Abstract
Objective
To provide an overview of global directives, standards, guidelines, and best practices in the management of hazardous medicinal products (HMPs) with closed system drug transfer devices (CSTDs).
Data sources
A literature review was performed using PubMed on CSTD performance, use, and cost-effectiveness. The authors provided information on directives, standards, and guidelines. A roadmap was proposed, which is the expert opinion of the authors.
Data summary
Despite evidence supporting CSTD use and their successful implementation in the United States, their usage remains inconsistent in Europe. Additionally, directives, standards, and guidelines on the safe management of HMPs vary in their recommendations. A unified approach to hazardous medicinal product management using CSTDs is therefore needed which includes training, action by professional societies, clear stances from professional bodies on which CSTDs are recommended, development and wider use of testing protocols, identification of current variables in directives and guidelines, and general alignment on these.
Conclusions
There is a need for global alignment on HMP management and when to use CSTDs. Implementation of the proposed roadmap will lead to improvements in the safety of healthcare practitioners exposed to HMPs.
Keywords
Introduction
Any healthcare practitioner (HCP), including nurses and pharmacy personnel involved in the preparation or administration of hazardous medicinal products (HMPs: which the authors have outlined in Figure 1), experience occupational exposure to HMPs. These include chemotherapy, antiviral drugs, hormones, and bioengineered drugs. 1 This exposure affects 1.8 million HCPs in the European Union (EU) and 8 million in the United States (US).2–4 Traditionally, syringes and needles, and spikes have been used for the preparation of HMPs, when transferring hazardous drugs from drug vials to dosing equipment such as infusion bags, bottles, or pumps.1,2,5 However, these methods are widely regarded as outdated technologies, and have an associated risk of accidental leakage, spills, and contamination.1,2,5 Such accidents can lead to exposure of HCPs to HMPs, resulting in a range of harmful health effects.1,2,6 Acute effects due to HMP exposure may last for weeks or months and include nausea, dermatitis, headache, dizziness, and menstrual problems.7,8 Chronic effects can persist for years, and may involve kidney and liver damage, damage to the lungs and heart, damage to bone marrow, infertility, effects on reproduction and the developing foetus, hearing impairment, and cancer. 7 Preventing exposure to HMPs is therefore crucial to protect HCPs from these serious health risks.

Personnel who can be exposed to hazardous medicinal products.
Hazardous medicinal products should be routinely prepared in the pharmacy using containment primary engineering controls (C-PECs), such as a biological safety cabinets and isolators.7,9 These are housed within containment secondary engineering controls (C-SECs), specifically the room in which the C-PEC is placed. 9 Closed system drug transfer devices (CSTDs) serve as supplemental engineering controls, primarily used to protect HCPs and environmental workers from exposure to HMPs, with secondary effects of preventing contamination of the HMP.1,9
Globally, there are various directives, standards, and guidelines that outline strategies for protecting HCPs from HMP exposure, some of which include guidance on CSTD use. Notably, the number of HMPs that are known or suspected to cause adverse health effects is increasing,10,11 further underscoring the importance of protecting HCPs.
This discussion paper presents an overview of global directives, policies, and best practices in the management of HMPs with CSTDs. Recent evidence on the use of CSTDs and the effectiveness of CSTDs, is also examined, along with a discussion of the regional differences and practical challenges in CSTD use. Finally, a roadmap is outlined for global alignment in HMP management with CSTD utilization.
Definitions of HMPs vary worldwide and lists of HMPs can be outdated
The definition of HMPs varies across worldwide regions (Table 1),1,9–16 although most definitions include drugs that are carcinogenic, reprotoxic, or mutagenic. In the US, a drug is considered hazardous if it fulfils one or more of the following toxicity criteria in humans, animal models, or in vitro systems: carcinogenicity, developmental toxicity (including teratogenicity), reproductive toxicity, genotoxicity, low-dose organ toxicity, or if it has a structure and toxicity profile that mimics existing drugs determined hazardous by these criteria.1,9,10 Japan uses criteria very similar to those in the US. 15 In contrast, in the EU, HMPs are defined as medicinal products that contain one or more substances that meet the criteria for classification in accordance with Regulation (EC) No 1272/2008 (the Classification, Labelling and Packaging [CLP] Regulation) of carcinogenic, mutagenic, or toxic for reproduction.11–13 There is no globally uniform classification of HMPs, which can cause confusion as to when to use a CSTD. The National Institute for Occupational Safety and Health (NIOSH) in the US, the European Trade Union Institute (ETUI) as well as the European Commission have published lists of HMPs (EU HMP).3,10,11 The EU HMP list takes into account HMPs included in the NIOSH list, with additional sources including the European Chemicals Agency and European Medicines Agency database, as well as the ETUI list. The European HMP list was published in 2025 11 and the NIOSH list was last updated in 2024; however, new HMPs are being developed all the time, highlighting the importance of clear definitions of what a HMP is. In addition, the latest NIOSH list does not include biologicals as they are approved by the U.S. Food and Drug Administration's Center for Biological Evaluation and Research, not the Center for the Drug Evaluation and Research. 10 Importantly, the NIOSH list does not include any drugs approved by the US Food and Drug Administration (FDA) since December 2015, 10 underscoring the need for a globally clear and harmonized definition of HMPs.
Select definitions of hazardous medicinal products.

CDER: Center for Drug Evaluation and Research; CLP: classification, labelling and packaging; EAHP: European Association of Hospital Pharmacists; EC: European Commission; EU: European Union; FDA: Food and Drug Administration; HMP: hazardous medicinal product; NIOSH: National Institute for Occupational Safety and Health; US: United States
Directives, standards, and guidelines vary in their recommendations on the safe management of HMPs
Various controls and levels of protection are recommended in the EU, US, and other international directives, standards, and guidelines (Supplementary Table 1). A directive is a legislative act, particularly in the context of the EU, that establishes specific goals that member states must achieve. 17 In the EU, directives must be transposed into the individual countries. A standard refers to a set rule or guideline that helps determine what is acceptable or expected in a certain situation and to improve best practice by standardization/harmonization. 18 In comparison, a guideline generally offers suggestions or recommendations by the organization involved. 19 In the US, standards are enforceable 20 whereas in the EU, it is up to the national authorities or EU commission to adopt global standards. 21
All guidelines, standards, and directives listed in Supplementary Table 1 uniformly emphasize the importance of protecting HCPs from the risks of HMPs. These recommendations include identifying HMPs, maintaining a list of HMPs, health monitoring, prevention measures, regular surface contamination testing, as well as education and training for HCPs involved in their handling. However, these guidelines, standards, and directives show considerable variability regarding the use of protective measures including the use of CSTDs. For instance, EU Directive 2022/431, 12 which is an amendment of the Carcinogen, Mutagen (CM) Directive 2004/37/EC, 22 states that if a HMP cannot be replaced, then a closed system or other measures to reduce exposure should be used. In the US, the Occupational Health and Safety (OSHA) standards directive23,24 advises that the performance claims of marketed CSTDs should be evaluated, whereas the US Pharmacopeia (USP) General Chapter 800 (USP 800) 9 practice and quality standards highlights that until a published universal performance standard for evaluation of CSTD containment is available, users should carefully evaluate the performance claims associated with available CSTDs based on independent, peer-reviewed studies and demonstrated contamination reduction. USP 800 is the only standard that mandates CSTD use in HMP administration. 9 USP 800 is adjunct to USP 795 (general compounding – nonsterile preparations) and USP 797 (pharmaceutical compounding – sterile preparations) which are standards that may be federally enforced. In states that have adopted USP 800, the state boards and accrediting bodies can enforce the sections related to HMP compounding.9,25,26 Regarding guidelines, recommendations on the use of CSTDs vary. For example, NIOSH guidelines recommend using devices such as CSTDs, glove bags, and needleless systems when transferring HMPs from primary packaging (e.g., vials) to dosing equipment (infusion bags, bottles, or pumps). 1 In contrast, the European Agency for Safety and Health at Work (EU-OSHA) highlights that there is differing information in the literature on the effectiveness of CSTDs for reducing the risks in the preparation of HMPs and that it is the decision of the management/staff as to whether CSTDs are to be used. 27 In addition, the European Biosafety Network highlights that CSTDs are effective in reducing exposure to HMPs and should be used in the whole life cycle of HMPs. 6
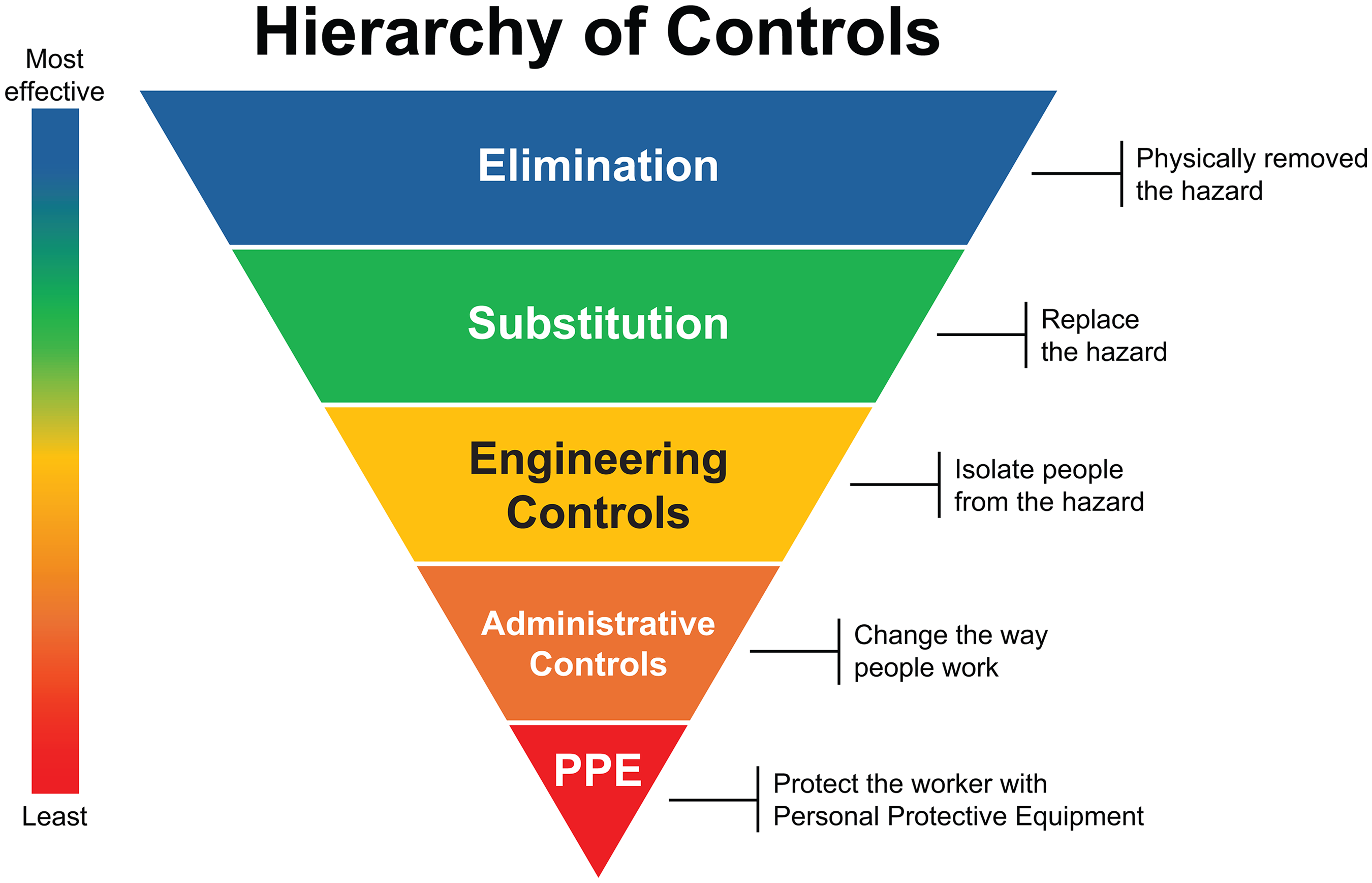
The use of CSTDs is recommended in guidelines published in Australia,16,28,29 Japan, 15 the US,1,9,30,31 European countries,6,27 and in global guidelines (specifically the International Society of Oncology Pharmacy Practitioners guidelines) (Supplementary Table 1). 32 Among the EU countries that have specific guidance, including Belgium, France, Hungary, Israel, Spain, Austria, Italy, and Sweden, only Italy have mandated the use of CSTDs.33–37 The NIOSH guidelines also outline the universal hierarchy of controls, which contain practical and effective methods to reduce exposure to help protect workers and are applicable in all workplace settings (Figure 2). 38 All of the prevention measures outlined in EU Directive 2022/431 replicate the universal hierarchy of controls.6,38
There is a general lack of consistency in the definition of a CSTD
A CSTD has been formally defined in the US and EU, but overall, there is a lack of consistency in how CSTDs are defined globally. In the US, NIOSH defines a CSTD as a drug transfer device that mechanically prohibits the transfer of environmental contaminants into the system and the escape of the HMP or vapour concentrations outside the system. 1 Whereas the FDA product code for CSTDs (the FDA ONB code) defines CSTDs as products that reconstitute and transfer antineoplastic and other HMPs in the healthcare setting and is indicated to reduce the exposure of healthcare personnel to chemotherapy agents. 39 In the EU, the European Commission defines a CSTD as a medicine transfer device that mechanically prohibits the transfer of environmental contaminants into the system and the escape of the HMP or vapour concentrations outside the system. 27
There are various types of CSTDs, all designed to protect users during the medication preparation and use preparation process
CSTDs protect HCPs during the medication use or preparation process by ensuring secure, leak-proof, connections when syringes are linked to drug vials, intravenous (IV) bags, IV administration sets, or infusion lines. 40 They consist of a vial adaptor, syringe adaptor, and IV bag (spike port) adapter (Figure 3). Depending on their design, they minimize or eliminate the risk of contamination of the HMPs or the risk of the HMPs escaping as vapours, aerosols, or liquids. 40
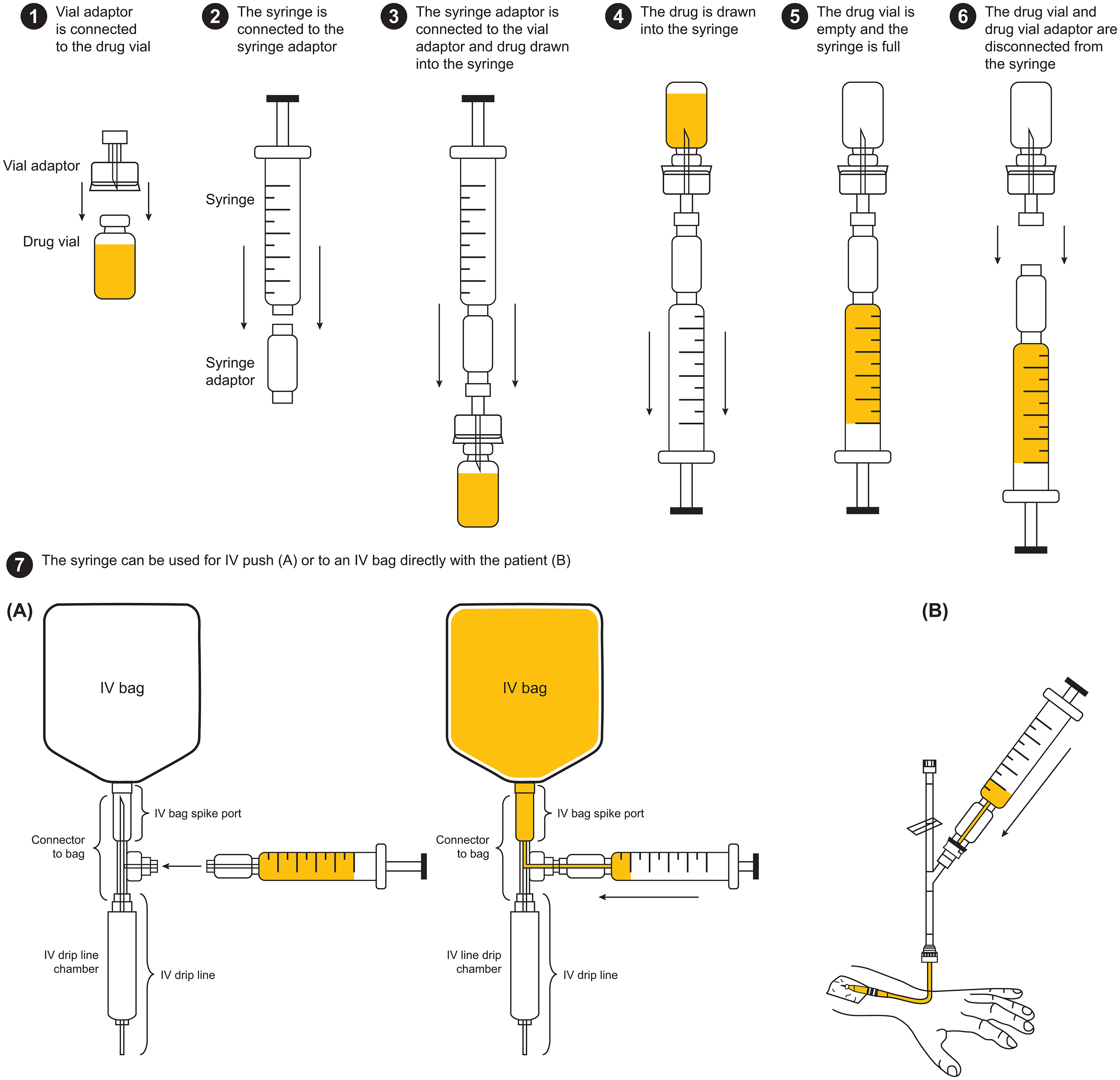
General components of a closed system transfer device and how they are used.
There are two primary types of CSTDs: air filtration and physical barrier systems (Figure 4).40–43 Air-filtration CSTDs clean the air that passes between the environment and the HMP vial. 43 This aims to prevent the unintended release of HMPs into the surrounding environment or the intake of environmental contaminants into the sterile drug pathway. 43 Some air filtration CSTDs have an additional activated carbon filter. 44 The physical barrier types use a closed barrier to block release into the surrounding environment or the intake of environmental contaminants into the sterile drug pathway. 43 CSTDs also vary in several design aspects, including closing mechanism, component types, intended end use, and fluid containment mechanism.45,46 Since the FDA ONB medical device product code was created in January 2013, a wide range of CSTDs have been developed and subsequently cleared by the FDA.47,48
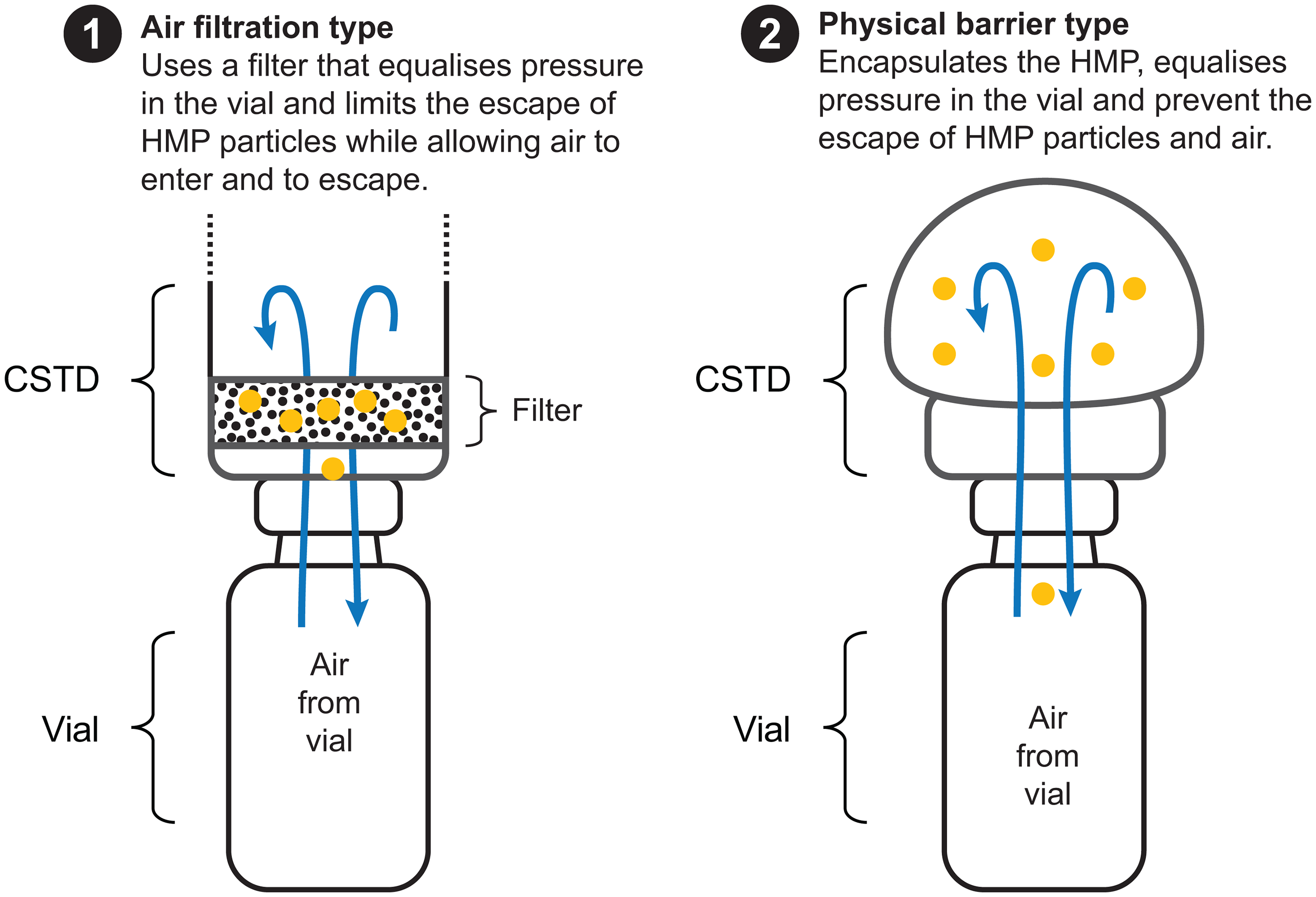
The two main types of closed system transfer device.
Limitations of available standard protocols to test CSTDs
When selecting which CSTD to use, it is important to recognize that these devices differ in design characteristics and protective efficacy, with varying levels of evidence supporting their use. Currently available CSTDs use different mechanisms to achieve ‘closedness’ and may therefore not be directly comparable. 49 Additionally, different methods of assessment have been used to support claims of being a closed system. 49 Accordingly, NIOSH developed a test protocol for testing CSTDs − the NIOSH 2015 protocol − in response to the need for an independent testing method for barrier-type CSTD performance.40,50 This protocol is designed to test containment performance of physical-barrier type CSTDs using 70% isopropyl alcohol (IPA) as a surrogate for HMPs, but it is not designed to test air-filtering type CSTDs. 41 Using this protocol, a maximum leak performance threshold of 1.0 parts per million of IPA vapour was established as a feasible performance benchmark for CSTDs. 50 However, this test cannot distinguish between different types of leakage. i.e., aerosols, liquids, or vapour. 43 Also, the NIOSH protocol does not use chemotherapy drugs but uses a surrogate, which does not resemble the specific characteristics of active hazardous pharmaceuticals such as molecular size and a unique vapour pressure. 44 At present, this is the only globally-accepted CSTD containment testing protocol until further standards are developed. In response to feedback from industry experts, researchers and manufacturers, NIOSH drafted an improved protocol with different challenge agents in attempt to allow testing of all types of CSTDs.40,43 This draft unified CSTD test protocol is under development by NIOSH but has not been finalized.41,51
To further complicate matters, there is currently no standard for comparing the performance of CSTDs that have been cleared by the FDA in the US or recognized under the Conformité Européene (CE) marking system in the EU. In the US, CSTDs that receive FDA 510(k) clearance are classified under the ONB product code, indicating that they meet the NIOSH definition of a CSTD.52,53 However, the ONB product code does not include any performance standards and there is no unified testing protocol, making comparison of different devices challenging. 53 In addition, the FDA does not test any of the CSTDs; rather, they rely on the manufacturers’ descriptions. Similarly, in Europe, CSTDs require a CE mark which indicates that a product has been assessed by the manufacturer and is deemed to meet EU safety, health, and environmental protection requirements.54,55 Ultimately, the absence of independent testing and standards, not covered by the ONB code or CE mark for comparing performance across CSTDs, means that users are reliant on manufacturer-provided data and published studies to compare efficacy of air-filtering CSTDs.
Many studies have evaluated the performance of CSTDs (Table 2). Given this is a rapidly evolving field and some earlier CSTDs are no longer available, we have focused on studies published over the past six years.40,44,56–63 Of the five identified studies comparing the performance of CSTDs using the NIOSH 2015 protocol,40,56–59 one study by Armistead et al. using the NIOSH 2015 protocol reported that the PhaSeal or PhaSeal Optima CSTD demonstrated full or partial effectiveness in IPA vapour containment (confidence interval [CI] = 0.00 or 0.00–0.17 ppm, respectively) whereas other comparator CSTDs (Equashield, Yukon Arisure, ICU Medical Chemolock) demonstrated CSTD IPA leakage (CI = 0.08–3.47, 1.69–2.89, and >5 ppm, respectively). 40 A second study by Forshay et al. using the NIOSH 2015 protocol reported that two out of six CSTDs, PhaSeal and Equashield, adequately contained IPA vapor and passed predefined testing criteria. 56 A third study by Skildaz et al. using a modified NIOSH protocol reported that three CSTDs (PhaSeal, ChemoLock, Equashield) had equivalent performance in containing IPA. 57 A fourth study, by Halloush et al. using a modified NIOSH protocol reported that Equashield, HALO, and PhaSeal devices met the protocol quantifiable performance threshold in containing IPA and were truly closed systems. 58 A fifth study by Tang et al. using a modified NIOSH protocol reported that unnamed CSTDs significantly reduced cyclophosphamide contamination exposure versus a traditional syringe technique. 59 All remaining studies, which used alternative techniques to the NIOSH 2015 protocol to assess CSTD performance, demonstrated that CSTDs significantly reduced surface contamination.42,44,60–64
Key studies evaluating the performance of CSTDs over the last six years. a
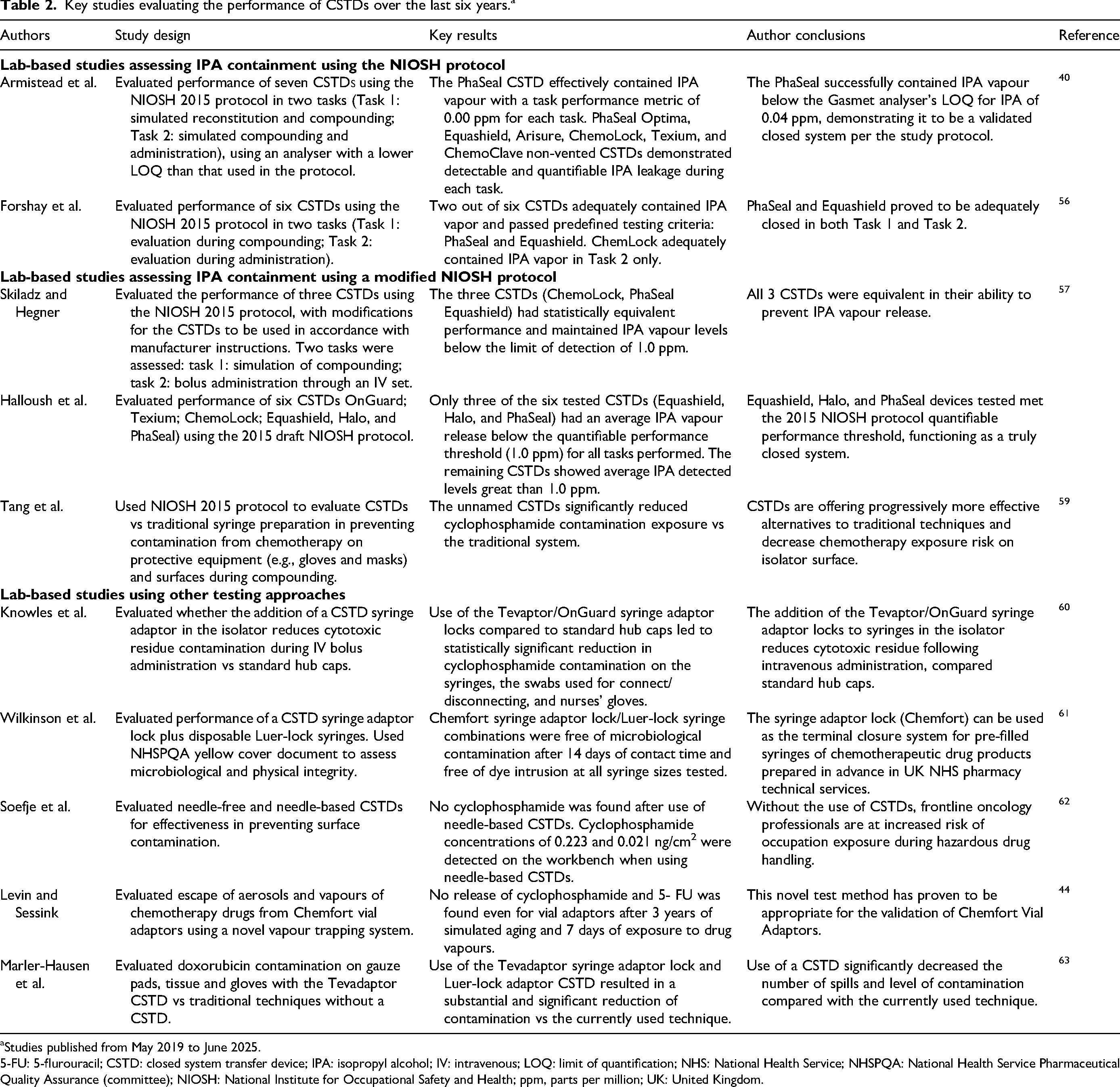
Studies published from May 2019 to June 2025.
5-FU: 5-flurouracil; CSTD: closed system transfer device; IPA: isopropyl alcohol; IV: intravenous; LOQ: limit of quantification; NHS: National Health Service; NHSPQA: National Health Service Pharmaceutical Quality Assurance (committee); NIOSH: National Institute for Occupational Safety and Health; ppm, parts per million; UK: United Kingdom.
In 2018, a systematic review by Gurusamy et al. assessing the effectiveness of CSTDs concluded that there was no evidence to support or refute the routine use of CSTDs in addition to safe handling of HMPs, as there was no evidence of differences in exposure between CSTDs plus safe handling versus safe handling alone. 65 However, this systematic review attracted criticisms based on study limitations, which resulted in revisions to the systematic review. 66 Despite the implemented revisions, the initial criticisms were considered to remain valid. 67 Limitations of the systematic review included that the studies were not controlled for the type of CSTD used and only two studies included nurses as subjects when nurses were the target population.66,67 The systematic review claimed to find no difference in health outcomes when none of the included studies actually measured health outcomes.66,67 Also, safe handling alone was poorly defined and not consistent across studies; therefore like was not compared with like. Also, the systematic review did not include any studies performed in a laboratory setting (e.g., in vitro or mechanistic studies) to validate the effectiveness of CSTDs.66,67 Finally, most of the CSTDs evaluated are no longer on the market, and those that are still available have been upgraded and improved since.
Aside from assessment of performance, the decision on which CSTD to use may also be driven by other factors, such as whether a drug is listed as a HMP (e.g., the drug is included in the EU HMP, NIOSH list, or other national guidelines), health authority decision-making, information on the drug manufacturer's product label, local opinions such as nurse and pharmacy leaders, how time-consuming the CSTD is to use, training needs, lack of information on compatibility of a CSTD with the drug product, and costs to the institution.46,49,68 There is considerable variability in influencing factors on decision-making at all levels of procurement. Also, decisions on which CSTD to use are made locally in some countries and nationally in others. For example, the Italian Society of Hospital Pharmacy (SIFO) provides national recommendations, 69 whereas in the US and United Kingdom (UK), all decisions are made locally.70,71 It would therefore be helpful to have a focus on safety information at a national level through governing bodies such as the National Health Service England in the UK to allow for consistent local decisions. All of these factors highlight the need for a decision to be made at a national and local level on which CSTDs are recommended.
CSTD use differs across regions and countries
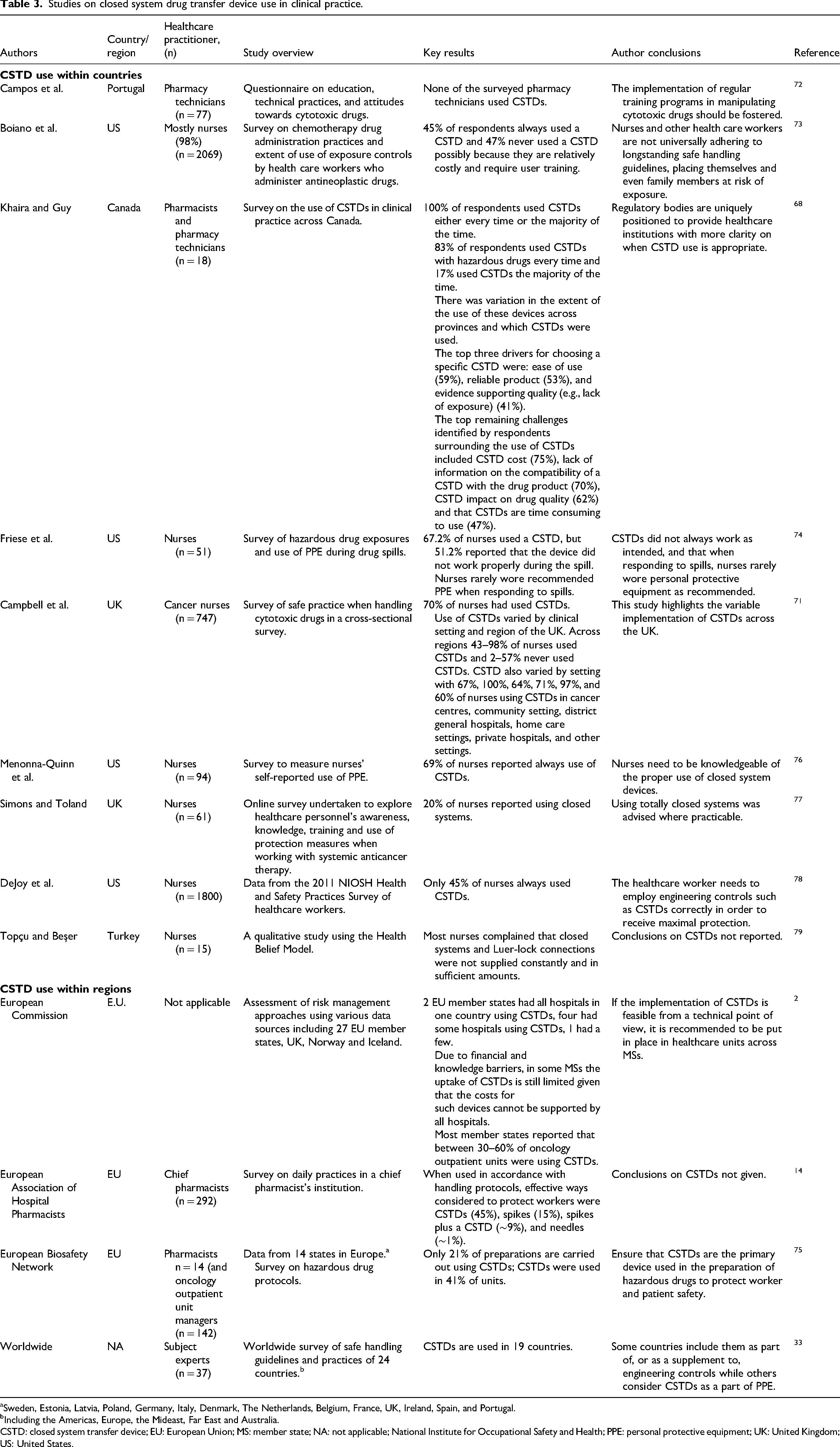
To date, few studies have examined how frequently CSTDs are used in clinical practice. In addition, the lack of a clear definition of CSTDs make interpreting studies and even selecting which CSTDs to compare challenging. Existing studies report variability in CSTD use within countries, with reported usage rates among pharmacists, pharmacy technicians, and nurses in these studies ranging from 0 to 100% during the preparation or administration of a HMP (Table 3).2,14,33,68,71–79 Across studies, frequently cited factors affecting CSTD use were the cost of devices and lack of training. Notably, a survey of CSTD use by oncology pharmacists and pharmacy technicians in clinical practice in Canada, who were frequent CSTD users, reported variation in the extent of use across provinces. 68 Factors that may influence CSTD use include differences in practice settings (academic vs community settings), differences in the definition of HMPs across institutions, years of user experience, and stakeholder influence (influence by the local health authority, the FDA, NIOSH). 68 In the UK, there is evidence of variation by region for nurses using CSTDs, ranging from 43 to 98%; overall, 30% of nurses in the UK did not use CSTDs at all. 71 This variation has been attributed to local decision making. In Scotland in 2019, variable use of CSTDs across cancer centres and National Health Service boards was reported, which was attributed to varying local guidelines and local CSTD preferences based on ease of use, compatibility with other equipment (e.g., infusion pumps), and features of the individual components of the CSTD. 80 In contrast, it is mandatory to use CSTDs in Italy. 36 Additionally, in the US, hospitals must use CSTDs in administration and they should be used in preparation of antineoplastics to comply with USP 800; otherwise, they can be cited and fined. 70 However, there are still issues of non-compliance, with a survey of pharmacy directors in the US in 2021 reporting that although 31% of pharmacy directors reported compliance with USP 800, 63% indicated they were not yet fully compliant but were working on compliance, and 6% were not fully compliant but were not working on compliance. 81 Of the hospitals that were non-compliant, 17.6% of pharmacies reported gaps in the use of CSTDs.
Studies on closed system drug transfer device use in clinical practice.
Sweden, Estonia, Latvia, Poland, Germany, Italy, Denmark, The Netherlands, Belgium, France, UK, Ireland, Spain, and Portugal.
Including the Americas, Europe, the Mideast, Far East and Australia.
CSTD: closed system transfer device; EU: European Union; MS: member state; NA: not applicable; National Institute for Occupational Safety and Health; PPE: personal protective equipment; UK: United Kingdom; US: United States.
CSTD use also varies across worldwide regions (Table 3). An analysis of EU member state authorities reported that between 30% and 60% of member state authorities oncology outpatient units were using CSTDs. 2 A European study of 27 EU member states plus the UK, Norway, Iceland, and Lichtenstein in 2021 reported that CSTDs were used in Denmark and Estonia in 100% of oncology outpatient units within the country, whereas in Latvia and Ireland, none of the oncology outpatient units were using CSTDs. 2 In the same study, two EU member states had all hospitals in one country using CSTDs, four had some hospitals using CSTDs, and one had a few hospitals using CSTDs. 2 In some member states, limited uptake was due to financial and knowledge barriers. A survey by the European Association of Hospital Pharmacists (EAHP) also demonstrated varying thoughts on the use of CSTDs by chief pharmacists in daily practice, with 45% considering a CSTD an effective way to protect HCPs from potential exposure to HMPs, while 15% favoured spikes, ∼9% preferred spikes plus a CSTD, and ∼1% preferred needles. 14 Additionally, a worldwide survey of guidelines and current practices for safe handling of antineoplastic and other HMPs in 24 countries in 2019 reported considerable variation in the recommended position of CSTDs in the hierarchy of protection controls and in which countries CSTDs were used. 33 According to the survey, CSTDs were used in 19 of 24 countries. Also, in some countries such as the US, CSTDs are considered supplemental engineering controls. Whereas, in Brazil, Chile, Japan, and the UK, they are designated as personal protective equipment (PPE). 33
A potential explanation for the variation in CSTD use2,33 could be the challenges faced by HCPs in some countries, such as the UK, where determining the most suitable and practical CSTD remains a complex and unresolved issue. 71 This may be due to individualized and local care group implementation processes. Although patient safety is consistently prioritized by policymakers and budget holders, worker safety often receives less attention. In reality, safety should be comprehensive, encompassing both patients and HCPs alike.23,82 Further local and global studies are therefore needed to understand how CSTDs are being used. Such studies should include criteria for decisions to use/not use CSTDs, categories of drugs that require CSTDs, and where CSTDs are applied in the clinical setting, e.g., in the hospital pharmacy or on the ward.
Closed system drug transfer device work practices can make a difference on how effective they are in preventing exposure to HMPs
Although CSTDs are designed to avoid contamination from and to HMPs and to shield HCPs during the preparation and administration of HMPs, evidence suggests that they are sometimes being used incorrectly in terms of where the drugs are being prepared and by whom. For example, a study of cancer nurses in Europe reported that 64% of oncology nurses reported that they never prepared hazardous cancer drugs at their workplaces, while 21% reported that it very rarely happened, 2% of nurses reported monthly occurrences, while 12% indicated that it happened weekly. 83 Similarly, a study in Spain found that nurses using CSTDs were still engaged in spiking infusion bags and attaching CSTDs to syringes just before administration, which is an identified exposure risk. 84 These practices pose a risk for exposure to HMPs.
To effectively integrate CSTDs within clinical practice, healthcare organizations should strive to eliminate exposure risks through comprehensive workflow integration, beginning at the pharmacy and continuing through to administration units through interdisciplinary communication and education. This approach will minimize risk at the earliest stage and ensure protection of HCPs. 71 Use of occupational exposure registries, such as that developed by the Hazardous Drug Safety Center in the United States could also help raise awareness of the extent of exposure to HMPs. 85 A combination of education and appropriate safety inductions, including use of CSTDs will help ensure HCP safety.
Use of biological drugs and CSTDs
Biological drugs are a broad class of complex medicines from living sources (e.g., cells, proteins, sugars) which include hormones, enzymes, blood-derived products, sera, and vaccines, immunoglobulins, allergens, and monoclonal antibodies (Supplementary Table 2).86–90 Biotechnological drugs, a subset of biological medicines, are produced using advanced techniques such as recombinant DNA technology, controlled gene expression in prokaryotic and eukaryotic cells, and hybridoma technology for monoclonal antibody production.86,87 Unlike chemically synthesized drugs, biological medicines consist of large, structurally complex molecules with unique stability and impurity profiles.86,87 Their production relies on living systems, which introduces variability, such as differences in glycosylation, that can affect immunogenicity. This complexity makes characterization challenging and closely ties the product to its manufacturing process, leading to the principle that “the product is the process”.86,87 The increasing use of biological drugs has significantly increased their use across therapeutic areas. 91 As outlined by the Clinical Oncology Society of Australia, these products are often perceived as being less hazardous than cytotoxics but they do require assessment for clinical risk to the patient and occupational exposure risk to staff handling the molecule. 91 These risk assessment processes ensure that the appropriate prevention strategies and protective measures are implemented. In Europe, close attention should be paid to Directive 98/24/EC (on the protection of workers from risks related to chemical agents), 92 Directive 2022/431 (on carcinogens, mutagens, and reprotoxic substances, and an amendment of Directive 2004/37/EC),12,22 and EC Regulation EC No 1907/2006 (on the Registration, Evaluation, Authorisation, and Restriction of Chemicals [REACH]). 93 These regulations collectively require that exposure to hazardous substances, which include biological drugs, be avoided wherever possible. This precautionary approach underscores the importance of proactive risk management and the implementation of protective measures, as the full extent of potential harm from biological drugs is not yet fully understood.
Cost-effectiveness of CSTDs
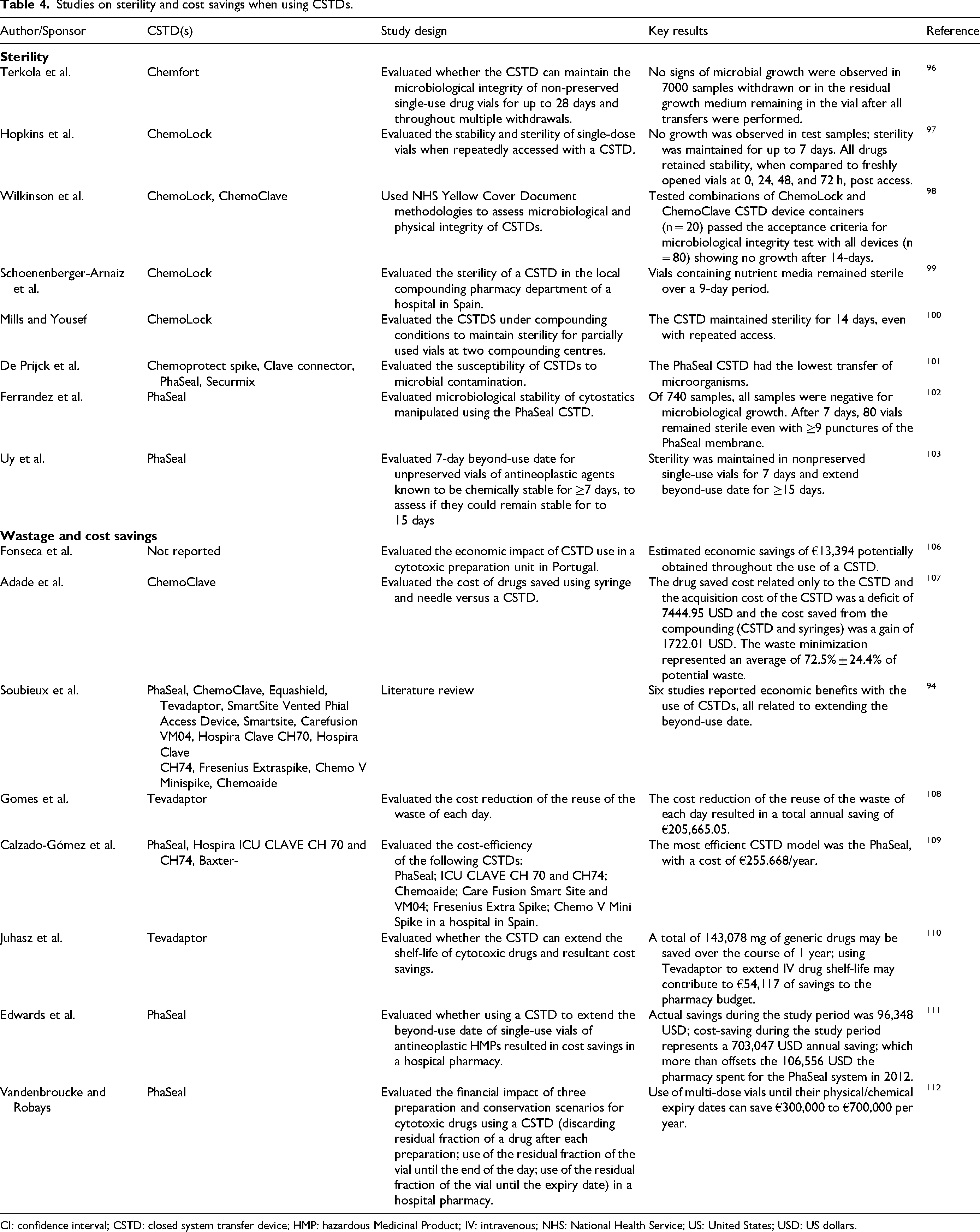
A key impediment to the use of CSTDs is their cost. 94 The traditional method of transferring drugs from vials to infusion bags or sets, using a needle syringe or spike, often leaves residual HMP in the vial. European Medicines Agency (EMA) and 95 USP 797 guidelines 25 limit the use of open sterile products to 12 − 24 h, often resulting in significant drug waste. However, using CSTDs can guarantee sterility of the HMP for at least seven days, extending the beyond-use date (Table 4).96–104 This ensures full utilization of HMPs, mitigates shortages and generates substantial savings, especially for high-cost medications such as monoclonal antibodies which can offset the cost of the CSTD itself (Table 4).94,102,105–112 Overall, this supports the financial case for the implementation of CSTDs.
Studies on sterility and cost savings when using CSTDs.
CI: confidence interval; CSTD: closed system transfer device; HMP: hazardous Medicinal Product; IV: intravenous; NHS: National Health Service; US: United States; USD: US dollars.
Need to adhere to directives and guidelines, particularly outside of the US
As described earlier, although the Gurusamy et al. systematic review 65 has potentially caused confusion, there is substantial evidence supporting the effectiveness of CSTDs in protecting HCPs from exposure to HMPs, while also demonstrating the risks of exposure and its associated sequelae.1,2,40,44,57–63,113 In addition, various guidelines, standards, and directives exist that, in principle, should enable consistent and comprehensive implementation of CSTDs across all stages of HMP handling − at the local, regional, and global levels. However, adherence is inconsistent, particularly when comparing practice between the EU and US.
The US has set a benchmark in implementing contamination testing and ensuring the use of CSTDs. A recent survey reported that 85% of facilities in the US in 2025 are using CSTDs for administration of HMPs. 114 Additionally, 51% of pharmacies are performing wipe analysis for surface contamination of HMPs. 115 USP 800 states that environmental wipe sampling for HMP surface residue should be performed routinely, at least every six months, and that CSTDs must be used when administering antineoplastic HMPs, when the dosage form allows (typically by injection or infusion). 9 Many state boards of pharmacies and other regulators can cite organizations for failure to comply with USP 800. In addition, the Joint Commission, which has accredited ∼15,000 healthcare organizations in the US, 116 also surveys healthcare organizations for compliance with USP 795 and USP 797. 117 Failure to comply can result in loss of accreditation, which is tied to reimbursement by Medicare. Improving HCP safety globally will require other countries to adopt and implement similar standards to those used in the US. In the EU, existing directives and guidelines, such as Directive 2022/431, already recommend regular contamination testing and health surveillance. Therefore, consistent adherence, rather than revisions to EU directives or guidelines, should be the main priority. This should be guided by the hierarchy of controls, which emphasize the use of better engineering controls. Adoption of CSTDs is likely to increase as contamination testing would be expected to show the status of current exposure of HCPs to HMPs, as has already been demonstrated in multiple studies to date.5,118–120
A joint statement from the European Association for Hospital Pharmacists (EAHP) and the European Society of Oncology Pharmacy (ESOP), in 2021, 121 strongly recommended support at the European level, ensuring that HCPs working with HMPs are familiar with the associated risks and the necessary precautions. While they do not specify CSTDs, they suggested that consistency was needed in the handling of HMPs. They also emphasized the importance of continuous education of HCPs, the use of appropriate personal protective and drug handling equipment, and standardization of training. 121 In the US, personnel training is one of the standards in USP 800 9 that accreditors, state boards of pharmacy and Medicare surveyors evaluate.19,122 This must occur based on job function and should address exposure risks, relevant policies and the correct use of PPE and equipment. Pharmacy and nursing professional organizations in the US agree with this approach. 24 The benefits of training have been seen in Europe, 27 whereby training on cleaning practices for HMPs substantially reduced contamination in the workplace but did not completely eliminate HMPs.118,123
Taken together, established directives and guidelines demonstrate that, with coordinated EU-level training, consistent implementation across European countries is achievable, making a global, unified approach to HMP management both realistic and within reach, as evidenced by what has been achieved in the US.
Proposing a roadmap for global alignment in HMP management with CSTDs
Based on the evidence discussed in this review, there is a clear unmet need for global alignment on HMP management with CSTDs and when to use CSTDs across regions and countries. Specific recommended actions are shown in the roadmap in Figure 5. These include the need for research on the latest guidance worldwide, including details of guidance by country and level of guidance (i.e., high-level to low-level guidance). On a local level, there should be interprofessional collaboration between users of CSTDs (nurses and pharmacy personnel) when deciding which CSTD to select. In addition, at the local level, good work practices related to implementation and CSTD use should be established, training should be provided by the CSTD manufacturers and clear policies and procedures for using CSTDs are needed. CSTD users should also be involved in determining the adequate supply of CSTDs to a workplace based on how much they are used. On a national level, there should be action by professional organizations to advocate for members’ HMP safety and regional or national resourcing to reduce costs and ensure daily availability of CSTDs (e.g., to avoid running out of CSTDs due to the correct amount not being procured), as local resourcing makes CSTD procurement more expensive. This highlights how safety should not be tied to costs; rather, decisions should be based on which CSTD to use based on specific uses, routes of administration, specific patient populations, and ease of use HMPs, in a specific setting, determined by being listed in the EU HMP or NIOSH lists. Ideally, efficacy testing will guide decisions in the future. At both local and national levels, countries and states may be able to help economically through tax breaks, while medical device companies may be able to apply discounts through centralized purchasing. At a national level, responsible bodies must adopt clear positions on which CSTDs they recommend; this also applies at local level. Also, clear guidance on implementation and maintenance of CSTDs, and which CSTD is more useful for specific applications is needed at national level. Coordinated training across HCP professions is also needed. On an international level, there is the need for the development and wide acceptance of standardized testing protocols to assess the containment effectiveness of both filter-type and barrier-type CSTDs. This should be accompanied with the identification of inconsistencies and shortcomings in current directives, national legislations, and guidelines, with the ultimate goal of achieving alignment and delivering consistent messaging to HCPs and above all ensuring HCP safety against HMP exposure.

Steps recommended to ensure global alignment in hazardous drug management.
Summary
CSTD use has been successfully implemented in the US but remains inconsistent in the EU. If established directives and guidelines can be adhered to in European countries through training led at an EU level, a global, unified approach to CSTD use is feasible. A roadmap is proposed at the local, national and international levels to help promote a unified approach to CSTD testing worldwide. The suggested unified approach is proposed to include training, action by professional societies, clear stances from professional bodies on CSTD recommendations, development and wider use of testing protocols, and identification of current variables in directives and guidelines, as well as a joint alignment on these. Implementation of this roadmap will improve the safety of HCPs exposed to HMPs.
Supplemental Material
sj-docx-1-opp-10.1177_10781552261434870 - Supplemental material for Development of a unified approach to hazardous medicinal product management using closed system drug transfer devices
Supplemental material, sj-docx-1-opp-10.1177_10781552261434870 for Development of a unified approach to hazardous medicinal product management using closed system drug transfer devices by Paolo Amari, Karen Campbell, Mark Henry, Martha Polovich and Paul Sessink in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgments
Medical writing support was provided by Mark English, PhD, and editorial review was provided by Celia Diez de los Rios, PhD, both of COR2ED, Basel, Switzerland, supported by an educational grant from BD, Sàrl, Switzerland.
Ethical approval and informed consent statements
Not applicable.
Author contribution statements
Paolo Amari: Conceptualization, Writing – Original Draft, Writing – Review and Editing.
Karen Campbell: Conceptualization, Writing – Original Draft, Writing – Review and Editing.
Mark Henry: Conceptualization, Writing – Original Draft, Writing – Review and Editing.
Marty Polovich: Conceptualization, Writing – Original Draft, Writing – Review and Editing.
Paul Sessink: Conceptualization, Writing – Original Draft, Writing – Review and Editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MH has received consultancy fees from Pfizer and Takeda; PS, has received consultancy fees from Gasgon Medical Ireland, Simplivia Healthcare Israel, ICU Medical USA, RPA Europa Czech Republic, Equashield Medical Israel, and Becton Dickinson and Company USA and UK; KC, has received consultancy fees from ICU Medical USA, MP and PA declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Not applicable.
Supplemental material
Supplemental material for this article is available online.
