Abstract
Organophosphates are a class of highly toxic chemicals that includes many pesticides and chemical weapons. Exposure to organophosphates, either through accidents or acts of terrorism, poses a significant risk to human health and safety. Existing antidotes, in use for over 50 years, have modest efficacy and undesirable toxicities. Therefore, discovering new organophosphate antidotes is a high priority. Early life stage zebrafish exposed to organophosphates exhibit several phenotypes that parallel the human response to organophosphates, including behavioral deficits, paralysis, and eventual death. Here, we have developed a high-throughput zebrafish screen in a 96-well plate format to find new antidotes that counteract organophosphate-induced lethality. In a pilot screen of 1200 known drugs, we identified 16 compounds that suppress organophosphate toxicity in zebrafish. Several in vitro assays coupled with liquid chromatography/tandem mass spectrometry–based metabolite profiling enabled determination of mechanisms of action for several of the antidotes, including reversible acetylcholinesterase inhibition, cholinergic receptor antagonism, and inhibition of bioactivation. Therefore, the in vivo screen is capable of discovering organophosphate antidotes that intervene in distinct pathways. These findings suggest that zebrafish screens might be a broadly applicable approach for discovering compounds that counteract the toxic effects of accidental or malicious poisonous exposures.
Introduction
Organophosphates (OPs) are a class acetylcholinesterase (AChE) inhibitors used as pesticides and as chemical warfare nerve agents.1–4 Human exposure to organophosphates causes a spectrum of behavioral and physiological effects, ranging from nausea, vomiting, lachrymation, and bradycardia at lower doses to seizures, muscle fasciculation, cardiovascular and respiratory collapse, paralysis, and death at higher doses.5,6 OPs are among the highest priority terror threats owing to their widespread availability and accessibility.2,7 In addition, the use of organophosphates as pesticides poses a significant risk of accidental exposure.8,9 Therefore, the development of improved organophosphate countermeasures will have a positive impact on human health and safety.
The major existing OP countermeasure is the combined administration of atropine and pralidoxime (2-PAM). OPs inhibit AChE, preventing the breakdown of the neurotransmitter acetylcholine.5,6 The accumulation of acetylcholine near cholinergic receptor sites causes continuous stimulation of cholinergic nerve fibers of the central and peripheral nervous systems, leading to the symptoms of toxicity. Drugs such as atropine antagonize cholinergic receptors, countering the effects of acetylcholine accumulation, but their toxicity limits their use.4,10 2-PAM, often used in conjunction with atropine, is an AChE reactivator. Organophosphates inhibit AChE by covalently linking a phosphoryl group to the enzyme’s active site serine hydroxyl group. 2-PAM reactivates the AChE by attacking the phosphorylated serine residue, removing the phosphate moiety, regenerating active enzyme.11,12 Some OPs are known to sterically hinder reactivation, thus reducing the efficacy of 2-PAM. Furthermore, toxicity associated with 2-PAM administration could lead to dangerous hypertension and tachycardia. 10 The development of additional antidotes that work via other mechanisms might therefore substantially improve clinical care and safety.1,4,13
We sought to develop an unbiased, screening-based approach to discover additional OP antidotes with diverse and potentially novel mechanisms. Our approach uses an in vivo chemical screen in zebrafish. The close relationship between zebrafish and human genome, anatomy, and physiology has already been demonstrated in a large body of literature.14,15 Zebrafish larvae exposed to OPs exhibit phenotypes analogous to the human response to OP exposure, including behavioral deficits, paralysis, and eventual death.16,17 Zebrafish offer several unique advantages, including the ease of handling and maintenance, rapid ex utero development, and the optical transparency of the larvae.18–20 Moreover, compounds identified by a zebrafish-based screen have already exhibited efficacy and lack of toxicity in a whole vertebrate animal, a significant clinical advantage over in vitro or cell–based screens. 21
We have identified conditions in which the prototypical OP azinphos-methyl causes consistent lethality of early life stage zebrafish. Using these conditions, with death as an end point, we conducted a pilot screen of 1200 known drugs in a 96-well format. Sixteen drugs exhibited the ability to prevent death in organophosphate-exposed zebrafish. Through subsequent biochemical and metabolite profiling assays, we have demonstrated that these compounds function through multiple diverse mechanisms. Therefore, an in vivo zebrafish screen could potentially identify new countermeasures with novel mechanisms of action and enhance our ability to respond to the threats of chemical exposure.
Materials and Methods
Materials
The chemical library was purchased from Prestwick Chemical (Illkirch, France). The drug compounds for retesting and ammonium formate of liquid chromatography–mass spectrometry (LC-MS) grade were purchased from Sigma-Aldrich (St. Louis, MO). The isotope labeled acetylcholine-d9 chloride (N,N,N-trimethyl-d9) and choline-1,1,2,2-d4 chloride were purchased from CDN Isotopes, Inc. (Point-Claire, Quebec, Canada). Methanol and acetonitrile of LC-MS grade were obtained from Fisher Scientific (Waltham, MA). Water used for high-performance liquid chromatography (HPLC) was purified by Milli-Q system (Millipore, Billerica, MA).
Instruments
A PerkinElmer 2030 Multilabel Reader (Victor X3; PerkinElmer, Waltham, MA) was used as the fluorescent microplate reader. An Agilent 1200 system (Agilent, Santa Clara, CA) was applied as the LC system. The analytical column was a Waters XBridge HILIC, 2.5 µm, 50 × 2.1 mm (Waters, Milford, MA). An ABI 4000 Q Trap (AB SCIEX, Framingham, MA) with an electrospray ionization (ESI) source was used as the mass spectrometer. The nitrogen and zero air were supplied by a Source 5000 LC/MS Gas Generator (Parker Balston Analytical Gas System; Parker Balston, Haverhill, MA). The infusion was performed by using a KD Scientific (model KDS-100; KD Scientific, Holliston, MA) syringe pump. A Leap HTS PAL system (Leap Technologies, Carrboro, NC) was used as the autosampler for injection. The data acquisition and processing were accomplished using Analyst software, version 1.5.1 (AB SCIEX, Framingham, MA).
Aquaculture
Fertilized eggs (up to 1000 embryos per day) were collected from group matings of TuAB zebrafish (a hybrid of Tubingen and AB zebrafish lines). Embryos were raised in HEPES (10 mM)–buffered E3 medium in a dark incubator at 28 °C until the experiment.
Zebrafish Chemical Screening Protocol
Three to five zebrafish embryos, 8 days postfertilization (dpf), were distributed into wells of clear flat-bottom 96-well microplates purchased from Fisher Scientific (cat. 260836) in 360 µL E3 buffer. Compounds from chemical libraries were added to each well at a concentration of 50 µM, before treating with 65 µM azinphos-methyl for 16 h. The animals were then observed under a Leica KL 200 optical microscope (Leica, Wetzlar, Germany) to check for heartbeat. The absence of heartbeat was used as an indicator death, and the percentage mortality was determined.
Ellman Assay
The AChE activity was assayed in vitro as described. 22
Cell Culture of SH-SY5Y Cells
Cell culture was carried out as described in detail by Ridley et al. 23 In brief, SH-SY5Y human neuroblastoma stock cultures were routinely maintained in Dulbecco’s modified Eagle’s medium (DMEM)/Ham’s F12 (1:1) modified medium containing 1% nonessential amino acids (NEAAs) and supplemented with fetal calf serum (FCS, 15%), glutamine (2 mM), penicillin (50 µg mL−1), and streptomycin (50 µg mL−1). Stock cultures were passaged 1:5 weekly and fed twice weekly.
Fluorescence Assay for Cholinergic Activity
The assay was performed as described in Ridley et al. 23 Culture medium was removed from the wells, and the cells were washed twice with Tyrode’s salt solution (TSS). Fluo-3 AM (10 µM) and 0.02% pluronic acid were added in TSS (40 µL per well), and the cells were incubated at room temperature for 1 h in the dark. The cells were washed twice with TSS and 80 µL TSS alone or OS1–OS6, and the plate was transferred to a PerkinElmer 2030 Multilabel Reader. After 10 min preincubation, nicotine (final concentration 30 µM) was added (20 µL per well), and fluorescence was monitored at a 538-nM wavelength. Maximum and minimum fluorescence was determined by the sequential addition of 20 µL Triton ×100 (2%, final concentration) and 20 µL MnCl2 (40 mM, final concentration). Changes in intracellular calcium were calculated as a percentage of the difference between the minimum and maximum fluorescence.
Preparation of Zebrafish Samples for Metabolite Profiling Experiments
Zebrafish larvae were collected at 8 dpf. The fish were treated with drugs (50 µM of azinphos-methyl and hit compounds) for 5 min before sample collection. For each study, 50 larvae (volume about 11 µL) were mixed with 89 µL Milli-Q water (Millipore), 600 µL methanol, and 100 µL internal standard (IS; 0.5 ng/mL d9-Ach and 20 ng/mL d4-Ch in methanol) solution. The larvae were homogenized by 45 s of sonication and followed by 20 s of vortex to get the lysates. Each 50 µL of fish lysate was mixed with 450 µL MeOH. After 1 min of vortex, the mixtures were centrifuged at 14 000 rpm for 5 min at 4 °C. Then, 400 µL of clear supernatant was taken and 5 µL of the solution was injected into LC-MS/MS system each time.
Mass Spectroscopy and High-Pressure Liquid Chromatography
The chromatographic separation was achieved by using a gradient program of mobile phase A (0.1% formic acid in 10 mM ammonium formate buffer) and B (0.1% formic acid in 95% MeCN with 5 mM ammonium formate) on an XBridge HILIC (Waters) at a flow rate of 150 µL/min. Gradient elution: 0–0.5 min, 100%B; 0.5–6 min, 100%–40%B; 6–7 min, 40%B; 7–8 min, 40%–100%B; and 8–15 min, 100%B. Each run of this LC method was 15 min. The detection was monitored by a 4000 Q Trap mass spectrometer with ESI in negative mode. The mass spectrometer measurement was conducted in the multiple-reaction monitoring mode (MRM). A Turbo Spray system (AB SCIEX, Framingham, MA) was used as ion source. The compound optimization of operation parameters was performed through the infusion and flow injection analysis (FIA) of the pure compounds (diluted in LC–mobile phase condition). The transitions m/z and optimized compound parameters are shown in
Results and Discussion
Organophosphates Cause Dose-Dependent Lethality in Zebrafish
We treated zebrafish larvae with two representative OP compounds, aziphos-methyl and parathion, for 16 h ( Fig. 1 ). The absence of heartbeat was used as a reliable indicator of death, and mortality was defined as the percentage of dead animals at the end of the treatment period. The LC50s of azinphos-methyl and parathion were determined to be 54 µM and 13.5 µM, respectively.
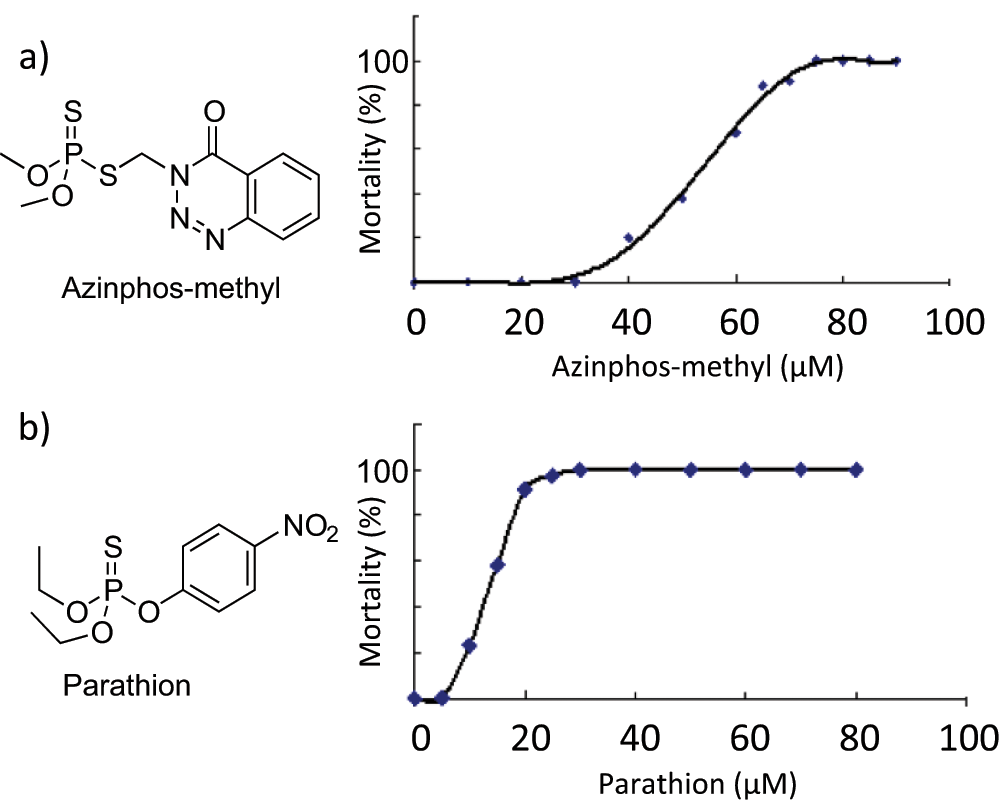
The effect of organophosphate (OP) treatment on zebrafish mortality. Zebrafish larvae (8 days postfertilization) were exposed to different concentrations (0–100 µM) of OPs for 16 h: (
Atropine/2-PAM Promotes Survival in Zebrafish following OP Exposure
A mixture of atropine (50 µM) and 2-PAM (50 µM), a known antidote for OP toxicity, significantly reduces lethality of zebrafish embryos and provides a positive control for our chemical screen ( Fig. 2 ).
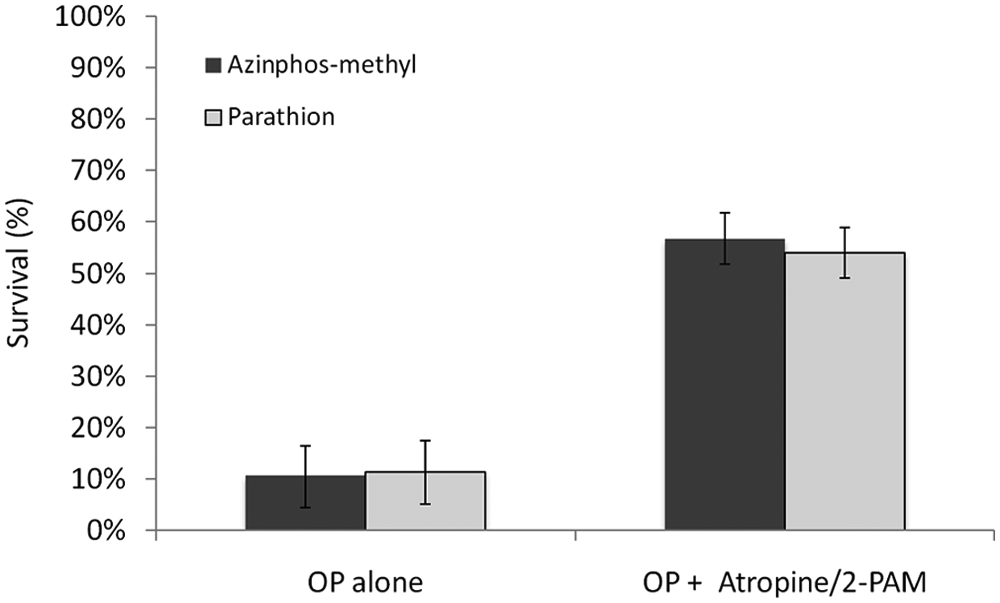
Atropine (50 µM)/pralidoxime (2-PAM) (50 µM) counteracts organophosphate (OP)–induced toxicity and improves survival. Zebrafish larvae (8 days postfertilization) were treated with azinphos-methyl (65 µM) and parathion (20 µM) alone and with a mixture of atropine (50 µM) and 2-PAM (50 µM) for 16 h.
Development of a Screen for Novel OP Antidotes
The lethality of zebrafish larvae following OP treatment provided the basis for our high-throughput screen. For chemical screening, three to five zebrafish larvae (8 dpf) were transferred by pipette to each well of a 96-well plate. Compounds from chemical libraries were added by multichannel pipette to each well at a concentration of 50 µM, before treating with 65 µM azinphos-methyl for 16 h. At this concentration, azinphos-methyl induced approximately 90% mortality ( Fig. 1a ).
Using the conditions described above, we screened the Prestwick library (1200 compounds) for those that could protect zebrafish from OP-induced lethality. The pilot screen was repeated, and only compounds that improved survival to 50% or higher in both assays were chosen for retesting. Sixteen compounds were confirmed as positive hits under these criteria (organophosphate suppressors, OS1–OS16) ( Table 1 ).
Pilot Screen of a Library of 1200 Compounds Identifies 16 Hits That Counteract Organophosphate Toxicity

For chemical screening, zebrafish larvae were treated with 65 µM azinphos-methyl for 16 h, reducing survival to 10%. Compounds that improved survival to 50% or higher were selected as hits for further investigation.
We also tested OS1–OS16 (50 µM) with a second OP compound, parathion (20 µM). All 16 of the compounds suppressed the toxicity of parathion to a similar degree as azinphos-methyl, demonstrating the broad applicability of these compounds against OPs ( Fig. 3 ).
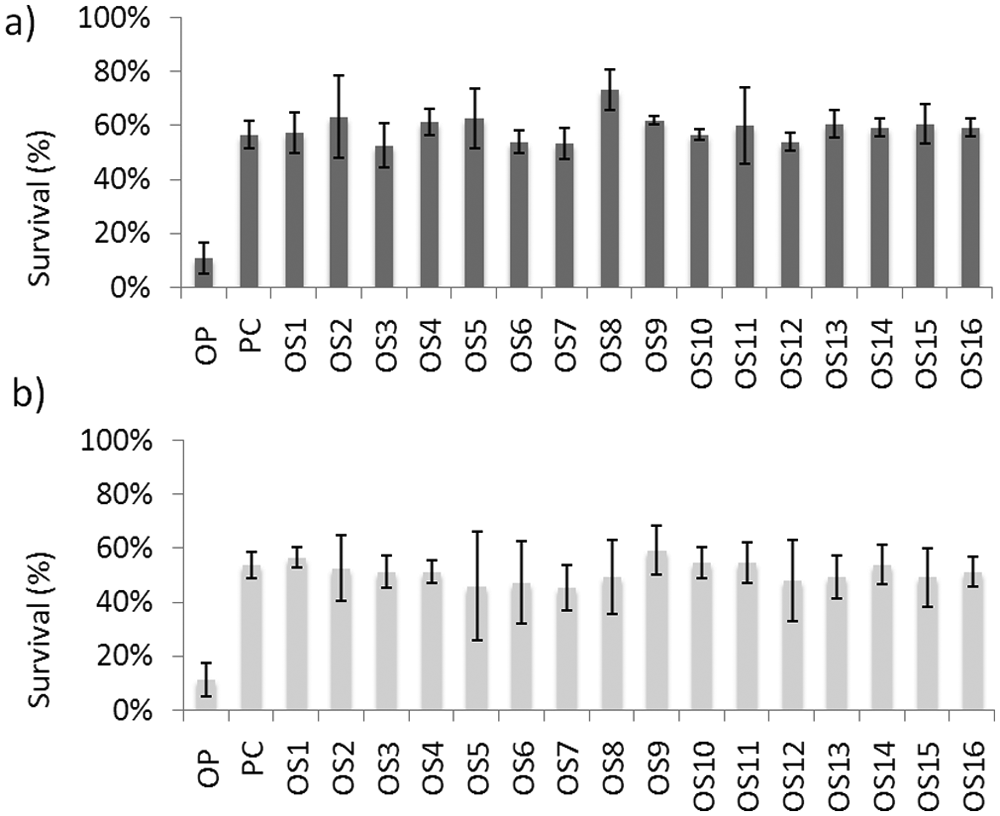
OS1 to OS16 (50 µM) are equally effective against the toxicity induced by (
Neostigmine Competes with OP for AChE Inhibition
Reversible (noncovalent) AChE antagonists have been shown to be partially effective as OP antidotes because they compete with OPs for access to AChE binding and thus reduce the amount of AChE permanently inhibited by OP. To determine if any of the 16 hits functions as a reversible AChE antagonist, we investigated the effect of OS1 to OS16 on AChE activity using the Ellman method. 22 One of the compounds, OS1 (neostigmine bromide), a known reversible AChE inhibitor, exhibited AChE antagonism (data not shown). Therefore, the in vivo zebrafish screen was able to identify OP antidotes that function by reversible antagonism of AChE.
Telenzepine, Glycopyrrolate, Metoclopramide Monohydrochloride, and Emetine Dihydrochloride Exhibit Anticholinergic Activity
Atropine is effective as an OP countermeasure because of its anticholinergic activity. To determine if any of the 16 hits functions through a similar mechanism, we used a cell-based fluorescence assay to measure the anticholinergic activity of the hit compounds. 23 Four compounds, OS2 to OS5, show significant anticholinergic activity ( Fig. 4 ). OS2 (telenzepine dihydrochloride) and OS3 (glycopyrrolate) are known acetylcholine receptor antagonists.24,25 However, the anticholinergic activity of OS4 (metoclopramide monohydrochloride) and OS5 (emetine dihydrochloride) had not been previously reported. Therefore, the in vivo zebrafish screen was able to identify OP antidotes that function via an anticholinergic mechanism, including both known and novel anticholinergic compounds.
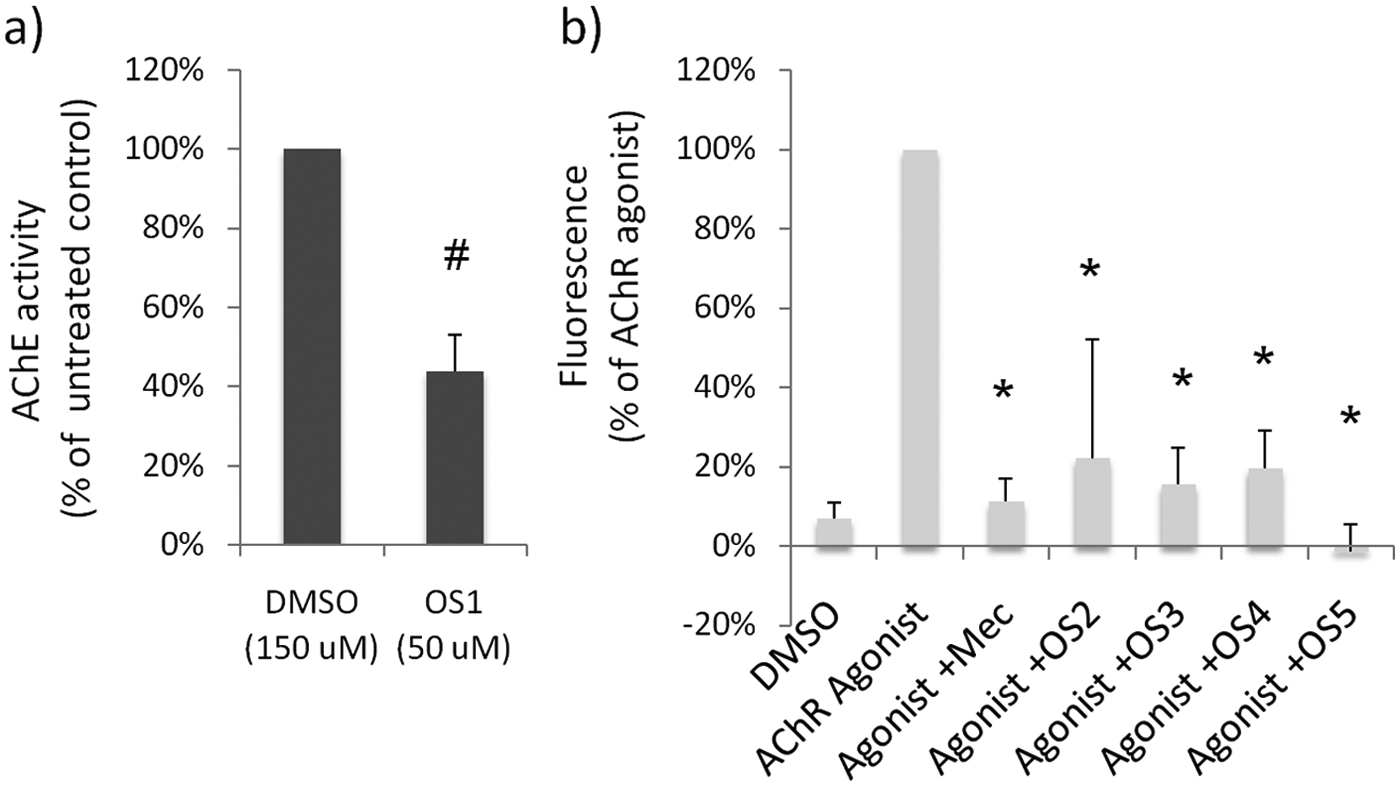
(
Metabolite Profiling Offers Additional Hypotheses for Mechanisms of Action of Hit Compounds
Although our in vitro assays provided mechanistic insights for OS1 to OS5, the mechanism of action of the remaining compounds remained unknown. We used a metabolite profiling approach to generate new hypotheses for further investigation. An LC-MS/MS metabolite profiling approach was used to detect and quantify neurotransmitters as well as the metabolites of the OP and counteracting drugs at a global level in samples of zebrafish lysates.
Azinphos-methyl (AZIN) is a relatively inert OP itself but is converted to azinphos-methyl oxon (AZOX), the bioactive form of the OP, in the liver ( Fig. 5a ). 24 Our preliminary metabolite profiling results indicate that OS6 and OS7 could function by inhibiting this bioactivation of AZIN ( Fig. 5b ). Zebrafish treated with AZIN and OS6 or OS7 exhibited significantly diminished levels of AZOX compared with zebrafish treated with AZIN alone or in combination with one of the other OP antidotes. Therefore, the in vivo zebrafish screen was able to identify OP antidotes that appear to function by inhibiting the bioactivation of AZIN. Such compounds would be of limited use as antidotes for nerve agents or other OPs that do not require bioactivation. However, these compounds demonstrate that the in vivo assay can discover compounds that function through multiple mechanisms of action, and they illustrate the importance and challenges of determining mechanisms of action for compounds discovered through phenotype-based screens.
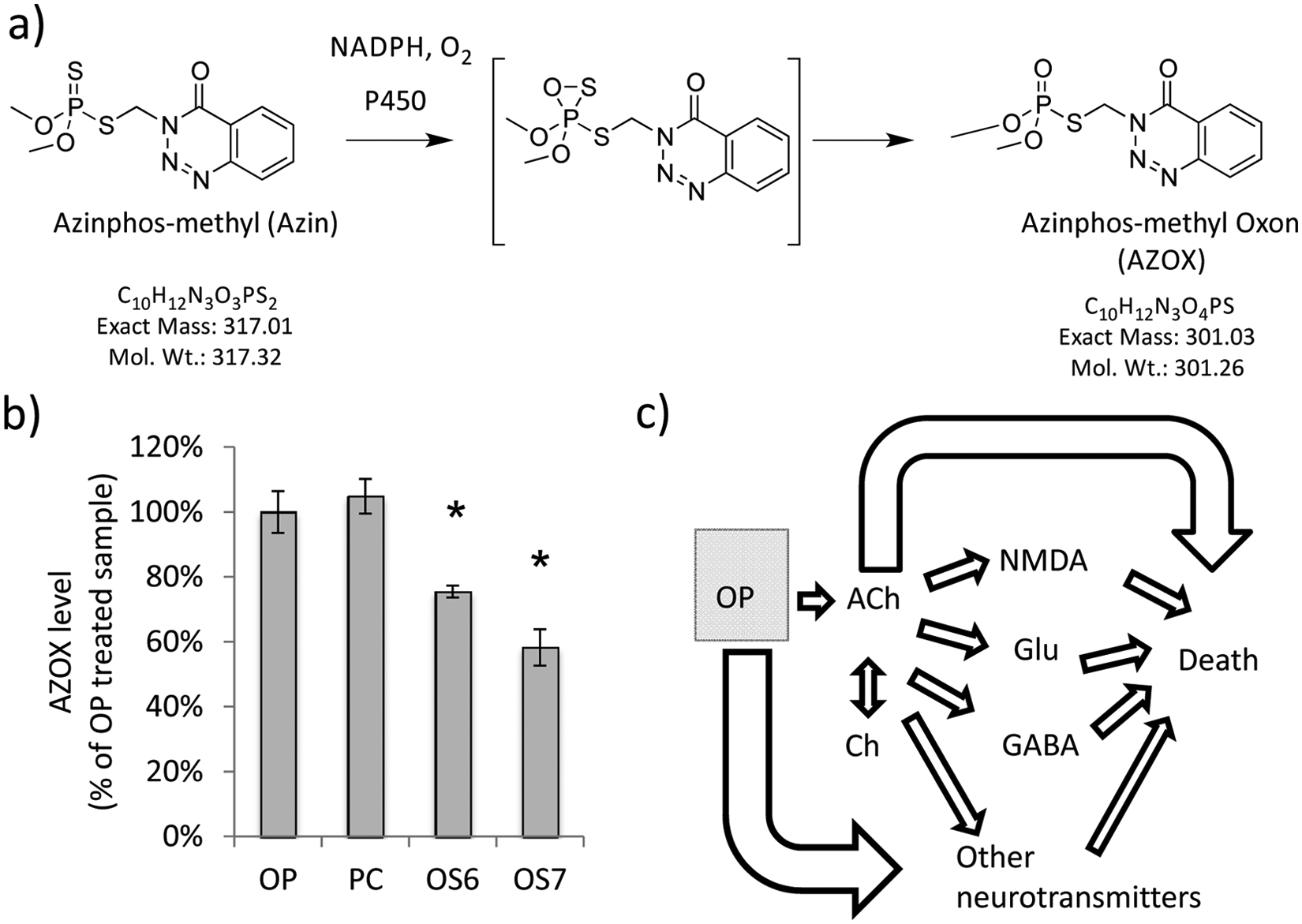
Metabolite profiling approach yields new mechanistic hypotheses. (
The studies described above were able to direct us to likely mechanisms of action for almost half (7/16) of the hits from the screen. However, the mechanisms of action of compounds OS8 to OS16 remain unknown. Future studies will focus on identifying these mechanisms. Fortunately, metabolite profiling identified numerous changes in the neurotransmitter profile after treatment with OP and suppressor compounds (
Pathway Prediction Based on Metabolomics Profiling of Neurotransmitters
Liquid chromatography/tandem mass spectrometry (LC-MS/MS)–based metabolomics studies detected significant changes in the in vivo levels of certain neurotransmitters in response to treatment with OS8-OS16. These results could potentially yield new hypotheses regarding their mechanism of action. GABA, γ-aminobutyric acid; NMDA, N-methyl-d-aspartate.
We have developed a rapid high-throughput method for discovering antidotes of OP toxicity in a zebrafish model. Our pilot chemical screen of a library of 1200 compounds has successfully identified 16 hits that rescue OP-induced mortality of zebrafish through a variety of known and potentially novel mechanisms, demonstrating the effectiveness of our model. The small molecules discovered by our screen could be excellent lead compounds for developing a diverse collection of antidotes for OP exposure. In addition, these compounds could also be used as powerful tools for dissecting toxicity pathways and improve our understanding of the underlying biology.
One of the major challenges of this phenotype-based approach is elucidating the mechanisms of action of the hit compounds. The mechanisms of several of our hit compounds remain unknown. Future work will involve further dissection of these mechanisms, investigating the efficacy of our hits in a mammalian model, as well as starting a large-scale screen for discovering additional countermeasures.
Footnotes
Acknowledgements
We thank Chung Yan J. Cheung, Andrew Rennekamp, and members of our research groups for helpful advice, raising zebrafish, maintaining cell cultures, and instrumental support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by the CounterACT Program, Office of the Director, National Institutes of Health (OD) and the National Institute of Neurological Disorders and Stroke (NINDS), Grant Number U01 NS063733 to RTP.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
