Abstract
The Wilderness Medical Society (WMS) convened an expert panel to develop evidence-based guidelines for the management of pain in austere environments. Recommendations are graded based on the quality of supporting evidence as defined by criteria put forth by the American College of Chest Physicians. This is an update of the 2014 version of the “WMS Practice Guidelines for the Treatment of Acute Pain in Remote Environments” published in Wilderness & Environmental Medicine 2014; 25:41–49.
Keywords
Introduction
The treatment of pain is important in the management of illness and injury in any context. Beyond the ethical considerations of compassion and management of suffering, effective management of acute pain in austere settings can facilitate the evaluation, packaging, and transportation of patients whose management would otherwise be challenging or impossible without pain control.
Management of acute pain is a multifaceted challenge that can involve physical stabilization of an acute musculoskeletal injury or wound, medical management of peripherally and centrally mediated pain responses, and management of psychological distress and anxiety. Any treatment of pain carries with it risks and benefits that may be amplified when these treatments are used in an austere environment. Limited patient access, difficulty of reevaluation during transport, the need to protect the patient from environmental insults, lack of monitoring equipment, and limited means to respond to adverse effects of treatments can amplify risks. Consequently, treatment of pain in an austere environment requires a thoughtful and progressive approach that minimizes harm while managing the needs of the patient and treating team.
For the purposes of this guideline, austere environments are defined as wilderness, remote, or highly resource-limited settings where access to typical hospital or EMS resources is unavailable, and evacuation may be challenging or prolonged. We include wilderness and expedition settings and other travel to regions where parties must be self-sufficient in their medical care. We exclude from this guideline pain management in the military setting, where resources and availability of evacuation are generally greater and for which guidelines already exist. We also exclude management of acute pain in space travel, where physiology and resource limitations are truly unique. The term “austere” has been chosen to replace “remote” in this guideline as it better describes a setting of limited resources and environmental challenges. When terrain, weather, or altitude are challenges to care, resources do not need to be geographically remote to be functionally unavailable.
The legal specifics of the acquisition, storage, and administration of controlled substances are beyond the scope of this guideline. Variation between and even within nations and jurisdictions is too great to facilitate recommendations specific to all settings. Further, we do not limit the recommendations in this guideline to a particular class, certification, or training level of responder. Whether amateur recreational backcountry users or trained medical responders, users of these guidelines must employ judgment as to which treatment modalities are within the scope of their training and appropriate to their treatment setting and available resources. In each of the sections to follow, we present the best available evidence identified by the panel as to the safety, efficacy, and austere-specific considerations for available pain treatment modalities.
Methods
An expert panel was convened during the 2013 annual winter meeting of the Wilderness Medical Society (WMS) in Park City, UT, and an original guideline was published in Wilderness and Environmental Medicine in 2014. A subsequent expert panel was convened in 2019, and a partial unpublished revision of the guideline was completed, integrating updated literature sources. To complete this updated literature review, a panel was convened in July 2022, participation in which was based on the individual's relevant clinical, research, and/or field experience. The panel included representatives from emergency medicine, anesthesiology, surgery, and the field of prehospital emergency medical services (EMS).
A review of relevant research articles was completed by a keyword search of the MEDLINE and PubMed Central databases via the PubMed search interface in addition to queries of SCOPUS and Google Scholar using keywords targeted to each subject area, with filters applied to remove non-English language sources. Searches were completed during June–August 2022. Keywords searched were generated by section authors. Abstracts were screened by section authors for relevance to austere settings, fundamental relevance to the understanding and treatment of pain, or the pharmacology and pharmacodynamics of medications discussed. Additional sources were identified by reviewing citations from identified sources and review articles. Where evidence directly pertinent to austere settings was not available, the best available evidence was included from EMS, tactical medicine, disaster medicine, emergency medicine, and global health literature sources. Pain control modalities were included based on expert consensus, and any modalities suggested by any member of the panel were subjected to a literature search for relevant evidence and applications.
Articles were excluded for lack of relevance as determined by expert review. Additional relevant review articles obtained for background, as well as relevant guidelines on the management of acute pain and traumatic injuries, were also selected for inclusion.
Recommendations were developed by section authors and revised using a consensus approach. Where evidence relevant to austere environments was limited or of low quality, recommendations were made by the consensus of the group based on the clinical experience of panel members and relevant literature from hospital and prehospital medicine. Recommendations were graded according to the criteria developed by the American College of Chest Physicians (Supplemental Table). 1 Final recommendations included were approved unanimously by all members of the panel.
Terms used in the guideline to discuss pain, such as “pain,” “nociception,” and “sensitization,” are used in a manner consistent with the International Association for the Study of Pain terms and definitions. 2
General Approach to Pain Management in an Austere Environment
Musculoskeletal injuries, including strains, sprains, dislocations, and fractures, are the most common cause of pain treated in austere environments. Other circumstances that may require pain management include acute medical ailments and environmental injuries such as cold injuries, bites, stings, and burns.3,4,5
In all cases, the management of pain is a secondary consideration to the management of immediate threats to life, including compromise of airway, breathing, or circulation, and secondary to scene safety for both the patient and the treating party. After scene safety and a primary survey have been addressed, a secondary survey should be undertaken to evaluate the cause and severity of pain. 6
Traumatic tissue injury, heat, cold, and ischemia can cause pain by different mechanisms, and understanding the mechanism of injury or illness informs appropriate treatment. In an alert patient, a description of mechanism, location, quality, and severity of pain can guide treatment. In an uncooperative or unresponsive patient, evaluation of the patient setting and the completion of a thorough secondary survey can identify traumatic as well as environmental injuries on the basis of deformity and tissue appearance. 7 Pain control should not be withheld out of concern for masking injury or limiting examination quality, and myths to the contrary have been addressed by quality evidence and expert recommendations.8,9,10,11
If possible, prior to treatment of pain, providers should evaluate pain severity to be able to evaluate response to treatment. A numerical rating scale (eg, Numerical Rating Scale [NRS-11] or Visual Analog Scale [VAS]) or the patient's subjective description of their pain severity should be used to evaluate response to treatments. 12 However, protocols should not compel obligatory use of medications or other interventions in response to specific scores, as a rigid approach requiring treatment in response to a specific threshold has been shown to increase medication-related adverse events.13,14
Except for some psychosocial interventions, all pain treatment modalities discussed henceforth are accompanied by both benefits and risks to the patient. In general, centrally acting pharmacologic agents (eg, opioid medications, benzodiazepines) carry greater risk than peripherally acting agents (nonsteroidal medications, local anesthetics) due to their potential to affect hemodynamics and respiration. Parenteral routes of administration (intranasal [IN]/intramuscular [IM]/intravenous [IV]/intraosseous [IO]) carry more risk of adverse effects than enteral or topical routes on account of accelerated absorption and shorter time to peak serum concentration. Importantly, risk and benefit are not directly correlated in all cases, and some low-risk interventions like psychological support or injury immobilization can be substantially effective in the treatment of pain.
While previous WMS guidelines have recommended a stepwise and escalating approach to pain control, we update this approach to consider situations in which the treating clinician recognizes an inherently severe cause of pain and immediately initiates more aggressive treatment measures. 15 Accordingly, we recommend an approach to pain treatment that selects and simultaneously applies pain treatment modalities based on the clinician's experience, the cause of pain, and appraisal of reported and anticipated pain severity. When possible, we still recommend the use of lower-risk interventions before or in concert with higher-risk interventions. Figure 1 provides a conceptual schematic of how different modalities might be selected based on pain severity and available resources.
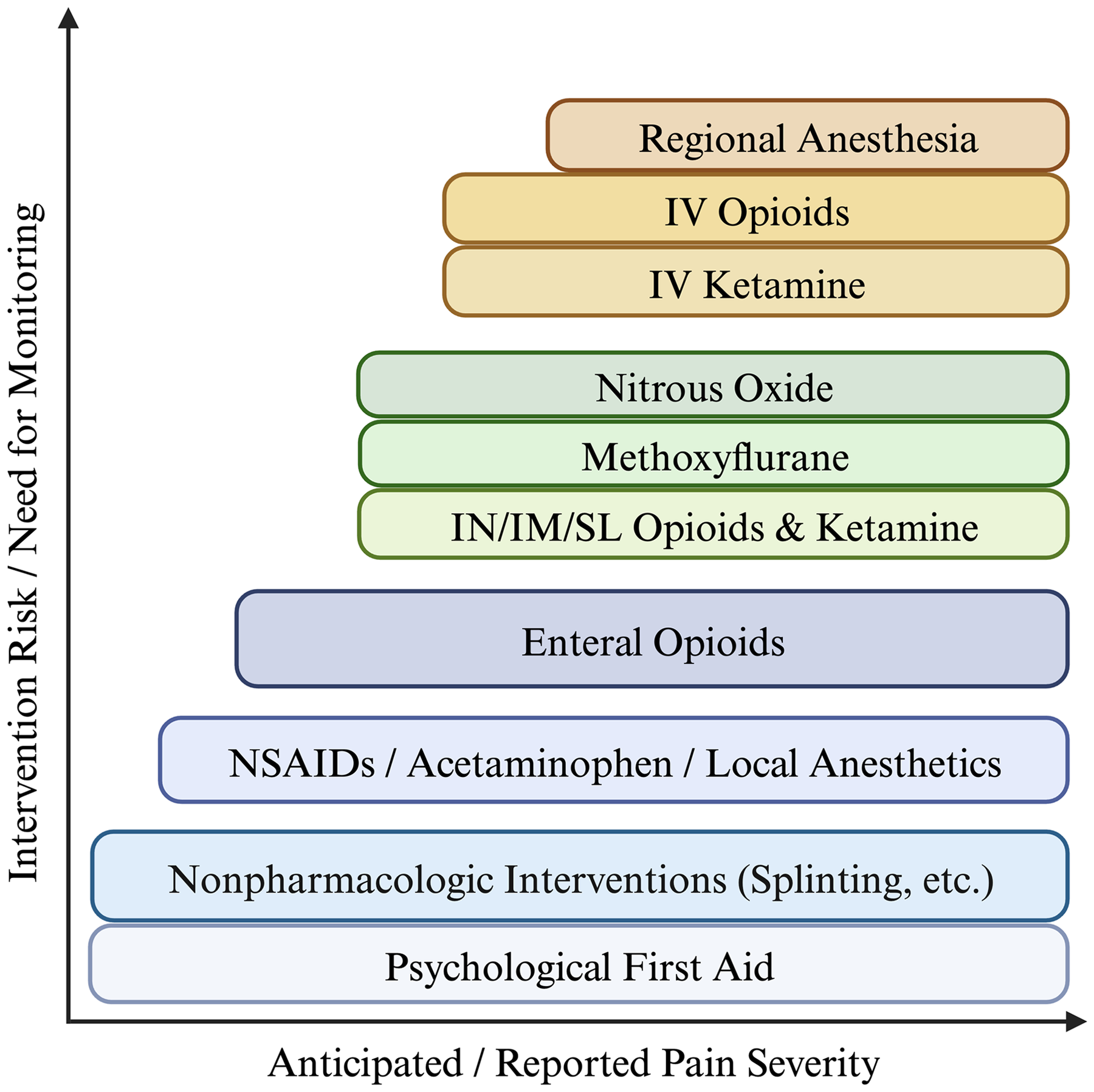
Conceptual schematic of escalating pain treatment stratified by risk posed to the patient and the need for monitoring when used. With greater anticipated or reported pain severity, a clinician may elect to use one or more modalities appropriate to that degree of severity. IV, intravenous; IM, intramuscular; IN, intranasal; SL, sublingual; NSAID, nonsteroidal anti-inflammatory drug.
After application of a given treatment, the patient's pain level should be reevaluated, and the treating provider should determine if pain control is adequate to meet the needs of the patient and the treating provider or team. Given that different modalities of pain control are appropriate for different mechanisms of injury and illness, there can be no clear prescriptive approach or linear road map for the treatment of pain. In some patients, anxiety and psychological distress may significantly amplify a response to a minor injury, while in others, an isolated orthopedic injury may be amenable to a specific intervention (eg, hematoma block), which is inappropriate in other cases. Experience and judgment, paired with frequent reevaluation of the patient and environment, are critical.
Medical responses in an austere environment are inherently limited, so one must evaluate and select pain treatments to minimize packed weight and bulk. An ideal medication for austere settings is compact; lightweight; durable against crush, abrasion, heat, and cold; nonsedating; has a large therapeutic index; works across multiple routes of administration; and has no potential for abuse or dependence.16,17,18 Since no such medication exists, providers must weigh the advantages and disadvantages of each medication carried considering the previous constraints.
A survey of providers at the WMS summer conference in 2016 demonstrated that among practicing medical provider respondents, 90% carried oral (PO) nonsteroidal anti-inflammatory drugs (NSAIDs), 81% carried acetaminophen (APAP), and 47% carried PO opioids when selecting medications for organized treks. 19 These medications, in addition to training in nonpharmacologic pain management, are likely to suffice in many settings, as minor orthopedic injuries and soft tissue wounds comprise the majority of cases. 5 In the management of severe acute pain, however, these medications may be inadequate.
In the following sections, we provide further discussion of pain control modalities. In each section, we discuss the risks and benefits of these interventions as well as the applications to which they are best suited. Additionally, we discuss treatment considerations such as the need to monitor for adverse effects. Some of these treatments are advanced and require experience that can only be developed in high-volume clinical environments such as hospitals and EMS services. These treatments provide opportunities for organized rescue teams and advanced medical providers to further refine their treatment of pain, but they are not necessary per se and, in some settings, may be inappropriate or excessive.
Recommendation: We recommend that providers should manage pain to a level that provides adequate relief, meets the needs of the treating team, and recognizes the limitations imposed on the team by the austere environment (strong recommendation, moderate-quality evidence).
Recommendation: We recommend that, when appropriate to the clinical setting, providers should evaluate patient response to low-risk pain treatments before progressing to higher-risk treatment modalities or routes of administration to minimize harm in the austere environment (strong recommendation, low-quality evidence).
Recommendation: We recommend that, when appropriate to the clinical setting, providers may initiate multiple simultaneous treatments to control the pain of greater reported or anticipated severity (strong recommendation, low-quality evidence).
Nonpharmacologic Pain Management
Psychosocial Interventions
The experience of pain is a product of nociceptive nerve signals from the peripheral nervous system that are interpreted and given emotional valence in the central nervous system. Practically, this means that the psychological state of the patient can affect their experience of pain.20,21 Laboratory studies have demonstrated that empathetic statements, caring touch, and the presence of a partner perceived as caring can modulate the experience of pain, reduce pain scores, and affect the autonomic response to pain.22,23,24
Anxiety, a threat-detection function mediated by the amygdala, is also a potent mediator of pain. High-anxiety situations induced in the laboratory setting are associated with higher pain scores for a given pain stimulus. 25 Additionally, greater acute pain intensity in the emergency department has been associated with elevated anxiety states, and postoperative pain has been correlated with preoperative anxiety levels.26,27 Indeed, perioperative behavioral interventions have been demonstrated to reduce postoperative pain and pain-related disability. 28 Attention can also be manipulated to treat pain; distraction from a noxious stimulus is associated with lower acute pain scores, while attention focused on the source is associated with higher pain scores.20,25,29,30
As empathy, caring, and distraction can mitigate pain, and conversely, coincident anxiety can increase pain, psychosocial interventions are potentially useful in the treatment of pain. Further, they are well-suited to an austere context, as they are weightless, durable, portable, and cost nothing. The most robust literature targeting the acute reaction to stress and its mitigation comes from work around posttraumatic stress disorder (PTSD) stemming from combat. Treatment modalities targeting reactions to stress in survivors of trauma have been codified into a toolset referred to collectively as psychological first aid (PFA). 31 PFA, while developed to treat survivors of combat, has more recently been directed toward wilderness responders and expedition members and has been integrated into wilderness medical texts and curricula.32,33,34
The full extent of PFA is beyond the scope of this guideline. However, its principal components provide a framework and shared vocabulary for psychosocial interventions that are likely to aid in the treatment of pain through mitigation of negative emotional states, limitation of stress response, and facilitation of adaptive behaviors. 21 These components have been adapted by the authors into a useful mnemonic for the application of PFA in austere environments (Table 1). 35
The ABCDE mnemonic for the psychosocial treatment of pain: Suggested mnemonic for the field application of psychological first aid to reduce acute pain.

Recommendation: We recommend that providers train in and apply relevant psychological first-aid techniques for treating pain in austere environments (strong recommendation, low-quality evidence).
Splinting, Bandaging, Immobilization, Cryotherapy
Treatment of acute musculoskeletal injuries has traditionally involved variations of the “protection, relative rest, ice, compression, and elevation” (PRICE) treatment protocol. Limited evidence supports this practice. However, PRICE remains in widespread use based on moderate-quality evidence and expert opinion.36,37
The physiological basis underlying this approach is to reduce edema formation. Extensive edema can be both painful and detrimental to mobilization and tissue healing.38,39 Currently, the PRICE protocol is considered the best initial treatment regimen for suspected soft-tissue injury and fractures. An alternative strategy called the “movement, exercise, analgesics, and treatment/therapy” (MEAT) protocol has been proposed for managing isolated ligament and tendon injury when fractures have been excluded. The MEAT protocol embraces inflammation, as the inflammatory process promotes healing and a quicker return to normal function. One randomized trial showed that early therapeutic exercise resulted in a quicker return to normal function versus PRICE therapy. 40 If an isolated ligament or tendon injury is highly suspected and fracture is unlikely, MEAT therapy can be substituted. In an austere environment, however, providers may be unable to differentiate between an acute fracture or isolated soft tissue injury, and thus, PRICE remains the recommended therapy for most acute musculoskeletal injuries.
The following treatment strategies are recommended for optimal outcomes when utilizing the PRICE protocol:
Protection from further injury and providing additional stability with taping, bracing, and/or splinting.
41
Relative rest will reduce inflammation and pain. If necessary for evacuation, patients can ambulate. Ice or snow application, when available, will decrease skin temperature below 15 °C, which is the temperature at which nerve conduction is inhibited and pain decreases.
42
Lower tissue temperature also reduces edema formation.
39
Cooling should be performed with cycles of 10 min of cooling followed by 10 min of passive rewarming, whenever practical, for the first 24 to 48 h post-injury.
43
This schedule may not be logistically feasible during an evacuation. The tissues should not be cooled to a degree that might result in frostbite or hypothermia.7,44 When ice or snow is not available, cold water may be substituted for cooling therapy. Evidence for cryotherapy is limited, so the risks of exposing the extremity and patient to potential frostbite and hypothermia must be weighed against the modest benefits of cryotherapy.
37
Compression with an elastic bandage or compression stocking can be used with the intent to reduce swelling. Some evidence exists to support this practice, though only a small number of trials have been published and results are mixed.
45
The compression should be form-fitting while still allowing adequate muscle expansion and sufficient blood flow. Capillary refill, sensation, and movement of the distal extremities should be checked regularly to monitor for excessive compression. Elevation of the injured area above the level of the heart will reduce potential swelling.
Recommendation: We recommend that applicable elements of the PRICE treatment protocol should be used for acute musculoskeletal injury as first-line pain therapy in austere environments. Injuries in which fracture is thought unlikely should use the MEAT treatment protocol (strong recommendation, low-quality evidence).
Acetaminophen, Anti-inflammatories, and NSAIDs
Acetaminophen (Paracetamol)
Acetaminophen (APAP), also known as paracetamol, is a widely used analgesic and antipyretic agent. It acts by inhibiting cyclooxygenase (COX) pathways in the CNS, enzyme pathways required for the production of prostaglandins and prostacyclins. 46 Both the analgesic and antipyretic effects of APAP are attributed to a reduction in prostaglandin production. 47 More recently, research suggests that APAP may also act as a cannabinoid system modulator. 48 The analgesic and antipyretic actions of APAP appear to be limited to the CNS, as the medication has not been shown to act on peripheral COX pathways. For this reason, APAP is ineffective as an anti-inflammatory. 48
The most commonly reported side effect of APAP administration, though rare, is GI discomfort.49,50,51 The most common severe effects of APAP ingestion are seen with hepatotoxicity from significant overdose. APAP has been associated with hepatocellular damage and death. This is usually seen in high doses, typically a single administration >4 g/day in healthy adults, or in chronic use of high doses (though toxicity from a single administration <150 mg/kg for adults or 200 mg/kg for children is rare). Care should be taken in chronic alcoholics and other patients with hepatic dysfunction, where the maximum safe daily dose of APAP may not be known.50,51,49
APAP has the advantage of being lightweight and low bulk. APAP is available in PO formulations (tablets, capsules, syrup, and suspension or solution), as a rectal suppository (PR), and as an IV infusion. Multiple studies have evaluated the efficacy of IV APAP against PO and rectal (PR) administrations and have found clinical equipoise among all formulations.52,53 The analgesic activity of all formulations of APAP diminishes over 6 h. 54
PO administration of APAP is generally the preferred route in most environments if the patient can swallow. The IV route may be utilized with good effect, but no benefit has been shown with this route over others. If the patient cannot take medications PO, PR administration is likely faster, easier, and equally effective. No evidence has suggested teratogenic effects from APAP use in pregnancy. 55
Recommendation: We recommend that APAP, in any formulation or route, should be limited to a maximum daily dose of 60 mg/kg or 4 g in four divided doses (strong recommendation, moderate-quality evidence).
Recommendation: We recommend that in the austere setting, APAP should be administered PO if possible. If a patient cannot tolerate PO medications, we recommend rectal administration of APAP as second line with suppository (preferred) or PO tablet given rectally (second line) (strong recommendation, moderate-quality evidence).
Nonsteroidal Anti-inflammatories
Non-steroidal anti-inflammatory drugs (NSAIDs) are a class of medications that act by inhibiting COX to produce anti-inflammatory as well as antipyretic and analgesic effects.56,57 They are further classified into salicylates (eg, aspirin), propionic acids (eg, naproxen, ibuprofen), acetic acids (eg, diclofenac, indomethacin), enolic acids (eg, meloxicam), and selective COX-2 inhibitors (eg, celecoxib). Except for the selective COX-2 inhibitors, NSAIDs are nonselective inhibitors of both COX enzymes: COX-1 and COX-2. COX-1 expression is involved in renal function, platelet aggregation, and maintenance of the gastrointestinal (GI) lining. COX-2 expression is generally quiescent until induced during an inflammatory response. 58 NSAIDs have been demonstrated to be effective analgesic agents that avoid or reduce the requirement for opiate medications.59,60
NSAIDs have numerous potential adverse effects, mainly related to their nonselective inhibition of COX-1 activity. While appropriately dosed short-term use of over-the-counter NSAIDs has been shown to be safe and well-tolerated, prolonged use and/or high doses of NSAIDs may lead to gastritis and peptic ulcer disease.61,62,63 COX inhibition may lead to diminished platelet aggregation that may be compounded by concurrent NSAID-induced gastritis and/or underlying coagulopathies. 64 However, multiple studies have shown that no increase in postoperative bleeding occurs with short-term use of NSAIDs.65,66 Patients with reduced renal function or altered hemodynamics due to acute volume depletion or underlying disease may be at risk for renal injury from renal papillary necrosis leading to further renal dysfunction, electrolyte disturbances, and nephrotic or nephritic syndromes. 67 While generally well-tolerated in those with normal renal function, recent data suggest that ibuprofen may have a nephrotoxic effect even in those with no prior renal disease. 68
Studies of multiple NSAIDs have suggested an analgesic ceiling at submaximal doses—for example, 400 mg PO for ibuprofen.69,70 The question of whether an additional anti-inflammatory effect is seen at higher doses is unclear. A 2015 Cochrane review could not find evidence of “clinically important differences in analgesic efficacy between NSAIDS and other PO analgesics,” suggesting that any additional anti-inflammatory effect for doses above the analgesic ceiling has no significant effect on pain, though this is based on low- or very low-quality evidence. 71
Ibuprofen and naproxen are the two most common PO NSAIDs used for analgesia. Aspirin, though a common medication, is usually eschewed for analgesic use, particularly in traumatic injury, due to its anticoagulant properties. The recommended analgesic dose of ibuprofen is 200–400 mg as needed every 6 h PO for adults or 5–10 mg/kg for children. In short-term applications, an ibuprofen dose of 1200 mg/day or less has been shown to lead to no further adverse GI effects than placebo in otherwise healthy patients. 72 Naproxen is also administered PO and is available in either immediate and extended-release tablets or suspension forms. Recommended analgesic dosing is generally a 550 mg loading dose, followed by 250 mg every 12 h PO for adults, and 5 mg/kg every 12 h for children (maximum 10 mg/kg/d). Meloxicam is another PO NSAID notable for austere use in that its recommended analgesic dosing for adults is 7.5–15 mg PO as needed only once daily.
Ketorolac is an NSAID that is available PO and in a parenteral form, given IV or IM. Studies show no difference in the reduction of pain scores when IM ketorolac is compared with PO ibuprofen, though the IM form may be useful when PO medications cannot be administered.73,74 Doses for adults are 15 mg IV or 30 mg IM every 6 h. 69
Topical NSAIDs, though proven to be effective analgesics, have a limited role in the treatment of significant injury in austere environments, given the increased weight, bulk, and limited stability of these preparations. 75
Recommendation: We recommend NSAIDs as an effective class of analgesic agents that should be employed as a first-line treatment for mild to severe pain in the austere environment in the absence of contraindications (strong recommendation, moderate-quality evidence).
Recommendation: We recommend that treatment strategies with NSAIDs should employ the lowest effective dose for the shortest possible duration to minimize adverse effects (strong recommendation, high-quality evidence).
Recommendation: We recommend that NSAIDs should not be used with known renal dysfunction, pregnancy, history of bariatric surgery, and/or history of GI bleeding (strong recommendation, high-quality evidence).
Synergistic Effects of Non-opioid Analgesics
Combinations of NSAIDs with APAP have been demonstrated to provide superior pain control to either drug alone or in combination with a PO opioid. The side effects are fewer, and patient satisfaction is higher. These benefits have been seen in a variety of injury patterns and in postoperative patients.76,77,78 A systematic review of 21 studies over 10 years found a combination of APAP and an NSAID was superior to either drug alone. 79
Military applications of NSAIDs/APAP combinations have been successful.79,80 US military units give combat-deployed soldiers a “pill pack” to be taken immediately after a penetrating extremity wound of any type. These packs contain APAP, meloxicam, and an antibiotic. 81
Acute traumatic pain can be treated with a combination of APAP and an NSAID effectively and, in many cases, without opioids. For severe pain, the initial doses of APAP and an NSAID should be simultaneous, after which the medications should be administered according to their appropriate dosing intervals. As the foundation of medical therapy for pain due to acute injury, these medications should be regularly administered throughout the acute injury period.
Recommendation: We recommend that a combination of APAP/NSAID should be considered in the treatment of moderate to severe acute pain in the austere environment (strong recommendation, moderate-quality evidence).
Opioid Analgesia
If psychological first aid, PRICE therapy, and non-opioid therapy are inadequate, or pain is sufficiently severe, an escalation of analgesic agents may be considered. Providing opioid analgesia must be undertaken with care in the austere environment as opioid use can be associated with a wide range of side effects, most concerningly respiratory depression and loss of airway protective reflexes. Other opioid side effects include dysphoria, euphoria, pruritus, nausea, vomiting, and constipation..82,83,84 Nonetheless, opioids have an established role in the treatment of severe acute pain in the wilderness or austere environment.82,85,86 Choice of opioid, route of administration, and dosage are dependent upon many factors, including scope of practice, mechanism and extent of injuries, logistical issues of extraction and transport of the patient, comorbidities, and presence of hypovolemic shock.
Respiratory depression and loss of protective airway reflexes caused by opioids should always be considered when administering opioid analgesia, especially when using fast-acting or potent formulations. With careful titration and administration by experienced physicians and other providers, these problems can usually be avoided.82,85 Whether opioids are part of the analgesic plan for an expedition, personal use, or wilderness EMS, consideration should be given to management of complications, and a risk/benefit analysis must be performed in advance of carrying and administering opioids. At a minimum, providers administering opioid analgesia should have basic training in airway management as this is the primary means of managing respiratory depression due to opioid use. Naloxone is a competitive opioid receptor antagonist that is useful in temporarily reversing respiratory depression and sedation due to opioids but also carries the risk of precipitating acute opioid withdrawal in chronic users. Carrying naloxone as a rescue medication should be considered when more potent forms of opioid medications are in use.
Recommendation: We suggest that opioid analgesia can be an effective component of acute pain management in austere environments; however, consideration of risk should be weighed against the benefit of these medications (weak recommendation, moderate-quality evidence).
Recommendation: We recommend that providers who administer opioid analgesics be prepared to recognize and manage respiratory depression. Naloxone availability should be considered when nonoral opioids are in use (strong recommendation, low-quality evidence).
Oral Opioids
Compared with parenteral preparations, PO formulations of opioids have the benefit of being easier to carry, store, and administer. These medications carry the least concern among opioid formulations for acute respiratory depression and are generally considered safe for unmonitored use. However, caution should be used in opioid-naïve patients, and short-acting opioids should be chosen over long-acting formulations. 87 Durability and ease of administration make PO opioids an attractive form of analgesia for the austere environment.
PO opioids are often combined by manufacturers with other analgesics, such as APAP, to provide synergistic pain relief. 88 When using these combined medications, care should be taken to monitor the total dose of both medications. It is preferable to separate the medications when possible and carry opioid and non-opioid medications independently to allow for titration of each medication. Non-opioid PO analgesics have been shown to be equally effective as or noninferior to PO opioid analgesics and opioid combination medications, and they generally have a better side effect profile.89,90,91 This knowledge should prompt a serious consideration of whether PO opioid analgesia provides any additional benefit over non-opioids in an austere environment. Psychological first aid, PRICE, and non-opioid medications should also be employed where appropriate to reduce the requirement for opioids.
Recommendation: We recommend that PO opioids or opioid/APAP combination drugs are effective for the treatment of acute severe pain in austere settings. However, non-opioid PO analgesic regimens may be preferred for their equivalent efficacy and superior safety profile (strong recommendation, moderate-quality evidence).
Intranasal, Transmucosal, Sublingual, and Transdermal Opioids
Noninvasive methods of parenteral opioid administration include IN, transmucosal (TM), SL, and transdermal (TD) routes. Fentanyl is the most frequently utilized opioid medication for non-IV parenteral use, given its relatively predictable pharmacokinetics.92,93 PO fentanyl is generally ineffective due to extensive first-pass metabolism. However, IN, TM, and SL opioids are not subject to this metabolism and have the advantage of providing acute pain relief without the requirement for IV access. TM and SL formulations have traditionally been utilized for the treatment of breakthrough cancer pain with great success.94,95
Intranasal opioid administration also provides a method for acute pain relief without the need for IV access. The IN route provides for rapid absorption of opioids. IN Fentanyl is most widely utilized in EDs, by EMS systems, and in wilderness settings such as on-hill ski patrol care.96,97,98,99,100 It has also been found to be safe for pediatric use in multiple settings.101,102,103,104,105 IN opioids are best delivered in a volume of 0.2–0.3 ml into each nostril. 106 Volumes greater than 1 ml per nostril are not absorbed nasally but rather swallowed, undergoing first-pass metabolism. The drug dose administered should be split between the two nostrils to maximize the absorptive area available and to keep volumes as low as possible. IN medications are most effectively administered through use of an atomizer device.107,108 The standard 50 ug/ml concentration IV fentanyl is used for IN administration.
When larger doses of opioids are desired IN, the volume of fentanyl required must be given as multiple doses over time to limit volumes per nostril. To overcome this volume limitation and provide greater analgesia in small volume doses, the more potent fentanyl analog sufentanil has been utilized. Sufentanil is nearly 100% bioavailable when given IN and at 20 min achieves equivalency with similar IV doses. 109 It has been successfully and safely utilized in clinic and ED settings.110,111
Oral transmucosal fentanyl citrate (OTFC) in lozenge form delivers fentanyl by TM absorption. OTFC was originally developed for treatment of cancer-related pain. OTFC has been used extensively by the US military and is safe when utilized in appropriate doses in a healthy military population.112,113 Safe use in the military has also led to safe and effective OTFC use in prehospital environments and recommendations for use in mountain rescue.18,114
Sublingual fentanyl administration has been shown to have pharmacokinetics similar to IV fentanyl. 115 Recently, sufentanil SL has been approved for treatment of acute pain in opiate naive patients in a monitored setting, developed with support from the United States Department of Defense to provide a non-IV method of battlefield pain control. The SL formulation is safe and effective and has a decreased side-effect and euphoric profile in comparison to fentanyl. 116 Studies of sufentanil SL use have been limited to ED- and hospital-based settings, but pooled studies of 436 and 806 patients suggest that it is safe and efficacious in monitored healthcare settings.117,118,119 There have not yet been any published studies on prehospital use.
Transdermal opioids are designed for long-term maintenance of pain control in opioid-tolerant individuals and have limited utility in the wilderness setting due to their slow onset of action. Due to a paucity of evidence for the treatment of acute pain, transdermal opioids are not recommended for use in austere environments.
Recommendation: We recommend OTFC for safe and effective treatment of pain in austere settings (strong recommendation, moderate-quality evidence).
Recommendation: We recommend intranasal fentanyl for safe and effective pain control in austere environments (strong recommendation, moderate-quality evidence).
Recommendation: We suggest that despite limited documented prehospital use, sublingual fentanyl and sufentanil are likely safe and effective for austere use, provided that patient monitoring is available (weak recommendation, moderate-quality evidence).
Intravenous, Intramuscular, and Intraosseous Opioids
IV opioid administration can provide fast and effective analgesia.82,86,120,121,122 However, obtaining and maintaining IV access may be difficult in austere environments as it requires appropriate skills, suitable equipment, and ongoing maintenance. Environmental factors such as cold or freezing temperatures may also weigh on the decision to pursue IV access.
IV opioids carry an increased licensing and regulatory burden and fall outside the scope of practice for many austere medical providers. The availability of IV medications may be more reasonable within organized wilderness EMS systems or in expedition base camps where prolonged stays and delayed evacuations may occur. Consideration should be given to the potential complications of parenteral opioid use, and all providers administering these medications should be proficient in basic airway management.84,85,121 Naloxone can be considered as rescue medication.
Intraosseous access (IO) is an alternative route that is more easily established in certain situations and should be considered when IV access is needed and cannot be readily obtained. 123 IV and IO routes have several advantages over the PO route: they provide rapid pain relief and reliable drug delivery, are titratable, and may be a familiar mode of administration for healthcare professionals.
Fentanyl has been recommended as the drug of choice for prehospital IV pain control in the EMS setting.85,122,124 In austere or prolonged settings, a longer-acting opioid might be preferred. Although the invasive nature of the IV route makes it impractical for many austere environments, once IV/IO access is established, these medications are still considered the gold standard for control of acute, severe pain. 85 The IM route avoids the need to establish IV or IO access, but due to variable absorption, onset and efficacy may be inferior. IM fentanyl and morphine have been compared prospectively and proven equivalent in terms of analgesia and side effects when given in equipotent doses. 125
Recommendation: We recommend that parenteral opioids be used to provide analgesia for acute severe pain in austere environments when benefits outweigh risks and when more conservative modalities do not provide sufficient analgesia (strong recommendation, moderate-quality evidence).
Ketamine
Ketamine is a dissociative anesthetic agent that is an N-methyl-D-aspartate (NMDA) antagonist suspected to block afferent effects of pain perception in the medullary reticular formation, alter certain CNS transmitter systems, and suppress spinal cord activity. 126 In common formulation, ketamine is a 50:50 racemic mixture of s- and r-ketamine. The s-ketamine enantiomer is a more potent analgesic agent and, in subdissociative doses, has fewer psychiatric side effects than the racemic mixture, which has led to its growing use as an analgesic in some countries. 127 The more widely available racemic form is discussed here.
In lower doses, ketamine has a long history of prehospital use for acute pain. Ketamine can be administered via several routes, including IM, IV/IO, IN, PO, SL, and PR. 128 Most wilderness applications involve IM, IN, and IV/IO routes.
Ketamine is unique among pain control medications in that it has variable effects at different doses, and care must be taken to remain within the range of “pain dose” or “low dose” ketamine (0.1–0.5 mg/kg IV or 1–2 mg/kg IM).128,129 At higher doses (1–2 mg/kg IV or 3–5 mg/kg IM), ketamine causes dissociation, a type of moderate sedation often employed in hospital use. 130 Between these dosing ranges, ketamine can cause hallucinations, euphoria/dysphoria, delirium, and other undesirable effects.
For pain control, ketamine is administered within the low dose range as above by slow IV push, IV infusion, or IM injection. If repeat dosing is undertaken, care must be taken to avoid rapid redosing and induction of hallucinations or sedation. Ketamine can be safely used in combination with opioids to enhance pain control if multiple modalities are needed. 131 Ketamine is well-tolerated and has been shown to be safe when administered in dissociative doses by nonphysician practitioners to facilitate procedures, which reinforces its safety when used in pain control doses. 132
Ketamine has been reported to be safe and useful and to have few associated adverse events when used by physicians working in an air medical mountain rescue service. 133 It has also been safely used at dissociative doses for procedures at high altitude (>3900 m) by primary care physicians without anesthesia training. This application in 11 cases found no significant complications or hypoxia associated with ketamine use. 134
In appropriately titrated pain doses, patients can maintain their own airway, as pharyngeal reflexes and spontaneous ventilation are usually preserved. However, salivation is often increased, and airway obstruction has been reported with dissociative doses. 135 Unlike many anesthetic and analgesic agents, cardiovascular function is often well-preserved in the setting of ketamine administration. This makes ketamine a suitable choice for trauma patients if hemorrhagic shock is suspected. 136
Recommendation: We recommend ketamine for the control of acute severe pain in the austere setting using pain-control doses (0.1–0.5 mg/kg IV, 1–2 mg/kg IM). Providers should be experienced in using the medication and equipped to manage airway complications should they arise (strong recommendation, moderate-quality evidence).
Local Anesthetics
Local anesthetic (LA) agents act by prolonging deactivation of voltage-gated sodium channels. When acting on peripheral neurons, they reduce or reversibly block neuronal transmission, leading to local analgesia or anesthesia. However, when absorbed systemically, these medications can cause severe adverse effects by acting on sodium channels in the myocardium or CNS, such as cardiac arrest and seizure. Attention must thus be given to weight-based maximum doses to avoid toxicity (eg, lidocaine 4–5 mg/kg, maximum 300 mg/dose and 2400 mg/day), and care must be taken to avoid intravascular or intraneural injection.
Worldwide, the most used LAs are lidocaine (lignocaine) and bupivacaine, although there are many variations and preparations within the category. The onset, duration of action, and density of the local analgesia or anesthesia produced by LAs depend upon multiple medication factors, including the lipid solubility, dissociation constant, concentration, and total dose. Typically, agents with more-rapid onset tend to have shorter duration of action, whereas longer-acting drugs carry higher risks of toxicity. 137
LAs can be applied topically to the skin or mucous membranes, injected directly into peripheral tissues, or adjacent to regional nerves or even the spinal cord to produce regional or neuraxial analgesia or anesthesia.
Topical Local Anesthesia
Topical anesthetic agents are directly applied on or around peripheral sites of nociception in gel, liquid, ointment, or cream form, blocking noxious stimuli at their source.138,139,140,141 Administration is simple, and the low systemic absorption reduces the risk of side effects. 142
There are many available formulations of anesthetics for topical use, including combination products and viscous formulations to facilitate easy application (eg, lidocaine/epinephrine/tetracaine [LET] and the eutectic mixture of lidocaine/prilocaine [EMLA]). The efficacy and safety of these formulations have been demonstrated for the treatment of wounds and stings and for the facilitation of laceration repairs.143,144 The efficacy of these formulations outside of recommended storage temperatures of 0–30° C has not been studied and may limit their use in some austere environments.
Recommendation: We recommend locally instilled and topically applied local anesthetic agents for safe and effective treatment of pain related to burns, stings, and soft tissue injuries in austere environments (strong recommendation, moderate-quality evidence).
Ophthalmic/Otic Solutions
Ophthalmic and otic solutions of topical anesthetics are useful in the treatment of conditions including corneal abrasions or ulcerations, ultraviolet keratitis, otitis media, and otitis externa.145,146 These preparations differ from plain aqueous solutions of LAs in that they often contain preservatives, buffers, viscosity agents, and other additives to make them more effective and tolerable at the target site. 147 Lacking alternatives, an aqueous solution of lidocaine, as used for local infiltration, can be used in topical ocular application. However, the lack of pH buffering reduces penetration into the tear film and causes significant discomfort on instillation, reducing efficacy and tolerability. 148 Ophthalmic and otic solutions are generally similar in composition, though ophthalmic solutions are sterile and free of particulate matter, so ophthalmic solutions may be instilled in the ear, but otic solutions should not be used in the eye. Otic solutions are generally less expensive. 149
Ophthalmic solutions can be of great use in the care of a patient who has become debilitated by a corneal abrasion or keratitis, as analgesia can facilitate eye opening and allow the patient to participate in their evacuation. Concerns over delayed corneal healing have led many providers to withhold repeated use in the setting of corneal injury. 150 However, a meta-analysis pooling two randomized, prospective studies in the emergency department showed no clinically significant adverse effects in the repeated use of ophthalmic proparacaine for less than 72 h. 151 Accordingly, it is reasonable to use a short course of ophthalmic anesthetics to aid in the evacuation of otherwise debilitated patients. Due to reduced capacity for self-monitoring, care should be taken to protect an anesthetized eye from insults, including dryness or exposure to foreign bodies or to UV radiation. 152
Recommendation: We suggest that ophthalmic anesthetic solutions are, when used appropriately, safe and useful for the treatment of acute ocular and auricular pain in austere environments (strong recommendation, low-quality evidence).
Regional Analgesia
Regional analgesia employs LA injection near and about nerves or nerve plexi to achieve broad areas of analgesia and anesthesia. The use of regional analgesia/anesthesia in the austere environment has the potential to meet many goals of austere pain management, such as avoiding sedation, respiratory depression, cardiovascular instability, and other unwanted side effects.153,154 Regional anesthesia also uses a limited amount of compact equipment to provide rapid and predictable onset of excellent pain control. However, such approaches require additional equipment, pharmacological agents, training, and experience. Selection of patient, setting, and technique must thus be carefully considered. 154 Neuraxial (intrathecal/spinal and epidural) injections are unlikely to be suited to austere conditions outside of specific circumstances and thus fall beyond the scope of these guidelines.
Field-Expedient Regional Blocks
In contrast to neuraxial techniques, regional anesthesia and analgesia can be achieved in many settings with minimal equipment and limited training while still retaining an acceptable margin of safety. Well-executed nerve blocks can provide excellent and long-lasting analgesia or full anesthesia of a limb or region to allow interventions and even mobilization and self-rescue. However, these advantages must be balanced with safety; nerve blocks may also hamper self-rescue or render a limb unusable for hours.
The concept of field-expedient regional blocks (FERBs) refers to nerve block techniques that:
Have consistent approaches using reliable anatomical landmarks; Can be performed reliably with a predictable distribution of effect; Avoid structures that carry high risk or consequences of injury (eg, pleura or major vessels); Have a low risk of unwanted effects (eg, phrenic nerve block and diaphragmatic paralysis, which frequently accompanies interscalene brachial plexus block); Require drug volumes that are well within the maximum safe dose of the selected LA; Tend toward slower drug absorption, reducing the risk of systemic toxicity; Are easily learned and can be practiced by austere medicine practitioners working in a variety of settings to become proficient before using them in the field; Do not require highly specialized equipment.
155
While a full discussion of suitable FERBs is beyond the scope of these guidelines, examples of the concept include certain orbital, dental, and occipital blocks in the head and neck; coracoid, wrist, and interdigital blocks in the upper limb; the fascia iliaca plane block, popliteal, peroneal, ankle, and intermetatarsal blocks in the lower limb; and ring blockade of any digit.156,157,158,159,160,161
Ultrasound has become the standard of in-hospital care for many regional blocks and is mandatory for some techniques. It allows visualization of tissue planes, nerves, blood vessels, and other structures with minimal discomfort and has many other uses in austere medicine. 153 However, ultrasound requires additional training and practice to be used effectively for regional blocks, which should be obtained in a controlled setting.
Practitioners should know the potential risks and complications of regional nerve blocks and seek patient consent wherever possible before performing a block. Knowledge of the signs, symptoms, and management of local anesthetic systemic toxicity (LAST) is essential. IV access should be obtained before all but the simplest blocks to facilitate treatment of complications as needed, and equipment and drugs for resuscitation should always be available before commencing a regional block. 162 These techniques are, therefore, limited to well-equipped and trained rescue teams and responders.
Recommendation: We suggest that when performed by experienced practitioners with necessary administration and monitoring equipment, field-expedient regional nerve blocks may be an effective tool to manage pain and facilitate procedures in an austere environment (weak recommendation, low-quality evidence).
Recommendation: We recommend that portable ultrasound be used for FERBs in austere environments when appropriate equipment and skills are available (strong recommendation, moderate-quality evidence).
Care of Patients with Nerve Blocks in the Field
Local and regional analgesia may facilitate (but should not delay) life- and limb-saving interventions. Well-executed regional analgesia may improve a patient's ability to communicate or partake in self-care and self-rescue activities, but it does not negate the responsibility to perform ongoing monitoring and assessment. Care should be given to assessing for symptoms and signs of LAST and to the care of the region under analgesia/anesthesia to prevent further injury. Patients with well-executed blocks may not be able to feel pressure points, control an anesthetized limb, or perceive inadequate perfusion from compartment syndrome, dressings, or immobilization measures that are too restrictive.
Recommendations: We recommend immobilization with adequate padding, pressure care, and frequent assessments for perfusion and absence of clinical signs of compartment syndrome as necessary for patients who have received regional blocks in austere environments (strong recommendation, low-quality evidence).
Hematoma Blocks
In contrast to all other modern regional analgesia techniques, a hematoma block aims to deliberately aspirate blood by advancing a 20G needle directly into the hematoma surrounding a fracture using sterile technique. It is nearly exclusively used for reduction of distal radius/ulna fractures, although its use has also been described for fractures of the tibia.163,164 As fracture hematomas can be considered contiguous with the bone marrow space, absorption of LAs via hematoma could theoretically be as rapid as IO drug administration. 165 For this reason, only lidocaine is used for this block, at a maximum dose of 1.5–2 mg/kg. For most adult patients, a volume of 5–10 ml of 1% lidocaine solution is safe and effective.166,167
Recommendations: We suggest that hematoma blocks with plain lidocaine can be performed by suitably experienced practitioners in austere environments (weak recommendation, moderate-quality evidence).
Field Infiltration of Local Anesthetics
Although not a true regional block, infiltration of LAs around the margins of a wound or in an injured region is an effective means of providing analgesia and can extend to procedural locoregional anesthesia. In this setting, there is little advantage to using concentrated LA, and dilutions of 0.5% to 1% lidocaine and as little as 0.1% of long-acting agents can be effective. Use of a narrow-gauge needle and injection with smooth continuous withdrawal reduces the risk of intravascular injection. Caution should still be employed in calculating the maximum safe dose; particularly when providing repeated injections within a limited timeframe.
Recommendations: We recommend field infiltration of dilute LA for safe management of acute pain due to soft tissue wounds in austere environments (strong recommendation, moderate-quality evidence).
Infiltration of Diphenhydramine
Diphenhydramine (DPH) is an antihistamine that is an inverse agonist at the H1 receptor. It is usually employed in the treatment of allergy or anaphylaxis and may be carried as a component of austere medical kits for this reason. While PO formulations of DPH are limited to these traditional indications, a 1% aqueous DPH formulation diluted from the 5% typical for IV use is also useful as an LA, particularly in those patients with allergies to typical LAs such as lidocaine. Local injection of 1% DPH achieves adequate analgesia for wound care within 5 min in 80% of patients.168,169,170 In comparison to lidocaine, the injection of DPH is more painful, the analgesic effect slightly less than that of lidocaine but adequate for suturing and wound care, and DPH analgesia has shorter duration than that of lidocaine.171,172 DHP infiltration can cause dose-dependent mild sedation, and this should be taken into account if the patient is expected to ambulate or participate in self-rescue. 168 The potential adverse effects of intraarterial injection DPH are minimal, particularly in comparison to lidocaine. Local DPH infiltration can, rarely, cause skin irritation.168,172 To date, DPH has not been studied for use in nerve, regional, and hematoma blocks.
Recommendation: We recommend aqueous DPH solution as safe and effective for local infiltration and wound care as an alternative to lidocaine in the austere environment when lidocaine is not available or allergy limits its use. Consideration should be given to its sedating effects (strong recommendation, moderate-quality evidence).
Intravenous Lidocaine
IV lidocaine use as an infusion for the treatment of pain has been widely reported in hospital-based applications and has shown equivalent effect relative to opioids in some applications, as well as opioid sparing effects.173,174 However, anesthesia guidelines describe the use of IV lidocaine as “high risk” even in the hospital setting due to its narrow therapeutic window. 175 In hospital application, access to lipid emulsion therapy is considered necessary to treat possible overdose. In a systematic review of its use in hospital application, of 289 patients treated with IV lidocaine, 44 adverse events were reported, of which 36 (12.4%) were “serious,” including altered mental status and cardiac dysrhythmias. 174 Meta-analysis of its use in the emergency department yielded similar adverse event rates but showed equivocal performance in comparison to opioids. 173
A case report is available describing two patients treated with IV lidocaine in an austere setting. Both patients experienced improved pain; however, both patients were treated without the availability of lipid emulsion and both experienced prodromal symptoms of LAST. 176 This highlights the hazard of employing this method without hospital-level resources. Because of the degree of monitoring and resuscitation equipment required, and given the very poor therapeutic index of this method, IV use of LAs is not recommended.
Recommendation: We recommend that IV lidocaine should not be used in the austere environment (strong recommendation, low-quality evidence).
Inhalational Analgesics
Inhalational analgesia is provided by breathing specific gases or volatile agents that induce analgesic effects after absorption in the lung. This should be distinguished from topical/TM administration of agents using atomization or nebulization.
Inhalational analgesia is characterized by rapid on- and offset, ease of administration, lack of immediate requirement of IV access, and comparable efficacy to other systemic agents. Currently, there are only two inhalational analgesics in clinical use: nitrous oxide (a gas), and methoxyflurane (a vapor). Both require specific equipment and methods to administer and are discussed further below.
Nitrous Oxide
Nitrous oxide (N2O) is a nonflammable, colorless gas with a slightly sweet scent that has euphoric and dissociative anesthetic properties at high partial pressures and significant analgesia at lower doses. N2O is typically self-administered by breathing through a demand-valve and mouthpiece that is held between the teeth or directly through an anesthetic face mask. Onset of action is rapid, within several breaths; clinically significant analgesia occurs within 5 min. 177 While the precise mechanism of action is not fully understood, anesthetic effects are likely through noncompetitive central NMDA-receptor antagonism, while analgesic effects are mediated by endogenous opioid release in the midbrain. 178 It is commonly used as a synergistic agent during induction and maintenance of general anesthesia but has also been extensively administered in combination with oxygen (O2) as an analgesic for painful conditions and procedures such as labor and delivery or dental work. Outside of the operating theater, it is most commonly available in an equal, preblended (50%) mixture with oxygen, known as Entonox. N2O can also be mixed with a separate supply of pure O2 using a blender, though this is more cumbersome for prehospital use. N2O has been used with considerable success as an analgesic in prehospital care for many years.177,179
Patient-administered N2O analgesia has been shown to have a low rate of adverse events and sedation in both children and adults.180,181 The most common side effects, such as nausea, dizziness, and mild sedation, are normally short-lived. At analgesic doses, it exhibits cardiovascular stability and no respiratory depression and can provide analgesia on par with systemic opiate administration.178,182,183,184 The rapid diffusion of N2O can lead to accumulation in gas-filled spaces within the body, and it should be used cautiously in head and chest trauma victims so as not to worsen pneumothorax or pneumocephalus, and its use is contraindicated in recently ascended divers. 185
Three important limitations for the use of N2O in the austere setting exist. First, it is stored in bulky, heavy, pressurized cylinders (eg, Entonox), which require the use of regulators and a demand valve, making transport and storage challenging.
Second, the nature of the pressurized gas mixture reduces the range of ambient temperatures in which it can be used. Direct sunlight and hot temperatures can risk cylinder rupture due to overpressure, and subzero (below −5.5 °C) temperatures will cause the N2O to liquify. Initially, this can result in the inhalation of oxygen with limited N2O in unpredictable concentrations. However, as the cylinder empties, an increasing concentration of N2O is delivered, and the mixture becomes dangerously hypoxic. 210 Accordingly, N2O/O2 mixtures must be administered with care at below-freezing temperatures.
Third, N2O is delivered as a gas, and increasing altitude will cause a corresponding decrease in the inhaled partial pressure of gases. This in turn decreases the analgesic effect of N2O at altitude. 186 Thus, while it is a useful agent at sea level, the value of N2O is greatly decreased at high altitude.187,188,189 Additionally, N2O can accumulate in small and poorly ventilated spaces, so adequate ventilation should be assured to prevent unintentional inhalation among bystanders.
These restrictions on the portability of the agent and use under adverse conditions has discouraged austere use. Although N2O has been employed among ski patrols in Canada, Australia, and the United States, its use on the ski hill, including efficacy and safety, has not been reported. 190
Recommendation: We suggest N2O as a safe and effective analgesic in austere environments for short painful procedures or for limited periods of time. Although evidence pertinent to austere use is limited, it can be safely used at low altitude, with appropriate monitoring, and where appropriate storage of cylinders can be assured (weak recommendation, low-quality evidence).
Methoxyflurane
Methoxyflurane is a halogenated volatile anesthetic with analgesic properties. 191 Its mechanism of action is not well understood, but it is a known GABA receptor agonist and reduces gap junction function in the brain. Although it is no longer used for anesthesia, it has been widely used as an analgesic in Australia and New Zealand by paramedic, military, and civilian first aid providers for over 30 years. 192
Methoxyflurane is self-administered by inhalation through a hand-held, whistle-like device that functions as a simple, disposable draw-over vaporizer. Onset of analgesia is rapid (within 5 min) and continues while the patient breathes through the device. 193 When used continuously, a single dose (3 ml) lasts for approximately 25–30 min. 194 Its effects are rapidly reversed once inhalation stops.
Changes in cardiovascular, respiratory, and neurological function during methoxyflurane administration are not clinically significant, and therefore IV access is not essential.193,195 The most common side effects are dizziness, headache, and feeling somnolent.194,196 Serious adverse effects are rare, but methoxyflurane is contraindicated in patients with renal or hepatic impairment as well as those who have a personal or family history of malignant hyperthermia.98,196
Concerns have been raised as to whether methoxyflurane will suffer the same decrease in efficacy at altitude that is experienced with N2O. 197 However, as saturated vapor pressure is not influenced by ambient pressure, altitude should not affect the partial pressure delivered by the device, a theory that has been borne out in initial laboratory work.186,198 Questions that remain to be studied are the effect of low ambient temperatures on device performance and whether environmental factors influence clinical efficacy of methoxyflurane in the field. Efficacy for analgo-sedation at high altitude (4470 m) appears to be preserved in one case study. 199
Methoxyflurane has been shown to have more rapid onset and comparable or better initial analgesia compared to IV opiates or APAP.200,201,202,203 In addition, its portability, stability, and ease of administration make it highly suitable for use in austere settings.98,204,205 The rapid onset of action and limited side effect profile make methoxyflurane useful as a temporizing measure until other modalities can be established or as a definitive short-term analgesic.192,206
Recommendation: We recommend methoxyflurane as safe and effective to treat pain in subanesthetic doses in austere environments (strong recommendation, moderate-quality evidence).
Adjuncts
Benzodiazepines
Benzodiazepines (BZDs), which have long been used in the treatment of anxiety, have previously been recommended as a possible analgesic adjunct. 15 This was rational because, as previously discussed, there is a correlation between anxiety states and increased perception of acute pain. However, no quality evidence has emerged to support the administration of BZDs for analgesia.
BZDs act centrally on GABA-A receptors, resulting in CNS depression. 207 Additionally, the combination of BZDs with other CNS depressants can potentiate their concerning adverse effects. The combination of BZDs with opioids has been well studied and has the potential to induce significant respiratory depression.208,209,210
Recommendation: We recommend that BZDs should not be used for the treatment of pain in austere settings (strong recommendation, high-quality evidence).
Further Modalities and Adjuncts
A variety of additional medication classes and techniques have been studied as adjuncts for pain in both the hospital an outpatient environment. These include antihistamines, battlefield acupuncture, antipsychotics, non-BZD muscle relaxants, capsaicin, and antidepressant medications. While there is heterogeneous but generally supportive evidence for the use of these medications in the long-term treatment of chronic pain, there is inadequate evidence to support their use for the treatment of acute pain in the austere environment, and austere use cannot be recommended.
Conclusion
In all cases, the comfort of the patient must be balanced with the risk of any tools used to address pain. In comparison to hospital-based or EMS medicine, evidence for pain control modalities specific to the austere environment is generally limited. Accordingly, the experience of the responder, consideration for available resources, and clinical judgment are of paramount importance in the selection of pain control modalities appropriate to the patient and the setting. Some interventions may enable the patient to participate in their own rescue while others may generate unacceptable risk to the patient or team. Backcountry recreationalists, expedition leaders, and professional rescuers alike should consider the resources, hazards, and limitations specific to their unique settings and should both plan for appropriate modalities to manage acute pain and for the training, monitors, equipment, and antidotes needed to manage the risks of those interventions. The paucity of austere-specific research also opens numerous opportunities for further study.
Supplemental Material
sj-pdf-2-wem-10.1177_10806032241248422 - Supplemental material for Wilderness Medical Society Clinical Practice Guidelines for the Treatment of Acute Pain in Austere Environments: 2024 Update
Supplemental material, sj-pdf-2-wem-10.1177_10806032241248422 for Wilderness Medical Society Clinical Practice Guidelines for the Treatment of Acute Pain in Austere Environments: 2024 Update by Patrick B. Fink, Albert R. Wheeler, William R. Smith, Graham Brant-Zawadzki, James R. Lieberman, Scott E. McIntosh, Christopher Van Tilburg, Ian S. Wedmore, Jeremy S. Windsor, Ross Hofmeyr and David Weber in Wilderness & Environmental Medicine
Footnotes
Acknowledgments
The authors would like to acknowledge the contributions of Katarzyna A. Grela, MBBS, FANZCA; Katie W. Russell, MD; Courtney L. Scaife, MD; and Rom A. Stevens, MD, to the unpublished 2019 revision of this clinical practice guideline, which informed the current text.
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The opinions of the authors expressed in this manuscript are their own and do not reflect official policy of the United States Army or the Department of Defense.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
