Abstract
The cystic fibrosis transmembrane conductance regulator (CFTR) protein is an important regulator of ion transport and fluid secretion in humans. Mutations to CFTR cause cystic fibrosis, which is a common recessive genetic disorder in Caucasians. Involvement of CFTR has been noted in other important diseases, such as secretory diarrhea and polycystic kidney disease. The assays to monitor CFTR function that have been described to date either are complicated or require specialized instrumentation and training for execution. In this report, we describe a rapid FlexStation-based membrane potential assay to monitor CFTR function. In this assay, agonist-mediated activation of CFTR results in membrane depolarization that can be monitored using a fluorescent membrane potential probe. Availability of a simple mix-and-read assay to monitor the function of this important protein might accelerate the discovery of CFTR ligands to study a variety of conditions.
Introduction
Cystic fibrosis (CF) is one of the most common autosomal recessive genetic disorders in Caucasians, with a reported incidence of 1 out of every 3000 live births in the United States. 1 This disease is caused by mutations within a single gene, coding for the cystic fibrosis transmembrane conductance regulator (CFTR) protein. The phenotypic hallmark of CF is chronic lung infection and associated inflammation from opportunistic microbes such as Pseudomonas aeruginosa, Haemophilus influenzae, and Staphylococcus aureus. 1 This eventually leads to deterioration of lung function and death in most patients with CF. 1 Although initially characterized as an anion (chloride) channel, it is now clear that the physiological role of CFTR extends beyond ion transport to the regulation of several important cytological processes. 1 There is emerging evidence to suggest that CFTR inhibitors may find additional applications beyond CF in diseases such as secretory diarrhea and autosomal dominant polycystic kidney disease, where cAMP-mediated ion flux and water transport regulated by CFTR play a vital role. Inhibitors of CFTR may be useful in the development of pharmacological models of CF and to understand the function of this protein. Very few compounds inhibit CFTR with high potency and specificity. Furthermore, some of these described compounds need further preclinical validation. 2
The search for CFTR inhibitors and activators has been hampered by the lack of readily available and easy to perform assays to measure CFTR function in a microplate format. The earliest approaches to study CFTR involved radioisotopes in flux assays and electrophysiological methods. The former approach has been all but abandoned due to the hazards involving radioisotope use. Electrophysiological assays require specialized instrumentation and trained personnel, and they are typically low throughput. Fluorescent halide probes, while available, are not ideal for studying CFTR activity for a number of reasons, including the need of multiple buffers for introduction into cells and during assay performance, poor retention in cells, high production costs, poor quantum yields, long incubation and assay times, and toxicity.
A few assays have been described over the years to measure CFTR function. 3 Jayaraman and colleagues 4 reported the use of a mutated version of yellow fluorescent protein (mYFP) to monitor CFTR function in double-transfected cells expressing mYFP and CFTR. Limitations of this assay include the need to wash cells multiple times prior to assay, requirement of different buffers for assay execution, and the need to use surrogate ions (iodide instead of chloride) for best assay performance. Recently, Vertex Pharmaceuticals (Cambridge, MA) reported a fluorescence resonance energy transfer (FRET)–based assay to monitor ΔF508-CFTR localization and function. 5 This assay also requires a two-buffer system during assay performance with multiple wash steps in between. Furthermore, FRET-based assays typically produce a low signal-to-noise ratio, often requiring data transformation, which can affect data quality. 6 Three additional assays have been described recently to monitor CFTR and ΔF508-CFTR (a common mutant form of CFTR) localization and function.7–9 One of these approaches has limited utility as the assay was designed to monitor localization of ΔF508-CFTR exclusively. 8 The other assays require specialized instrumentation; need considerable sample manipulation prior, during, and after the assay; and are difficult to perform in a general lab setting.7,9 Also, with the exception of the high-throughput screening (HTS) assay developed by Jayaraman et al., 4 none of the other assays has been used to identify CFTR inhibitors, and their utility in this regard remains untested.
Past studies indicate that CFTR activation results in altered membrane potential, which can be detected using an appropriate probe. 10 In this report, a rapid FlexStation-based fluorescent membrane potential (FMP) assay to monitor CFTR function in a microplate format is described. The assay can be performed using a commercially available FMP probe formulation, which represents a significant improvement over existing approaches to monitor CFTR activity. These probe formulations are typically bis-barbituric acid oxonols, along with a lipohilic quencher dye. The probe enters cells and binds to intracellular proteins or membranes and exhibits enhanced fluorescence. Increased depolarization results in further influx of the probe into cells and increase in fluorescence. Conversely, hyperpolarization is indicated by a decrease in fluorescence. In the proposed CFTR assay, agonist-mediated activation of CFTR results in membrane depolarization. 10 This increases FMP probe-mediated cellular fluorescence that can be measured using a plate reader in a kinetic assay, which is demonstrated in this report.
Materials and Methods
Materials
All chemicals, including cell culture media, supplies, and plasticware, were purchased from Sigma-Aldrich (St. Louis, MO) unless otherwise specified. The CFTR inhibitors N-(2-Naphthalenyl)-((3,5-dibromo-2,4-dihydroxyphenyl)methylene)glycine hydrazide (GlyH-101) and 5-[(4-Carboxyphenyl)methylene]-2-thioxo-3-[(3-trifluoromethyl)phenyl]-4-thiazolidinone (CFinh-172) were generously donated by the North American Cystic Fibrosis Foundation (Bethesda, MD). Both these compounds were originally developed by Dr. Alan Verkman at the University of California at San Francisco. 11 Of these, GlyH-101 is a cell-permeable glycinyl hydrazone compound that acts as a potent, selective, and reversible open-channel blocker of CFTR with intermediate speed (<1 min; 95% inhibition at 50 µM; Ki = 4.3 µM in CFTR-expressing FRT cells for apical membrane Cl– current), and CFinh-172 is a cell-permeable 2-thio-4-thiazolidinone compound that acts as a potent, reversible, rapid, and voltage-independent inhibitor of CFTR (Ki ~300 nM). 11
Cell Culture and Treatment
Mammalian expression plasmids for GFP-CFTR and GFP–ΔF508-CFTR were donated by Dr. Bruce Stanton (Dartmouth Medical School, Hanover, NH). 12 The complementary DNA (cDNA) inserts were re-cloned into the lentiviral pLENTI6 vector backbone (Invitrogen, Carlsbad, CA) and sequenced. The vectors were used to transfect HEK293 cells obtained from ATCC (Manassas, VA). Stable clones were isolated under selection in media containing blasticidin (5 µg/mL). Clones were tested using immunoblotting 13 for expression of target proteins, and a single clone expressing either protein was used for subsequent studies.
Immunoblotting and Immunofluorescence
Immunoblotting of CFTR was performed using the M3A7 antibody as previously described. 13 For immunofluorescence studies, cells were grown for 72 h and then fixed lightly using 1% formalin. Cells were then stained with Texas red–conjugated wheat germ agglutinin (WGA; Molecular Probes, Eugene, OR) at a concentration of 0.1 mg/mL in phosphate-buffered saline (PBS) for 5 min. Cells were washed three times in an excess of PBS and imaged under ×40 magnification using a Discovery 1 imaging system (Molecular Devices, Sunnyvale, CA) using appropriate filter sets. Images were processed using ImageJ (National Institutes of Health [NIH], Bethesda, MD).
Membrane Potential Assay
For a typical assay, parental HEK293, HEK293/CFTR, and HEK293/ΔF508-CFTR cells were detached using nonenzymatic means and plated in optical-bottom black 96-well plates coated with poly-D-lysine at a concentration of 5000 cells per well in 100 µL Dulbecco’s modified Eagle’s medium (DMEM)/F12 media containing 2% fetal bovine serum (FBS) and antibiotics. After the cells reached confluence (typically 72 h), cells were loaded by adding 100 µL FLIPR Blue FMP dye (Molecular Devices) dissolved in cell culture media (2% FBS) to the preexisting media for 30 min at 37 °C. All agonists and inhibitors were made in the dye solution. For inhibitor assays, cells were incubated for an additional 15 min before stimulation with the agonist cocktail. A cocktail of CFTR agonists containing 8-cpt–cAMP and genistein was directly added to the plate at a 10× concentration. Cells were analyzed in a FlexStation II instrument (Molecular Devices) at ex 530/em 565. Baseline was established by reading the plates for ~15 s. Readings were taken at ~1.5-s intervals. For the assay validation exercise, the NIH Clinical Collection of ~400 pharmacologically active compounds was used (http://www.nihclinicalcollection.com/). Compounds were added to each individual well at a 10× concentration and preincubated for 15 min prior to stimulation with the agonist cocktail (15 µM 8-cpt–cAMP + 10 µM genistein). The concentration of 8-cpt–cAMP was at ~EC75 of CFTR activation. All test compounds were screened at 10 µM for CFTR inhibition. Intra-assay variability (standard deviation) was typically below 20%. Signal-to-noise ratio was between 4- to 6-fold for all assays reported. Baseline fluorescence was automatically normalized for all assays using the Softmax software program provided with FlexStation devices (Molecular Devices). All data were analyzed using GraphPad Prism Version 6.0 (GraphPad Software, La Jolla, CA).
Results and Discussion
A robust assay to monitor CFTR function has been developed. To attain this goal, HEK293 cell lines stably expressing CFTR and ΔF508-CFTR ( Fig. 1 ) were engineered. The choice of HEK293 cells was dictated by past studies indicating that CFTR expression, function, and localization were not compromised in these cells. 14 The cDNAs encoding GFP-tagged CFTR and ΔF508-CFTR were cloned into pLENTI6 vectors (Invitrogen), sequenced, and transfected into HEK293 cells. Clonal cell lines were selected using antibiotic resistance and analyzed for CFTR or ΔF508-CFTR expression using immunoblotting, and two cell lines were designated for further evaluation and assay development. Localization of CFTR and ΔF508-CFTR was verified using immunofluorescence ( Fig. 1A ). Immunofluorescence analyses of each cell type demonstrated that a population of GFP-CFTR localized to the plasma membrane with characteristic rim-like fluorescence (top left panel) as expected, whereas GFP–ΔF508-CFTR expression was generally restricted to the cytoplasm (bottom left panel). Fixed cells were also stained with Texas red–conjugated WGA to delineate cellular membranes (middle panels, pseudocolored red). Merged images are shown in the far right panels (pseudocolored yellow). Co-localization of GFP-CFTR with the plasma membrane was clearly noted. By contrast, GFP–ΔF508-CFTR demonstrated little to no co-localization with the plasma membrane. Figure 1B demonstrates expression of CFTR and ΔF508-CFTR in engineered HEK cells. In immunoblot analysis, maturation of CFTR was noted (appearance of fully glycosylated band C) versus restricted maturation of ΔF508-CFTR (band B) in agreement with past literature. 14
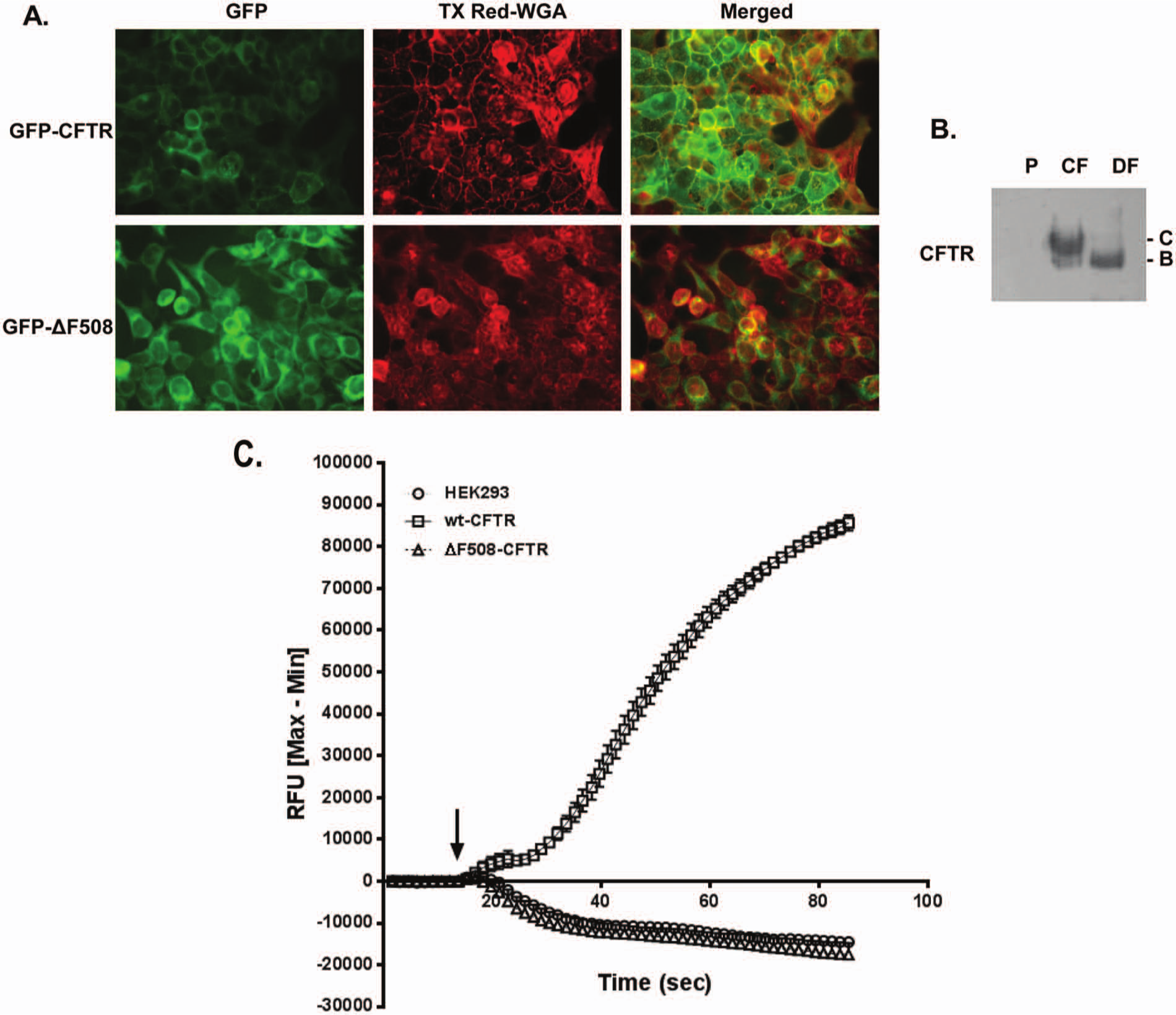
Membrane potential assay to measure cystic fibrosis transmembrane conductance regulator (CFTR) function. (
Having created stable cells expressing CFTR or ΔF508-CFTR, feasibility studies were performed next to monitor CFTR function using the membrane potential assay. Based on past literature and through preliminary studies, the FLIPR Blue membrane potential dye (Molecular Devices) was deemed most suitable for further evaluation and assay development. As demonstrated in Figure 1C , stimulation of CFTR cells with an agonist cocktail made of 8-cpt–cAMP (10 µM) and genistein (10 µM) led to membrane depolarization, which resulted in increased fluorescence. As expected, an increase in fluorescence was noted only in HEK cells expressing CFTR but not in parental HEK cells or those expressing ΔF508-CFTR, as functional CFTR was absent in the latter two cell types. In fact, mild hyperpolarization was noted in these two cell types possibly due to nonspecific activation of other ion channels or receptors by the agonist cocktail. These studies indicated that CFTR was correctly localized in HEK cells and was functional.
Further characterization of the HEK293/CFTR cell line was performed through concentration-response studies with the CFTR agonist 8-cpt–cAMP in the presence of genistein (10 µM) and also by blocking CFTR activity by using the CFTR antagonist CFinh-172. As demonstrated in Figure 2 , CFTRinh-172 blocked activation of CFTR upon stimulation with the agonist cocktail in a concentration-dependent fashion. Similar data were obtained with GlyH-101, another CFTR inhibitor (data not shown).
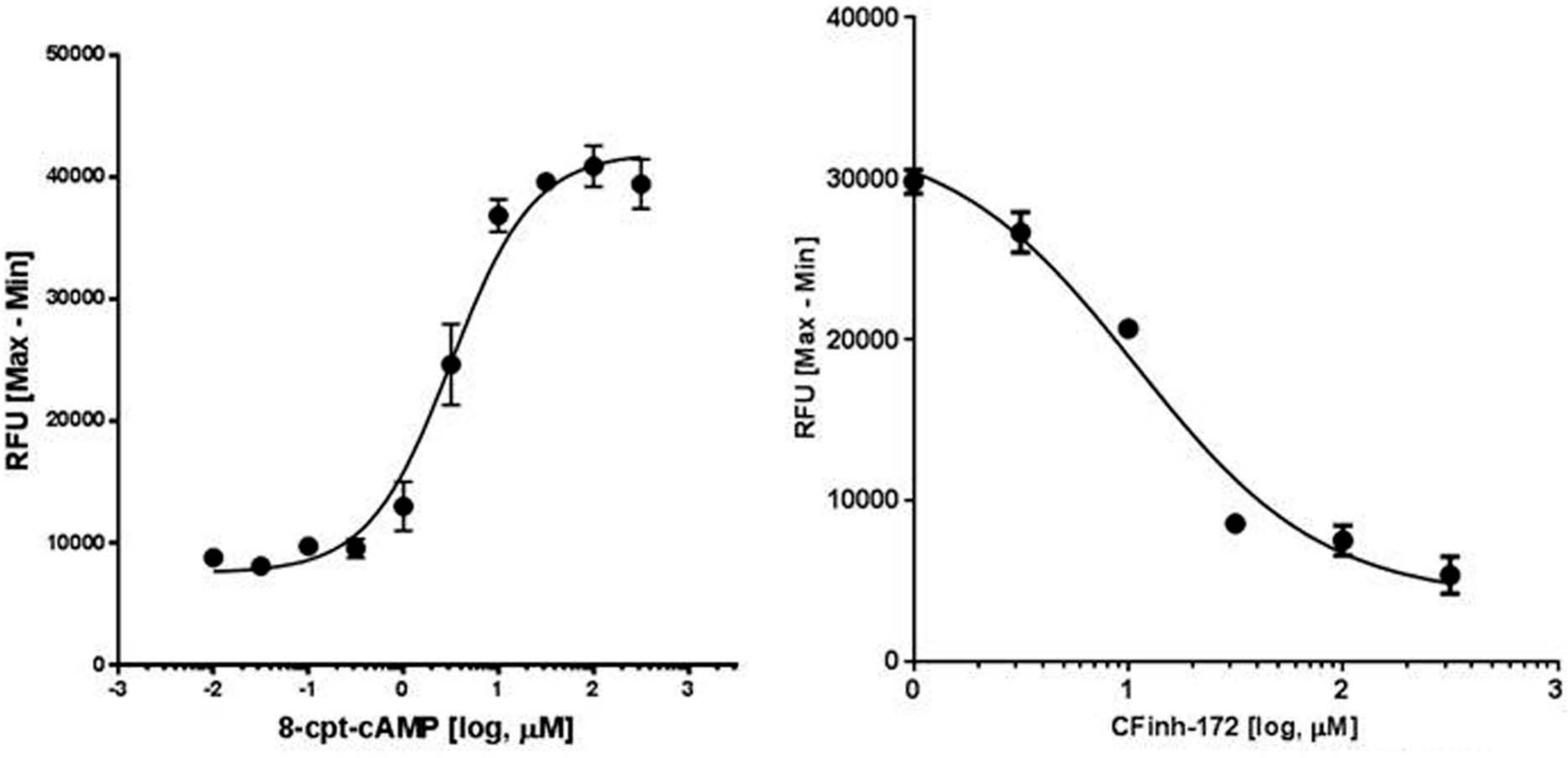
Activation and inhibition of cystic fibrosis transmembrane conductance regulator (CFTR). (Left) HEK293/CFTR cells (n = 4) were stimulated with various concentrations of the agonist cocktail as described in the Materials and Methods section, and CFTR-mediated depolarization was measured. Concentration-dependent increase in fluorescence was noted with 8-cpt–cAMP stimulation of CFTR (EC50 = 3.2 µM). (Right) Preincubation of HEK293/CFTR cells (n = 3) with CFinh-172 inhibited CFTR activity and agonist-mediated depolarization of cells in a concentration-dependent fashion (IC50 = 10.9 µM). RFU, relative fluorescence units.
Next the sensitivity of this assay to DMSO, which is a commonly used solvent, was tested. For these assays, HEK293/CFTR cells were incubated with various concentrations of DMSO to 1% and then stimulated with the agonist cocktail (8-cpt–cAMP + genistein) at increasing concentrations. Concentrations of DMSO up to 1% had a minimal effect on assay performance, as noted in Figure 3 . Thus, this assay is suitable for routine screening of compounds dissolved in DMSO.
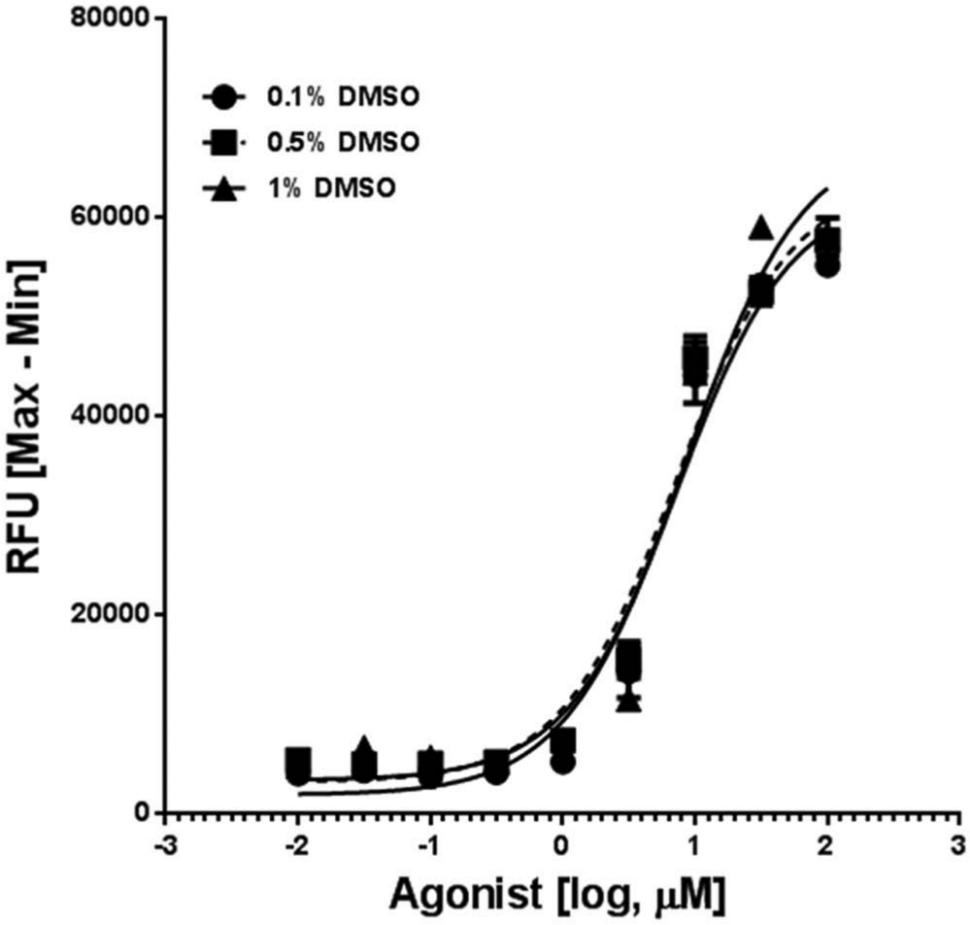
Tolerance of the cystic fibrosis transmembrane conductance regulator (CFTR) membrane potential assay to DMSO. Wells of HEK293/CFTR cells (n = 3) were incubated in duplicate with various concentrations of DMSO up to 1% final concentration and then stimulated with increasing concentrations of an agonist cocktail comprising of 8-cpt–cAMP and genistein as described in the Materials and Methods section. CFTR-mediated membrane depolarization was not significantly affected by DMSO at the concentrations tested (EC50 = 7.2 µM, 7.2 µM, and 8.6 µM at 0.1%, 0.5%, and 1% DMSO, respectively). RFU, relative fluorescence units.
Finally, a small set of pharmacologically active compounds (NIH Clinical Collection, ~400 compounds) was tested for CFTR inhibitory activity as a validation exercise. Compounds that inhibited CFTR activity by >50% were identified. Of these compounds, some were summarily rejected as they were known cytotoxic agents. This produced a total of 21 compounds for further validation. These 21 compounds were subsequently retested to rule out false-positive hits. Retesting led to confirmation of 4 compounds as hits. These compounds are listed in Table 1 . Unfortunately, none of these compounds were potent inhibitors, and only nitrendipine showed concentration-dependent CFTR inhibition at concentrations tested (IC50 >20 µM). Interestingly, nitrendipine is an L-type calcium channel blocker, and one of the adverse effects associated with its use in patients is edema. Therefore, these data emphasize the need to screen a larger collection of compounds to identify new scaffolds for optimization.
Initial Hits for Cystic Fibrosis Transmembrane Conductance Regulator Inhibition.

In summary, a simple membrane potential assay to monitor function of CFTR has been developed. In this assay, the biological response of CFTR to an agonist as reflected through membrane depolarization is measured using an FMP dye in a “no-wash” format. This was possible due to two important factors. First, high-quality CFTR-expressing cells were created that produce a strong anion flux upon agonist stimulation without the need to establish a concentration gradient. Second, the FMP probe formulation from Molecular Devices proposed for this assay is one of the most sensitive reagents among those commercially available, as determined through independent testing. 15 Most of the assays to monitor CFTR function that have been reported to date require considerable technical expertise and/or sample manipulation to get acceptable data.4,5 The most commonly used fluorometric assays of this protein require a chloride-containing buffer for equilibration and a chloride-free buffer to artificially induce a chloride gradient followed by stimulation with an agonist cocktail to activate chloride transport through CFTR.4,5 The presence of the chloride gradient amplifies the signal in cells that express modest levels of CFTR. This two-buffer system is necessary to get an acceptable signal-to-noise ratio but also renders the assays difficult to perform and may compromise data quality as well. The technical advancement reported here represents a clear improvement over previously reported methods as a single buffer system can be effectively used to monitor CFTR function.
This assay was developed using a FlexStation plate reader capable of automated liquid addition and kinetic assays. However, in theory, this assay could be further simplified and implemented in a general laboratory equipped with a dye-compatible fluorescent plate reader. In such a scenario, after prereading the baseline, the cells could be stimulated with an agonist cocktail and a single reading could be taken 2 min or so after stimulation. This approach might transform the kinetic assay described in this article into a simple end-point assay. Finally, further miniaturization of this assay to a higher density format (384 wells or higher) will allow for large-scale HTS campaigns to identify CFTR activators and inhibitors. Assay miniaturization and optimization for HTS are planned as future goals of this project.
Footnotes
Acknowledgements
We thank Mr. Tim Morris for excellent technical assistance during cell line development.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors declared receipt of the following financial support for the research, authorship, and/or publication of this article: Work was supported by internal funding from RTI International and 1R21NS064848-01A1 from NIH.
