Abstract
There is an urgent need to develop new, safe, and affordable macrofilaricidal drugs for onchocerciasis and lymphatic filariasis treatment and control. The Anti-Wolbachia Consortium (A·WOL) aims to provide a novel treatment with macrofilaricidal activity by targeting the essential bacterial symbiont Wolbachia. The consortium is currently screening a diverse range of compounds to find new chemical space to drive this drug discovery initiative and address this unmet demand. To increase the throughput and capacity of the A·WOL cell-based screen, we have developed a 384-well format assay using a high-content imaging system (Operetta) in conjunction with optimized Wolbachia growth dynamics in the C6/36 Aedes albopictus mosquito cell line. This assay uses texture analysis of cells stained with SYTO 11 as a direct measure of bacterial load. This validated assay has dramatically increased the capacity and throughput of the A·WOL compound library screening program 25-fold, enriching the number of new anti-Wolbachia hits identified for further development as potential macrofilaricides for onchocerciasis and lymphatic filariasis.
Introduction
Lymphatic filariasis and onchocerciasis are highly disabling diseases that affect more than 150 million people throughout the tropics. Both of these diseases are due to infection by filarial nematodes: Wuchereria bancrofti, Brugia malayi, and Brugia timori for lymphatic filariasis and Onchocerca volvulus for onchocerciasis. These filarial worms all contain endosymbiotic bacteria, Wolbachia, which are vital for nematode survival and fertility.1–5
Current treatments for these diseases include diethylcarbamazine, albendazole, and ivermectin. Although these drugs are effective in killing the microfilariae (mf, or juvenile worms), they have poor macrofilaricidal activity (adult worm death). The antibiotic doxycycline is the first drug to deliver substantial macrofilaricidal activity safely, acting by depletion of the endosymbiotic bacteria (Wolbachia) from the nematode. This anti-Wolbachia treatment has multiple benefits, including safe macrofilaricidal kinetics and sterilization of the adult worms leading to a sustained clearance of mf.3,6 In addition, this treatment has been shown to reduce the pathology of lymphedema and hydrocele associated with lymphatic filariasis.7 –9 Although doxycycline provides superior therapeutic activity compared to standard antifilarial drugs, the logistics of a 4–6-week course of treatment and the contraindication in children and pregnant women are barriers to its use in wide-scale mass drug administration (MDA) campaigns. The Anti-Wolbachia Consortium (A·WOL) was established to discover and develop new anti-Wolbachia drugs to overcome these barriers.1–4
A·WOL has screened in excess of 18,000 compounds in a cell-based assay that used Wolbachia-infected insect cells [C6/36 (wAlbB)] cultured for 9 days in the presence of test compounds. Anti-Wolbachia activity was assessed by Wolbachia reduction, which was determined using a quantitative PCR (qPCR) assay targeting the 16S ribosomal RNA (rRNA) gene.1–3,10 The development of this assay, as well as the screening of the complete human pharmacopeia against Wolbachia, is described in Johnston et al. 1 Although robust and fully validated for use as a screen, the qPCR approach lacked throughput. To increase screening throughput and capacity, we have modified this assay to exploit the benefits of the Operetta high-content automated imaging system.
Here, we demonstrate the development of the first anti-Wolbachia high-content screen (HCS) fully validated for a high-throughput drug discovery screening program—including edge effect analysis, drift analysis, Z′ analysis, and dose–response curve (to show sensitivity and EC50 results). Compounds with known anti-Wolbachia activity throughout our screening pipeline, from activity in vitro to efficacy in clinical trials (e.g., doxycycline), were used to both validate the assay system and evaluate library compounds with previously unknown activity. This anti-Wolbachia HCS assay has dramatically increased the capacity of the A·WOL drug discovery program greater than 25-fold.1–3
Materials and Methods
Cell Culture
The C6/36 (wAlbB) cell line is a mosquito (Aedes albopictus) derived cell line stably infected with Wolbachia pipientis (wAlbB). To create this cell line, the supernatant from cultured Aa23 cells (A. albopictus) naturally infected with the W. pipientis strain wAlbB was harvested and filtered to remove whole cells. This supernatant was used to inoculate C6/36 cells (ECACC No. 89051705), resulting in a stably Wolbachia-infected cell line C6/36 (wAlbB).1,11–12 Cells were incubated at 26 °C and subpassaged every 7 days using a 1-in-4 dilution in Leibovitz media (Life Technologies, Loughborough, UK) supplemented with 20% fetal bovine serum (FBS; Fisher Scientific, Loughborough, UK), 2% tryptose phosphate broth (Sigma-Aldrich, Poole, UK), and 1% non-essential amino acids (Sigma-Aldrich).
Anti-Wolbachia HCS Assay Setup
C6/36 (wAlbB) cells were subpassaged 6–8 days before plating out at a density of 2000 viable cells per well in a 384-well CellCarrier plate (PerkinElmer, Llantrisant, UK), suspended in Leibovitz media with the additives described in the “Cell Culture” section. All compounds were dissolved in DMSO with each compound added to a single well at a final concentration of 5 µM (resulting in <1% final DMSO concentration). Control samples per plate consisted of 12 wells of vehicle control (DMSO) and 6 wells of the following controls: 5 µM doxycycline (positive control—the gold standard for Wolbachia reduction; SigmaAldrich) and a suboptimal 50 nM doxycycline concentration. Each well held a final volume of 100 µl, with the exception of the outer wells, which contained 130 µl of phosphate-buffered saline (PBS; SigmaAldrich).
After 7 days of 26 °C sterile incubation, 25 µl of staining media containing 60 µM of SYTO 11 DNA dye (Life Technologies) was added to each well. After a 15 min incubation, all media was removed from each well and replaced with fresh media (no stain). Using the Operetta high-content automated imaging system (PerkinElmer), five fields per well were imaged using a confocal 60× objective with the Fluorescein filter (excitation filter: 460–490; emission filter: 500–550). The PerkinElmer software Harmony was trained to first identify the cell nucleus and cytoplasm, followed by the spot edge ridge (SER) texture analysis, which was used to score each intact cell on the complexity of the cytoplasm (
Fig. 1A
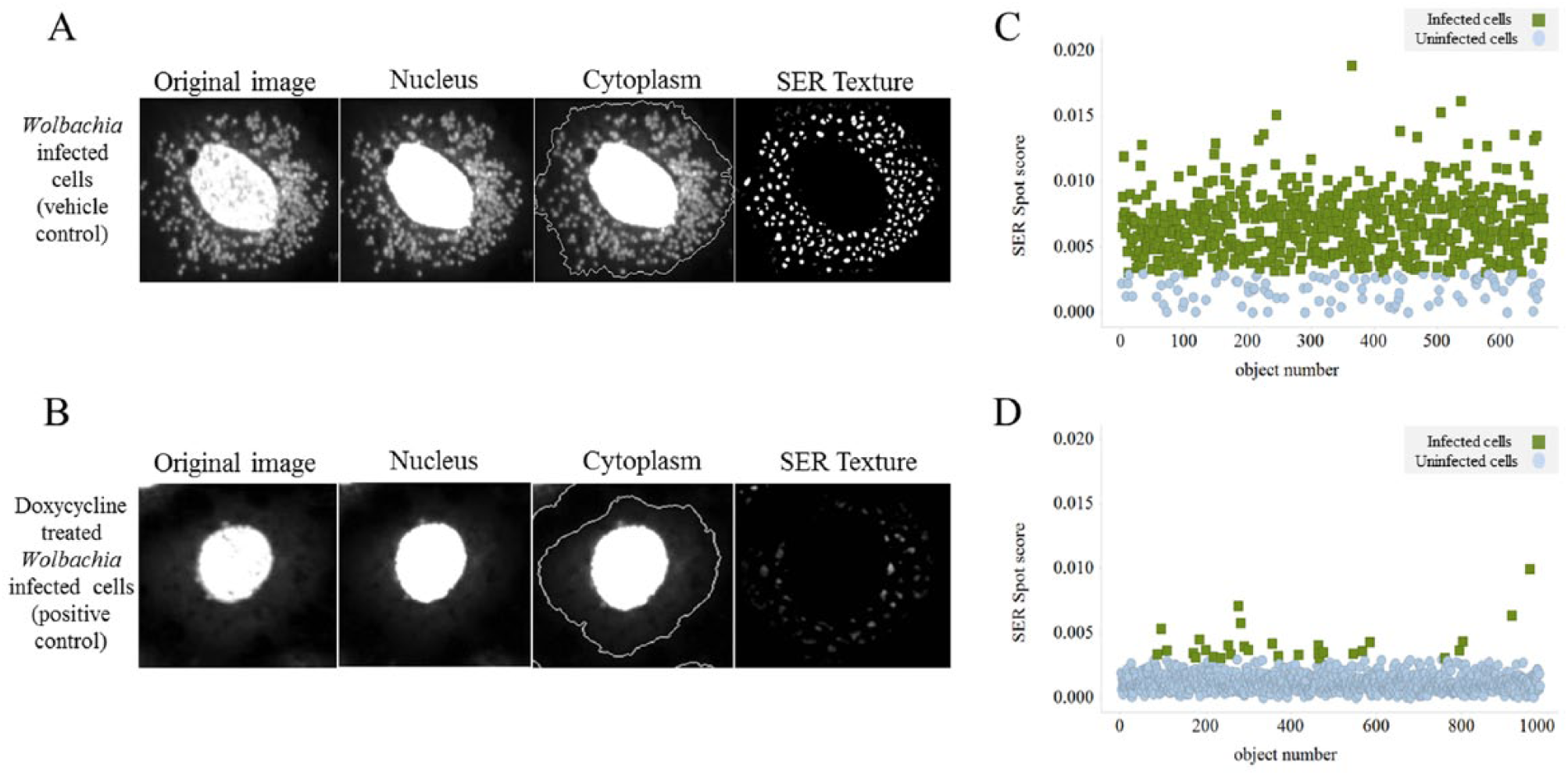
Anti-Wolbachia Consortium (A·WOL) whole-cell anti-Wolbachia screen. (
Anti-Wolbachia HCS Assay Results Analysis
Using the vehicle and positive (Wolbachia reduction) controls, a threshold was set to indicate if each cell was classed as infected or uninfected. Wolbachia-infected cells (vehicle control) have a complex cytoplasm texture (high SER texture score), whereas Wolbachia-uninfected cells (doxycycline-treated positive control) have a uniform cytoplasm texture (low SER texture score). From this analysis, the following readouts were calculated per well: cell number, SER texture score, and percentage of Wolbachia-infected cells.
Z' factor (Z') validation of each plate was calculated using the percentage of Wolbachia-infected cells value from the vehicle and positive controls.
14
Vehicle controls have a high Wolbachia load and therefore a high percentage of cells classed as infected with Wolbachia. Positive control (doxycycline-treated) cells have a low Wolbachia load and therefore a low percentage of Wolbachia-infected cells (
Fig. 1C
All compound sample wells were then analyzed and normalized (along with the positive controls) against the vehicle (untreated) control to give a percentage reduction of Wolbachia-infected cells. In addition, using the cell number analysis, any compounds with a host cell number amounting to less than 50% of the vehicle control were classed as toxic and retested at a reduced compound concentration.
All compounds that were >90% of the positive control’s percentage reduction of Wolbachia-infected cells were classed as strong hits (because they were similar to or greater than the 5 µM doxycycline positive control). Compounds that yield infection rates between 50% and 90% of the positive control were classed as moderate hits [because they were similar to the suboptimal (50 nM) doxycycline control]. All hit compounds were then reconfirmed in a full dose response to define their potency.
Results
Wolbachia-Infected C6/36 (wAlbB) Cell Line Dynamics
For image-based assays that use a per cell analysis, it is important to optimize the homogeneity of infection levels between cells. Initial analysis using the Operetta system highlighted that Wolbachia-infected cells can exhibit variable bacterial loads from cell to cell, with some cells actually appearing to be uninfected (data not shown). Such variation reduces the signal window, thus reducing the quality of any resulting assay and preventing assay validation (see the “Assay Validation” section). We therefore optimized the seeding density and media supplements to provide a robust method for producing cells with high homogeneous Wolbachia loads. The main parameter that affected Wolbachia infection homogeneity was the percentage of FBS used in the culture media. Switching from the standard 5% FBS in the culture media to 20% typically increased the percentage of infected cells from 14–60% to more than 70% among replicate wells. Overall, cells 6–8-day post passage, seeded at 2000 viable cells per well of a 384-well plate, and maintained in media containing 20% FBS provided the optimized conditions.
Assay Optimization
Various settings were optimized on the Operetta system to increase the throughput of the assay. SYTO 11 green fluorescent nucleic acid stain was used to visualize both the Wolbachia and the host cell C6/36 (wAlbB) nuclei. The use of SYTO 11 as a live cell stain further enhances the simplicity of this method by reducing the number of steps involved in the staining process.
A 60× objective was used for this assay because it gave the optimum cellular detail to assess the intracellular Wolbachia load while maximizing the number of cells analyzed per field and image. The final image from each field is a composite from three images taken at separate focal planes. This composite image proved robust in capturing the majority of cells in focus. The optimization of the number of fields and number of planes was based on iterative Z' analysis.
Once the images were obtained, Harmony was optimized to identify the cell nuclei and cytoplasm (
Fig. 1A
Harmony Texture Analysis
Initial attempts to count the number of Wolbachia per cell proved fruitless because the density in cells maintained with 20% FBS culture media was too high to achieve any accuracy through spot identification. Therefore, the Wolbachia infection was determined by the texture analysis function available within Harmony using spot edge ridge (SER) texture analysis. This analysis used the cytoplasm (nucleus excluded) as our image region of interest. Wolbachia-infected cells have a complex pattern to their cytoplasm that results in a high SER texture score for each cell (
Fig. 1A
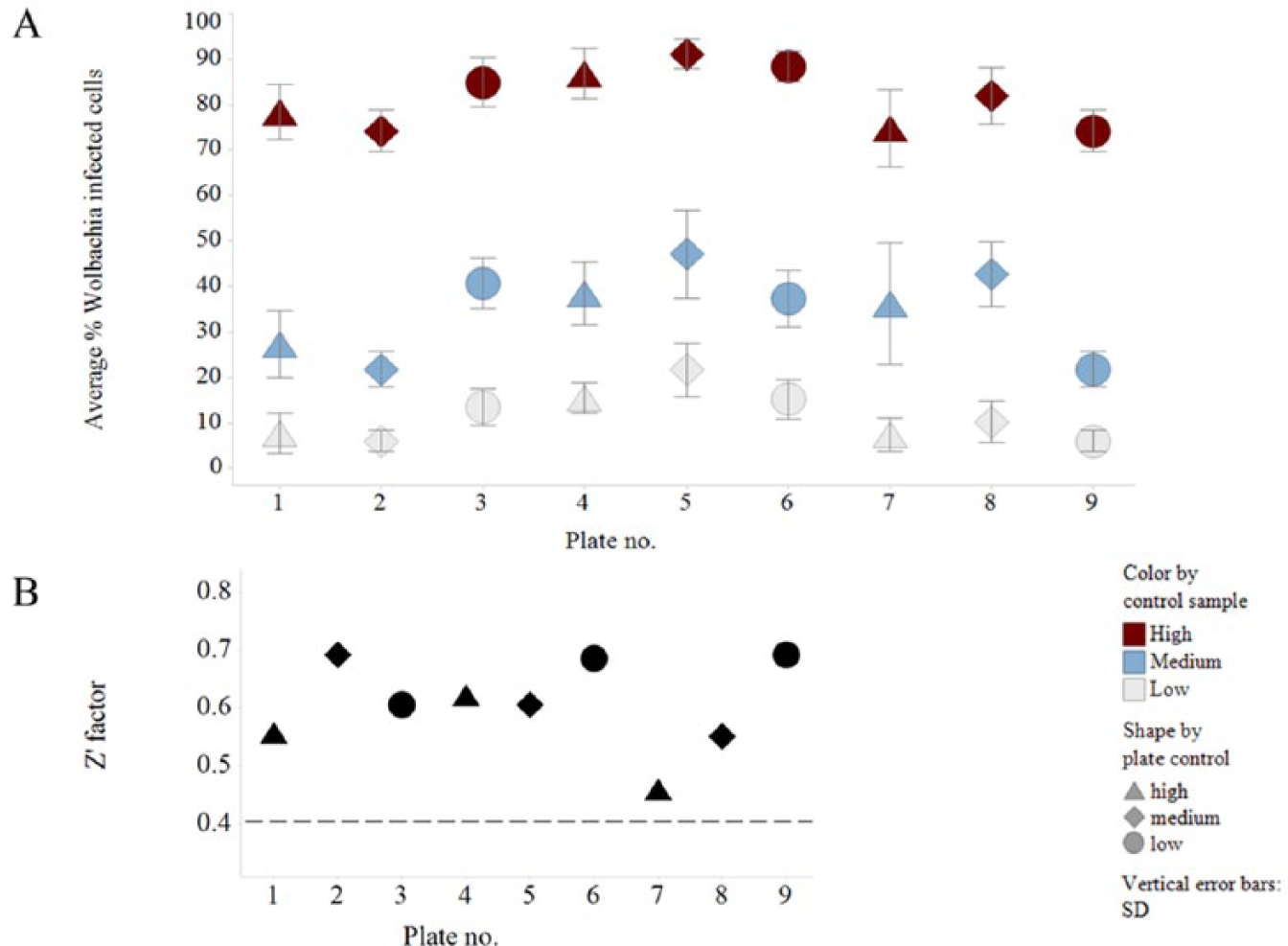
Assay validation data. (
Assay Validation
The vehicle (Wolbachia-infected) and positive (doxycycline-treated) controls were used to calculate the Z' score. The Z' measures the ratio of the signal window to the signal dynamic range of the assay based on a particular number of sample replicates. 14 Within this assay, we achieved satisfactory Z' scores (greater than 0.4) with a single sample replicate ( Fig. 2B ). Next, we validated the assay, including testing for edge or drift effects within each plate, as well as inter- and intraday variability. Three plates were prepared containing columns of cells with high (H; vehicle), medium (M; 50 nM suboptimal dose of doxycycline), and low (L; 5 µM doxycycline) levels of Wolbachia infection. Each plate contained the same repeating column pattern of H, M, and L; however, each plate started at a different point in the pattern. 15 Using this method among all three plates, a single well position will gain a result for each treatment. This was then repeated on 3 different days. We obtained consistent and robust results in this validation with all plates passing the Z' ( Fig. 2 ), with little interday or intraday variation between the plates, or drift and edge effects within each plate ( Fig. 3 ).
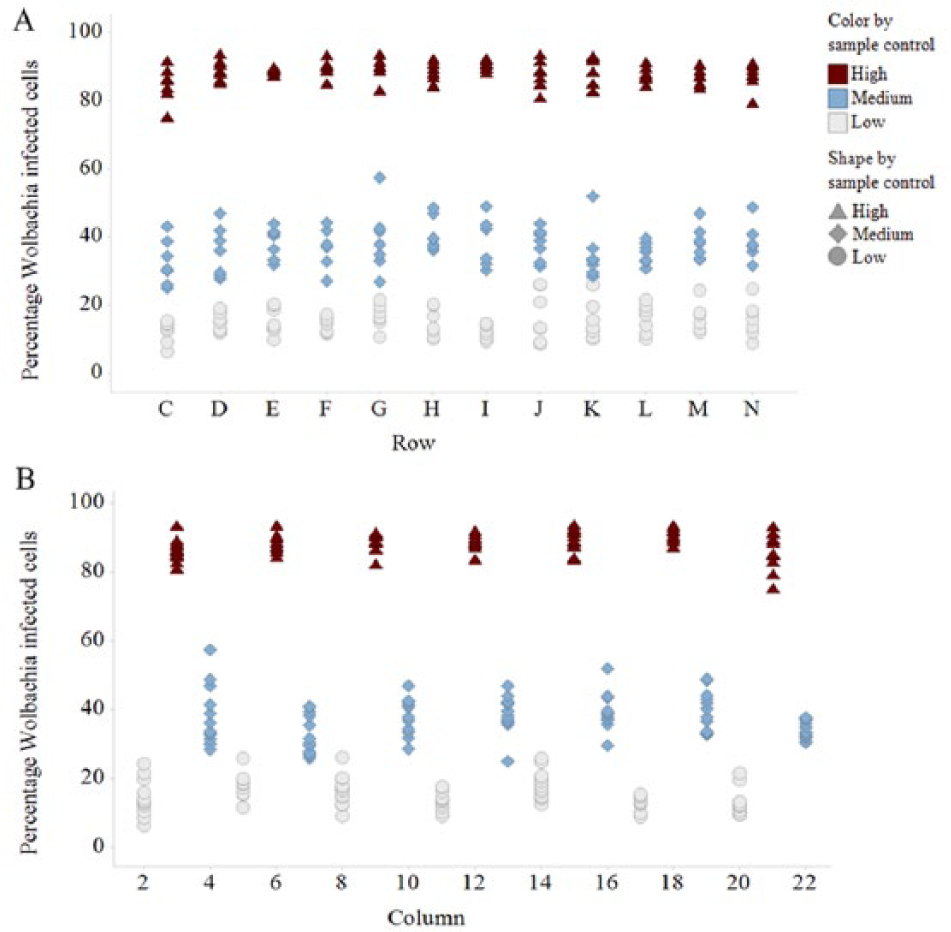
Drift and edge effects validation data: an example from plate 6 showing there is no drift across (
Hit Dose Responses
A significant benefit of this assay is that we now have the capabilities to produce EC50 dose–response curves on our hits. These curves are now routinely obtained in our primary screen for our smaller libraries and as a secondary screen for our larger library hits. Doxycycline dose–response curves are also run as a reference in these routine screens ( Fig. 4 ). This capability has allowed us to initiate a medicinal chemistry “hit-to-lead” and lead optimization development phase in the A·WOL II Macrofilaricide Drug Discovery program.2–3
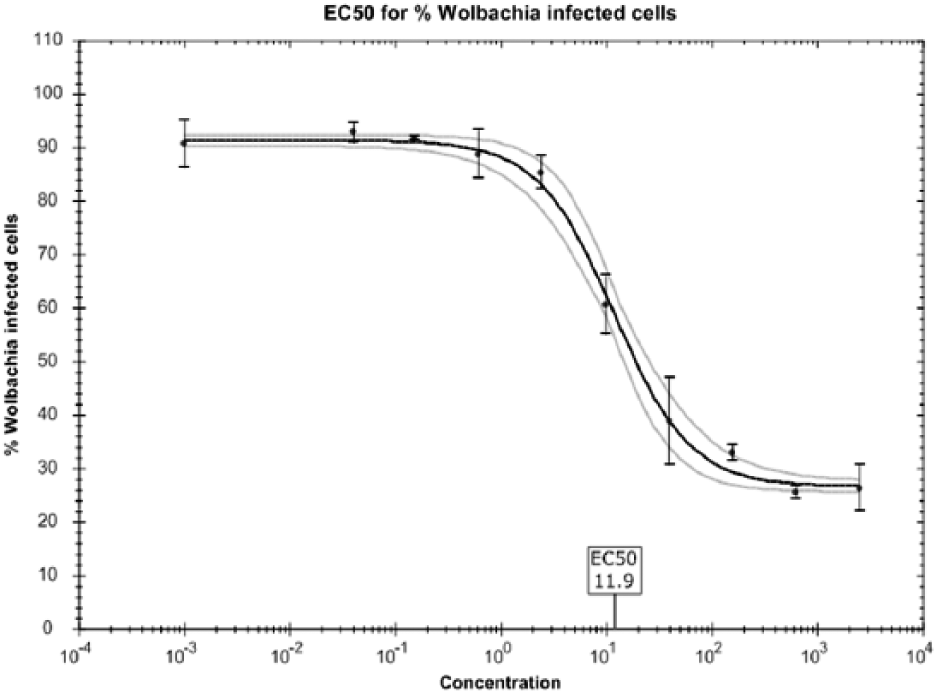
Dose response for doxycycline in the Anti-Wolbachia Consortium (A·WOL) whole-cell anti-Wolbachia screen. This is an example of a dose–response curve generated using doxycycline in the validated assay described in this article, resulting in an EC50 of 11.9 nM.
Toxicity
Toxicity is assessed in this assay by comparing the host cell number from compound samples to that of the vehicle control. This toxicity can result from either cell death or reduced cell replication due to exposure to a compound. In this assay, a compound with a cell number less than 50% of the vehicle control cell number is classed as toxic.
Discussion
Through optimizing cell growth dynamics and using image-based detection with the Operetta high-content automated imaging system, we have developed and validated a cell-based screening assay with a higher throughput and capacity for the primary screening of anti-Wolbachia drugs. Improvements have also been made in evaluating cell toxicity using Harmony. The miniaturization from the 96-well qPCR format to a 384-well imaging format and the simplification of the staining method from a multistep DNA quantification method have dramatically improved not only the throughput but also the screening cost per drug. 1
All of these improvements have resulted in a 25-fold increase in throughput compared with the qPCR method, through which we were able to screen approximately 1000 compounds per month. Using this new HCS assay, we are able to screen more than 25,000 compounds per month.
Increasing the FBS content in the incubation media has reduced the sensitivity of our cells, therefore reducing the number of hits discarded due to toxicity as well as reducing the number of retests required to confirm that the toxicity was not masking a potential hit. An additional benefit of this screen is the improved data quality we obtain, because we now have results from an individual cellular level rather than a single whole sample readout. This has allowed us to directly monitor Wolbachia and host cell dynamics within a population, as demonstrated in our initial tests to compare Wolbachia levels in 5% and 20% FBS culture media. This was an extension on work by O’Neill et al., 12 which stated that increasing the percentage of FBS can increase the intracellular Wolbachia load as well as the number of cells infected.
There is scope to miniaturize this assay further to a 1536-well format, which would again increase throughput but would require liquid-handling robotics and cell concentration optimizations. Time-kill assays are also now possible to determine how rapidly compounds have anti-Wolbachia activity.
This assay should be easily transferable to other laboratories with similar equipment. This transfer is currently being tested at AstraZeneca in the first open-access HTS project for the World Intellectual Property Organization’s (WIPO) Re:Search program against neglected tropical diseases. Through this collaboration, staff from A·WOL are using the leading automation, technology, and expertise at AstraZeneca’s Global High Throughput Screening Centre to scale up the A·WOL high-throughput HCS screen. The successful validation of this new assay could enable screening against AstraZeneca’s full compound library in a single screening activity.
Our validated cell-based HCS screen mirrors the move toward HCS of whole organisms or cell-based assays becoming a lot more popular in both the pharmaceutical industry and academia, including the neglected tropical disease field.2,16–22 This change is partly due to the high rate of attrition from early hits to robust lead compounds, because many of the molecular target-based hits are not active at a whole-cell level. Due to HCS phenotypic whole-cell and whole-organism screens being initiated at this whole-cell rather than molecular level, this should increase the chance of obtaining robust lead compounds.23–25
Improvements to our primary screening assay have created the opportunity to increase the size and diversity of our library screening activities to improve the prospects of finding new anti-Wolbachia drugs as macrofilaricidal therapies against filarial nematodes of medical and veterinary importance that rely on Wolbachia symbiosis: onchocerciasis, lymphatic filariasis, and dog heartworm (Dirofilaria immitis). 4
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A·WOL is supported by a grant from the Bill and Melinda Gates Foundation to the Liverpool School of Tropical Medicine.
