Abstract
The identification of hits and the generation of viable leads is an early and yet crucial step in drug discovery. In the West, the main players of drug discovery are pharmaceutical and biotechnology companies, while in China, academic institutions remain central in the field of drug discovery. There has been a tremendous amount of investment from the public as well as private sectors to support infrastructure buildup and expertise consolidation relative to drug discovery and development in the past two decades. A large-scale compound library has been established in China, and a series of high-impact discoveries of lead compounds have been made by integrating information obtained from different technology-based strategies. Natural products are a major source in China’s drug discovery efforts. Knowledge has been enhanced via disruptive breakthroughs such as the discovery of Boc5 as a nonpeptidic agonist of glucagon-like peptide 1 receptor (GLP-1R), one of the class B G protein–coupled receptors (GPCRs). Most of the original hit identification and lead generation were carried out by academic institutions, including universities and specialized research institutes. The Chinese pharmaceutical industry is gradually transforming itself from manufacturing low-end generics and active pharmaceutical ingredients to inventing new drugs.
Drug discovery is a capital-intensive complex process that bridges the gap between a promising target derived from basic research and drug development for use in the clinic. Once a promising drug target is identified, the steps involved in actual drug discovery are diverse and challenging, which may include identification of screening hits, hit-to-lead optimization, and lead characterization. 1 The quality and structural diversity of leads are essential to chemistry efforts in generating first-in-class or best-in-class medicines. 2
The whole process of drug discovery is quite resource intensive that needs a lot of capital, technologies, and talent pool but with a very limited successful outcome. 3 Because of a close relationship between drug development and human health care, innovation of this nature is strictly regulated by government agencies in every major country. In the past two decades, we have witnessed tremendous economic growth in China, which in turn has led to a marked progress in the quality of this nation’s research and development (R&D) activities. Meanwhile, policy adjustments have also been directed toward improvements on knowledge creation, intellectual property protection, and market-oriented commercialization mechanisms. 4 However, there still exists a large gap between China and Western countries in terms of the maturation of R&D system and the quality of its outputs. In the West and Japan, drug R&D has a clear division of labor, and the main players are pharmaceutical and biotechnology companies, 3 while in China, academic institutions remain central in the field of drug discovery.
Current Status of Drug Discovery Endeavor
The identification of hits and leads is an early and yet crucial step in drug discovery. While routinely seen in major pharmaceutical companies in the West, it is relatively new to Chinese indigenous pharmaceutical industry, whose growth is still largely dependent on generic production, distribution, and sales. 4 In the past more than 20 years, these enterprises have come under a continuous pressure from both the society and the government to innovate. This is coupled with a tremendous amount of investment from the public as well as private sectors to support infrastructure buildup and expertise consolidation relative to drug discovery and development. Since the Patent Cooperation Treaty 5 took effect in China in 1993, the government has launched a series of national mega-research and development programs to encourage drug innovation, such as the medical technical innovation project “1035,” 6 which focused on establishment of a number of technology platforms based on the value chain of drug discovery. For instance, the “Drug Innovation and Traditional Chinese Medicine Modernization” project was aimed at the construction of platform centers covering the whole drug discovery and development workflow, 7 and the “Scientific and Technological Major Special Project for Significant Formulation of New Drugs” 8 emphasized capability enhancement and product development, to name a few. These multiyear, renewable, and adequately funded projects usually involved several unrelated research teams of both academic and industrial origins and led to the formation of China’s independent drug discovery and development system that is helping the country’s transformation from imitation to innovation. As a result, a number of integrated technology platforms have been operational involving both national centers and/or state key laboratories conferring good laboratory practice (GLP) guidelines. 6 With more than two decades of concerted efforts, a drug innovation system with Chinese characteristics has been formed.
The establishment of large-scale compound libraries is a long-cherished ambition in China’s endeavor for innovation. The National Center for Drug Screening (NCDS), 9 which is located at the Pharma Valley of China–Shanghai Zhangjiang High-tech Park, is the largest of its kind in China. In fact, the NCDS has been accumulating small molecules since its foundation in 1997. This effort received a tremendous boost in 2008, when Novo Nordisk A/S of Denmark donated its entire compound library to China and a new public-private partnership was formed as a result to screen active hits against an array of neglected tropical infectious diseases.10,11 Based on this development, the Chinese authorities, both at central and provincial levels, committed three rounds of intensive investment to build a resource center for drug discovery: the Chinese National Compound Library (CNCL), which has been operational since 2012. 12 With a dozen satellite libraries across the country, the CNCL now possesses approximately 1.4 million small-molecule compounds of diverse structures, a large portion of which is proprietary. In conjunction with the NCDS, the CNCL is quickly becoming a model of “open innovation” whereby a variety of drug discovery projects, originated domestically as well as internationally, are performed within its state-of-the-art research facilities based in Zhangjiang, Shanghai, or through provision of compound samples. Indeed, some of the high-throughput screening (HTS) hits highlighted in this review were initially identified via this mechanism.
Viable Leads Generated with Different Strategies
Target-based approaches have been the major focus of drug screening in the past two decades, generally with application of various molecular and cellular bioassays in conjunction with HTS technologies.13–15 However, empirical phenotypic screening is still an important and even more productive strategy to find new chemical start points for drug development. 16 Besides HTS, sometimes people directly modified the structures of natural products to obtain ideal compounds as hits. 11 Scientists often exert their influence by integrating information obtained from different technology-based strategies to achieve a better understanding as to which hit is most suitable for optimization. In this regard, Chinese scientists have been diligently monitoring recent trends in pharmaceutical development, and by tapping abundant resources in terms of available research and development grants, international connections, talent pool, and newly established infrastructures and networks, among others, they made a series of high-impact discoveries in this area. Some representative leads are summarized ( Tables 1 – 5 ), from which one might find certain specific characteristics relevant to drug discovery efforts in China.
Representative Lead Compounds Identified in China for the Treatment of Central Nervous System Diseases.
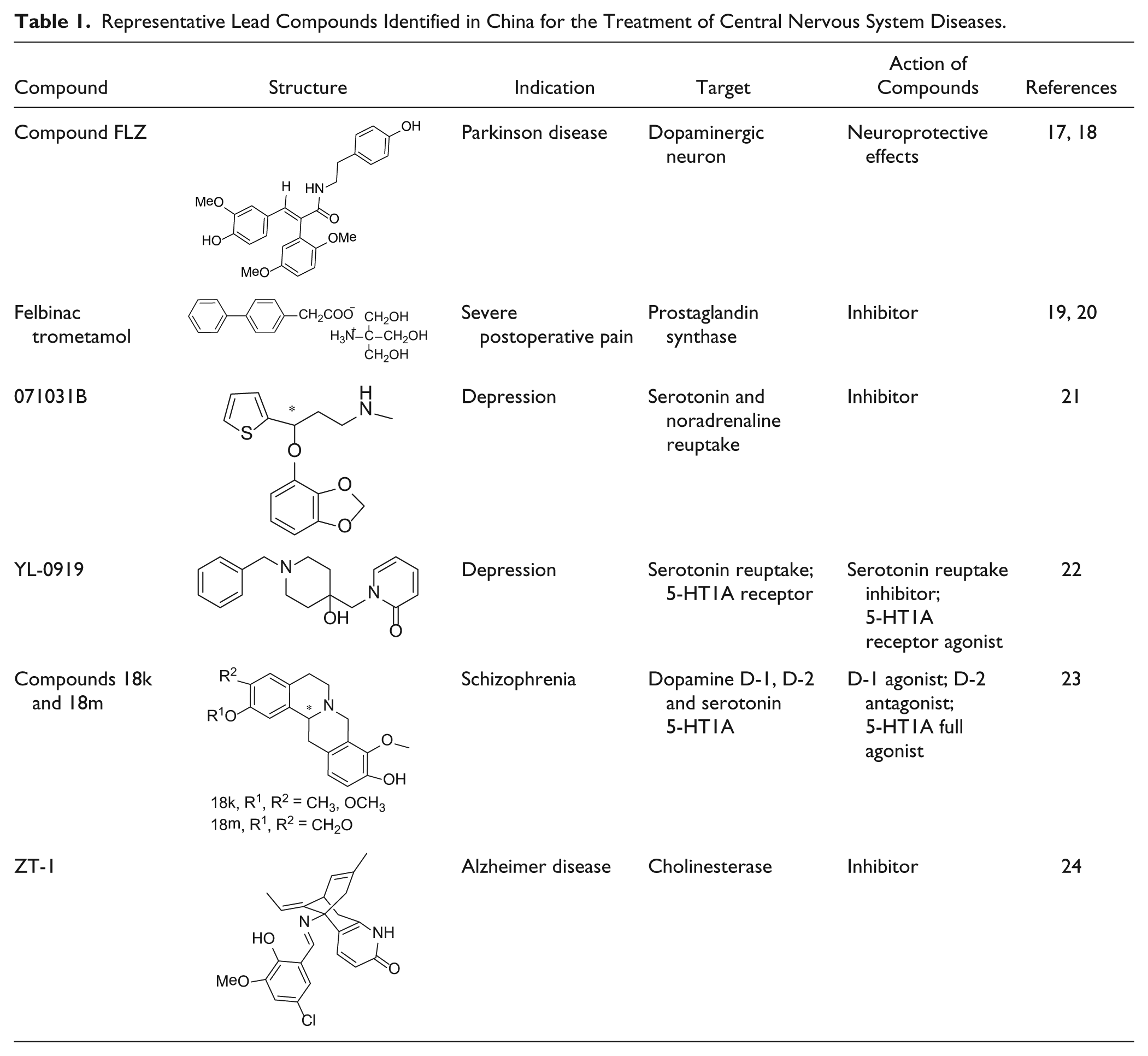
Representative Lead Compounds in China for Metabolic Disorders.
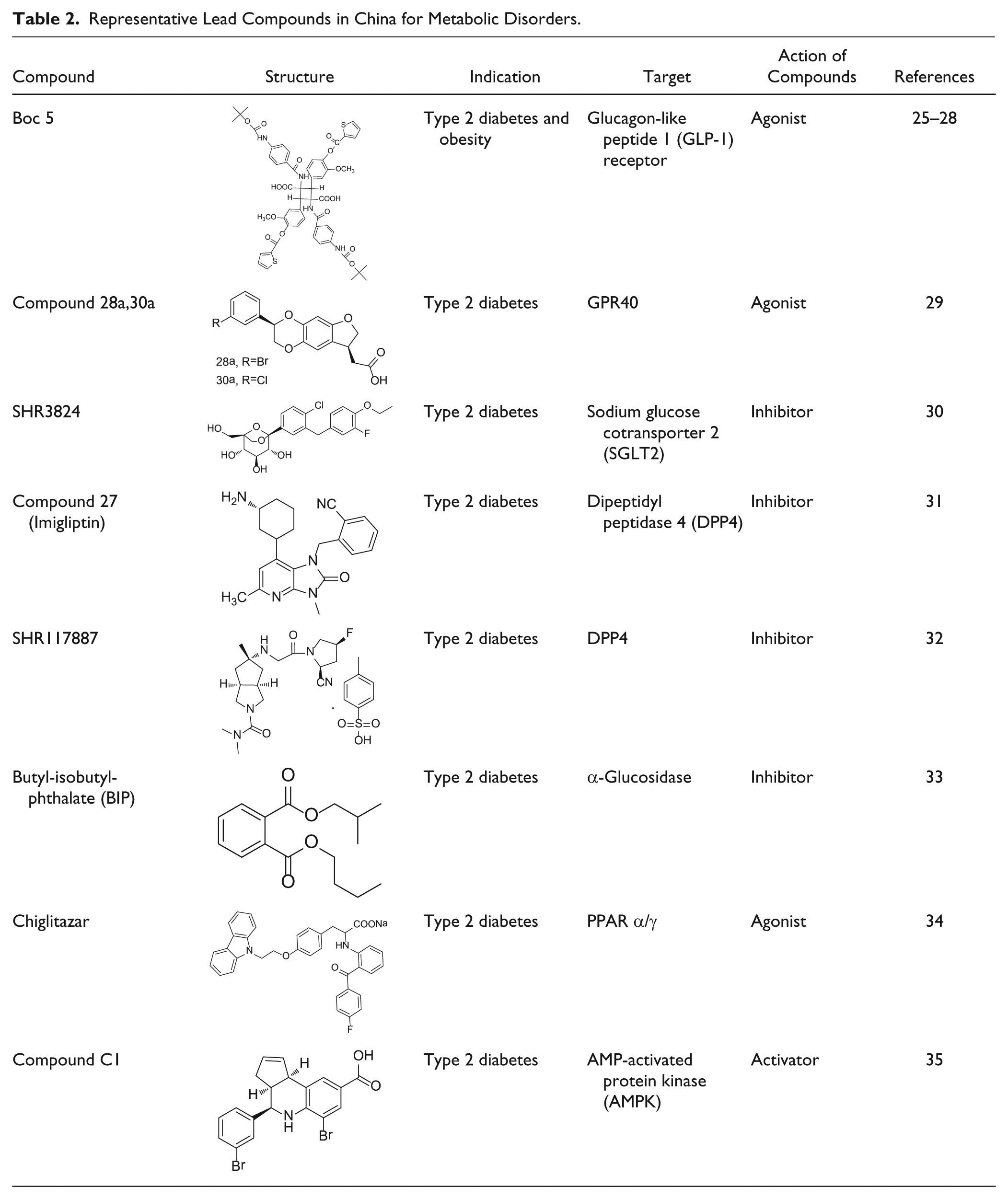
Representative Lead Compounds in China for the Treatment of Allergy, Infection, and Inflammation.
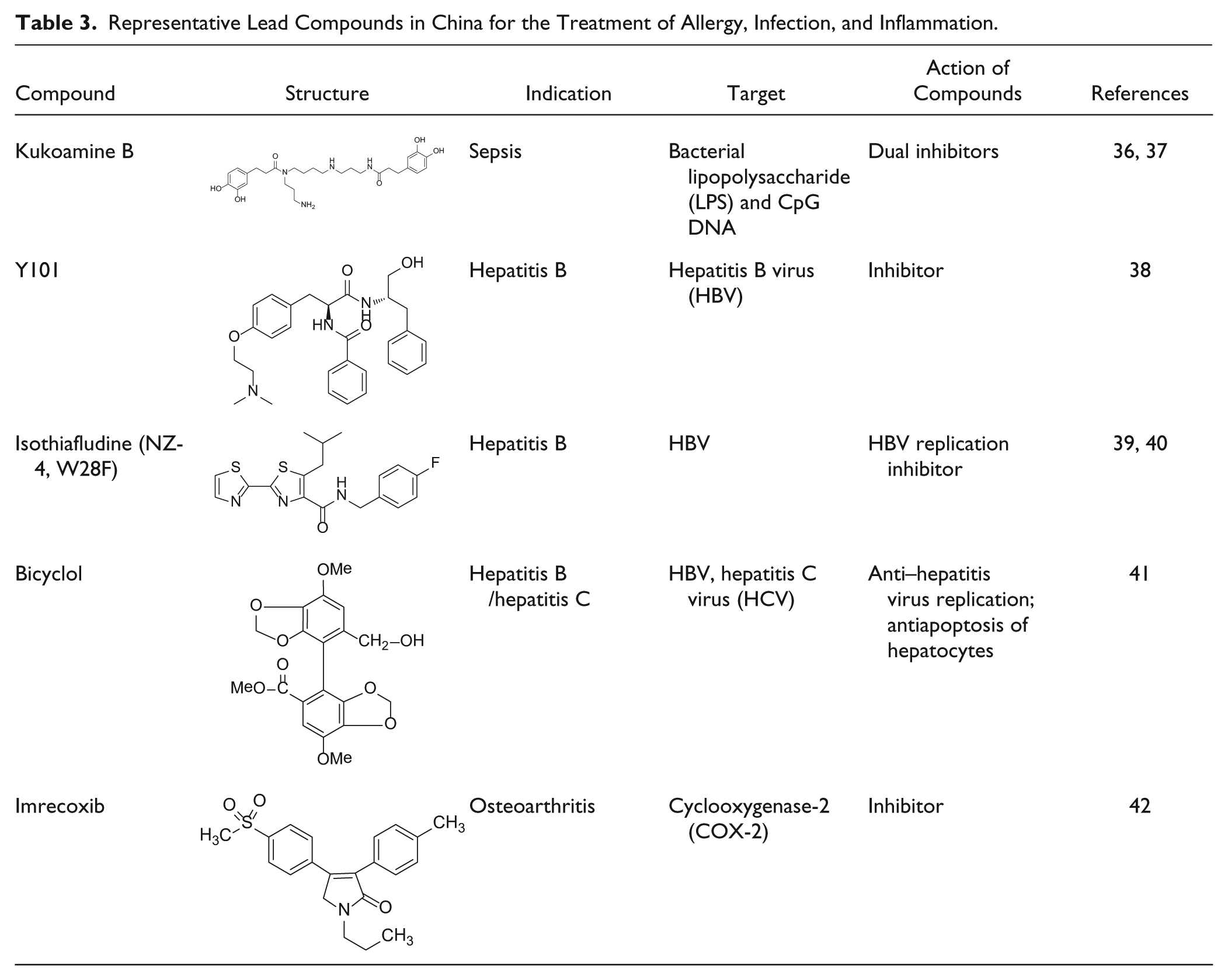
Representative Lead Compounds in China for Cardiovascular Diseases.
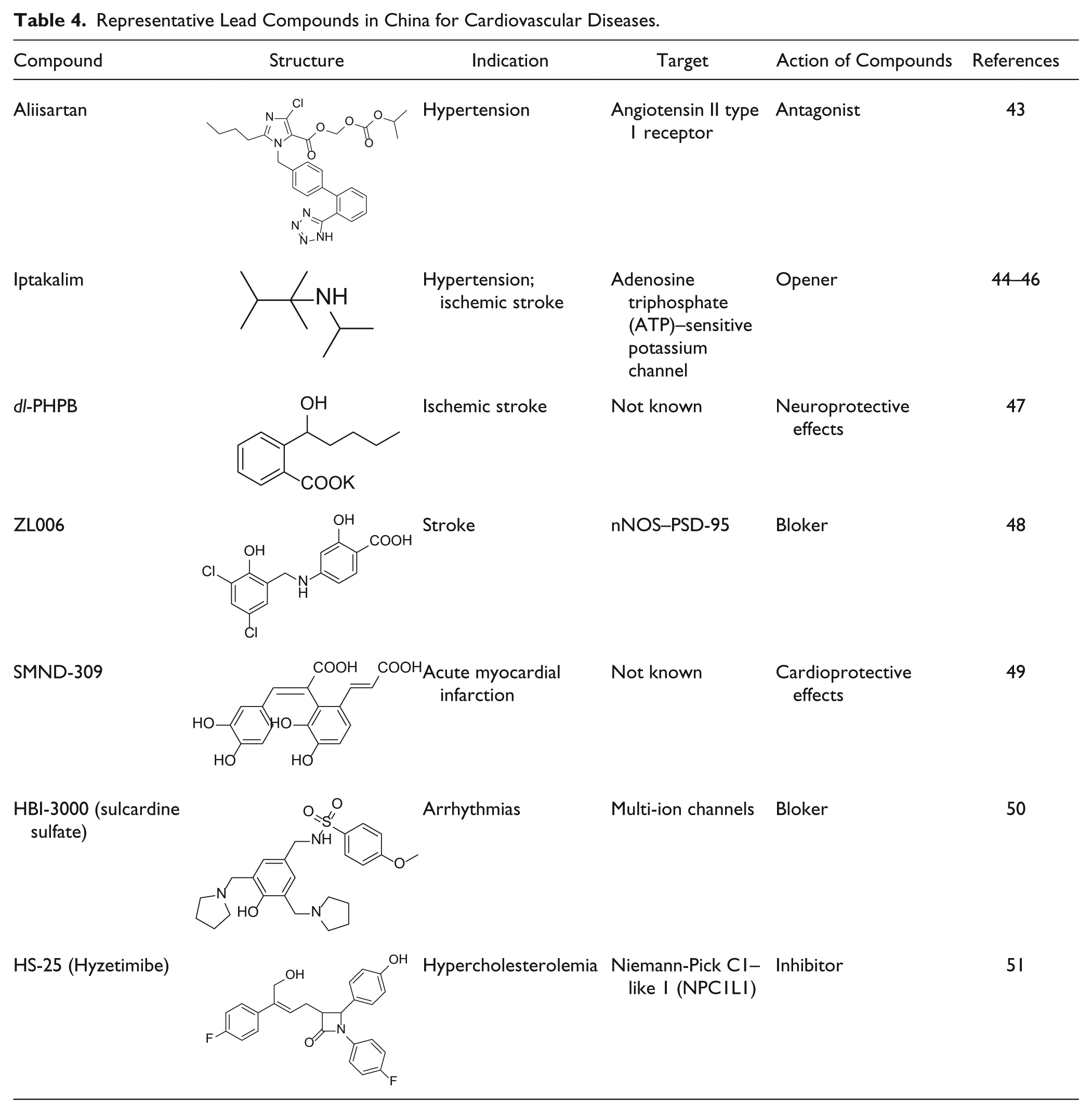
Representative Oncology Lead Compounds Identified in China.
Natural Products as a Focal Point
Searching for active compounds from natural products for pharmaceutical development is a tradition in China, mainly due to the existence of traditional Chinese medicine (TCM).61,62 Despite long-lasting debate about its effectiveness and modernization, TCM still serves as an alternative choice of health care in this most populated country on Earth. There are more than 12,000 kinds of medicinal plants in China, 63 many of which are endemic species. Isolation of active components from Chinese herbal medicines is a common approach to discover structural diversity even for synthetic medicinal chemists. It is known that some isolated compounds may fall under an HTS approach, but most are usually available only in small quantities or as mixtures, making downstream development extremely difficult. 64 Therefore, they are now largely excluded from standardized corporate collections of synthetic small molecules. 65 However, the chemical diversity of natural products is useful in the design of drug-like compounds, and natural products are still a major source in the Chinese drug discovery arena.
When an active component derived from TCM is isolated or synthesized, it can be screened alone or in conjunction with a diverse compound library against a variety of bioassays.66–68 Parkinson disease (PD) is a neurodegenerative disease affecting up to 80% of dopaminergic neurons in the nigrostriatal pathway. Compound FLZ ( Table 1 ), a novel synthetic squamosamide derivative originated from a Chinese herb, was found to have strong protective effects on cultured rat neurons from damage and apoptosis induced by hydrogen peroxide, glutamate, N-methyl-D-aspartate (NMDA), hemoglobin, and ischemia-reoxygenation. 17 This action was subsequently verified in experimental PD animal models: apart from marked improvement in animal behavior, both dopamine levels in the striatal region and the number of dopamine neurons in the substantial nigra increased significantly. 18 FLZ was also shown to inhibit apoptosis, thereby promoting the survival of dopaminergic neurons and their release of dopamine. This lead is presently geared toward a full-scale clinical development.
If therapeutic effects of natural products are well known and their activities can be evaluated with specific bioassays, 63 then hits or leads may be generated by a variety of techniques, including bioactivity-guided isolation strategy. For example, sepsis is a systemic inflammatory response induced by severe infections. Bacterial lipopolysaccharide (LPS) and CpG DNA are important pathogenic molecules and hence drug targets for sepsis. It was therefore envisaged that sepsis might be mitigated by agents capable of neutralizing LPS and CpG DNA simultaneously. Liu and coworkers developed a biosensor screening platform with LPS and CpG DNA immobilized on the surfaces of cuvettes in the biosensor.36,37 Aqueous extracts of Cortex lycii were selected from 114 traditional Chinese herbs for their higher binding affinities to both LPS and CpG DNA. In subsequent experiments, chromatography was used and coupled with the biosensor to purify fractions with high affinities for LPS and CpG DNA. The screening resulted in the discovery of Kukoamine B (KB, Table 3 ), which neutralized LPS and CpG DNA in vitro. KB was further proven to be a novel antiseptic agent and has achieved a total chemical synthesis. It is now undergoing a phase 1 clinical trial. 69
In addition to random screening, use of more focused libraries would improve the efficiency of HTS and save costs. Focused libraries can be constructed by creating subsets of these scaffolds according to certain chemoinformatic criteria or by design and synthesis of target-biased molecules. 70 Leucamide A, a cyclic heptapeptide isolated from the Australian marine sponge Leucetta microraphis, was found to have moderate cytotoxic activity in several tumor cell lines. In an attempted total synthesis of leucamide A, Chen and colleagues 39 made a library of 4,2-bisheterocycle tandem derivatives and identified compound 1 as a lead compound against hepatitis B virus (HBV) in vitro. To diversify the bioactive scaffold, several series of compounds were designed and synthesized, the anti-HBV activity of which was evaluated in HBV-transfected HepG2.2.15 cells. Isothiafludine (NZ-4, Table 3 ), a derivative of bis-heterocycle tandem pairs, was identified as a potent HBV inhibitor. Isothiafludine was effective against HBV DNA replication by blocking the encapsidation of pregenomic RNA (pgRNA) and interfering with the nucleocapsids assembly. 40 Furthermore, NZ-4 efficiently inhibited the replication of HBV mutants resistant to nucleoside and nucleotide analogues. Relevant patent applications have been filed in a number of counties, including China, the United States, and Japan, and a drug candidate based on this structural series is undergoing clinical trials both in China and Europe. 71
Due to the molecular complexity of compounds derived from natural products, some may have problems in absorption, distribution, metabolism, and excretion (ADME). The prodrug strategy sometimes attracts concerns of practicality. 72 Huperzine A is a lycopodium alkaloid isolated from a Chinese herb Huperzia serrata and a reversible and selective acetylcholinesterase (AChE) inhibitor. It was approved in China to treat Alzheimer disease (AD) in 1994. 73 ZT-1 ( Table 1 ), a novel analogue of huperzine A, is rapidly degraded to huperzine A. Phase 2 clinical trials of oral ZT-1 in 35 European hospitals have achieved satisfactory end points. 74 A sustained-release dosage form (administered once a month) has concluded phase 1 clinical studies demonstrating expected blood drug concentrations in a month. 24
Another prodrug is camptothecin (CPT). It is a cytotoxic quinoline alkaloid that inhibits topoisomerase I (topo I) and currently is the only topoisomerase I inhibitor employed in clinical oncology. 75 It was isolated from the bark and stem of Camptotheca acuminata, a plant native to China. Chimmitecan (L-2-Z, Table 5 ) is a novel 9-small-alkyl-substituted lipophilic camptothecin capable of inhibiting topo I catalytic activity as well as trapping and stabilizing covalent topo I–DNA complexes. 76 It displays antitumor activities both in vitro and in vivo. Simmitecan (L-P) 52 is a water-soluble prodrug of L-2-Z, and its clinical development commenced recently with submission of an investigational drug application (IND) in China.
Stroke is a major public health problem, leading to high rates of death and disability in elderly populations. It is known that excessive stimulation of N-methyl-D-aspartate receptors (NMDARs) and the resulting neuronal nitric oxide synthase (nNOS) activation are crucial for neuronal injury after a stroke insult. However, direct suppression of NMDARs or nNOS can cause severe side effects because of their key physiological roles in the central nervous system. Cerebral ischemia induces the interaction of nNOS with postsynaptic density protein 95 (PSD-95). In recent years, disruption of the nNOS–PSD-95 interaction has become a new point of interest to develop drugs preventing cerebral ischemic damage 77 . Zhou et al. 48 , proposed and confirmed that disrupting the ischemia-induced nNOS–PSD-95 interaction may be a strategy to treat stroke that not only prevents ischemic damage but also avoids the undesirable effects of directly blocking NMDAR function and inhibiting nNOS activity. Given this molecular mechanism, a variety of compounds were designed and synthesized to prevent the interaction of the nNOS PDZ domain to PSD-95. A small-molecule inhibitor, ZL006 ( Table 4 ), was identified as the most potent one. 48 It blocked the ischemia-induced nNOS–PSD-95 association selectively, exerted a potent neuroprotective activity in vitro, and ameliorated focal cerebral ischemic damage in rodents subjected to middle cerebral artery occlusion (MCAO) and reperfusion. Moreover, it was capable of crossing the blood-brain barrier while leaving NMDAR function and nNOS catalytic activity unaffected. ZL006 is now undergoing a development program supported by a reputable Chinese pharmaceutical company. 78
Knowledge Enhancement via Disruptive Breakthroughs
G protein–coupled receptors (GPCRs) are the largest group of cell surface receptors characteristic of a common architecture of seven membrane–spanning helical domains. 79 They play an essential role in transducing extracellular signals across the cell membrane via G proteins with high specificity and sensitivity. GPCRs have been the most important and widely used targets for pharmacological intervention. 80 There are three main groups of GPCRs: classes A, B, and C. Class B GPCRs include receptors for several peptide hormones involved in a host of physiological functions from bone maintenance and glucose regulation to immune function and pain transmission. Compared with class A GPCRs, there is a deeper extracellular cavity in the receptor core of the class B GPCR proteins that presumably forms part of the peptide-binding sites, resulting in great difficulty for the identification of small-molecule ligands, especially small-molecule activators or agonists. 81
Most GPCRs have distinct signal transduction pathway(s). HTS assays targeting these receptors can be developed to measure relevant signals along a given pathway, such as calcium mobilization, cAMP accumulation, or other convenient readouts upon ligand binding.82–84 The discovery of Boc5 as a nonpeptidic agonist of glucagon-like peptide 1 receptor (GLP-1R), one of the class B GPCRs, was considered a disruptive breakthrough for this difficult target as well as for this class of GPCRs in general. 85 In one of our attempts to discover small-molecule GLP-1R modulators, 86 we developed a robust bioassay in HEK293 cells stably transfected with a rat GLP-1R vector and a cAMP response element–driven luciferase reporter vector. Following an HTS campaign against 48,160 small-molecule compounds, five initial hits were identified, and two were confirmed to be GLP-1R agonists. One derivative of the hit compound SH14800, NC133909, formed a substituted cyclobutane structure, Boc5 ( Table 2 ), which exhibited a whole spectrum of biological and therapeutic properties similar to those of the native hormone, both in vitro and in vivo.25–27 Unfortunately, follow-up studies on this molecule have rendered it “nondruggable” in terms of oral bioavailability and large-scale production of active pharmaceutical ingredients (APIs). 86
Academic Institutions Still Leading the Way
Unlike in the developed countries, where drug discovery traditionally resides with the private sector (namely, the pharmaceutical industry and, in recent decades, biotechnology companies), in China, such efforts are mainly made in academic institutions, including universities and specialized research institutes, due partially to historical reasons and current funding mechanisms. 3 As a matter of fact, many of the hits or lead compounds presented in this review were discovered by a handful of state-sponsored research entities such as the Shanghai Institute of Materia Medica, Chinese Academy of Sciences (SIMM, CAS), Institute of Materia Medica, Chinese Academy of Medical Sciences (IMM, CAMS), Tianjin Institute of Pharmaceutical Research (TIPR), China State Institute of Pharmaceutical Industry (CSIPI), and Academy of Military Medical Sciences (AMMS). Because these organizations have established some state-of-the-art infrastructure along the value chain of drug discovery and development, most preclinical studies are carried out internally prior to out-licensing deals. In this regard, local pharmaceutical and biotechnology companies have maintained their conventional role as observers in this early stage of drug discovery.
Cooperation between academic research and commercial development ensures that promising leads eventually enter the candidate pipeline of drug makers. Generally, there are two popular ways of collaboration: joint development or technology transfer. The former seems more common these days, as many self-proclaimed academic entrepreneurs wish to mitigate risk should their leads become a “blockbuster” down the road, unless spinning off their own startups. The latter would be a typical transaction seen elsewhere. For instance, sulcardine sulfate 50 ( Table 4 ) is a novel antiarrhythmic agent discovered by SIMM with patents allowed in China, the United States, Japan, and some European countries. In 2010, SIMM executed out-licensing deals, with a domestic pharmaceutical company and an international technology broker, for clinical development and eventual marketing in China and abroad, respectively. 87 Phase 1 clinical studies conducted in the United States and China suggest that this drug candidate has a good safety profile, and now it is undergoing phase 2 clinical studies. Imrecoxib 42 ( Table 3 ) is a new and moderately selective cyclooxygenase-2 (COX-2) inhibitor initially identified by the Institute of Materia Medica (IMM, CAMS), which obtained market clearance in 2011 through a codevelopment arrangement with a large private drug maker based in China. 88
Since Novo Nordisk first set up its research and development center in Beijing in 1997, Roche, AstraZeneca, Novartis, GlaxoSmithKline, and Eli Lilly, to name a few, all have followed the lead and established their drug discovery presence in China, aimed at tapping the local talent pool and reducing development expenditure. Both original research (such as target validation,89–92 lead generation,93–95 and preclinical evaluation96–99) and outsourcing projects are routinely performed in these centers. Obviously, their presence has raised the standard of research, disseminated expertise of invention, and trained a new pool of specialists, all extremely useful to China’s endeavor for innovation. Meanwhile, collaborative programs have been devised between multinational pharmaceutical companies and various Chinese academic institutions. For example, SIMM began to collaborate with French drug maker, Servier, in 1997, and over the years the scope of collaboration expanded such that a joint laboratory has been set up to pursue “early drug metabolism and toxicity evaluation” in addition to conventional single-project collaboration and out-licensing transactions. 100 This win-win arrangement resulted in the foundation of a world-class drug safety evaluation center within SIMM where AstraZeneca was instrumental to its passage to the Organisation for Economic Cooperation and Development (OECD) GLP accreditation.
Role of Private Sector Becoming Visible
With unwavering support from various government agencies with attractive tax incentives, the Chinese pharmaceutical industry is gradually transforming itself from manufacturing low-end generics and APIs to inventing new drugs. Although most companies are still focusing on me-too development, me-betters have become a preferred choice for many. Many newly identified drug targets, such as GPR40,101–103 sodium glucose cotransporter 2 (SGLT2),104,105 and Niemann-Pick C1–like 1 (NPC1L1),106,107 have all been adopted for the screening of me-too or me-better drugs. One would hope that the knowledge and experience obtained in their me-too and me-better successes will sooner or later be transformed into their action in discovering first-in-class drug leads. 108 As a result, the number of drug candidates (class 1.1) reviewed by the China Food and Drug Administration (CFDA) for clinical trials has increased steadily and had a sharp rise in 2013 ( Figure 1 ). 109 Most of the clinical studies of these compounds were carried out by private pharmaceutical companies.
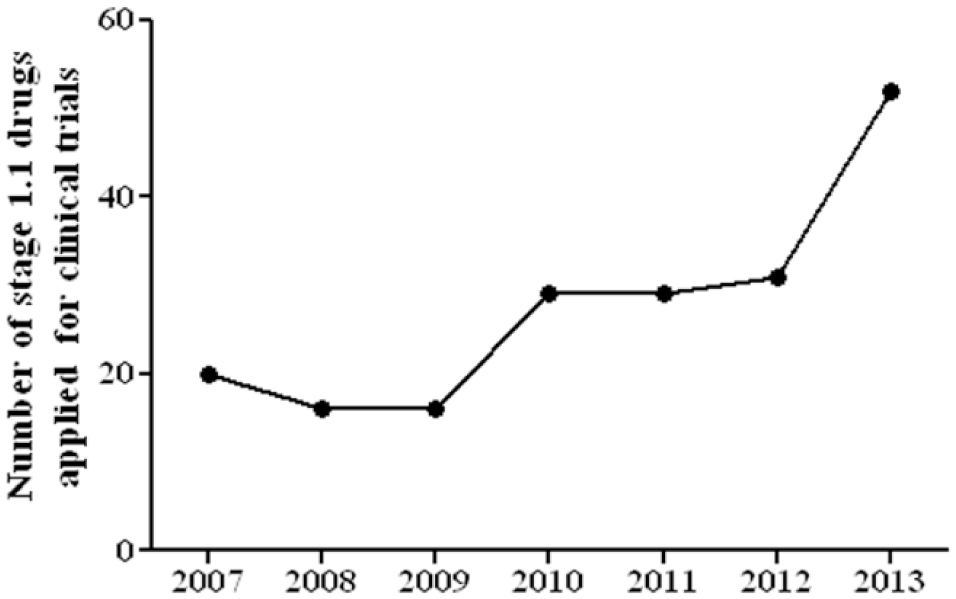
Number of class 1.1 drug candidates undertaken by the China Food and Drug Administration for investigational drug application (IND). Data were derived from the Database of Drug Registration and Acceptance.
In conclusion, a large amount of money has been invested in the past two decades to establish infrastructures for drug development. As a result, a series of leads derived from target-based or phenotypic screenings have entered into clinical trials. Most of the original hit identification and lead generation were carried out by academic institutions, but the role of industry is increasing.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by grants from the National Health and Family Planning Commission of China (2012ZX09304-011, 2013ZX09401003-005, 2013ZX09507001, and 2013ZX09507-002), Shanghai Science and Technology Development Fund (13DZ2290300), and the Thousand Talents Program in China.
