Abstract
Inducible gene expression systems are particularly useful for the functional characterization of genes with putative toxic properties. In the course of studying the role of hypoxia-regulated gene expression on cell survival using the tetracycline-inducible (tet-on) system, the author noted that exposure to the inducing ligand doxycycline (dox) inhibited caspase-3 cleavage in control samples. To limit this confounding off-target effect, he devised an in vitro pulse dose, delayed-injury protocol testing both dox and a novel tetracycline analog 9-t-butyl doxycycline (9-TB). Although 9-TB induced higher transgene levels compared to matched concentrations of dox, continuous exposure to both drugs inhibited caspase-3 cleavage in hypoxic samples. Conversely, a 6-h pulse dose of 9-TB followed by a 40-h washout period prior to hypoxic challenge activated robust transgene expression and lessened the inhibitory effects on caspase-3 processing. It is anticipated that these protocol modifications will improve the performance of tet-regulated genetic screens, particularly in situations where cell death is used as a primary end point.
Introduction
T
Although their use is contraindicated in childhood and pregnancy, tetracycline compounds have been used to treat a variety of medical conditions and may ultimately be suitable for use in clinical protocols involving regulated expression of therapeutic genes expressed from viral and cell-based vectors. Interestingly, in addition to their broad-spectrum antibacterial and antiprotozoal effects, tetracyclines exert both neuroprotective and anti-inflammatory effects on somatic tissues. 7 For example, both doxycycline and minocyline have been linked with inhibition of matrix metalloproteases and pro-caspase-3 activation. 5 These observations have led to speculations that these compounds may benefit patients with noninfectious, chronic degenerative disorders, including multiple sclerosis, amyotrophic lateral sclerosis (ALS), and stroke. 1–17,20
One of the principal advantages of an inducible gene expression system is having the ability to generate stable transfectants harboring toxic genes where conventional constitutive gene expression systems might otherwise fail. 11–15 In studying the role of hypoxia-induced gene expression on cell survival, we discovered that dox exposure itself inhibited stress-induced caspase-3 cleavage in control cell lines, confounding our ability to isolate effects of the gene of interest. To address this problem, we devised a pulse-dosing strategy comparing the effects of doxycycline with a novel tetracycline analog, 9-TB. As mentioned earlier, 9-TB is of particular interest given its hydrophobic properties, demonstrated central nervous system (CNS) penetration, and 10-fold higher binding affinity for TetR and rtTA relative to other related compounds. 19 Our results show that in addition to achieving higher levels of transgene expression at comparable doses, 9-TB performed better than dox after pulse dosing, inducing reasonable transgene expression while at the same time minimizing the inhibitory effects of ligand exposure on caspase-3 activation.
Materials and Methods
Reagents
Doxycycline (4-[Dimethylamino]-1,4,4α,5,5a,6,11,12α-octahydro-3,5,10,12,12α-pentahydroxy-6-methyl-1,11-dioxo-2-naphthacenecarboxamide; Sigma-Aldrich, St. Louis, MO) and the derivative 9-t-butyl Dox (9-TB; a kind gift from Mark Nelson, Paratek Pharmaceuticals, Boston, MA) were dissolved in water to a final concentration of 10 mg/mL. 12 Hygromycin was obtained from Invitrogen (Carlsbad, CA).
Cloning and stable cell line production
Inducible green fluorescent (GFP) expression was achieved by subcloning the IRES-GFP fragment into the tetracycline responsive bidirectional vector pBig2i kindly provided by Dr. Craig Strathdee (Amgen, Inc., Cambridge, MA). 16 This construct, designated pBig2i-MCS-IRES-GFP, was initially tested for inducible transgene expression by transfection into the HN33 line (a gift from Bruce Wainer, Emory, GA) passaged in Dulbecco’s modified Eagle’s medium (DMEM)/high glucose (HG)/5%. 9,10 The HN33 line was created by crossing mouse hippocampal neurons with the immortalized N18TG2 line to maintain neuronal properties and has been used to study neuronal responses to hypoxic stress. 6–14 HN33 stable lines were generated by batch transfection and selection with hygromycin according to standard protocols maintained in DMEM/8% serum containing 200 µg/mL hygromycin for maintenance. Stripped serum was used throughout to minimize basal pBig2i activity (Innovative Research, Novi, MI).
In vitro hypoxia model
In vitro hypoxia was achieved using a triple gas incubator (Binder, Marietta, OH), and oxygen tension was monitored via an on-board zirconium oxide sensor, which exhibits linear sensing proportional to oxygen concentration. Cells were exposed to 0.5% O2 for 18 h unless otherwise specified.
Western blotting
Cell lysates were obtained by rinsing monolayers with ice-cold phosphate-buffered saline (PBS) 1× followed by the addition of RIPA buffer containing protease inhibitors (Sigma-Aldrich). Samples were boiled in Laemmeli buffer for 1 min under reducing conditions and resolved by electrophoresis on polyacrylamide gels. Proteins were transferred to PVDF membranes and blocked in TBS-T (50 nM Tris-HCl [pH 8.0], 0.9% NaCl, and 0.1% Tween-20) containing 5% nonfat dry milk for 1 h at room temperature. Antibodies used in this study included GFP (1:2000; NB-100-131, Novus Biologicals, Littleton, CO), cleaved caspase-3 (1:1000; CST, Danvers, MA), and actin (1:10 000; Santa Cruz Biologicals, CA). Blots were exposed to the appropriate secondary antibodies conjugated with horseradish peroxidase (HRP; 1:5000; Santa Cruz Biologicals) prior to ECL detection (Amersham/GE Life Sciences, Piscataway, NJ). Densitometry was performed using 8-bit TIFF files and the Java-based application ImageJ (http://rsbweb.nih.gov/ij/). The area under the sample curves was determined using the analyze gel function, setting the relative background intensities with the line tool. The fold-activation reflects the level of hypoxia-induced caspase-3 cleavage compared to normoxic samples (n = 2 for each comparison group).
Results
We first compared the dose–response behavior of the HN33 tet-inducible pBig2i-MCS-IRES-GFP reporter line to a range of dox and 9-TB concentrations. After plating, cells were exposed to either drug (0.25-10 µg/mL) continuously for 40 h prior to analysis ( Fig. 1A ). Neither drug produced clear effects on cell morphology at intermediate concentrations ( Fig. 1B ). As expected, 9-TB stimulated higher levels of GFP expression from the TetO promoter relative to dox exposure. Lower levels of GFP expression observed with high-dose 9-TB (10 µg/mL) were associated with a slight decrease in actin levels ( Fig. 1C ) and changes in the growth and morphology of cultured cells (data not shown).
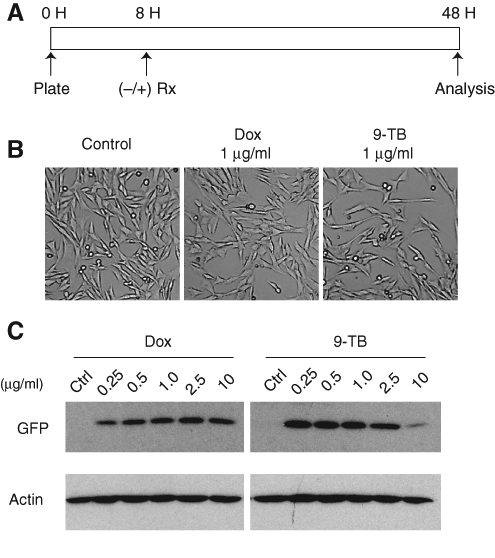
Comparison of doxycycline and 9-t-butyl doxycycline (9-TB) on inducible green fluorescent protein (GFP) expression from a tet-on reporter cell line. (
In addition to exhibiting the morphological and electrophysiological characteristics of the parental hippocampal neurons, 9 the HN33.11 line also remains sensitive to hypoxic challenge and is a reasonable model to study the apoptotic potential of candidate neuronal genes. In vitro hypoxia (0.5% O2, 24 h) induces robust cleavage of the master apoptotic regulator pro-caspase-3 ( Fig. 2 ; normoxia vs hypoxia). However, although both tet-analogs induced GFP expression as expected, continuous exposure to either dox or 9-TB also inhibited hypoxia-induced caspase-3 cleavage. This off-target activity was of particular concern because it confounds the analysis of genes with putative apoptotic effects.
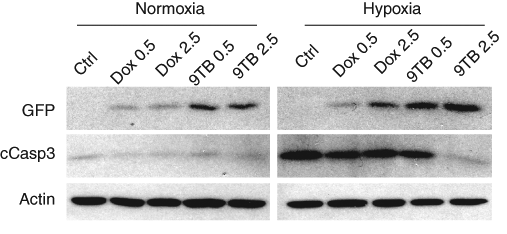
Tetracycline derivative compounds inhibit hypoxia-induced levels of cleaved caspase-3 (cCasp3). The tet-on inducible stable cell line was exposed to varying concentrations of ligand (0.5 vs 2.5 µg/mL), and parallel sister was exposed to either normoxic or hypoxic (0.5% O2) conditions for 18 h. Lysates were analyzed by Western blotting for both transgene induction (green fluorescent protein [GFP]) and cCasp3 levels.
In our experience, the kinetics of the tet-regulated system is delayed with respect to the timing of both gene induction after drug exposure and transgene silencing upon removal of ligand. In light of this, we hypothesized that the observed drug effect on gene induction could be temporally isolated from its effects on caspase activity. To test this, we devised an approach using overnight drug exposure (12 h) to trigger gene expression, followed by a washout period (16 h) accomplished by full-volume replacement of the culture media prior to hypoxic injury ( Fig. 3A ). We found that although continuous exposure with dox (2.5 µg/mL) produced levels of GFP expression comparable to that seen with equivalent 9-TB dosing, pulse dosing with 9-TB produced dramatically increased expression levels ( Fig. 3B ; GFP continuous vs pulse). As previously observed, both drugs interfered with caspase-3 cleavage. Importantly, despite higher overall levels of caspase activity after pulse dosing, the modified protocol ameliorated dox-mediated suppression of hypoxia-induced caspase-3 cleavage seen after continuous ligand exposure. This effect was less impressive in 9-TB pulse-dosed samples ( Fig. 3B , right panel, lane 4 vs 6).
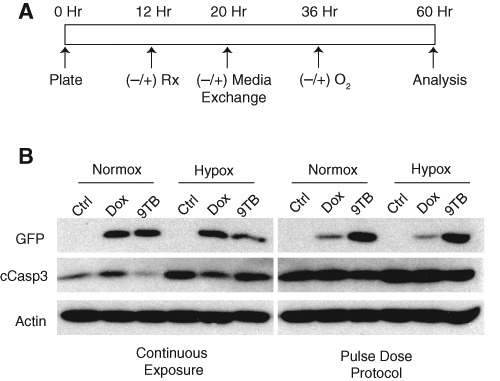
Pulse dosing with 9-t-butyl doxycycline (9-TB) supports transgene expression and mitigates the inhibitory effects of ligand on hypoxia-induced caspase-3 cleavage. (
To improve on this paradigm, we next tested whether varying the washout period would have a beneficial effect on GFP and caspase-3 levels in 9-TB-treated cultures. As expected, longer chase periods were associated with higher levels of transgene expression ( Fig. 4A ). Importantly, the suppressive effects of 2.5 µg/mL 9-TB on caspase-3 cleavage were reduced with longer washout periods ( Fig. 4B ; 12 vs 24 h). Because even low-dose 9-TB could support transgene expression given a long enough washout period, we tested whether lowering the 9-TB concentration would be beneficial. Continuous 9-TB dosing resulted in nearly equivalent transgene induction with 0.25- and 2.5-µg/mL doses after a 48-h exposure ( Fig. 5B , paradigm 3). Importantly, transient exposure (6 h) to 0.25 µg/mL 9-TB produced detectible amounts of expressed GFP and permitted a reasonable degree of caspase-3 cleavage after hypoxic challenge relative to control cultures ( Fig. 5C ; 5.2-fold vs 10.6-fold).
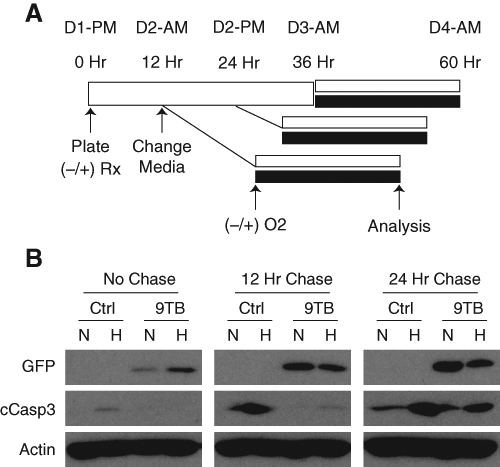
Length of the drug washout period influences green fluorescent protein (GFP) expression and caspase-3 cleavage. (
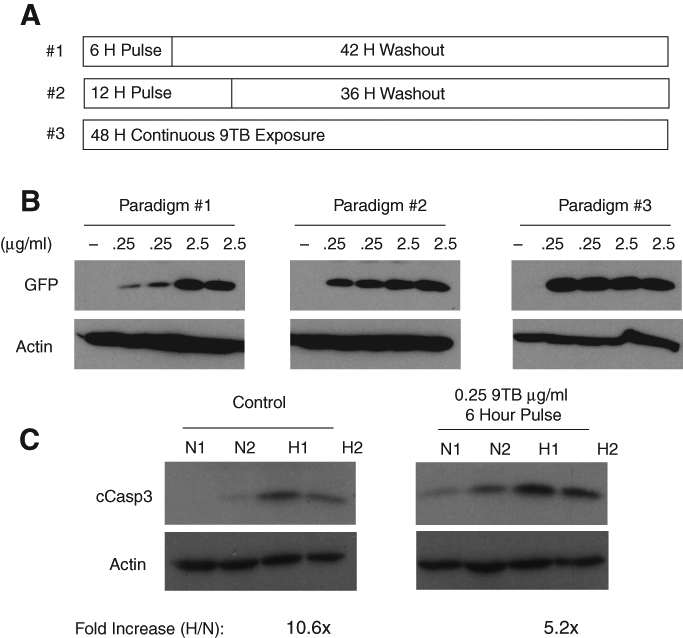
Optimization of 9-t-butyl doxycycline (9-TB) pulse-dosing protocol. (
Discussion
In addition to their potent antimicrobial properties, tetracycline analogs also have protective effects on eukaryotic cells in part through effects on inflammatory and apoptotic signaling cascades. In the course of analyzing the effects of immediate early gene expression after in vitro hypoxia, we found that in addition to inducing rtTA-regulated expression from a TetO–cytomegalovirus (CMV) promoter, continuous dox exposure inhibited apoptotic signaling as measured by cumulative levels of caspase-3 cleavage. Given our interest in understanding the influence of hypoxia-induced transcription on cell injury, this unintended off-target effect represented a technical hurdle. The principal finding of this study is that a relatively brief pulse-dosing protocol using the dox analog 9-TB can support tet-on inducible gene expression without blocking endogenous apoptotic signaling cascades typically seen when using continuous dosing regimens and common ligands.
Having tight control over the “on” and “off” state of regulated gene expression systems is of great interest, and efforts to identify tet transactivator mutants with tighter drug-like behaviors are ongoing. 8 One potential benefit of this approach would be the identification of rtTA variants with ligand binding constants several orders of magnitude lower than the affinity for ligand toward components of the apoptosome. However, as we have demonstrated, further optimization of these drug-responsive systems can also be achieved through the characterization of novel small-molecule ligands. As reported, 9-TB was more potent than doxycycline with regard to its ability to activate rtTA-driven gene expression from a stable reporter cell line. Presumably, this effect was related to its increased hydrophobicity and affinity for the tet transactivators. 19 Regarding the robust yet delayed kinetics of transgene expression observed with 9-TB, it was this property of the drug that allowed us to overcome ligand-mediated inhibition of hypoxia-induced apoptotic signaling seen with continuous dosing. Of note, although pulse dosing with doxycycline did ameliorate its suppressive effects on caspase cleavage, we were unable to stimulate comparable delayed transgene expression to acceptable levels using doxycycline after comparable brief pulse dosing (data not shown). Although there are other tetracycline derivative compounds available, we cannot comment on their potential activity in the pulse-chase paradigm. It should be noted that although inclusion of the washout period in our protocol lessened the ligand-mediated effects on cell death signaling machinery, this approach may not translate generically to other genes expressed in the context of the tet-on system for the following reason. In contrast to many of the cellular transcripts involved in the acute phase response and cell death signaling, GFP is relatively stable, exhibiting a half-life of 26 h in its unmodified state. Although not tested directly, it is possible that the pulse-delayed exposure paradigm is best suited to study proteins whose stability mirrors that of the expressed transgene.
We recognize the potential to capitalize on the “off-target” effects of dox and related compounds with regard to human disease. 9-TB has been used successfully in vivo to study the dynamic behavior of tet-responsive transgenic sequences in the central nervous system. 19 Given recent advances in the fields of cell- and viral-based therapeutics for neurodegenerative diseases and stroke, it is not far-fetched to consider using tetracycline-based gene regulation cassettes as drug delivery devices. In this context, having access to a drug capable of stimulating therapeutic gene expression while at the same time inhibiting endogenous apoptotic signaling programs represents a novel opportunity for therapeutic synergy. However, at the present time, tet-regulated gene expression systems are commonly used for commercial applications, including the large-scale production of therapeutically useful biologicals and in drug screening programs. Our observations have important implications in the latter setting where tightly regulated stable cell line systems are required for complex drug screening campaigns to identify pro- and antiapoptotic compounds for cancer and neurodegenerative conditions. We anticipate that the protocol modifications described herein will be easily translated to a wide variety of cell-based assays.
Footnotes
Acknowledgements
I thank Rita Giuliano for technical assistance in establishing the doxycycline-regulated HN33.11 stable cell line, Allison Hendershot for her assistance in characterizing the dose–response curves for doxycycline and 9-TB, and Arthur Totten for his work refining the pulse-dosing protocol. I also am grateful to Dr. Nina Schor for her thoughtful advice and review of the manuscript. Funding as provided by grants from the NIH/NINDS to MWH (R00NS060764).
