Abstract
HIV-1 envelope glycoprotein (Env) is a major determinant of viral pathogenicity. The evaluation of the biological properties of patient-derived envelopes by comparing two effector cell lines (293T and HeLa) is reported. A standard cell-to-cell fusion assay was used to evaluate fusogenicity, whereas a coculture with CD4+ cells was used to evaluate absolute cell loss, single cell death, and hemifusion events. Fusion and absolute cell loss assays showed that Env-expressing 293T and HeLa cells had different fusion efficiencies; fusion was magnified in 293T cells despite a significantly lower cell-surface Env expression. Conversely, gp41-mediated single cell death and hemifusion induced in CD4+ cells by 293T-Env-positive cells were significantly lower than that induced by HeLa-Env-positive cells. These data showed that the effector cell line used in the in vitro assays is crucial, and a combination of assays is recommended to evaluate the biological properties of patient-derived envelope glycoproteins: preferentially, 293T-Env-positive cells for the evaluation of fusogenicity and HeLa-Env-positive cells for the evaluation of cell death parameters. The combination of assays described in our work could be a valuable tool for dual screenings of large collections of primary Envs or Env mutants and drugs acting on these Envs.
Introduction
Human immunodeficiency virus (HIV) infection leads to a gradual depletion of CD4+ cells that results in immunodeficiency. 1 This process involves several highly debated mechanisms: the direct killing of infected cells, the indirect killing of uninfected cells, a defect in the capacity for lymphocyte proliferation, and/or an overactive chronic immune response and immune activation. 2 Although the contribution of these mechanisms to CD4 depletion in vivo is not yet fully understood, it has been established that the number of infected cells detected in individuals infected with HIV is much lower than the number that would be needed to achieve the profound loss of CD4+ cells seen with disease progression, suggesting that death of bystander cells by apoptosis, autophagy, or abortive infection is one of the major mechanisms of cell destruction.3–5 In fact, most CD4+ cells undergoing apoptosis in the peripheral blood and lymph nodes of patients infected with HIV are uninfected. 6
Although the function of the HIV envelope glycoprotein complex (gp120/gp41, Env) is to facilitate the entry of the viral nucleocapsid into the target cell by mediating fusion of the viral and cellular membranes, it has also been shown that this glycoprotein plays a crucial role in the depletion of CD4+ cells.
Viral Env glycoproteins expressed on infected cells may induce cell depletion by cell-to-cell fusion and syncytium formation, 7 which results in the activation of autophagic and intrinsic apoptotic pathways8,9 or the death of single uninfected bystander cells.3,10 Env-induced bystander single cell death is mediated by gp41 and is associated with the membrane hemifusion processes between the envelope-expressing cells and the target cells.11,12 Hemifusion is a phenomenon in which the outer leaflets of the membranes of the effector and target cells interact transiently without the formation of a fusion pore or progression to the formation of syncytia. This process results in an exchange of lipids between hemifused cells, which can be measured by the transfer of lipophilic dyes from the effector cells to the target cells; this process may lead to membrane destabilization and may trigger apoptotic signals during cell-to-cell contact. Hemifusion has been well characterized for influenza hemagglutinin 13 and has also been reported for HIV.11,12,14 A number of cell-based surrogate assay systems have been reported to mimic the HIV fusion and hemifusion processes, most of them based on a reporter gene under the control of the T7 polymerase or HIV-1 long terminal repeat (LTR).15–17 Nevertheless, these assays only measure fusion capacity, and mainly laboratory-adapted 11 or point mutant envelopes have been evaluated. 12 Few efforts have been dedicated to measure clinically relevant envelopes, and no standard method is available to measure envelope-induced cytopathicity. 18
Because of the central role of gp41 in mediating CD4 cell death, it is important to address the pathogenesis of primary, rather than laboratory-adapted, gp41 proteins to evaluate how changes in gp41 could modify the pathogenesis of the envelope protein. Moreover, because gp41 is involved in CD4+ cell loss by different mechanisms, it is important to evaluate absolute loss and bystander CD4+ cell apoptosis together with fusion capacity to fully characterize gp41-induced cytopathicity. In this study, the biological properties of patient-derived Env/gp41 glycoproteins were evaluated using two envelope-expressing cell lines (293T and HeLa). The results showed a differential behavior between envelopes expressed in both cell lines, highlighting the relevance of the selection of the envelope-expressing cell line used in the in vitro assays, especially when the cytopathic properties of impaired or patient-derived gp41 glycoproteins are evaluated.
The standardization of a methodology capable of quantifying the total cytopathicity of HIV envelopes can be used for screening of HIV-1 active compounds in the first steps of HIV replication and determining their mode of action. In addition, our patient-derived envelope cloning system would allow determining changes in the envelope proteins present in HIV-infected patients and identifying potential active compounds against these viral variants circulating in the population.
Materials and Methods
Cell Cultures and Reagents
The 293T cell line was obtained from American Type Culture Collection (ATCC; LGC Standards, Middlesex, UK). The HeLa and TZM-bl cell lines were supplied by the National Institutes of Health (NIH) AIDS Research and Reference Program. Cell lines were grown in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% heat-inactivated fetal calf serum (FCS) and maintained at 37 °C in a 5% CO2 incubator. Peripheral blood mononuclear cells (PBMCs) were freshly isolated from buffy coats obtained at a local blood bank (Banc de Sang i Teixits [BST], Barcelona, Spain) and used to purify primary CD4+ cells by negative immunomagnetic selection (Miltenyi Biotec, Madrid, Spain). Final preparations were >95% of CD4+ T cells as assessed by flow cytometry. The isolated CD4+ T cells were incubated overnight at 37 °C in RPMI/10% FCS prior to use. All media were purchased from Invitrogen (Madrid, Spain).
The fusion inhibitor C34, the CXCR4 antagonist JM-2987 (hydrobromide salt of AMD-3100), and the antibody IgGb12 were obtained through the NIH AIDS Research and Reference Program.19–21 2G12, a broadly gp120-neutralizing antibody, and the secondary antibody goat anti-human IgG were obtained from the Polymun (Vienna, Austria) and Jackson ImmunoResearch Laboratories (West Grove, PA), respectively. The Tat expression plasmid, pcTat, was obtained through the NIH AIDS Research and Reference Reagent Program. 22
The cell tracker, dichloro-dimethylacridin-one (DDAO), and the lipophilic probe, DiI, were purchased from Molecular Probes (Invitrogen). The cationic fluorescent dye, propidium iodide, and the potentiometric mitochondrial probe, DIOC6, were purchased from Sigma (Madrid, Spain) and Invitrogen, respectively.
Envelope Expression Vectors and Mutagenesis
Env expression vectors constructed previously in our laboratory were used to develop a functional assay. Briefly, gp41 coding regions derived from plasma samples collected at the different time points of three antiretroviral-experienced patients infected with HIV-1 were amplified by PCR using Platinum Taq DNA Polymerase High Fidelity (Invitrogen) and the following primers: MluF2 and RNANestedR (nucleotides 7726–7747 and 8882–8904 of the HIVHXB2 numbering system, respectively). A fragment corresponding to the gp120 protein was amplified from a NL4-3 plasmid (primers RNANestedF and MluR2: nucleotides 5954–5983 and 7727–7747 of the HIVHXB2 numbering system, respectively). The purified gp41 and gp120 products, which overlapped each other in 22 bases, were combined and assembled by PCR and purified to obtain hybrid envelopes (gp120 from NL4-3 and gp41 from patients) that were directionally cloned into the vector, pcDNA.3.1D/V5/His-TOPO (Invitrogen), obtaining 48 Env recombinant expression vectors. As controls, constructs coding for the Env of NL4-3, LaI, and the fusion-defective mutant, 41.2, 23 were cloned into the pcDNA3.1 vector. The additional Env mutant, D589L, described as a hemifusion-active but fusion-defective control, 12 was constructed by introducing a point mutation in the NL4-3 and LaI Env backgrounds using the GeneTailor Site-Directed Mutagenesis Kit (Invitrogen) with specific primers.
Hela and 293T Transfections
The two cell lines selected to express Env constructs (293T and HeLa cells) were plated in parallel at a density of 8 × 105 cells/well in six-well plates and allowed to grow overnight. To obtain Env-positive cells, both cell lines were transfected with 1.3 µg of the Env-expressing plasmids for the cocultures with primary cells or were cotransfected with the Env-expressing plasmids and 2.7 µg of pcTat for the fusion assays. Transient transfections of 293T cells were accomplished using the Calphos Mammalian transfection kit (Clontech Laboratories, Mountain View, CA), whereas Lipofectamine 2000 (Invitrogen) was used for HeLa cell transfections. Twenty-four hours posttransfection, cells were collected for further analyses. As negative controls, cells were mock-transfected (pcDNA 3.1 vector) or transfected only with pcTat.
Cell Surface Envelope Expression
Twenty-four hours posttransfection, 2 × 105 293T- or HeLa-Env-positive cells were harvested and incubated with the anti-gp120 monoclonal antibodies, 2G12, or IgGb12 (4 µg/mL) for 20 min at 37 °C. After being washed twice, the cells were incubated with phycoerythrin-labeled goat anti-human IgG at room temperature (RT) for 15 min. Cells were washed, fixed in formaldehyde 1%, acquired in a fluorescence-activated cell sorting (FACS) LSRII flow cytometer, and analyzed using the FACSDiva software (BD Biosciences, San Jose, CA). Mock-transfected cells were used as a negative staining control. The percentage of Env-positive cells and the geometric mean fluorescence intensity (geoMFI) of these cells were considered as individual parameters or used to calculate the relative fluorescence intensity (RFI = % of Env-positive cells × geoMFI of Env-positive cells) as described previously. 18
Cell-to-Cell Fusion Assay
Env/pcTat- and pcTat-transfected 293T and HeLa cells (3 × 104 cells) were cocultured 24 hours posttransfection with the reporter cell line, TZM-bl (3 × 104 cells), for 6 h or in kinetic experiments for 2, 4, 6, 12, 24, and 52 h in 96-well plates in the presence and absence of the CXCR4 coreceptor inhibitor, JM-2987 (1 µg/mL). The fusion efficiency of each clone was quantified by assessing luminescence (Britelite kit; PerkinElmer, Waltham, MA) with a Luminoskan Ascent luminometer (Labsystems, Barcelona, Spain).
Envelope-Induced Cytopathicity in Primary CD4+ T Cells: Absolute Cell Loss and Bystander Apoptosis
Env-induced cytopathicity was evaluated using a coculture system of Env-expressing 293T or HeLa cells as effector cells and labeled primary CD4+ T cells as target cells. Primary CD4+ T cells were stained with 10 µg/mL of the far red cell tracker, DDAO, for 1 h at 37 °C. Env-expressing 293T or HeLa cells (7.5 × 104) were cocultured with 5 × 104 of extensively washed CD4+/DDAO+ T cells on 96-well plates in the absence or presence of the inhibitor, JM-2987 (1 µg/mL). Cocultures were incubated for 24 h and then stained with DiOC6 (40 nM) and propidium iodide (5 µg/mL) for 1 h at 37 °C. MicroBeads (Beads Perfect Count; Invitrogen) were added to quantify the absolute loss of the CD4+/DDAO+ T cells by flow cytometry.
Hemifusion Assay
Twenty-four hours posttransfection, 293T and HeLa effector cells expressing NL4-3, D589L, and 41.2 Envs were labeled with 1 µL of the lipophilic probe, DiI (1 mM), and incubated for 5 min at 37 °C. CD4+/DDAO+ cells, prepared as described above, were used as target cells for the hemifusion assays. The labeled effector cells (7.5 × 104) were cocultured with the target cells (5 × 104) in the presence and absence of the C34 inhibitor (5 µg/mL). After 24 h at 37 °C, the cocultures were analyzed by flow cytometry. In all of the flow cytometric analyses, CD4+ T cells were identified by morphological parameters (forward and side scatter) and by DDAO staining.
Statistical Analyses
Variables were compared using nonparametric Mann-Whitney tests. All statistical analyses were performed using GraphPad Prism version 5.01 for Windows (GraphPad Software, San Diego, CA). A p-value of 0.05 was considered significant for these studies.
Results
Cell-Surface Expression of HIV-1 Recombinant Envelope Glycoproteins
To measure the envelope-induced cytopathicity, two effector cell lines were selected for analysis. The 293T 16 and, to a lesser extent, HeLa 24 cell lines have been used previously as effector cells in several cell-to-cell fusion assays, although a comparison between both cellular models has never been made. The potential differences in transfection efficiency, Env processing, delivery to cell surface, and other cellular factors between the cell lines could make one of them more suitable for the quantification of Env activity. After optimization of the transfection and cell surface staining procedures, both cell lines were transiently transfected with 48 hybrid Env-expressing plasmids (bearing NL4-3 gp120 and patient-derived gp41 sequences). Accordingly, the tropism of these Envs was consistent (X4) because of the gp120 subunit that is common to all of them. Cells were harvested and analyzed for Env expression with the human anti-gp120 antibody, 2G12. Env expression, as determined by the percentage of positive cells, was different in both cell lines, and a significantly higher frequency of Env-positive cells was observed in the HeLa cells than in the 293T cells for the tested envelopes (p < 0.0001) ( Fig. 1A ). Because of the low expression observed in 293T, the processing and expression in 293T were confirmed by Western blot analysis (data not shown). Similar statistically significant differences were observed when the Env expression level, as measured by the geoMean fluorescence intensity ( Fig. 1B ) or the relative fluorescence intensity, which is a measure of total Env expression ( Fig. 1C ), was evaluated. The different patterns of expression were further confirmed using another human anti-gp120 antibody, IgG1 b12, which binds to the CD4-binding site epitope in the gp120 protein, and therefore ruling out differences in envelope glycosylation as a potential cause of different expression between the two cell lines (data not shown).
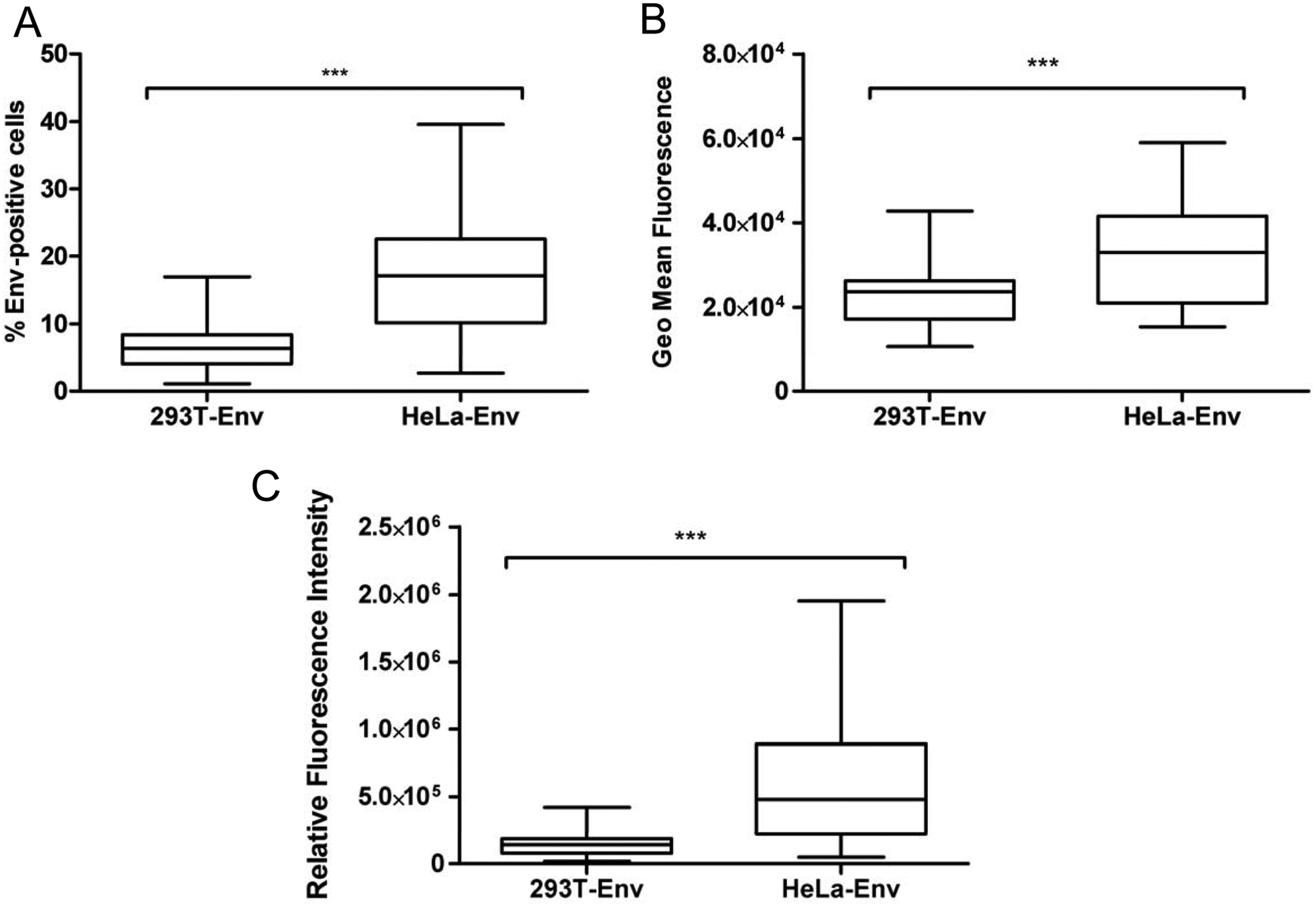
Quantification of Env expression in transfected 293T and HeLa cells. Forty-eight Env clones were transfected in 293T and HeLa cells and analyzed for their surface expression 24 h posttransfection with the 2G12 antibody. Envelope expression was determined by the percentage of Env-positive cells (
Analysis of Envelope Fusogenicity
In the past decade, a number of different fusion assay systems have been reported that use a variety of methods to measure cell fusion, including the counting of syncytia by light microscopy or fluorescence-based cytometric methods.25,26 However, reporter genes are widely used because they are sensitive and quantitative.15–17 In this study, a luciferase reporter gene assay was selected using TZM-bl cells, which contained an HIV-LTR-driven luciferase reporter cassette. As previously shown, 6 h after coculturing Env-expressing cells with reporter TZM-bl cells, luminescence was measured to determine the fusion capacity of each recombinant envelope. 12 This time point provides detectable levels of luciferase activity that are not influenced by the death of syncytia and are, therefore, the direct result of fusion. 16 This fusion assay showed significantly different fusion values when Envs were expressed in the 293T cells and in the HeLa cells (p = 0.018; Fig. 2A , gray bars), being higher in the 293T cells. This finding cannot be due to a higher cell surface expression in the 293T cells because the expression was significantly lower than in the HeLa cells ( Fig. 1 ). Alternatively, a faster fusion kinetic in the 293T-Env-positive cells could have resulted in the observed higher luminescence values at shorter times. However, when the fusion capacity was analyzed for two wild-type (wt) envelopes (NL4-3 and LaI) over time in a 52-h coculture, the same kinetic was observed in both effector cells; however, Env-expressing 293T cells maintained an increase in absolute fusion values over the entire period. The signal was detectable as early as 2 h in both Env-expressing cells, and it reached a plateau at 12 to 24 h in both cocultures ( Fig. 2B ). The increased fusion efficiency in the 293T cells, as measured by luminometry, paralleled a higher induction of syncytia in the culture, as determined by optical microscopy, with destruction of most of the culture after 24 h of coculture (data not shown). To check for the reproducibility of this fusion assay, a control envelope (the NL4-3 wt) was transfected in each experiment. Importantly, when the data of the primary envelope were normalized to the NL4-3 envelope controls (100%) of each experiment, the fusogenicity was comparable between both cell lines (p = 0.915; Fig. 2A , white bars). As expected, all the clones were inhibited by addition of the CXCR4 inhibitor JM-2987 ( Fig. 2A ).
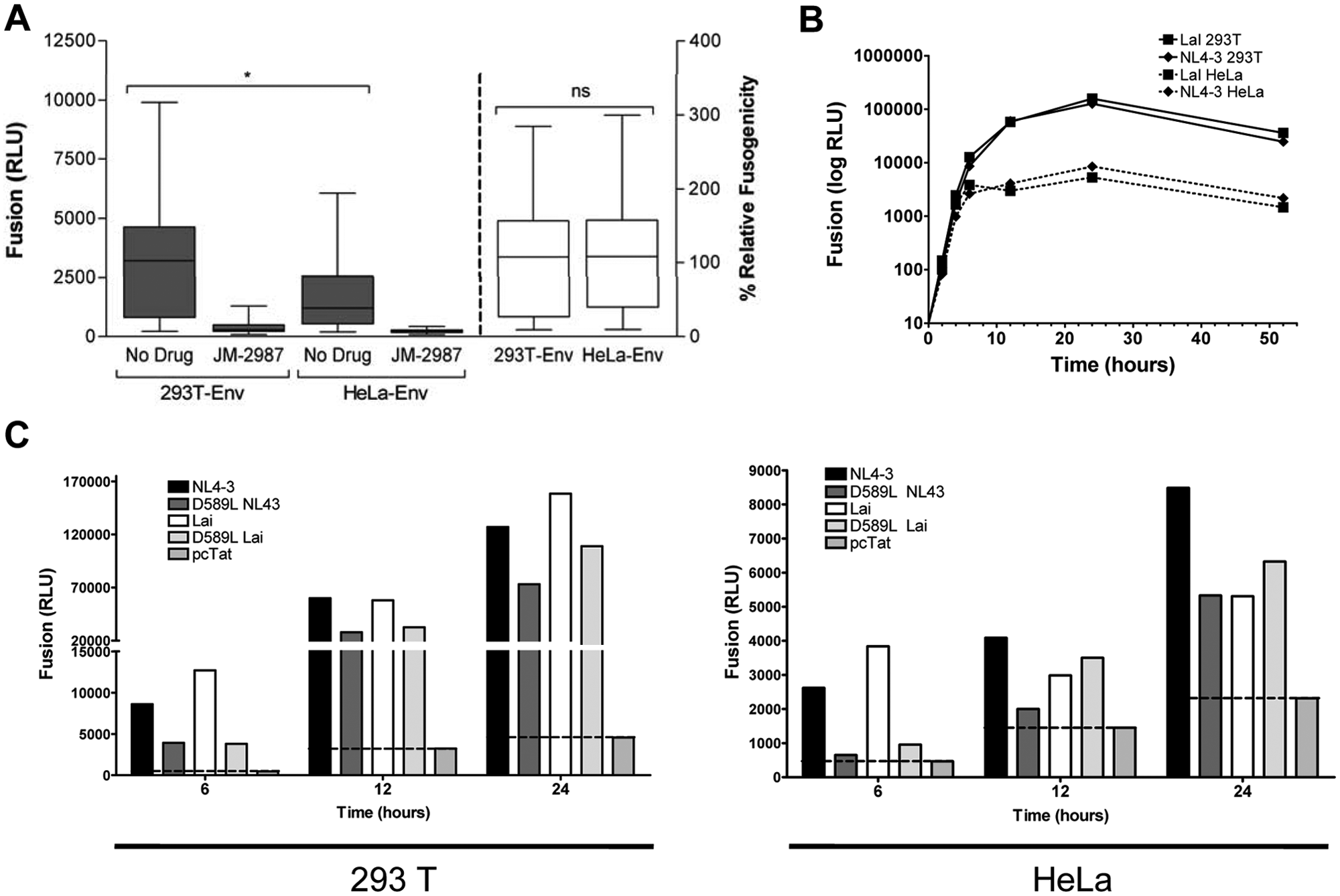
Fusion mediated by envelope glycoproteins expressed in 293T and HeLa cells. (
The relevance of those differences in absolute fusion was evaluated using an Env mutant (D589L) described previously as defective in cell-to-cell fusion. 12 This mutation, located in the distal end of HR1, seems to affect the formation and dilation of fusion pores. Two different backgrounds (NL4-3 and LaI) were used to introduce that point mutation to rule out the effect of other changes along the envelope sequence. At 6 h of coculture, neither of the two D589L variants (D589L-NL43 and D589L-Lai) fused or, at least, the fusion was highly impaired when the envelopes were expressed in the HeLa cells (9% and 14% normalized to NL4-3 and LaI, respectively), as shown previously 12 ( Fig. 2C ). Nevertheless, both variants (D589L-NL43 and D589L-Lai) showed fusion activity (43% and 27% normalized to NL4-3 and LaI, respectively) when the coculture was performed with 293T-Env-positive cells. Longer durations of coculture showed that the D589L mutants were fusogenic in both cell lines, showing, in general, lower but comparable fusion activity than their respective wt forms ( Fig. 2C ). Thus, these results highlight the importance of the effector cell line in fusion readout and suggest a higher sensitivity of the 293T-based assay.
Quantification of the Envelope-Induced Cytopathicity in CD4+ T Cells
Analysis of envelope-induced bystander apoptosis
In addition to fusion, it has been shown that the Env glycoprotein plays an important role in the depletion of CD4+ T cells by inducing single uninfected bystander cell death.
The total envelope-induced cytopathicity of patient-derived envelopes was evaluated by quantifying the Env-induced single cell death in cocultures of primary CD4+ T cells with 293T- or HeLa-Env-positive cells. Because of the key role of gp41 in CD4+ cell death,11,27 the Env-expressing plasmids were constructed as gp160 recombinant proteins bearing gp120 from NL4-3 and gp41 derived from patients, thus allowing for the specific effects of changes in the gp41 subunit. Cocultures of both 293T and HeLa Env-transfected cells with purified CD4+ T cells were performed and analyzed at 24 h by flow cytometry after staining with propidium iodide and DiOC6 to determine plasma membrane function and mitochondrial transmembrane potential, respectively. CD4+ T cells that entered into the apoptotic pathway lose DiOC6 positivity and gain propidium iodide staining because of the loss of mitochondrial transmembrane potential and a progressive loss of plasma membrane integrity ( Fig. 3A ). Apoptosis culminates in the development of secondary necrosis, which is characterized by propidium iodide–positivity and DiOC6-negativity, similar to primary necrotic cells ( Fig. 3A ). Each Env was analyzed using both effector cell lines in the absence or presence of JM-2987. The background death detected in the presence of this anti-CXCR4 inhibitor determined the specific gp41-mediated apoptosis in CD4+ T cells ( Fig. 3A ). When envelope-mediated bystander apoptosis was analyzed for 37 recombinant envelopes, a significantly lower apoptosis induction was observed for Env/gp41 expressed on the 293T cells than on the HeLa cells (p < 0.0001; Fig. 3B ).
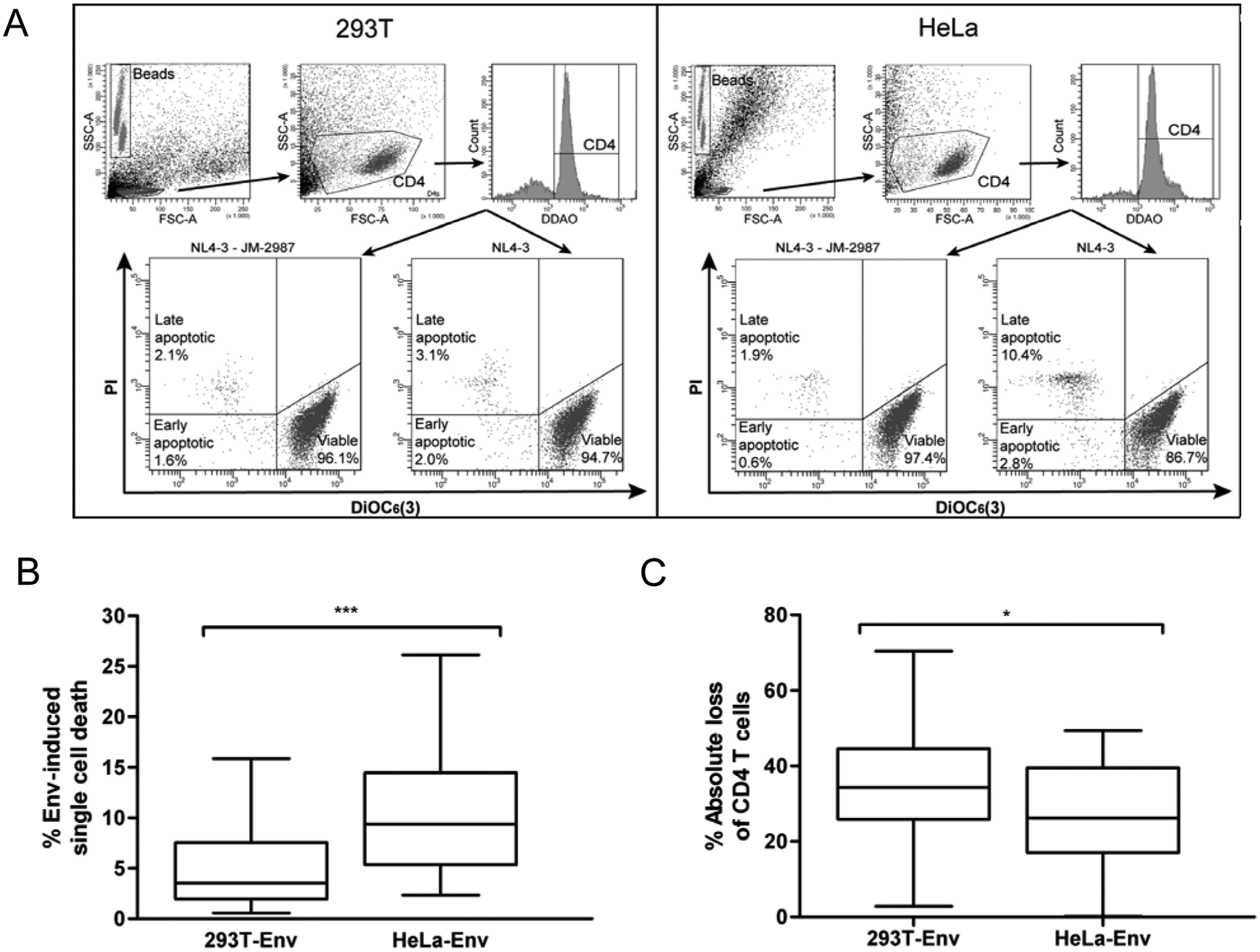
Envelope-induced cytopathicity in CD4+ T cells. (
Analysis of the envelope-induced absolute loss of CD4+ cells
To fully characterize Env-induced cytopathicity, in addition to Env-induced single cell death, the total destruction of CD4+ T cells should be evaluated to take into account the disappearance of CD4+ cells due to syncytium formation. In this experimental approach using both effector cell lines and the same gating strategy in the analysis of envelope-induced single cell death, the number of viable CD4+ T cells remaining in the culture was quantified and the absolute loss was calculated using the amount of acquired fluorescent microbeads in each sample ( Fig. 3A ). Contrary to apoptosis induction and in correlation with the absolute fusion data, the absolute loss of CD4+ cells was significantly higher when the envelopes were expressed on 293T cells than on HeLa cells (p = 0.037; Fig. 3C ), again suggesting a difference in the hemifusion/fusion processes between both cell lines.
Analysis of envelope-induced hemifusion activity
Envelope-induced single cell death has been associated with a specific gp41-mediated transfer of lipids from the membranes of Env-expressing cells to the target cells in the absence of detectable cell-to-cell fusion (hemifusion process).11,12 To further analyze the different behavior of Env expressed on these two effector cell lines, the hemifusion activity of the three following envelopes was evaluated: the NL4-3 wt and two point mutant envelopes, the D589L Env-mutant, which was described previously as defective in cell-to-cell fusion but able to mediate hemifusion, 12 and a 41.2 envelope, which could not fuse or hemifuse. 23 Before analyzing hemifusion, we confirmed that the 41.2 mutant was unable to mediate fusion in both the 293T and HeLa effector cells (data not shown) and that the D589L mutant was able to fuse ( Fig. 2 ) when expressed in the 293T cells and in long-term cultures (>6 h) in the HeLa cells. A lipophilic dye transfer assay was performed between the transiently transfected Env-positive effector (293T and HeLa) cells and CD4+ T cells. Cocultures of both of the labeled cells were performed in the presence and absence of the fusion inhibitor, C34, and flow cytometry was used to determine the transfer of lipids from the effector cells to the target cells. Primary CD4+ T dead cells were morphologically identified and specifically selected by positive DDAO staining. Cocultures resulted in the appearance of double-labeled CD4+ T dead cells (DDAO+DiI+), which were the primary cells that hemifused and that partially mixed their membrane lipids with the Env-expressing cells ( Fig. 4A ). As shown in Figure 4A , B , although both effector cells induced detectable levels of hemifusion, the transfer of dye was significantly higher in the HeLa-Env-positive cells when compared with the 293T-Env-positive cells. The higher signal-to-noise ratio that was obtained when the HeLa-Env-positive cells were used as effector cells allowed for the quantification of hemifusion events induced by the D589L mutant in that effector cell line but not when 293T-Env-positive cells were used. Overall, these data suggested that HeLa-Env-positive cells provide higher sensitivity in hemifusion assays.
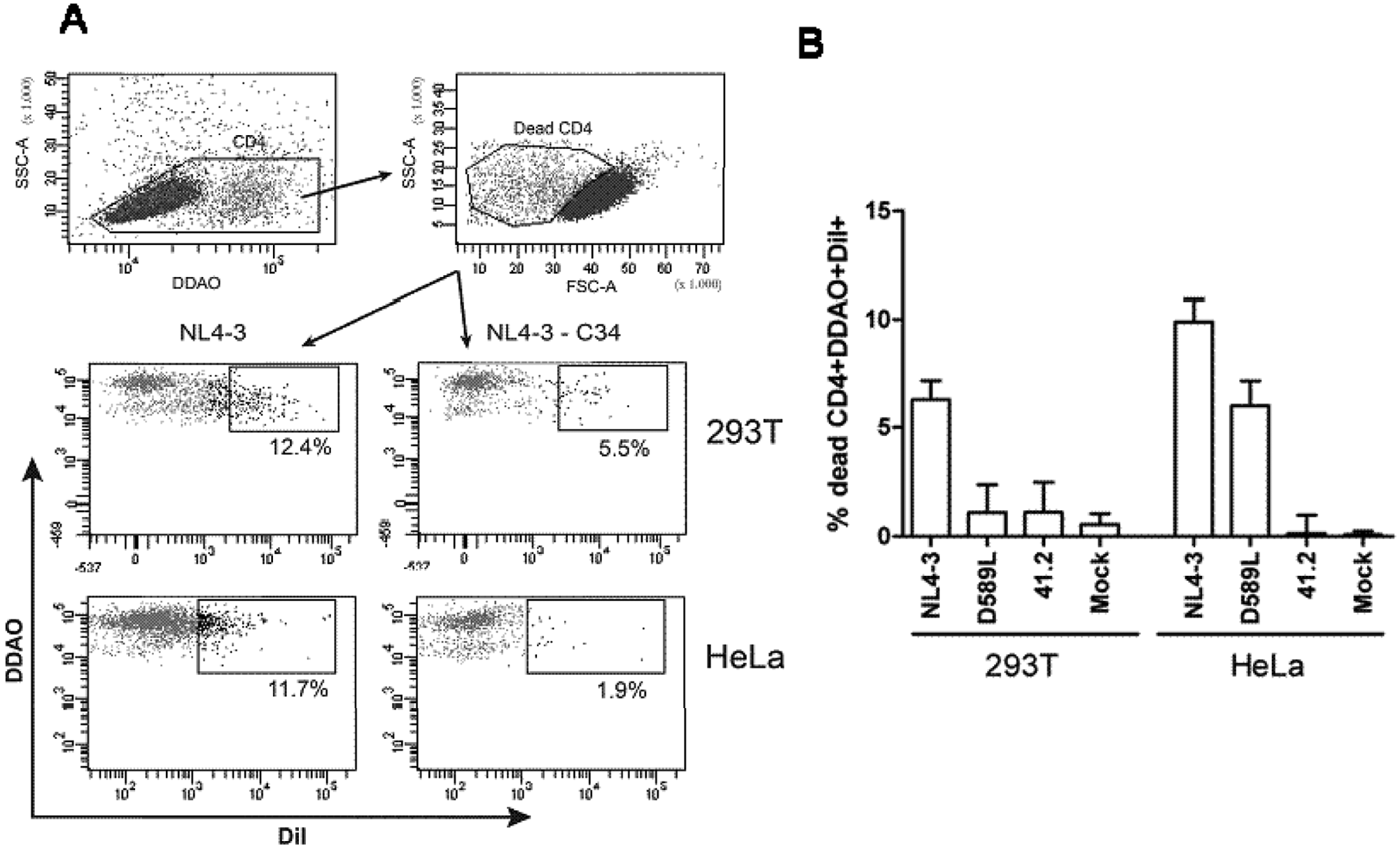
Hemifusion mediated by different HIV-1 envelopes with CD4+ T cells. (
Discussion
The fusogenic activity of HIV Env has long been assessed15–17,25,26 and associated with pathogenesis both in vitro and in vivo.28,29 Nonetheless, it has been observed that the amount of T cell depletion in patients infected with HIV far exceeds the number of infected T cells, suggesting an indirect mechanism of HIV pathogenesis by bystander cell death. 4 HIV Env expressed in infected cells may interact with target CD4+ cells, induce cell depletion by syncytium formation, or mediate the killing of innocent bystander cells.10,30,31
In the present study, the biological properties (fusion capacity and single cell death induction) of envelopes derived from patients expressed on the surface of two cell lines are reported. A cell-to-cell fusion reporter gene assay and a coculture with labeled primary CD4+ T cells were performed to evaluate absolute cell loss, single cell death, and hemifusion induced by the envelope. To use the best effector cells, two cell lines, 293T and HeLa, previously used in the evaluation of HIV Env function,16,24 were compared and transiently transfected with Env-hybrid-expressing vectors (expressing gp120 from NL4-3 and gp41 isolated from patients infected with HIV).
First, the fusion capacity was evaluated using the TZM-bl as the reporter cell line. This Tat transactivable system was chosen by its high sensitivity and wide dynamic range. 16 Consistently, fusion was detected after 2 h of cell mixing and increased over time until a plateau was reached at 12 to 24 h. Despite lower Env expression levels, higher absolute values were obtained when the envelopes were expressed on 293T cells. However, when values were normalized to the NL4-3 reference envelope, they became comparable, suggesting that envelopes showed a similar qualitative behavior in both cell lines.
To evaluate whether the differences in the fusion efficiency observed between the two cell lines was relevant for the characterization of the Env functions, the Env point mutant, D589L, described previously as defective in cell-to-cell fusion, 12 was used. This envelope showed highly impaired fusion in the standard 6 h of coculture when expressed in HeLa cells but functioned when expressed in 293T cells. The Env fusion capacity of this mutant was confirmed in both cell lines when the coculture was maintained over a longer period of time. Overall, these data suggest that in a standard 6-h coculture, the differences between both effector cells are significant enough to suggest that use of HeLa cells could miscategorize fusion-defective envelopes, and it would be more appropriate to use the more efficient cell line, 293T, as the effector cell line. Alternatively, fusion kinetic assays, although time-consuming, may also be useful to evaluate accurately the fusion capacity of impaired Env glycoproteins or Env derived from patients infected with HIV.
In addition to the fusion capacity, the Env-induced cytopathicity in primary cells was evaluated in both effector cells. For the analysis of envelope-mediated single cell death and hemifusion, a coculture of Env-expressing cells with primary CD4+ T cells isolated from healthy donors as target cells was established. Unexpectedly, the Env-mediated single cell death induced in CD4+ T cells by 293T-Env-positive cells was significantly lower than that induced by HeLa-Env-positive cells. Based on the differences between fusion and single cell death induction, the intermediate link between both processes, hemifusion, was examined. Sometimes, the fusion process would not result in a complete fusion of cells and would be interrupted at the hemifusion step. This situation could occur often, especially in vivo, where Env-induced syncytia are rarely seen in patients infected with HIV. It has been described that gp41-mediated hemifusion, defined as a membrane fusion event characterized by the mixing of the outer leaflets of the lipid bilayer without progression to fusion pore formation, is both required and sufficient to induce apoptosis in bystander cells.11,27 Consequently, to fully evaluate the behavior of the two effector cells, a hemifusion assay of lipophilic dye transfer was performed in the presence and absence of C34, which blocked efficiently syncytia and single cell death, 11 and the obtained values were compared. The results showed higher levels of specific lipid mixing between the primary and HeLa cells than the 293T effector cells, which correlates with the single cell death data. According to these results and previously reported models, 32 293T effector cells seem to follow a major direct pathway, having a direct transition to the formation of the fusion pore and generating high levels of fusion and lower levels of hemifusion and single cell death. In contrast, HeLa effector cells may mainly follow a slow indirect pathway and will fuse their external leaflets with target cells that have a large contact area by inducing greater extents of hemifusion and, therefore, single cell death and low levels of fusion.
Overall, these data suggest that, depending on the effector cell used, envelopes may mediate more hemifusion versus fusion or vice versa. The rate of fusion may depend on the lipid composition of the viral and target membranes. 33 It was reported previously that the propensity of lipid bilayers to hemifuse and develop fusion pores depends on lipid composition, cytoskeleton components, and transmembrane osmotic pressure. 34 In addition, as reported for receptor recruitment, 35 the envelope mobility in effector cells would be expected to have severe consequences in fusion.
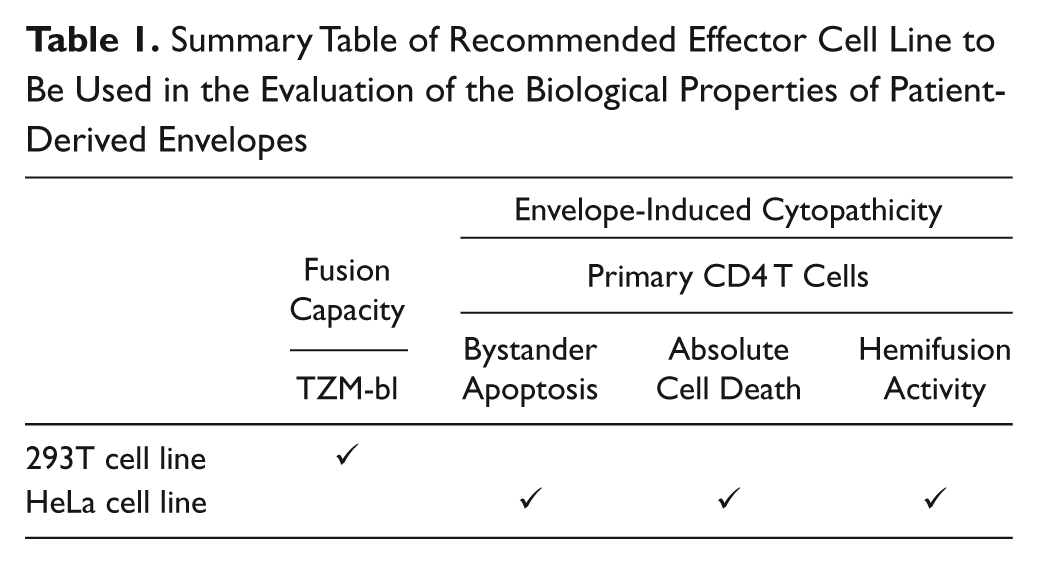
In conclusion, our results showed that the effector cell line used in different assays is crucial to avoid skewed results when the functional characterization of the biological properties of patient-derived envelopes is evaluated. Thus, a combination of assays is recommended to achieve an overall characterization of Env-mediated cytotoxic events, especially when envelopes with low fusogenic capacity, or envelopes derived from patients, are evaluated. 293T-expressing cells should be used as the effector cell line in the evaluation of fusion capacity, and HeLa-Env-expressing cells should be preferentially chosen for the evaluation of cell death parameters or intermediate fusion processes ( Table 1 ). This combination of assays yields relevant data of the cytopathicity of multiple envelopes in parallel, and it could be used for the screening of HIV-1 antiviral drugs acting in different Env functional steps, against viral envelopes that are currently circulating in the population of patients infected with HIV.
Summary Table of Recommended Effector Cell Line to Be Used in the Evaluation of the Biological Properties of Patient-Derived Envelopes
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for research and/or authorship of this article: This work was supported by the FIS project 07/0418 (to C. Cabrera) and the Spanish AIDS network “RIS, Red Temática Cooperativa de Investigación en SIDA (RD06/0006).” C. Cabrera and J. Blanco are researchers from Fundació Institut de Recerca en Ciències de la Salut Germans Trias i Pujol supported by the Health Department of the Catalan Government (Generalitat de Catalunya). F. Cunyat is supported by the FIS project 07/0418. This work is part of the Ph.D. thesis by F. Cunyat at Universitat Autònoma de Barcelona, Barcelona, Spain. M. Curriu is supported by an RIS contract.
