Abstract
Takayasu arteritis (TA) is now recognized worldwide and is a disease that mainly affects the aorta and its main branches. TA rarely involves the small or medium-sized vessels. Certain vascular lesions, such as arterial stenosis, occlusion, and aneurysm are common with TA. However, patients with new-onset TA who present with left main trunk acute non-ST segment elevation myocardial infarction are extremely rare. We report a 16-year-old female patient with non-ST segment elevation myocardial infarction due to severe stenosis of the left main coronary artery that was caused by TA. She was eventually diagnosed with TA and underwent successful coronary artery stenting combined with glucocorticoids and folate reductase inhibitor therapy. Over the 1-year follow-up, she experienced two episodes of chest pain and was admitted to the hospital. During the second hospitalization, coronary angiography (CAG) revealed 90% stenosis of the original left main trunk (LM) stent. Following percutaneous coronary angiography (PTCA), drug-coated balloon (DCB) angioplasty was performed. Fortunately, a clear diagnosis of TA was made, and treatment was initiated with an interleukin-6 (IL-6) receptor inhibitor. Early diagnosis and therapy for TA are emphasized.
Keywords
Introduction
TA, which is also called “pulseless disease”, is a rare systemic granulomatous inflammatory vasculitis with an unknown etiology. 1 TA is rare and occurs at a rate of <3 per million people in Western countries. 2 TA is more prevalent in women of childbearing age and in Asian populations. Studies have shown that in adults, the clinical prognosis is relatively poor after coronary artery involvement, and the mortality rate is high.3,4 Angiographic and pathological studies have demonstrated that the incidence of coronary involvement is low in patients with TA, accounting for 9% of all TA cases. However, despite the low incidence, myocardial ischemia is a major cause of death in TA, with a rate of up to 50% at 5 years. Therefore, early diagnosis and therapy is essential.5,6 We report a 16-year-old female patient with non-ST segment elevation myocardial infarction due to severe stenosis of the left main artery that was caused by TA.
Case report
A 16-year-old female patient was admitted to our outpatient service on 14 August 2021 with a 2-day history of chest tightness and pain after walking for tens of meters. She had no history of recurrent oral ulcer or vulvar ulcer. There was no history of infectious diseases, such as hepatitis, typhoid, or tuberculosis, and no family history of coronary heart disease or other related diseases. Hemodynamics on presentation were stable, with blood pressure of 93/63 mmHg and no significant difference between the arms, pulse of 88 beats/minute, respiratory rate of 20 breaths/minute, arterial blood oxygen saturation of 99% on room air, and temperature of 35.7°C. Breath sounds could be heard over both lungs with trace wet rales. There was no heart murmur. There was also no murmur over the subclavian artery, abdominal aorta, and renal artery, with good fluctuation in the radial artery and dorsalis pedis artery. An electrocardiogram (ECG) revealed ST-segment elevation in the aVR lead and ST-segment depression in leads I, II, III, aVF, aVL, and V1 to V6 (Figure 1).

Electrocardiogram (ECG) obtained during the first hospitalization showing ST-segment elevation in the aVR lead and ST-segment depression in leads I, II, III, aVF, aVL, and V1 to V6.
Troponin T (TnT) and creatine kinase-MB (CK-MB) levels were within the respective reference ranges. The next day, laboratory testing revealed the following: TnT: 0.365 ng/mL (upper limit of normal: 0.1 ng/L); CK-MB: 88.5 IU/L (upper limit of normal: 24 IU/L); white blood cell (WBC) count: 9.24 × 109/L (reference range, 4–10 × 109/L); neutrophils: 7.11 × 109/L (reference range, 2–7 × 109/L); and red blood cell (RBC) count: 4.65 × 1012/L (reference range, 3.5–5.5 × 1012/L). The lipid levels were as follows: total cholesterol: 2.82 mmol/L, triglyceride: 0.54 mmol/L, high-density lipoprotein cholesterol: 0.82 mmol/L, and low-density lipoprotein cholesterol: 1.86 mmol/L. The urea and creatinine levels were within the respective reference ranges. Transthoracic echocardiography revealed hypokinesia of the posterior and inferior walls of the left ventricle, with a left ventricular ejection fraction (LVEF) of 50% and mean pulmonary artery pressure (PASP) of 75 mmHg.
On the basis of the clinical findings, we diagnosed acute non-ST segment elevation myocardial infarction. The treatment strategy and timing for non-ST segment elevation coronary syndrome was made on the basis of the initial risk stratification. After obtaining consent from the patient and her family, we performed urgent coronary angiography (CAG) (Figure 2a), which revealed 99% stenosis in the ostium of the left main coronary artery (LMCA), which resulted in collateral circulation to the distal right coronary artery. Intravascular ultrasonography (IVUS) (model: P/N 807520-001; Volcano Corporation, San Diego, CA, USA) revealed the absence of calcification and that the three-layered structure (intima, tunica media, adventitia) was unclear and concentrically thickened (Figure 2b). Therefore, with the results of the CAG, we decided to place a single stent; i.e., we performed a crossover technique from the LMCA to the proximal segment of the anterior descending branch, which crossed the circumflex branch opening. A drug-eluting stent (Firebird2, 3.5 ×18 mm; MicroPort, Shanghai, China) was implanted into the LM trunk and the initial segment of the anterior descending branch (Figure 2c). After coronary stent implantation, a non-compliant balloon (Quantum Maverick 3.75 × 12 mm; Boston Scientific, Natick, MA, USA) was inserted, and dilatation was repeated. Post-procedure CAG confirmed that the LCX ostium was not affected, and that the stent had expanded well (Figure 2d). The patient’s chest tightness was relieved after the procedure, and follow-up ECG showed that the ST segment and T wave of the precordial lead had returned to the respective baseline levels. Considering the patient’s young age, we investigated further to identify the underlying mechanism for the ECG abnormalities. Additional laboratory testing revealed a high erythrocyte sedimentation rate (ESR; 36 mm/h) and elevated C-reactive protein level (CRP; 14 mg/L). Immunoglobulin testing revealed the following: complement 3 (C3): 1.430 g/L (reference range, 0.8–1.2 g/L), with unremarkable levels of C1q, C4, immunoglobulin (Ig)G, IgA, IgM, anti-neutrophil cytoplasmic antibodies, antinuclear antibody profile, anti-double-strand (ds)DNA antibody, lupus anticoagulant, antiphospholipid antibody, and antistreptolysin O. Pulmonary computed tomography angiography (PCTA) revealed that the trunk of the left pulmonary artery was slightly narrowed, and the left superior lobe artery, lingual artery, and apical and posterior segments of the right superior lobe artery were not visible, indicating the lumen occlusion (Figure 3a and b). Pulmonary perfusion/ventilation imaging revealed perfusion deficiency in the upper lobe of the left lung and the middle and upper lobes of the right lung (Figure 3c and d). To determine the cause, we performed pulmonary arteriography, which revealed dilation of the main pulmonary artery, slight decrease in local perfusion from the right upper pulmonary artery, occlusion of the left upper pulmonary artery, seen as a loss of distal perfusion, and localized stenosis of the left lower pulmonary artery and decreased distal perfusion (Figure 4). Furthermore, angiography of the thoracic and abdominal aorta revealed that the lumen of the main trunk and primary branch of the right renal artery were thinner compared with the left renal artery (Figure 5a). The enhancement degree of most of the right renal parenchyma was lower than that of the left (Figure 5b). CTA of the other sections of the aorta revealed no abnormalities, including in the common carotid artery, abdominal aorta, and intracranial artery.

Coronary angiography (CAG) during the first hospitalization showing 99% stenosis in the ostium of the left main coronary artery (LMCA) (a). A drug-eluting stent was implanted (b). Intravascular ultrasonography (IVUS) showing no calcification; however, the three-layered structure (intima, tunica media, adventitia) is unclear and concentrically thickened. The area of the external elastic membrane (EEM) of the ostium of the LMCA measures 3.11 mm2, and negative remodeling is visible (c). After implantation of the drug-eluting stent, the minimum area in the stent measures 12.56 mm2, and the stent is well-adhered to the vessel wall (d).

Pulmonary computed tomography angiography (PCTA) performed during the first hospitalization showing that the trunk of the left pulmonary artery is slightly narrowed, and the left superior lobe is not visible (a). The lingual artery and apical and posterior segments of the right superior lobe artery are also not visible (b). Pulmonary perfusion/ventilation imaging showed minimal perfusion in the upper lobe of the left lung (c) and the middle and upper lobes of the right lung (d).
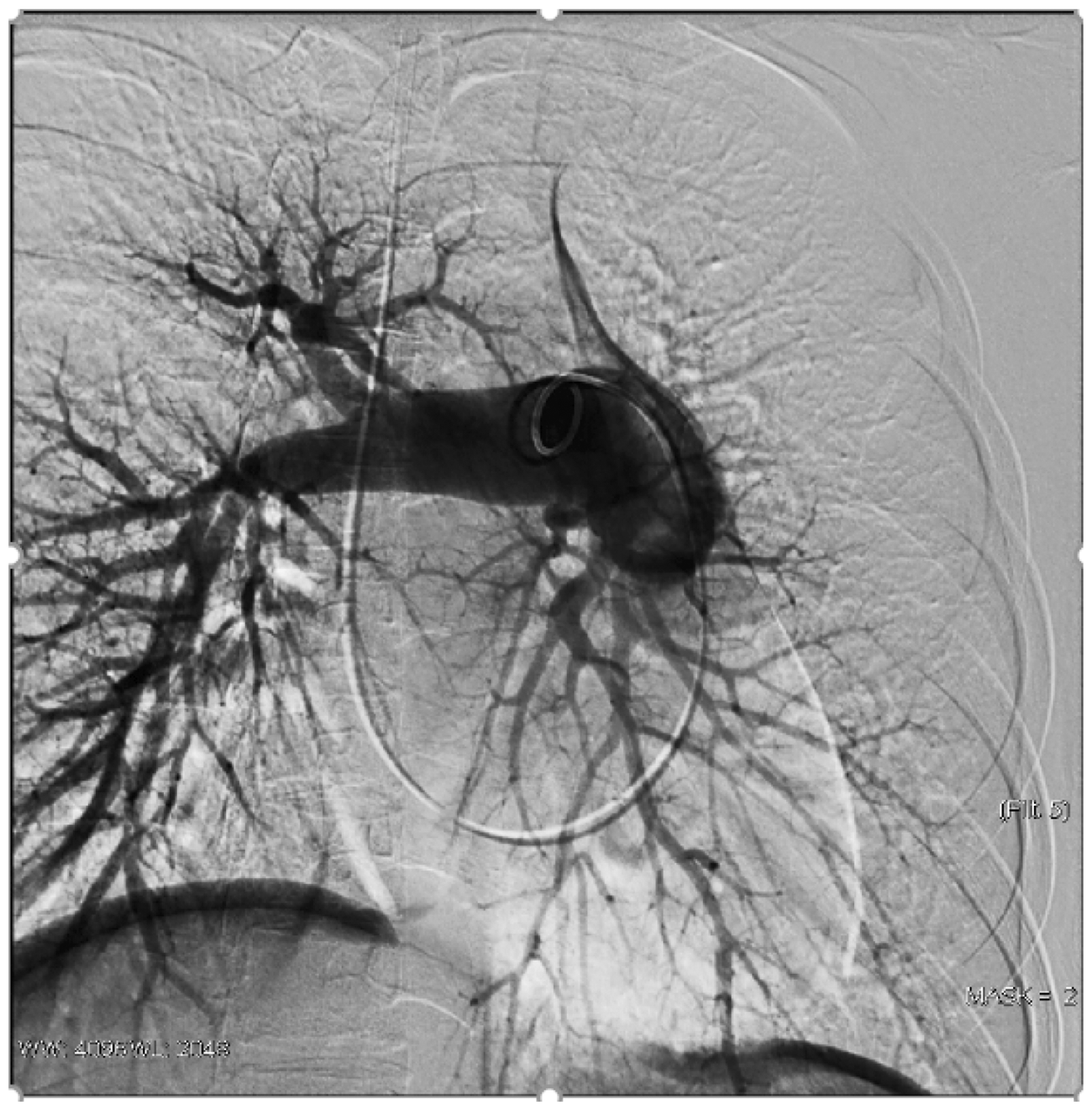
Pulmonary arteriography performed during the first hospitalization showing markedly decreased blood flow through the vessel. The left upper pulmonary artery is occluded, and the left lower pulmonary artery has focal stenosis, with reduced perfusion distally.
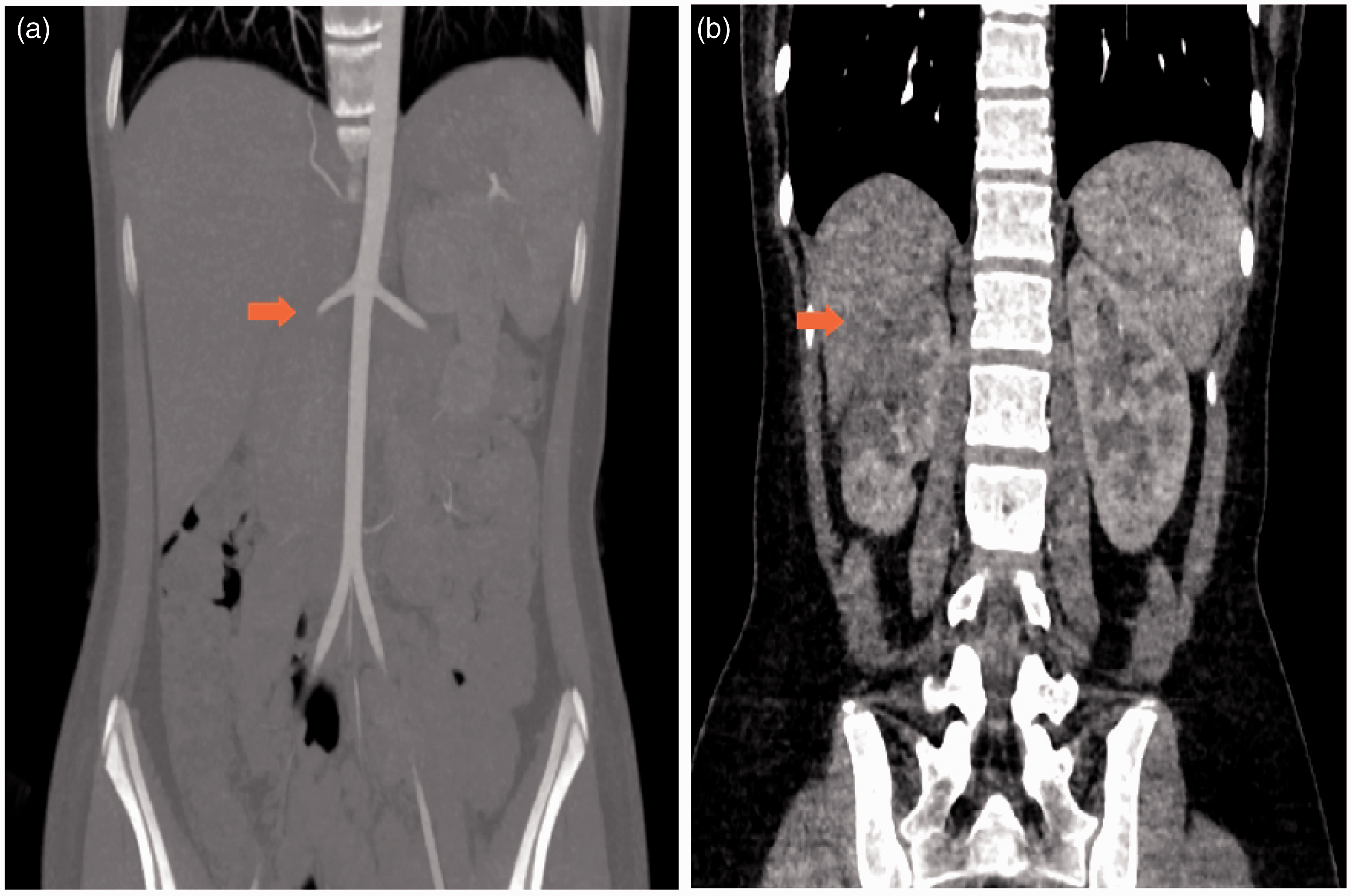
Angiography of the thoracic and abdominal aorta performed during the first hospitalization showing that the lumen of the main trunk and primary branch of the right renal artery are thinner compared with the left (a) and The enhancement degree of most of the right renal parenchyma is lower than that of the left (b).
Positron emission tomography-CT (PET-CT) revealed no abnormally high metabolism or active inflammation in the large blood vessels. However, there was significantly high metabolism in the cardiomyocytes; therefore, we considered myocardial ischemia (Figure 6). On the basis of the patient’s medical history, and the physical and laboratory examination findings, we considered the possibility of TA, with the complications of pulmonary artery occlusion, pulmonary hypertension, and renal artery stenosis. Regarding the pulmonary artery occlusion, pulmonary hypertension, and renal artery stenosis, transthoracic echocardiography and renal function markers were re-evaluated every 2 months. Ten days after the intravascular procedures, the patient was discharged with aspirin (100 mg, qd), tigrillo (90 mg, bid), atorvastatin (20 mg, qn), prednisone (1 mg/kg, qd), and methotrexate (10 mg, qw).

Positron emission tomography-computed tomography (PET-CT) performed during the first hospitalization showing no abnormally high metabolism or active inflammation in the large blood vessels, namely, the common carotid artery (CCA; central arrows) (a), subclavian artery (arrow) (b), iliac artery (arrows) (c), and aorta (arrows) (d). There was significantly high metabolism in the cardiomyocytes.
The patient was scheduled for follow-up every 2 months, and she remained asymptomatic until 10 months post-stent implantation. On 29 May 2022, she experienced chest tightness and shortness of breath, which was associated with activity and fatigue and relieved after 5 minutes of rest, and she was re-hospitalized. ECG revealed myocardial ischemia, as with the previous ECG. Repeat CAG revealed severe in-stent restenosis (ISR) of the ostium of the LMCA (Figure 7a). We then performed mild dilatation of the LMCA using a percutaneous coronary balloon (10 atm × 40 s). Post-procedure IVUS revealed that the minimal luminal area of the LMCA was 4.91 mm2, and the cross-sectional area of the stent was 12.56 mm2. Neointima was visible in the stent, and its thickness was 1.5 mm, with an area of 7.65 mm2 (Figure 7b). Our planned strategy was to use drug-coated balloon treatment, which was performed after sufficient ischemic preconditioning (Figure 7c). Post-procedure IVUS revealed that the minimal luminal area of the LMCA was 12.56 mm2 (Figure 7d), and ECG revealed that the ST-segment had returned to the baseline level.

Coronary angiography (CAG) performed during the second hospitalization showing 99% in-stent restenosis (ISR) in the ostium of the left main coronary artery (LMCA); coronary artery stents are visible (a). Stenosis improved markedly after drug-coated balloon dilatation (b). Intravascular ultrasonography (IVUS) showing that the neointima is visible in the stent, with a thickness of 1.5 mm and area of 7.65 mm2 (the difference between the cross-sectional area of the stent and the minimum cross-sectional area of the lumen) (c). After treatment, the cross-sectional area of the lumen improved markedly, and the minimum luminal area was 12.56 mm2 (d).
Unfortunately, the patient experienced another episode of chest pain on 19 October 2022. We performed PET-CT, which revealed diffuse hypermetabolism in the walls of the large vessels, namely the origin of the left common carotid artery, ascending aorta, right pulmonary trunk, stent implantation site in the LMCA, and left anterior descending branch (Figure 8). Transthoracic echocardiography revealed hypokinesia of the inferior wall of the left ventricle, with an LVEF of 53% and mean PASP of 44 mmHg. Subsequently, the cause of recurrent stenosis in the left main coronary artery was clarified. On the basis of the original treatment, we initiated interleukin-6 (IL-6) receptor inhibitor therapy (tocilizumab, 8 mg/kg for 4 w). Interventional therapy was performed after the macrovasculitis stabilized. A recent follow-up of the patient revealed that the ESR and CRP levels had not returned to normal levels. Therefore, the patient was still unable to undergo percutaneous coronary intervention. We are currently following the patient in the Department of Rheumatic Immunology.
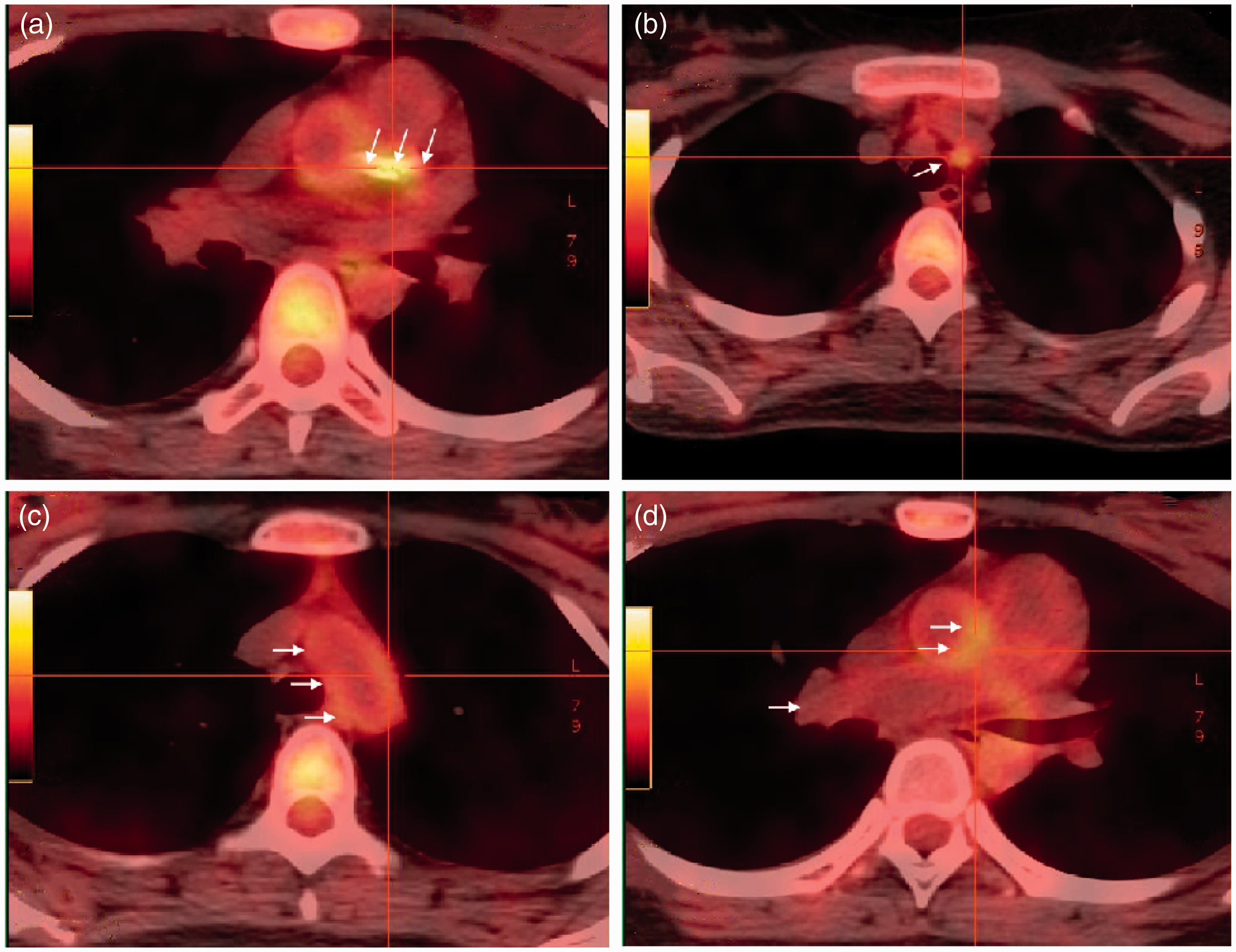
Positron emission tomography-computed tomography (PET-CT) performed during the third hospitalization showing diffuse hypermetabolism in the walls of the large vessels (arrows in each image), namely, the root of the aorta (a), ascending aorta (b), aortic arch (c), and brachiocephalic trunk (d).
The reporting of this study conforms to the CARE guidelines. 7
Discussion
Clinically, TA leads to stenosis, occlusion, or aneurysmal degeneration of large arteries, mainly the aorta and its major branches.8,9 Coronary arteries are affected much more rarely, at a rate of 9% to 11%.10 There are several clinical types of TA: stenosis or occlusion of proximal coronary segments involving the coronary ostia (type 1; most common), diffuse or focal coronary arteritis (type 2), and aneurysms of the coronary vessels (type 3; most rare).10,11 Our patient had Type 1 TA. The lesion in the coronary artery involved the ostium of the LMCA, and CAG revealed complete occlusion of the opening of the left trunk. There were no abnormalities in the other coronary arteries. It is widely believed that abnormal cell-mediated immunity is the main pathogenesis in TA.12
Our patient developed severe coronary heart disease without the expected risk factors; therefore, we must be alert for non-atherosclerotic coronary artery disease. TA must be considered first; however, on the basis of the underlying pathological process, the clinical picture can be divided into an early pre-pulseless systemic phase and a late occlusive phase. In up to 50% of patients with TA, a previous inflammatory history cannot be identified. We combined our patient’s medical history with the results of relevant diagnostics tests, such as arteriography and PET-CT, which we repeated several times, and eventually made a successful diagnosis.
Medical management using corticosteroids remains the basis of therapy in TA; however, some patients also require surgical treatment.8,12 Because the disease is prone to progression and recurrence, corticosteroids combined with immunosuppressive therapy (methotrexate, mycophenolate mofetil, or cyclophosphamide) has been the main means of inducing remission. However, studies have shown that biotherapies, such as tumor necrosis factor- (infliximab) and IL-6 receptor inhibitors (tocilizumab), have been widely used in TA patients unresponsive to corticosteroids and/or conventional immunosuppressive agents, because of their safety and efficacy.13,14 In our case, IL-6 receptor inhibitor therapy did not achieve the expected effect. TA gradually became a chronic disease, and our patient required long-term drug treatment, which was consistent with the findings of Lei et al4 and Kim et al.15 These studies show that TA may be self-limiting in a small number of patients; however, most patients will worsen and develop chronic progressive disease and require long-term immunosuppressive therapy.4,15 Our patient received long-term immunosuppressive treatment and regular follow-up; however, she developed ISR, which is consistent with previous studies stating that coronary stent therapy is prone to the development of ISR.16 Similarly, Bulanov et al reported this situation in a single patient; therefore, the treatment of patients with TA, especially percutaneous coronary intervention, must be carefully considered. 17 For young patients, the long-term effect of CABG is unsatisfactory. Macedo et al. 18 reported that a 23-year-old female patient with TA underwent emergency CABG because of complete occlusion of the LAD and LCX. At the end of the 8-month follow-up, the LM was completely occluded. CABG is traumatic, and it is difficult for young patients to agree to undergo this procedure. Additionally, TA is a chronic progressive disease, and corticosteroids, immunosuppressants, and biotherapies are required long-term. Therefore, it is necessary to follow patients closely and monitor the relevant inflammatory indicators, such as IL-6, ESR, and CRP. However, there is a lack of specific clinical laboratory indicators, which makes patient management a challenge.2,19
In summary, although doctors’ awareness of the diagnosis of TA has recently increased, making this diagnosis is still a challenge for cardiologists and vascular surgeons because of the rarity of the disease. This report aimed to help clinicians consider TA when they encounter patients with acute coronary syndrome as the first symptom. Better awareness will improve the diagnosis rate of TA, and improve the rate of timely treatment.
Footnotes
Author contributions
Yanqing Wang managed the patient’s care, reviewed the literature, and wrote the manuscript. Yawei Duan, Hanqi Su, and Yanling Wang managed the patient’s care and provided guidance on writing the manuscript. Peng Gao and Wenlou Bai interpreted the patient’s imaging results. Hua Chen and Dongxia Liu devised the treatment plan, managed the patient’s care, and provided guidance on writing the manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in the published article.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
This study was approved by the Institutional Review Board of Hebei General Hospital, and written informed consent for publication was obtained from the patient and her family. The corresponding author had full access to all of the data and accepts final responsibility for submission of the manuscript for publication.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
