Abstract
Current pharmaceutical compound screening systems rely on cell-based assays to identify therapeutic candidates and potential toxicities. However, cells grown on 2D substrata or in suspension do not exhibit the mechanical or physiological properties of cells in vivo. To address this limitation, the authors developed an in vitro, high-throughput, 3D hydrogel tissue construct (HTC)–based assay system to quantify cell and tissue mechanical properties and multiple parameters of physiology. HTC mechanics was quantified using an automated device, and physiological status was assessed using spectroscopy-based indicators that were read on microplate readers. To demonstrate the application of this system, the authors screened 4 test compounds—rotenone (ROT), cytochalasin D (CD), 2,4-dinitrophenol (DNP), and Rho kinase inhibitor (H-1152)—for their ability to modulate HTC contractility without affecting actin integrity, mitochondrial membrane potential (MMP), or viability. All 4 compounds dose-dependently reduced HTC contractility. However, ROT was toxic, DNP dissipated MMP, and CD reduced both intracellular F-actin and viability. H-1152 was found to be the best candidate compound since it reduced HTC contractility with minimal side effects. The authors propose that their HTC-based assay system can be used to screen for compounds that modulate HTC contractility and assess the underlying physiological mechanism(s) of compound activity and toxicity.
Keywords
Introduction
T
We have developed in vitro 3D hydrogel tissue constructs (HTCs) as a more natural environment for investigating cellular traction and contractile force. HTCs are formed by the extracellular-matrix remodeling activity of the cells, and different cell types (e.g., fibroblasts) can form HTCs that exhibit mechanical behaviors similar to in vivo tissues.
9
We demonstrated that the contractile force of the HTCs can be decomposed into active and passive components attributable to intracellular actomyosin activities and extracellular matrix stiffness, respectively.
10
To increase the throughput of HTC assays, we developed miniaturized HTCs and an automatic device, the Palpator™ (

Mechanical quantitation of hydrogel tissue construct (HTC) contractile force. (
To demonstrate the utility of this high-content screening system, we profiled HTC mechanical and physiological responses to 4 representative compounds that reduce cellular contractile force: Rho kinase inhibitor H-1152, actin filament disruptor cytochalasin D (CD), MMP uncoupler 2,4-dinitrophenol (DNP), and oxidative phosphorylation inhibitor rotenone (ROT). The effect of these compounds on HTC contractility was assessed using the Palpator™, whereas effects on the actin cytoskeleton, MMP, and HTC viability were assessed using spectroscopic indicator dyes. We envision that our HTC-based high-content screening system will enable the discovery of novel nontoxic compounds that can mediate cellular and tissue contractile forces for the treatment of fibrotic diseases.
Materials and Methods
Cell culture and HTC formation
Rat embryo fibroblasts (REF-52) were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Fisher Scientific, Pittsburgh, PA) supplemented with 10% fetal bovine serum (FBS; Atlanta Biologicals, Lawrenceville, GA). Cells were subcultured every 2 to 3 days and used between passages 40 and 70 to form HTCs that exhibit comparable force activity. To make HTCs, REF-52 cells were dissociated by incubating with 0.05% trypsin (Fisher Scientific) for 10 to 15 min, pelletized by centrifugation at 1000 g for 10 min, resuspended in 10% FBS DMEM, and diluted in HTC tissue solution to 8 × 105 cells per mL. The HTC tissue solution consisted of 10% FBS DMEM, 1 mg/mL type 1 collagen (BD Biosciences, San Jose, CA) in 0.02 N acetic acid, sodium hydroxide (NaOH) to neutralize the acid, and 5× DMEM to compensate for the volume of collagen and NaOH. The HTC tissue solution was aliquoted (300 µL/well) into 8-well mini-construct chambers (MC-8™, InvivoSciences, McFarland, WI) 11 and incubated at 37°C and 5% CO2 for 30 min. Then, 350 µL of 10% FBS DMEM medium was added to each well for an additional 48 h of incubation.
HTC force measurement
The MC-8™s were placed on the stage of the Palpator™, which automatically inserted a probe into each well and stretched the individual HTC. The probe was connected to a force transducer that measured the resistance force induced in the HTCs. These forces were recorded, and a custom Matlab algorithm 11 was used to analyze the data and quantify the active cell contractile force. To obtain stable measurements of contractile force, HTCs were preconditioned prior to force measurements. 11 HTCs were preconditioned by being stretched 3 consecutive times on the Palpator™ device. Preconditioning was not necessary if subsequent force measurements were within 30 min of the previous stretch.
HTC TMRE labeling and MTT assay
HTCs were incubated in 100 nM tetramethylrhodamine (TMRE; Invitrogen, Carlsbad, CA) for 30 min and then in phenol red–free 10% FBS DMEM for 60 min. HTC TMRE signal was read on a Synergy HT plate reader (Biotek Instruments, Winooski, VT). A custom adapter plate was used to position the MC-8™s on the plate-holding rack, and the 543/590 filter set was used with a gain setting of 50.
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium (MTT; Invitrogen) diluted in DMEM was added to each well to achieve 0.5 mg/mL and then incubated for 2 h. The formazan dye that forms in the cells was then extracted using 500 µL of isopropanol (Fisher Scientific) containing 0.1 N hydrochloric acid. Then, 200 µL of this solution was transferred to a 96-well plate and read on the Synergy HT plate reader. Absorbance at 570 and 650 nm was recorded, and the difference (A570 – A650) was used as the formazan signal.
HTC drug treatment
All chemicals were from Sigma-Aldrich (St. Louis, MO) unless otherwise noted. CD, ROT, DNP, and Rho kinase inhibitor (S)-(+)-2-methyl-1-[(4-methyl-5-isoquinolinyl)sulfonyl]homopiperazine (Calbiochem, Gibbstown, NJ) were suspended in DMSO. Stock solutions of CD, ROT, and H-1152 at 1 mM were serially diluted to 100, 10, and 1 µM in DMEM. DNP was diluted to 1 M in DMSO and then to 100, 10, and 1 mM in DMEM. Following HTC preconditioning and background force (i.e., predrug) measurement, 50 µL of the drug solutions was added to each well. The final DMSO concentrations for all drugs were at 1%, 0.1%, and 0.01% (respective to the drug concentrations), which did not exhibit significant toxicity effects on the HTCs (Suppl. Fig. 1). Then, 50 µL of DMEM was added to the control wells. Upon drug addition, medium in the wells was mixed by pipetting 4 times.
HTC fixing and F-actin quantification
Drug-treated HTCs were incubated in 0.5 mL of 4% paraformaldehyde (Sigma) solution (in PBS) for 30 min. Fixed HTCs were rinsed twice and then permeabilized by incubation in 0.5 mL of 0.1% Triton X-100 (Fisher Scientific) solution (in PBS) for 30 min. Permeabilized HTCs were rinsed twice with 0.5 mL Tris-buffered saline Tween-20 (TBST) and stained for 45 min with 0.5 mL of 1:200 diluted Alexa 568–conjugated phalloidin (A12380, Invitrogen) in TBST. HTCs were then rinsed thrice with TBST. Alexa-phalloidin fluorescence intensities were read on a Synergy HT plate reader using the TRITC filter set.
REF-52 cell treatment and fixing
REF-52 cells were plated on 35-mm culture dishes (50,000 cells) in 10% FBS DMEM (2 mL). Cells were incubated overnight and then treated with compounds. Concentrated CD (20 µM), H-1152 (10 µM), DNP (30 mM), and ROT (100 µM) were dissolved in DMEM and then diluted 10× into each plate of cells (220 µL per 2 mL medium). Cells were treated for 24 h, rinsed once with 2 mL of phosphate-buffered saline (PBS), and then fixed in 1 mL of 4% paraformaldehyde (Sigma) solution (in PBS) for 30 min. Fixed cells were rinsed twice and then stored in 2 mL of PBS.
REF-52 cell labeling and imaging
Fixed cells were permeabilized by incubation in 1 mL of 0.1% Triton X-100 (Fisher Scientific) in PBS for 15 min. Permeabilized cells were rinsed twice with 1 mL TBST buffer and then blocked with 1 mL of 5% goat serum in TBST for 1 h. Cells were stained with 1 mL of TBST containing 1:200 diluted Alexa 568–conjugated phalloidin (A12380, Invitrogen), 2% goat serum, and 400 nM of DAPI. Cells were stained for 30 min and then rinsed twice with TBST. Labeled cells were mounted with Vectashield (Vector Laboratories, Burlingame, CA), covered with a cover glass, and then sealed with nail polish. The plate was then inverted onto a SP5 confocal microscope (Leica Microsystems, Bannockburn, IL) and imaged with 63× water immersion objective. Alexa 568 was excited using the 543 laser line, and DAPI was excited using a MaiTai multiphoton laser.
Statistical analysis
Student’s t-test, 2-tailed, was used to assess the significance (p ≤ 0.05) of CD-treated versus control HTCs (
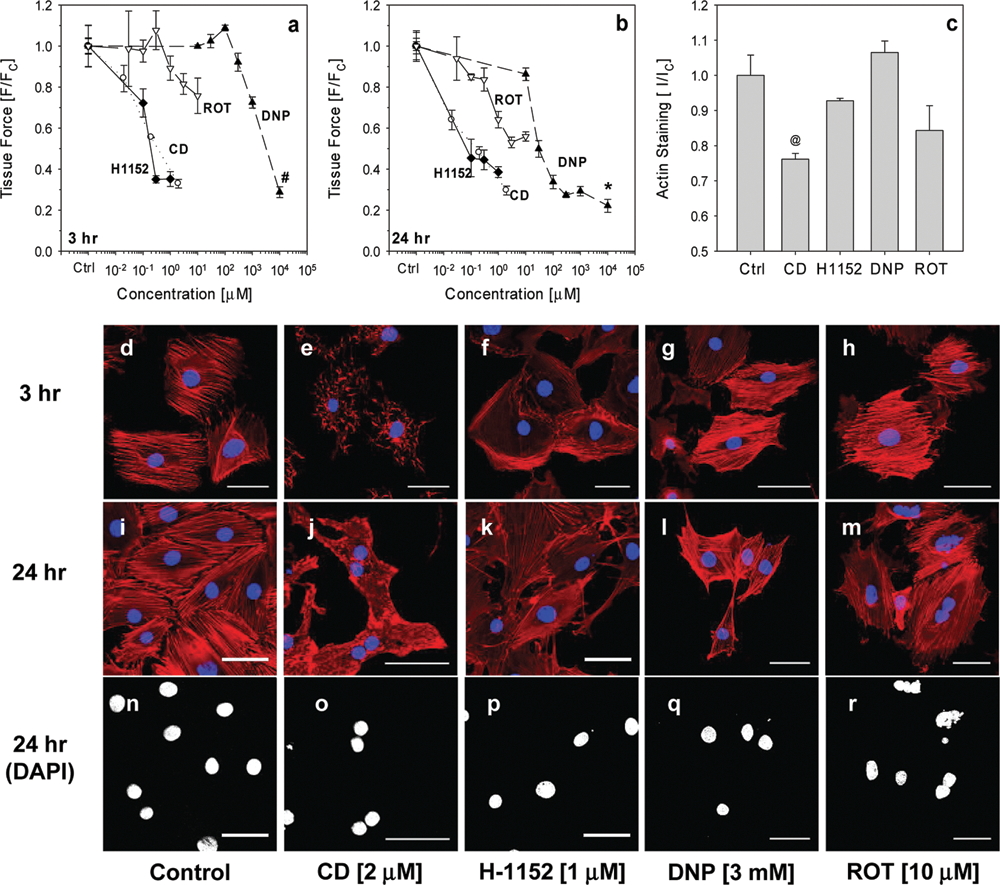
Drug induced dose-dependent reduction in hydrogel tissue construct (HTC) contractile force. Rho kinase inhibitor H-1152, cytochalasin D (CD), rotenone (ROT), and 2,4-dinitrophenol (DNP) dose-dependently reduced HTC contractile force. Forces were measured at (
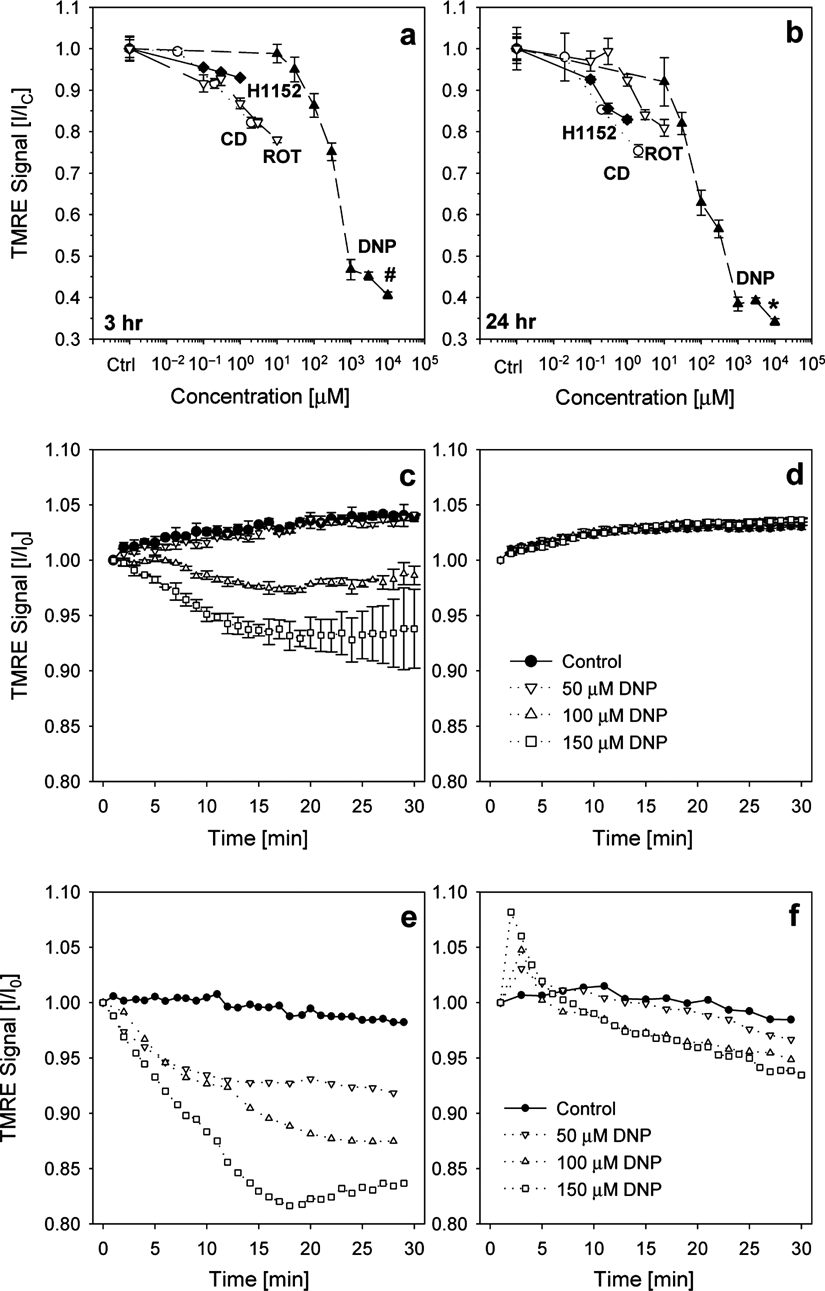
Live fluorescence signal detection in hydrogel tissue construct (HTCs). (
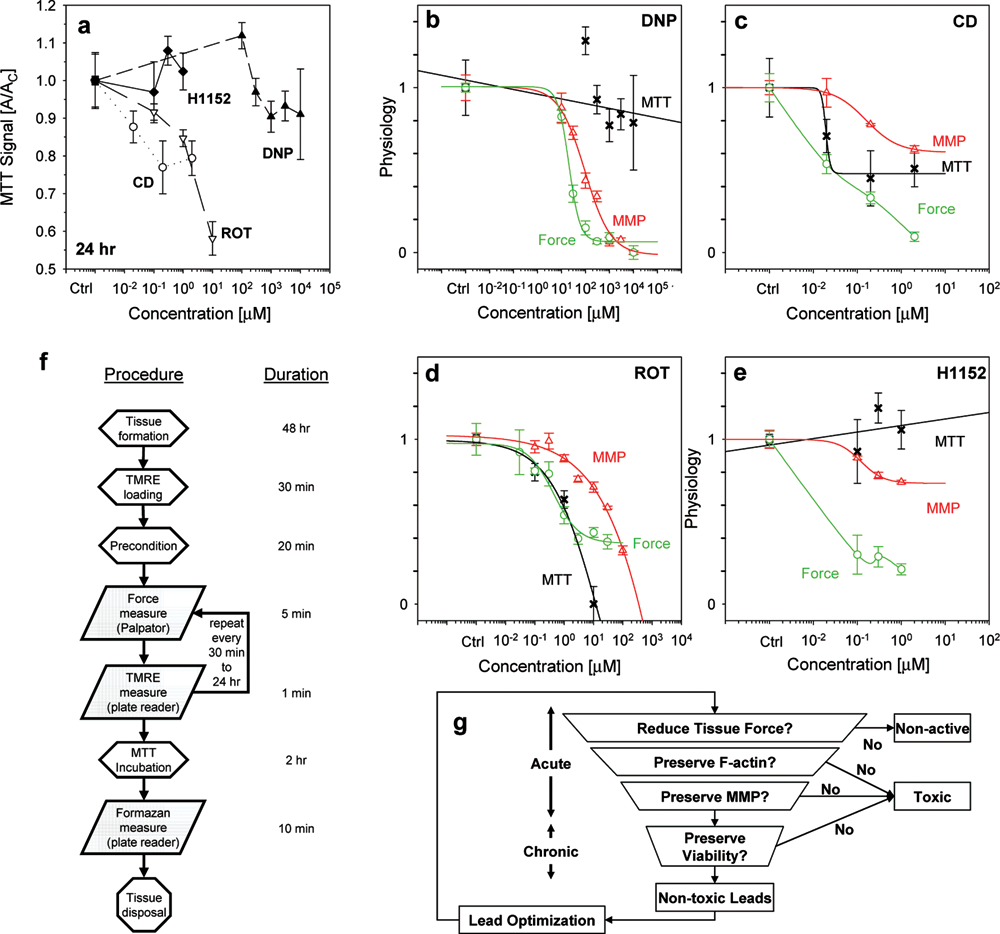
Hydrogel tissue construct (HTC) contractile force and physiological phenotype-based screening of compounds. (
Results and Discussion
Testing compounds for reduced HTC contractile force
We treated HTCs with CD, H-1152, DNP, and ROT for 3 and 24 h and profiled their effects on HTC mechanics. Three-hour treatment with H-1152 and CD dose-dependently reduced HTC contractile force with EC50 ≈0.1 µM (
Twenty-four-hour treatments enhanced the effects observed at 3 h (
Filamentous actin and HTC mechanics
Tissue force is maintained through the integrity of the cytoskeleton, especially through interactions between filamentous actin (F-actin) and myosin.
10
To quantify the amount of F-actin, compound-treated HTCs were stained with Alexa-phalloidin and measured on a plate reader. F-actin content in HTCs treated with CD (2 µM) was significantly reduced (
To confirm the loss in F-actin, compound-treated monolayer cells were stained with Alexa-phalloidin and examined with a confocal microscope. Extensive loss in F-actin was observed by 3 h post CD treatment (
HTP assessment of mitochondrial membrane potential
To further identify the mechanisms by which HTC contractility can be mediated, we assessed changes in cellular MMP. A plate reader was used to measure the intensity of the fluorescent dye TMRE, which accumulates in the mitochondria depending on MMP. Three hours of DNP treatment uncoupled MMP and dose-dependently reduced TMRE signals with an EC50 of ~340 µM (
Results from these experiments also demonstrated superior sensitivity in detecting TMRE/MMP in the HTCs as compared to cell monolayers grown on 2D substrata. DNP-induced concentration-dependent reduction in TMRE signal was readily detectable in HTCs using a plate reader (
Quantification of HTC viability
The compound-induced reduction in HTC contractile force could have been due to toxicity. To investigate the compounds’ toxic effects, we performed HTC viability assays using MTT. Intracellular succinate dehydrogenase converts the yellow MTT to a purple-colored formazan. This change was quantified by differential absorbance spectroscopy (A570-A650) using a plate reader. Three-hour treatment with any compound did not result in a significant loss of viability (results not shown). In 24 h, CD and ROT treatments dose-dependently reduced HTC viability (
HTC contractile force and physiological phenotype-based screening of compounds
To simplify compound screening decisions, experimental results were summarized as phenotypic profiles that reflect the impact of each compound on HTC physiology (
Classify chemical compounds through physiological parameters obtained using HTC-based assays
As demonstrated, in addition to the HTC contractile force, MMP and viability were incorporated into the assay workflow to concurrently monitor multiple physiological parameters. Additional assays, including intracellular calcium levels, oxygen consumption, and free radical production, can be inserted at the end point of the workflow (
A decision tree illustrates how HTC-based assays can be applied to identify drug candidates that exhibit similar characteristics to the Rho kinase inhibitor (H-1152) to progressively and systematically reduce the number of candidate compounds (
There are currently no other means to easily simultaneously assess the mechanics and physiology of HTCs and not in high-throughput format. One potential alternative is the micro-post-based technique proposed by Vandenburgh et al. 4 that quantifies tissue contractile force by the extent of micro-post deflection. However, this approach is not as flexible in that the sensitivity range of force measurement is limited to ~1.2 mN, which is determined by the stiffness, width, and the maximum amount of micro-post deflection. The sensitivity range of the transducer used in our technique is approximately 4 times larger, from 0 to 49 mN. Furthermore, only our technology offers researchers the ability to stretch the tissues and investigate strain-induced changes in cell physiology, including morphology, growth, differentiation, and responses. In particular, the ability to reconstitute and measure the Frank-Starling relationship in engineered myocardium 9 is of great importance to cardiovascular research.
Summary
In this report, we demonstrated how HTCs can be used to phenotypically screen for compounds that reduce tissue contractile force yet have minimal effects on the actin cytoskeleton, mitochondrial membrane potential, and cell viability. The HTCs provided cells a more in vivo–like 3D microenvironment to recapitulate the morphology and physiology observed in native tissues. In addition, optical assays using HTCs demonstrated superior sensitivity for detecting fluorescent indicators since emission signals were integrated from multiple cell layers. Most Z′ factors of the HTC contractility, MMP, and MTT assays satisfied NIH’s criteria (≥0.4) for use in HTP screening applications. We envision that the HTC-based assay system can serve to identify drug candidates that mediate tissue mechanics and facilitate the identification of their efficacy, toxicity, and mechanism of action in the early stages of drug discovery.
Footnotes
Acknowledgements
We thank Matthew Staniszewski for his technical assistance and Dr. Amy (Cayemberg) McQuade for her discussions and editing. This work was partly funded by NIH GM069072 and GM087784.
Dr. V. Lam and M. Staniszewski are employees of InvivoSciences, LLC. Dr. T. Wakatsuki is coinventor of the Palpator™ and cofounder of InvivoSciences LLC.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
