Abstract
Compound sample preparation and delivery are the most critical steps in high-throughput screening (HTS) campaigns. Historically, several methods of compound delivery to assays have been used for HTS, including intermediate plates with prediluted compounds, assay-ready plates (ARPs) using either preplated dried compound films or nanoliter DMSO spots of compounds, as well as pin tool–delivered compounds. We and others have observed differences in apparent compound potency depending on the compound delivery method. To quantitatively measure compound potency differences due to the chosen delivery methods, we conducted a controlled study using a validated biochemical luciferase assay and compared potencies when compounds were delivered in either ARPs (using acoustic dispensed nanoliter spots) or by pin tool. Here we compare hit rates, confirmation rates, false-positive rates, and false-negative rates between the two delivery methods using the luciferase assay. We compared polystyrene (PS) and cyclic olefin copolymer (COC) plates using both delivery methods and examined whether ARPs stored at 4 °C were superior to those stored frozen at −20 °C. The data show that the choice of compound delivery method to the assay has an effect on the apparent IC50’s and that pin tool delivery results in more confirmed hits than preplated compounds, resulting in a lower false-negative rate. However, this effect is minimized through the use of COC plates and by obtaining plates in a “just-in-time” mode. Overall, this report provides guidance on using assay-ready compound plates and has affected the way HTS campaigns are using acoustically dispensed plates in our department.
Keywords
Introduction
Compound sample preparation is a critical step for high-throughput screening (HTS) operations. 1 Several different methods to deliver compounds to assay plates currently exist. All of these methods attempt to maximize efficient dissolution of compounds using an organic solvent while minimizing the percentage of this solvent delivered to the biological assay system. Most bioassays will not tolerate more than a few percent of solvent, so the goal is to provide the appropriate compound concentration in as little solvent volume as possible. Although methods exist in which compounds are predissolved in high-vapor-pressure solvents such as 80/20 methanol/water allowing for the addition of compounds to assay plates followed by complete removal of the solvent through evaporation, 2 the primary method has been to deliver compounds from DMSO solutions. For DMSO solutions in HTS assays, there primarily exists three ways in which compounds can be delivered from a highly concentrated DMSO source plate into an assay plate for evaluation. The first method of compound addition uses pin tool technology, which employs metal or plastic pins to directly transfer a set volume (usually ranging between 2 nL–5 µL) from the compound source plates to the assay plates containing the biochemical or cell-based assay mixture. 3 Second, there is noncontact preplating of compounds directly into assay plates by transferring a set volume from the source plate to an assay plate to which the biochemical reagents are added either as quickly as possible or at a later stage following storage (reviewed by Koltay and Ernst 4 ). Some common examples include ultrasonic or acoustic droplet ejection technology5-6 or piezo sample distribution.7,8 Third, there is an indirect dilution method, whereby a concentrated compound stock solution dissolved in DMSO is diluted into an aqueous intermediate buffer, effectively diluting the compound as well as the associated solvent to an assay-compatible concentration. This aqueous dilution is then transferred into a separate assay plate (thus, an indirect dilution of compound, also sometimes referred to as an “intermediate dilution” delivery).
Obviously, there exist advantages and disadvantages for each of these three methods. Pin tools offer a rapid approach to compound delivery, with accuracies often better than 5%, precisions within 10%, and highly reproducible transfers even after years of continuous use. 9 Compared with an intermediate dilution method, there is also significant cost-savings by eliminating disposable pipette tips and plates from the compound transfer step as well as reducing the quantity of compound required to prepare the dilution. The chief limitation of the pin tool is that this is a contact-based method, delivering a fixed volume whose accuracy is a function of many factors, including pin and assay plate surface properties, source and assay plate well depth, removal speed from the compound source plate, and the method of washing and drying of the pins between additions. 3 The fixed-volume nature of the pin tool also means that different sizes of pins must be available or assay volume must be adjusted to obtain the desired final compound concentration. In addition, reliable transfer is limited to wet delivery, because dry-dispense methods using pin tool technology results in very poor accuracy and precision, and the pin tool must be efficiently cleaned between source plates to prevent compound carryover.3,8
With respect to noncontact compound delivery, preplating of the compounds in this manner as assay-ready plates (ARPs) offers some advantages over pin tool and indirect compound transfer. For example, preplated compounds can be made to order on any set of compound plates and compounds can be cherry-picked and added to well positions in a completely flexible manner. In addition, ARPs can enable certain screening operations such as biosafety hazardous screens, whereby all dispensing steps must be contained in a biosafety cabinet and a single disposable assay plate is used to prevent any potential contamination of the compound source plate. Last, the opportunity exists to prepare the screening plates ahead of time, thereby decoupling the compound delivery from the screening process and allowing for extended storage prior to screening. Preplating compounds into assay plates is not without some concern, however. It has been reported that the physical appearance and size of acoustic dispensed droplets into 1536w polystyrene (PS) assay plates were influenced by the initial volume plated, time of storage, storage temperature, and relative humidity. 5 In addition, the activity of some compounds in several biochemical assays appear sensitive to the above conditions. 5 Similarly, complete drying of compounds down onto the surface of assay plates in the absence of suitable excipients poses the risk of poor recovery efficiencies. 10
Intermediate dilution methods, although historically one of the most commonly used bench-top methods of assessing compound activity, suffer from potential compound precipitation effects in assay buffer and potential losses due to adsorption of compounds onto microtiter plates or plastic pipette tips. 11 Decreased potencies of test compounds and increased false-negative results are found when the compound addition methods include an aqueous serial dilution process6,12; thus, this method is not currently in use in our HTS platforms.
Recently, in our laboratory, when evaluating biochemical assay results generated from ARPs compared with pin tool–delivered compounds, we noticed several examples in which differences in the apparent potency of compounds were obtained depending on the method of compound delivery. Reduced hit rates were often observed where compounds were delivered from ARPs compared with pin tool–delivered compounds across multiple assay technologies, including two different fluorescence intensity–based assays (see examples in Fig. 1 ), as well as a luminescence assay and an AlphaScreen-based assay (data not shown). However, as these assays were not originally designed for a precise comparative analysis, we set forth to conduct a carefully controlled comparative study of the current two most common methods of compound delivery, using a highly reproducible and robust biochemical assay carried out in two common microtiter plate types (PS and cyclic olefin copolymer [COC]) to determine if these observations were reproducible.
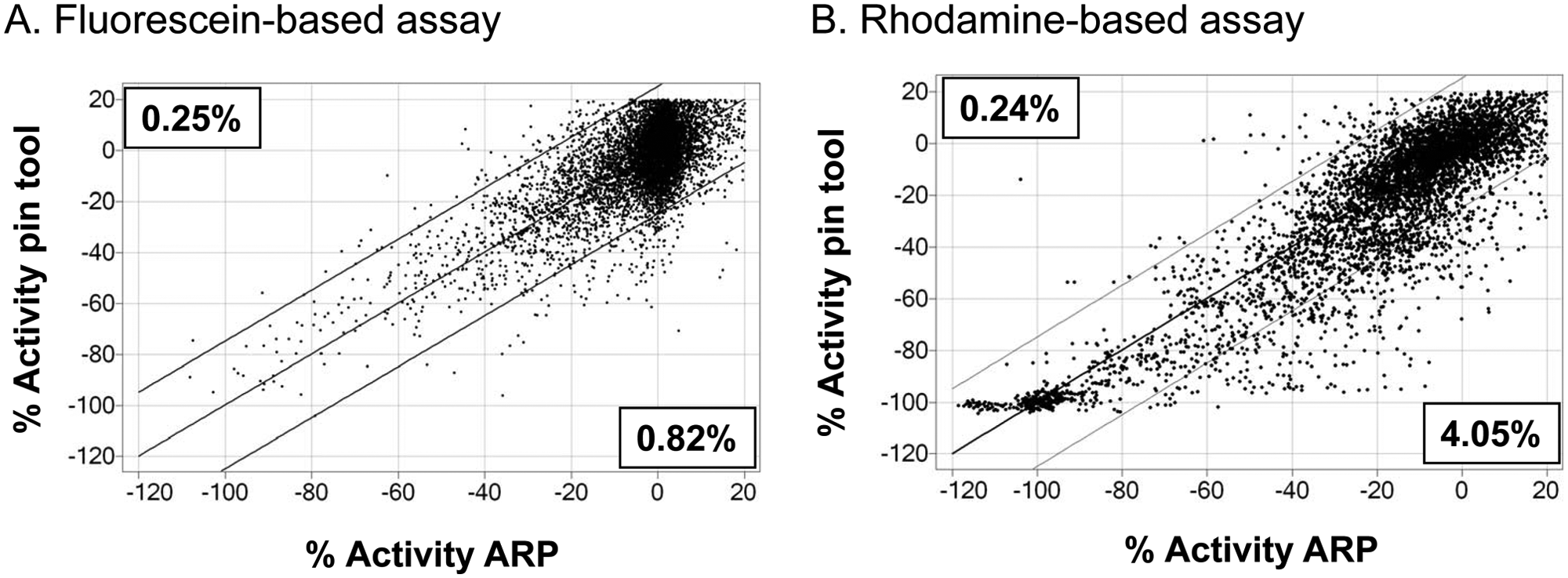
Impact of the compound dilution method on the apparent potency of a diversity collection of compounds in two different fluorescence-based peptide hydrolysis assays. (
Materials and Methods
Materials
Microtiter plates used in this study are as follows: 1536-well, flat-bottom, white polystyrene, high-base plates, catalog No. 782075 from Greiner Bio-One (Germany) and 1536-well, flat-bottom, white COC, high-base plates, catalog No. 19120 from Aurora/Nexus Biosystems (Poway, CA) were used for the luciferase studies. The 1536-well, flat-bottom, black PS, high-base plates, catalog No. 782076 from Greiner Bio-One were used for the peptide thioester hydrolysis assay and rhodamine peptide cleavage assay.
Reagents
For luciferease studies, ATP, bovine serum albumin (BSA), D-luciferin, magnesium acetate, Tris acetate, and Tween 20 were purchased from Sigma-Aldrich (St. Louis, MO). DMSO, certified ACS grade, was purchased from Fisher (Waltham, MA). Purified wild-type Photinus pyralis firefly luciferase (FLuc) was purchased from Sigma-Aldrich (catalog No. L9506).
PURE151 compound collection
The PURE151 compound collection is a diverse set of 8800 compounds available from the Novartis Sample Management Group. It consists of synthetically prepared compounds as well as purified natural products. Compounds are stored at 4 °C under 20% relative humidity in a 90% DMSO/10% water mixture in COC plates to avoid freeze-thaw cycles and water uptake and to allow rapid plate replication.13,14
ARPs
Preparation of all ARPs was performed in an ambient environment (21–22 °C), using the same 1536- compound source plates for all test sets for a screen. The standard process is completely automated and uses liquid transfer by acoustic droplet ejection using an Echo 555 (Labcyte, Sunnyvale, CA). The sample transfer time was between 5 and 6 min per plate. Both primary screening plates and confirmation concentration response screening plates were prepared in duplicate, in PS or COC plates.
Preparation of primary test sets of ARPs
Sets of ARPs were prepared from the same compound source plates, using the Echo 555. After dispensing, the ARPs were lidded within 1 min and stored at 4 °C, 50% RH, by the system for approximately 1 h. These plates were then manually placed in heat/vacuum–sealed bags to prevent evaporation of the samples or any potential water uptake. In addition, stacks of plates were sealed with a “sacrificial” DMSO-filled plate added to the top and also to the bottom of the plate stack prior to bagging. This was added to help minimize any potential evaporation issues in the plates. ARPs were stored under two different conditions: 4 °C or −20 °C. Also within 1 h, the source plates used for ARP production and destined for the pin tool use were lidded and stored in a heat/vacuum–sealed bag at 4 °C until just before use. All ARPs for each assay were screened the following day to simulate actual working conditions and to eliminate day-to-day variability in the assay performance and to facilitate comparison of test sets. Prior to assay, 4 °C and −20 °C bagged plates were allowed to reach room temperature (~1 h) prior to opening to prevent potential water uptake from the atmosphere.
Preparation of confirmation test sets of ARPs
For the luciferase assay screen, dilution plates from the source plate (2 mM) were prepared. Each compound was represented by an eight-point, 3.16-fold dilution series in a 384-well dilution plate. Samples of 4.5 µL from four of the dilution plates were then compressed in different quadrants of COC source plates in 1536 format. ARPs were generated from these source plates as described above, using the Labcyte 555 Acoustic Droplet Ejection method. All source and assay plates were lidded and stored in a heat/vacuum–sealed bag within 1 h from the completion of the sample dispense, including the addition of DMSO-filled plates at the top and bottom of the stacks to mitigate any potential evaporation issues. ARPs were then stored under the two different conditions (4 °C or −20 °C) as described in the previous section.
Echo and pin tool precision and accuracy testing and pin tool wash protocol
The Echo 555 and Janus (PerkinElmer, Waltham, MA) with fitted 1536-pin array 20 and 50 nL slotted pin tools (V&P Scientific, San Diego, CA) were QC’d prior to use in all experiments. Specific details on the QC process and pin tool wash protocol can be found in the
Luciferase assay
A total of 3 µL/well of substrate/buffer concentrate (10 µM D-luciferin, 10 µM ATP, 0.05% BSA, 13.3 mM magnesium acetate, 50 mM Tris acetate, pH 7.3, 0.01% Tween 20, final concentration) was dispensed into PS or COC white 1536-well high-base plates by using a Flexdrop Plus Dispenser (PerkinElmer). For pin tool addition of 20 nL/well compound solution, a V&P Scientific pin tool equipped with a 1536-pin array containing 20 nL slotted pins was used. After compound transfer, 1 µL/well of luciferase/buffer concentrate (10 µM luciferase in 1× PBS pH 7.3, 0.05% BSA, 0.01% Tween 20, final concentration) was dispensed into the assay plate by using a Flexdrop, and then the assay plate was centrifuged for 1 min at 1000 rpm by using VSpin (Agilent Technology, Santa Clara, CA). After a 10 min incubation at ambient temperature in a Liconic STX220, luminescence was detected by a ViewLux (Perkin Elmer) using a 10 to 60 s exposure time with clear filter and 1× binning. All screening operations were performed by using a fully integrated Thermo CRS Dim4 robotic system (Thermo, Waltham, MA) with Polara system software (Thermo).
Peptide thioester hydrolysis assay
The self-hydrolyzing thioester tetrapeptide, z-GLAR-SBzl, was purchased from Biosyntan (Berlin, Germany) and kept as a 50 mM stock solution in 95% DMSO/5% water at −20 °C. At room temperature, this peptide spontaneously hydrolyzes to release the newly formed thiol benzyl mercaptan, which is detected using 2,4-dinitrobenzenesulfonyl-fluorescein, a fluorescent analog of Ellman’s reagent. 15 The BES-thio-analog was synthesized at Novartis and kept as a 10 mM stock solution in 90% DMSO/10% water at −20 °C. Reactions were conducted in PBS assay buffer containing 0.01% Tween-20 detergent (PBST buffer) with a final concentration of 100 µM z-GLAR-SBzl peptide and 12.5 µM BES-thio-analog and 20 µfinal compound concentration. All stock solutions were kept on ice at 0 °C to 4 °C during the screening to prevent the spontaneous hydrolysis of the peptide. For evaluating the effects of the compound delivery method, the 8800 compound Novartis PURE151 collection was tested in the assay as follows: For pin tool delivery, z-GLAR-SBzl peptide was dissolved to a concentration of 200 µM in PBST buffer immediately prior to use, and 1 µL was dispensed into the wells of Greiner 1536w black polystyrene assay plates using a FlexDrop. To these assay plates, 20 nL from 1536-well source plates of compounds was added using the 20 nL pin tool. Immediately following the compound delivery via pin tool, 1 µL of 25 µM BES-thio-analog dissolved in PBST buffer was added. Plates were then centrifuged at 1000 rpm for 1 min using a VSpin (Agilent Technology) and incubated at 25 °C, 99% humidity, in a temperature- and humidity-controlled incubator for 60 min to allow the peptide hydrolysis to occur. Fluorescence product was detected through the use of an Envision microplate reader with 485 nm excitation filter and 535 emission filter. For ARP, 20 nL final volume of compounds from the above 1536-well source plates was added via Echo to Greiner 1536-well black polystyrene assay plates and stored at −20 °C for 24 h prior to testing. After thawing the plate storage bags at room temperature, the plates were centrifuged at 1000 rpm for 1 min in a VSpin prior to screening. The assay was run in these plates directly by adding 1 µL z-GLAR-SBzl peptide dissolved to a concentration of 200 µM in PBST buffer using a FlexDrop, followed by a 5 min offset delay to account for the timing needed for the comparator assay pin tool delivery and washing steps. One microliter of 25 µM BES-thio-analog dissolved in PBST buffer was then added, plates were centrifuged at 1000 rpm for 1 min using the VSpin, and the reaction was initiated at 25 °C, 99% humidity, for 60 min. As for the pin tool–delivered assay, the fluorescence product was detected through the use of an Envision microplate reader with 485 nm excitation filter and 535 emission filter.
Rhodamine peptide cleavage assay
The rhodamine- containing tetrapeptide D-Pro-Phe-Arg-Rho110-Glu was purchased from Biosyntan (Berlin, Germany) and kept as a 5 mM stock solution in 95% DMSO/5% water at −20 °C. A commercially available trypsin-like serine protease (Calbiochem/EMD Chemicals) was used as the enzyme in the assay. The rhodamine peptide is cleaved by the protease, which releases the fluorescent rhodamine product. All stock solutions were kept at 4 °C during the screening. Reactions were conducted in assay buffer (50 mM Hepes/HCl, pH 7.8, 150 mM NaCl, 1 mM EDTA, 0.05% CHAPS) using 0.5 µM D-Pro-Phe-Arg-Rho110-Glu peptide and 40 pM serine protease using Greiner black PS plates. For evaluating the compound delivery method, the same method used for the peptide thioester hydrolysis described above was used, except after ARP production, compound plates (20 nL volume, 2 mM stock concentration) were stored at 4 °C overnight prior to testing. The assay was run in these plates directly by adding reaction mixture, to yield a final compound concentration of 10 µM. All reactions were run for 60 min at ambient temperature and fluorescence product detected through use of a Viewlux reader with 485 nm excitation filter and 520 nm emission filter.
Data analysis
Data analysis was conducted using Novartis proprietary data evaluation software, TIBCO Spotfire version 3.1, and GraphPad Prism. Further details can be found in the
Results
Comparison of Activity of Pin Tool–Delivered Compounds to ARPs
While evaluating the results of several HTS assays in our department where compounds were delivered to the assay via pin tool or with ARP using nL DMSO acoustically delivered spots, we noticed differences in apparent compound potency. An example of two simple fluorescence-based assays in which compound delivery methods were compared is shown in Figure 1A - B . There is a clear reduction in apparent compound potency depending on the method of compound administration used. In the first example using a fluorescein-based assay, there is a larger percentage of compounds that appear less active using ARP compared with pin tool delivery ( Figure 1A ). Similarly, using a rhodamine-based fluorescence assay, there is also a larger percentage of compounds that appear less active with ARP delivery compared with pin tool–delivered compound plates ( Figure 1B ).
Luciferase Assay
Based on several examples found in our laboratory, such as that shown in
Figure 1
, we set up a controlled study using a highly robust and reproducible FLuc enzyme assay.
17
The luciferase assay was chosen for the test as this is a common reporter enzyme used in HTS applications and is highly sensitive and reproducible. In addition, all reagents are commercially available, and the overall HTS protocol contains few liquid-handling steps, which limits possible artifacts due to the process. The protocol for the assay is shown in
The workflow for compound handling and plate production is shown in Figure 2 . The 8800 compounds present in the PURE151 library were used to create duplicate copies of ARP in both PS and COC 1536-well plates. Each set of plates was then stored overnight at either 4 °C or −20 °C prior to assay. Plates were removed from the compound storage facility, and after a brief period at room temperature to thaw the −20 °C plates and bring them to room temperature, they were used immediately for screening. Pin tool–delivered compounds were delivered from the same set of source plates used to make the ARP; thus, differences in compound batch, concentration, purity, or integrity were eliminated as potential variables.
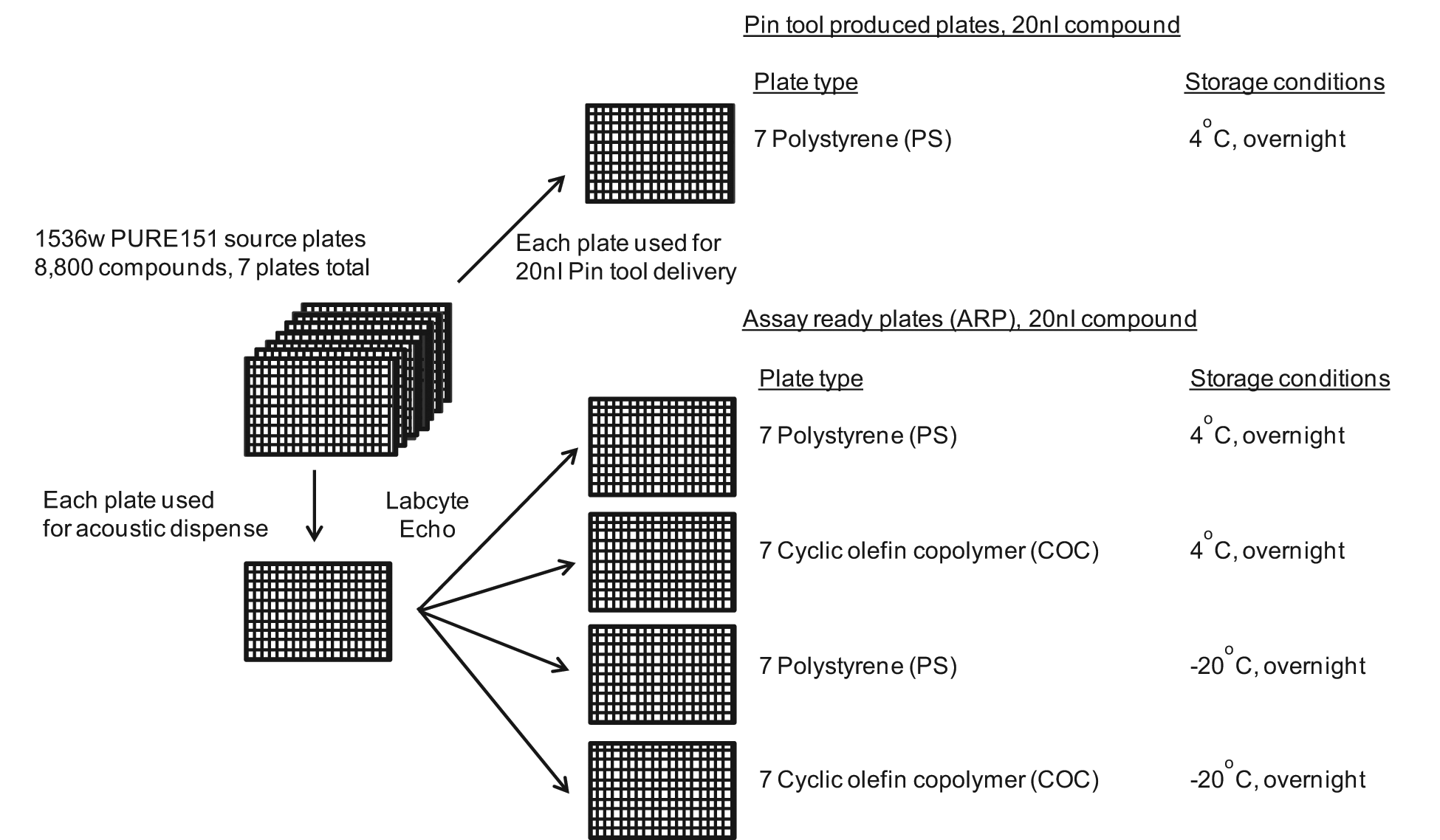
Workflow for compound handling and assay-ready plate (ARP) production. A total of 8800 compounds comprising the Novartis PURE151 diversity collection were used for ARP production and as a source for the pin tool delivery. The seven 1536-well source plates were processed individually in the Labcyte Echo 555 and used to create seven copies containing 20 nL of each compound in the respective polystyrene (PS) and cyclic olefin copolymer plates. ARPs were kept overnight at 4 °C or overnight at −20 °C prior to use. Duplicate sets of plates (14 per condition) were created for all ARPs. The seven 1536w compound source plates used to create the ARPs were kept overnight at 4 °C and then used for the pin tool delivery into PS plates.
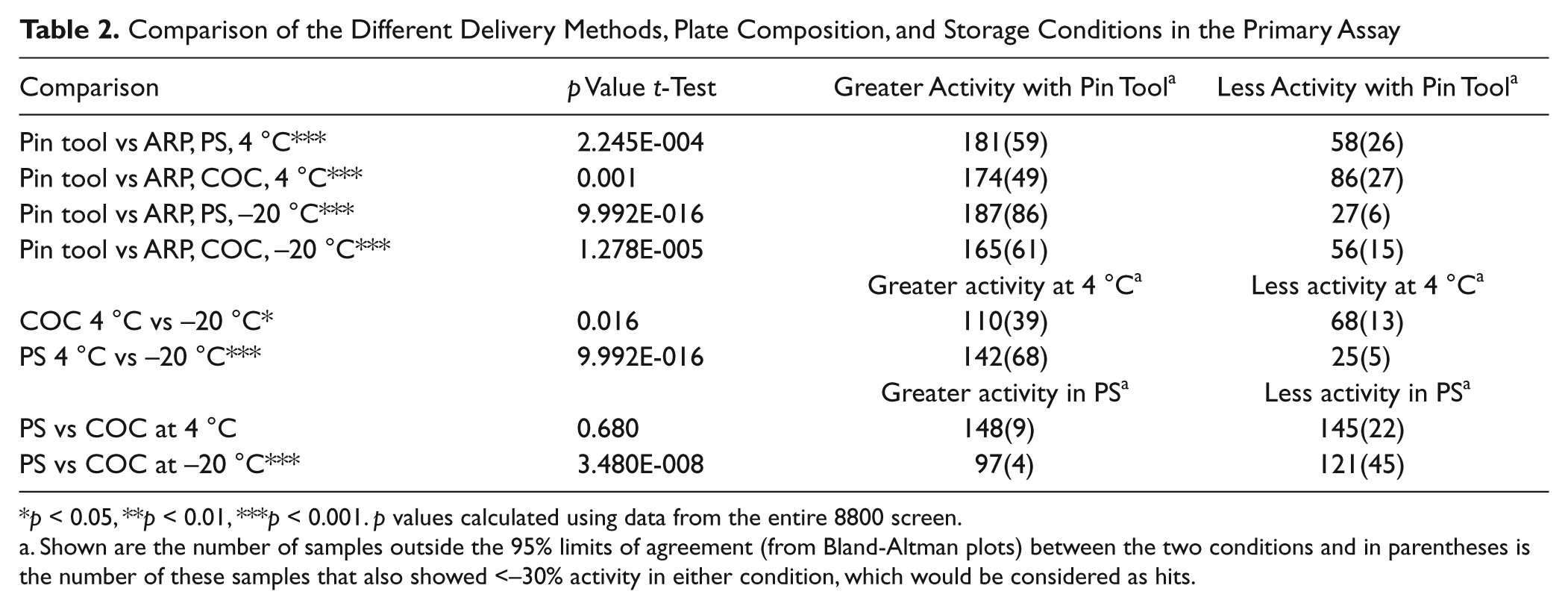
The luciferase assay showed excellent reproducibility from duplicate screens that used the same direct compound addition method. The performance and results of the replicate testing are shown in
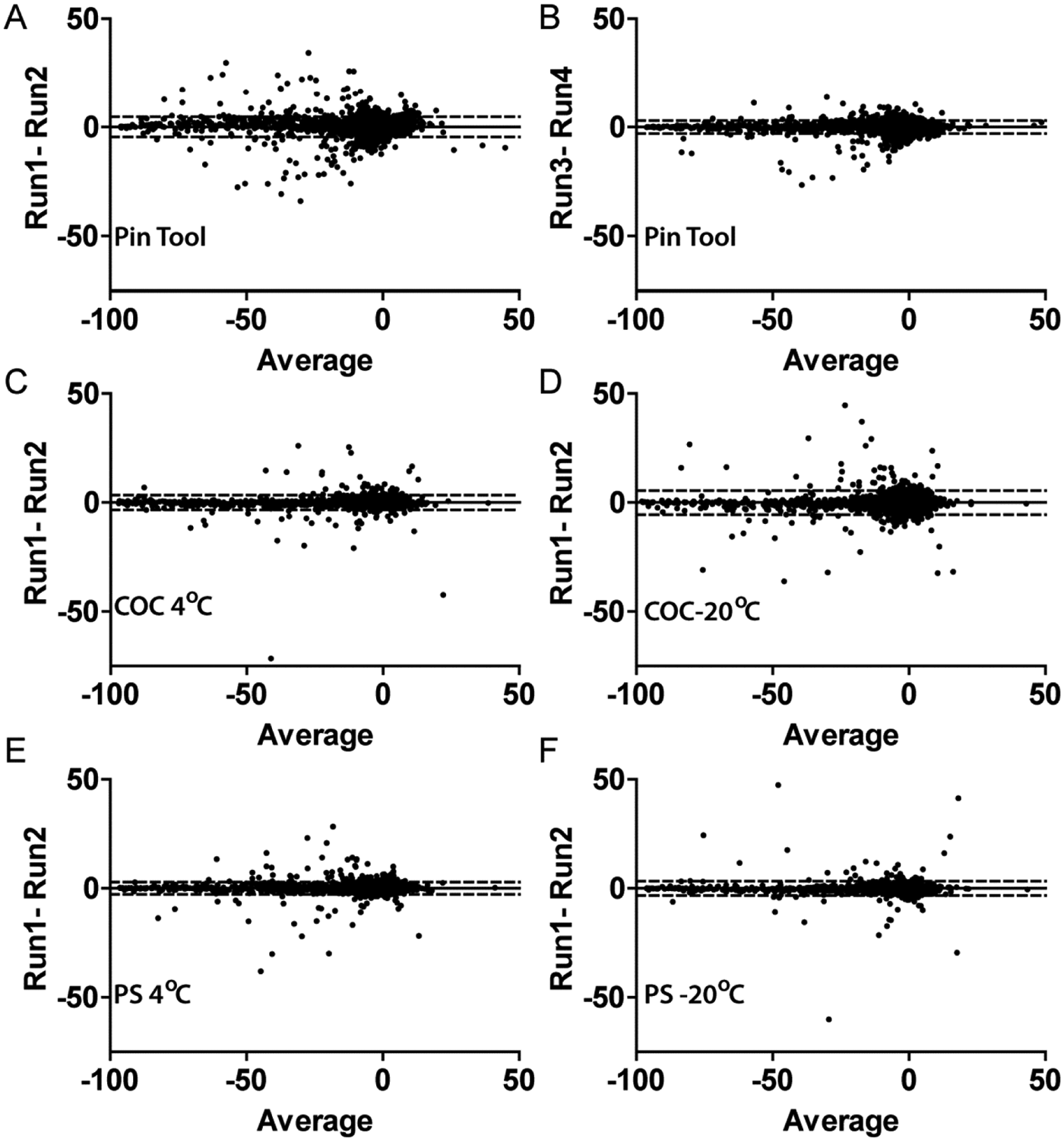
Intramethod variability analysis for the conditions under study. Bland-Altman plots are shown. (
Summary of Hit Rates and Analysis of Intramethod Reproducibility
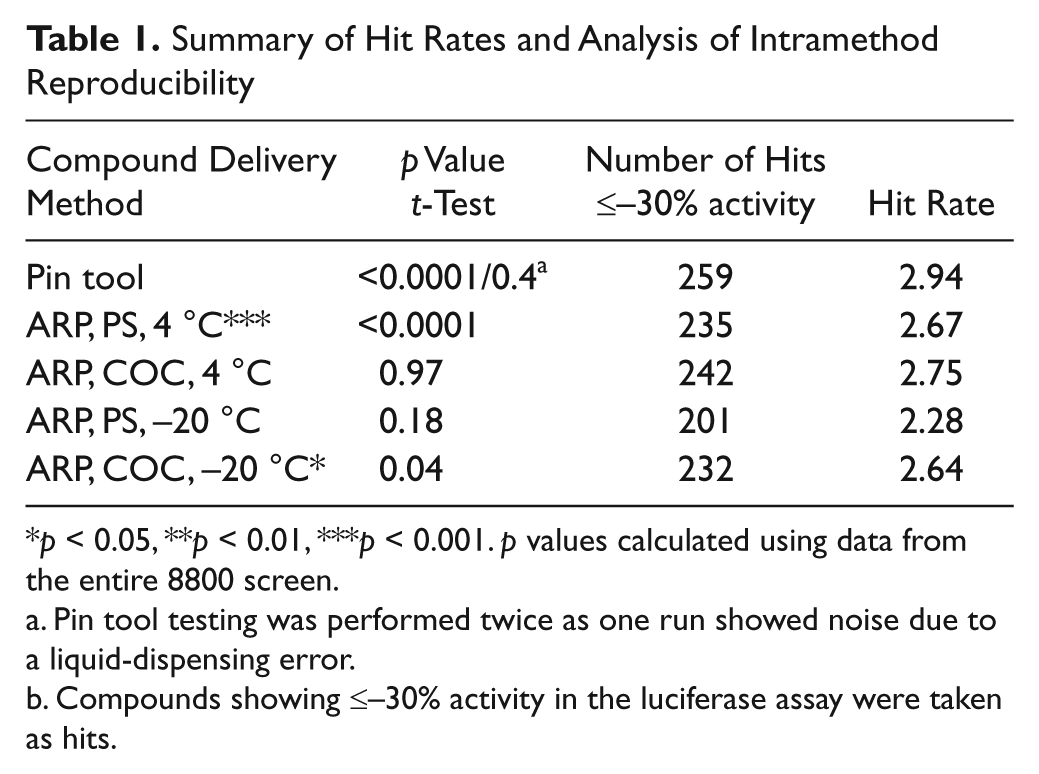
p < 0.05, **p < 0.01, ***p < 0.001. p values calculated using data from the entire 8800 screen.
Pin tool testing was performed twice as one run showed noise due to a liquid-dispensing error.
Compounds showing ≤–30% activity in the luciferase assay were taken as hits.
Luciferase Inhibitor Hit Identification Using the Different Screening Methods
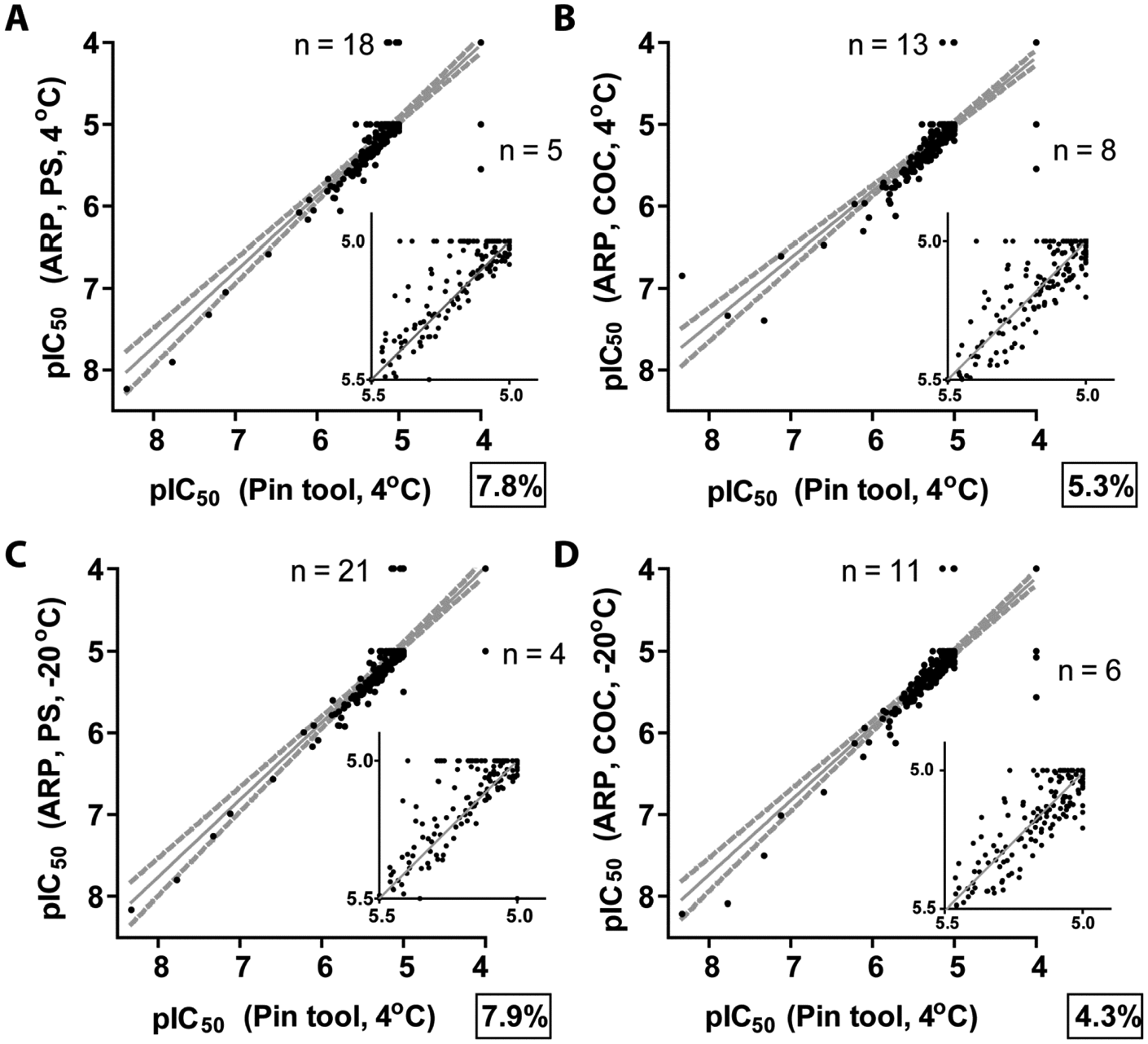
We used three standard deviations of the screening data as a guide, which was approximated by a ≤–30% activity cutoff for hit selection for each of the different methods. For the purpose of comparison of one method of compound delivery to another, compound data sets were averaged and hits from each method were then compared to one another. The number of hits identified in each of the different methods from the 8800 compound collection is shown in Table 1 . Pin tool–delivered compounds clearly resulted in the greatest number of hits found (reflected in the highest hit rate), followed by PS and COC ARPs stored at 4 °C and then PS and COC ARPs stored at −20 °C. All comparisons of pin tool delivery to ARP methods showed significant differences in activity (p < 0.001; Table 2 ). The majority of the activity differences were due to samples showing more potent activity with pin tool compared with ARP ( Figure 4 ; Table 2 ). Overall, this resulted in a larger number of unique compounds being identified using the pin tool method, especially when comparing with PS ARPs stored at −20 °C, which showed the largest activity differences, with the majority of samples showing more potent activity with the pin tool ( Table 2 ).
Comparison of the Different Delivery Methods, Plate Composition, and Storage Conditions in the Primary Assay
p < 0.05, **p < 0.01, ***p < 0.001. p values calculated using data from the entire 8800 screen.
Shown are the number of samples outside the 95% limits of agreement (from Bland-Altman plots) between the two conditions and in parentheses is the number of these samples that also showed <–30% activity in either condition, which would be considered as hits.
Intermethod variability analysis. Bland-Altman plots of the average run data. Methods shown are pin tool versus assay-ready plates (ARPs) prepared in cyclic olefin copolymer plates and used either as fresh (4 °C;
We also compared various conditions using ARP plates to each other. This showed that storing ARP plates as frozen copies had a detrimental effect on activity ( Table 2 ). Samples prepared as ARP and subsequently kept frozen showed less activity than those stored at 4 °C, and this difference appeared greater when PS plates were used compared with COC plates ( Table 2 ). No significant difference in activity was observed between ARP prepared in PS and COC with storage at 4 °C (p = 0.67), suggesting that the type of plate plastic may be a concern that is secondary to storage temperature.
Luciferase Inhibitor Hit Confirmation from the Different Screening Methods
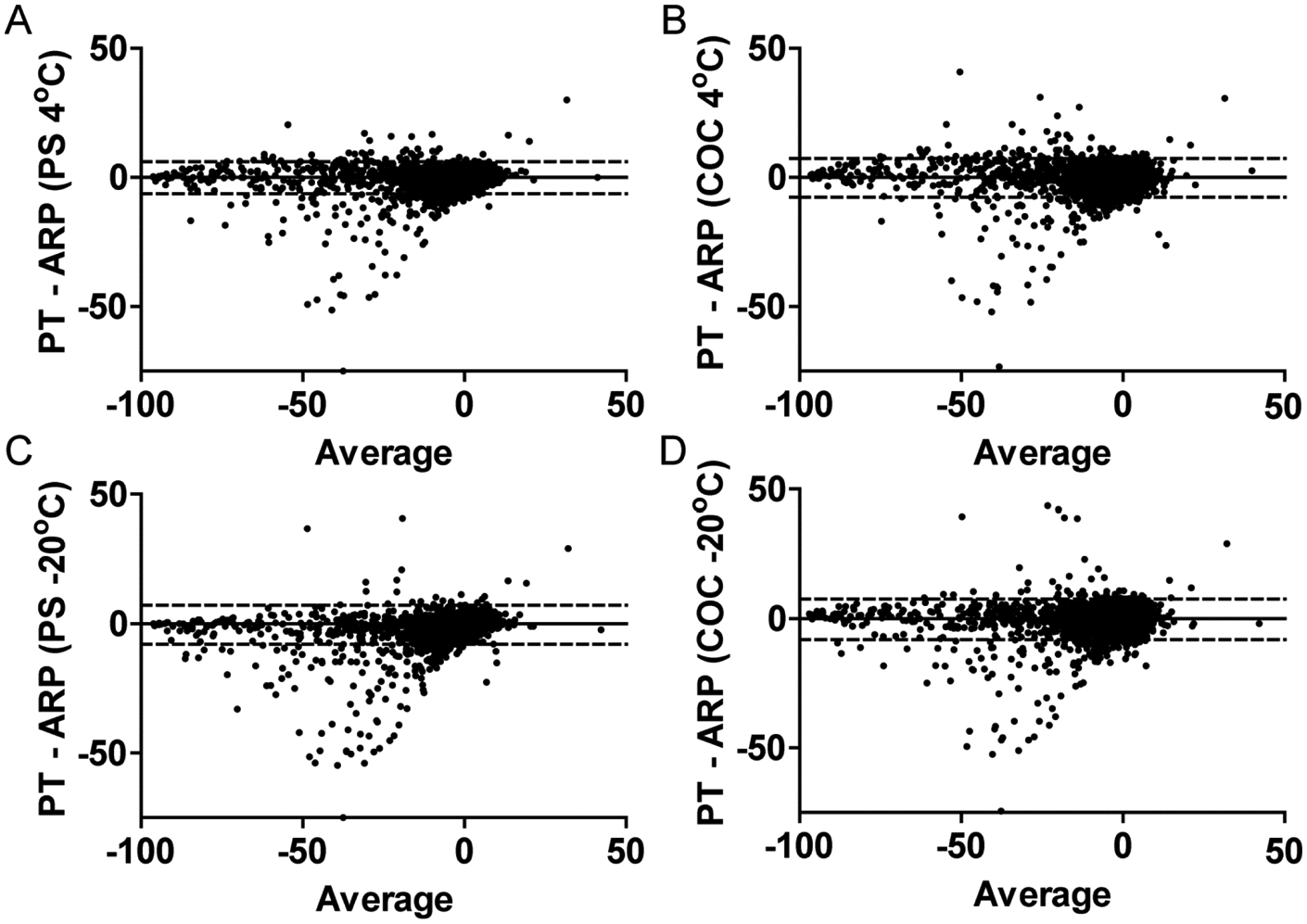
To verify that the observed differences in hits between the different conditions was due to genuine inhibition, compounds identified as hits from all of the test conditions shown in Figure 3 were picked for confirmation studies. This resulted in the selection of 322 total compounds, which were then tested at eight concentrations to calculate IC50’s. The eight-point dilution series was prepared in 90% DMSO/10% water using a 3.16-fold dilution between each concentration. The top starting concentration of compound for confirmation was 10 µM. As the original hits from the different test conditions were picked at ≤–30% activity, compounds were considered confirmed if the concentration response curves yielded an IC50 of ≤10 µM or for the less potent compounds if the data at the highest compound concentration (10 µM) showed ≤–30% activity. The confirmation data are shown in Figure 5 . In PS plates, 7.8% and 7.9% of the active compounds stored at 4 °C and −20 °C ( Figure 5A , C , respectively), showed a ≥twofold shift in IC50 compared with the pin tool delivery method. In contrast, only 2.1% and 1.8% of the confirmed hits showed a ≥twofold shift in IC50 compared with the PS ARP. In COC plates, 5.3% and 4.3% of the active compounds stored at 4 °C and −20 °C ( Figure 5B , D ), respectively, showed a ≥twofold shift in IC50 compared with the pin tool delivery method (compared with 1.8% and 2.5% shift using the pin tool, respectively).
Confirmation (validation) results of the hits picked from pin tool delivery or assay-ready plates (ARPs). Eight-point dilution series were prepared and tested, and IC50 determinations were made using four parametric curve fits. Compounds that were inactive were assigned a pIC50 of 4, whereas those less potent compounds that showed an IC50 ≥10 µM (% activity ≤–30% at 10 µM compound) were assigned a pIC50 of 5. In all panels, the bold center line represents linear fit to the data, and the two gray dotted lines represent the 95% confidence intervals. The inset graph is a magnified view of the graph with the line representing equal activity. The percentage of the compounds that showed a ≥twofold IC50 shift are shown in a box for each respective delivery condition compared with the pin tool delivery method. Concentration values are in molar.
The primary and confirmation data were then used to calculate the false-negative and false-positive rates for the multiple methods. These results are shown in Table 3 . The false-positive rate among all the different conditions remained essentially constant with a value between 7.7% and 9.0%. The false-negative rate, however, showed a very different result. Compounds delivered via pin tool showed the lowest false-negative rate (14.6%), whereas those from ARP stored at 4 °C (both PS and COC) showed the next lowest amount ( Table 3 ). By far, however, the delivery method, plate type, and storage conditions with the largest false-negative rate were those of the ARP PS plates stored at −20 °C (34.7%), which is more than twice the rate of the pin tool–delivered compounds in PS plates.
Validation Results for Different Delivery Methods, Plate Composition, and Storage Conditions a
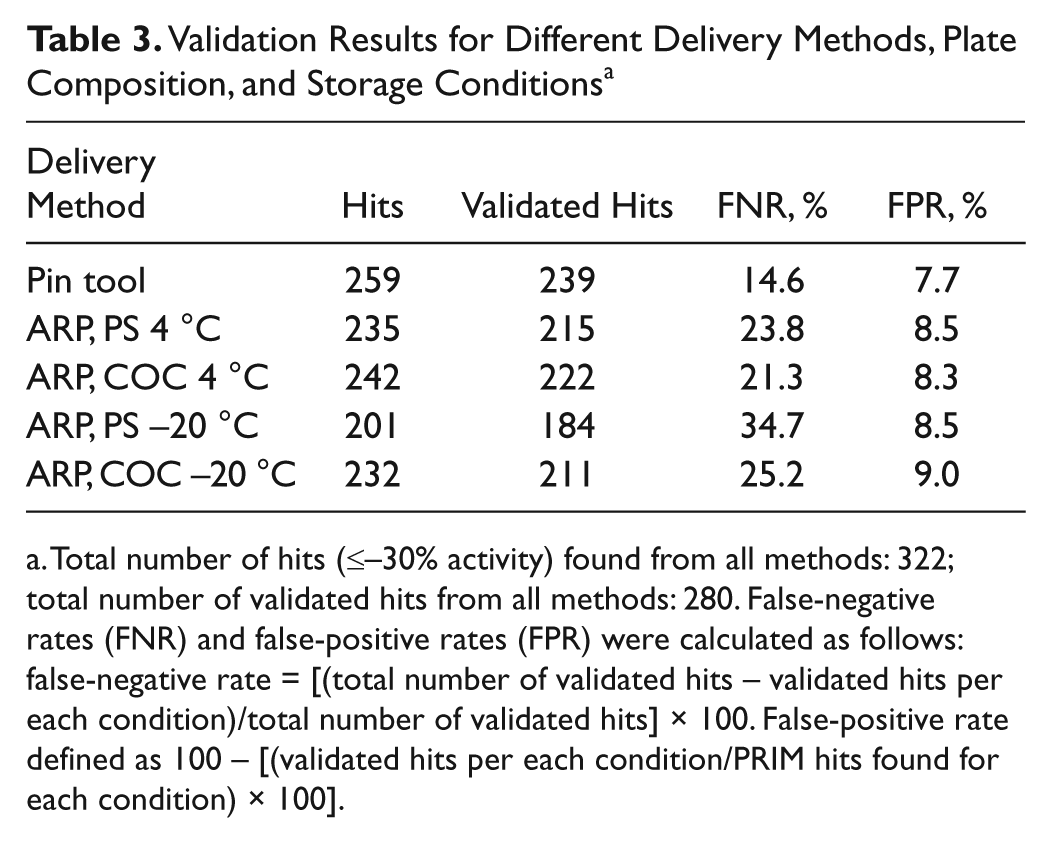
Total number of hits (≤–30% activity) found from all methods: 322; total number of validated hits from all methods: 280. False-negative rates (FNR) and false-positive rates (FPR) were calculated as follows: false-negative rate = [(total number of validated hits – validated hits per each condition)/total number of validated hits] × 100. False-positive rate defined as 100 – [(validated hits per each condition/PRIM hits found for each condition) × 100].
Discussion
As the HTS process continually evolves to tackle challenging targets while increasing throughput in miniaturized assay volumes, new mechanisms to ensure the full assessment of compound activity against desired targets continue to be developed. This includes new microtiter plate formats with new chemical compositions, new liquid-handling and delivery systems, as well as new methods of compound storage and delivery. To maximize the chances of identifying all of the active compounds in a screening campaign, we sought to determine how compound presentation influences compound potency in a biochemical assay. Using a commonly used reporter enzyme, we assessed compound inhibition potency by screening a diverse collection of 8800 compounds in a biochemical luminescence assay format. This was conducted using our typical screening default microtiter plates composed of PS as well as COC plates and analyzing the method of compound delivery to the assay using acoustic dispensing compared with pin tool delivery ( Figure 2 ).
In this study, we found a clear difference in the apparent inhibition potency for compounds when delivered via pin tool directly into an assay or when compounds were preplated as ARP. As shown in Table 1 , pin tool delivery results in a higher hit rate (2.94%) than did ARP prepared and stored overnight at 4 °C. This was true for both PS plates and COC plates. The differences in hit rate became more apparent, however, when analyzing ARP prepared and stored overnight at −20 °C. PS ARPs showed a much lower hit rate compared with COC plates when stored frozen. Examination of the difference in this compound delivery scenario showed that this condition showed the greatest number of samples exhibiting more potent activity in pin tool versus ARP ( Table 2 ). Comparative analysis of temperature and type of plate plastic (COC or PS) also supports that ARPs prepared using 20 nL spots should be used in a just-in-time mode and never frozen, with the type of plate plastic (PS or COC) having less of an effect on compound potency when the plates are not frozen.
As this study was carried out in duplicate for all ARPs (and quadruplicate for the pin tool delivery), and because the correlation plots for the FLuc assay were in excellent agreement, we picked hits from all tests to confirm the activity. From the validation data shown in Figure 5 for the different methods, it is clear that more of these hits confirmed with pin tool addition compared with ARP. Table 3 summarizes these findings and shows that although the false-positive rate did not change substantially from method to method (supporting the high reproducibility of the luciferase assay), the false-negative rate did change between methods. These missed opportunities became very evident in the PS ARPs, with a false-negative rate of ~24% when stored at −20°C. When PS ARPs were stored at −20°C, the false-negative rate was ~35%, with 97 validated compounds missed. Extending this to a 1 million sample screen, we would expect ~11K false negatives for the luciferase assay using PS ARPs with storage at −20° compared with using pin tool delivery of compounds.
There are several possibilities that could explain the differences in hit rate between the different compound addition methods. One could be that the differences arise from the sample source. However, in our controlled study, we used the same compound source plate for both pin tool delivery and ARP production to eliminate differences due to the compound samples (e.g., concentration or purity). Another reason could be that acoustic dispensing provides a different amount of compound sample than pin tool addition. However, before initiating the experiments and prior to conducting all testing, the pin tool and acoustic dispenser were checked for delivery volume accuracy and precision using a standard fluorescein stock solution in 90% DMSO/10% water to confirm that both methods were delivering nearly equal volumes. Thus, these trivial explanations seem unlikely to explain the differences in activity, especially when comparing ARPs using either PS or COC plates.
A possibility for the differences observed is that the single −20 °C overnight freeze-thaw cycle in the ARPs decreased the compound purity (e.g., the compounds were degraded) and thus potential FLuc inhibitors were lost. We did indeed observe that storage of ARPs at −20 °C overnight showed significantly less activity. Although freeze-thaw effects on compound purity were not carried out as part of this study, at least four other compound management groups within the pharmaceutical industry have looked at the effects of freeze-thaw cycles on purity. Blaxill and colleagues at GlaxoSmithKline analyzed 160 compound samples for stability and found no evidence of increased compound degradation with increased number of freeze-thaws and that freeze-thawing is not correlated with sample stability. 19 Cheng and colleagues at Abbott Laboratories reported in a similar study of 644 repository compounds that no significant compound degradation was observed after 11 freeze-thaw cycles assessed by LC/UV/MS, although with a small amount of compounds (2%), there was visible precipitation after 1 freeze-thaw cycle. 20 Another study at Proctor and Gamble identified the maximum number of freeze-thaw cycles that were allowable to maintain compound purity, with 1 freeze-thaw not predicted to result in any compound losses. 21 A study at the National Institutes of Health on the PubChem library showed no significant loss of compound integrity up to 37 weeks with storage at −80 °C. 22 Lastly, in a study carried out at Bristol-Myers Squibb, Matson and colleagues reported that after freeze-thaws, only 1 of 11 chemotypes analyzed had >50% decrease in purity, although the authors concluded that loss of sample could have been a result of degradation or precipitation caused by water uptake. 23
Another possibility to explain the apparent potency differences that compounds displayed using ARPs is that compounds either precipitated onto or were sequestered by the plate plastic and therefore not available in the assay system. This could be due to concentration of the compound due to evaporation of the 20 nL DMSO spot and subsequent insolubility in the assay buffer or increased water uptake leading to precipitation and/or adsorption to the microtiter plate plastic, which, as noted by the studies cited above, could be worsened by the storage at low temperature.
There are some studies reporting the loss of DMSO droplets and corresponding loss of active compound activity in preplated ARPs over time in storage. In particular, some compounds dried down onto PS 1536-well plates (Evotec NanoCarrier) are very poorly recovered. 10 The study examined 15 known drugs and showed that 100 nL compound spots could be dried on 1536-well plates within 20 min at 30 °C. When attempting to recover the compounds in a typical assay buffer (50 mM HEPES, pH 7.4, 50 mM NaCl, 0.1% Pluronic F-127) for 1 h at 20 °C in a humidified atmosphere, compound recoveries ranged from 100% to less than 10%. The study concluded that the use of certain excipients greatly facilitated compound recovery, but in the absence of excipients, compound losses could be expected. Similarly, using 229 compounds with a range of inhibition profiles, Matson and colleagues showed that evaporating these compounds to dryness in microtiter plates and then attempting to redissolve and assess the compound activity in biological assays, 7.8% of the inhibitory compounds lost activity. 23 In another study using PS ARPs stored under different conditions and then used in several biochemical assays, the authors demonstrated that although most compounds showed similar potencies across the conditions tested, there was an apparent decline of activity for some compounds in PS plates stored for 13 days at either room temperature or −18 °C, as well for compounds stored 6 days at −18 °C. 5 Lastly, similar to the data presented in our study, Turner and colleagues saw less of an effect on biological assay performance using long-term storage of assay-ready COC plates, although that study was conducted using a lower percentage of DMSO (20 nL of 75% DMSO/25% water) than this study (90% DMSO/10% water), which potentially affects the evaporation rate of the 20 nL droplets. 7 In addition, the water-DMSO phase diagram 24 predicts that a solution composed of 90% DMSO/10% water will be frozen at −20 °C, but if the percentage water actually increased in the 20 nL spot by twofold, the solution would be close to the phase transition and compounds would be expected to be less soluble under these conditions. Our process in which plates are lidded and vacuum/heat sealed within 1 h of plating should limit water absorption, but although it is not studied here, one possibility is to place ARPs at −80 °C until ready to use to ensure the sample is frozen, which could improve storage performance. An option worth considering is the inclusion of excipients in ARP, which, as just discussed, could facilitate the recovery of some dried-down compound samples in ARP. One would need to evaluate the effects of the excipient chosen on each assay under consideration, however, prior to the large-scale production of ARPs for HTS, and a cellular or biochemical assay sensitive to such excipient would need an alternative approach.
Similarly, problems related to absorption/precipitation to the plate plastic could be lessened if larger DMSO spots are used to prepare ARPs. In a follow-up experiment in our laboratory comparing 50 nL compound spots to 20 nL compound spots in PS plates, we found that a significantly larger number of luciferase inhibitor compounds could be found in the 50 nL droplets, even though the final compound concentrations (10 µM) were the same (data not shown). Thus, one possibility to mitigate the loss of potency observed here is to increase the size of the compound spot; however, for 1536-well ARPs, increasing the compound spot volume will likely result in unacceptable compound-screening concentrations and DMSO percentages.
The study described here was sparked by several historical observations suggesting a difference in compound potency when ARPs or pin tools were used to deliver compounds. We constructed a controlled study with a robust biochemical assay to test this notion using our current screening workflow. The results show that pin-tool delivery results in more validated hits than ARPs. Nonetheless, acoustic dispensing is a powerful technology that when properly used can be very enabling. Acoustic dispensing can be used to create low-volume (~100–500 nL) source plates for the pin tool, which can enable high-throughput cherry-picking operations. However, from the results of this study, we would recommend that if ARPs are employed, these should be prepared in a “just-in-time” mode and that COC plates should be used if cold storage is required.
Footnotes
Acknowledgements
We wish to thank Melissa Grippo and John Peltier from the Hit Discovery Group for compound quality control analysis, Meir Glick from Lead Discovery Informatics for hit list assessments, and Manori Turmel, Scott Bowes, and Greg Wendel from the Sample Management Group for helpful suggestions during the study. We thank Chun-Hao Chiu and Vincent Yu from the Hit Discovery Group for early pin tool and ARP assay evaluations. We thank Reinhard Bergmann from Global Scientific Operations for evaluation of the statistical analyses. We also thank John Davies and Dejan Bojanic of the Lead Finding Platform for support of this study and critical discussions.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support for the research, authorship, and/or publication of this article as employees of Novartis.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
