Abstract
Vascular injury mobilizes bone marrow–derived proangiogenic cells into the circulation, where these cells can facilitate vascular repair and new vessel formation. We sought to determine the relationship between a new biomarker of circulating bone marrow–derived proangiogenic cell activity, the presence of atherosclerotic cardiovascular disease (CVD) and its risk factors, and clinical outcomes. Circulating proangiogenic cell activity was estimated using a reproducible angiogenic colony-forming unit (CFU-A) assay in 532 clinically stable subjects aged 20 to 90 years and ranging in the CVD risk spectrum from those who are healthy without risk factors to those with active CVD. CFU-A counts increased with the burden of CVD risk factors (p < 0.001). CFU-A counts were higher in subjects with symptomatic CVD than in those without (p < 0.001). During follow-up of 232 subjects with CVD, CFU-A counts were higher in those with death, myocardial infarction, or stroke than in those without (110 [70–173] vs 84 [51–136], p = 0.01). Therefore, we conclude that circulating proangiogenic cell activity, as estimated by CFU-A counts, increases with CVD risk factor burden and in the presence of established CVD. Furthermore, higher circulating proangiogenic cell activity is associated with worse clinical outcome in those with CVD.
Atherosclerosis is initiated by endothelial cell injury due to factors such as smoking, diabetes, hypertension, and hypercholesterolemia. Arterial intimal inflammation, hyperplasia, and thrombosis result, leading to tissue ischemia, infarction, and death. Experimental models have demonstrated a robust physiologic response to endothelial injury and tissue ischemia characterized by mobilization of bone marrow–derived cells with the capacity to stimulate new endothelial and/or blood vessel growth into the circulation.1,2 The role of these proangiogenic cells—collectively referred to as endothelial progenitor cells (EPCs)—in determining clinical atherosclerotic cardiovascular disease (CVD) outcome has recently been studied.1–4 These studies have generally involved small numbers of heterogeneous patients with risk factors or established CVD. Furthermore, these studies have been hampered by challenges in accurately defining and measuring EPCs. 3 Flow cytometric methods designed to count EPCs are impaired by the lack of a specific cell marker. 5 Culture assays that rely on the growth of colonies from EPCs vary importantly in technique, with different assays giving rise to different cell types, ranging from bone marrow–derived proangiogenic hematopoietic cells to vessel wall–derived endothelial cells.6,7 Thus, clinical studies performed to date have yielded conflicting data regarding the association of circulating proangiogenic cells with CVD risk factors and disease burden.8,9 Furthermore, circulating proangiogenic cells have not been well explored as a predictive CVD biomarker.
In this study, we sought to understand the relationship between CVD and circulating bone marrow–derived proangiogenic cells, as assessed by a culture-based colony formation assay in a large population of subjects, ranging from individuals who are healthy and without cardiovascular risk factors to patients with established CVD. The details of this assay, called the angiogenic colony-forming unit (CFU-A) assay, have been described. 10 Specifically, we investigated whether the frequency of CFU-As cultured from peripheral blood are (a) modulated by risk factors for CVD and whether their numbers predict (b) the presence of CVD and (c) future adverse CVD events.
Materials and Methods
Study Subjects
Subjects with and without CVD were recruited from advertisements and ambulatory care units. Subjects with CVD had a >50% angiographic stenosis of one or more major epicardial coronary or peripheral arteries, with clinical evidence of myocardial or peripheral ischemia based on symptoms and/or noninvasive stress testing. Patients without CVD had no history of symptomatic or asymptomatic myocardial or peripheral ischemia and/or infarction. Patients were excluded if they had a myocardial infarction (creatine kinase–myocardial band [CK-MB] ≥2× upper limit of normal), change in statin therapy, surgery, infection, steroid use within the prior 3 months, or any chronic inflammatory disease, cancer, treatment with bone marrow stimulants/chemotherapy, or chronic renal insufficiency (creatinine >2.5 mg/dL). Blood for the CFU-A assay was drawn in a fasting state. The study was approved by the Emory University Institutional Review Board, and all subjects gave informed consent.
Definition of Risk Factors for CVD
In addition to age, risk factors assessed included hypertension, hypercholesterolemia, diabetes, and active smoking (within 1 month). Hypertension was defined as blood pressure >140 mm Hg systolic or >90 diastolic, or medical therapy for documented hypertension. Hypercholesterolemia was defined as low-density lipoprotein (LDL) >120 mg/dL or medical therapy for hypercholesterolemia. Diabetes was defined as plasma glucose ≥126 mg/dL or medical therapy for diabetes.
Proangiogenic Cell Assay
The CFU-A assay is a culture assay that quantifies the number of mononuclear proangiogenic cells and their functional ability to grow and form colonies in vitro.8–11 In brief, mononuclear cells were isolated by density-gradient centrifugation from a 20-mL sample of blood using CPT (Becton Dickinson, Franklin Lakes, NJ) tubes, suspended in growth medium (Dulbecco’s modified Eagle’s medium supplemented with 20% fetal bovine serum and 6.5% endothelial cell growth supplement), and plated on dishes coated with human fibronectin (Biocoat; Becton Dickinson). To eliminate mature circulating endothelial cells, cells adherent after 24 h were discarded, and nonadherent cells were replated onto new fibronectin-coated six-well plates (1 million cells/well). Growth medium was changed every 2 days. Colonies, identified as multiple thin, flat cells emanating from central clusters of rounded cells, were counted 7 days after plating.8,12 Colonies were counted manually in six wells by an observer who was blinded to the clinical data and reported as the mean number of CFU-As per 1 million cells. Immunocytochemistry and flow cytometry of CFU-A cells demonstrated cells expressing both leukocyte and endothelial antigens (80% of cells expressing CD45, 20% of cells expressing endothelial nitric oxide synthase and von Willebrand’s factor), reverse transcriptase demonstrated high levels of expression of proangiogenic factors, and capillary-like tubes formed in culture. Reproducibility was tested by measuring CFU-A count in 15 blood samples drawn 1 week apart from the same individuals. The overall correlation between the repeated assays was 0.84, and the mean number of colonies in each of the duplicates was 48.4 ± 4.1 versus 50.4 ± 2.9.
Clinical Outcome Data Collection
Adverse outcome assessments for death, myocardial infarction, stroke, or coronary revascularization were obtained by telephone interview, examination of hospital and clinic encounters, and linkage with the Social Security Index for death records in subjects with CVD. Cardiovascular death was defined as death due to a myocardial infarction, cerebrovascular accident, sudden cardiac death, or congestive heart failure. Myocardial infarction was defined as an elevation of cardiac necrosis markers (troponin and/or CK-MB levels two times the upper limit of normal) plus ischemic symptoms and/or new ST-segment elevation (0.1 mV) in at least two contiguous leads. Stroke was defined as clinical symptoms in addition to radiological evidence of stroke without intracranial hemorrhage. Coronary revascularization was defined as percutaneous coronary intervention or coronary artery bypass grafting. For subjects experiencing two or more different acute events, all events were considered in the analysis. All events were independently verified by two experienced clinicians using available medical records.
Statistical Analysis
Continuous variables are expressed as mean value ± SD, whereas categorical variables are expressed as absolute and/or relative frequencies. All continuous variables were tested for homogeneity of variance and normal distribution before any statistical analysis was applied, by using the Kolmogorov-Smirnov criterion. For skewed variables, data are expressed as median value (25th–75th percentile), and logarithmic transformation was performed prior to analysis.
To test for differences among groups with respect to continuous variables, we used the unpaired t-test, Mann-Whitney U-test, or one-way analysis of variance (ANOVA) as appropriate. Categorical variables were tested with the χ2 test. Correlations between continuous variables were evaluated by calculation of the Pearson correlation coefficient. Multivariable analysis adjusting for potential confounders was performed by either analysis of covariance (ANCOVA) or linear regression analysis (backward elimination method). The assumptions for linearity and homoscedasticity were tested based on the standardized residuals plots.
Receiver-operator characteristic (ROC) curve analysis was used to identify the cutoff level for CFU-As best associated with presence and absence of CVD. In addition, ROC curve analysis was used to identify the optimal level of CFU-As for predicting the occurrence of future cardiovascular events during follow-up.
The area under the ROC curve was used as an estimate of the overall performance, considering that an area of 0.5 indicates no ability to discriminate CVD. The association of CFU-A count with the presence of CVD or the occurrence of cardiovascular events was assessed with estimation of the value that maximizes sensitivity and specificity, corresponding to the point of the ROC curve that is closest to the left upper angle. Specifically, for outcomes during the follow-up, separate ROC curve analyses were plotted for (1) the composite end point of death plus myocardial infarction (MI) plus stroke and (2) the composite end point of death plus MI plus stroke plus revascularization. Then, cumulative event-free survival was evaluated both by univariate analysis (log rank test and Kaplan-Meier analysis) and multivariate analysis (Cox proportional regression analysis). Crude and adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) were used to evaluate the relative risk for adverse cardiac events during the follow-up. Exact p-values ≤0.05 were considered statistically significant. Data analysis was performed with SPSS software, version 14.0 (SPSS, Inc., an IBM Company, Chicago, IL).
Results
Subject Characterization
Of the 532 subjects (age 55.0 ± 13.6 years; range, 20–90 years), 243 had a history of CVD ( Table 1 ). One hundred seventy-nine subjects had no CVD risk factors (diabetes, hypertension, hyperlipidemia, and smoking), 99 had 1 risk factor, 111 had 2 risk factors, 103 had 3 risk factors, and 40 subjects had 4 risk factors. CFU-A counts ranged from 1 to 406 (median, 57; interquartile range, 25–99).
Clinical Characteristics of Subjects
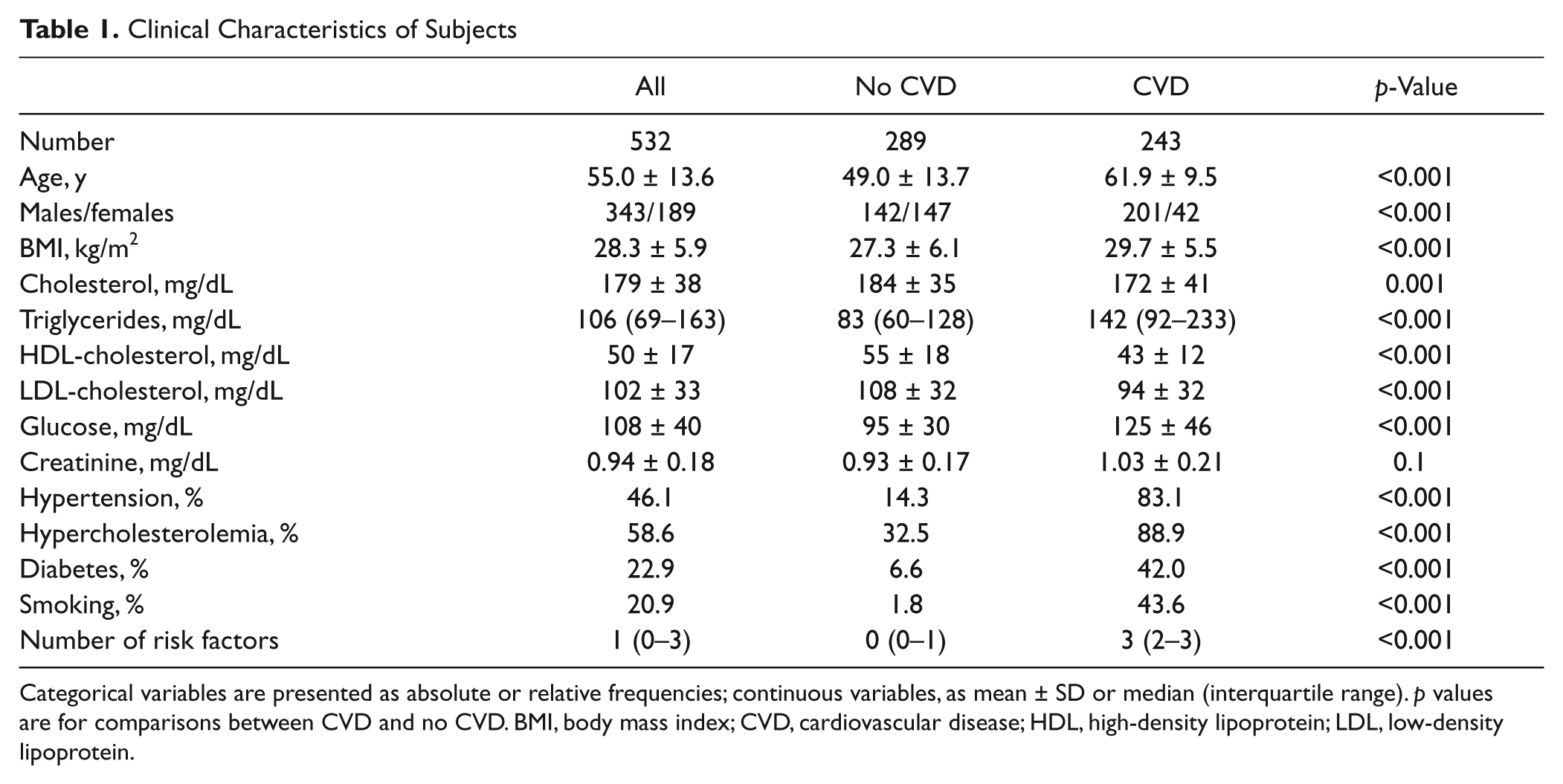
Categorical variables are presented as absolute or relative frequencies; continuous variables, as mean ± SD or median (interquartile range). p values are for comparisons between CVD and no CVD. BMI, body mass index; CVD, cardiovascular disease; HDL, high-density lipoprotein; LDL, low-density lipoprotein.
CFU-A Association with CVD and Its Risk Factors
CFU-A counts increased significantly with age (r = 0.50, p < 0.001; Fig. 1 ) and were significantly higher in men than in women (p < 0.001). CFU-A counts were also significantly higher in subjects with hypercholesterolemia (p < 0.001), diabetes (p < 0.001), smoking (p < 0.001), and hypertension (p < 0.001) ( Fig. 1 ), but the correlation with body mass index (BMI) was weak (r = 0.09, p = 0.057). After correction for age and sex, the association between CFU-A count and hypercholesterolemia, smoking, diabetes, and hypertension remained significant (all p < 0.001).
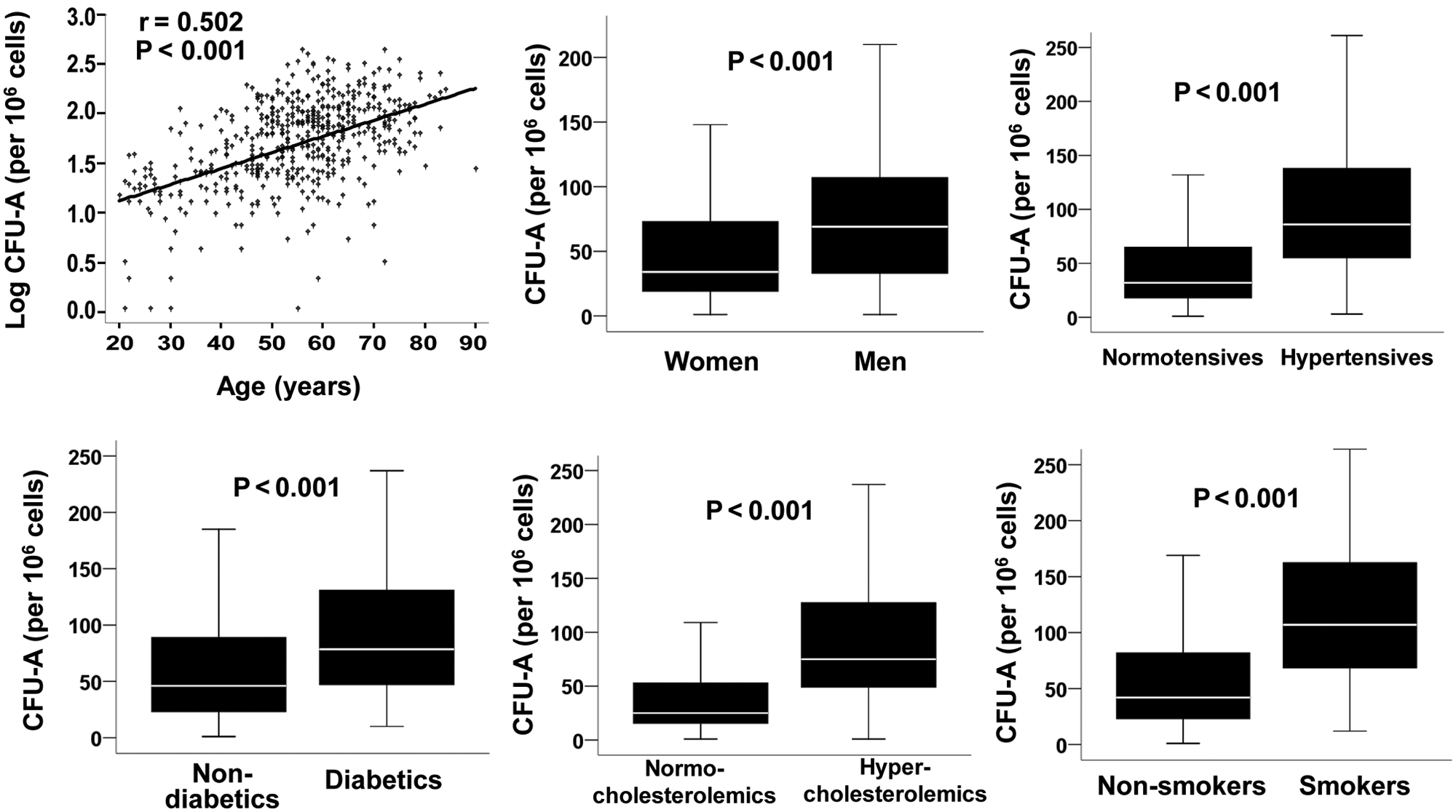
Relationship between angiogenic colony-forming unit (CFU-A) count and age, sex, and risk factors for cardiovascular disease (CVD). In the box-and-whisker plots, the centerline denotes the median value; the extremes of the box, the interquartile range; and the bars, the upper and lower limits of 95% of the data.
CFU-A counts were higher in subjects with CVD compared with those without (p < 0.001; Fig. 2A ), and this association remained significant after adjustment for age, sex, and risk factors (p < 0.001). The area under the ROC curve of CFU-As for the prediction of CVD was 0.850 (95% CI, 0.818–0.882; p < 0.001; Fig. 2C ). The CFU-A value that maximized the sum of sensitivity and specificity (69) was associated with a sensitivity of 72.0%, specificity of 82.0%, a positive predictive value of 77.1%, and a negative predictive value of 77.7% for CVD diagnosis.
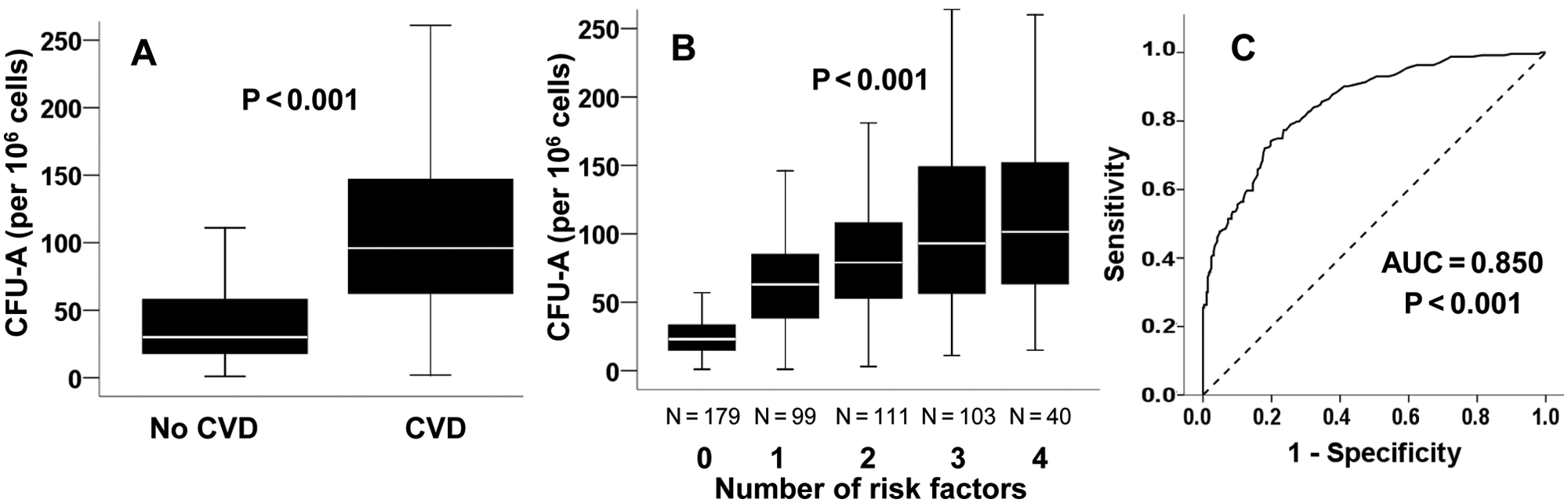
(
When the entire population was categorized by the number of risk factors for CVD (hypertension, hyperlipidemia, current smoking, diabetes), CFU-A counts were higher in subjects with increasing risk factor burden ( Fig. 2B ). Multivariate regression analysis confirmed that this association with risk factor burden was independent of age, sex, and presence of CVD ( Table 2 ). The risk factor burden was also associated with a stepwise increase of CFU-A counts in subjects without CVD, a relationship that remained significant even after adjustment for age and sex after multivariate regression analysis (p < 0.001; Table 2 ).
Multivariable Regression Analysis Evaluating the Association of CFU-A Count with the Number of Risk Factors in the Total Population and in the Subgroup without CVD
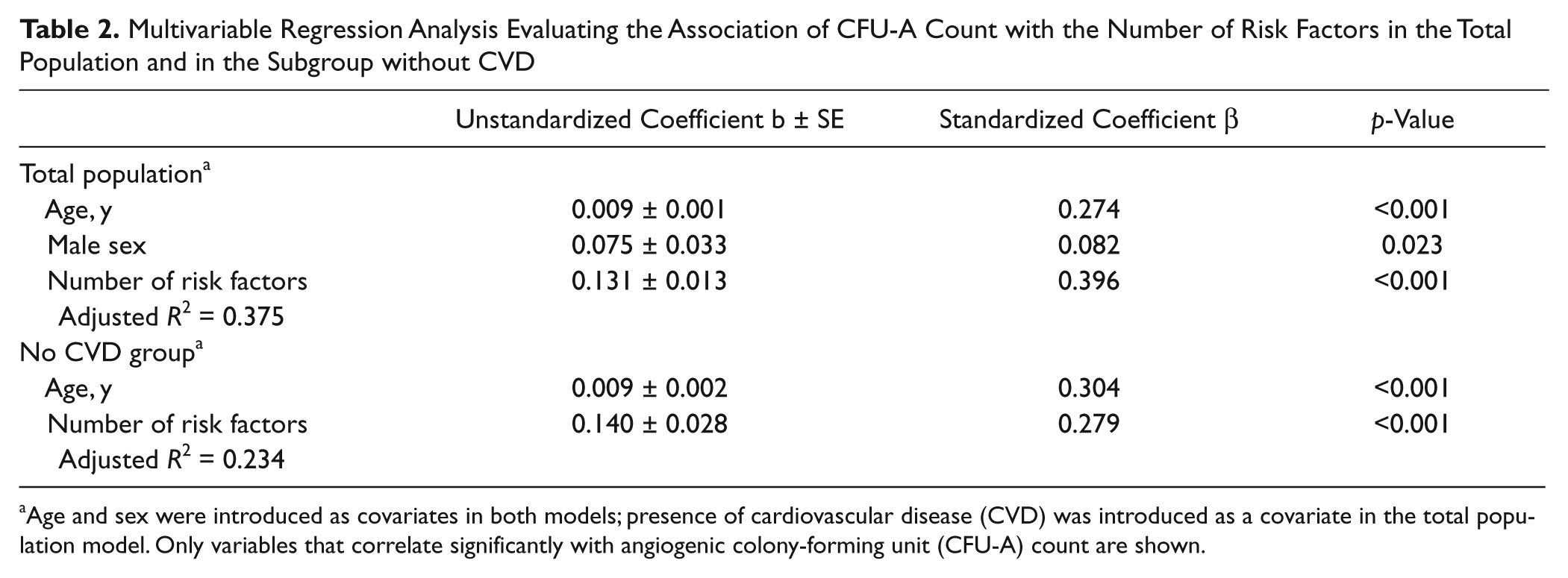
Age and sex were introduced as covariates in both models; presence of cardiovascular disease (CVD) was introduced as a covariate in the total population model. Only variables that correlate significantly with angiogenic colony-forming unit (CFU-A) count are shown.
CFU-A Counts as Predictors of Adverse Outcome
The 243 subjects with CVD were followed for a median of 888 days (range, 24–1429 days; interquartile range, 498–1092 days). A total of 59 subjects (24.3%) experienced an adverse cardiovascular event (death, MI or stroke, or revascularization) ( Table 3 ). Subjects who experienced an adverse event during follow-up had significantly higher CFU-As as compared with those who were free of events (110 [70–173] vs 84 [51–136]; p = 0.01).
Cardiovascular Events during Follow-up of 243 Subjects with Cardiovascular Disease
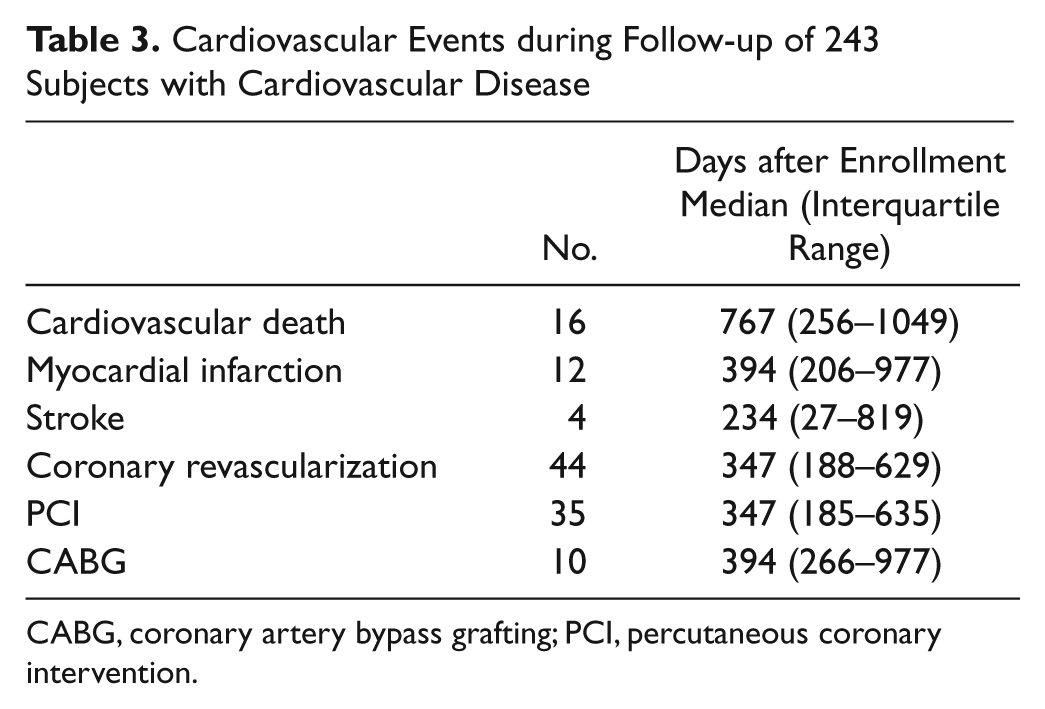
CABG, coronary artery bypass grafting; PCI, percutaneous coronary intervention.
Cardiovascular death, myocardial infarction, and stroke are considered to be strong evidence of cardiovascular disease progression. ROC curve analysis identified a CFU-A count of 103 with the highest ability to predict the composite end point of cardiovascular death, MI, or stroke during follow-up. Kaplan-Meier analysis showed that subjects with >103 CFU-As had a significantly higher incidence of cardiovascular death, MI, or stroke ( Fig. 3 ). Cox proportional hazard regression analysis confirmed that the ability of the CFU-A count to predict the end point of cardiovascular death, MI, or stroke was independent of age, sex, and CVD risk factors (adjusted HR, 2.6; 95% CI, 1.2–5.7; p = 0.017; see Data Supplement). When revascularization was considered along with death, MI, and stroke, the CFU-A count did not discriminate between subjects with and without events (HR, 1.5; 95% CI, 0.9–2.6; p = 0.12; see Data Supplement).
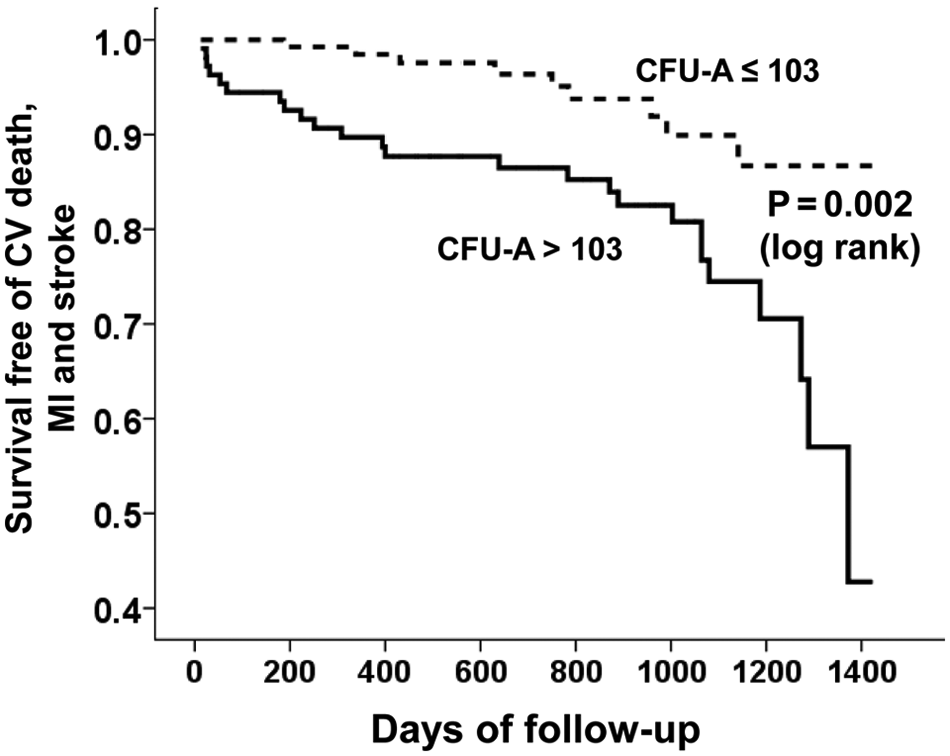
Survival free from death, myocardial infarction (MI), and stroke in subjects with cardiovascular (CV) disease according to angiogenic colony-forming unit (CFU-A) count.
Discussion
The present study evaluates the clinical importance of circulating proangiogenic cells in a large, broad-spectrum population (ranging from relatively healthy subjects to patients with advanced CVD) with close to a 3-year follow-up period. Using the CFU-A assay, we demonstrate that aging and the presence of risk factors for CVD are accompanied by increasing circulating proangiogenic cell activity, with higher CFU-A counts in those with a greater risk factor burden. In addition, an increased CFU-A level was independently associated with the presence of CVD. Finally, in a prospective study of patients with CVD, the CFU-A count is an independent predictor of adverse CVD events.
The CFU-A assay is one of several assays that identifies circulating, proangiogenic cells called endothelial progenitor cells (EPCs). Despite the name, the term EPC is now known to refer to cells of both endothelial and hematopoietic lineage, which contribute synergistically to vascular repair and angiogenesis either directly or through paracine effects.13–15 Previously used assays appear to measure different populations of cells. 16 Flow cytometry–identified EPCs, using the CD34, CD133, and vascular endothelial growth factor receptor 2 surface antigens, have been shown to develop in vitro into cells of the monocyte-macrophage lineage. 5 EPC culture assays, which may have an advantage over flow cytometry in that they measure both cell number and function (via total cell or colony growth), have yielded cells of either primarily hematopoietic lineage6–8 or endothelial lineage, depending on the methodology used.6,17,18 Furthermore, the cells were not consistently of bone marrow origin.6,17,18 The CFU-A assay measures bone marrow–derived precursor cells that mature into both proangiogenic hematopoietic- and endothelial-like cells, serving as a biomarker of a “combined” proangiogenic cell activity.10,19
The relationship between circulating proangiogenic cells and the burden of CVD risk factors and CVD has varied in the different clinical studies performed so far.8,9,20,21 Flow cytometrically enumerated CD34+/KDR+ cells have generally been inversely associated with a number of cardiovascular risk factors, the presence of CVD, and the incidence of cardiovascular events.20–22 Hill et al. 8 showed their CFU counts to be inversely associated with cardiovascular risk factor burden as well as endothelial function in 45 CVD-free men. On the other hand, Guven et al. 9 found the number of circulating “late outgrowth” CFUs to be positively correlated with the severity of coronary disease in 48 patients undergoing angiography. Our study of CVD and EPCs is one of the largest to date and includes a broad spectrum of CVD risk while carefully eliminating many potentially confounding acute and chronic conditions (acute MI, inflammatory disease, etc). The positive correlation of CFU-As with CVD risk and prognosis is in line with the findings by Guven et al. 9 Differences in our findings compared with those of other EPC studies are potentially attributable to the different cell types measured due to the different assay methods used. For example, unlike CD34+/VEGF+ cell enumeration, the CFU-A assay reflects both circulating cell number and function via colony growth. Therefore, despite a reduced circulating pool of EPCs, the growth potential or activity of the cells, measured by outgrowth of colonies, may be greater in these patients. Another cause of the differences in our findings compared with those of other EPC studies may be the smaller, more varied study populations previously studied.
Our finding of increasing circulating proangiogenic cell activity associated with increasing age, cardiovascular risk factor burden, and clinical CVD suggests that these cells are part of a physiological vascular repair response that parallels ongoing vascular injury. Experimental and human studies have shown that bone marrow–derived proangiogenic cells are mobilized into the peripheral circulation by single episodes of acute vascular injury.23–25 Our present observations extend this concept of injury-induced mobilization to chronic or ongoing, repeated vascular injury. Tissue hypoxia secondary to severe CVD may also contribute to the increase in proangiogenic cell activity observed in patients with CVD above and beyond their cardiovascular risk factor burden.26–28 Importantly, a higher CFU-A count not only was associated with the presence of CVD but was also predictive of adverse CVD events in patients with CVD, a finding that was independent of other risk factors and age. This suggests that CFU-A colony formation is directly tied to the amount of ongoing vascular injury, although as a reparative response, it is “insufficient.” It is also possible that circulating proangiogenic cells can actually predispose to increased vascular disease through the promotion of intimal hyperplasia, plaque neovascularization, inflammation, or thrombosis, mechanisms that have all been supported by recent studies.29–31 Its independence of age and other risk factors suggests it could serve as a unique biomarker of cardiovascular risk. Furthermore, as an in vitro cell culture assay, it could also serve to test compounds designed to mitigate CVD risk via their effects on angiogenesis.
Our study has particular strengths, such as the large population studied and the confirmation of our findings both in cross-sectional and prospective settings. However, we recognize certain limitations. Although the subject population studied spanned the continuum of risk for CVD, certain patient populations may have been underrepresented, including females. Another limitation, as acknowledged in the discussion above, is that causality cannot be inferred since this was an observational study. Further mechanistic studies investigating the causes and consequences of the increase in CFU-As are required. Direct correlation of the CFU-A assay with other EPC assays and clinical outcomes would also be of interest. In addition, our study does not provide evidence of the predictive ability of the CFU-A count with regard to adverse outcome in patients without established CVD.
Despite the above-mentioned limitations, our study demonstrates some important findings. Circulating proangiogenic cell activity, as measured by the CFU-A count, is increased in the presence of cardiovascular risk factors and CVD in humans. Furthermore, increased CFU-A levels predict an increased risk of death, MI, and stroke in patients with CVD. This strongly suggests that circulating proangiogenic cell activity reflects the level of ongoing vascular injury.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported with funding from Eli Lilly and Company, Indianapolis, Indiana; a gift from the Marcus Foundation, Atlanta, Georgia; the Georgia Tech/Emory Center for the Engineering of Living Tissues (GTEC) National Science Foundation (NSF) Grant EEC-9731643; and PHS Grant UL1 RR025008 from the Clinical and Translational Science Award Program, National Institutes of Health, National Center for Research Resources.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
