Abstract
BCDIN3D is an RNA-methyltransferase that O-methylates the 5′ phosphate of RNA and regulates microRNA maturation. To discover small-molecule inhibitors of BCDIN3D, a suite of biochemical assays was developed. A radiometric methyltransferase assay and fluorescence polarization–based S-adenosylmethionine and RNA displacement assays are described. In addition, differential scanning fluorimetry and surface plasmon resonance were used to characterize binding. These assays provide a comprehensive package for the development of small-molecule modulators of BCDIN3D activity.
Introduction
Noncoding microRNAs (miRNAs) are a potent cellular mechanism to regulate gene expression. These RNA elements are produced by processing of pri/pre-miRNAs by the Drosha and Dicer nucleases to active ~22 nucleotide miRNAs, which are subsequently loaded into the RNA-induced silencing complex (RISC). This multiprotein complex uses the miRNA as a selection mechanism to specifically target complementary mRNAs for degradation. The details of these mechanisms have been reviewed thoroughly in the literature.1–4
Aberrations in miRNA levels have been implicated in a variety of disease states.5–9 For example, expression of miR-145 is decreased in several human cancers, including breast,10,11 prostate, 12 thyroid, 13 oral squamous cell, 14 and colorectal adenocarcinoma.15,16 The biological half-lives of miRNAs vary, 17 as the intracellular concentrations of mature miRNAs are generally controlled at both the expression and degradation levels. Cellular levels of miR-145 are controlled by the P53 and phosphoinositide 3-kinase/Akt pathways, 18 which regulate pri-miRNA-145 expression, the Drosher/Dicer pathway that produces the mature miR-145, 19 and the miRNA clearance pathway. 20 A potent regulator of miR-145 maturation is the recently discovered O-methyltransferase BCDIN3D. This enzyme catalyzes the dimethylation of the 5′ monophosphate of the maturing miRNA, thus removing the negative charge required for further processing by Dicer. 19 Genetic depletion of BCDIN3D significantly upregulates the expression of miR-145 and inhibits the invasive potential of the MDA-MB-231 cell line, 19 suggesting that inhibition of BCDIN3D activity may afford therapeutic benefit in certain malignancies.
Pharmacological manipulation of miRNA-dependent pathways is a particularly exciting area of drug discovery that presents a unique profile of challenges. While advances in nucleic acid–based therapeutics are promising, direct delivery of miRNA is not trivial, requiring specialized delivery mechanisms and stabilizers. 21 These challenges have led us and others to adopt a more traditional small molecule–based approach to modulate miRNA levels by targeting enzymes in the miRNA biogenesis or degradation pathways.22–26 To this end, we describe herein a suite of biochemical assays for the discovery and characterization of small-molecule modulators of BCDIN3D methyltransferase activity.
Materials and Methods
Reagents
S-adenosylmethionine (SAM) was purchased from AK scientific (Union City, CA; cat. S018), and S-adenosylhomocysteine (SAH) and sinefungin were purchased from Cayman Chemical (Ann Arbor, Mi; cat. 13603 and 13829, respectively). All other chemicals were purchased from Sigma-Aldrich (St. Louis, MO) or Bioshop (Burlington, Ontario, Canada) or as mentioned elsewhere in the text and were reagent grade or higher. FL-NAH has been previously described. 27 [ 3 H]S-adenosylmethionine (12–18 Ci/mmol) was obtained from PerkinElmer (Waltham, MA).
Cloning and Protein Expression
A DNA fragment encoding human BCDIN3D (1-292) was amplified by PCR and subcloned into pFBOH-MHL (Addgene plasmid 62304; Addgene, Cambridge, MA, USA). The resulting construct was transformed into DH10Bac Competent E. coli (Invitrogen, Carlsbad, CA, USA), and a recombinant viral DNA bacmid was purified, followed by recombinant baculovirus generation in Sf9 insect cells (yield of 3 mg/L). Sf9 cells grown in HyQ SFX insect serum-free medium (Thermo Scientific, Waltham, MA) were infected with 10 mL of P3 viral stock per 0.8 L of suspension cell culture and incubated at 27 °C using a platform shaker set at 100 rpm. The cells were collected 72 h postinfection, when viability dropped to 70% to 80%.
Harvested cells were resuspended in 20 mM Tris-HCl (pH 7.5) containing 500 mM NaCl, 5 mM imidazole and 5% glycerol, 1× protease inhibitor cocktail (2.5 µg/mL each of aprotinin, leupeptin, pepstatin A, and E-64), or Roche cOmplete EDTA-free protease inhibitor cocktail tablet (Roche Applied Science, Penzberg, Germany). The cells were lysed chemically by rotating 30 min with NP40 (final concentration of 0.6%) in the presence of benzonase nuclease followed by sonication at a frequency of 7 (10 s on and 10 s off) for 4 min (Sonicator 3000; Misoni, Farmingdale, NY, USA). The crude extract was clarified by high-speed centrifugation (60 min at 36,000 g at 4 °C).
The lysate was then loaded onto a Hispur Ni-NTA resin (Thermo Scientific). The column was washed and eluted with 20 mM Tris-HCl (pH 7.5), 500 mM NaCl, and 5% glycerol, containing 30 mM and 250 mM imidazole, respectively. The protein was further purified by gel filtration on a Superdex 200 (26 × 60) using an ÄKTA FPLC (GE Healthcare, Little Chalfont, UK) equilibrated with 20 mM Tris-HCl (pH 8.0), 250 mM NaCl, 5% glycerol, and 1 mM dithiothreitol (DTT). Protein purity was confirmed via sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and the pure fractions were pooled, concentrated, and flash frozen. For SAM competition assay, the protein was washed on a column with 3.6 L of buffer (20 mM Tris, 500 mM NaCl, pH 7.5) at 3 mL/min monitoring A260/280. Our competition data showed that a significant portion of the protein is capable of binding to the probe, consistent with the SAM binding site being available.
Radiometric Methyltransferase Assay
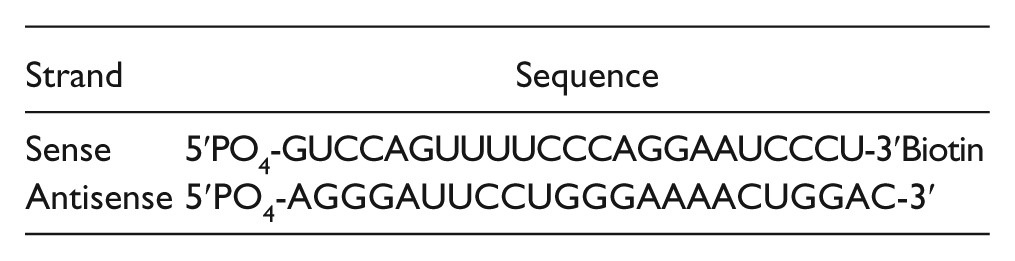
The methyltransferase activity of BCDIN3D was measured using a radiometric assay that detects transfer of a [ 3 H]methyl from the radiolabeled cofactor S-[Methyl- 3 H]-adenosyl-L-methionine (PerkinElmer; cat. NET155001MC) to a biotinylated 23-bp double-stranded RNA (dsRNA) substrate. Reactions were performed in a buffer consisting of 20 mM Tris (pH 8.0), 10 mM DTT, 0.01% Triton X-100, and 0.4 U/µL RNaseOUT (Life Technologies, Carlsbad, CA). The biotinylated double-stranded miR-145 substrate was synthesized by IDT (Coralville, IA). The sequences are as follows:
Reactions (10 µL) were allowed to proceed in standard 96- or 384-well polypropylene plates and were quenched by the addition of excess 7.5 M guanidinium hydrochloride (GuHCl). For kinetic characterization, samples were spotted on SAM2 Biotin Capture Membranes (Promega, Madison, WI), washed with 2 M NaCl (2× 5 minutes) and with water (2× 5 minutes), and allowed to air dry. Incorporated radioactivity was quantitated by liquid scintillation analysis using a Tricarb scintillation counter (PerkinElmer). All kinetic measurements were performed under initial rate conditions using 30 nM BCDIN3D, a saturating concentration of one substrate, and varying concentrations of the other substrate (SAM or biotinylated miR-145).
The optimized radiometric screening assay conditions were as follows: 30 nM BCDIN3D, 200 nM biotinylated miR-145, 3 µM [ 3 H]-SAM in a buffer consisting of 20 mM Tris (pH 8.0), 10 mM DTT, 0.01% Triton X-100, and 0.4 U/µL RNaseOUT. The reaction was allowed to proceed at 37 °C for 60 min before being quenched with an equal volume of 7.5 M GuHCl. The quenched reaction mixture was transferred to 384-well Streptavidin FlashPlates HTS Plus (PerkinElmer; SMP410A001PK) for scintillation counting in a TopCount NXT (Perkin Elmer). Z′ factor (0.7) for performing a radiometric assay in the 384-well format was determined using the optimized condition in the presence (100 µM) and absence of SAH. Mean high and low signal in the presence and absence of SAH was 3050 cpm and 218 cpm, respectively.
Fluorescence Polarization Assay
General
All binding experiments were performed in a total volume of 10 µL binding buffer (20 mM Tris [pH 8.0] supplemented with 0.01% Triton X-100 and 5 mM DTT) in 384-well black polypropylene PCR plates (Axygen, Tewksbury, MA, USA; cat. PCR-384-BK). For RNA-based fluorescence polarization experiments, RNAseOUT (Life Technologies) was added to the assay to a final concentration of 0.4 U/µL. Fluorescence polarization (FP) was measured using a BioTek Synergy 4 (BioTek, Winooski, VT) after 30 min of incubation at ambient temperature. The excitation and emission wavelengths were 485 nm and 528 nm, respectively.
Saturation of FL-NAH and miR-145-FL with BCDIN3D
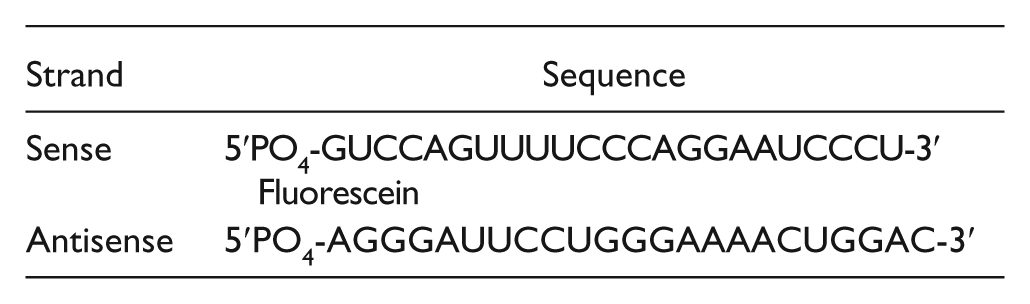
Increasing concentrations of BCDIN3D were incubated with 50 nM of the SAM binding site fluorescent ligand FI-NAH or 40 nM miR-145-FL, and the resulting FP was measured as described above. The miR-145-FL is a synthetic fluorescently labeled double-stranded RNA modified with a 5′ monophosphate (IDT). The miRNA has the following sequence:
The binding affinity Kd and maximal FP signal (Bmax) were calculated using nonlinear least squares regression to a single-site binding model in GraphPad Prism v. 6.05 (GraphPad Software, La Jolla, CA).
Fluorescent ligand displacement experiments
In total, 9 µL of a mixture of BCDIN3D (RNA Site FP: 300 nM, SAM Site FP: 5 µM) and a fluorescent ligand (40 nM FL-miR145 or 50 nM FL-NAH) was added to each well of an assay plate. Then, 1 µL of a competitor dilution or vehicle control was added to every well and mixed thoroughly using a Bravo liquid handler (Agilent, Santa Clara, CA). After a brief centrifugation at 250 g to remove any bubbles, the assay was allowed to incubate at ambient temperature for 30 min before FP analysis. DMSO concentration was held constant (0.5%) across all assay wells for all experiments except the DMSO tolerance tests.
Z′ factor analysis
For the SAM binding site FP assay, BCDIN3D (5 µM) was incubated with 50 nM FI-NAH-1 in the presence or absence of 250 µM SAH for 30 min at ambient temperature, and fluorescence polarization was measured as described above. For the RNA binding site FP assay, BCDIN3D (300 nM) was incubated with 40 nM FL-miR-145 in the presence or absence of 5 µM unlabeled miR-145 for 30 min at ambient temperature, and fluorescence polarization was measured as described above. Z′ values were calculated as previously described. 28
Statistical Analysis and Data Fitting
All data analysis was performed in Prism GraphPad v. 6.05 or Microsoft Excel (Microsoft Corp., Redmond, WA). Kinetic parameters were determined by fitting the experimental data to the standard Michaelis-Menten model using nonlinear least squares regression analysis. IC50 and Kdisp values were calculated using nonlinear least squares regression analysis to a four-parameter concentration-response curve model.
Differential Scanning Fluorimetry
BCDIN3D was diluted to 0.1 mg/mL in 100 mM HEPES, 100 mM NaCl (pH 8.0) in the presence of 5× SYPRO Orange dye (Life Technologies; cat. S-6650), and increasing concentrations of SAM, SAH, or the fungal metabolite sinefungin in a total volume of 20 µL in a white polypropylene 384-well plate (Axygen; cat. UC500). Differential scanning fluorimetry (DSF) was performed in a Light Cycler 480 II (Roche Applied Science) using a 4 °C/min temperature gradient from 20 °C to 95 °C. Data points were collected at 1 °C intervals. DSF data were fitted to a Boltzmann sigmoid function and Tm values were determined as previously described. 29
Surface Plasmon Resonance
Surface plasmon resonance (SPR) studies were performed using a Biacore T200 (GE Health Sciences, Little Chalfont, UK) at 20 °C. Approximately 5700 response units (RU) of BCDIN3D were amino-coupled onto one flow cell of a CM5 chip (according to the manufacturer’s protocol) while another flow cell was left empty for reference subtraction. SAM was dissolved in 12 mM HCl (100 mM stock), and 3-fold serial dilutions were prepared in 12 mM HCl. For SPR analysis, the serially titrated compound was diluted 1:1000 in HBS-EP (20 mM HEPES [pH 7.4], 150 mM NaCl, 3 mM EDTA, 0.05% Tween-20) + 5% DMSO. Kd determination experiments were performed using single-cycle kinetics with an contact time of 60 s, off time of 120 s, and a flow rate of 75 µL/min. Kd calculations were done using Steady State Affinity Fitting and the Biacore T200 Evaluation software (GE Health Sciences). Using SAM to assess the binding competence of the amino-coupled BCDIN3D indicated that the protein was 60% functional for SAM binding.
Results and Discussion
Biochemical Characterization of BCDIN3D Methyltransferase Activity
Recombinant full-length BCDIN3D was expressed in Sf9 cells and purified to apparent homogeneity using standard chromatographic methods as described in the Materials and Methods. The purified protein demonstrated robust in vitro RNA methyltransferase activity using a biotinylated duplex miR-145 miRNA substrate. The assay was further optimized (
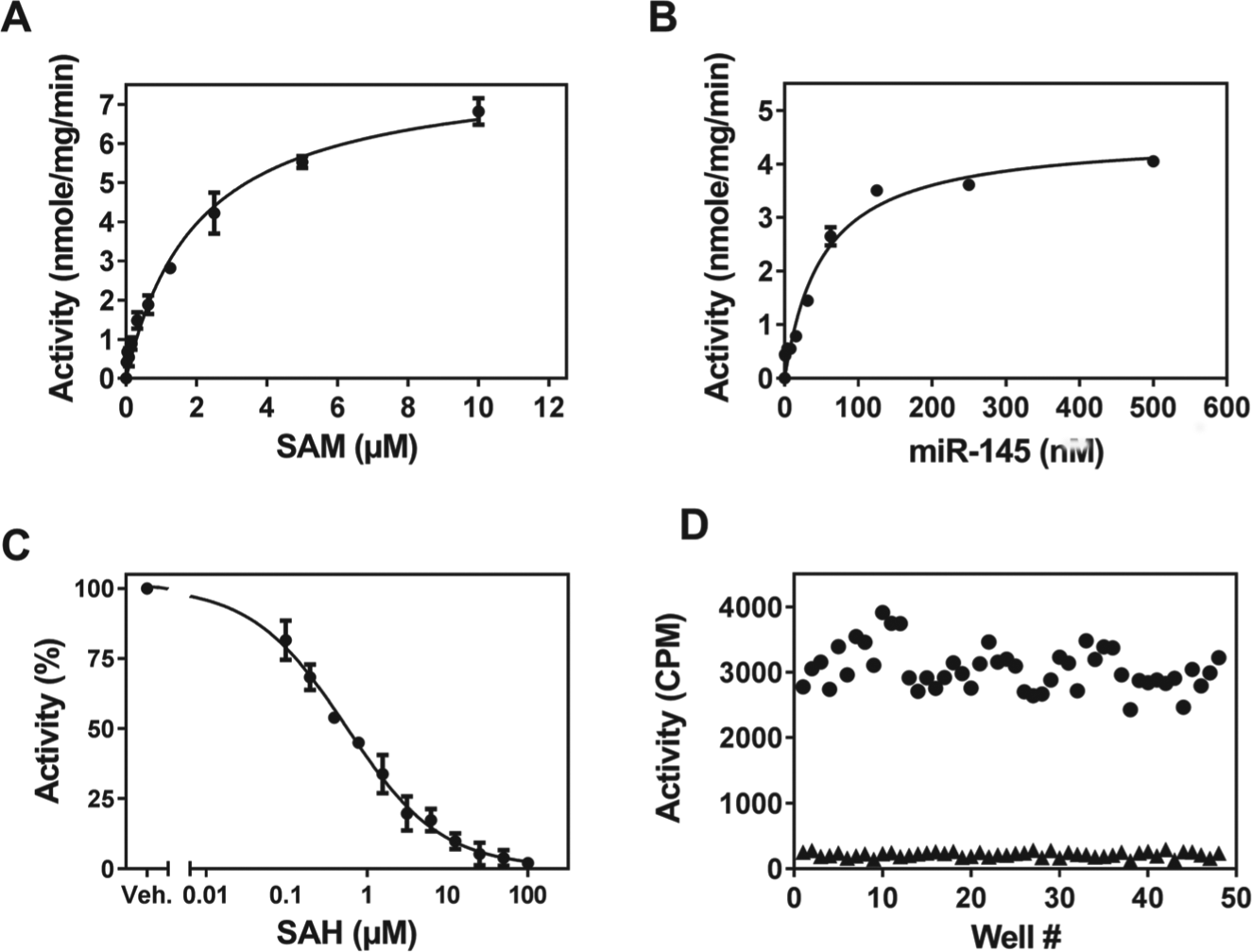
Kinetic parameter determination of BCDIN3D.
To identify small-molecule modulators of BCDIN3D, we modified the radiometric assay to increase throughput and sensitivity to inhibitors. Initially, the assay was performed in a 96-well format using 20 µL of reaction mixture and was miniaturized to a 10-µL reaction volume in a 384-well format and was automated using a Bravo liquid handler (Agilent). The optimized assay was designed under pseudo-balanced conditions, using the
Development of FP Assays for the SAM and RNA Binding Sites of BCDIN3D
We recently reported a novel fluorescein-labeled SAM analogue that binds to the SAM binding site of MLL1.
27
This ligand also binds tightly to the SAM binding site of BCDIN3D in a saturable manner (
Fig. 2A
) with a Kd of 3 ± 0.2 µM and a Bmax of 230 ± 9 mp (
Table 1
). Furthermore, FL-NAH also inhibits BCDIN3D using a radiometric methyltransferase assay with an IC50 value of 30 ± 5 µM, further confirming that it binds at the active site of the enzyme (
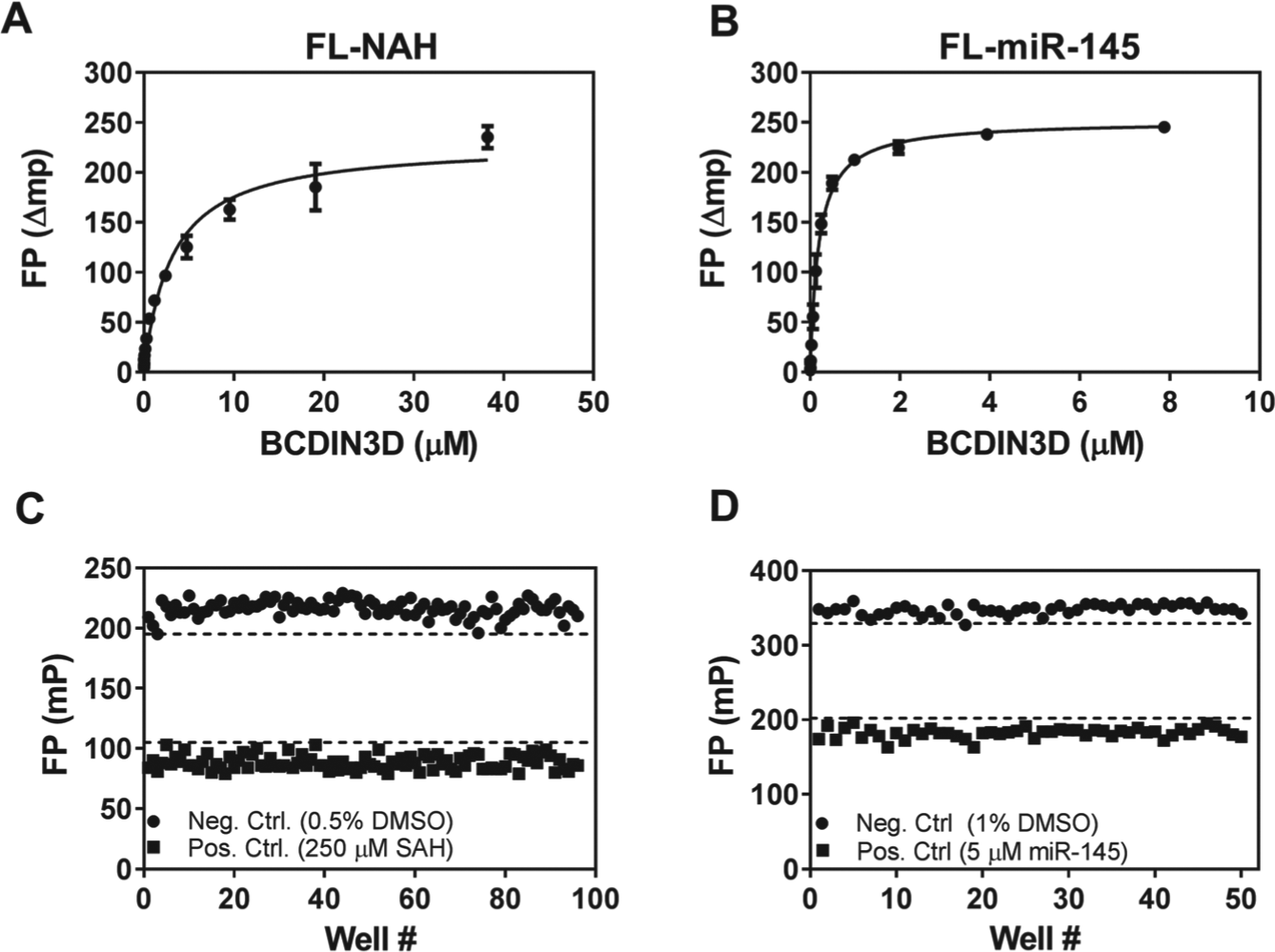
Fluorescence polarization binding assays for BCDIN3D. BCDIN3D induces a saturable increase in fluorescence polarization of (
Fluorescence Polarization–Based Displacement Assays.
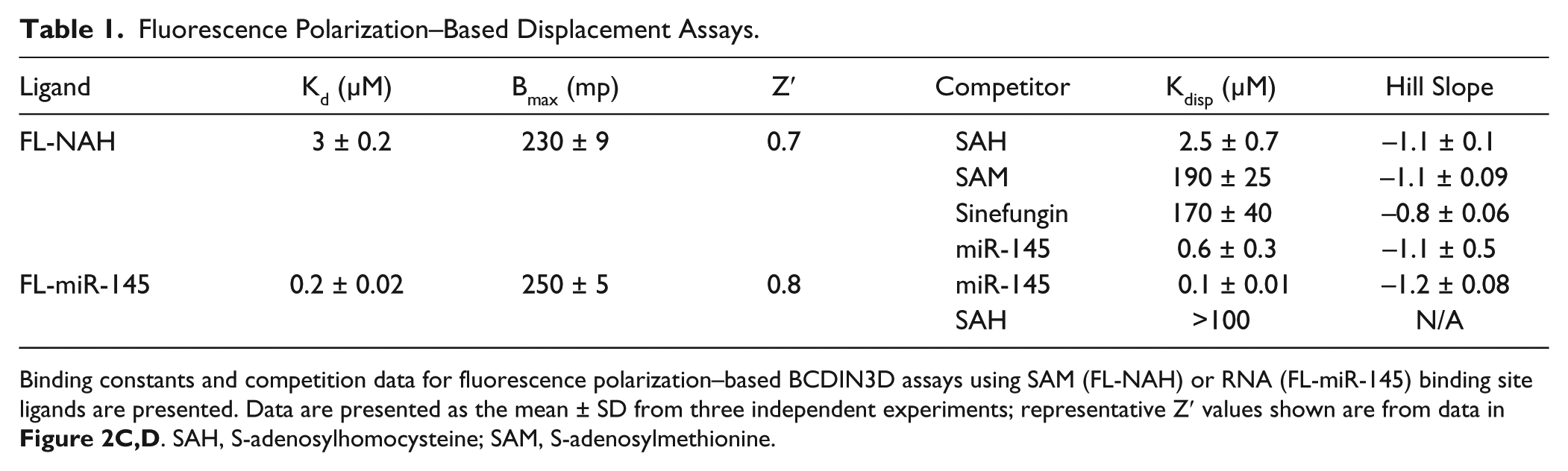
Binding constants and competition data for fluorescence polarization–based BCDIN3D assays using SAM (FL-NAH) or RNA (FL-miR-145) binding site ligands are presented. Data are presented as the mean ± SD from three independent experiments; representative Z′ values shown are from data in Figure 2C , D . SAH, S-adenosylhomocysteine; SAM, S-adenosylmethionine.
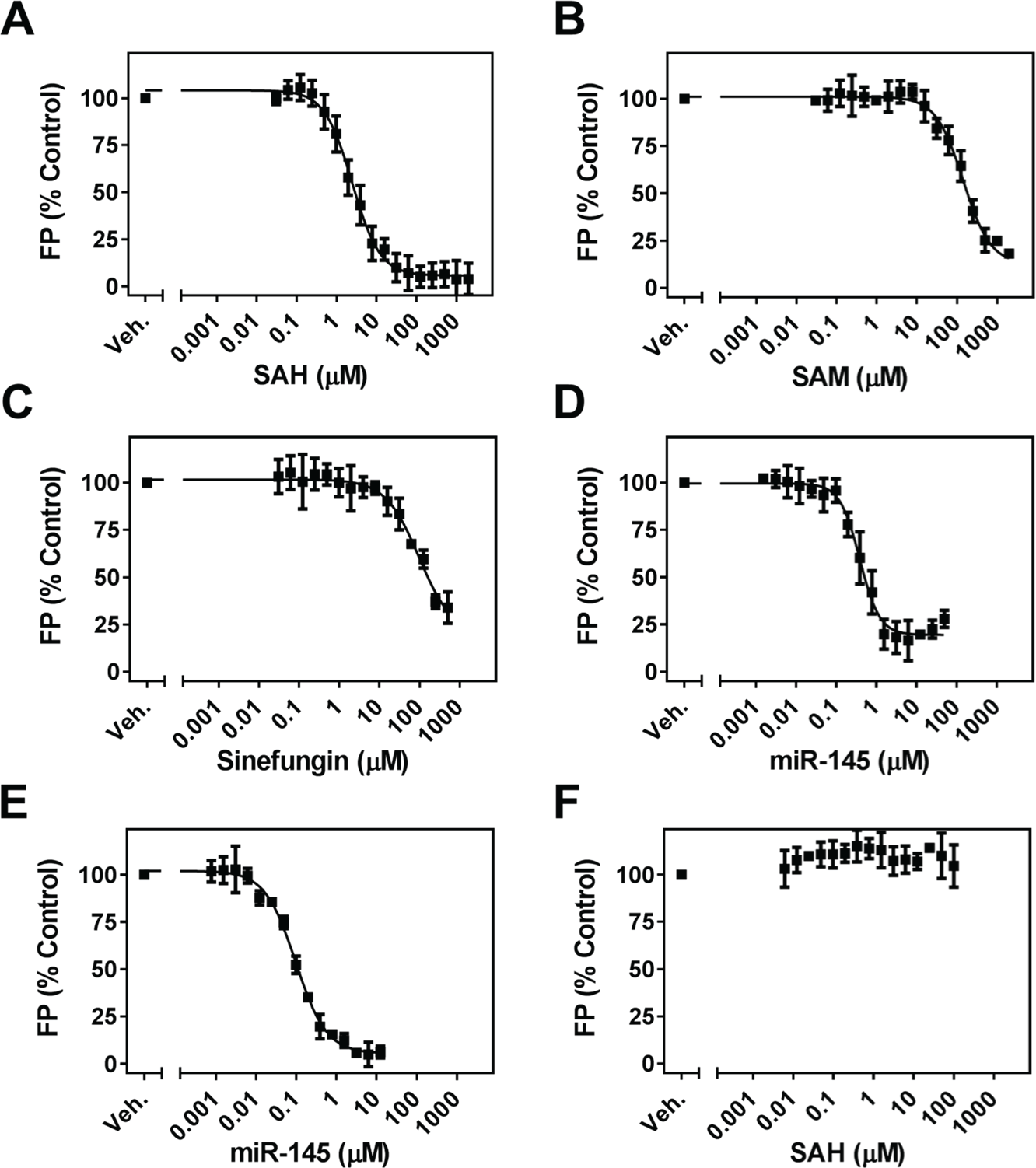
Characterization of FL-NAH and FL-miR-145 displacement. FL-NAH is displaced by (
Thermal Stability Assays for BCDIN3D
Thermal stability assays are a useful method to study protein-ligand interactions. Indeed, this approach has been routinely adapted for compound screening in a 384-well format using either differential scanning fluorimetry or differential static light-scattering methods.29,34 We have optimized a differential scanning fluorimetry assay to monitor the change in thermal stability of BCDIN3D in response to binding of small-molecule ligands. Using this assay, BCDIN3D (0.1 mg/mL) was thermally denatured with a melting temperature (Tm) of 53.6 ± 0.2 °C and was stabilized by SAM, SAH, and sinefungin in a concentration-dependent manner ( Fig. 4 ). SAM (2.5 mM), sinefungin (2.5 mM), and SAH (2.5 mM) stabilized BCDIN3D by 1.9 ± 0.2 °C, 2.4 ± 0.3 °C, and 9.2 ± 0.1 °C, respectively. This assay is suitable for screening to detect ligands regardless of where they bind on BCDIN3D and is also a useful orthogonal method for hit confirmation.
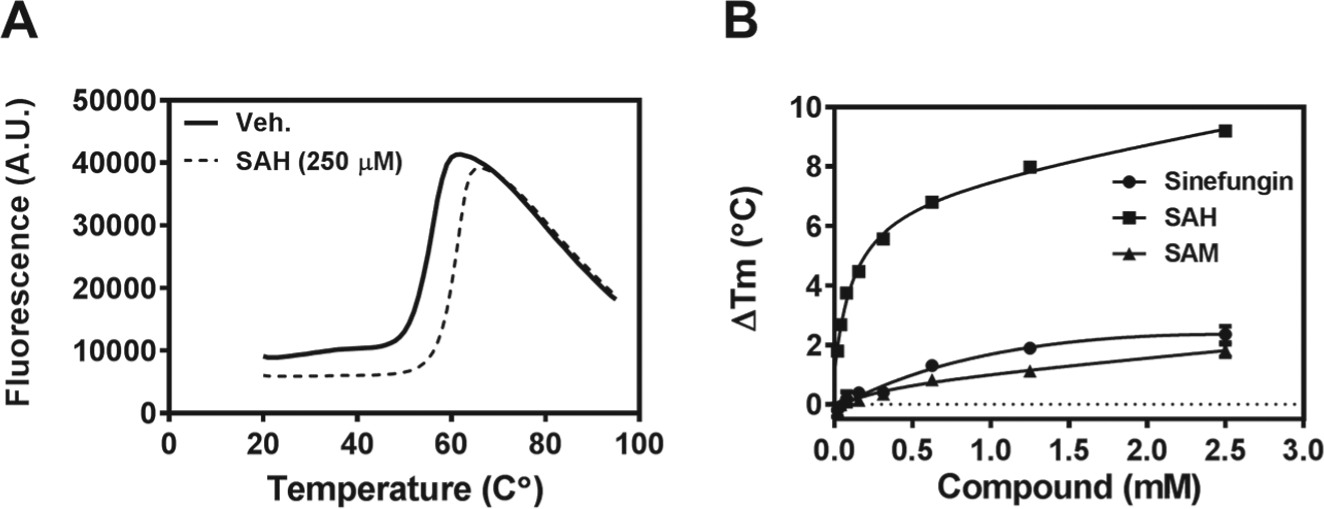
BCDIN3D is thermally stabilized by S-adenosylmethionine (SAM). Thermal stability of BCDIN3D was assessed in the presence and absence of ligands using differential scanning fluorimetry (DSF). (
Use of SPR as an Orthogonal Assay for BCDIN3D
The medium- to high-throughput methods described above provide substantial information regarding the potency and mechanism of action of novel BCDIN3D ligands. However, they do not provide a direct measurement of binding affinity or nuanced information regarding the manner of the binding event (e.g., stoichiometry). To this end, we have optimized a complementary assay for BCDIN3D that directly measures compound binding via SPR. While SPR is a relatively low-throughput screening method, it provides high-quality data suitable for the biochemical characterization of the interaction between BCDIN3D and novel ligands. To validate the method, BCDIN3D was amino-coupled onto a CM5 chip and a serial titration of SAM injected across the protein as described in the Materials and Methods. BCDIN3D shows robust binding to SAM and returns a steady-state affinity (Kd) of 7 ± 0.5 µM ( Fig. 5 ).
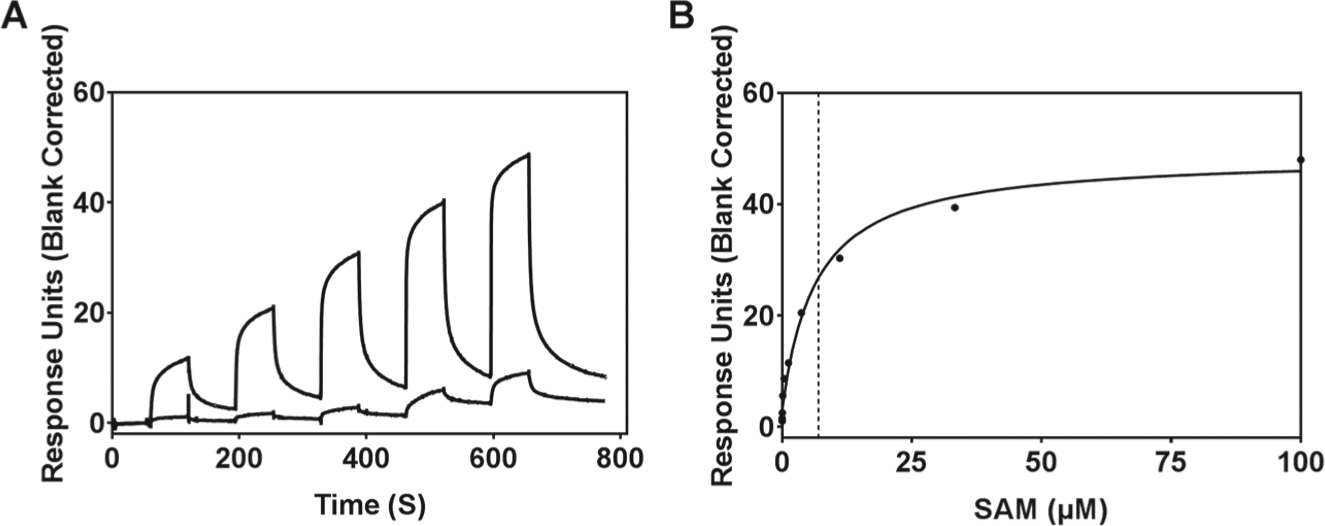
Assessing the binding of S-adenosylmethionine (SAM) to BCDIN3D using surface plasmon resonance (SPR). (
In conclusion, BCDIN3D is a potent regulator of miRNA processing that has been shown to be dysregulated in a variety of malignancies. The physiological role of BCDIN3D is only beginning to be understood. Selective chemical probes that target this methyltransferase would be valuable tools to further elucidate the biological functions and disease relevance of BCDIN3D activity. To this end, we present here a suite of biochemical assays for BCDIN3D activity or substrate/cofactor binding that is suitable for either primary screening or orthogonal confirmation of small-molecule modulators of BCDIN3D activity. During the course of these studies, we have also characterized the kinetic parameters and substrate/cofactor binding constants of BCDIN3D. This panel of assays will accelerate the development of small-molecule modulators of BCDIN3D activity.
Footnotes
Acknowledgements
We thank Alma Seitova for protein expression and Elisa Gibson and Taraneh Hajian for protein purification.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The SGC is a registered charity (number 1097737) that receives funds from AbbVie, Bayer Pharma AG, Boehringer Ingelheim, Canada Foundation for Innovation, Eshelman Institute for Innovation, Genome Canada, Innovative Medicines Initiative (EU/EFPIA), Janssen, Merck & Co., Novartis Pharma AG, Ontario Ministry of Economic Development and Innovation, Pfizer, São Paulo Research Foundation-FAPESP, Takeda, and Wellcome Trust. YGZ was supported by a National Institutes of Health grant R01GM086717.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
