Abstract
Introduction
Accidental hypothermia increases mortality in patients with traumatic injury, making hypothermia management essential in prehospital trauma care. Chemical heat blankets are commonly used for this purpose. These blankets require time to get warm, with agitation often used to accelerate the exothermic reaction. Many search and rescue teams use chemical heat blankets, but optimal activation of the blankets remains uncertain. This study investigated how varying oxygen exposure durations affect the thermal performance of these blankets to guide their optimal use in search and rescue scenarios.
Methods
In room-temperature conditions, a full-body manikin was wrapped in a standardized hypothermia model, including a chemical heat blanket, and evaluated under 3 conditions: 1) no shaking, 2) shaking for 2 min, and 3) shaking for 15 min before wrapping. Thermometers were placed inside the heating panel pockets and secured in place with tape to ensure consistent measurements. Temperature data were collected over 6 h, and each scenario was performed 4 times.
Results
Blankets shaken for 2 min achieved higher initial temperatures than those not shaken, with a significant difference sustained for 88 min. Shaking for 15 min resulted in higher initial temperatures than 2-min shaking, with no long-term difference. All scenarios converged to the same temperature (36°C) after 6 h.
Conclusion
Shaking the blanket for 2 min significantly improved early heating performance, whereas longer shaking offered minimal benefit. These findings suggest that short preparation time may be adequate in optimizing thermal delivery and reducing the time to treatment for patients with prehospital accidental hypothermia.
Introduction
Accidental hypothermia is defined as an unintentional drop in core temperature below 35°C. 1 It is categorized into mild (35–32°C), moderate (32–28°C), and severe (<28°C) hypothermia. 2 Even at an ambient temperature of 10°C, individuals may experience numbness, shivering, frostbite, and hypothermia, particularly without adequate protective clothing. 3 Mild hypothermia impairs blood coagulation, and moderate hypothermia can cause severe physiologic effects, including arrhythmias, impaired cognition, respiratory depression, and cardiac arrest. Even mild hypothermia is associated with decreased survival in several patient groups, particularly patients with traumatic injury. 2
In prehospital settings, hypothermia poses a risk for all patients regardless of the underlying illness or injury. Hypothermia is part of the triad of death in trauma alongside coagulopathy and acidosis, which exacerbate one another and increase mortality risk. 2 In trauma cases, reduced core temperature is an independent risk factor for mortality. 4 Therefore, detecting hypothermia early and preventing further cooling by minimizing heat loss, improving cardiovascular stability, and maintaining fluid volume are crucial. 2
Critically ill or injured patients in mountain environments are particularly susceptible to hypothermia due to ambient environmental conditions such as wind, low temperatures, and wet conditions. They also may suffer from trauma that restricts movement, decreasing endogenous heat production and accelerating cooling. Therefore, insulating these patients from the environment quickly to prevent further heat loss is crucial. International hypothermia guidelines recommend a multilayer wrapping model: an inner vapor barrier to prevent evaporative heat loss, an insulating layer to minimize conductive and radiative heat loss, and an outer waterproof and windproof layer to prevent convective heat loss. Additionally, wet clothing should be removed before wrapping, and patients should receive active external warming.5-7
Various active external warming methods are available, including electrically heated garments and blankets and chemical heat blankets. Chemical heat blankets usually consist of a nylon blanket with multiple heating elements containing an iron-based powder that generates heat through oxidation. These blankets are packaged in polybags that insulate against air and water vapor until use. When the polybag is opened, the iron powder reacts with oxygen, releasing heat. 3 Manufacturers recommend removing the blanket from the polybag 15 min before use to allow exposure to oxygen and give the blanket time to heat up and reach the appropriate temperature. 1
Chemical heat blankets are often standard equipment in search and rescue teams. When the victim's location is known, rescuers can open the polybag in advance to ensure that the blanket reaches optimal temperature on arrival. However, in large-scale search missions with multiple search and rescue teams and where the victim's exact location is unknown, the heating blanket will have to be opened and activated when the victim is located. In this case, rescuers must decide how much time they will allow for the chemical heating blanket to activate before use.
This study aimed to describe heat production of an oxygen-reactive chemical heat blanket in 3 different exposure times to room air.
Method
Setup and Equipment
A full-body Resusci Anne QCPR training manikin (Laerdal Medical, Stavanger, Norway) was used to simulate the volume and body surface area of an actual patient. Although evaporation and burns were not study objectives, a vapor barrier (ASAP Jona 200; ASAP Norway, Skien, Norway) was included as the innermost layer to enhance realism. A chemical heat blanket, Ready-Heat II 12-panel blanket (86×122 cm; TechTrade LLC, Orlando, FL), was placed on the anterior surface of the manikin, external to the vapor barrier. Over the heat blanket, a standard ambulance cotton blanket (310 g/m2) was added for insulation. The outermost layer, an insulated mountain shelter (Jerven Fjellduken Extreme Primaloft 170 g/m2; Jerven, Odda, Norway), covered the model as a protective and insulating shell. This wrapping method was standardized for all test scenarios. Experiments were conducted indoors at a room temperature of 18 to 19°C.
Measurements/Procedure
Temperature measurements were obtained by placing 2 digital thermometers connected to a single Corpuls3 multimonitor (Corpuls, GS Elektromedizinische Geräte G. Stemple, Kaufering, Germany) in separate heating panel pockets over the thorax, and the measurements were taken from the anterior pockets. The thermometers were attached with tape to the heating panel side facing the manikin. Data were collected every minute over a 6-h period starting from closure of the mountain shelter zipper.
Scenarios
The study included 3 scenarios designed to mimic realistic situations in search and rescue operations:
The chemical heat blanket was unpacked from the polybag and immediately placed in the wrapping model. This simulates a situation where a patient with hypothermia is rescued, the heat blanket is opened, and the patient is wrapped immediately. The chemical heat blanket was unpacked from the polybag, shaken for 2 min, and then placed in the wrap. This simulates a situation where a patient with hypothermia is rescued, the heat blanket is opened, and 1 team member shakes the blanket for 2 min while the others are performing trauma assessments and other interventions. The chemical heat blanket was unpacked from the polybag, shaken for 15 min, and then placed in the wrap. This simulates a situation where a patient with hypothermia is located with a 15-min response time, and the blanket is opened in the helicopter/car so that it can be applied immediately on arrival.
Statistical Analysis
Preliminary power analysis showed that assuming a minimal clinically relevant difference between the blankets of 1°C and a standard deviation of 0.3, a sample size of 3.64 blankets in each of the 3 scenarios would be required to achieve a power of 0.9 with a significance level of 0.05. We therefore decided to use 4 blankets in each scenario. Statistical analyses were performed using unpaired t tests for all time points because our main aim was to investigate the specific time points where the scenarios were no longer different rather than general differences between the blankets.
Results
The results demonstrated varying temperature developments across the 3 scenarios. The blankets in the 0-min scenario had the lowest starting temperature at 23°C (Table 1). The graph for this scenario shows a steady rise toward 36°C, although at a slower rate than the other scenarios. The curve demonstrates a continuous temperature rise throughout the 6-h period (Figure 1). The blankets in the 2-min scenario showed a steep temperature increase within the first 30 min, before it stabilized and remained steady around 36°C for the next 6 h. The blankets in the 15-min scenario, which started with the highest temperature, reached their peak of just under 38°C within the first 10 min. The temperature then dropped to ∼35°C during the first hour, before it gradually rose again to 36°C, where it remained stable. Although the 3 scenarios began at different temperatures, they all converged toward the same temperature after 6 h.
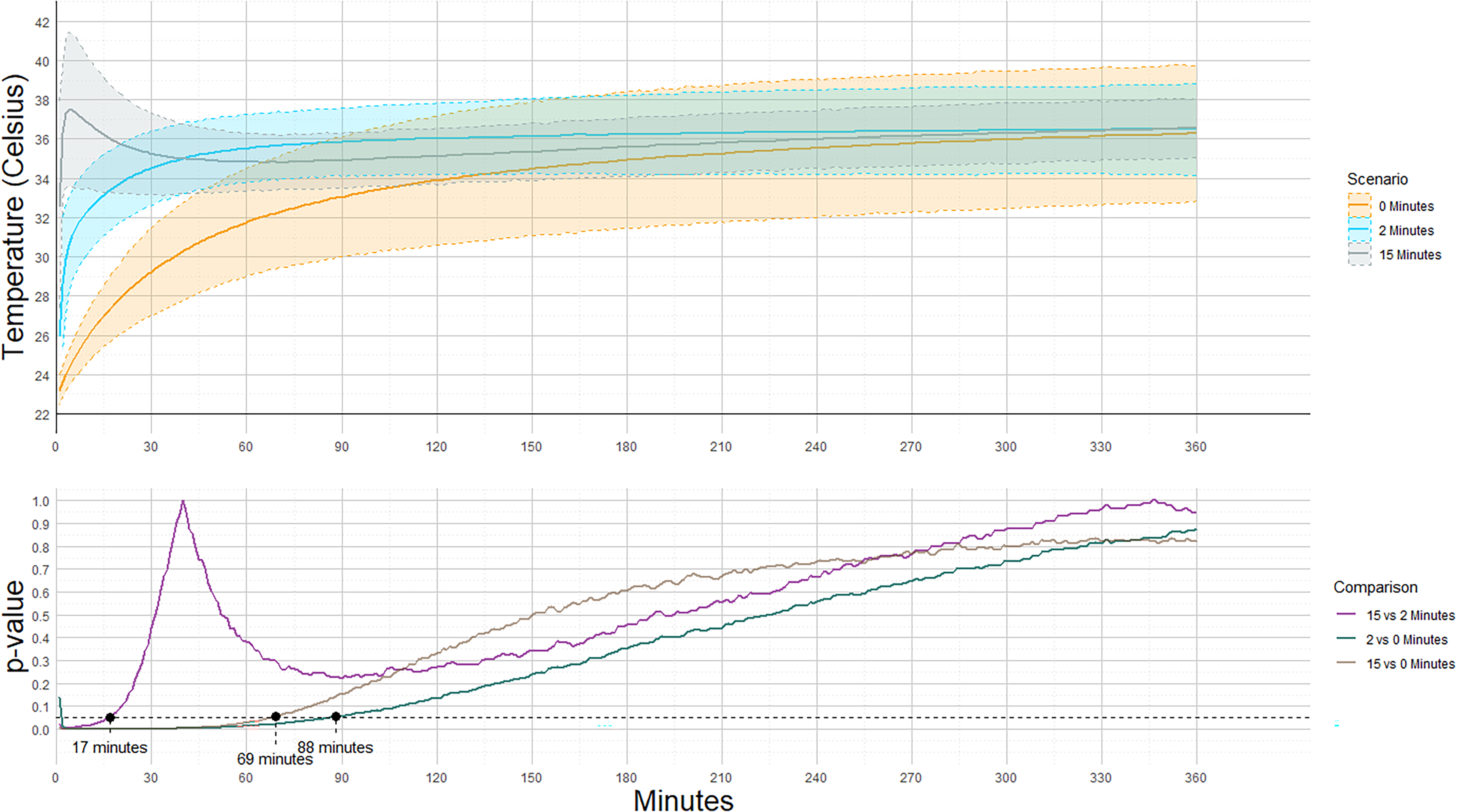
Temperature development of chemical heating blankets based on air exposure time.
Average temperature levels of the different scenarios during the experiments.

Note. This table shows the mean temperature and standard deviation of the chemical heat blankets at various time intervals across the 3 scenarios.
The figure also highlights the points at which the P values of the temperature differences exceeded 0.05. This occurred between the 2- and 15-min scenarios after 17 min, between the 15- and 0-min scenarios after 69 min, and finally, between the 0- and 2-min scenarios after 88 min.
Discussion
The results revealed significant differences between all scenarios, particularly between the 2- and 0-min scenarios. These differences are especially relevant because the difference between the 2 scenarios persist for a long time. There was a significant difference for 88 min, meaning that the blanket temperature was significantly lower in the 0-min scenario until this point. The 0-min scenario had a significantly lower starting temperature and did not fully catch up to the average temperature of the other scenarios within the 6-h study period. In contrast, the 2-min scenario began at a higher temperature and showed a moderate increase before stabilizing at ∼36°C for the following 6 h. This indicates that shaking the blanket for 2 min has a substantial effect compared with not shaking. Clinically, this difference could be important because achieving a higher and more stable temperature enhances the treatment of prehospital accidental hypothermia. Allocating 2 min to shake the blanket before wrapping therefore could have a significant impact on treatment with minimal time investment. In a prehospital search and rescue situation, 1 team member can activate the blanket by shaking it while others perform a primary survey.
The difference between the 2- and 15-min scenarios was less pronounced. There was no observable difference after 17 min, suggesting limited benefit of shaking the blanket for the additional 13 min. Choosing a 2-min shaking period instead of 15 min could save time and resources, especially given that preparing the blanket 15 min before patient contact is impractical. This shorter preparation time enables earlier active rewarming of patients with accidental hypothermia. Additionally, it reduces staff workload and optimizes resource allocation without severely compromising the effectiveness of the warming intervention.
The Ready-Heat II instructions advise that heating begins immediately on opening the polybag and exposing the blanket to air, reaching “operating temperature” after 15 min. Comparing the 0- and 15-min scenarios revealed substantial differences in both starting temperature and temperature stability. There was no observable difference after 69 min. Although shaking the blanket for 15 min may seem time consuming, it is clearly more effective than not shaking. Overall, not shaking the blanket appears to be the least effective method for using the heat blanket. This scenario shows a lower initial temperature, and the average temperature does not seem to reach the temperatures of the other scenarios, even after 6 h. The slower rate of temperature increase in this scenario may lead to less effective rewarming. Therefore, shaking the blanket for any duration, whether during a short or long preparation period, seems to be far more effective than not shaking.
Despite the differences in initial performance, all scenarios converged to the same temperature after 6 h and maintained stability without any reduction in temperature. This suggests that the blankets, regardless of initial exposure duration (15, 2, or 0 min), will eventually reach the same temperature over time. Additionally, the stability indicates that there is sufficient oxygen within a mountain rescue bag to maintain temperature throughout a 6-h period. This aligns with the findings of Greene et al, 1 who examined temperature changes in a chemical heat blanket under dry, damp, and wet conditions. In their study, the temperature of the chemical heat blanket also remained stable without any reduction for more than 7 h.
Some search and rescue teams may hesitate to open the blanket too early out of concern that they might use up the blanket's heat, fearing that it will not remain warm over an extended period, such as during prolonged rescue. Our findings demonstrated that the blanket would remain stable for more than 6 h without a reduction in temperature. In a situation where the injured patient's location is known, search and rescue teams can activate the thermal blankets well in advance of their arrival to ensure optimal blanket activation when active rewarming is initiated.
In the 15-min shaking scenario, where the blanket was exposed to air longer, a faster temperature increase was observed after 10 min, followed by a drop to the same level as the other scenarios. This indicates that prolonged shaking initially warms the blanket more effectively but that the temperature decreases when wrapped in a sealed rescue bag. Although the mountain rescue bag contains sufficient oxygen to maintain a stable temperature over 6 h, the available oxygen may be limited relative to oxygen in room air. After the drop, the temperature of this scenario converged to the same temperature as the other scenarios, confirming the similar conditions within the rescue bag. Additionally, the temperature drop may be because of the environment's thermal gradient between the heat blanket and the other components of the rescue bag. The components of the rescue wrap will absorb heat from the heating blanket. Consequently, when the heat blanket is packed in the rescue bag, it will experience both a reduction in oxygen availability and a drop in temperature, leading to the observed decrease in temperature.
Burn injury risk increases with rising temperatures and prolonged contact with the heat source. The manufacturer of the Ready-Heat II chemical heat blanket discourages direct contact with the skin due to burn injury risk. Our findings revealed a maximum temperature that never exceeded 38°C, which cannot cause burn injuries. However, other clinical factors, such as endogenous heat production, ambient temperature, and oxygen concentration, may result in higher temperatures than those observed in our study.
The inclusion of the vapor barrier is also pertinent because it effectively prevents evaporation, a critical factor in rapid cooling in clinical settings. Although evaporation does not affect a manikin, the vapor barrier ensured that the experimental setup closely mirrored real-world clinical practice. Additionally, the use of a standardized wrapping method with a cotton blanket and mountain quilt as insulating layers reflects current rescue practices, enhancing the study's clinical relevance.
Limitations
This study had several limitations that must be considered when interpreting the results. First, the experiments were conducted under controlled indoor conditions at a stable room temperature of 18 to 19°C. Although this approach ensured consistent measurements and allowed for standardized scenario comparisons, it did not account for the dynamic and often significantly colder environments encountered by rescue personnel. External factors such as lower temperatures, wind, altitude, and moisture could greatly impact both the patient's heat loss and the effectiveness of the chemical heat blanket.
Unfortunately, preliminary power calculations were performed for paired t tests, which have higher statistical power than unpaired t tests. This may have led to the study being underpowered, but it does not affect the results of the experiment.
Another limitation is the use of an “Anne” manikin, which does not produce endogenous heat. The metabolic heat produced by a real patient could influence the blanket's temperature, potentially resulting in higher temperatures than observed in this study. Conversely, oxygen availability for the chemical heat blanket may be reduced in a clinical setting because a real patient would consume oxygen. This could diminish the blanket's heat production and result in lower temperatures than recorded here. Temperatures inside the vapor barrier were not measured. Although clinically relevant, this was deemed out of scope for this experiment. It is also difficult to comment on the generalizability of our results regarding different heating blankets than the ones evaluated in our experiment. These limitations highlight the importance of caution when applying the study results to clinical practice because real-world factors may lead to variations in the blanket's performance.
Conclusion
This study examined the thermal performance of a chemical heat blanket under varying exposure times to air before wrapping, simulating rescue scenarios. Shaking the blanket for 2 min before wrapping significantly improved its initial temperature compared with no shaking, whereas shaking for 15 min offered minimal additional benefit. Regardless of the shaking duration, all scenarios converged toward the same temperature after 6 h, indicating that extended shaking initially accelerates heating without influencing long-term effects.
In rescue scenarios where the patient's location is known, search and rescue teams should activate the blanket before reaching the patient because it will remain warm for several hours. If the patient's location is unknown, allocating 2 min to shake the blanket before wrapping the patient seems to increase initial blanket performance significantly. However, shaking the blanket for 15 min yields little additional benefit and will delay active warming of the patient. These findings offer practical guidance for the efficient use of chemical heat blankets in prehospital care.
Footnotes
Acknowledgments
The authors acknowledge the effort of Jörg Assmus for his statistical assistance before, during, and after the experiments. The authors also acknowledge the contribution of Editage Author Services for improving the language quality and grammar of the manuscript.
Author Contribution(s)
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The expenses of this study were covered in their entirety by the Norwegian Air Ambulance Foundation.
Data Accessibility Statement
All data are available from the corresponding author on reasonable request.
