Abstract
Although multiple culture assays have been designed to identify endothelial progenitor cells (EPCs), the phenotype of cells grown in culture often remains undefined. We sought to define and characterize the proangiogenic cell population within human peripheral blood mononuclear cells. Mononuclear cells were isolated from peripheral blood and grown under angiogenic conditions for 7 days. Formed colonies (CFU-As) were identified and analyzed for proliferation, mRNA and surface antigen expression, tube-forming ability, and chromosomal content. Colonies were composed of a heterogeneous group of cells expressing the leukocyte antigens CD45, CD14, and CD3, as well as the endothelial proteins vascular endothelial (VE) cadherin, von Willebrand’s factor (vWF), CD31, and endothelial nitric oxide synthase (eNOS). Colony cells expressed increased levels of proangiogenic growth factors, and they formed tubes in Matrigel. In comparison with colonies from the CFU-Hill assay, our assay resulted in a greater number of colonies (19 ± 9 vs. 13 ± 7; p < 0.0001) with a substantial number of cells expressing an endothelial phenotype (20.2% ± 7.4% vs. 2.2% ± 1.2% expressing eNOS, p = 0.0006). Chromosomal analysis indicated the colony cells were bone marrow derived. We, therefore, describe a colony-forming unit assay that measures bone marrow–derived circulating mononuclear cells with the capacity to proliferate and mature into proangiogenic leukocytic and endothelial-like cells. This assay, therefore, reflects circulating, bone marrow–derived proangiogenic activity.
Atherosclerosis is initiated by endothelial injury due to cardiovascular risk factors such as diabetes, hypertension, and hypercholesterolemia that results in arterial intimal inflammation, hyperplasia, and thrombosis. 1 It has been proposed that bone marrow–derived cells circulating in peripheral blood participate in both the repair of the injured endothelium and in neovascularization of ischemic tissue through either direct incorporation or via paracine effects. These cells have been collectively referred to as endothelial progenitor cells (EPCs).2–5
Different methods of identifying EPCs have been developed. 2 Colonies of cells with endothelial characteristics grown from human peripheral blood mononuclear cells in vitro under angiogenic conditions were first described by Asahara et al., 6 who regarded the colony-forming units (CFUs) to be circulating EPCs. Since then, various culture assays and flow cytometry have been developed to detect circulating EPCs, and they have been associated with endothelial function, atherosclerosis burden, and cardiovascular clinical outcomes, suggesting that they play a role in vascular health.7–9
Despite this common association with atherosclerotic disease, both the assays and the cells being measured are heterogeneous. Different blood cell isolation procedures and culture conditions result in growth varying from independent, isolated cells to colonies with various morphologies. One culture assay, the “late-outgrowth” assay, relies on long incubation periods and gives rise to rare colonies of endothelial cells.10,11 A shorter term culture assay, the CFU-Hill, gives rise to cells of a monocyte-macrophage lineage with potent proangiogenic paracrine effects but no apparent endothelial cells.10,12 EPCs identified using flow cytometry via the markers CD34, CD133, and vascular endothelial growth factor receptor 2 (VEGFR2) also appear to be of the monocyte-macrophage lineage, and no consensus on cell markers specific for EPCs has been reached. 13
We sought to develop an assay to identify cells circulating in the peripheral blood with angiogenic and vascular repair capacity. Herein, we describe the angiogenic colony-forming unit (CFU-A) assay, which identifies bone marrow–derived peripheral blood mononuclear cells capable of proliferating into colonies of cells that express endothelial and leukocyte phenotypes and produce angiogenic factors.
Materials and Methods
Study subjects
Healthy volunteers without chronic diseases, hypercholesterolemia (low-density lipoprotein [LDL] cholesterol level <120 mg/dL), hyperglycemia (glucose <99 mg/dL), hypertension (blood pressure <135 mm Hg systolic and <85 mm Hg diastolic), or obesity (body mass index 19–26) and with at least a 5-year nonsmoking status were recruited. In addition, 4 patients who had sex-mismatched bone marrow transplants were studied. Venous blood was drawn after an overnight fast. The study was approved by the Emory University Institutional Review Board, and all subjects gave informed consent.
CFU assay
Our CFU-A assay was modified from previous assay descriptions.7,14 Mononuclear cells were isolated from 16 mL of whole blood by density-gradient centrifugation using CPT tubes (Becton Dickinson, Franklin Lakes, NJ), washed, and resuspended in growth medium (Dulbecco’s modified Eagle’s medium [DMEM] supplemented with 20% fetal bovine serum and 6.5% endothelial cell growth supplement [ECGS]; Becton Dickinson). To eliminate potential contamination by mature circulating endothelial cells, we plated the cells in 6-well culture dishes coated with human fibronectin (Biocoat; Becton Dickinson), and after 24 h, nonadherent cells were replated onto new fibronectin-coated 24-well dishes (1 million cells/well). Growth medium was changed every 2 days, and colonies/well were counted 7 days after plating. A colony was identified as multiple thin, flat cells emanating from a central cluster of rounded cells.6,7 Reproducibility was tested in 15 blood samples drawn 1 week apart from the same individuals. The overall correlation between the repeated assays was 0.84 (p < 0.001).
The commercially available CFU-Hill assay (Endocult; Stem Cell Technologies, Vancouver, Canada) was performed per the manufacturer’s directions for comparison.7,15 Both assay colonies were counted by a single, blinded observer in a minimum of four wells. Average number of colonies per 1 million mononuclear cells is reported.
Proliferation assay
To determine whether the colonies were derived from mononuclear precursor cells via proliferation, 5-bromo-2′-deoxyuridine (BrdU; Sigma, St. Louis, MO) (1 µg/mL) was used to label cells for 24 h on day 3 or 6 of growth. Subsequently, immunostaining with anti-BrdU (DAKO, Carpinteria, CA) was used to detect proliferating cells within the colonies (see below).
Reverse transcriptase polymerase chain reaction (RT-PCR)
RNA was isolated from freshly isolated mononuclear cells and from 7-day colonies using the Qiagen RNeasy Mini Kit (Qiagen, Valencia, CA). cDNA was prepared from RNA samples and RT-PCR was performed in an ABI 7900 (Applied Biosystems, Foster City, CA). RT-PCR was performed in an ABI 7900 (Applied Biosystems, Foster City, CA) instrument using pathway-focused gene expression PCR arrays from Super Array (human endothelial biology, angiogenesis). mRNA expression levels were determined using SYBR Green–based real-time PCR. Results were analyzed using the PCR Array data analysis web portal to convert threshold cycles into fold-changes.
Immunocytochemistry
We performed immunocytochemistry for leukocyte and endothelial antigens. 7 Colonies were fixed in 2% paraformaldehyde for 1 h, washed in phosphate-buffered saline (PBS), and blocked with 5% normal serum in 2% bovine serum albumin (BSA) in PBS for 30 min. Cells were incubated with the primary antibody (mouse anti-CD31 1:300; eBioscience, San Diego, CA), mouse anti-CD45 1:5000 (eBioscience), mouse anti-CD3 1:300 (eBioscience), mouse anti-CD14 1:200 (Santa Cruz Biotechnology, Santa Cruz, CA), mouse anti–vascular endothelial (VE) cadherin 1:50 (Abcam, Cambridge, UK), rabbit polyclonal anti–endothelial nitric oxide synthase (eNOS) 1:1000 (Santa Cruz Biotechnology), and rabbit polyclonal anti–von Willibrand’s factor (vWF) 1:1000 (Chemicon, Temecula, CA) in 2% BSA for 1 h and then with biotinylated horse anti-mouse IgG for monoclonal primary antibodies and biotinylated goat anti-rabbit IgG for polyclonal antibodies (1:200) (Vector Laboratories, Burlingame, CA) for 30 min. After another PBS wash, cells were stained with streptavidin-conjugated quantum dots (QDot 605; Invitrogen, Carlsbad, CA), 1:100, for 1 h and counterstained with Hoechst nuclear stain. Images were acquired on a Zeiss LSM 510 confocal microscope (Carl Zeiss, Jena, Germany).
Flow cytometry
To detect the surface expression of endothelial and hematopoietic lineage marker proteins, harvested colonies from 24-well plates were suspended in PBS after washing and incubated for 1 h in the presence of the anti-CD14, -CD3, -CD45, -eNOS, -vWF, or -VE cadherin and then conjugated with biotinylated secondary antibodies. After washing with PBS, cells were stained with streptavidin-conjugated quantum dots (QDot 605; Invitrogen), 1:100, and analyzed by flow cytometry (Facs Calibur; Becton-Dickinson).
Matrigel tube formation assay
In 24-well fibronectin-coated culture dishes, 250 µL of the 1:1 Matrigel:PBS solution was applied per well and allowed to polymerize at 37 °C for 30 min to 2 h. After polymerization, 106 preplated, nonadherent mononuclear cells or cells from 4-day-old colonies were plated in each well with 1 mL DMEM and 20% FBS, 6.5% ECGS and observed for tube formation for up to 5 days.
Cell migration assay
CFU cell migration from 16 subjects was assayed using the BD Biocoat Angiogenesis Endothelial Cell Invasion System (Becton Dickinson). CFU cells (50 000/well) in culture medium were distributed into a 24-well multiwell insert/receiver plate containing a fluorescence-blocking microporous polyethylene terephthalate membrane (3.0 µm pore size) evenly coated with human fibronectin, vascular endothelial growth factor (VEGF; 50 ng/mL), and stromal cell–derived factor 1 alpha (SDF-1α) (50 ng/well), which were used as chemoattractants. To quantitate the number of cells that migrated through the pores and attached to the underside of the insert membrane, cells were labeled with a fluorescent dye and were measured using a bottom-reading fluorescent plate reader.
Fluorescent in situ hybridization
Peripheral blood mononuclear cells from four sex-mismatched bone marrow transplant recipients were cultured via the CFU-A colony assay protocol above. At 7 days, fluorescent in situ hybridization (FISH) was performed on the colonies and on interphase cells from a concurrent peripheral blood chromosome culture. The Vysis CEP X Spectrum Orange/Y Spectrum Green Direct Labeled Fluorescent DNA Probe Kit (#30-161059; Abbott Laboratories, Abbott Park, IL) was used to detect the presence of X and/or Y chromosomes in the colonies and interphase cells. A G-banded metaphase karyotype was also produced as a confirmation of FISH results.
Statistical analysis
To test for differences among groups with respect to continuous variables, we used the paired or unpaired t-test, as appropriate. Correlations between cell counts were made using linear regression analysis; p-values ≤0.05 were considered statistically significant. Data analysis was performed with SPSS software, version 14.0 (SPSS, Inc., an IBM Company, Chicago, IL).
Results
CFU assay comparison
We compared the CFU-A assay with the commercial CFU-Hill assay, which has been previously studied.10,16 Mononuclear cells were isolated from peripheral blood from 59 subjects (mean age, 45 ± 11 years) and grown in vitro using both assays. Colonies were identified as clusters of rounded cells surrounded by spindle-shaped cells at the periphery under the microscope and were similar in morphology in both assays ( Fig. 1A ). Colony counts ranged from 3 to 48 for the CFU-A assay and from 1 to 34 for the CFU-Hill assay. Significantly more colonies grew from the CFU-A assay than the CFU-Hill assay (19 ± 9 vs. 13 ± 7; p < 0.0001). Correlation was poor (r2 = 0.08, p = 0.03), suggesting potentially important differences between the two assays.
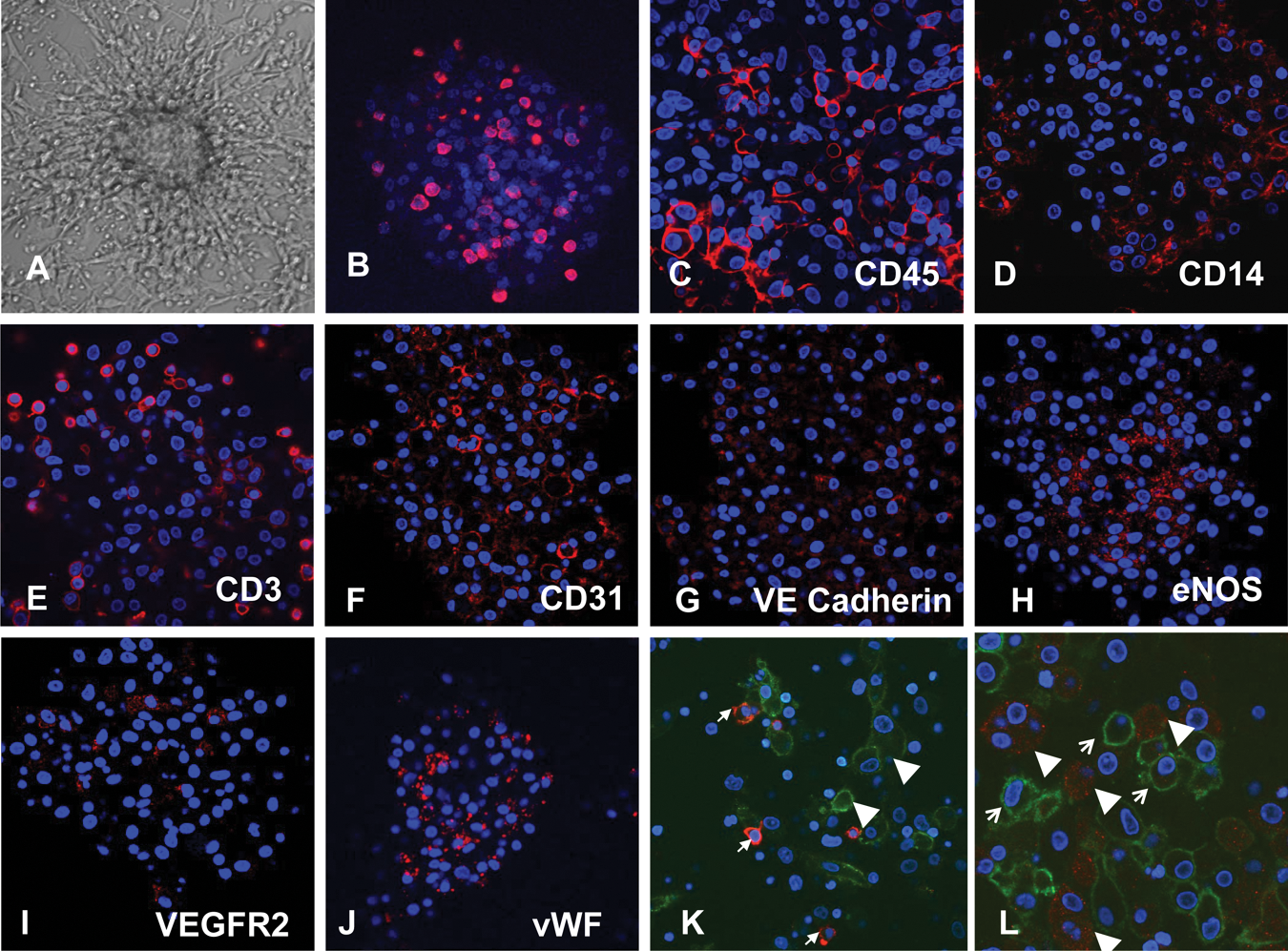
Angiogenic colony-forming unit (CFU-A). (
Colony cell proliferation
Proliferative potential is considered one criterion for progenitor cells. To determine whether CFU-A cells proliferate, we studied Brd-U incorporation culture days 3 and 6. Brd-U immunostaining on both days 4 and 7 revealed proliferating cells interspersed throughout the colony ( Fig. 1B ). Levels of proliferating cells appeared similar at both time points (data not shown).
RT-PCR
To analyze the proangiogenic potential and endothelial lineage of the developing CFU-A colonies, we compared RNA expression of stem cell–, angiogenesis-, and endothelium-related proteins in the isolated mononuclear cells with the mature CFU-A colonies. The expression of 30 different proangiogenic or endothelial genes was upregulated up to 210-fold in the CFU-A cells compared with mononuclear cells ( Table 1 ). In particular, we found a 5-, 7-, and 11-fold increase in expression of the endothelial-specific proteins CD31, eNOS, and vWF RNA, respectively, in the mature colony as compared with mononuclear cells. These data support the notion that CFU-A colonies contain cells with endothelial and proangiogenic characteristics. In addition, we found that the expression of certain stem cell–related genes was downregulated (AGC1 [0.12, p = 0.002], CD8A [0.10, p = 0.01], CD8B [0.09, p = 0.01], COL2A1 [0.27, p = 0.02], DLL [0.13, p = 0.14], FRAT1 [0.23, p = 0.04], MYC [0.17, p = 0.04], and NCAM1 [0.11, p = 0.04]), suggesting that progenitor cell differentiation occurring during the culture resulted in the endothelial and proangiogenic cells.
RNA Expression of Select Proteins in CFU-A Colonies as Compared with Circulating Mononuclear Cells in Volunteer Subjects
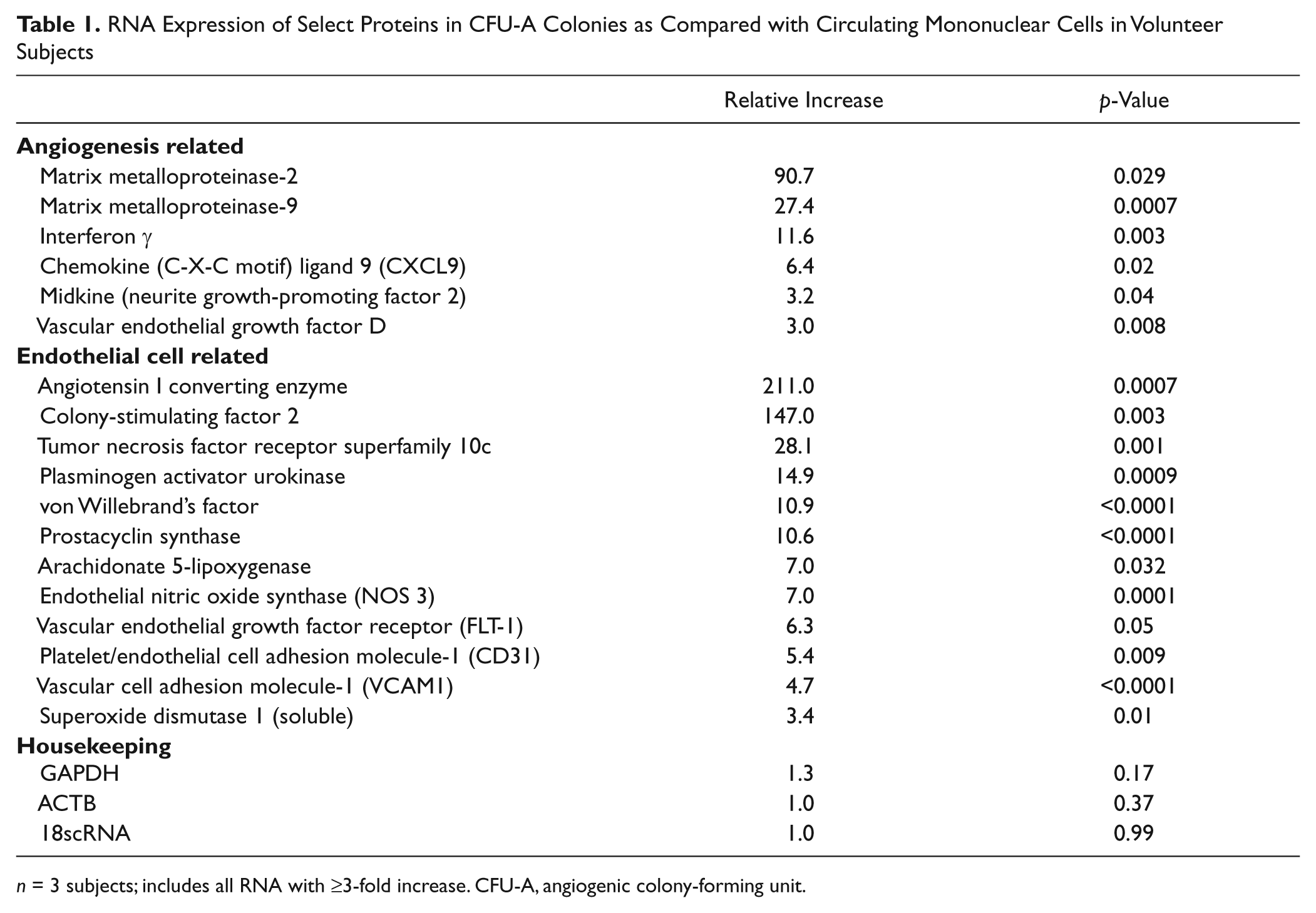
n = 3 subjects; includes all RNA with ≥3-fold increase. CFU-A, angiogenic colony-forming unit.
Immunocytochemistry
Immunocytochemistry for leukocyte and endothelial proteins was performed on the colonies. Many CFU-A colony cells stained for the leukocyte antigen CD45, whereas subgroups of cells, primarily located at the periphery, stained for the monocyte-antigen CD14 and lymphocyte-antigen CD3. Staining for endothelial antigens CD31, VEGFR2, VE-cadherin, and vWF also occurred throughout the colony, whereas eNOS staining occurred primarily centrally ( Fig. 1C – J ). Double staining demonstrated that cells that stained for vWF or eNOS did not stain for CD14 ( Fig. 1K , L ). These observations suggest that CFU-A colonies are composed of leukocytes of monocytic and lymphocytic lineage, as well as of endothelial-like cells. In comparison, the CFU-Hill colonies subjectively appeared to stain primarily for monocyte and lymphocyte markers and only sparsely for eNOS ( Fig. 2 ).
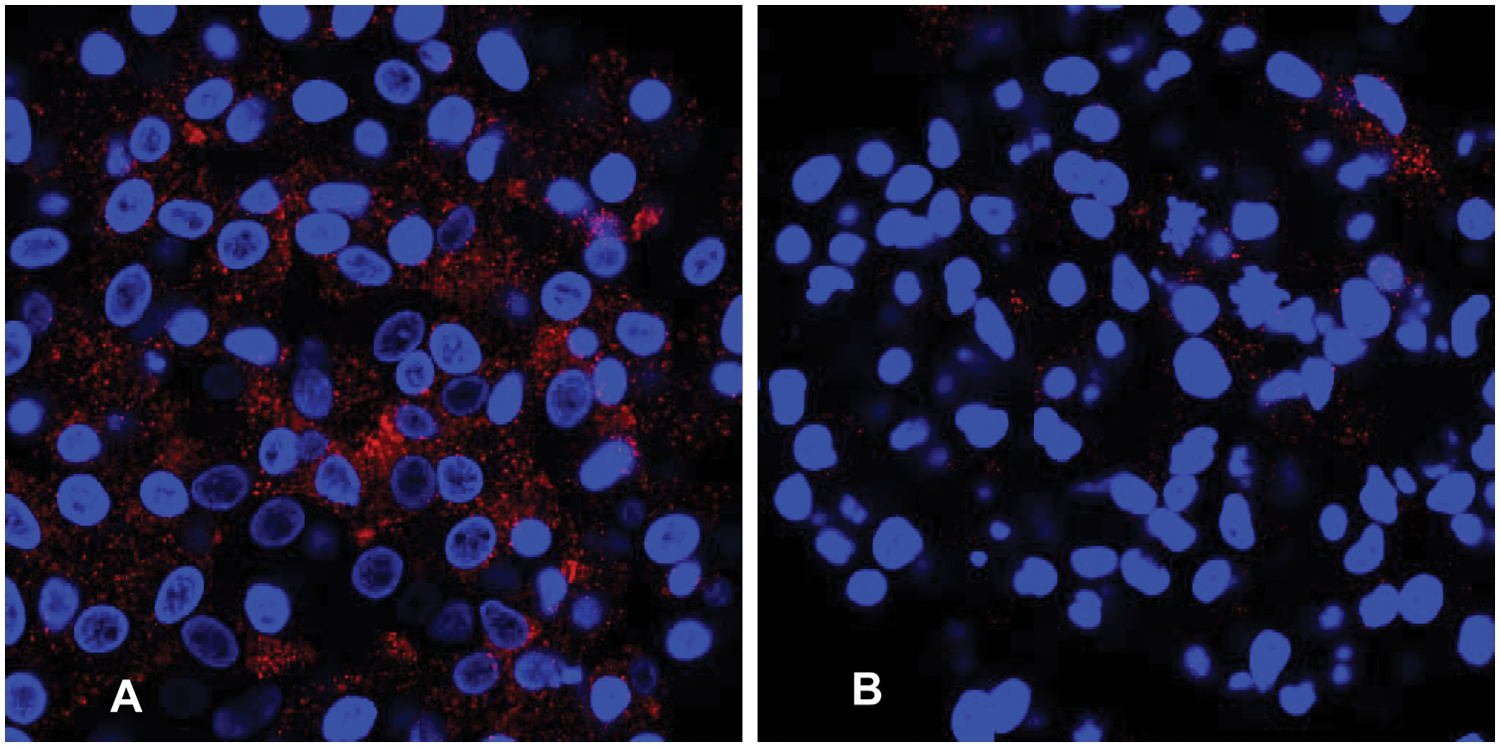
Immunostaining for endothelial nitric oxide synthase (eNOS) demonstrating increased staining in the angiogenic colony-forming unit (CFU-A) (
Flow cytometry
Quantification of cell types in the CFU-A colonies at day 7 using flow cytometry demonstrated that the majority of cells expressed CD45 (80%) and CD3 (60%) antigens. However, a sizable minority (20%) also expressed the endothelial markers eNOS and vWF. In contrast, only 2% of cells in the CFU-Hill colonies expressed the endothelial-specific proteins eNOS and vWF (p < 0.001; Table 2 ).
Comparison of Cell Frequency in CFU-A vs. CFU-Hill Colonies from Volunteer Subjects Measured by Flow Cytometry
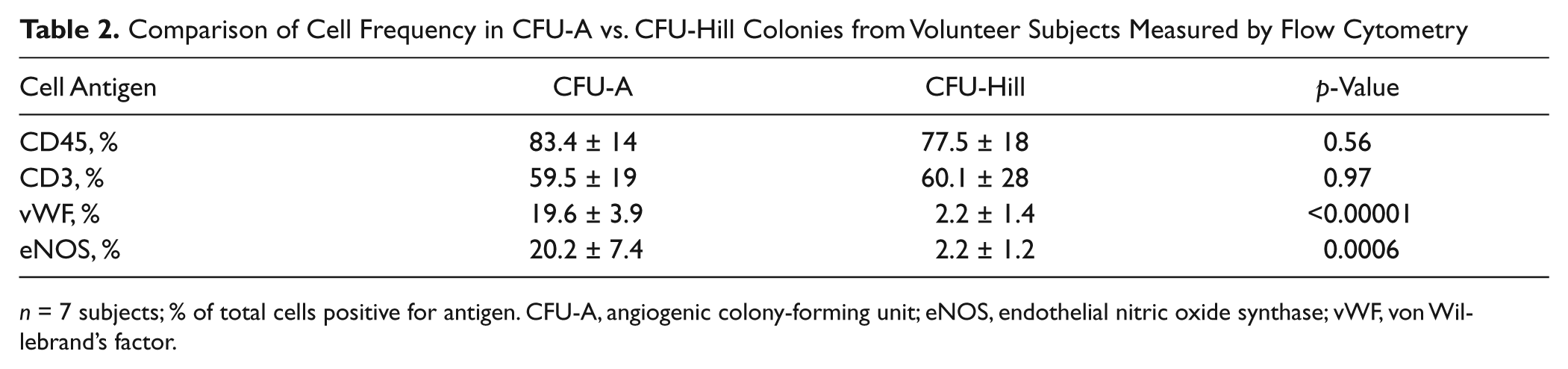
n = 7 subjects; % of total cells positive for antigen. CFU-A, angiogenic colony-forming unit; eNOS, endothelial nitric oxide synthase; vWF, von Willebrand’s factor.
Tube-forming assay
To assess the angiogenic capacity of the circulating mononuclear cells and the resultant CFU-A colony-derived cells, we performed Matrigel tube-forming assays on nonadherent mononuclear cells as well as maturing colonies. The mononuclear cells formed networks of tubes that contained distinct lumens as assessed by electron microscopy. Furthermore, when individual maturing colonies were transferred to Matrigel, tube-like structures emerged from their centers at 24 h ( Fig. 3 ).
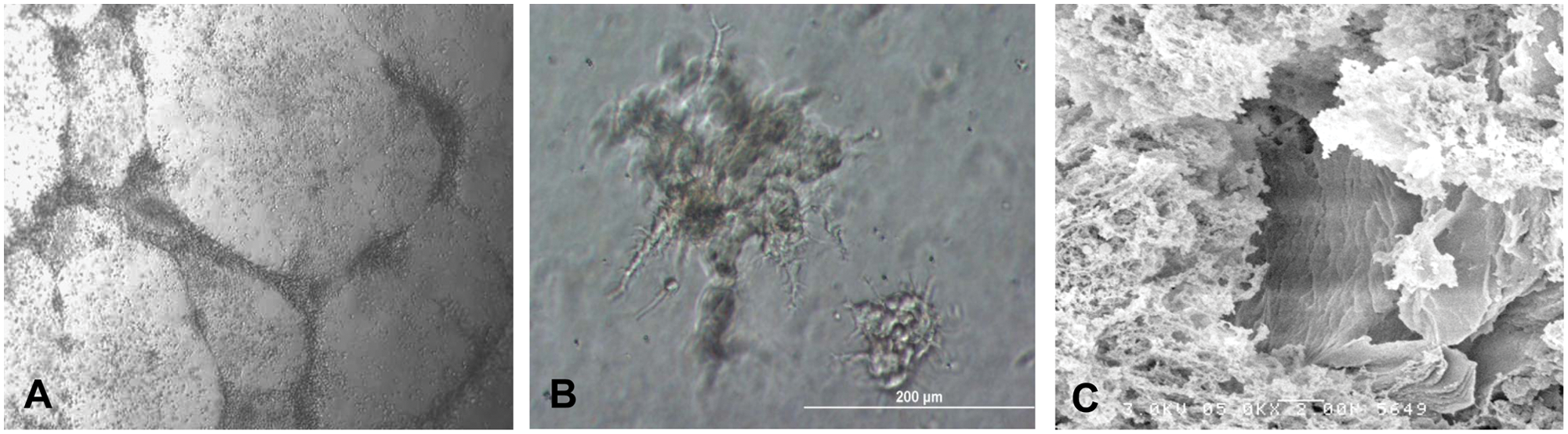
Matrigel tube-forming assay. (
Cell migration assay
To assess the response to specific proangiogenic stimuli, we measured migration of CFU cells across a porous membrane. Cell migration was significantly stimulated by VEGF and SDF-1α as compared with control (21 [p < 0.00001] and 41 [p = 0.04] vs 15 cells/well).
Sex-mismatched bone marrow transplant (BMT) subjects
To determine the origin of the CFU-A cells, we studied three female BMT recipients who had male donors. All cells in the colonies as well as the interphase and metaphase cells were male, having an X and a Y chromosome ( Fig. 4 ). A fourth male BMT recipient who had a female donor was studied. All of this patient’s cells in colonies, interphase cells, and metaphase cells were female, having two X chromosomes. These results confirm the bone marrow origin of the CFU-A colonies.
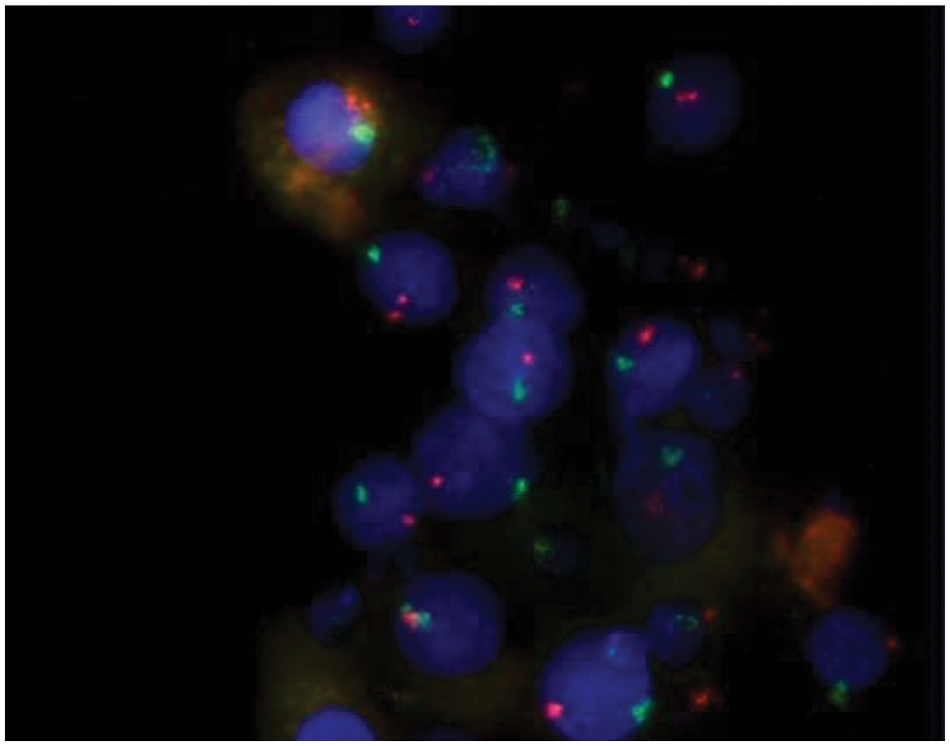
Fluorescent in situ hybridization demonstrating the bone marrow origin of angiogenic colony-forming unit (CFU-A) colony cells. Colony cells staining for Y chromosomes (green) indicate they are from the bone marrow of a female subject who received a male donor bone marrow transplant. X chromosome = orange. Nuclear counterstaining with Hoechst (blue). Typical examples from n = 4 volunteer subjects (magnification ×63).
Discussion
Peripheral blood mononuclear cells contribute to angiogenesis and vascular repair, processes important in the pathogenesis of atherosclerosis and ischemic heart disease, and have been collectively called EPCs. Culture assays have been used to identify these cells, but culture conditions and outgrowth morphology have varied considerably between the assays, and not all assays have been optimally characterized. We sought to characterize a distinct culture assay, the CFU-A assay. We demonstrate that CFU-A are peripheral blood mononuclear cells that (1) are bone marrow derived, (2) are proliferative, and (3) give rise to heterogeneous cell colonies containing both endothelial- and hematopoietic-like cells, which have (4) significantly upregulated proangiogenic protein expression and (5) the ability to form capillary-like tubes. Thus, the CFU-A assay reflects circulating, bone marrow–derived, proangiogenic cell activity.
The CFU-A assay appears to be different from other popular EPC culture assays. The “late outgrowth” assay identifies circulating proliferative cells that form homogeneous cobblestone colonies of endothelial cells.10,11 However, these circulating cells are extremely rare, if present at all, in the adult human circulation and may originate from the vessel wall instead of the bone marrow.17,18 Hill et al. 7 described a shorter duration CFU assay based on the work of Asahara et al., 6 and a commercial variant of this assay, called the CFU-Hill, was developed. This assay was originally believed to reflect true EPCs and was associated with endothelial function and atherosclerotic disease in patients. 9 However, since then, investigators have demonstrated that the CFU-Hill assay yields almost exclusively hematopoietic-like cell colonies, with almost no eNOS expression and no in vitro capillary tube formation capability.10,16 Instead, these bone marrow–derived progenitor cells facilitate angiogenesis through paracrine mechanisms. 12 Although the CFU-A assay grows colonies morphologically similar to the CFU-Hill assay, the CFU-A colonies contain both hematopoietic-like cells as well as endothelial-like cells. Importantly, almost 20% of the cells express endothelial-specific antigens vWF and eNOS (compared with 2% of the CFU-Hill colony cells), and the cells migrate in response to the angiogenic stimulant VEFG and form capillary-like tubes in Matrigel, indicating their vasculogenic potential. Our immunocytochemical double-staining experiments further supported endothelial lineage of some colony cells by showing that they were not monocyte/macrophage lineage cells expressing an endothelial phenotype. In addition, almost 60% more colonies formed using the CFU-A than the CFU-Hill assay, with little correlation in CFU number between these assays. We have also shown a robust and distinct relationship between the CFU-A assay and clinical atherosclerosis, which differs substantially from that of the CFU-Hill assay. 19 Finally, unlike the late-outgrowth colony assay cells, all the CFU-A colony cells are bone marrow derived.
The differences between the CFU-A and the other EPC assays are likely due to different isolation and culture conditions resulting in different circulating cell selection, growth pattern, and phenotype manifestation. Important differences include (1) the culture duration (7 vs. 5 days for the CFU-Hill assay vs. ≥2 weeks for the late-outgrowth assay), (2) the elimination of early adherent cells (potential mature endothelial cells) (24 h vs. 48 h for the CFU-Hill assay vs. no elimination for the late-outgrowth assay), and (3) the culture media used (DMEM with 20% FBS and 6.5% EGCS vs. Endocult media for the CFU-Hill with a proprietary formulation). These differences have likely resulted in the substantially different colony characteristics we observed.
The different cell phenotypes found in CFU-A colonies support the potential proangiogenic role of CFU-A. An effective bone marrow–derived reparative response to vascular injury involves multiple cell types, including both hematopoietic and endothelial-like cells, with some cells offering only paracrine stimulation and others incorporating directly into the injured vessel.16,20–22 The appearance of both hematopoietic and endothelial-like cells in a common colony may reflect their common precursor, the hemangioblast, which can differentiate into both lineages. 23 Alternatively, the CFU-A colonies may be derived cooperatively from more differentiated precursor cells, potentially including monocytes that develop a vascular phenotype and have been shown to be involved in angiogenesis and vascular repair. 24 Either way, the culture technique and the resultant biphenotypic CFU-A colony growth may reflect the bone marrow–derived vascular repair activity more comprehensively than other more selective techniques.
There are some limitations of this study. Endothelial-like cells were identified based on some, but not all, relevant endothelial markers. Double immunostaining showed that cells expressing endothelial antigens did not coexpress monocyte-antigen CD14, but we do not definitively distinguish between endothelial-like cells derived directly from their primitive endothelial progenitors and those derived from more differentiated myeloid cells.10,16,25 We have shown some differences between the CFU-A and the CFU-Hill assays, but further comparison of molecular expression profiling could provide additional insight. We have not quantified whether the amount of protein being expressed in this in vitro assay correlates with in vivo angiogenesis, nor have we demonstrated any in vivo evidence that CFU-A are directly involved in angiogenesis or vascular repair, both subjects that need further study. However, we do provide compelling evidence of the association between the CFU-A and the development and outcomes of clinical atherosclerotic disease. 19
In conclusion, we describe a culture-based assay that is able to reproducibly measure circulating, bone marrow–derived, proangiogenic cells and is distinct from the other EPC assays. We believe it will be useful in measuring vascular health, the risk of developing cardiovascular disease, and the testing of proangiogenic compounds.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Eli Lilly and Company, Indianapolis, Indiana; a gift from the Marcus Foundation, Atlanta, Georgia; the Georgia Tech/Emory Center for the Engineering of Living Tissues (GTEC) National Science Foundation (NSF) Grant EEC-9731643; PHS Grant UL1 RR025008 from the Clinical and Translational Science Award program, National Institutes of Health, National Center for Research Resources; and the Woodruff Fund.
