Abstract
Two-pore domain K+ (K2P) channels are thought to be druggable targets. However, only a few agents specific for K2P channels have been identified, presumably due to the lack of an efficient screening system. To develop a new high-throughput screening (HTS) system targeting these channels, we have established a HEK293-based “test cell” expressing a mutated Na+ channel (Nav1.5) with markedly slowed inactivation, as well as a K+ channel (Kir2.1) that sets the membrane potential quite negative, close to K+ equilibrium potential. We found in this system that Kir2.1 block by 100 μM Ba2+ application consistently elicited a large depolarization like a long-lasting action potential. This maneuver resulted in cell death, presumably due to the sustained Na+ influx. When either the TWIK-related acid-sensitive K+ (TASK)-1 or TASK-3 channel was expressed in the test cells, Ba2+-induced cell death was markedly weakened. Stronger activation of TASK-1 by extracellular acidification further decreased the cell death. In contrast, the presence of K2P channel blockers enhanced cell death. IC50 values for TASK-1 and/or TASK-3 blockers acquired by measurements of relative cell viability were comparable to those obtained using patch-clamp recordings. Both blockers and openers of K2P channels can be accurately assessed with high efficiency and throughput by this novel HTS system.
Introduction
Two-pore domain K+ (K2P) channels generate significant time-independent or background currents and play key roles in the regulation of cellular functions by modulating the resting membrane potentials (RMPs) in variety of cell types. 1 The K2P channel family consists of 6 subfamilies and total 15 members. These K+ channels are regulated by a wide variety of voltage-independent factors, including physical stimuli and bioactive substances. 1 Recent studies on K2P channels have also revealed their possible involvement in pathophysiological conditions and chronic diseases, such as cardiovascular, neurological, and metabolic disorders.2–7 Accordingly, K2P channels are considered to be attractive drug targets. However, very few agents specific to K2P channels have been reported so far. One of the reasons for this may be a lack of appropriate high-throughput screening (HTS) systems for K2P channel modulators.
At present, several different HTS methods for drug discovery targeting on K+ channels, for example, thallium flux assay,8–10 potential-sensitive fluorescence assay, 11 rubidium-flux measurement, 12 and electrophysiological assays, 13 are available. However, none of them completely fulfill the requirements for high-throughput assays. Some of them do not convincingly monitor the appropriate movement of K+ ions through specific channels in the plasma membrane and the resulting changes in membrane potential. An automated patch-clamp platform certainly enables the direct measurement of membrane currents. For many applications, this method possibly costs too much to use for the primary screening of more than 100,000 test compounds.
Previously, we have established a recombinant cell line derived from HEK293 cells (IFM/Q3+Kir2.1; also called test cell), 14 in which a single action potential induces cell death. This test cell is based on the ability to express stably the mutated Na+ channels (IFM/Q3) in which a large part of inactivation is markedly slowed or removed. These Na+ channels are co-expressed with wild-type inward rectifier K+ channels (Kir2.1). In this cell line, electrical stimulation (ES) gives rise to sustained opening of IFM/Q3 and Na+ overload, which leads to cell death. The extent of this cell death can be easily measured by one of universal methods, such as MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) assay. Additional co-expression of voltage-dependent K+ (Kv) channels including hERG, Kv1.5, or Kv1.3 to the test cell shortened the ES-induced action potential and suppressed cell death.14,15 In the presence of Kv channel blockers, ES evoked prolonged action potential and cell death again. Thus, this approach succeeded in offering accurate and reproducible evaluations of the efficiency of Kv channel blockers without using any electrophysiological techniques.14,15 One drawback of this system is that additional equipment for ES of cells in each well of a 96- or 384-well plate is required to activate voltage-dependent channels effectively.
The present study was undertaken to simplify this assay procedure. Here, we used Ba2+ instead of ES in an effect to include cell excitation as a part of the HTS system that can be used for K2P channel drug discovery. We have succeeded in developing an ES-free and automated screening system that provides reliable evaluation of compounds acting on background K2P channels, such as TWIK-related acid-sensitive K+ (TASK)-1 and TASK-3 channels, with high efficiency.
Materials and Methods
Establishment of the Test Cell
Mutated human cardiac Na+ channel Nav1.5 (IFM/Q3) was made by substituting 1485IFM1487 in the III–IV interdomain linker (NM_198056.2) with QQQ (Q3) as described previously. 14 IFM/Q3 and human Kir2.1 (NM_000891.2) were subcloned into pcDNA3.1/Neo(+) and pcDNA3.1(+)/Zeo (Invitrogen, Carlsbad, CA), respectively. HEK293 cells were maintained at 37 °C in 5% CO2 with high-glucose Dulbecco’s modified Eagle’s medium (DMEM) (FUJIFILM Wako Pure Chemical, Osaka, Japan) containing 10% fetal bovine serum (FBS; Sigma-Aldrich, St. Louis, MO), 100 U/ mL penicillin (FUJIFILM Wako Pure Chemical), and 0.1 mg/mL streptomycin (Meiji Seika, Tokyo, Japan). Double stable expression of IFM/Q3 and Kir2.1 was achieved with calcium phosphate co-precipitation transfection techniques and following G418 (FUJIFILM Wako Pure Chemical)/zeocin (Invitrogen) selection.
Preparation of Recombinant Viral Vectors
Recombinant baculoviral vectors were constructed by using plasmid vectors derived from pFastBac1 (Thermo Fisher Scientific, Waltham, MA), which contains CMV-IE promoter, woodchuck hepatitis virus posttranscriptional regulatory element (WPRE), and vesicular stomatitis virus G protein (VSV-G) to achieve high expression in mammalian cells. 16 Human TASK-1 (NM_002246) Δi20 mutant, 16 human TASK-3 (NM_001282534), or GFP from pAcGFP-N1 (Clontech, Mountain View, CA) was inserted into the baculoviral vectors. Recombinant baculoviruses were generated by using the Bac-to-Bac system (Thermo Fisher Scientific). Recombinant viruses were amplified in Sf9 cells (Thermo Fisher Scientific). Amplified viruses were harvested from the culture supernatant and the debris of cells was removed through centrifugation (1000g; 10 min). The supernatant was used for infection. The expression of K2P channels was achieved by the addition of the supernatants at the amount a 10% of the culture medium. The efficiency of infection was estimated by membrane current recordings using the whole-cell patch-clamp techniques. The currents of K2P channels were confirmed by their electrophysiological and pharmacological characteristics in 16 cells out of 16 from two sets of the infection experiments.
Electrophysiological Recordings
The whole-cell patch clamp was performed using the CEZ-2400 amplifier (Nihon Kohden, Tokyo, Japan) and a data acquisition/analysis system (Digidata 1440A and pClamp software v10; Molecular Devices, Foster City, CA), as has been reported previously. 14 The sampling rate was 10 kHz. Cells were grown on small pieces of coverslips in a petri dish and transferred to a recording bath chamber filled with a bathing solution on the stage of an inverted microscope (TE300; Nikon, Tokyo, Japan). Cells were continuously perfused with the bathing solution. Whole-cell recordings were carried out at room temperature. The bathing solution had an ionic composition of 137 mM NaCl, 5.9 mM KCl, 2.2 mM CaCl2, 1.2 mM MgCl2, 14 mM glucose, and 10 mM HEPES. The pH of the bathing solution was adjusted to 7.4 or 8.4 with NaOH. The pipette-filling solution for whole-cell recordings consisted of 140 mM KCl, 4 mM MgCl2, 10 mM HEPES, 2 mM Na2ATP, and 0.05 mM EGTA. The pH of the solution was adjusted to 7.2 with KOH.
MTT Assay
Cell viability was quantified by the colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazoliumbromide (MTT; Sigma-Aldrich) assay as described previously. 14 The stock MTT solution was prepared in phosphate-buffered saline at the concentration of 5 mg/mL. Cells were seeded onto 24-well plates (40,000–50,000 cells/well) and cultured for 48–72 h. DMEM supplemented with 10% FBS and 300–600 µM mexiletine (Sigma-Aldrich) was used to enhance mutated Nav1.5 expression.17,18 Previous reports have shown that the incubation with mexiletine partially rescues the defective expression of Nav1.5.17,18 When TASK-1 or TASK-3 was transiently introduced to the test cells by baculoviral vectors, the virus was applied to the wells approximately 24 h before the experiments. On the day of assay, the cells were washed with the bathing solution, and test compounds were added to the wells. After 10 min of incubation at room temperature, cells were treated with 200 μM BaCl2 and incubated for 12–16 h at 37 °C. MTT solution was added at the amount of 10% of the culture medium (10 μL/100 μL medium), and then the plates were incubated for 4 h at 37 °C. After 4 h, a solubilization solution containing 20 w/v% sodium dodecyl sulfate (SDS) and 50 v/v% N,N-dimethylformamide was added to the well at the same amount of the medium, and the plate was incubated for 4 h at 37 °C. When the solubilization was completed, these plates were analyzed for absorption on a Multiscan JX (v1.1 system; Thermo Labsystems, Beverly, MA), using a test wave length of 595 nm and a reference wavelength of 650 nm.
Membrane Potential Measurements Using Voltage-Sensitive Fluorescent Dye
Membrane potential changes were measured using DiBAC4(3) (DOJINDO, Kumamoto, Japan) as has been reported previously. 14 Data were collected and analyzed using a laser scanning confocal fluorescent microscope (A1R, Nikon). The excitation wavelength was 488 nm. The emission light was collected by a band-pass filter (DM514/BA525–555). The sampling interval of DiBAC4(3) fluorescence measurements was 5 s. The data were acquired as the relative fluorescent intensity of DiBAC4(3), taking that in 140 mM K+ solution as unity (ratio of F/F140K).
Automated Assay Using 96-Well Plates
Cells were seeded onto poly-
Randomized Assay Test
Five compounds, PK-THPP, ML365, losartan potassium, atropine sulfate, and NS309, were initially diluted in dimethyl sulfoxide (DMSO, FUJIFILM Wako Pure Chemical) to a stock concentration of 1 mM. These compounds were assigned random numbers by another person who did not perform any other steps of the experiments for blind test assay. The efficacy of these randomized compounds against TASK-3 at a concentration of 1 μM was evaluated without knowing which they were.
Reagents
Barium chloride was obtained from FUJIFILM Wako Pure Chemical. PK-THPP and ML365 were obtained from Tocris Bioscience (Bristol, UK). Losartan potassium was obtained from Tokyo Chemical Industry (Tokyo, Japan). Atropine sulfate was obtained from Nacalai Tesque (Kyoto, Japan). NS309 was obtained from Alomone Labs (Jerusalem, Israel). These compounds were dissolved with distilled water or DMSO.
Statistics
Aggregated data sets are shown as mean ± SEM. In this study, n is the number of independent cells in the patch-clamp and imaging experiments, and the number of wells in the MTT assay using 24-well or 96-well plates. N is the number of independent experiments. The statistical significance between two groups and among multiple groups was evaluated using Student’s t test and Tukey’s test after the F test and one-way analysis of variance, respectively. The Z′ factor was calculated as follows: Z′ factor = 1 – 3 (SD of positive controls + SD of negative controls)/(mean of positive controls − mean of negative controls). The concentration–inhibition relationship was determined by measuring the relative viability of cells and fitting a curve to each set of data based on the Hill equation: y = 1/[1 + (D/[IC50])n], where D is the drug concentration, IC50 is the half-maximal inhibition, and n is the Hill coefficient.
Results
Block of Kir2.1 by Ba2+ Depolarizes Test Cells Expressing IFM/QQQ
In the test cells (IFM/Q3+Kir2.1 cells), the RMP measured using the current clamp mode of a whole-cell patch clamp was –78.4 ± 1.6 mV (n = 6) ( Fig. 1A,B ). As expected, this was close to the reversal potential for K+ (–80.9 mV) calculated based on the experimental conditions. Application of 100 μM Ba2+ consistently elicited a large and persistent membrane depolarization including an overshoot, somewhat resembling a long-lasting action potential. The peak membrane potential at the overshoot was +40.0 ± 2.1 mV (n = 6) ( Fig. 1A,B ), approaching the calculated reversal potential of Na+ (+90.8 mV). As shown, the membrane potential remained at +13.4 ± 3.9 mV even 30 s after the addition of Ba2+. These results show that the block of Kir2.1 by submillimolar Ba2+ evokes large and long-lasting membrane depolarization, and this results in persistent opening of IFM/Q3 Na+ channels.
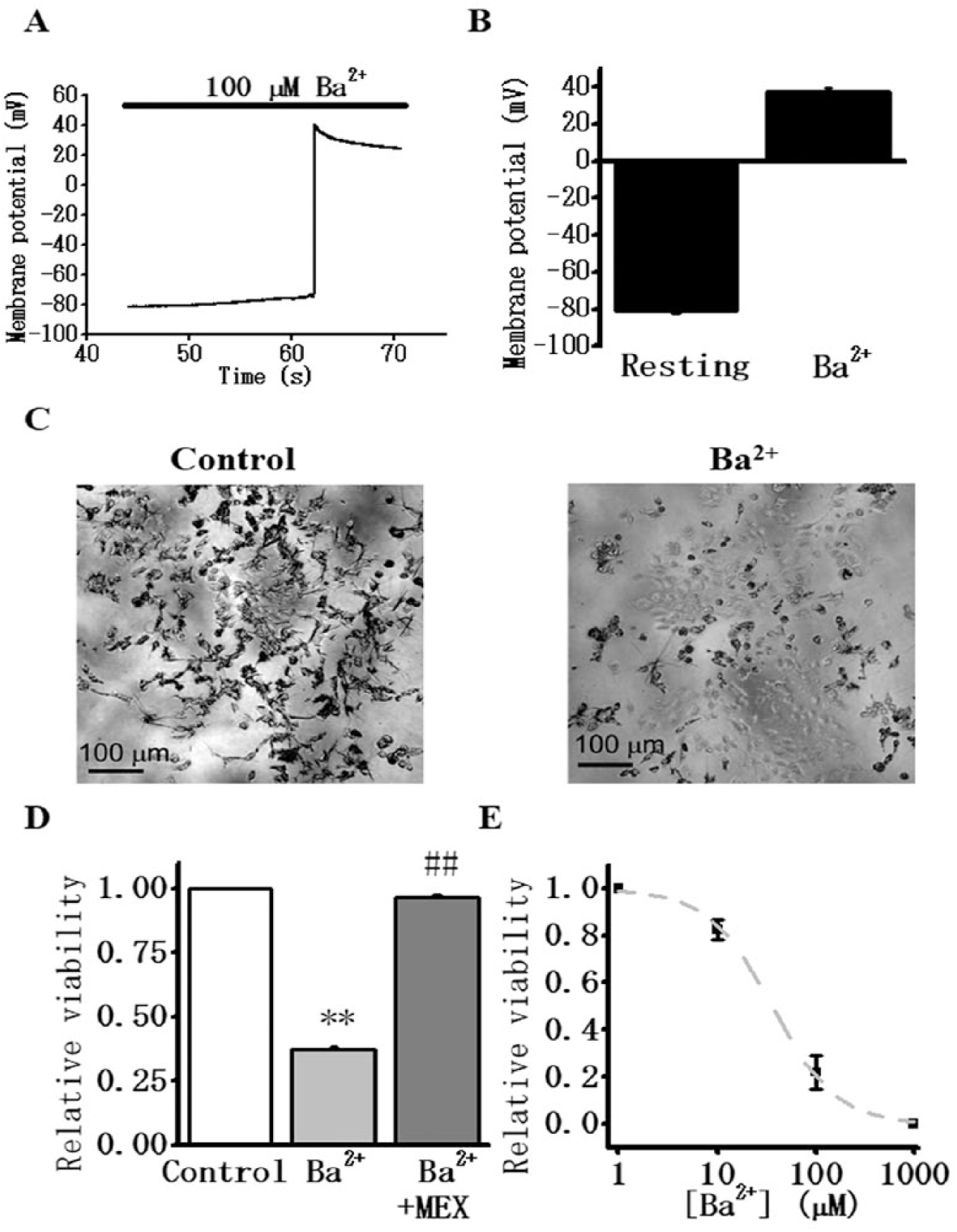
Ba2+-induced depolarization and cell death of test cells (Nav1.5 mutated in IFM/Q3 + Kir2.1).
Ba2+ Causes Death of the Test Cells in a Dose-Dependent Manner
The effect of Ba2+ on cell viability was examined using a population of test cells cultured in 24-well plates. These cells were treated with MTT to measure cell viability ( Fig. 1C ). 14 The relative viability of test cells was 0.37 ± 0.0071 (n = 6); that is, 63% of the cells died after the addition of 200 µM Ba2+, when taking the value in the control without Ba2+ as unity (1.0) ( Fig. 1C,D ). This measured cell death was almost completely suppressed by the presence of 300 μM mexiletine (0.97 ± 0.011 viability, n = 6) ( Fig. 1D ). Further examinations revealed that Ba2+ caused cell death in a dose-dependent manner. The IC50 for Ba2+, as determined with this test cell viability, was 36.0 ± 3.0 μM (n = 3) ( Fig. 1E ). We note that this value is close to the literal IC50 obtained previously from Kir2.1 current inhibition by voltage-clamp experiments (16.2 μM). 19 These results show that the block of Kir2.1 by Ba2+ elicits large Na+ influx through activated IFM/Q3. Presumably Na+ overload led to subsequent cell death.
Additional Co-Expression of K2P Channels in the Test Cells Reduced Ba2+-Induced Cell Death
TASK-1 and TASK-3 are members of the K2P channel family that produce leak K+ current in a variety of cell types. Generally, K2P channels are less sensitive to block by Ba2+ than Kir2.x. The IC50 of the Ba2+ block of TASK-1 and TASK-3 has been reported to be 400 and 300 μM, respectively (see Suppl. Table S1 ).20,21 The blockade of K2P channels by Ba2+ is often incomplete. We hypothesized that the additional co-expression of TASK-1 or TASK-3 in the test cells would keep RMP at a negative value even after the blockade of Kir2.1 by Ba2+. Thus, this maneuver could prevent the cell death ( Fig. 2A ). If a test compound has a blocking effect on K2P channels, the RMP would become less negative and a sustained depolarization and large Na+ influx would occur depending on the RMP. In contrast, if a test compound is an opener of K2P channels, the RMP would become more negative, resulting in higher cell viability ( Suppl. Fig. S1 ).
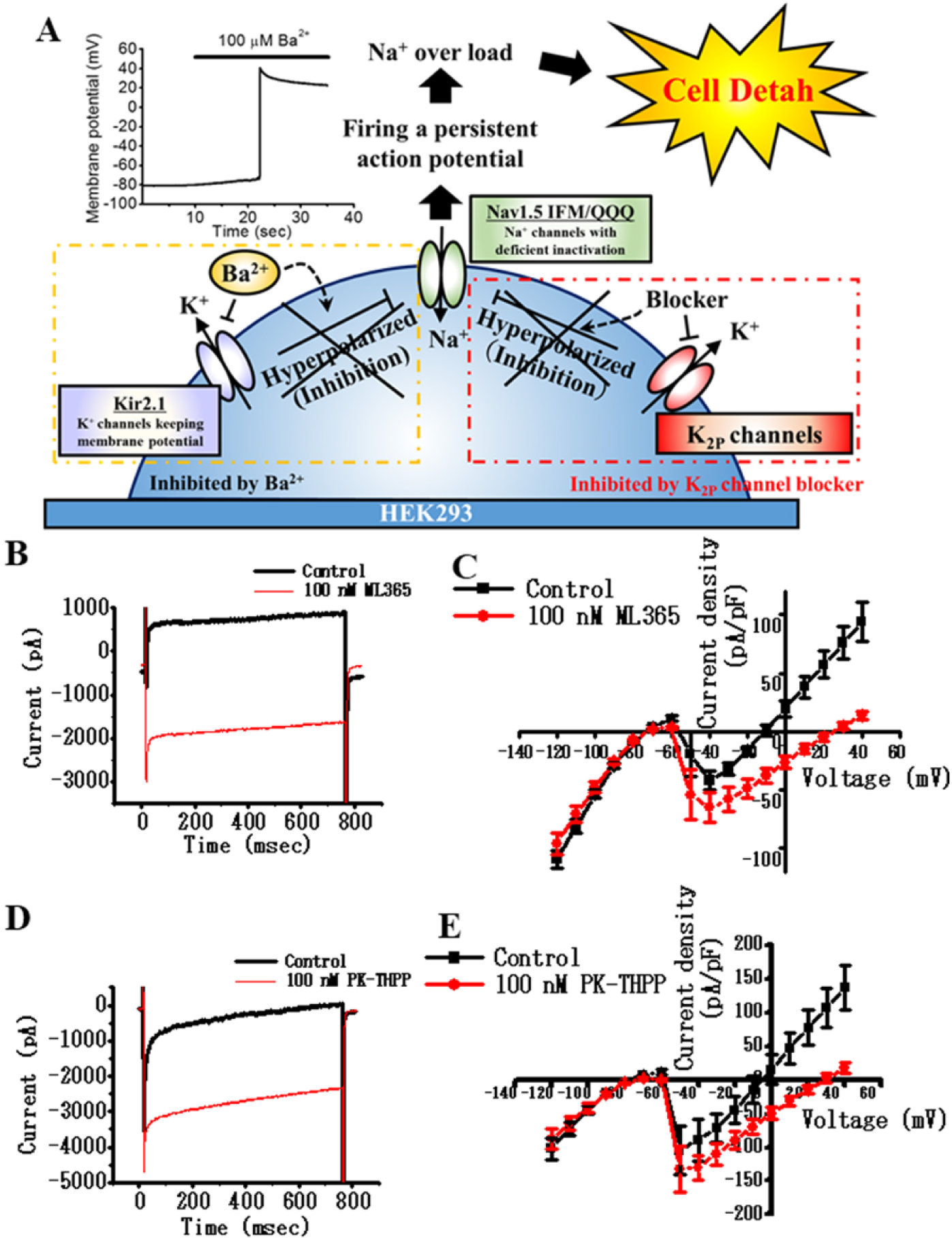
Introducing the K2P channel to test cells to evaluate the potency of test compounds modulating K2P channel activity by cell viability. (
To achieve the high expression of K2P channels, recombinant baculoviral vectors that contain genes coding TASK-1 or TASK-3 were utilized. We note, however, that TASK-1 contains an endoplasmic reticulum retention signal (i20 domain, 295G-314W) 22 and exhibits robust intracellular localization. Accordingly, a TASK-1 mutant that lacks the i20 domain (i.e., TASK-1Δi20) was used in this study. In the experiments for this study, either TASK-1Δi20 or TASK-3 was additionally expressed in the test cells. This combination yielded the desired TASK-1 or TASK-3 screening cell systems, respectively. Whole-cell patch-clamp recordings confirmed the high and reproducible functional expression of these channels in the test cells ( Fig. 2B–E ). Importantly, the outward current due to TASK-1 or TASK-3 channel activity was larger than the sustained component of the inward Na+ current through IFM/Q3 during the depolarization. The addition of ML365 or PK-THPP as TASK-1 or TASK-3 blockers unmasked the inward Na+ currents ( Fig. 2B–E ).
To confirm these results in multicell preparation, we examined the effects of these drugs on membrane potential using membrane potential-sensitive dye, DiBAC4(3) 14 ( Fig. 3 ). Application of Ba2+ elicited the expected persistent depolarization in the test cells ( Fig. 3A,B ). In TASK-1 or TASK-3 screening cells, the addition of Ba2+ did not evoke significant depolarization. However, subsequent addition of ML365 or PK-THPP did elicit a large and sustained depolarization ( Fig. 3C–F ). These findings confirmed that the additional co-expression of TASK-1 or TASK-3 maintained the hyperpolarized RMP value and thus prevented the Ba2+-induced opening of IFM/Q3 and subsequent cell death.
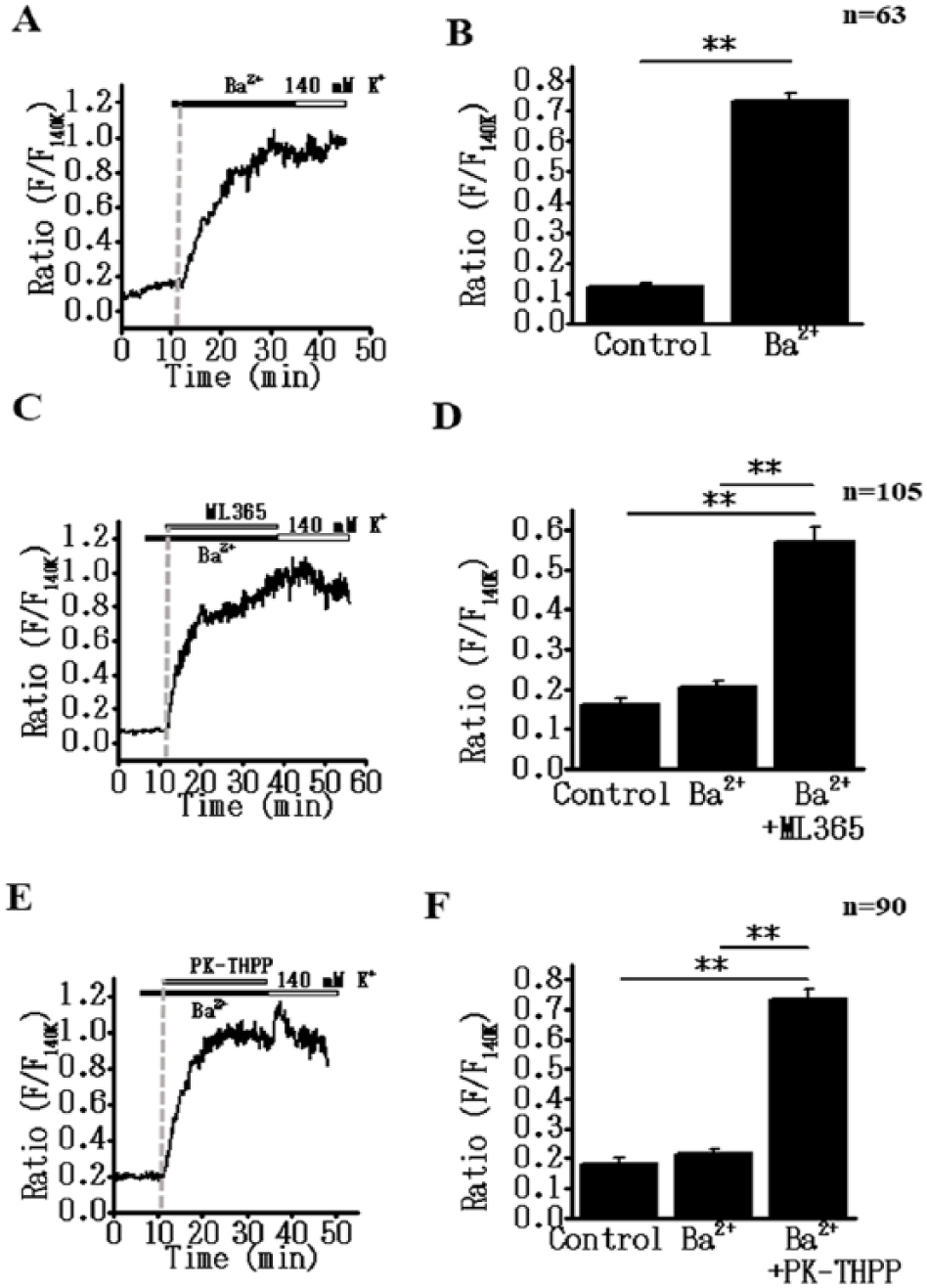
K2P channel inhibition can induce a sustained membrane depolarization in IFM/Q3+Kir2.1+K2P cells in the presence of Ba2+. Changes in membrane potential were monitored using DiBAC4(3). Fluorescence intensity (F) was normalized to that obtained by exposure to 140 mM K+ solution (F140K). (
Figure 4A consists of a flowchart depicting the procedure of K2P channel modulator screening by measuring cell viability. As shown in Figure 2A , we hypothesized that the activity of K2P channels expressed in test cells (TASK-1 or TASK-3 in this study) could be detected and quantified as their protecting effects on Ba2+-induced cell death. Indeed, as shown in Figure 4B,C , K2P channel activity compensates Ba2+-induced Kir2.1 channel inhibition in the regulation of RMPs, and thus prevents the overshoot depolarization and subsequent cell death ( Fig. 4A ).
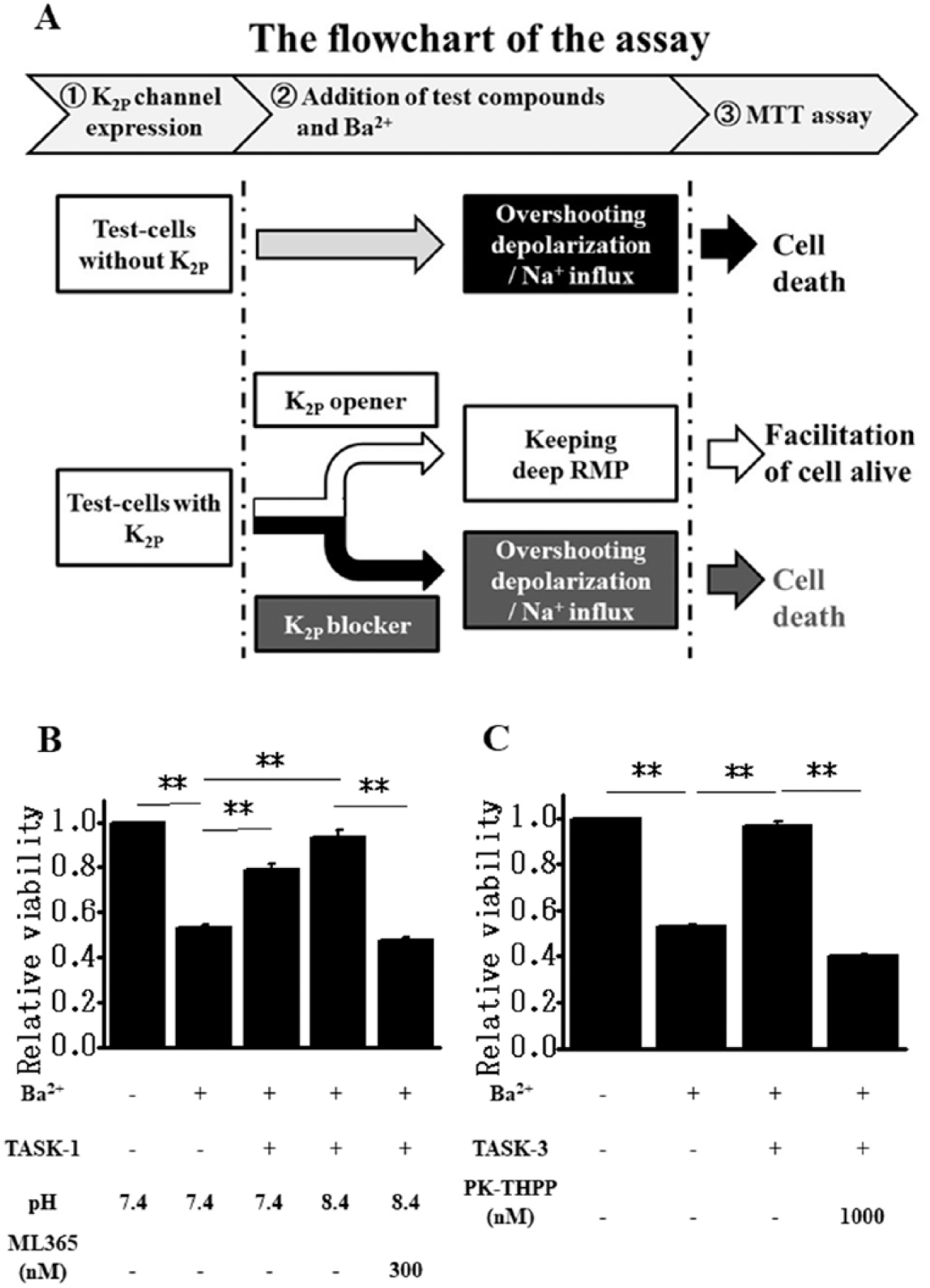
Modulation of K2P channel activity can be evaluated by the measurements of cell viability. (
Guided by the rationale in the flowchart, the evaluation of channel blockers and/or openers in TASK-1 or TASK-3 screening cells was performed using either 24- or 96-well plates and the MTT assay. The additional expression of TASK-1Δi20 in test cells as TASK-1 screening cells in 24-well plates consistently enhanced the cell viability following the Ba2+ application from 0.534 ± 0.012 (n = 6) to 0.787 ± 0.031 (n = 6) (pH 7.4, p < 0.01) ( Fig. 4B ). We also noted that since the IC50 of TASK-1 for extracellular pH is 7.3, TASK-1 is not fully activated at pH 7.4. Thus, TASK-1 can respond to subtle changes in extracellular pH within the physiological range. 23 As expected from the pH sensitivity, alkalization of the bathing solution to pH 8.4 in TASK-1 screening cells increased cell viability following Ba2+ application (n = 6, 0.936 ± 0.033, p < 0.01 vs 0.534) ( Fig. 4B ). The inhibition of cell death by TASK-1 expression in test cells was removed by the application of 300 nM ML365 (0.474 ± 0.015, n = 6, p < 0.01 vs 0.936) ( Fig. 4B ). The viability of test cells following Ba2+ application was also returned to very near the control value, from 0.532 ± 0.006 to 0.965 ± 0.020 (n = 6, p < 0.01), by the additional expression of TASK-3 ( Fig. 4C ). The inhibition of cell death by TASK-3 expression was canceled by the application of 1 μM PK-THPP (0.402 ± 0.007, n = 6, p < 0.01 vs 0.965).
Taken together, these results make clear that the application of K2P blocker cancels the compensatory action of the K2P channel on Ba2+-induced Kir2.1 inhibition. In contrast, the application of K2P channel openers (pH 8.4 in the case of TASK-1 in this study) presumably keeps the well-hyperpolarized RMP and more effectively prevents Ba2+-induced cell death. Under specific conditions, where the functional activity of the corresponding K2P channel is set, and thus not high enough to completely prevent the Ba2+-induced cell death, the K2P opening effect can be detected by significantly enhanced cell viability.
Evaluation of Dose-Dependent Inhibition of K2P Channel by Blockers
The dose-dependent effects of K2P channel blockers, ML365 and PK-THPP, on TASK-1 and TASK-3 were analyzed using the TASK-1/TASK-3 screening cells cultured in 24-well plates. As shown in Figure 5A , the viability of TASK-1 screening cells was dose dependently decreased by either ML365 or PK-THPP. The IC50 values of ML365 and PK-THPP for the viability inhibition were 20.6 ± 2.5 nM (n = 4) and 3.0 ± 0.3 nM (n = 4), respectively. The IC50 values of these blockers for TASK-1 current inhibition recorded using the patch-clamp technique are 12 and 17 nM, respectively. 8 Dose-dependent inhibition of cell viability by the compounds on TASK-3 was also examined ( Fig. 5B ). The dose dependency of the inhibition by ML365 was not well fitted by the Hill equation and the IC50 was >150 nM (n = 3). The IC50 of PK-THPP was 10.1 ± 1.6 nM (n = 4). The IC50 values of ML365 and PK-THPP for TASK-3 current inhibition measured in a patch-clamp study in a heterologous expression system have been reported as 480 and 35 nM, respectively. 8 Thus, the IC50 values obtained by use of K2P screening cells were close to, but slightly lower than, those obtained with the conventional electrophysiological method.
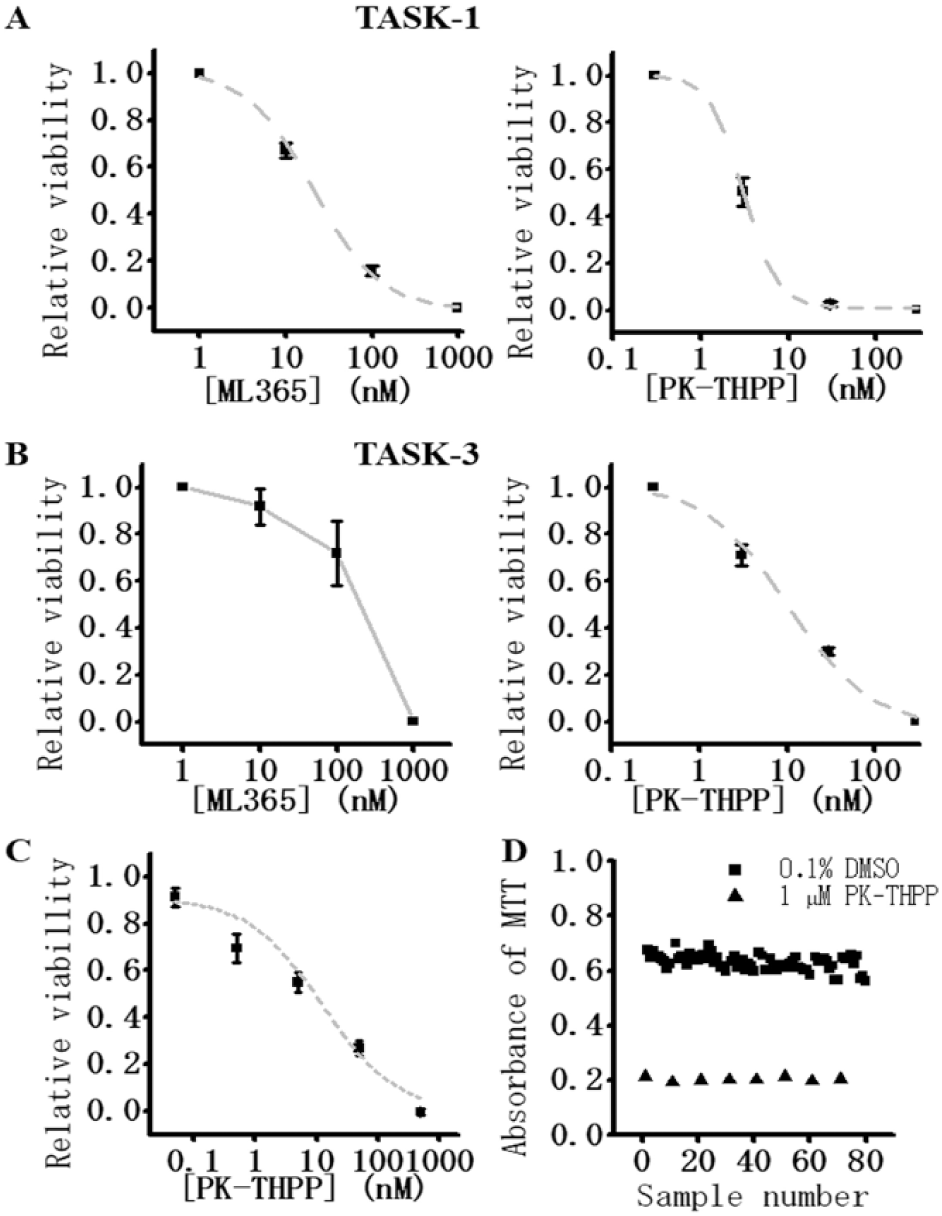
Validation of the assay systems for K2P channels. (
Evaluation of K2P Channel Modulators by Using an Automated Screening System
We examined whether this novel screening system can be automated for HTS. Here the seeding of TASK-3 screening cells onto 96-well plates, the exchange of experimental solutions including washout of mexiletine, and the application of test compound (PK-THPP) were all performed using automated pipetting systems (see Materials and Methods). PK-THPP at concentrations of 0.05, 0.5, 5, 50, and 500 nM was applied to TASK-3 screening cells, and the IC50 obtained by this automated system was 12.2 ± 4.2 nM (n = 9) ( Fig. 5C ), very close to that obtained in Figure 5B (10.1 nM). In order to assess the quantitative accuracy of this system, we calculated the Z′ factor. 24 When the Z′ factor is between 0.5 and 1.0, the assay is considered to be acceptable. The Z′ factors measured in the assays using the screening cells for TASK-1 or TASK-3 in 24-well plates with manual operation were 0.50 and 0.61, respectively (data not shown). In Figure 5D , the TASK-3 screening cells were seeded on 96-well plates, and cells treated with 200 µM Ba2++ 0.1% DMSO were used as a negative control and those treated with 200 µM Ba2++ 1 µM PK-THPP were used as a positive control. Data obtained using this automated process were shown in Figure 5D . The calculated Z′ factor from the data in Figure 5D was 0.75, and the averaged Z′ factor was 0.66 ± 0.05 (the mean of three trials performed on three separate dates with each as an independent assay; the three Z′ values were 0.58, 0.75, and 0.65). These results demonstrated that our automated assay system can achieve the reliability required for HTS.
Finally, a randomized blind test was performed to further evaluate this screening system utility using TASK-3 screening cells and our automated system mentioned above. In this trial, we used five compounds, each at 1 μM PK-THPP (TASK-3 blocker), ML365 (TASK-3 blocker), losartan (angiotensin receptor antagonist), atropine (muscarinic receptor antagonist), and NS309 (small conductance Ca2+-activated K+ channel activator). These compounds were assigned a number (T1–T5) at random and the experiment was performed in a blind fashion. Figure 6A illustrates the absorption values of the MTT assay in the alignment of five drugs and vehicle control (DMSO) in a 96-well plate. T1 and T3 decreased cell viability by 0.610 ± 0.051 (n = 12, p < 0.01 vs DMSO) and 0.875 ± 0.042 (n = 12, p < 0.01 vs DMSO), respectively. T2, T4, and T5 had no effect on cell viability ( Fig. 6A,B ). The correspondence of numbers and compounds is shown in Figure 6C : T1: ML365, T2: NS309, T3:PK-THPP, T4: losartan, and T5: atropine. Note that based on these results, two TASK-3 blockers could be clearly identified from five test compounds by this randomized blind test.
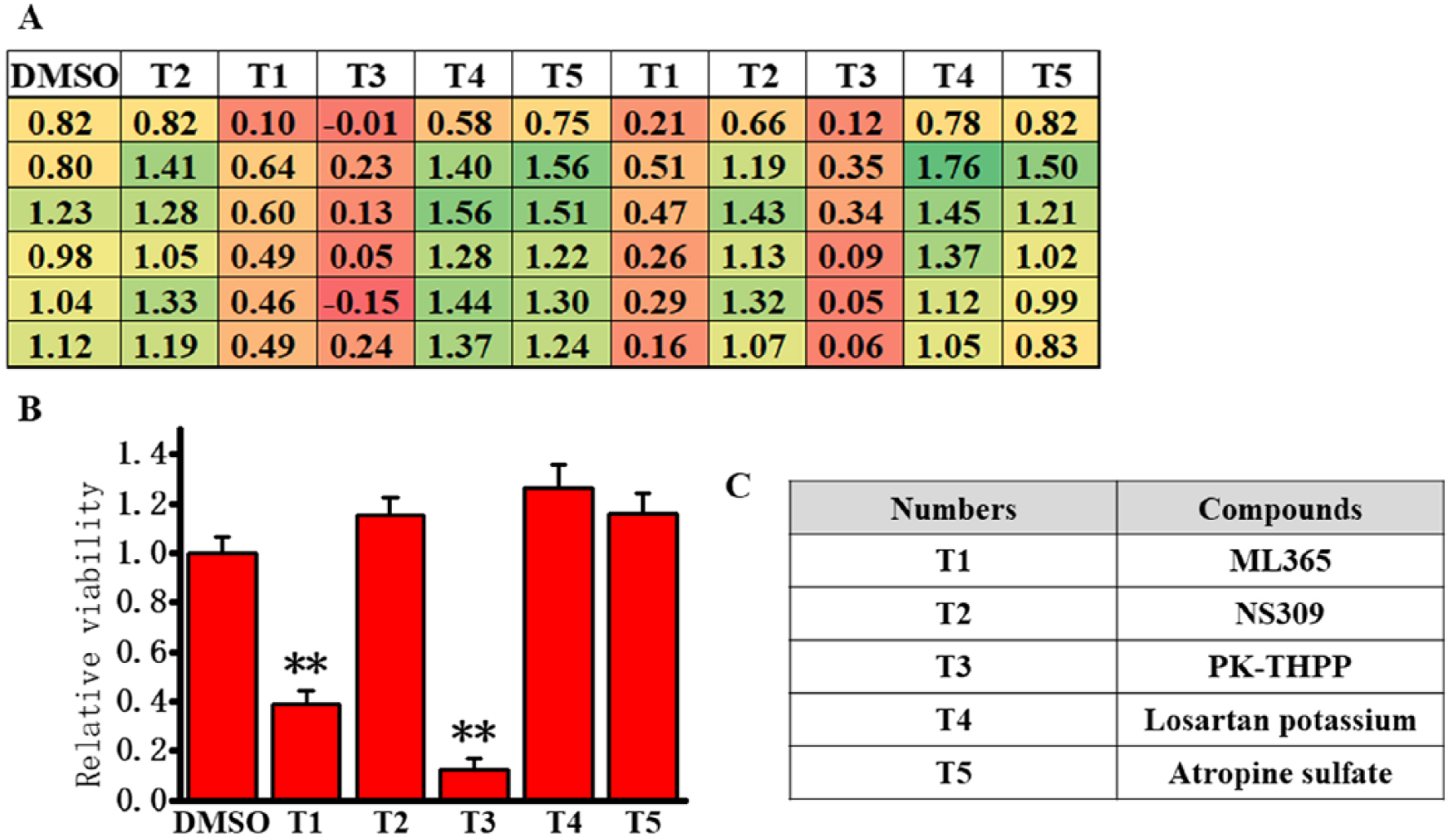
Randomized blind test for the evaluation of TASK-3 modulators using the new cell-based assay.
Discussion
In the present study, we present a novel cell-based assay system that is suitable for drug discovery targeting on ion channels, particularly background K2P channels. Importantly, the viability of the screening cells, which express Kir2.1+IFM/Q3+targeted K2P and are treated with low concentrations of Ba2+, is strongly regulated by the activity of background K2P channels. The viability was substantially modulated by the presence of a test compound acting on K2P channels. The quantitative data sets depicting the efficiency of K2P channel modulators obtained by this system were as precise as those from membrane current recordings under patch clamp. Single-cell patch-clamp assays are the gold standard method for assessing the effects of drug candidates on ion channel activity. Thus, this new system allows us to evaluate the modulating effects of compounds on K2P channel activity simply by monitoring cell viability, and it offers high time and cost performance, which is best suited for HTS.
Mechanisms of the New Assay System
In our test cells (Kir2.1+IFM/Q3), the average RMP was –78.4 mV ( Fig. 1A,B ). Thus, it is obvious that the expressed Kir2.1 channel activity contributes to keeping the RMP close to the K+ equilibrium potential (–80.9 mV). This consequence of Kir2.1 expression is responsible for keeping the IFM/Q3 Na+ channels at a resting inactive state. The addition of 100–200 μM Ba2+ quite selectively inhibits Kir2.1 channels and evokes a sustained depolarization including an overshoot ( Fig. 1 ). This overshoot is due to Na+ channel opening, because the peak potential (+40.0 mV) ( Fig. 1A,B ) is closer to the reversal potential for Na+ (+90.8 mV) and the large depolarization was blocked by 300 μM mexiletine (not shown directly in this study). Importantly, the application of Ba2+ induced cell death in a dose-dependent manner. The cell death was also diminished by mexiletine ( Fig. 1D,E ). These results can be explained based on the Ba2+ inhibiting the Kir2.1 channel and causing membrane depolarization, resulting in the activation of the IFM/Q3 Na+ channels. Since the IFM/Q3 Na+ channel lacks inactivation gating mechamisms, 25 the resulting excess Na+ influx causes Na+ overload and subsequent cell death. A similar scenario has been reported using veratridine as an agent that markedly slows Na+ channel inactivation in neurons.26,27 The exact mechanism underlying the cell death in our system was not further examined in this study.
Ba2+-induced cell death in the test cells was suppressed or reduced by co-expression of TASK-1 or TASK-3 ( Fig. 4B,C ). These channels are known to be relatively insensitive to Ba2+ (IC50 for Ba2+: >400 μM in TASK-1 and >300 μM in TASK-3; see also Suppl. Table S1 ). Other K2P channels are also insensitive to Ba2+ or less sensitive than Kir2.1.20,21,28,29 For these reasons, K2P channels instead of Kir2.1 can keep the negative RMP even in the presence of submillimolar Ba2+. Importantly, the inhibition of K2P channels by TASK-1 and TASK-3 blockers, ML3658 and PK-THPP 11 , strongly enhanced the Ba2+-induced cell death of K2P screening cells ( Fig. 4 ). These results also provide a series of evidence revealing the protective effects of K2P channels on Kir2.1+IFM/Q3 cells. Of interest is that the alkalization of the bathing solution buffer enhanced the protective effect of TASK-1 ( Fig. 4B ). TASK-1 activity is highly sensitive to extracellular pH in a narrow physiological range. TASK-1 activity is enhanced in slightly alkaline conditions (approximately pH 8.4) compared with in neutral conditions (approximately pH 7.4). 23 These findings suggest that our assay system for K2P channels is applicable not only for the evaluation of candidate blockers but also for their openers, if the assay conditions including the functional expression level of the targeted K2P channel are adapted appropriately. In addition, this cell-based assay may also be potentially available for the HTS of drug discovery targeting on K+ channels such as Ca2+-activated K+ channels and ATP-sensitive K+ channels, which can contribute to keeping the RMP well negative and/or induce after-hyperpolarization of action potentials.
Validation of the Assay System
The IC50 (20.6 nM) for the ML365-induced block of TASK-1 obtained by our method was very similar to that (12 nM) determined by electrophysiological measurements. 8 Moreover, the present results confirm the previous observation concerning the selectivity of ML365 for TASK-1 apparently being over that for TASK-3. In this study, the IC50 for TASK-3 was >150 nM, and it was reported to be 480 nM in a previous report. 8 However, there is a lack of agreement between the results in the previous report and those in the present study about the selectivity of PK-THPP for TASK-1 over TASK-3. PK-THPP was originally reported as a TASK-3-selective blocker, exhibiting 10-fold selectivity over TASK-1 by use of automated patch-clamp methods; the IC50 values for TASK-1 and TASK-3 were 303 and 35 nM, respectively. 11 An independent study has reported that PK-THPP is a TASK-1-selective inhibitor over TASK-3 with 11.2-fold selectivity (16 and 180 nM, respectively) based on measurement with thallium flux assay (FDSS). 8 In addition, data revealing an apparent 2.1-fold selectivity have been obtained (17 and 35 nM, respectively) with automated patch-clamp assay (Q-Patch). 8 The present results are consistent with those obtained in the latter report with Q-patch, since the IC50 values of PK-THPP for TASK-1 and TASK-3 are 3.0 and 10.1 nM, respectively ( Fig. 5A,B ).
Z′ factor is a reliable index that has been used to validate the reproducibility of assay methods for HTS. Assays with a Z′ factor ranging between 0.5 and 1.0 are considered to be excellent for HTS. 24 The averaged Z′ factor of the assay with TASK-3 screening cells in 96-well plates under automated procedures was 0.66 ( Fig. 5D ). Thus, these results suggest that the new cell-based assay fulfills the requirements of HTS for K2P channel drug discovery.
False-positive or -negative findings from our system would be predicted in some cases, as has been discussed in the previous study, in which ES was used instead of Ba2+. 14 For example, a cytotoxic test compound would be evaluated as a false positive of the K2P channel blocker. If the test compound possesses a Na+ channel blocking effect, it would be evaluated as a false positive or a false negative of the K2P channel opener or blocker, respectively. These limitations can be easily assessed by comparing effects of a test compound on Ba2+-induced cell death in IFM/Q3+Kir and IFM/Q3+Kir+K2P, as has been suggested previously. 14
Application of an Automated Process Based on 96-Well Plates and Randomized Blind Test
For optimized practical HTS, it is also required that the whole screening process, including cell seeding, is performed by an automated operation system using 96- or 384-well assays. Accordingly, we have adapted this assay based on an automated operation system consisting of 96-well plates, including validation with respect to the IC50 and Z′ factor. Even under the demanding conditions of automated procedures, the IC50 of PK-THPP in the TASK-3 screening measurement was 12.2 nM. This was in close agreement with data obtained using the manual procedure with 24-well plates (10.1 nM). While it was slightly lower than the IC50 values by automated patch clamp (35 nM), 8 it was substantially different from the thallium assay value (180 nM). 8 The Z′ factor calculated from the results obtained by the automated operation system with 96-well plates in this study was as good as 0.75 ( Fig. 5D ).
Importantly, also the reproducibility of our automated operation system was confirmed by a randomized blind test ( Fig. 6 ). Among five test compounds at the concentration of 1 μM, only ML365 and PK-THPP were detected as potent TASK-3 blockers. These findings provide additive evidence that this K2P channel screening system is well qualified for real HTS.
K2P Channel Modulators as Drug Candidates
K2P channels are responsible for background leak currents that modulate the RMP in a variety of cell types.1,30 Changes in functional expression of K2P channels are known to be involved in many significant pathological settings. For example, TASK-1 is involved in chronic atrial fibrillation,2,3 pulmonary hypertension, 31 inflammation, 32 and neurodegeneration. 33 On the other hand, TASK-3 is associated with depression 34 and cancer progression.35,36 Accordingly, modulators of TASK-1 and TASK-3 have been suggested to be targets for new drug therapy for these diseases. Thus, the development of a novel assay method suitable for HTS that can be used to discover K2P channel modulators with high efficiency is much needed. Here, we provide a novel solution for the issue. The original technology developed in our previous study 14 has been patented in Japan, the United States, Europe, and China (WO/2012/002460). The important additional component of the screening method using Ba2+ instead of ES to elicit a sustained depolarization has been registered in another patent application (PCT/JP2017/039641).
In conclusion, we succeeded in developing a new drug screening system targeting the ion channels, which contribute to background K+ currents and can strongly regulate RMP, such as K2P channels. The underlying rationale and cellular mechanism of this screening system is significantly different from those of others. By monitoring cell viability, we indirectly but precisely detect membrane potential changes reflecting the activity of targeted K+ channels. In addition to convenience and reliability, our system also offers very high performance with respect to the initial cost for equipment and the operating cost. Thus, it is a novel and robust example of real HTS for drug discovery targeting selected ion channels.
Supplemental Material
DISC829745_SupplementalMaterial – Supplemental material for Development of a Novel Cell-Based Assay System for High-Throughput Screening of Compounds Acting on Background Two-Pore Domain K+ Channels
Supplemental material, DISC829745_SupplementalMaterial for Development of a Novel Cell-Based Assay System for High-Throughput Screening of Compounds Acting on Background Two-Pore Domain K+ Channels by Keisuke Kawasaki, Yoshiaki Suzuki, Hisao Yamamura and Yuji Imaizumi in SLAS Discovery
Footnotes
Acknowledgements
We acknowledge Dr. Takashi Murayama (Juntendo University, Tokyo, Japan) for his kind provision of the baculoviral vector. We also thank the Drug Discovery Initiative, University of Tokyo, for valuable technical support.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors disclosed the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study has been performed as a collaboration of the authors in Nagoya City University (NCU) with ChanneloSearch Technology Co. Ltd. (Nagoya, Japan), which is a venture company funded by some researchers in NCU, including the corresponding author, Y.I.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI grant numbers 26670039, 26293021, and 16K15128 to Y.I.; 16H06215 and 16K15127 to Y.S.; and 16K08278 and 17H05537 to H.Y.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
