Abstract
Hematopoietic stem and progenitor cells (HSPCs) generate all cell types of the blood and are crucial for homeostasis of all blood lineages in vertebrates. Hematopoietic stem cell transplantation (HSCT) is a rapidly evolving technique that offers potential cure for hematologic cancers, such as leukemia or lymphoma. HSCT may be autologous or allogenic. Successful HSCT depends critically on the abundance of engraftment-competent HSPCs, which are currently difficult to obtain in large numbers. Therefore, finding compounds that enhance either the number or the activity of HSPCs could improve prognosis for patients undergoing HSCT and is of great clinical interest. We developed a semiautomated screening method for whole zebrafish larvae using conventional liquid handling equipment and confocal microscopy. Applying this pipeline, we screened 550 compounds in triplicate for proliferation of HSPCs in vivo and identified several modulators of hematopoietic stem cell activity. One identified hit was valproic acid (VPA), which was further validated as a compound that expands and maintains the population of HSPCs isolated from human peripheral blood ex vivo. In summary, our in vivo zebrafish imaging screen identified several potential drug candidates with clinical relevance and could easily be further expanded to screen more compounds.
Introduction
Hematopoietic stem cells (HSCs) are the blood cells that give rise to all the cell lineages of the blood. They give rise to both the myeloid (monocytes, neutrophils erythrocytes, etc.) and lymphoid (T cells, B cells, and natural killer cells) lineages of blood cells. HSCs reside in the bone marrow, where they multiply and differentiate to a subset of the blood types and also mobilize out of the bone marrow to multiply and give rise to the other blood cell types. The therapy of some malignant and nonmalignant blood diseases consists of the transplantation of hematopoietic stem and progenitor cells (HSPCs) into patients. These cells consist of a mixture of pure HSC and committed progenitor cells that are isolated from either bone marrow, peripheral blood, or human cord blood (hCB). 1 Obtaining pure HSCs is technically challenging.
Hematopoietic stem cell transplantation (HSCT) has been developed over the past 40 years and is an important therapy, with approximately 50,000 patients undergoing this procedure every year with either autologous or allogenic cells. 2 In spite of its medical importance, HSCT remains a risky procedure, especially for allogenic sources of cells, leading to graft versus host disease, but also for autologous sources of cells, leading to poor recovery and mortality.
It is thought that one of the limiting factors for successful HSCT is the insufficient number of functional HSPCs capable of homing and engrafting after transplantation, leading to increased mortality. 3 Thus to improve the clinical outcome of autologous and allogenic HSPC transplantation, an expansion of HSPCs prior to transplantation is currently considered essential to increase the chances of successful engraftment. So far, most attempts to increase HSPC numbers ex vivo have been unsuccessful because of either inadequate cell numbers or loss of engraftment capacity of the HSPCs during expansion. 3 Current protocols mainly rely on the use of recombinant cytokines, mesenchymal feeder layers, and additional molecules, like Notch ligands or prostaglandin E2 (PGE2) derivatives.4,5 Unfortunately, these strategies have not been able to avoid loss of stem cell activity and induction of differentiation during ex vivo culture. There is therefore a need to discover substances that prevent HSPCs from losing their stem cell potential while stimulating significant ex vivo expansion in order to exert therapeutic benefits to patients undergoing transplantation.
Executing drug discovery screens in vertebrate systems is generally very costly, technically challenging, and time-consuming. Zebrafish presents many advantages as a vertebrate screening model, due to its short generation time, its transparency, the available genetic tools, and the number of progeny produced progeneration. 6 Since the regulation of HSPC development is a well-conserved process between mammals and fish, zebrafish represents a small molecule screening system with high translational potential. 7 Both in mammals and zebrafish, HSPCs are of endothelial origin and arise from the ventral wall of the dorsal aorta. In immature HSPC, the proto-oncogene cmyb is highly expressed and the expression decreases during differentiation to various hematopoietic cells in both mammals and zebrafish. Cmyb-deficient mouse embryos die during gestation due to defects of definitive hematopoiesis in fetal liver. 8 In zebrafish, cmyb-expressing HSPCs arise in the aorta-gonad-mesonephros (AGM) of the ventral wall of dorsal aorta and can be easily detected at 36 h postfertilization (hpf). The cmyb-expressing HSPCs in the AGM migrate to caudal hematopoietic tissue (CHT) and thymus and then translocate to the kidney marrow, which is the equivalent of the bone marrow of mammals. 9 Recently, a chemical screen measuring the expression of cmyb and runx1 was performed using whole-mount RNA in situ hybridization (WISH) in order to identify small molecules regulating HSPC formation in zebrafish embryos. In this study, embryos were exposed between 11 and 36 hpf to 2357 known bioactive compounds. PGE2 was identified as a regulator of HSPC self-renewal during embryogenesis and shown subsequently to enhance HSPC engraftment, as measured by competitive repopulation studies in mice after ex vivo treatment of HSPCs. 5 Furthermore, PGE2 was shown to enhance both expansion of hCB by hematopoietic colony formation assay in vitro and engraftment in xenotransplantation experiments. 10
In this article, we describe the development of a high-content assay for high-throughput in vivo chemical compound screening to assess compound effects on cmyb:EGFP-expressing HSPCs in transgenic zebrafish embryos. Adaptation of typical high-throughput screening automation technologies to zebrafish embryos allowed us to perform a 550-compound screen in triplicate. A semiautomated image analysis pipeline was developed to segment and quantify individual HSPCs. We found several compounds that increased the number of HSPCs in vivo in zebrafish and validated them for their ability to increase the population of HSPCs isolated from human peripheral blood in vitro.
Materials and Methods
Embryo Preparation
All animal experiments were carried out in strict accordance with European Union and German animal protection laws. All experimental procedures were approved by the animal ethics committee of the Dresden University of Technology (TUD) and the Landesdirektion Sachsen (approval number AZ 24D-9168.11-1/2008-4). Adult zebrafish homozygous in cmyb:gfp 5 (in this article referred to as cmyb:EGFP) were mated with zebrafish homozygous in flk1:ras-cherrys896 11 (in this article referred to as flk1:mCherry), and double transgenic embryos were collected in E3 medium (5 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl2 × 2H2O, 0.33 mM MgSO4 × 7H2O, 0.0002% methylene blue, pH 6.5) and incubated at 28.5 °C. At 10 hpf embryos were dechorionated by treatment with 10 mg/mL pronase for 3–4 min. This procedure was adapted from Trompouki and Zon 12 for dechorionation of 36 hpf embryos. Treatment of 10 hpf embryos with pronase did not harm the embryos, if the pronase was washed away immediately when the first embryos started to get out of their chorions. Dechorionation was crucial for imaging the embryos from the lateral side. Embryos were washed four times with E3 medium and once with E2 medium (15 mM NaCl, 0.5 mM KCl, 1 mM CaCl2 × 2H2O, 1 mM MgSO4, 0.15 mM KH2PO4, 0.05 mM Na2HPO4, 0.7 mM NaHCO3). Dechorionated embryos were manually transferred one by one using an elongated Pasteur pipette into a 384-well glass-bottom plate (SensoPlate, Greiner Bio-One, Monroe, NC) (one embryo per well).
Automated Chemical Screen of Embryos in 384-Well Plates
After manual transfer of embryos into the 384-well plate, the volumes in the wells were unequal. To obtain equal volumes, the E2 medium was aspirated with a 384-well plate washer (PW384, Tecan, Männedorf, Switzerland) to a 50 µL remaining volume. The acoustic dispenser Echo 550 (Labcyte Inc., Sunnyvale, CA) was used to add 100 nL of chemical compounds in DMSO to the embryos. In total, 550 compounds from two focused libraries (StemSelect and InhibitorSelect, Calbiochem, Darmstadt, Germany) were screened in triplicates (three wells per compound) at a concentration of 20 µM. At 36 hpf, the embryos were fixed overnight at 4 °C by addition of 50 µL of 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS) using the drop dispenser WellMate (Thermo Scientific, Waltham, MA; 2% PFA final concentration). On the next day, the plates were washed with PBST (0.1% Tween-20 in PBS) using the plate washer PW384. Tween-20 within PBST was used to reduce surface tension. Thereby, embryos did not stick to the liquid surface, and could sink down to the bottom of the plate. The PW384 plate washer performed a wash cycle of seven repeats. First, a two-step aspiration left 70 µL, and then 38 µL of liquid in a 384 well. Second, a dispense step added 80 µL of PBST to each well. After dispensing, the embryos were allowed to sink down to the bottom of the well for 1 s. This procedure reduced the risk of embryos being washed away during the automated washing. Finally, the embryos were stored in 0.02% of sodium azide in PBS to preserve samples for long-term storage.
Automated Imaging and Analysis
The embryos in the 384-well plates were imaged on the confocal, high-content screening system Opera (PerkinElmer, Waltham, MA) using the 10× objective (NA = 0.4). By tilting the plate, embryos were positioned on the bottom edge of the well. In some cases, the embryos still required to be manually oriented. Positioning and orientating the embryos within the wells ensured that the whole embryo was imaged by acquiring four positions at the bottom of each well (four fields in a row, no overlap). cmyb:EGFP was excited with a 488 nm laser and imaged with both a 520/35 emission filter and a 660/150 emission filter for autofluorescence (see below). flk1:mCherry was excited with a 561 nm laser and imaged with a 605/40 emission filter. Forty z planes with 10 µm steps were acquired at each position and each wavelength. Image processing and image analysis were done by execution of three custom-designed macros for the open-source image analysis software Fiji 13 (http://fiji.sc/Fiji). Further details about the installation and usage of the macros are available at https://github.com/stoeter/Fiji-Tools-for-HCS/wiki/Fish-Embryo-Screen. In brief, the first macro converts the images from the Opera by applying a background subtraction, tiling all fields, subtracting the autofluorescence channel from the green fluorescence protein (GFP) channel, and performing a maximum z projection for each channel. The second macro let the user specify a region of interest (ROI) around the AGM and CHT of the embryo. The third macro uses the z projection and the ROI to segment and measure GFP-positive objects (HSPCs) within each embryo.
Data Analysis
Data were analyzed by the open-source software KNIME
14
(http://www.knime.org) and plug-ins “HCS tools” and “Scripting Integration,” which support the handling of large screening data within KNIME. GFP-positive objects (HSPCs) were filtered for their size, roundness, and intensity to exclude false objects resulting from segmentation errors. Measurements, such as intensity, size, and roundness of all objects per well, were averaged first for each embryo and second for each compound. Data were normalized using the median measurement of DMSO-treated embryos for each fish pair (n = 4–9). For hit definition, the robust Z score
15
was calculated using the median and median absolute deviation (MAD) of DMSO wells for each fish pair. Compounds increasing the number of GFP-positive objects (mean Z score > 2.5) were considered a hit, and compounds with a mean Z score of less than −5 were considered a decreasing hit. Images of the hits were checked visually to exclude chemical compounds that showed a strong autofluorescence overlaying the fluorescent signal of the GFP fusion protein. Autofluorescent compounds can lead to false positives, and compounds were verified in wild-type embryos to exclude autofluorescence (
Bar plots and error bars visualize means and standard deviations. Where indicated, p values were calculated using unpaired one-tailed t test. For estimates and their deviations, the mean and the standard error of the mean (SEM) were calculated.
Whole-Mount RNA In Situ Hybridization
Cmyb in situ hybridization was performed as described previously. 12 Twenty to thirty embryos on a 25 mm glass vessel were treated from 10 hpf until 36 hpf in E2 media containing 20 μM of valproic acid (VPA) or PBS (control). At 36 hpf, the embryos were washed three times with E2 media and fixed in 4% PFA for whole-mount in situ hybridization (WISH) for cmyb.
Isolation and Culture of CD34+ Cells from Peripheral Blood
Mobilized peripheral blood collections were obtained from University Hospital Carl Gustav Carus Dresden. Informed consent was obtained in accordance with a research protocol approved by the local institutional review board. Peripheral blood of healthy donors was collected from leukapheresis products after the donors were treated with 7.5 μg/kg granulocyte colony-stimulating factor for 5 days. CD34+ cells were purified from leukapheresis samples using CD34 antibody-conjugated magnetic beads according to the manufacturer’s instructions (Miltenyi Biotec, Frankfurt, Germany). CD34+ cells have a purity of more than 95% as assessed by flow cytometry (fluorescence-activated cell sorting [FACS]) and a vitality of more than 96% as measured by trypan blue exclusion. CD34+ cells (5 × 104) were cultured in CellGro (CellGenix, Freiburg, Germany) serum-free medium supplemented with 10 ng/mL stem cell factor (SCF), 10 ng/mL Fms-related tyrosine kinase 3 ligand (FLT3 ligand), and 10 ng/mL interleukin 3 (IL-3) and incubated at 37 °C in 5% CO2. After 24 h, cells were exposed to 1 mM VPA or PBS (control) and incubated for another 7 days.
Flow Cytometry Analysis
Cells isolated from peripheral blood were labeled with CD34-FITC and CD90-APC monoclonal antibodies (1:25; Miltenyi Biotec). After staining, 1 × 104 cells were examined using FACS Calibur (BD Biosciences, Heidelberg, Germany) and analyzed with the open-source Flowing Software (http://www.flowingsoftware.com).
Results
Development of a High-Content Screening Assay to Monitor Hematopoietic Stem and Progenitor Cells
In zebrafish, cmyb is essential for development of HSPCs and one of the earliest markers expressed in definitive HSPCs. Definitive HSPCs in the AGM migrate to the CHT, which is an intermediate site of blood development, and subsequently colonize the kidney ( Fig. 1A ). To monitor cmyb-expressing HSPCs, a cmyb:EGFP line 5 was used. To help determine the region of the AGM and CHT during image analysis, a zebrafish line expressing the endothelial marker flk1:mCherry 11 was crossed to the cmyb:EGFP line. For screening, double transgenic embryos expressing both markers, cmyb:EGFP and flk1:mCherry, were used ( Fig. 1B–D ).
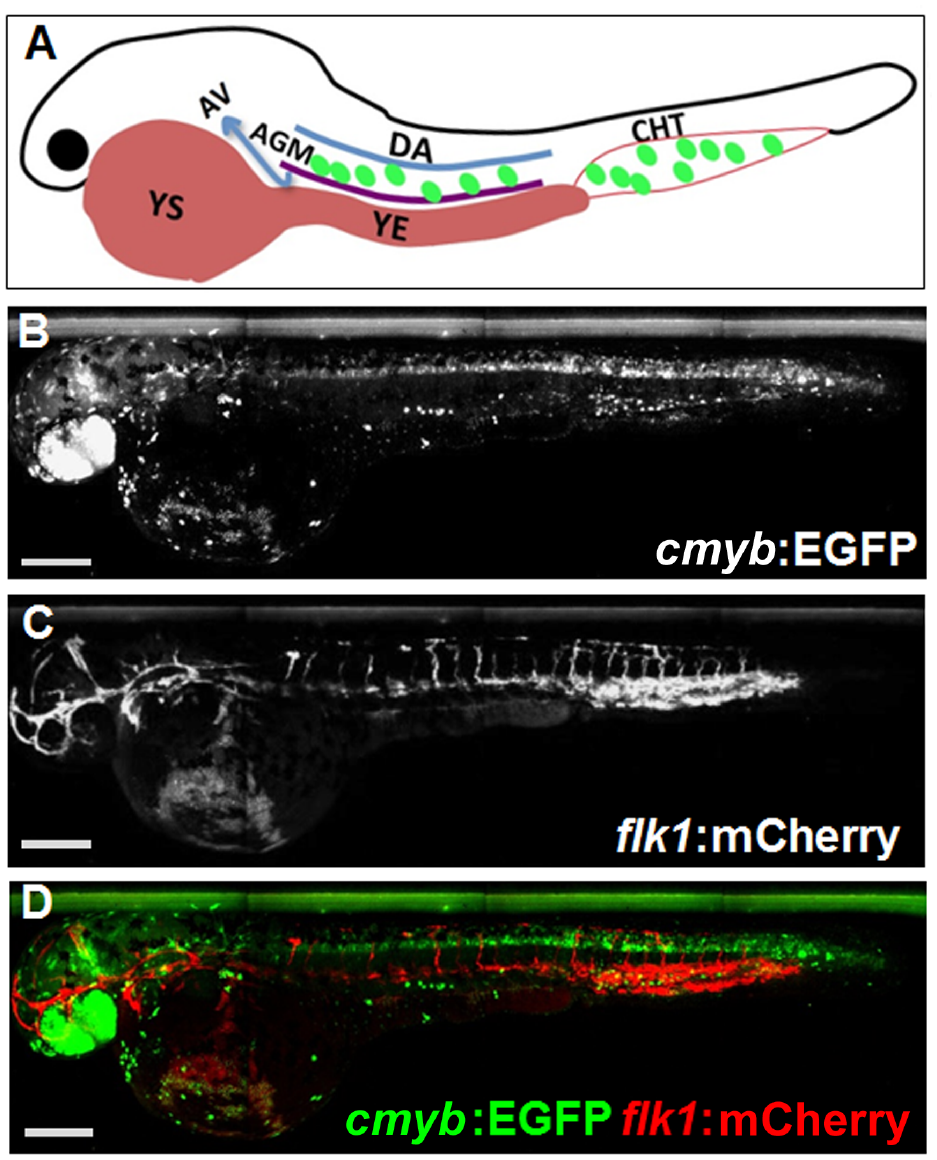
Expression of cmyb and flk1 in zebrafish embryos. (
Developing screening applications for zebrafish in 384-well plates is challenging, as the animals are not attached and will be washed away by typical wash protocols for attached cells. For imaging applications, a further challenge is to find the animal in the comparatively large area of a 384-well plate and perform whole animal imaging along its anterior–posterior axis. Several experimental steps were optimized to carry out in vivo zebrafish chemical compound screening using high-throughput screening automation and a minimum of manual steps (see Material and Methods,
Development of a Semiautomated Image Analysis Routine to Quantify HSPCs in Dechorionated Zebrafish Embryos
cmyb:EGFP-positive cells are found in the region of the AGM close to the ventral wall of dorsal aorta (DA) and in the region of the CHT ( Fig. 1 ), which likely represent definitive HSPCs. 9 We developed a semiautomated image analysis pipeline to detect and count the HSPC in the AGM and CHT.
The acquired enhanced green fluorescence protein (EGFP) images contained autofluorescence signal that would have led to the detection of false-positive objects ( Fig. 2 ). Therefore, an autofluorescence image was captured at a longer wavelength ( Fig. 2B ) and subtracted from the EGFP image ( Fig. 2A ), largely reducing the autofluorescence signal ( Fig. 2C ). Next, the image fields were tiled and the z planes were collapsed into a single image with a maximum z projection. Detection of the AGM and CHT region could not be done in an automated fashion in a reliable way. Thus, a user defined the ROI, where cmyb:EGFP-positive objects needed to be detected. Lastly, individual cmyb:EGFP-positive objects were automatically segmented with the method of Li ( Fig. 2D,E ). To accomplish these tasks, we wrote three macros in the open-source software Fiji 13 that allowed a semiautomated image analysis guided by the user. In this manner, the manual portion of the analysis for an entire plate required no more than 2–3 h.
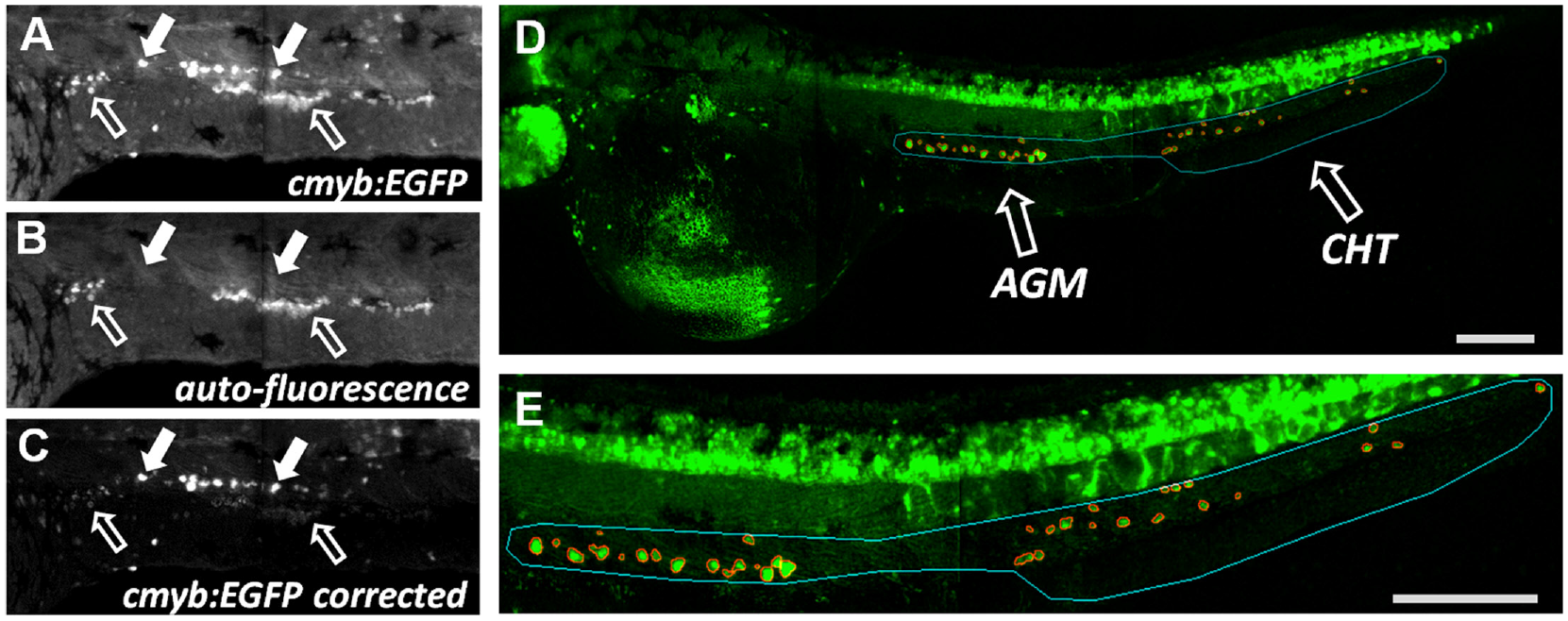
Image analysis and correction for autofluorescence signal. Signal of cmyb:EGFP excited with 488 nm laser and detected with a 520/35 filter (
Object data were analyzed with the open-source software KNIME. 14 Dim and small objects were filtered out, and finally object data were aggregated into well measurements and normalized to control (see Material and Methods). To test our high-content screening assay with the developed semiautomated image analysis and data analysis pipeline, we treated embryos with indomethacin, a COX1/2 inhibitor that was shown to reduce the expression of cmyb and the number of HSPCs. 5 We were able to reproducibly detect a reduction of cmyb:EGFP-expressing HSPCs in the AGM and CHT region of indomethacin-treated embryos compared to DMSO-treated embryos ( Fig. 3A ). This proof of concept demonstrated that with the help of a user-guided image analysis, a quantification of individual HSPCs in zebrafish embryos was possible.
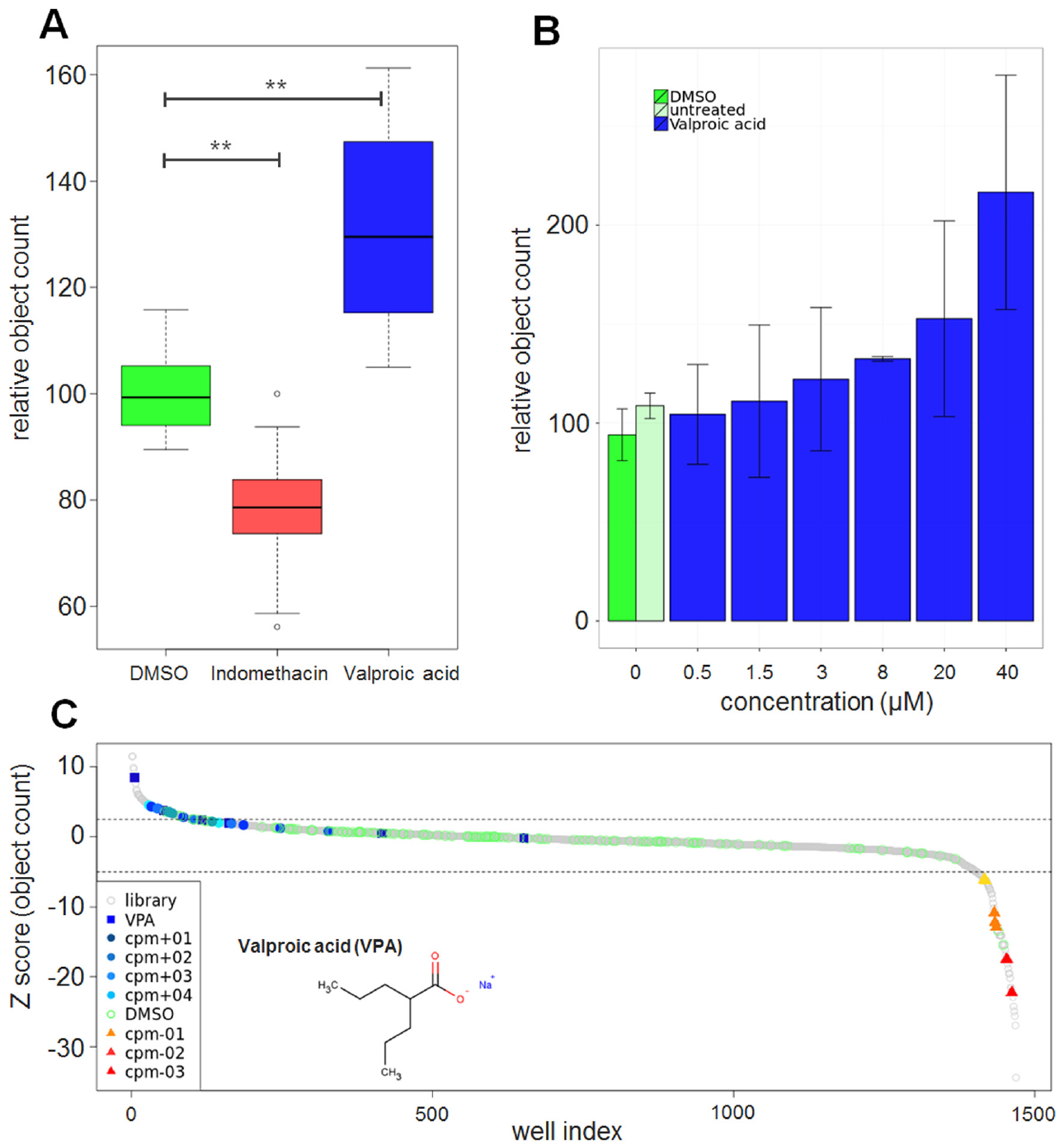
In vivo chemical screen for regulators of hematopoiesis in zebrafish embryos. Object data from image analysis were aggregated first per embryo and second per compound. (
Valproic Acid Increases the Number of cmyb:EGFP-Positive Cells
Using our high-content assay for hematopoietic stem and progenitor cells in zebrafish embryos and our semiautomated image analysis, we screened two chemical libraries containing compounds with known targets or known biological activities. Embryos were exposed to compounds at a concentration of 20 µM between 12 and 36 hpf. DMSO was used as a negative control and indomethacin was used as a control to decrease the number of HSPCs. When the screen was initiated, no inducer of HSPCs was known, and so no positive controls were included. Early during the screening campaign, VPA was identified as a strong inducer of HSPCs ( Fig. 4A,B ) and after hit confirmation, VPA was included as a positive control in the validation of the assay in the remainder of the screen. On each plate, several wells were used as a negative control (DMSO) and positive control (indomethacin, VPA) to monitor the performance of the assay. In DMSO-treated embryos about 50 ± 3 cmyb+ objects were identified, in indomethacin-treated embryos this number was decreased to 78% ± 4%, and in VPA-treated embryos it was increased to 131% ± 12% ( Fig. 3A ). To confirm the effects of VPA, embryos were treated with different concentrations ranging from 0.5 to 40 µM. As shown in Figure 3B , VPA increased the number of cmyb:EGFP-positive HSPCs in a dose-dependent manner with a two fold increase at the highest concentration. To further validate VPA, WISH of cmyb was performed in DMSO and 20 µM VPA-treated wild-type embryos. VPA strongly increased the expression of cmyb in the region of the AGM and the CHT compared to DMSO-treated embryos ( Fig. 4C,D ).
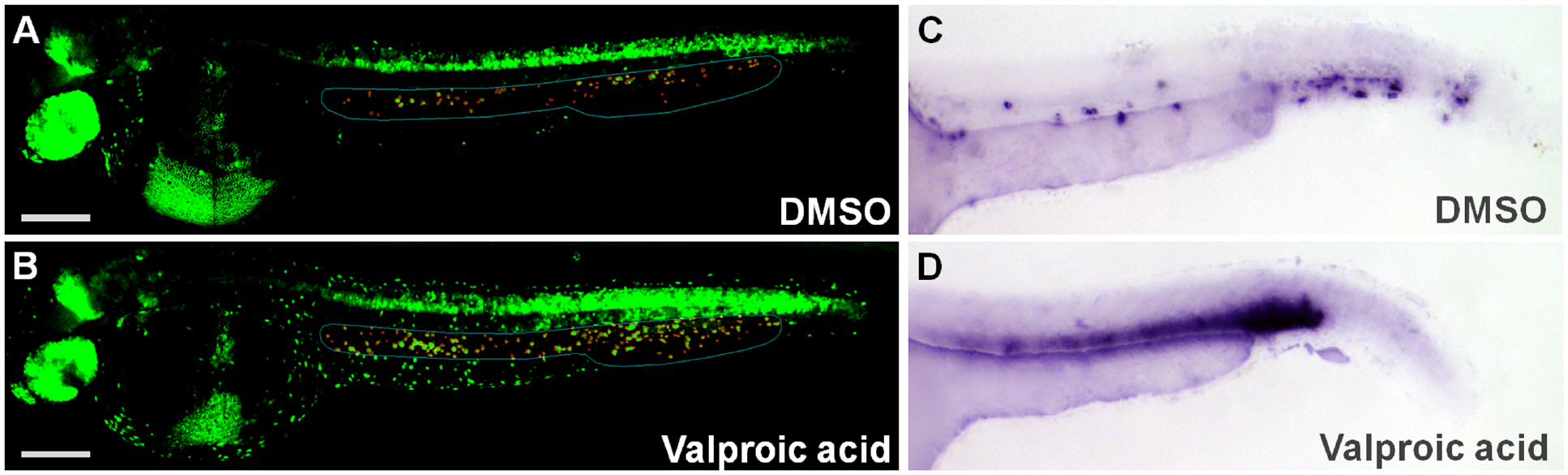
Effect of valproic acid on the expansion of HSPCs in the AGM and CHT of the zebrafish embryo. (
In total, 550 compounds were screened in triplicate and, including control wells, 2335 embryos were analyzed. A total of 601 embryos showed a severe toxic phenotype and were classified as “dead.” Five compounds were identified that strongly increased the number of HSPCs, and three compounds decreased the number of HSPCs (for hit selection criteria, see Materials and Methods, Fig. 3C ).
Therefore, using our in vivo vertebrate screening approach, we identified several compounds that expand HSPCs in zebrafish embryos.
Valproic Acid Increases the Population of CD34+ and CD90+ Cells from Human Peripheral Blood Ex Vivo
The HDAC inhibitor valproic acid was previously reported to stimulate proliferation of HSPCs. 16 In order to verify the clinical potential of VPA for expanding HSPCs, we investigated its effect on human CD34+ cells isolated from mobilized human peripheral blood. The cells were treated ex vivo for 7 days with 1 mM VPA and analyzed by flow cytometry. CD34 is a well-known surface marker for human immature HSPCs and, in combination with Thy-1 (CD90), defines a highly pluripotent subpopulation.17,18 It is well established that the CD90+ (Thy-1+) subpopulation of CD34+ cells contains more primitive HSPC and accounts for most of the long-term engraftment in animal transplant models.18,19 In our culture conditions, the total number of cells increased up to 20-fold. The proportion of CD34+ cells in VPA-treated conditions was 68 ± 5%, whereas in vehicle-treated controls the proportion was only 24 ± 3% ( Fig. 5 ). Importantly, in VPA-treated cultures, the amount of CD90+ cells was 75 ± 6% and the percentage of doubly positive CD34+CD90+ cells was 61 ± 4%. In contrast, in vehicle-treated controls, the CD34+CD90+ cells represented only 2 ± 2%, which were all also CD34+. This demonstrates that VPA expands the population of CD34+CD90+ cells isolated from human peripheral blood ex vivo, suggesting that it could be of critical benefit in HSCT.
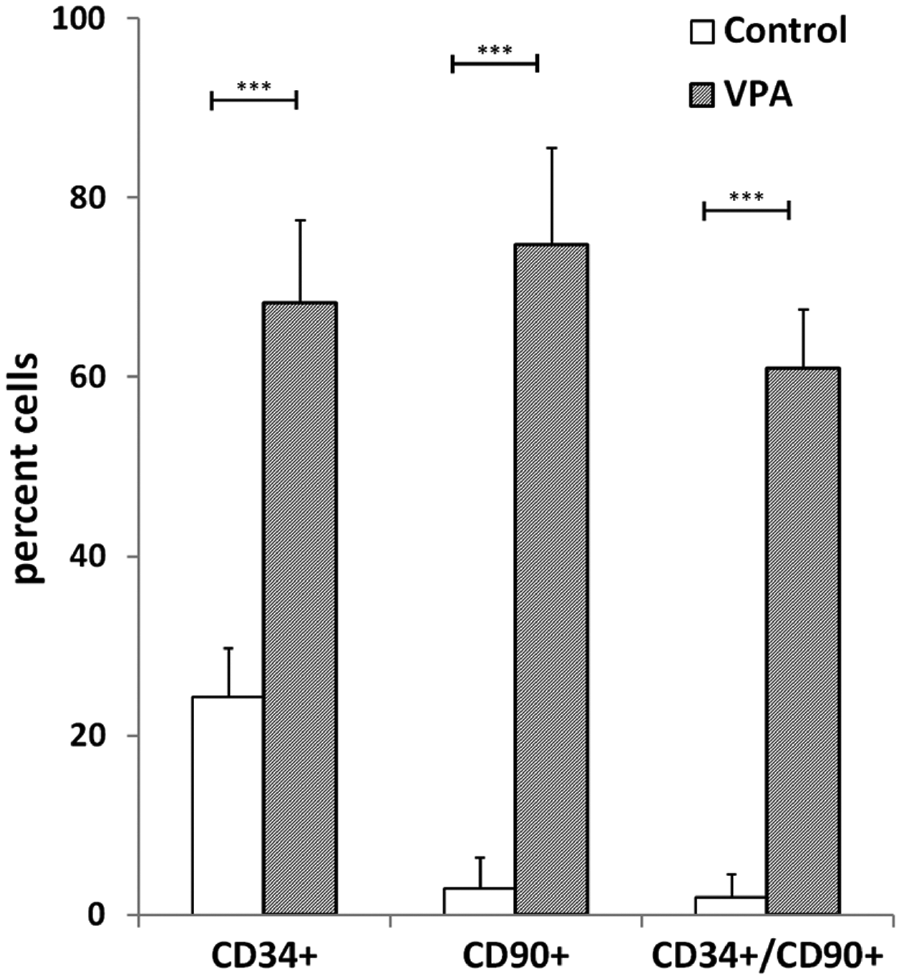
The population of human HSPCs isolated from human peripheral blood is expanded ex vivo after VPA treatment. CD34+ cells were isolated from four individual donors and treated with VPA (1 mM) or PBS. The effect on the percentage of CD34+, CD90+, and CD34+CD90+ cells was determined after 7 days of culture using FACS. VPA significantly increased the percentage of CD34+ cells (p < 0.001), CD90+ cells (p < 0.001), and CD34+CD90+ cells (p < 0.001) compared to control treatment (n = 4).
Discussion
All differentiated hematopoietic cells are derived from hematopoietic stem cells. Several pathologies, such as hematologic and lymphoid cancers, require repopulation by HSCT during therapy. A major limitation of HSCT is the paucity of engraftment-competent cells, reducing the success rate of this procedure.3,20 Therefore, a major goal in the field is to devise a method to expand engraftment-competent HSPCs ex vivo prior to HSCT. To achieve this, the identification of small molecules that expand HSCs in vivo or in vitro is crucial.
Screening for compounds expanding the HSPC population in mammals is not feasible due to technical, ethical, and financial limitations. Zebrafish is considered a suitable vertebrate model system for drug discovery and toxicology with a high degree of developmental and signaling pathway conservation for mammals. 21 Screening a large number of small molecules in zebrafish embryos is challenging for several reasons. Several zebrafish imaging screens have been published previously, but they were mostly carried out in 96-well plates and the resolution was low to allow imaging of the entire well.22–24 Screening in 384-well plates at higher magnification with automated image analysis is rare and particularly challenging.25,26 First, as zebrafish are nonadherent, robotic wash steps (e.g., after fixation) are at risk of washing the embryos away. Second, given their small size relative to the well, determining the location of the embryos for imaging is challenging. Lastly, the analysis of the rounded fish embryos in their chorion is nearly impossible for confocal microscopy and should be carried out using single plane illumination microscopy (SPIM). We adapted common plate washer protocols, allowing us to wash large three-dimensional (3D) objects without loss. By tipping the plates, we localized and oriented the embryos in a reproducible manner for imaging. This positioning and orientation step might be omitted using smart microscopy that could first localize the embryos at low resolution, determine location and orientation, and then allow higher magnification imaging. Lastly, we adapted a dechorionation protocol without mechanical intervention, 12 involving only liquid handling, allowing the rapid production of dechorionated fish embryos for plating in 384-well plates.
For image analysis, we created three macros with Fiji allowing a semiautomated quantification of the embryos. Analyzing a large number of embryos is possible with these macros, since the user-defined ROIs are rapidly created for segmenting the cells. These macros are publicly available and could be applied to different image analysis tasks.
In summary, our methods allow screening at single cell resolution in the context of the whole organism due to the use of higher magnification (10×) and 3D imaging (25–40 planes on a confocal microscope). Our adaptation of typical high-throughput screening automation technologies for compound addition, sample fixation, and sample washing minimizes manual work, reagent quantities, and time required for screening. To the best of our knowledge, such a fluorescent-based screening protocol for dechorionated zebrafish embryos in 384-well format has not been reported yet. The throughput of this approach allows screening close to a thousand embryos per week, but with some adaptation a higher throughput is possible. This protocol could also be easily adapted to other types of fluorescent-based in vivo assays and using other markers, as well as to other image analysis tasks, including 3D segmentation.
We used our procedure to perform a screen of 550 compounds from two focused libraries with known biological activities on important signaling pathways and differentiation. From the entire screen we identified five small molecules that increased the number of HSPCs in zebrafish, one of which was VPA. To validate the identified hits in a larger vessel and in wild-type embryos, WISH using a cmyb probe was performed. Four of the hits, including VPA, increase the expression of cmyb in the AGM and CHT (data shown only for VPA). Next, we demonstrated that four of the five hits increase the population of human peripheral blood stem cells (CD34+CD90+), which is considered the critical stem cell population for successful HSCT (data only shown for VPA).
Our screening approach is validated, since VPA has been previously reported to increase HSPCs. VPA was reported to induce the expansion of CD34+ progenitors in mice in a concentration-dependent manner. 27 Furthermore, colony-forming units–spleen (CFU-S) assays and competitive repopulation assays showed that VPA increases proliferation as well as self-renewal of HSCs. 16 Here, we show that VPA increases the population of CD34+CD90+ significantly. These findings are supported by a recent publication providing evidence that VPA also has the ability to increase the population of human cord blood stem cells (CD34+CD90+). 28
A well-known effect of VPA is the inhibition of histone deacetylase (HDAC). VPA induces the hyperacetylation of the N-terminal tails of histone 3 and histone 4 in vitro and in vivo. 29 In addition, VPA is indicated for treatment of epilepsy, bipolar disorders, and myelodysplastic syndromes (MDSs).30,31 However, the molecular mechanism underlying VPA’s effect on HSC expansion is still unclear.
The fact that VPA expands HSPCs from both peripheral and cord blood stem cells is of great interest, as one of the major issues in stem cell transplantation is to find a suitable cell source for each patient. The donor cells need to match the recipient immunologically, particularly the patient’s HLA antigens, rendering the search for appropriate donors challenging. 1 Peripheral blood stem cells represent an easily accessible source of immunologically matched HSPC for patients with various hematological and oncological conditions. Accordingly, the relative proportion of HSCTs using peripheral blood stem cells has increased compared to HSCTs using bone marrow. 32
Taken together, our screen identified VPA and four other small molecules that modulate hematopoiesis. These molecules are therefore candidate drugs for improving HSPC transplantation and will be further evaluated for their clinical applicability.
Footnotes
Acknowledgements
The authors would like to thank the fish facilities of CRTD and MPI-CBG for assistance with zebrafish maintenance and breeding. We thank Leonard I. Zon, Harvard Stem Cell Program, Boston Children’s Hospital, for supporting N.S. during her time as visiting scientist in his laboratory.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a CRTD seed grant and the Collaborative Research Center “SFB 655 Cells into Tissues.” Chemical compounds and libraries were kindly provided by the screening facility (TDS) of the Max Planck Institute of Molecular Cell Biology and Genetics (MPI-CBG).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
