Abstract
AMP-activated protein kinase (AMPK), a conserved heterotrimeric kinase, serves as an energy sensor maintaining energy balance at both cellular and whole-body levels and plays multiple beneficial roles in carbohydrate and lipid metabolism, which makes AMPK an attractive target for diabetes and other metabolic disorders. To date, establishment of the physiologically relevant biochemical assay for AMPK has not been reported. Here we developed a phosphorylated AMPK protection assay based on a time-resolved fluorescence resonance energy transfer (TR-FRET) assay, using the protein phosphatase 2A (PP2A) to dephosphorylate AMPK. The partially dephosphorylated AMPK by PP2A had lower activity than phosphorylated AMPK. This specific TR-FRET assay for AMPK was optimized in the 384-well format and produced similar EC50 values for AMPK activators AMP and A769662 and a similar IC50 value for AMPK inhibitor compound C, as previously reported. Under the optimized conditions, the assay Z′ factor calculated over 160 data points has an optimal value greater than 0.5, which is suitable for high-throughput screening. In conclusion, this phosphorylated AMPK protection assay we developed is very robust, sensitive, and simple to perform and may be useful as a high-throughput assay for identifying AMPK activators with the ability of preventing activated AMPK against dephosphorylation by phosphatase in the physiological conditions.
Introduction
Type 2 diabetes has become a major and growing health risk recent years. Insulin resistance is a characteristic of most of type 2 diabetes, which is associated with abnormality of glucose and lipid metabolism. 1 AMP-activated protein kinase (AMPK) serves as a key energy sensor and regulator, playing multiple beneficial roles in glucose and lipid metabolism. 2 Allosteric activation of AMPK is of interest as a feasible way for the treatment of metabolic disorders such as diabetes and obesity.
AMPK is a heterotrimeric serine/threonine kinase comprising a catalytic α subunit and two regulatory β and γ subunits. Each of its subunits exists in two or three isoforms (α1, α2, β1, β2, γ1, γ2, γ3) in mammals with different tissue expression, 3 suggesting multiple heterotrimeric complexes may play different roles in different tissues. Recent studies showed that the AMPK(α1β2γ1) was the predominant isoform in human liver, suggesting AMPK in human hepatocytes may be tissue-specifically activated. 4 AMPK can be activated by stimuli stress that increases the cellular AMP/ATP ratio. Once activated, it switches off ATP-consuming pathways such as fatty acid synthesis and gluconeogenesis and switches on ATP-generating pathways such as fatty acid oxidation and glycolysis. 5 The beneficial effects on glucose and lipid metabolism make AMPK an important pharmacologic target for the treatment of type 2 diabetes, obesity, and other metabolic disorders.
Under physiological conditions, cellular AMPK is directly regulated by activation through phosphorylation of threonine 172 (Thr-172) in the activation loop of the catalytic α subunit by upstream kinases and deactivation through dephosphorylation at this site by phosphatases. It is known that several distinct upstream kinases should be responsible for AMPK activation in vivo by the phosphorylation of Thr-172 in the α subunit, including LKB1 and calcium/calmod- ulin-dependent protein kinase kinase beta (CaMKKβ). 5 The dephosphorylation of AMPK is mediated by protein phosphatases, such as the protein phosphatase 2A (PP2A), which is one of the major phosphatases in eukaryotic cells and has been involved in AMPK deactivation. 6 Recently, Xiao et al. 7 have reported the structural basis for activator 991 binding to human AMPK at a site between the kinase domain and the carbohydrate-binding module, contributing to its allosteric activation and protection against dephosphorylation. 7 It provides the possibility of discovering novel AMPK activators in the assay system under the physiological regulation of AMPK.
Previous studies have found that the beneficial effects of two widely used antidiabetic agents, metformin and thiazolidinediones, and two adipose tissue–derived hormones, leptin and adiponectin, may be mediated, at least in part, via AMPK activation.8–10 However, these agents activate AMPK indirectly. AICAR (5-aminoimidazole-4-carboxamide riboside), a widely used pharmacologic agent for AMPK research in cells and in vivo, can be converted to ZMP, an AMP mimic, and then stimulates AMPK.
5
So AICAR also activates AMPK indirectly, and its cellular effects may not be specific. Abbott Laboratories has developed microarrayed compound screening (µARCS) technology for an AMPK high-throughput screening (HTS) assay and discovered a small-molecule AMPK activator A769662, which directly activates partially purified rat liver AMPK with an EC50 of 0.8 µM.11,12 Although the µARCS AMPK assay can increase screening throughput, it still requires multiple steps, as seen in the traditional filter assay for protein kinases. Merck Research Laboratories has developed the homogeneous time-resolved fluorescence and Alphascreen AMPK assays using both Anti-pS133-CREB antibody and ACC-CREB peptide.
13
Recently, the time-resolved fluorescence resonance energy transfer (TR-FRET) assay has been used as a high-throughput assay for protein kinase, such as the LANCE
For mimicking the physiological conditions and improving the druglike lead rates in HTS, we developed a novel AMPK(α1β1γ1, α1β2γ1) TR-FRET assay for HTS to obtain small-molecule AMPK activators capable of protecting activated AMPK against dephosphorylation induced by phosphatase PP2A. It depends on the phosphorylation level of AMPK, which can be dephosphorylated by AMPK co-incubation with PP2A. The results presented in this study suggest that our developed AMPK TR-FRET assay may facilitate identification of specific AMPK activators with the ability of activating AMPK and also preventing AMPK dephosphorylation from HTS.
Materials and Methods
Materials
The phosphorylated and active AMPK(α1β1γ1) or AMPK(α1β2γ1) were purchased from Carna Biosciences (Kobe, Japan). The LANCE
Development of AMPK TR-FRET Assay in the Absence of PP2A
For optimization of AMPK concentration and incubation time in 384-well plates, an 8-µL reaction system containing 30 nM Cy5-SAMS and 3 µM ATP in the 1× kinase buffer (50 mM HEPES, 1 mM EDTA, 10 mM MgCl2, 0.01% Brij35, and 2 mM DTT) was carried out with various concentrations of AMPK(α1β1γ1) or AMPK(α1β2γ1) in the range of 0 to 4 nM for 1 h at room temperature. Reactions were terminated at varying incubation times with the Stop Solution/Detection Mix containing 10 mM EDTA, 2 nM Eu-pSAMS-Ab antibody in the detection buffer (50 mM HEPES, 0.01% bovine serum albumin [BSA], and 0.01% Triton X-100) and incubated for another 1 h. Signal was detected using the EnVision Multilabel Reader (Perkin-Elmer) in TR-FRET mode with excitation at 320 nm and emission at 665 and 615 nm, respectively. Because of constant fluorescence signals at 615 nm (emission from the europium antibody), all data on AMPK activity were expressed as fluorescence signals at 665 nm (emission from phosphorylated SAMS peptide), instead of the ratio of fluorescence emissions at 665/615 nm.
To study other components’ (DTT, Mg2+, BSA) effects on AMPK assay, the assay was performed by varying concentrations of each reagent while the rest of the reagent concentrations were fixed.
Development of AMPK TR-FRET Assay in the Presence of PP2A
For further development of our novel phosphorylated AMPK protection assay, the optimized reaction condition was 1 nM AMPK(α1β1γ1) or AMPK(α1β2γ1), 30 nM Cy5-SAMS, and 3 µM ATP in the 1× kinase buffer with a total reaction volume of 8 µL. The varying concentrations of PP2A were preincubated with AMPK enzyme for 1 h at room temperature, and then the substrate solution containing Cy5-SAMS, ATP, and OA was added for incubation for another 1 h at room temperature.
To determine the optimal concentration of OA, a selective inhibitor of PP2A, in our novel phosphorylated AMPK protection assay, the PP2A concentration was held constant at 4 nM and the OA concentration was varied up to 10 nM under defined conditions as described above. For optimization of PP2A concentration, the OA concentration was fixed at 10 nM and the PP2A concentration was varied up to 16 nM. The optimal concentrations of OA and PP2A were selected based on the optimal signal-to-noise ratio.
To test AMPK activators or inhibitors in this assay, various dilutions of the compound stock solution in 100% DMSO were directly added to the kinase buffer to give a final DMSO concentration at 1%, which did not influence the reaction system. The tested compounds were preincubated with AMPK for 30 min, and then the indicated concentration of PP2A was added for incubation of 1 h, followed by incubation with OA/Cy5-SAMS/ATP substrate solution in the reaction system. All reactions were repeated in three independent experiments.
Results and Discussion
In this study, for the first time, we have developed a novel AMPK TR-FRET assay, which is physiologically relevant, simple and robust, to identify a small molecule that activates AMPK and protects AMPK Thr-172 against dephosphorylation induced by phosphatase PP2A under well-defined assay conditions. This in vitro assay is performed in white 384-well plates without any wash steps and is based on Cy5-labeled peptide substrate Cy5-SAMS bound to specific antibody. This TR-FRET AMPK assay format is illustrated in Figure 1 . Cy5-SAMS is phosphorylated by AMPK, which can be partially dephosphorylated by PP2A. The level of the substrate Cy5-SAMS phosphorylation represents the relative AMPK activity.
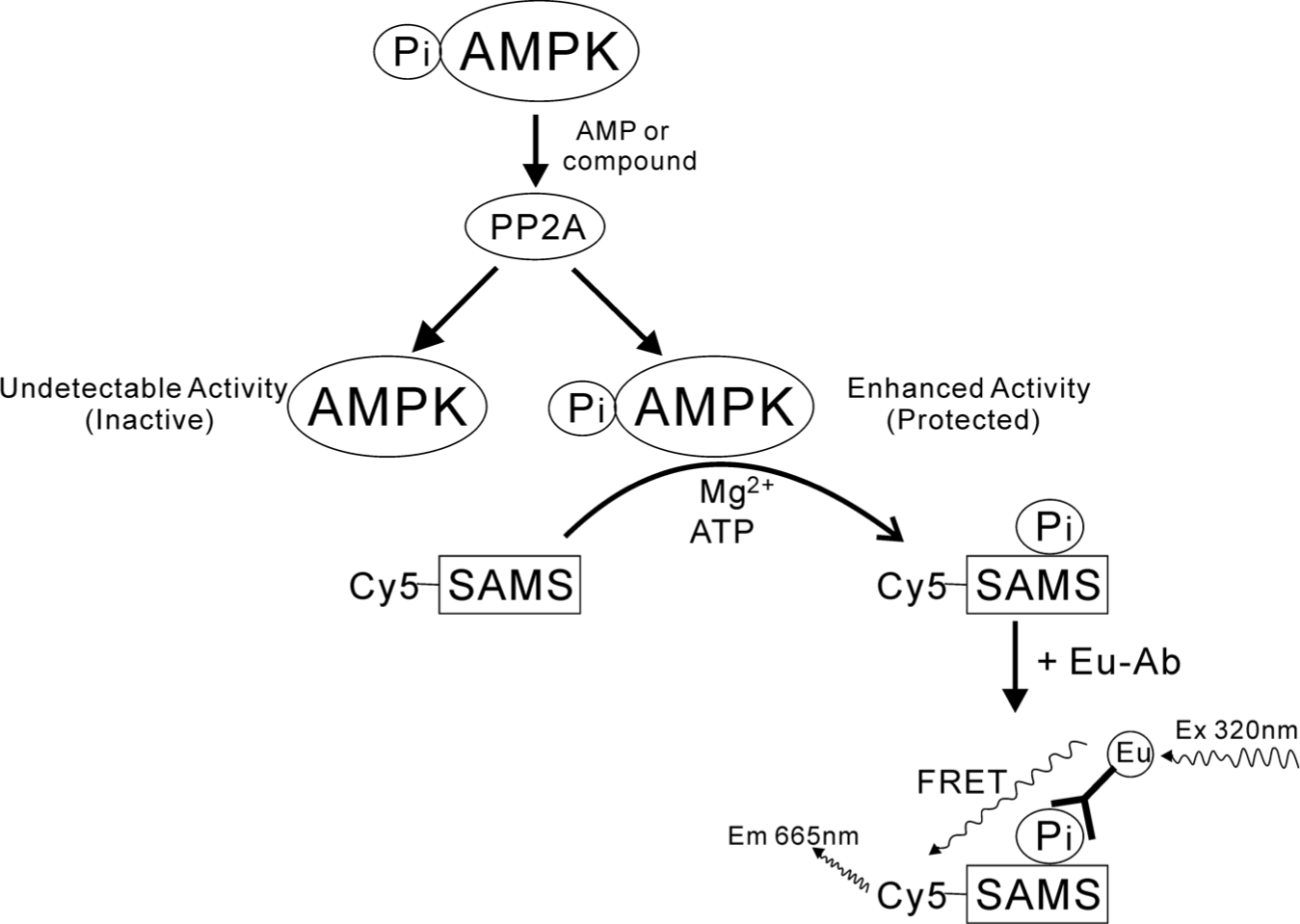
Schematic diagram of the phosphorylated AMP-activated protein kinase (AMPK) protection assay: time-resolved fluorescence resonance energy transfer assay for AMPK in the presence of protein phosphatase 2A.
For determination of the
To establish optimal assay conditions for the TR-FRET assay, the effect of concentration of AMPK enzyme and the time point of the AMPK reaction were first determined. As shown in
Two major factors, protein phosphatase PP2A and OA, would influence the signal/noise ratio in the phosphorylated AMPK assay. OA, a naturally occurring polyether toxin derived from marine dinoflagellates, is a reversible, potent, and selective inhibitor of PP2A. To investigate the effects of OA and PP2A on AMPK activity in this assay, we opted to use 10 nM OA to inhibit PP2A after AMPK co-incubation with PP2A. OA showed no interference on the AMPK reaction system in the absence of PP2A (
Fig. 2A
;
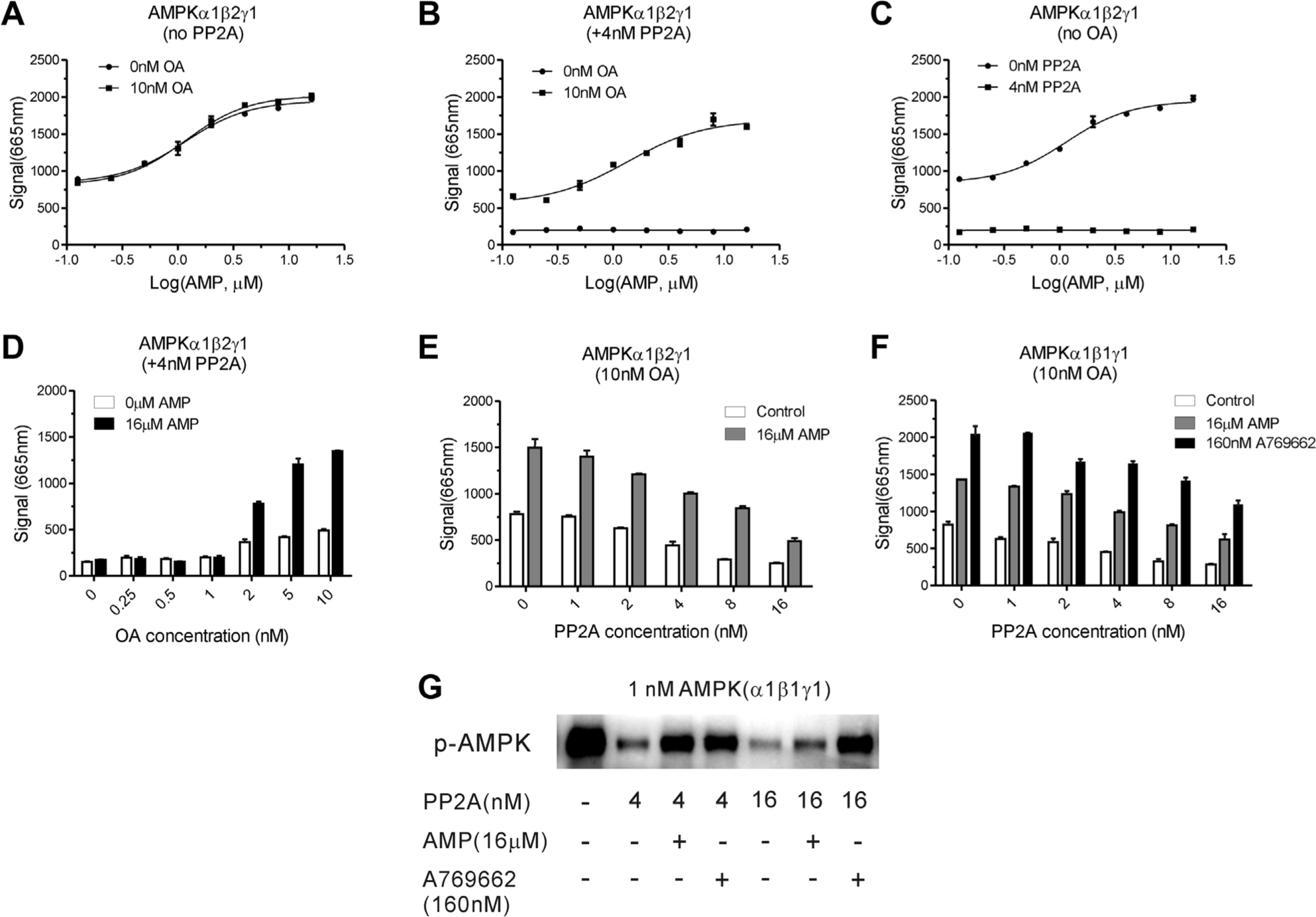
Effects of phosphatase protein phosphatase 2A (PP2A) and Okadaic acid (OA) on the AMP-activated protein kinase (AMPK) assay system. (
In this phosphorylated AMPK protection assay system, the protein phosphatase PP2A and its inhibitor OA play important roles for the measurement of AMPK activity. Our data suggest that PP2A can not only dephosphorylate the activated AMPK but also dephosphorylate the AMPK-phosphorylated substrate Cy5-SAMS, which results in lower AMPK activity signal. After AMPK co-incubation with PP2A and before substrate Cy5-SAMS addition in the AMPK reaction system, OA addition can inhibit PP2A activity for preventing dephosphorylation of substrate Cy5-SAMS. Therefore, OA addition was necessary and indispensable in this AMPK assay.
AMP is an allosteric activator of AMPK. With our novel screening system, AMP activated AMPK(α1β1γ1) and AMPK(α1β2γ1) by about 2- to 2.5-fold with an EC50 of 2.4 and 1.3 µM, respectively, in the absence of PP2A ( Fig. 3A, C ), which was similar to the published data. 15 The activation extent of AMP on both heterotrimers decreased by about one-third in the presence of 4 nM PP2A, with similar EC50 values as that without PP2A. A reported small-molecule AMPK activator, A769662, 12 stimulated AMPK(α1β1γ1) with an EC50 of about 19.8 nM in the absence of PP2A, comparable to an EC50 of about 25.9 nM in the presence of PP2A ( Fig. 3B ). Cool et al. 12 have reported that A769662 activates the baculovirus-expressed AMPK(α1β1γ1) with a potent EC50 value of 0.7 µM using the µARCS assay. This distinct difference of EC50 values for A769662 between the study by Cool et al. and our study may result from the different assay systems and AMPK sources. The activation extent of A769662 was reduced by about 20% when treated with PP2A ( Fig. 3B ). The lower background of this assay in the presence of PP2A may be caused by the partial dephosphorylation of substrate Cy5-SAMS by phosphatase, which will be further investigated in the next study. As expected, A769662, which specifically activated the AMPK-containing β1 subunit, had no influence on AMPK(α1β2γ1) activity ( Fig. 3D ). These results suggest that both AMP and A769662 activated AMPK and also protected AMPK against dephosphorylation by PP2A, which is in accord with a previous report. 12
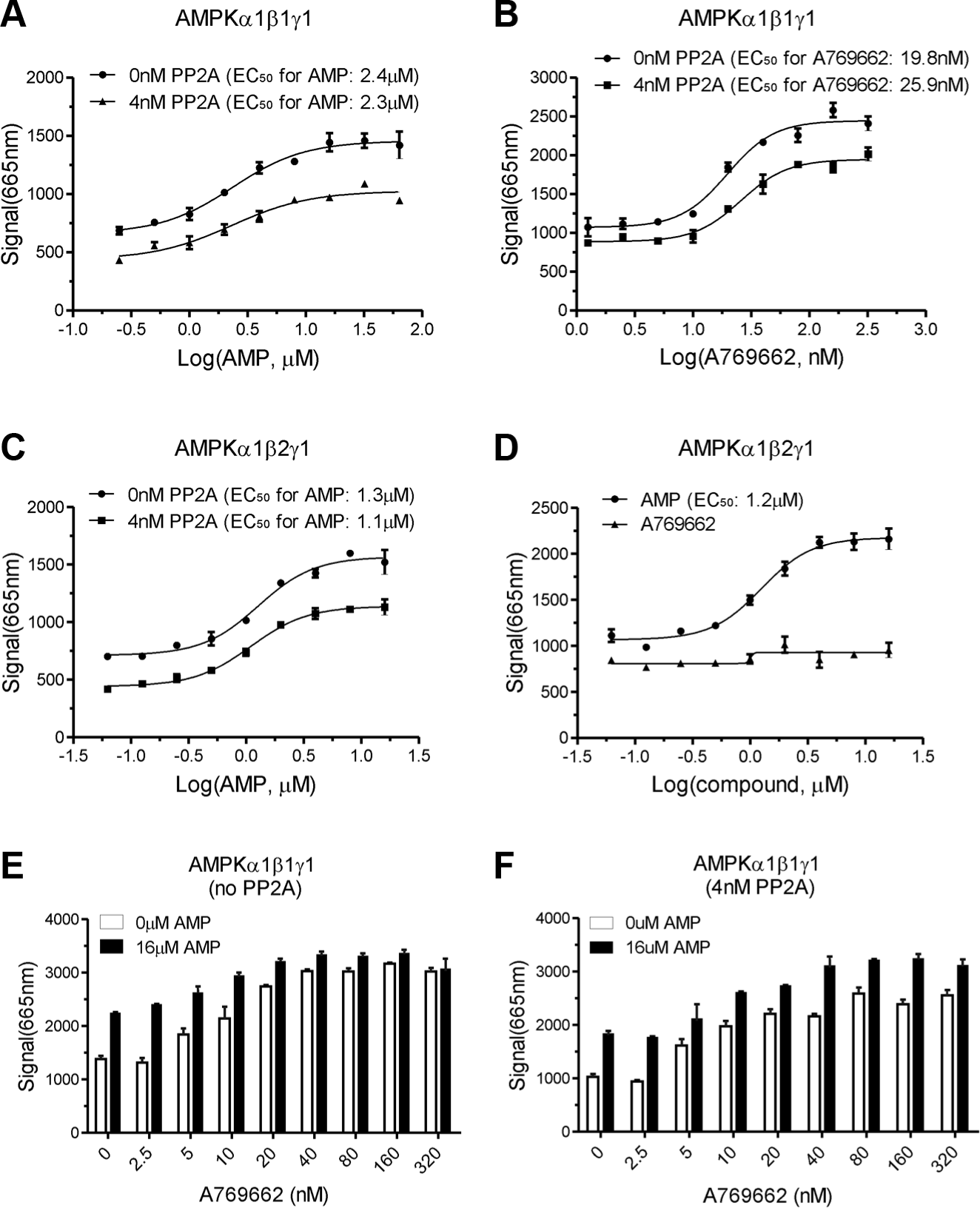
EC50 for AMP and A769662 in AMP-activated protein kinase (AMPK; α1β1γ1, α1β2γ1) in the absence or presence of protein phosphatase 2A (PP2A). (
To further demonstrate that A769662 stimulates AMPK activity in a manner that differs from AMP using this AMPK assay, we determined the effect of A769662 titration on AMPK activity under a saturated concentration of AMP (16 µM). A769662 stimulated AMPK dose dependently and had additive effects in the presence of 16 µM AMP (
Fig. 3E
), and similar trends were observed in the screening assay incubated with PP2A (
Fig. 3F
). It is suggested that both AMP and A769662 activated AMPK with different mechanisms and also protected AMPK against dephosphorylation by PP2A. In addition, a specific AMPK inhibitor compound C titration in both AMPK(α1β1γ1) and AMPK(α1β2γ1) generated similar inhibition curves with an IC50 value of approximately 350 nM (
After fixing all parameters in the optimized condition, we determined the Z′ factor for the assay. In 160 wells of a 384-well plate, we opted to replicate the assay with 16 µM AMP (positive controls), whereas in another 160 wells, the same reaction without AMP and AMPK (negative controls) was performed.
In conclusion, the phosphorylated AMPK protection assay we developed is a sensitive and robust assay that can be used as a screening tool for identifying AMPK activators capable of activating AMPK and protecting AMPK against dephosphorylation by phosphatase in physiological conditions. AMPK activators identified using the current screening assay should be further evaluated in cell-based assays and in vivo to test their abilities of activating AMPK and showing AMPK function.
Footnotes
Acknowledgements
We thank Yuanxue Gao, Sai Chang, and Kangyuan Hu for experimental work. We also thank the anonymous reviewers for their comments and some suggestions for improvement.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the National Natural Science Foundation of China (21402241, 81320108029), the Program for Jiangsu Province Innovative Research Team, the Natural Science Foundation of Jiangsu Province (BK20130653), the Open Project Program of the State Key Laboratory of Drug Research (SIMM1403KF-02), the Open Project Program of State Key Laboratory of Natural Medicines, China Pharmaceutical University (SKLNMKF201407), the Fundamental Research Funds for the Central Universities (JKZD2013006), and the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry (to Dr. Tao Pang).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
