Abstract
Pesticides currently in widespread use often lack species specificity and also become less effective as resistance emerges. Consequently, there is a pressing need to develop novel agents that are narrowly targeted and safe to humans. A cell-based screening platform was designed to discover compounds that are lethal to mosquito (Anopheles and Aedes) cells but show little or no activity against other insect (Drosophila) or human cell lines. Mosquito-specific, aqueous-stable cytotoxins were recovered at rare frequencies. Three of these were profiled for structure-activity relationships and also assessed in whole-animal toxicity assays. In at least one test case, species-specific cytotoxicity seen in culture effectively translated to the whole-animal level, with potent toxicity against Anopheles yet none against Drosophila. Therefore, this initiative has the potential to advance novel mosquitocidal agents and, in a broader sense, could establish a versatile platform for developing customized pesticides that selectively target other disease vectors as well.
Introduction
As vectors of both parasitic and viral diseases, mosquito populations are a global concern. There is a broad consensus that new agents for controlling this insect vector will be needed to ultimately eradicate diseases such as malaria, helminthiasis, hemorrhagic fevers, and encephalatidis.1–4 Anopheles gambiae, for example, is the predominant vector for malaria in Africa and contributes to the death of approximately one million people each year. 5 The use of pesticides on long-lasting impregnated nets and additional mosquitocidal strategies can successfully reduce mortality rates. 5 However, the efficacy of these strategies is limited because resistance emerges quickly,2,6 and this trait often confers cross-protection against multiple pesticides.4,7 Many pesticides also exhibit broad-spectrum toxicities. Therefore, major challenges that shape the development of new pesticides are ensuring human safety, limiting off-target effects in the ecosystem, and mitigating resistance. To confront these challenges, a high-throughput cell-based screening platform was developed. The underlying rationale for approach is straightforward: Compounds lethal to cultured mosquito cells but harmless to other Dipteran or human cells would be promising leads for the development of new mosquitocidal agents that are narrowly targeted and environmentally safe. Using this strategy, mosquito-specific, water-stable cytotoxins were found at rare frequencies (0.04%), and in at least one test case, targeted activity seen in culture predicted targeted toxicity in whole-animal studies.
Previously, Pridgeon et al 8 reported an efficient method for whole-animal screening of mosquito larva. However, relative to cell-based screening, throughput was limited, and extensive amounts of test compounds were needed. Furthermore, the platform did not distinguish nonselective from selective toxins. The proof-of-concept study outlined here was designed to confront two pivotal assumptions. First, we tested whether we could effectively use a cell-based strategy to capture species-specific cytotoxins. Second, we tested whether targeted cytotoxins found and characterized in culture would translate to whole-animal toxicity.
Materials and Methods
Cell Lines
The mosquito line 4A3A (Anopheles gambiae) was kindly provided by Mario Soberon. 9 The mosquito lines Sua 4.0 (MRA-921, Anopheles gambiae) and MSQ43 (MRA-858, Anopheles stephensi) were obtained from ATCC through the Malaria Research & Reference Reagent Resource Center (MR4), now Biodefense and Emerging Infections Research Resources Repository (BEI Resources). The two Drosophila lines Kc and S2R+ as well as the mosquito lines 4A3A and Sua 4.0 were cultured in Schneider’s media with 10% fetal bovine serum (FBS), 50 U ml–1 penicillin, and 50 µg ml–1 streptomycin at 25 °C with no CO2. The mosquito line Aag2 (Aedes aegypti) was kindly provided by Ann Fallon. 10 The mosquito lines MSQ43 and Aag2 were cultured in MEM media supplemented with 1 g glucose, 2.2 g NaHCO3, 10 mL L-glutamine 200 mM, 10 mL MEM vitamin solution 100×, 20 mL MEM nonessential amino acids, 5% FBS, 50 U ml–1 penicillin, and 50 µg ml–1 streptomycin at 29 °C with 5% CO2. The human line HCT116 was provided by Bert Vogelstein 11 and was cultured in McCoy’s media with 10% FBS, 50 U ml–1 penicillin, and 50 µg ml–1 streptomycin at 37 °C with 5% CO2. The hepatic line TPH-1 was provided by Ranjit Ray. 12 The human line T98G as well as TPH-1 were cultured in DMEM supplemented with 10% FBS, 50 U ml–1 penicillin, and 50 µg ml–1 streptomycin at 37 °C with 5% CO2.
Cell-Based Screening Platform
After a series of optimization trials, 4A3A cells were plated into 384-well plates at a density of 5000 cells/well and allowed to adhere for 4 h at 25 °C prior to automated delivery of the UT Southwestern chemical library compounds, all of which were at a final concentration of 5 µM (1% DMSO) in the assay. All assay plates were incubated at 25 °C for ~96 h. At the end of the incubation period, cell survival was measured using the CellTiter Glo system following the manufacturer’s protocol (Promega, Madison, WI). All assay plates included reference wells with either vehicle alone (columns 2 and 23) or a lethal dose of blasticidin (500 ng mL–1 in column 1). These controls enabled independent assessments of the dynamic range and assay quality of each plate as assessed using the Z′ value. 13 Plates with Z′ values <0.45 were classified as failed and repeated. From this primary screen, 176 compounds inducing potent mosquito cell lethality (Z scores ≤–3) were identified as candidates for retesting. In the second stage of the pilot study, all 176 candidates were retested in triplicate at a final compound concentration of 5 µM (1% DMSO) against the same Anopheles 4A3A cell line. In parallel, these compounds were also counterscreened in triplicate against two different Drosophila cell lines (S2R+ and Kc). The three cell lines S2R+, Kc, and 4A3A were all plated at the same density and assessed as in the original screen. All data were analyzed and quality controlled using the Screener software suite (version 10, GeneData, Inc., Basel, Switzerland).
Compound Acquisition and Assessment Strategies
Compounds were purchased anew from the commercial entities ChemBridge and ChemDivision (listed in
Stability Assay
Compounds are routinely stored as a powder, in 100% DMSO at 10 mM or 100% DMSO at 20 mM, all at −20 °C. Aliquots of the 10 mM freezer stocks were put into non–light-protecting Eppendorf tubes. In addition, these aliquots were used to make 1:50 dilutions in H2O (200 µM) in the same type of tube. These aliquots were stored at ambient temperature for a month prior to retesting.
IC50 Assay
Drosophila and mosquito cell lines were plated into 384-well plates at a density of 5000 cells/well and allowed to adhere for 4 h prior to compound addition. Mammalian cell lines were plated into 384-well plates at a density of 2000 cells/well and allowed to adhere for 24 h prior to compound addition. The Echo 555 Liquid Handler by Labcyte (Sunnyvale, CA) was used for precise automated delivery of compounds. Each compound was diluted by half-log intervals in triplicate from a top dose of 50 µM (SW1376580) or 500 µM (SW120412 and SW049753). Curve fitting was performed using the Condoseo module of the Genedata Screener (10.0.2) software suite (Genedata AG, Basel, Switzerland). The nonlinear curve- fitting algorithm in this module uses the four-parameter Hill equation. The parameters in this equation are defined as follows: the activity level at zero concentration of test compound; Sinf, the activity level at infinite concentration of compound; X, the concentration of the test compound in logarithmic units (varied in the experiment); IC50, the concentration of activity at which the activity is 50% of the maximum level; and the Hill coefficient (n), a measure of the slope at the IC50. The program provided a goodness of fit as determined by R2 values for each curve. In cases in which there was no dependence of activity on the dose of the test compound, a lower bound of the IC50 was set to the top dose in the experiment (e.g., >50 mM).
Whole-Animal Assay: Anopheles and Aedes
Mosquito eggs were placed in water to hatch and were maintained at 25 °C in a humidified room. After 24 h, the first instar larvae were counted and transferred into individual trays containing 500 mL water with 0.5% DMSO and the test compound at the required final concentration. Ground Tetramin tropical fish food was added each day in small amounts to ensure most of the food was eaten each day and adjusted as the larvae increased in size. Pupation was observed every 24 h, with pupae being counted each day and transferred to small containers, with the sex confirmed following eclosion.
Whole-Animal Assay: Drosophila
Fly food was prepared with Jazz Mix Drosophila food (Fisher Scientific, Waltham, MA). Powder was mixed with dH2O, boiled, and then placed in a 42 °C water bath to cool. Upon reaching 42 °C, 5 mL of the food was added to vials along with the compound and a dye indicator to demonstrate proper mixing. Food was allowed to cool 6 to 24 h at room temperature. Embryos were then collected and counted into groups of 50 on a bed of agar. Agar with a group of embryos on top was excised and placed into each of the food-containing vials. Two days later, the agar was removed, and unhatched embryos were counted. The vials were then monitored for pupation and eclosion rates based on the number of hatched embryos.
Results
The schematic in
Figure 1
details the workflow of the high-throughput screening platform. In the first phase, the mosquito cell line 4A3A (derived from Anopheles gambiae) was screened with a structurally diverse chemical library containing 8000 inert small molecules (referred to as the UTSW 8K library). From this primary screen, a total of 176 generic cytostatic or cytotoxic compounds were identified. In the second phase, each was retested against the same 4A3A cell line and, in parallel, counterscreened against two different Drosophila lines (S2R+ and Kc). Note that all three of these cell lines are cultured in the same media, thereby ensuring that the differences in sensitivity trace to the biology of the cells rather than the conditions under which they are cultured. The heat map in
Figure 1
illustrates compounds with activity specific for 4A3A cells. To prioritize these compounds, the stringency of the Z score thresholds was increased. In addition, legacy data on the UTSW 8K library was used to exclude compounds active against any of eight human cell lines that were previously screened. These analyses produced a panel of 16 compounds, and as shown in

A cell-based screening platform identifies targeted mosquito-specific cytotoxins. The workflow illustrated to the left is a high-throughput cell-based screening strategy designed to identify mosquitocidal cytotoxins. Lead compounds listed on the right were assessed for cross-species activity and long-term stability (legend below).
The panel of selective cytotoxins was newly acquired from a commercial vendor (except SW060760), and each was retested against the original mosquito line 4A3A and two Drosophila lines S2R+ and Kc. Thirteen compounds were confirmed for targeted activity. These were also empirically tested for activity in three human cell lines to affirm inferences based on legacy data
Quantitation of Cytotoxicity of the Three Lead Compounds. a
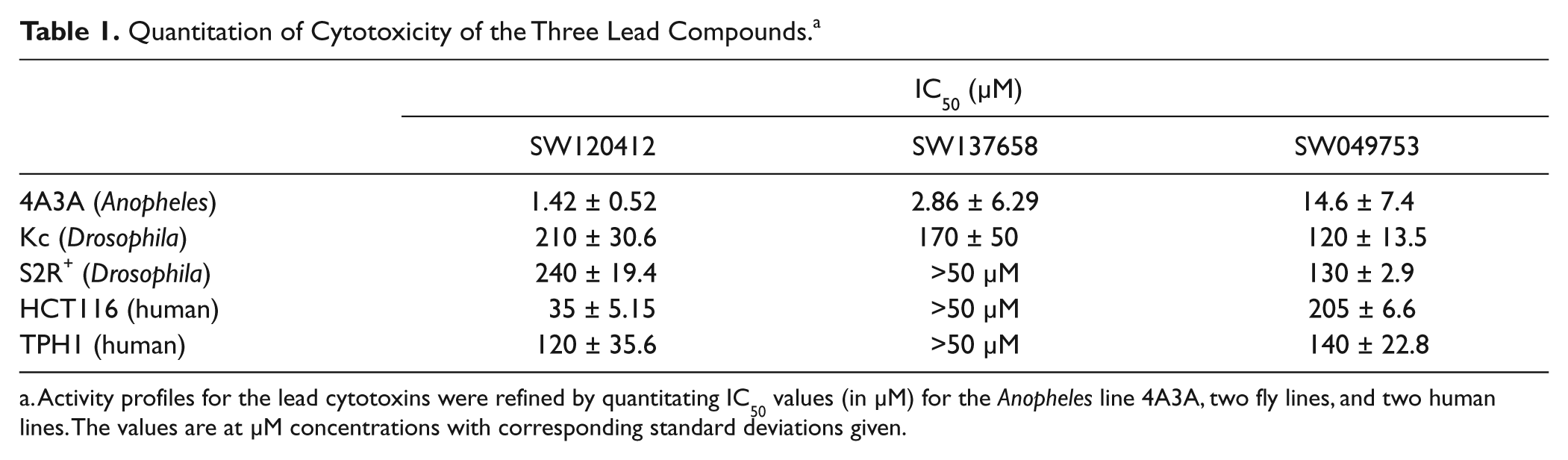
Activity profiles for the lead cytotoxins were refined by quantitating IC50 values (in µM) for the Anopheles line 4A3A, two fly lines, and two human lines. The values are at µM concentrations with corresponding standard deviations given.
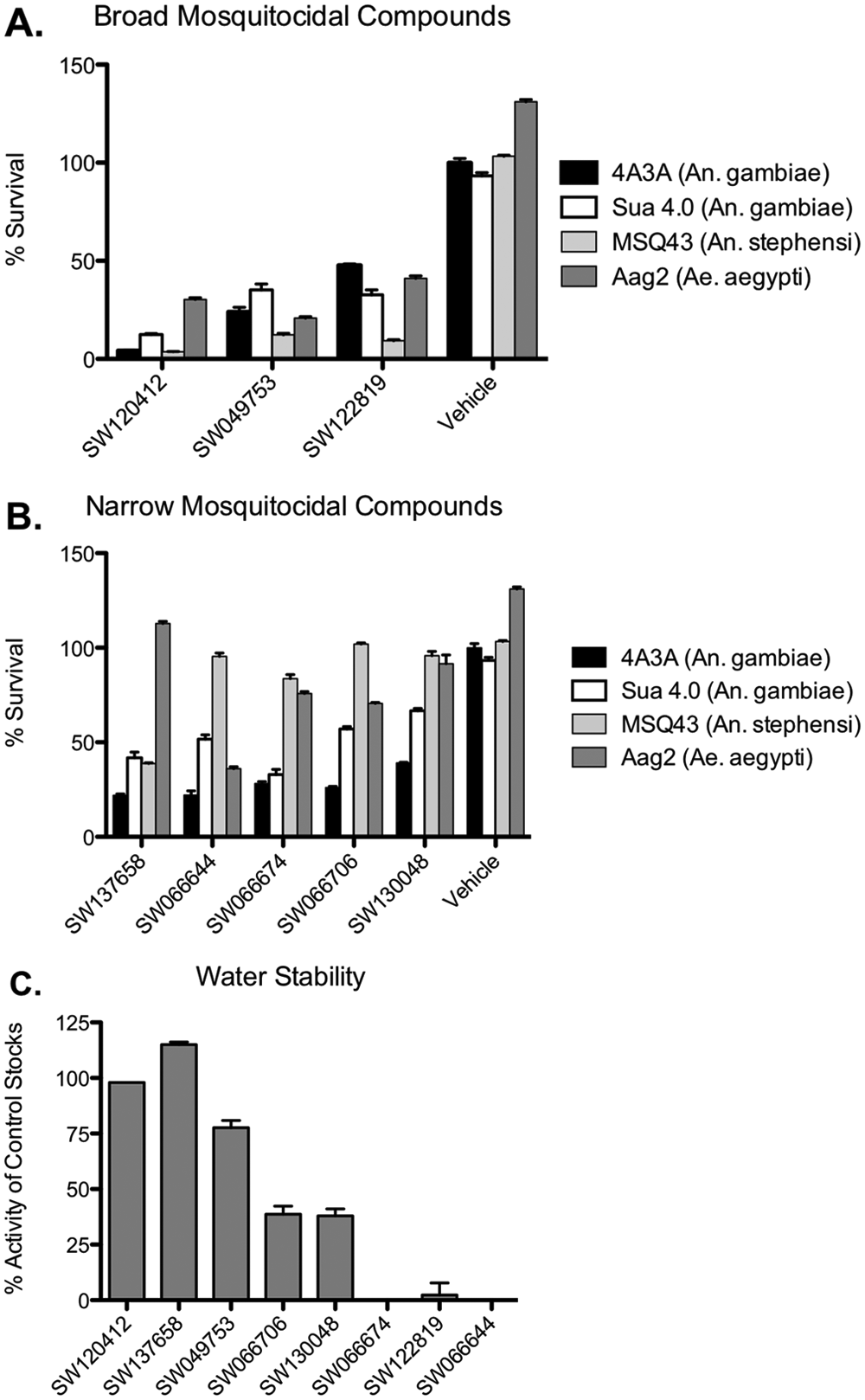
Assessment of selective cytotoxins.
Long-term stability is an important logistical consideration in the manufacturing and distribution of pesticides. Likewise, aqueous stability at ambient temperatures is particularly critical for efficacy in the field. Therefore, the remaining eight lead compounds (broad and narrow cytotoxicity) were tested after long-term storage for 1 mo at ambient temperatures. Of the eight lead compounds, two retained significant toxicity in both solvents (DMSO or H2O), and a third compound showed some reduction in cytotoxicity but retained >50% activity in water
The UTSW 8K library used in the primary screen is a representative subset of a larger institutional library at UT Southwestern containing 200 000 compounds (UTSW 200K library). To evaluate the potential for chemical optimization, compound families sharing core structures with each of the three lead cytotoxins were recovered from the UTSW 200K library and similarly tested for both toxicity and specificity
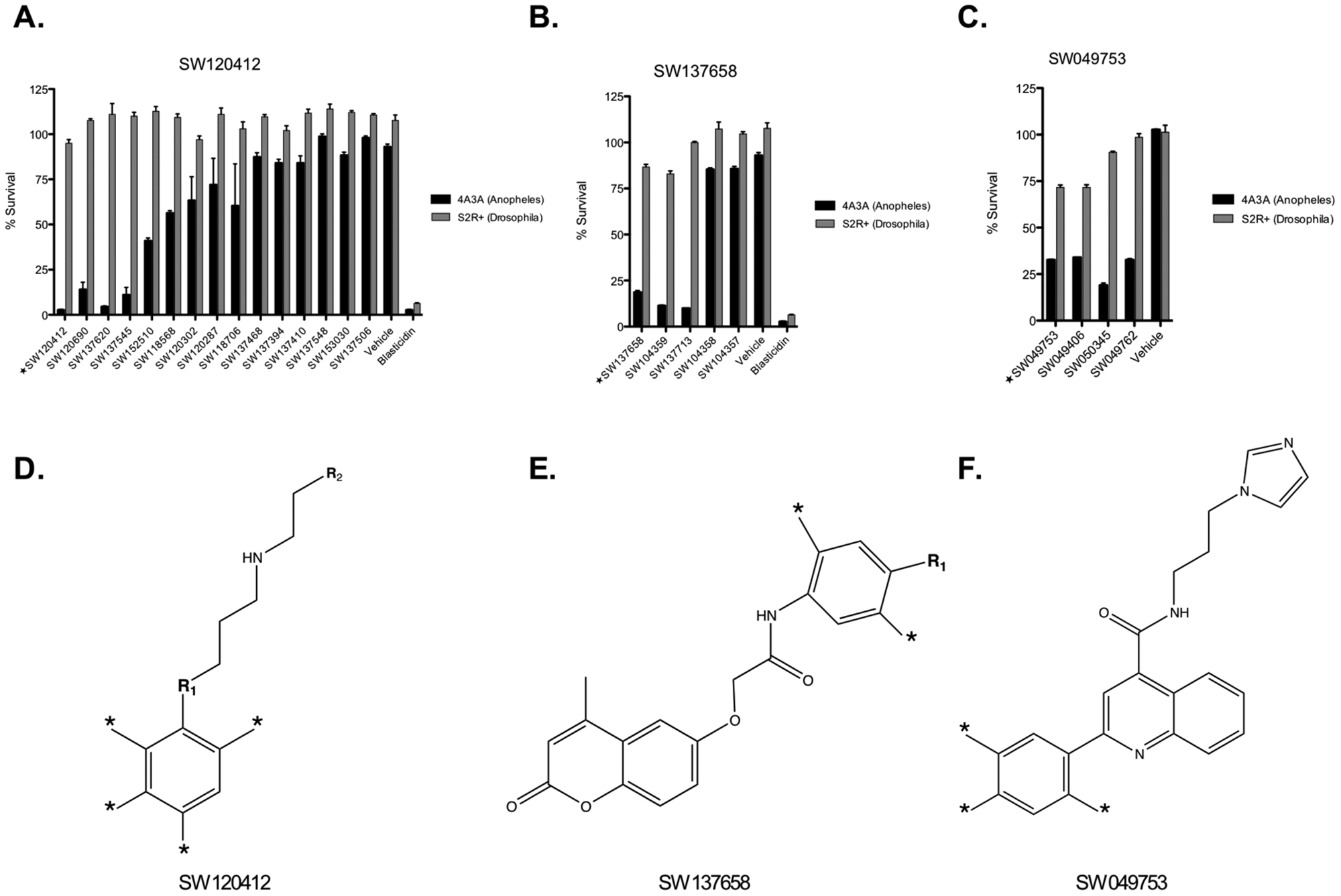
Structure activity analysis. Compound families sharing core structures in common with each lead cytotoxin were assembled and tested. (
To more precisely resolve IC50 values for the three lead cytotoxins seen in
Figure 2C
, each was assayed using an Echo 555 liquid-handling instrument to dispense half-log doses down from a top concentration of 50 µM or 500 µM. IC50 values for the mosquito line 4A3A, two Drosophila lines, and two human lines are seen in
Table 1
. Note that all showed potent selectivity windows with IC50 values representing 10- to 170-fold better activity in mosquito cells relative to the fly and human lines tested. One impressive candidate, SW137658, had an IC50 of 2.86 µM for the cell line 4A3A, but no IC50 value was detected in the human cell lines tested (see also
A premise built into the platform predicts that selection for mosquito-specific toxicity in culture should enrich for compounds with targeted toxicity at the whole-animal level. To test this prediction, the three water-stable, lead cytotoxins were assayed for larvicidal activity against Anopheles, Aedes, and Drosophila samples. SW120412 shows striking mosquito-specific toxicity
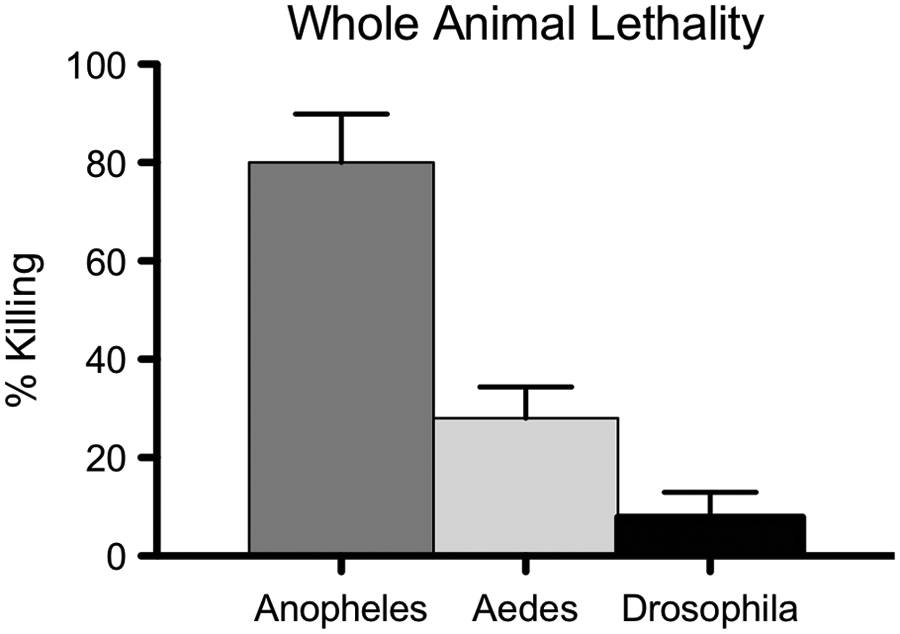
Identification of a targeted mosquitocidal compound. All three lead compounds from Table 1 were subsequently tested for larvicidal activity in whole-animal assays. SW120412 showed impressive potency against A. gambiae, whereas Drosophila larvae were unaffected for eclosion rates, even when exposed to 200 µM SW120412 (normalized eclosion rates of 109% and 97%).
Discussion
There is a broad consensus that eradication of mosquito-borne diseases will require new methods for controlling mosquitoes as resistance to one pesticide often yields cross-resistance to many pesticides.2,7 In addition, new threats such as West Nile Virus are creating epidemics in developed areas where residents are concerned that aerial spraying of pesticides could adversely affect public health and the ecosystem.
14
This proof-of-principle study demonstrates that a cell-based screening strategy can identify mosquito-specific, water-stable cytotoxins, which, in at least one case, translated to targeted killing in whole- animal assays (
Supplemental Material
Supplemental_JBS – Supplemental material for A Cell-Based Screening Platform Identifies Novel Mosquitocidal Toxins
Supplemental material, Supplemental_JBS for A Cell-Based Screening Platform Identifies Novel Mosquitocidal Toxins by Melissa A. O’Neal, Bruce A. Posner, Craig J. Coates and John M. Abrams in SLAS Discovery
Footnotes
Acknowledgements
We thank Steven McKnight and the HTS Core at UT Southwestern (especially Janie Life, Shuguang Wei, and Chun Hui Bu) for their support of the chemical screen. The HTS Core receives support from NCI (1P01CA95471-09), the Simmons Cancer Center (1P30CA142543-01), and UT Southwestern.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants to J.M.A. from the Welch Foundation (grant No. I-1727), the Gates Foundation (53134), and the National Institute of General Medical Sciences (R01GM072124).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
