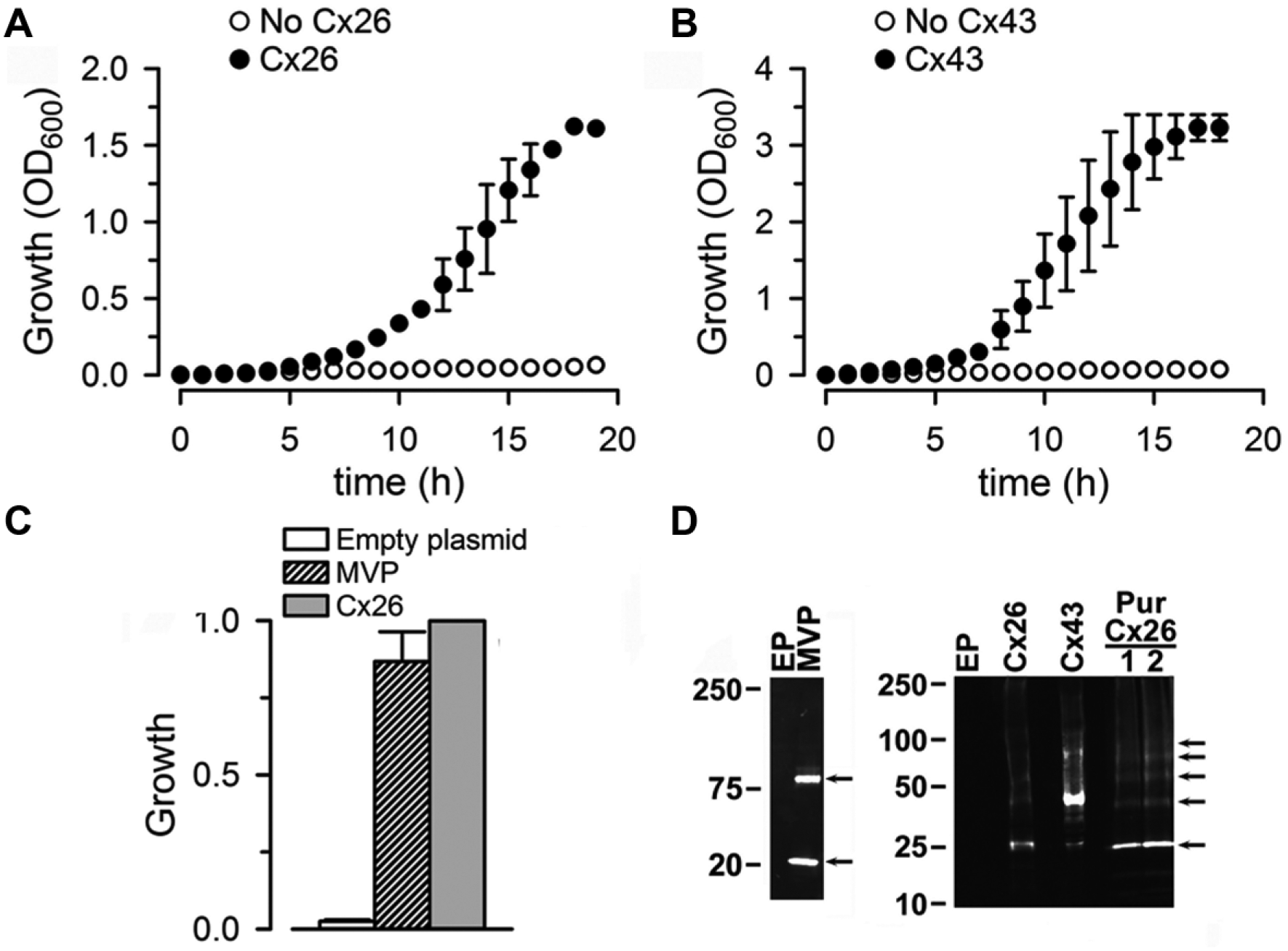
Growth complementation by connexins. (
From the results in this section, we decided on the following conditions for the Cx26-mediated growth complementation assay: single measurement 18 h after the cells were grown at 30 °C in NLM supplemented with 4 mM KCl, 0.4 mg/mL ampicillin, and 0.5 mM IPTG, with shaking at 250 rpm. For the Cx43 assay, the conditions were similar, except that [KCl] was 8 mM and IPTG was 10 µM. Although we did not optimize the assay for Cx46, LB2003 cells transformed with Cx46 DNA grew after 18 h to an OD600 of 1.18 ± 0.06 (n = 3; p < 0.001 vs. cells transformed with the EP). These results suggest that the basic protocol can be easily adapted to probe HCs formed by different connexin isoforms.
The growth complementation of connexin-expressing LB2003 cells grown in low-[K+] medium is likely the result of K+ influx through HCs. This seems to indeed be the case because the K+ content of LB2003 cells expressing Cx26 was higher than that of cells transformed with the EP ( Fig. 3 ). Assuming an intracellular water volume of 0.45 mL/g wet weight, 22 the increased K+ content corresponds to a difference in intracellular [K+] of 26 ± 3 mM.
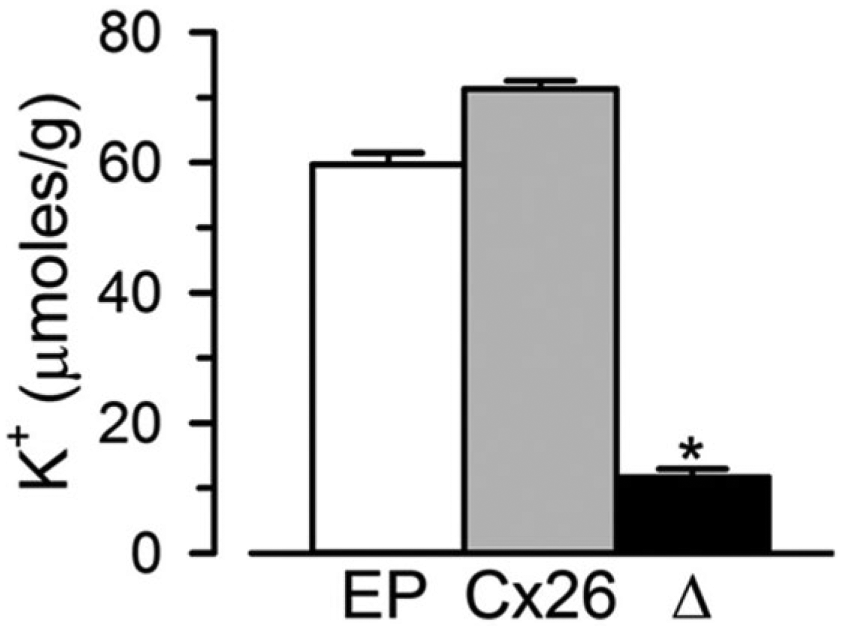
Increased K+ content in LB2003 cells expressing Cx26. LB2003 cells grown at 30°C in NLM + 4 mM K+ and 0.5 mM IPTG were harvested, resuspended in NLM, and lysed. The [K+] in the lysate was measured with a K+-selective electrode, and the data are presented as K+ content per gram of wet weight (mean ± SEM, n = 3 independent experiments). EP, cells transformed with the empty pQE60 plasmid; Cx26, cells transformed with pQE-Cx26; Δ, difference between Cx26 and EP. The difference between the Cx26 and EP values was statistically significant (p < 0.01).
Effects of HC Inhibitors and Cx26 Mutations on the Growth Complementation Elicited by Connexin Expression in LB2003 Cells
The studies in the previous section strongly suggest that the growth in low-[K+] medium of LB2003 cells expressing connexins is the result of K+ influx through HCs. To strengthen this notion, we tested the effects of known HC inhibitors and Cx26 mutations that display abnormal regulation by Ca2+. Figure 4A shows that the HC inhibitors 2-aminoethoxydiphenyl borate (2-APB) and 1-octanol and divalent cations commonly used to inhibit connexin HCs 23 reduced the grow complementation elicited by Cx26. At the concentrations used (see legend to Fig. 4A ), none of the inhibitors displayed toxicity, as demonstrated by the absence of an appreciable effect in the growth of LB2003 cells in high-[K+] medium. Growth as a percentage of the control value in the absence of 2-APB, octanol, or divalent cation addition was: 50 µM 2-APB = 92 ± 1%, 1 mM 1-octanol = 96 ± 1%, 1 mM Ca2+ = 100 ± 1%, 20 mM Mg2+ = 104 ± 1%, and 100 µM Zn2+ = 99 ± 1% (n = 3 independent experiments for each condition).
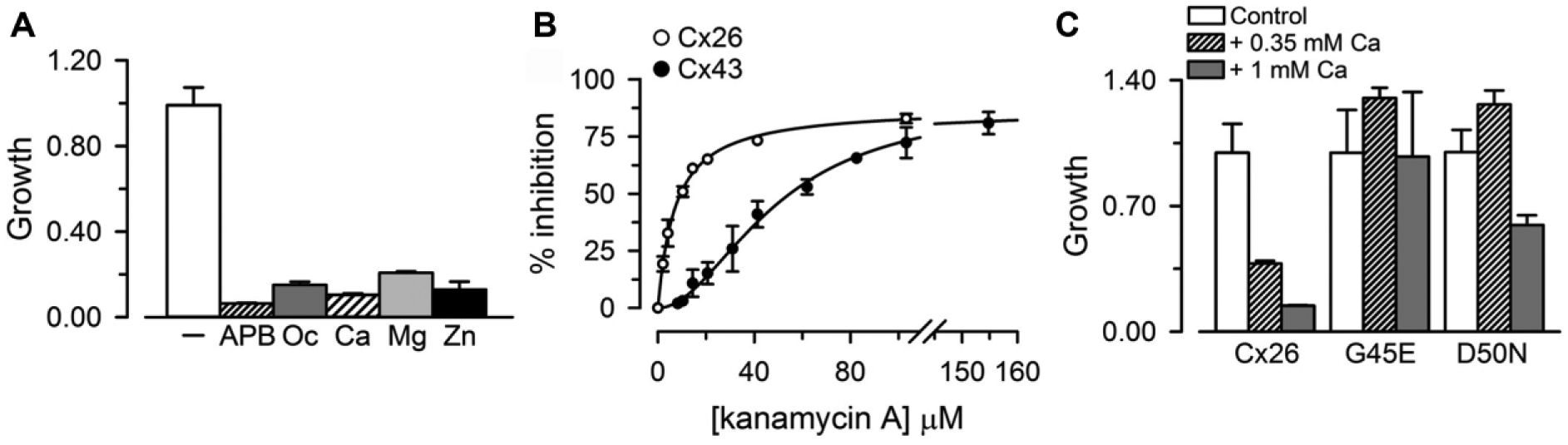
Effects of connexin HC inhibitors on Cx26-dependent growth complementation. (
The LB2003 cells that we used are kanamycin resistant because of their transformation with the pREP4 plasmid (see Materials and Methods); kanamycin A had no effect on LB2003 cells grown in KLM; growth in 150 µM kanamycin A was 105 ± 3% of the value in the absence of the aminoglycoside (n = 3; no significant difference). Since it has been recently shown that aminoglycosides inhibit connexin HCs,24,25 we tested for inhibition of growth complementation by Cx26 and Cx43 by kanamycin A. As expected for an effect mediated by HCs, kanamycin A inhibited growth complementation by Cx26 and Cx43 ( Fig. 4B ). The calculated IC50 for cells expressing Cx26 was 7.2 ± 0.4 µM (Hill coefficient of 1.0 ± 0.2), and for cells expressing Cx43 was 49 ± 3 µM (Hill coefficient of 1.9 ± 0.2). Kanamycin A (150 µM) had no effect on MVP-dependent growth complementation; growth in cells expressing MVP was 105 ± 3% of the value in the absence of the aminoglycoside (n = 3; no significant difference).
Since in most experiments we used human Cx26 followed by a His tag preceded by a cleavage sequence for the TEV protease (catalytic domain of the nuclear inclusion a protein of the tobacco etch virus), we performed control experiments using a plasmid with the Cx26 sequence without any tags. Transformation with this plasmid produced growth complementation equivalent to that by our tagged Cx26, with an IC50 for inhibition by kanamycin A of 7.2 ± 2.2 µM, indistinguishable from that in cells expressing our tagged Cx26 (see above). Overall, these results demonstrate that our assay can be used to screen for HC inhibitors, and that it has sufficient sensitivity and reproducibility for concentration–response studies.
We also studied the growth complementation by two Cx26 mutants, G45E and D50N. These mutants display low sensitivity to Ca2+ inhibition.26-28 Addition of 0.35 mM or 1 mM CaCl2 to the nominally Ca2+-free medium in cells that express Cx26 reduced growth complementation by ~60% and 90%, respectively ( Fig. 4C ). In cells expressing the G45E mutant the effect of Ca2+ was absent ( Fig. 4C ), whereas in those expressing the D50N mutant, 0.35 mM Ca2+ had no effect and 1 mM Ca2+ reduced growth complementation by only ~40% ( Fig. 4C ). In summary, mutations known to form functional HCs with low response to extracellular Ca2+ produced growth complementation with low sensitivity to Ca2+; that is, the mutants elicit growth complementation with the response to Ca2+ that is expected from their known properties in eukaryotic cells.26-28
Growth Complementation Assay for HTS
As mentioned in the introduction, there are no HC assays that are easily amenable to HTS scaling. Since our assay has a low cost and is simple and robust, we performed a basic analysis to determine its potential for HTS. We studied three conditions: (1) LB2003 cells transformed with EP as negative control (no growth), (2) LB2003 cells transformed with pQE-Cx26 (Cx26; growth complementation by Cx26), and (3) same as the previous condition, but in the presence of 10 µM kanamycin A (Cx26 + kanamycin A; ~50% inhibition of growth complementation by Cx26). For each condition, we run a full plate (24 wells) simultaneously, three separate days ( Fig. 5A ). The Z′ factor is a commonly used statistical parameter to assess the quality of an HTS assay. 20 It is a dimensionless parameter with a value between –∞ and ≤ 1 that takes into account the data variation of the sample (Cx26) and background (EP). An assay with Z′ ≥ 0.5 is considered excellent, values between 0 and 0.5 point to a marginal assay that requires optimization, and assays with Z′ < 0 are not suitable for HTS. 20 With a Z′ of 0.8, our assay has great potential for HTS. The Z′ value calculated using the Cx26 + kanamycin A sample (~50% inhibition) was 0.6, also an excellent value. It is also important to note that DMSO, a common solvent of small-compound libraries, did not inhibit growth complementation by Cx26. In fact, at the highest concentrations tested, DMSO increased the signal-to-background ratio of the HC-dependent complementation, suggesting that addition of DMSO to our basic assay medium may have a beneficial effect on the assay performance. The growth relative to Cx26 was 1.00 ± 0.02, 1.10 ± 0.03, 1.30 ± 0.04, and 1.68 ± 0.07 in the presence of 0.1%, 0.5%, 1%, and 2% DMSO, respectively (n = 3 independent experiments for each condition). This lack of inhibition by DMSO should allow for testing of high concentrations of compounds from small-compound libraries. These results support the use of our assay for HTS and also indicate that the assay can easily detect compounds that elicit partial HC inhibition.
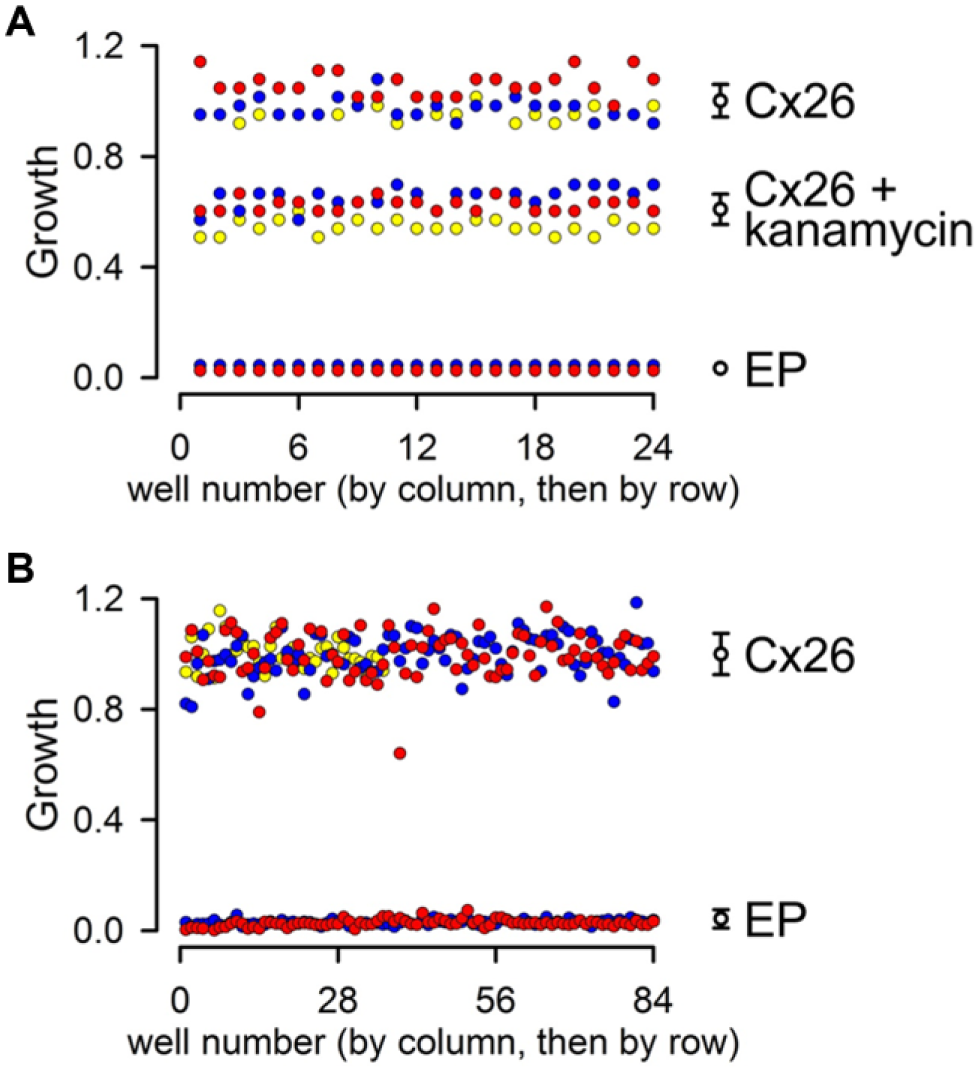
Evaluation of assay performance. (
To test for scalability of the assay, we determine the Z′ factor from experiments in 96-well plates ( Fig. 5B ). In two experiments, one plate contained LB2003 cells transformed with EP and the other plate cells transformed with pQE-Cx26, whereas in a third experiment, half of the plate contained LB2003 cells transformed with EP and the other half cells transformed with pQE-Cx26. The calculated Z′ factor from the three independent experiments was 0.75, strongly suggesting easy scalability of the assay.
Discussion
Abnormal opening of connexin HCs is implicated in many disorders, which makes HCs an attractive target for therapy. “Leaky” HCs have been proposed or demonstrated to have a role in the pathophysiology of disorders of the heart (e.g., cardiac infarction and arrhythmias), nervous system (e.g., cerebrovascular accidents, oculodentodigital dysplasia, and Charcot–Marie–Tooth disease), skin (e.g., forms of keratitis, keratodermia, and ectodermal dysplasia), and the eye (cataracts). 7 Currently, dye uptake and electrophysiology are the most commonly used methodologies to assess connexin HC function.1,13 These methodologies pose barriers for HTS because of their complexity and scaling-up cost. The assay presented here is simple, inexpensive, and robust, and has high potential as an HTS platform.
LB2003 is an E. coli strain with deletion of three K+ uptake systems (Kdp, Kup, and Trk).14,15 As a result, LB2003 cells do not grow in low-[K+] medium, but grow in high-[K+] medium, or in low-[K+] medium if the cells are transformed for expression of recombinant K+-selective channels (growth complementation).15,17 K+ is necessary for E. coli growth because of its involvement in maintaining turgor pressure, activation of enzymes, and intracellular pH regulation. Our previous data strongly suggested that human HCs expressed in LB2003 cells are able to complement cell growth, as the K+-selective channels do. 14 This was expected because they provide a pathway for K+ influx, although it was not obvious because these “large” and poorly selective channels can also have deleterious effects on the cells (e.g., depolarization and alterations in metabolite homeostasis). Because of the presence of a favorable electrochemical driving force and the permeability of HCs to K+, it is reasonable to assume that growth complementation is the result of K+ influx. This notion is supported by the increased K+ content of Cx26-expressing LB2003 cells grown in 4 mM [K+] compared with cells that do not express Cx26.
The results presented here confirmed our previous observation of growth complementation by expression of Cx26, and added the following: (1) Demonstration of growth complementation by Cx43 and Cx46. This is a very important addition because Cx26 and Cx43/Cx46 are some of the most dissimilar connexin isoforms. 1 Therefore, it seems likely that our growth complementation assay can be used to assess the function of HCs formed by most or all connexin isoforms. (2) Demonstration that the connexin-dependent growth complementation is the result of the expression of functional HCs. Two lines of evidence support this conclusion. The first line is the effects of known HC inhibitors. Growth complementation was sensitive to known HC inhibitors: the poorly selective divalent cations (they also affect the function of many other proteins), 9 2-APB and 1-octanol, 23 and the more selective aminoglycosides.24,25 The second line of evidence is the alterations in growth complementation by Cx26 mutants. Cx26 mutants that display specific regulatory alterations show growth complementation that has the corresponding properties. The Cx26 mutants G45E and D50N form functional HCs that display low Ca2+ sensitivity.26-28 The expression of G45E and D50N in LB2003 confers growth complementation, as expected from the formation of functional HCs by these mutants,26-28 but its sensitivity to Ca2+ was altered. In cells expressing wild-type Cx26, the decrease in growth complementation was ~60% and ~90% in response to 0.35 and 1 mM CaCl2, respectively, whereas the reduction was absent (LB2003 cells expressing G45E) or reduced (absent in cells expressing D50N at 0.35 mM Ca2+ and ~40% at 1 mM Ca2+) in cells expressing the mutants.
It is important to keep in mind that the assay presented here is simple, inexpensive, and can be scaled up for HTS, but is not designed to study HC function in detail. The readout of the assay is based on a measurement of a phenomenon (bacteria growth) that depends on HC function, but is not a direct function of it (as opposed to HC currents and solute transport). The functional HCs allow K+ influx, which is required for cell growth, but we do not know the degree of HC inhibition needed to reduce K+ sufficiently to inhibit growth. We expect that our assay will be useful for HTS, but that does not mean that it will not miss potential inhibitors for a variety of reasons, such as limited access to the periplasmic space because of the presence of a cell wall, incompatibility with growth medium, indirect effects that need proteins present in mammalian cells that are absent in bacteria, and HC inhibition that is insufficient to lower intracellular K+ to the level needed to reduce cell growth. The HC function assay presented here can be used as a low- to medium-throughput assay for studies of functional effects of mutations or to analyze the effects of HC inhibitors. It can be used to determine whether specific mutations produce functional HCs or whether compounds inhibit HCs formed by a specific isoform. The new assay complements more complex assays that provide far more detailed mechanistic information, such as permeability assays and electrophysiological studies. A major drive to develop the growth complementation assay was the possibility of having an assay that can be scaled up for HTS. Our analysis clearly shows that the assay has excellent potential in this area. It should be useful for the discovery of new and better HC inhibitors (and probably GJC inhibitors) because of its simplicity, low cost, easy scalability, reproducibility, and sensitivity.
Footnotes
Acknowledgements
We want to thank Dr. Luis Reuss for his critical evaluation of the manuscript and the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health grants R01GM79629, 3R01GM079629-03S1, and R01GM097159; American Heart Association Texas Affiliate Inc. grant 14GRNT18750014; Welch Foundation grant BI-1757; and a TTUHSC Preliminary Data Program grant.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
