Abstract
Introduction
We studied field rewarming using a typical winter sleeping bag versus two heated hypothermia wrap systems in a semi-realistic lab simulation.
Methods
10 participants (8 M, 2 F) were cooled to 36.1°C core temperature through 10.5-11.5°C water immersion, then performed 60 min of passive rewarming in 0°C air. The rewarming methods tested were: 1) a −9°C rated mummy-style Sleeping Bag; 2) Doctor Down Rescue Wrap; and 3) Thermal Yielding Vascular Airway Capsule (TYVAC) system; the latter two methods included vapor barriers and two heating pads. Rectal and skin temperatures, along with metabolic heat production calculated via indirect calorimetry, were measured throughout rewarming.
Results
One male participant was removed from analysis due to lack of sufficient cooling. Rectal temperature decreased in the remaining participants by ∼1.1–1.2°C to 36.1°C during the initial immersion phase. Over the 60 min of rewarming, rectal temperature changes were Δ0.0 ± 0.6°C in a sleeping bag, Δ+0.2 ± 0.3°C in Doctor Down, and Δ+0.2 ± 0.3°C in TYVAC, with no significant differences across methods. Mean skin temperatures, metabolic heat production, and perceptual measures were also similar across methods with no method×time interactions.
Conclusions
After 60 min of passive rewarming in cold conditions, all three rewarming methods were able to stall continued core cooling to levels at or slightly above post-immersion temperatures. With no differences in any physiological measures, it appears that all three rewarming methods are equally viable options for wilderness responders, and the choice should come down to environmetal conditions, availability, convenience, and ergonomics rather than rewarming efficacy.
Introduction
Many recreational, occupational, and military activities involve exposure to cold air or water environments, and a combination of environmental conditions along with inadequate clothing and/or metabolic heat generation can potentially result in frostbite, cold stress (core body temperature Tcore > 35°C), or clinical hypothermia (Tcore < 35°C). 1 Even after rescue from immediate danger, Tcore can continue to drop due to cold air and wet clothing, and incapacitation from cold stress also presents a considerable challenge for rescue, medical care, and transport of the individual.2,3 Therefore, it is recommended to commence rewarming techniques in the field to promote the stabilization/increase in Tcore and prevent continued heat loss and further hypothermia.4,5 Furthermore, if possible, external warming (using heat packs or external heat sources) is recommended to promote rewarming and water vapor barriers to reduce heat loss. 6
The current gold standard approach for field rewarming from being cold-stressed or mild hypothermia is to dry the individual, and place them into a sleeping bag with a vapor barrier (if possible) and to remove wet clothes if shelter or warm environment is available. 6 These steps allow the individual to produce internal metabolic heat through shivering and minimize further cooling. 6 However, a limitation to the field use of sleeping bags is they are often not waterproof, typically lack a vapor barrier, and cannot be placed directly on snow or ice. Furthermore, no additional external heat source is provided. An alternative approach is to use heated-hypothermia wrap systems, which are commercially available systems that include an insulating enclosure, vapor barrier, and a heat source. Recently, the Doctor Down® system reduced shivering and increased skin temperature compared to other commercially available heated-hypothermia wrap systems in −22°C air. 5 However, this comparison is limited as participants were normothermic and not pre-cooled prior to cold exposure, impacting the ability to understand how the product actually rewarms individuals in its intended use.
The purpose of this study was to test the ability of two heated-hypothermia wrap systems options versus a user-assembled sleeping bag ensemble (−9°C rated mummy) in a field simulation consisting of cold-water immersion and core cooling, followed by 60 min of rewarming in a cold air environment. For the heated-hypothermia wrap systems, we chose the Doctor Down due to its performance in Dutta et al. 5 along with a prototype Thermal Yielding Vascular Airway Capsule (TYVAC®) unit that offered potential ergonomic and user improvements (waterproof and semi-buoyant, helicopter lift compatibility). Our null hypotheses were that all three rewarming systems would facilitate rewarming and have similar rates of rewarming.
Methods
Participants
The experimental protocol was cleared by the Research Ethics Board at Brock University (REB# 23-035) and conformed to the latest revision of the Declaration of Helsinki. Ten healthy volunteers (see Table 1 for characteristics) who were free from cardiovascular, respiratory, neurological, and cold disorders and were not pregnant were recruited from the university and community population. One participant was removed from the overall data set due to achieving minimal amounts of cooling over the 120 min cold-water immersion; their individual data is provided when possible. All participants were informed of the experimental protocol and associated risks before participating in this experiment and provided both verbal and written informed consent.
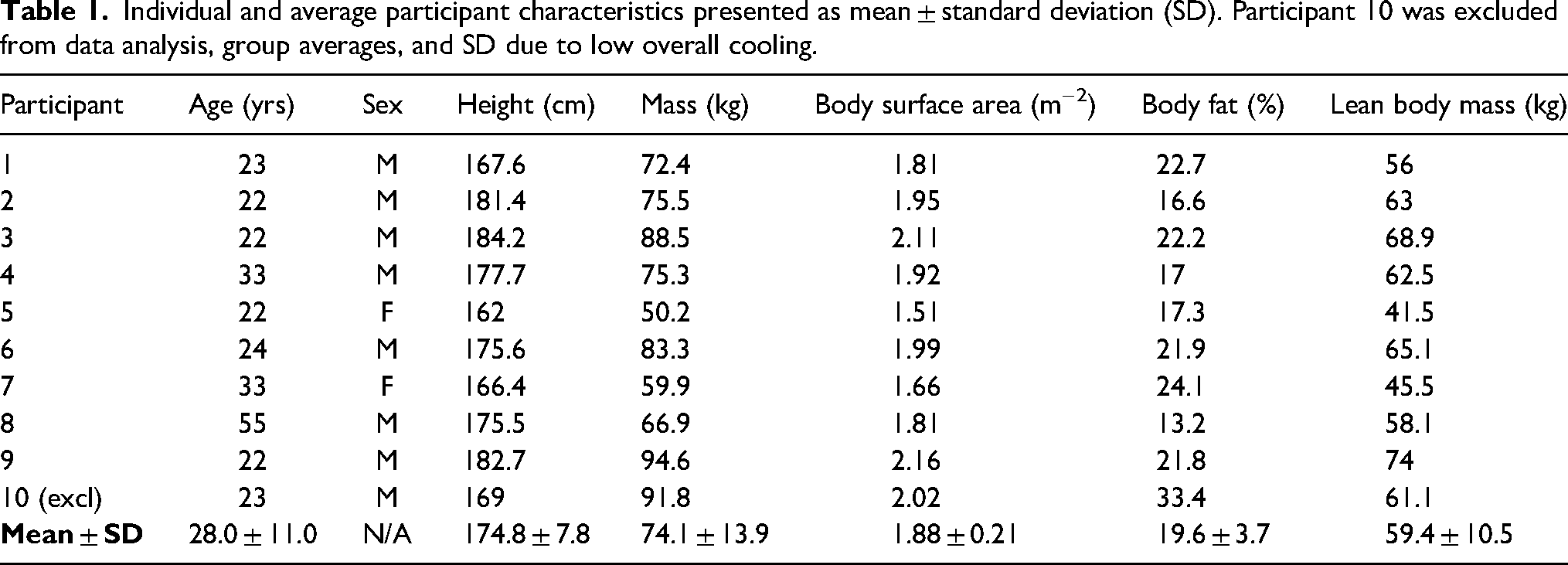
Individual and average participant characteristics presented as mean ± standard deviation (SD). Participant 10 was excluded from data analysis, group averages, and SD due to low overall cooling.
Experimental Design
The experiment was a randomized repeated measures design consisting of 3 experimental sessions differing by the rewarming system used. The experimental conditions were separated by 3–7 days to minimize the potential of cold acclimation and performed at the same time of day to control for circadian fluctuations in core temperature. Menstrual cycle was not controlled or monitored in the study. Participants were instructed to avoid vigorous exercise and alcohol consumption within 24 h and perform the same dietary practices prior to each trial.
Experimental Protocol
Each trial consisted of a 5 min baseline in an environmental chamber set to a thermoneutral (24°C, 50% relative humidity) air condition, a 10.5–11.5°C cold-water immersion to lower core temperature to 36.1°C, and a 60 min rewarming period in the environmental chamber with 0°C cold air. Upon arrival, participants voided their bladder and nude body mass (kg) was recorded. A urine sample was tested for urine specific gravity (PAL-10S, Atago, Japan) to determine hydration status. Participants were considered euhydrated if urine specific gravity was ≤ 1.020, or else the test was rescheduled (one trial was rescheduled due to inadequate hydration). For females, the urine sample was also used to perform a pregnancy test. All trials were identical except on the first trial, where height (cm) was measured using a stadiometer, and body mass (kg), % body fat, and lean body mass (kg) was measured using bioelectrical impedance analysis (InBody520, Biospace Co. Ltd., South Korea). Participants wore a t-shirt and their swim trunks (male) or two-piece swimsuits (female) for the baseline and removed the t-shirt for the cold immersion and rewarming phases.
Following baseline, participants were moved from the chamber and were placed in a harness connected to a ceiling lift and immersed to their collarbones, with their hands, toes, neck, and head out of the water. Water temperature was maintained between ∼10.5 and 11.5°C and controlled with a water chiller, while circulating jets maintained convective water flow. The immersion pool had ∼850 gallons of water as well as circulating jets to increase convective heat loss. Participants performed the immersion protocol until rectal temperature (Tre) was reduced to 36.1°C or 120 min. Participants were then removed from the pool and were dried off with towels, instrumented (3-lead electrocardiogram, metabolic mask), and placed in the rewarming condition (see details later) on a moveable table. Participants were then wheeled into the environmental chamber and performed the rewarming protocol.
The rewarming protocol consisted of 60 min of supine rest in each of the 3 rewarming systems in cold air (0°C, ∼60% relative humidity, ∼0.5 m·s−1 wind speed) to simulate an accidental water immersion when falling through ice. In this scenario, participants would be removed from the cold water and would still be in a cold air environment as emergency personnel provide first aid and evacuate the individual. Participants were fitted with a winter toque and continued to wear the same swim trunks/bathing suits from the water immersion, though we towel-dried the participants as best as possible prior to entering the rewarming bags. During the rewarming protocol, participants used the following rewarming systems.
Sleeping Bag
The sleeping bag was a −9°C rated mummy-style sleeping bag (comfort temperature rating −2°C) weighing 1.49 kg (Centaurus, Mountain Equipment Company, Canada). The sleeping bag has an outer shell lined with Pertex Quantum nylon with DWR treatment to repel water with a nylon lining. The sleeping bag is insulated with synthetic Hyperloft down. This sleeping bag has a hood that was tightened with a drawstring and shoulder flaps to cover the upper body and prevent heat loss from the torso. This rewarming system acted as the control condition.
Doctor Down
The Doctor Down Rescue Wrap (11-001, Hunter Black, Doctor Down Inc.) consists of an outer shell made of storm-tech (3-layer multipolymer) and is breathable, waterproof, and windproof. It includes a disposable vapor-proof liner that absorbs biological fluids. The bag uses Velcro to close the wrap system. Plastic clamps were used to pinch openings by the shoulders and neck to provide a tighter seal. The bag has two internal pockets (27.5 × 28 cm) inside the top layer at the chest and abdomen to hold 2 reusable gel chemical heat packs (sodium acetate salt solution; 20 × 20 cm, 0.6 kg; Doctor Down Reusable Heat Pack, Doctor Down Inc.). The gel packs were activated 10 min prior to use. We did not directly measure gel-pack temperature prior to or during cold exposure; however, previously, it has been demonstrated that this gel-pack temperature is 45°C when activated, and the temperature remains >40°C for at least 90 min after activation (tested in thermoneutral conditions, ∼23°C). 5
Thermal Yielding Vascular Airway Capsule (TYVAC)
The TYVAC System from SOLaaT Technologies (Port Colborne, Canada) is comprised of two main subcomponents: 1) a 6-layer, lightweight, multimaterial, full-body warming capsule consisting of abrasion-resistant, breathable, waterproof denier nylon sandwiching 5 layers of lightweight, heat retaining materials and 2) a multilayer groin vapor barrier using the same denier nylon designed to retain heat within the groin area. The system is fully encapsulated by zippers from head to toe, although a small square panel is exposed for the face. The capsule has two ∼61 × ∼46 cm pockets, one on each side, that hold two, one-time use, air-activated heat packs (Ready-Heat; 1 Panel, TechTrade, sized 15 × 20 cm, 0.2 kg) lasting up to 10 h each. We did not directly measure heat pack temperature, where previously it has been demonstrated that the gel-pack temperature is ∼35°C in the first 60 min of activation, peaking at ∼40°C within 120 min after activation (tested in thermoneutral conditions, ∼23°C). 5 Air-activated heat packs require oxygen to keep warm; however, we did not open the TYVAC or remove the heat packs over the course of the 60 min rewarming period potentially influencing the efficacy of the heating packs.
Insulation and Clo values were unavailable and were not measured in each of the 3 rewarming systems.
Physiological Measurements
Prior to baseline, participants self-instrumented with a flexible thermocouple (RET-1, Physitemp Instruments) 15 cm beyond the anal sphincter to measure rectal temperature (Tre) sampled at 4 Hz. Weighted mean skin temperature (
Metabolic heat production (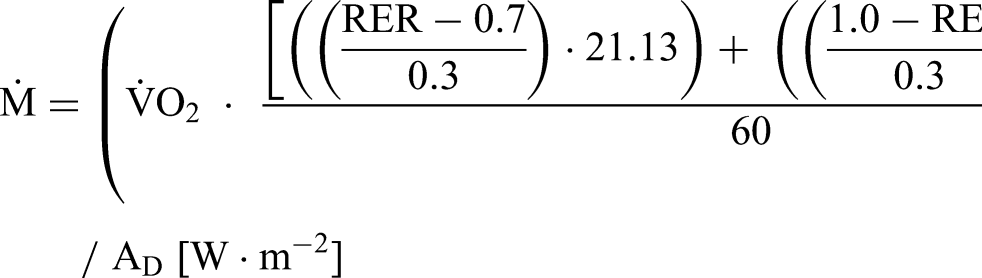
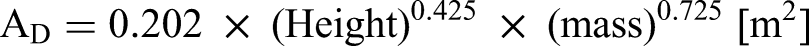
Indirect calorimetry assumes that metabolic heat production is due to oxidative, rather than non-oxidative (anaerobic), energy sources
8
; however, during passive cold exposure, RER has the potential to be ≥1 due to increased reliance on glycogen and carbohydrates to fuel shivering thermogenesis
9
and/or through increased lactate production and hyperventilation leading to increase carbon dioxide expired.
10
If RER ≥ 1, the following equation was used to account for the energy equivalent for carbohydrates only:
8
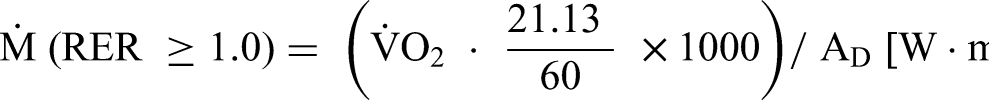
Perceptual Measures
At baseline, immersion end, 0 min. and every 15 min of the rewarming period, Likert-type scales were used to measure the following perceptual variables.
Whole-Body Cold Discomfort
Participants rated their discomfort related to cold strain on a 0–10 scale where 0 = no sensation of cold, 2 = slightly cold, 4 = fairly cold, 6 = moderately cold, 8 = very cold, and 10 = unbearable cold. 5
Overall Temperature
Participants rated their overall temperature on a 1–5 scale where 1 = very warm, 2 = warm, 3 = neutral, 4 = cold, and 5 = very cold. 5
Perceived Shivering Rating
Participants self-rated perceived shivering intensity on a 0–4 scale from 0 = no shivering, 1 = occasional mild tremors of the jaw and neck, 2 = intense tremors of the chest, 3 = intermittent vigorous generalized tremor, and 4 = continuous violent muscle activity.
Thermal Measures of Bag Surface
An estimate of inside surface bag temperature was performed using an iButton (DS1923-F5 Hydrochron, iButtonLink LLC) at the level of the chest on an area not covered by a heating pack. This was done in order to understand how the surface inside the rewarming systems changes over the course of 60 min while individuals are rewarming. Due to the mummy-style design and fit of the sleeping bag, the surface temperature sensor was in direct or partial contact with the participants’ skin, thus confounding the surface temperature measurement in this rewarming method. Therefore, inside surface bag temperature was recorded and compared only in Doctor Down and TYVAC, as there was no contact with participants’ skin.
Statistical Analysis
All statistical analyses were performed using GraphPad Prism (V.8.3.0, GraphPad Software). All physiological data is presented as mean ± SD with statistical significance set a P ≤ 0.05. Data were analyzed using either a 3-system (sleeping bag vs. Doctor Down vs. TYVAC) by 6-time (baseline vs. 0 min vs. 15 min vs. 30 min vs. 45 min vs. 60 min) repeated-measures ANOVA or an equivalent 3 × 6 linear mixed-model repeated measures ANOVA to account for any missing data. Additionally, comparisons between conditions were 1 × 3 system repeated measures ANOVA. Data were assessed for outliers and normality using visual inspection of Q-Q plots. Sphericity was not assumed, and the Greenhouse-Geisser was used in statistical analysis. When significant, a Bonferroni post-hoc analysis corrected for multiple comparisons was used to test specific main effects between systems and time.
Perceptual data were analyzed using 3 system × 7 time (baseline, immersion end, 0 min, 15 min, 30 min, 45 min, 60 min) repeated measures ANOVAs. Due to no system or system × time main effects, post-hoc comparisons for the main effect for time were analyzed using a Bonferroni post-hoc analysis corrected for multiple comparisons. All perceptual analyses are expressed as median (quartile 1–quartile 3).
Results
Immersion Phase
One participant was removed from all analyses (N = 9) due to no significant cooling (∼Δ-0.4°C) during immersion in all trials. One participant reached the 120 min in all 3 trials (∼Δ-0.9°C); furthermore, included in analyses were 2 additional participants reaching the 120 min cooling limit in a sleeping bag, 1 additional participant in Doctor Down, and 3 additional participants in TYVAC. Immersion times were similar (P = 0.225) across sleeping bag (74.0 ± 32.0 min), Doctor Down (77.0 ± 34.0 min), and TYVAC (90 ± 34.0 min). Overall, rectal temperature (Figure 1A) decreased from a baseline of 37.4°C by Δ-1.2 ± 0.4 (sleeping bag), Δ-1.2 ± 0.3°C (Doctor Down), and Δ-1.1 ± 0.4°C (TYVAC). There was no difference in transition time from immersion end to rewarming start (P = 0.288) between sleeping bag (9.0 ± 3.0 min), Doctor Down (11.0 ± 2.0 min), or TYVAC (10.0 ± 4.0 min).
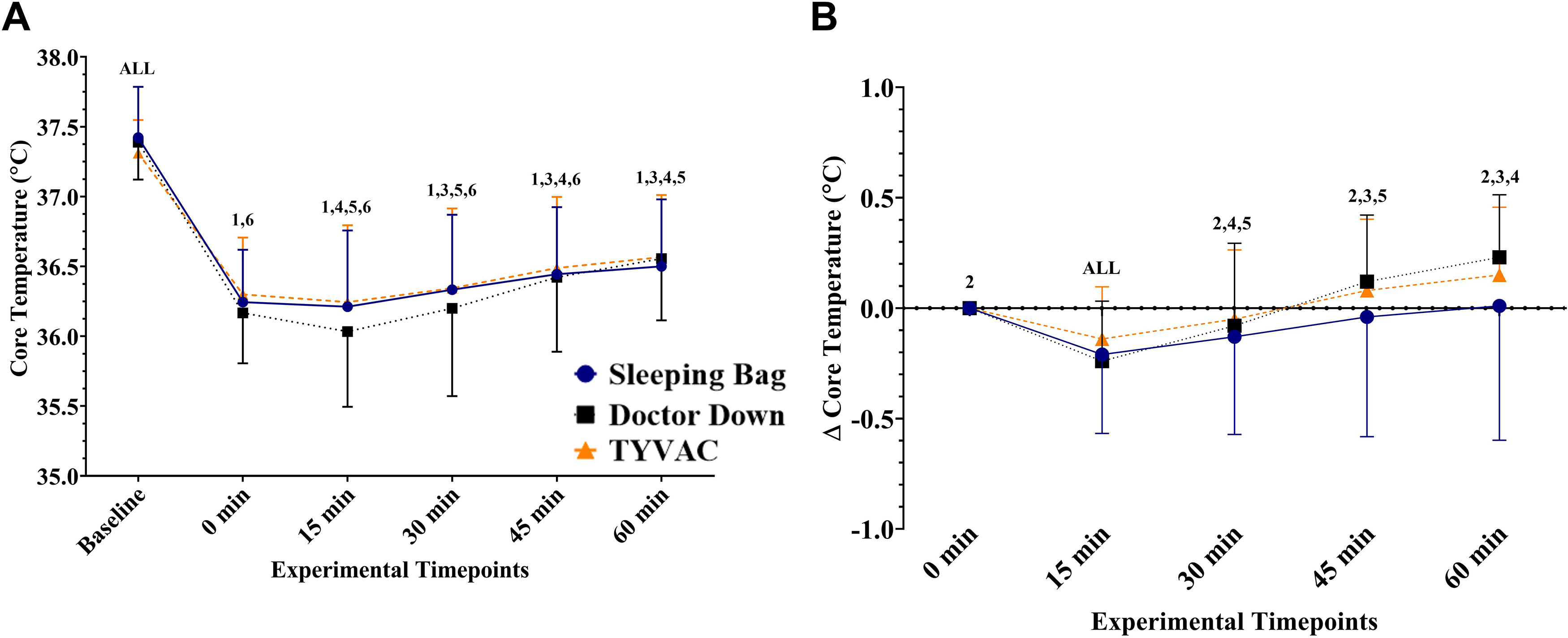
Absolute (A) and relative delta (B) core temperature responses presented as mean ± SD. For both, there was a significant time effect (both P ≤ 0.001) with no significant effect of rewarming system or interaction. Pairwise comparisons can be interpreted for Graph A as follows: All = different from all timepoints, 1 = different from baseline, 2 = different from 0 min, 3 = different from 15 min, 4 = different from 30 min, 5 = different from 45 min, 6 = different from 60 min. For Graph B, pairwise comparisons can be interpreted as follows: All = different from all timepoints, 1 = different from 0 min, 2 = different from 15 min, 3 = different from 30 min, 4 = different from 45 min, 5 = different from 60 min.
Rewarming Phase
There was no main effect of system or interaction (all P > 0.05) for any of the thermal measures. There was a significant time effect (both P < 0 .001) for absolute Tre (Figure 1A) and relative Tre (Figure 1B), where Tre increased over the course of the 60 min rewarming period. Overall, there was an average increase of Δ0.0 ± 0.6°C in sleeping a bag, Δ+0.2 ± 0.3°C in Doctor Down, and Δ+0.2 ± 0.3°C in TYVAC.
Whole-body
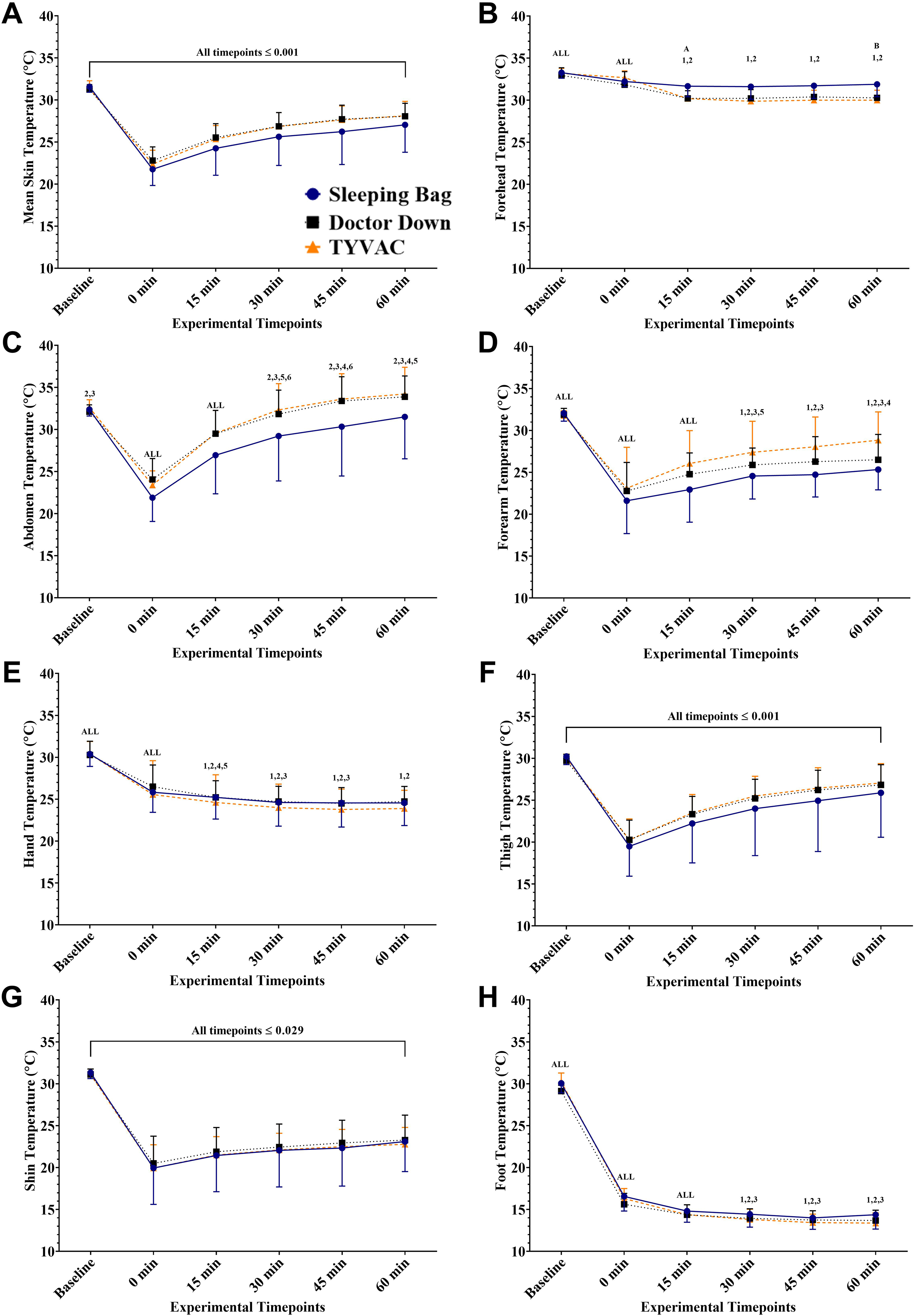
Overall mean skin temperature (A), and regional temperature for forehead (B), abdomen (C), forearm (D), hand (E), thigh (F), shin (G), and foot (H). For all, there was a significant time effect (both P ≤ 0.001) with no significant effect of the rewarming system or interaction, except for the forehead. Pairwise comparisons can be interpreted as follows: All = different from all timepoints, 1 = different from baseline, 2 = different from 0 min, 3 = different from 15 min, 4 = different from 30 min, 5 = different from 45 min, 6 = different from 60 min. For system differences, pairwise comparisons for the forehead temperature (B) can be interpreted as follows: A = difference between sleeping bag and Doctor Down, B = difference between sleeping bag and TYVAC, and C = difference between Doctor Down and TYVAC.
There was no difference between surface temperature (Figure 3, system effect was p = 0.390, system × time interaction was P = 0.618). There was a time effect (P ≤ 0.001) where surface temperature decreased progressively over the course of the 60 min.
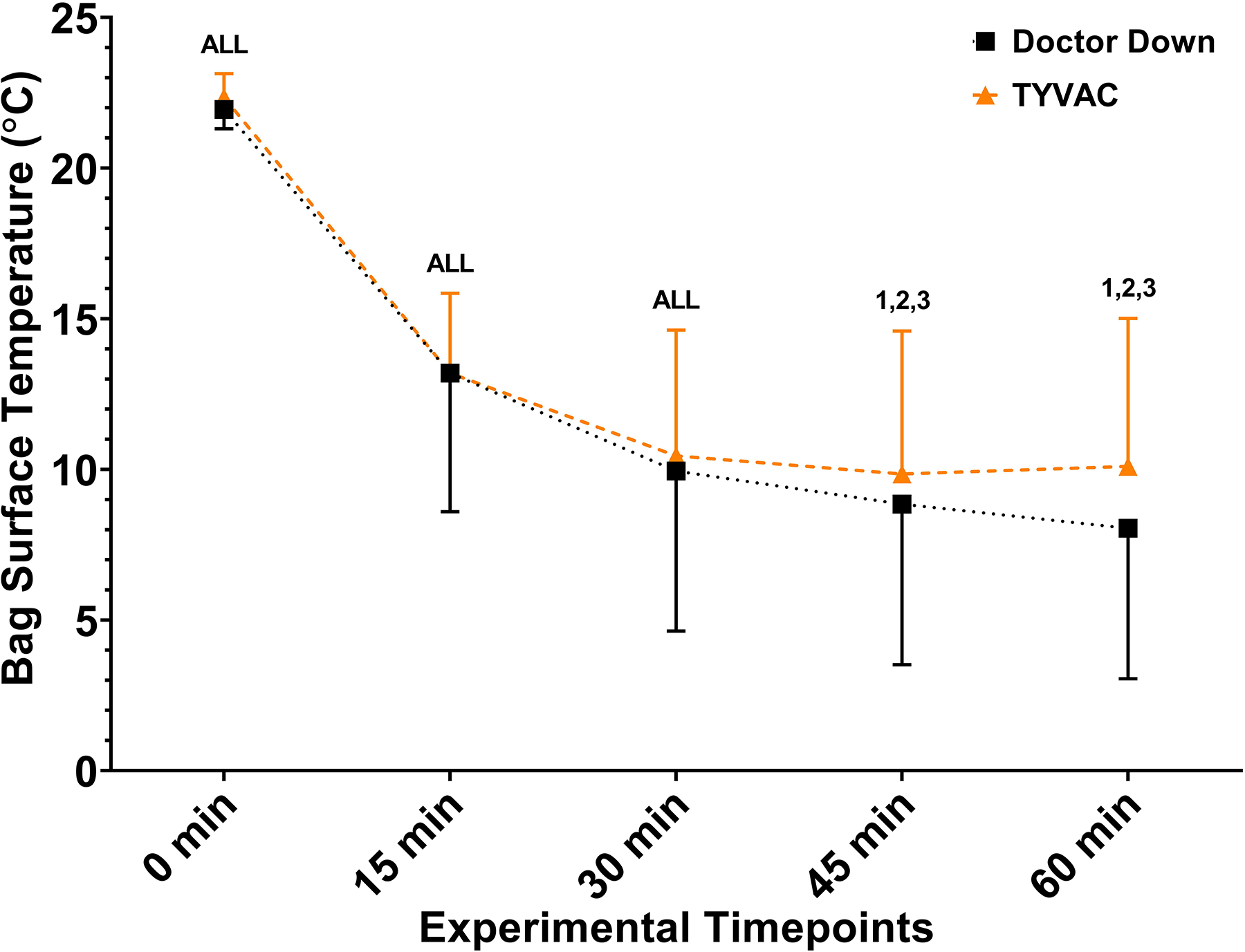
A comparison of inside surface bag temperature between Doctor Down and TYVAC. There was a significant time effect (P ≤ 0.001) with no differences between systems (P ≤ 0.390). Pairwise comparisons can be interpreted as follows: All = different from all timepoints, 1 = different from 0 min, 2 = different from 15 min, 3 = different from 30 min, 4 = different from 45 min, 5 = different from 60 min.
Due to technical errors, heart rate was unable to be recorded in one participant in Doctor Down, and metabolic data in one sleeping bag and one TYVAC trial. Heart rate (Figure 4A),
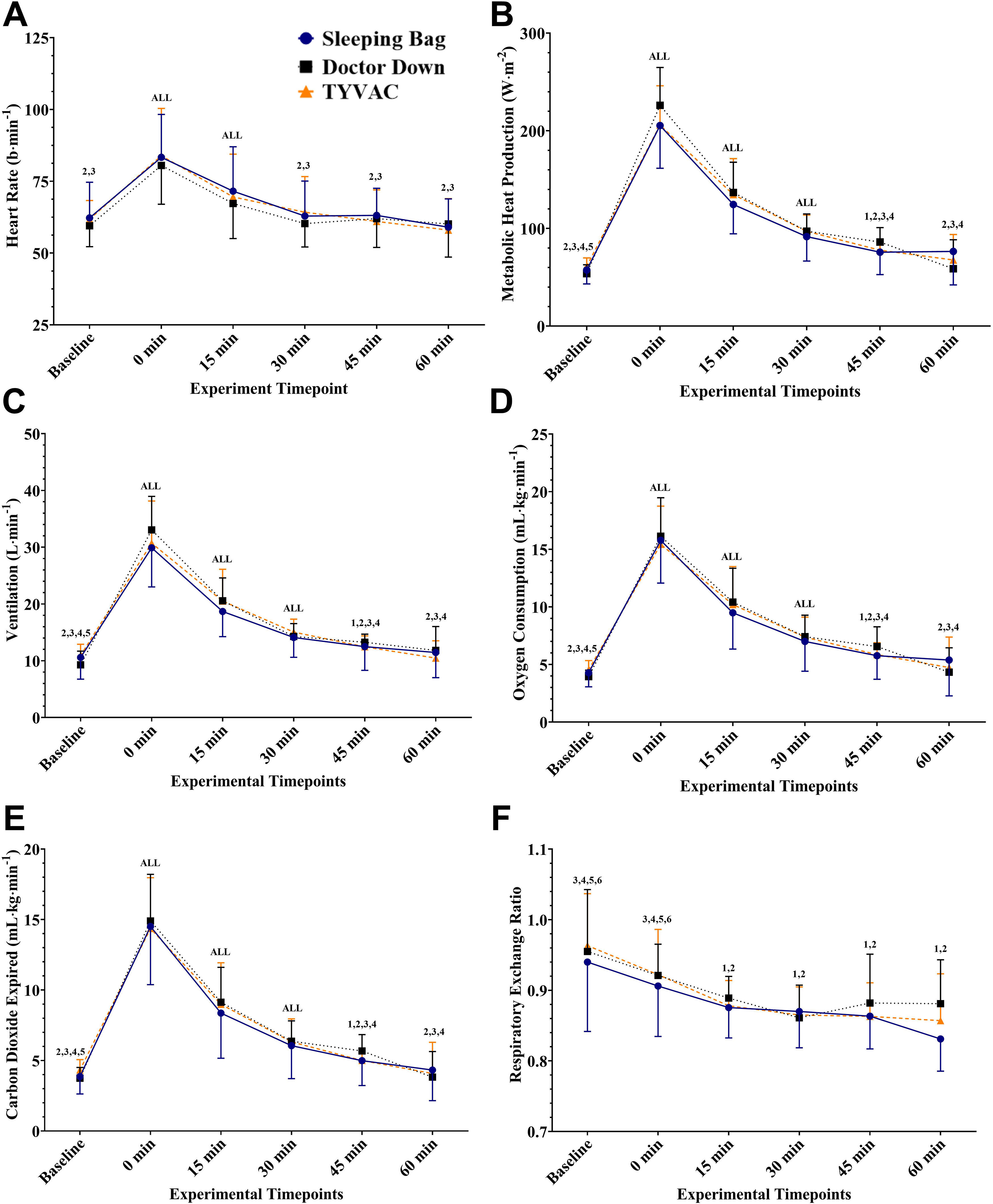
Heart rate (A) and metabolic responses to encapsulate the shivering and the participant's internal rewarming responses for heat rate (A), metabolic heat production (indexes shivering intensity) (B), ventilation (C), oxygen consumption (D), carbon dioxide expired (E), and RER (F). For all, there was a significant time effect (both P ≤ 0.001) with no significant effect of the rewarming system or interaction. Pairwise comparisons can be interpreted as follows: All = different from all timepoints, 1 = different from baseline, 2 = different from 0 min, 3 = different from 15 min, 4 = different from 30 min, 5 = different from 45 min, 6 = different from 60 min.
Perceptually, there were increases in whole-body cold discomfort, overall temperature, and perceived shivering intensity as each variable demonstrated a main effect for time (all P < 0.001) but no effect for system or system × time interaction (all P > 0.05) (Table 2). Overall, the pattern of responses was that participants felt coldest and perceived the most shivering at the end of the cold-water immersion and first 15 min of the trial. These values improved over the course of the rewarming period but were still elevated above baseline levels at the end of the 60 min rewarming period.
Perceptual responses for whole-body cold discomfort, overall temperature, and perceived shivering intensity over the course of the experimental protocol. Data is presented as median (Q1–Q3).
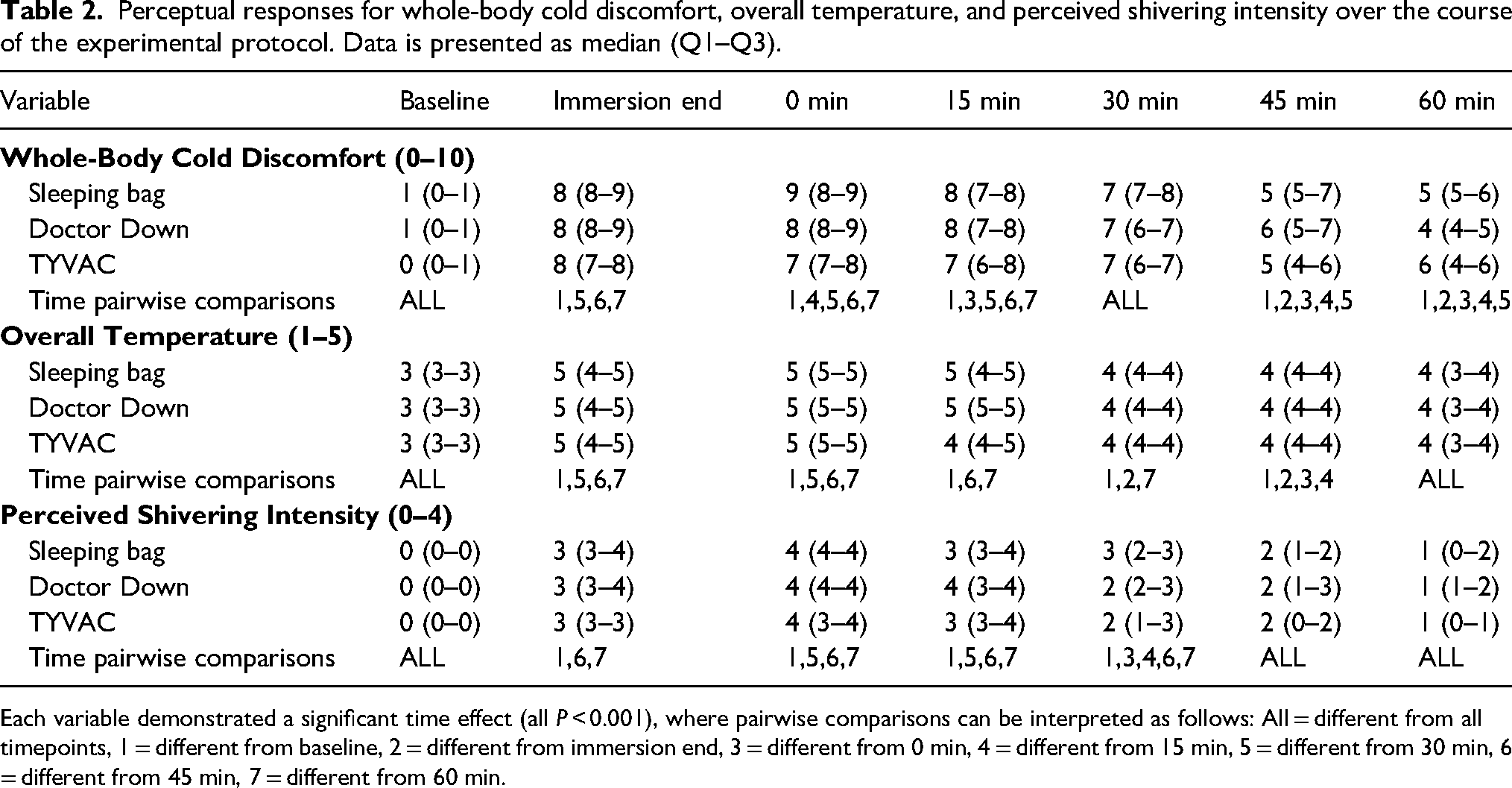
Each variable demonstrated a significant time effect (all P < 0.001), where pairwise comparisons can be interpreted as follows: All = different from all timepoints, 1 = different from baseline, 2 = different from immersion end, 3 = different from 0 min, 4 = different from 15 min, 5 = different from 30 min, 6 = different from 45 min, 7 = different from 60 min.
Discussion
Protection from further cooling is the priority for field management of cold stress and mild hypothermia, paired with supporting passive rewarming through shivering. 6 This study aimed to compare field rewarming using a typical winter sleeping bag versus two heated hypothermia wrap systems in a semi-realistic lab simulation consisting of 10.5–11.5°C cold water immersion and ∼1°C core temperature decrease, followed by 60 min of rewarming in a cold (0°C) air environment. As per our null hypothesis, all three systems rewarmed cold-stressed individuals, as rectal temperature was at or higher after 60 min of rewarming than following removal from cold water. Additionally, as per our null hypothesis, all three systems elicited similar rates of rewarming. Therefore, currently, the choice of field rewarming systems comes down to environmental conditions, availability, and ergonomics rather than any physiological advantages.
Our combination of cold-water immersion and cold air rewarming was sufficient to elicit intense to moderate shivering. Metabolic heat production was very high at the initiation of rewarming, at ∼200 W·m−2 or ∼3.5 times MET. Even from 30–60 min into rewarming, metabolic heat production remained ∼100 W·m−2 or ∼1.7 times MET. This is higher than the ∼1.2 MET during a 24-h sustained shivering study in 7.5°C air 11 and comparable to a study with passive cooling in 0°C air to a Tre decrease of 1.0°C. 12 While cold water immersion can elicit more intense shivering, with metabolic heat production exceeding 4 MET either with cooling to below clinical definitions of mild hypothermia of < 35.0°C 4 or even with only a mild 0.4°C core temperature decrease, 13 we are confident that our combination of low skin temperatures and high metabolic heat production during rewarming is representative of typical field rewarming situations and the results are transferable to individuals who are hypothermic where shivering is present. However, these results may not be transferable to patients with moderate or severe clinical hypothermia where shivering is suppressed or absent (Tcore < 32°C) 6 as participants are reliant on exogenous heat and the rewarming method without internal metabolic heat production. As ethical cutoffs for Tcore are typically ≥ 35°C, it is not feasible to perform the same study with participants starting rewarming at a lower Tcore. Future case reports or moderate/severe hypothermia simulation studies using pharmacological interventions that suppress shivering (e.g., meperidine) 14 are needed to determine the efficacy of heated-hypothermia wrap systems for moderate and severe hypothermia.
Aside from maintaining shivering, the other critical role of a field rewarming system is to provide protection from the elements along with insulation that prevents loss of endogenous heat production. The microclimate temperature within the Doctor Down and TYVAC was high upon the initiation of rewarming at ∼23°C (as systems were stored in room air) but dropped over the course of rewarming in 0°C air to 8–13°C from 15–60 min. We were not able to measure sleeping bag microclimate temperature due to direct sensor contact with the skin, but the similar core and skin temperature, along with heat production data, would suggest a similar microenvironment inside the sleeping bag. This intermediate temperature between skin and ambient temperature would suggest incomplete insulation from both wrap systems, such that there was a continued strong gradient for heat loss despite the TYVAC being fully encapsulating—including the head—with zippers and the Doctor Down being enclosed with Velcro. While the face was exposed in all conditions, this would be the case in all three ensembles in field use, though the TYVAC is intended to have a light mesh fabric screen to provide some protection from the elements (which was not used in the current study so that metabolic data could be collected). Overall, it appears important to reinforce education that insulation from the elements remains incomplete with sleeping bags or other passive rewarming wrap systems and that it is critical to maximize shelter and ambient temperature where feasible.
In our protocol, we tested a worst-case scenario for the control condition, whereby a −9°C rated mummy-style sleeping bag was used but without any vapor barrier or heating pads as added by Dutta et al. 5 Our primary addition was a toque, which was common across all three rewarming systems and would typically be available in the field. It is thus interesting that, despite the addition of a vapor barrier and two heating pads, neither the Doctor Down or TYVAC provided additional physiological rewarming benefits, and anecdotally participants only minimally sensed the presence of the heating pads. At the same time, the presence of heating pads did not alter either mean skin temperature or metabolic heat production, suggesting that they, at best, have a net neutral physiological impact. Based on our laboratory results, it does not appear essential to prioritize heating pads in field rewarming systems to enhance Tcore rewarming, which is in contrast to recommendations to apply external heat to the axillae, chest, and back for mild and moderate hypothermia as these areas have the highest potential for heat transfer and close proximity to vital organs. 6 The limited effectiveness may be due to our participants having the capacity to shiver. Previously, studies inducing mild hypothermia (esophageal temperature ∼35.0°C) following 8°C cold-water immersion with shivering suppressed (using meperidine) to simulate severe hypothermia have found torso rewarming using charcoal heaters,15,16 electrical heating pads, 17 chemical heat packs, 15 and hot water bags 15 to be more effective than no torso heating while rewarming in thermoneutral environments. However, in these scenarios, individuals are reliant on exogenous heat. Therefore, based on our results, when shivering is present, the types of heating packs used in the current study may not enhance Tre rewarming; however, they may provide perceptual benefits in thermal comfort and warming tissue near vital organs as opposed to enhanced Tcore rewarming. A limitation is we did not measure the heat pack temperature over the course of the rewarming period or following the experimental trials and cannot confirm if the heat packs retained their heat in the cold environment. The elemental conditions in the current study are more favorable than typical survival scenarios as there was no ground conduction with cold surfaces (i.e., snow, ice); no moisture/wetness on the bag's surface from snow, water, or ice; and no high wind speeds, where the use of vapor barriers and/or extra insulation is recommended to protect the individual from these conditions 6 and where heating packs may provide additional thermophysiological benefits.
Conclusions
In a semi-realistic lab simulation of a cold survival situation, this study found no difference in physiological rewarming between a standard winter sleeping bag and two commercial heated hypothermia wrap systems. We thus conclude that the choice of field rewarming systems should be based on availability, convenience, and environmental conditions, though the commercial systems could provide ergonomic advantages for first responders not tested in this study.
Footnotes
Acknowledgments
The authors express their gratitude to the participants for their efforts and enthusiasm throughout the study.
Author Contribution(s)
Data Availability Statement
The data is available upon reasonable request by contacting the corresponding author.
Declaration of Conflicting Interests
We declare no potential professional or commercial conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Saving One Life at a Time (SOLaaT), a company based in Port Colborne, Canada, provided in-kind contributions consisting of all three rewarming systems, the iButton skin temperature sensors, and participant honoraria. They did not have input in the study design, and none of the authors had any financial or personal conflicts with SOLaaT.
