Abstract
Introduction
The European Biosafety Network recommends that cytotoxic drug surface contamination in pharmacies and patient wards not exceed 0.1 ng/cm2. Among other mitigations, closed system transfer devices (CSTDs) are recommended in the US, Europe, and UK for the reduction of surface contamination. Although in the UK CSTDs are not part of standard cytotoxic preparation procedures in isolators, CSTD syringe adaptors are recommended for use with syringes for intravenous administration. We investigated whether the addition of a CSTD syringe adaptor in the isolator reduces cytotoxic residue contamination during intravenous bolus administration.
Methods
Twenty-five cyclophosphamide syringes were prepared with hub caps and twenty-five with SIMPLIVIA® (formerly Tevadaptor/OnGuard®) Syringe Adaptor Locks (SALs) in an isolator. Surface contamination of syringes, gauze pads placed at the administration site, and nurses’ gloves was compared between the practice of connecting hub caps in the isolator and removing them in the ward versus connecting SALs in the isolator during preparation. Cyclophosphamide contamination was quantified by liquid-chromatography mass spectrometry.
Results
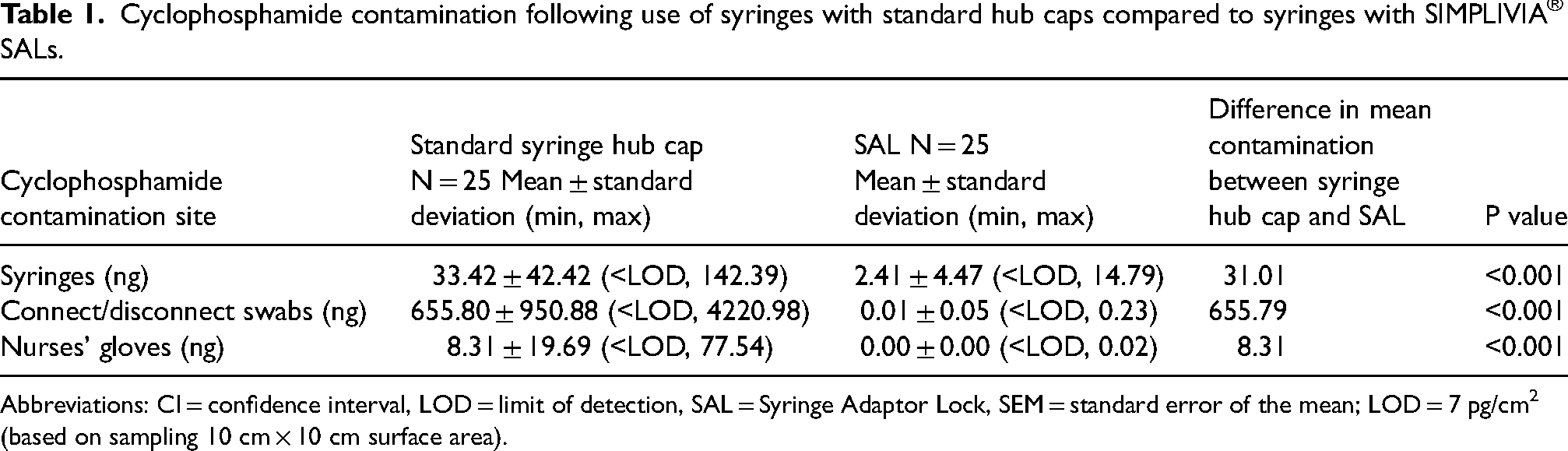
Use of SALs compared to standard hub caps led to statistically significant reduction in cyclophosphamide contamination on the syringes (2.41 ± 0.89 ng versus 33.42 ± 8.48 ng, p = 0.0007), the swabs used for connect/disconnect (0.01372 ± 0.01 ng versus 655.80 ± 190.18 ng, p = 0.001), and nurses’ gloves (0.0009 ± 0.0009 ng versus 8.31 ± 3.94 ng, p = 0.04). When hub caps were used, 48% of syringes, 76% of gauze pads, and 8% of gloves exceeded the recommended limit of 0.1 ng/cm2, while no samples exceeded this limit with SAL.
Conclusions
The implementation of CSTD syringe adaptors was beneficial for reducing cytotoxic drug exposure to nurses administering intravenous syringes.
Introduction
Cytotoxic drugs interfere with the cell cycle, triggering apoptosis in rapidly dividing cells. 1 Although this mechanism of action is crucial for fighting cancer, it also presents substantial health risks to individuals exposed to these compounds as cytotoxic drugs have the potential to cause harm to health if they are ingested, inhaled or absorbed through contact with the skin or other body membranes.2–9 Such exposure can occur throughout the various phases of drug handling, including their preparation, administration, transport, disposal, or when cleaning cytotoxic drug spills or handling patient waste.2,4,5,7,8,10–13 Anyone working with patients receiving cytotoxic drugs is at risk of exposure, including pharmacy, nursing and medical staff.2,4,8,12,13
The consequences of unintentional exposure to cytotoxic drugs can be severe, as they have been linked to acute and chronic toxicities, carcinogenicity, mutagenicity, and reproductive (CMR) toxicities.14–16 Healthcare workers who are exposed to cytotoxic drugs are at a higher risk of developing a range of health problems, including local effects (e.g., eye or skin irritation), nausea, dizziness, headaches and acute toxicity. The likelihood that a healthcare worker will experience adverse effects from cytotoxic drugs increases with the amount of drug, frequency of exposure, and lack of proper work practices.5,8 In the long term, they may also experience more serious health concerns and an increased risk for developing cancer.17–20
Due to the importance of this issue in occupational health, healthcare institutions and regulatory authorities have issued guidelines and recommendations to reduce the risks associated with the handling of cytotoxic drugs.2–5,8–12,21–29 Strategies for minimizing exposure include implementing engineering controls, such as isolators and closed system transfer devices (CSTDs). Administrative controls are also required, which include staff training, standard operating procedures, good hygiene measures, monitoring exposure in the workplace, health surveillance, safe management of cytotoxic spills and safe disposal of cytotoxic waste.2–5,8,10–12,22,23,27 Additionally, the use of personal protective equipment is recommended.2–5,8,11,12,22,23,26,27
The dual objectives of CSTDs are to preserve product sterility and to safeguard healthcare professionals from potential exposure to CMR substances.30–32 These devices employ various pressure equalization and containment technologies to facilitate the process of reconstituting drug powder and transferring a drug solution into empty or pre-filled containers, such as infusion bags, bottles or syringes, 33 while maintaining a closed system. Therefore, they reduce cytotoxic residue contamination when used for the aseptic preparation of cytotoxic drugs.26,34–52 Studies have shown a decrease in the percentage of surface sampling wipes with detectable levels of cytotoxic drugs in the preparation area and administration area.26,34–47,49–52 Some of these studies have also shown a decrease in the percentage of workers who had detectable levels of cytotoxic drugs in their urine when using CSTDs.40,46,49,50
In the UK, CSTDs are not part of standard cytotoxic preparation procedures in isolators, but the National Health Services (NHS) Pharmaceutical Quality Assurance Committee recommends the use of CSTD syringe adaptors with syringes used for intravenous administration in order to reduce nurses’ risk of cytotoxic drug exposure. At University Hospitals Birmingham, the standard practice is to connect Luer caps in the isolator and remove them for administration. 11
Simplivia® CSTD systems (Simplivia Healthcare, Ltd), use a unique Toxi-Guard® technology that allows automatic pressure equalization while maintaining a mechanical drug-binding barrier against microbial ingress and hazardous drug escape. Studies that have evaluated the physical and chemical compatibility of this CSTD system, as well as its syringe integrity, have shown that patient safety is not compromised when SIMPLIVIA® Syringe Adaptor Locks (SALs) are added in the isolator as part of the final container for the storage of certain cytotoxic drugs, including cyclophosphamide, cytarabine, ifosfamide, vinblastine, vincristine doxorubicin, epirubicin, 5-fluorouracil, methotrexate, carboplatin, cisplatin, etoposide, fludarabine, gemcitabine, irinotecan and oxaliplatin.53–56 SALs lock onto Luer lock syringes, such that once the two components are connected, they cannot be disconnected. 57 However, no studies to date have demonstrated the benefit of adding the SAL in the isolator to syringes for subsequent intravenous administration. One study showed an overall reduction in the level of doxorubicin contamination when SALs were added to syringes at ward level prior to intravenous bolus administration. 58 A significant reduction in doxorubicin contamination was observed during administration, mostly notably on the gauze pads, whereas reduction in contamination on the tissues or nurses’ gloves was not statistically significant. 58
We first confirmed the microbiological integrity of syringes connected to SALs for a period of 7 days, a prerequisite for the addition of SALs in the isolator. We then investigated whether such practice reduces cytotoxic residue contamination during intravenous bolus administration.
Materials and methods
Syringe integrity testing of the SALs attached to a Luer lock syringe
Three process validations were completed by three different operators to ensure the results obtained were not operator dependent. 59 During the validation, each operator aseptically prepared six different size syringes (1 mL, 3 mL, 5 mL, 10 mL, 20 mL and 50 mL; BD, Franklin Lakes, NJ, USA) in an isolator by drawing up tryptic soy broth into the syringe to the required volume (0.85 mL, 2.5 mL, 4.2 mL, 8.4 mL, 17 mL and 50 mL, respectively) as per standard operating procedures. A SAL was then added to each syringe before being removed from the isolator. To ensure the results obtained were applicable to all syringe sizes and to prevent the requirement for extrapolation or interpolation of the data between syringe sizes, each syringe size available for aseptic preparation in the aseptic unit was filled by each operator. The broth fill volumes in each syringe were chosen based on the guidance provided by the Pharmaceutical Aseptic Services Group (PASG) and the NHS Pharmaceutical Quality Assurance Committee, which recommends that syringes used as final containers should not be filled to more than 85% of their nominal capacity.60,61 The maximum fill volume was chosen for this validation, as it poses the greatest risk of microbial contamination due to flexing or bending of the extended plunger after filling.60,61 The results therefore represent worst case scenarios regarding fill volumes and can be interpolated to smaller volumes.
The syringes were then stored in the aseptic unit for 7 days. Each syringe was stored either at room temperature or in a refrigerator to mimic real-life practice in case the storage temperature influenced the syringes’ integrity when stored with the SAL. The 7-day storage period was chosen based on the maximum 7-day expiry at the time for aseptic units operating under Section 10 exemption of the Medicines Act 1968.
After the 7-day incubation period, the syringes were transferred for incubation and microbiological testing at an analytical laboratory (Quality Control NorthWest [QCNW], Stockport, England, UK). Each syringe was incubated at 20–25°C for 7 days followed by 30–35° for 7 days, before being tested for microbiological contamination. One syringe of each of the volumes 1 mL, 3 mL, 10 mL, 20 mL, and 50 mL containing tryptic soy broth inoculated with Staphylococcus aureus, Candida albicans, Bacillus subtilis, Aspergillus brasiliensis or Pseudomonas aerginosa species, respectively, and closed with a SAL comprised the positive control. This ensured the tryptic soy broth used in the validation would grow microorganisms if present. The microorganisms were chosen for fertility testing as per the Quality Assurance of Aseptic Preparation Services: Standards 2016, based on the common microorganisms expected to be found in the environment. 59 The incubation period complies with the NHS Pharmaceutical Quality Assurance Committee guidance on the Protocol for the Integrity Testing of Syringes and End of Session Broth Fill Technique for Sterility Assurance of Products Aseptically Prepared in Section 10 Units.61,62 The incubation temperature and duration ensured the growth of microorganisms and therefore, the validity of the results.
Cyclophosphamide syringe preparation
A total of 50 syringes containing cyclophosphamide (Baxter, Deerfield, IL, USA) for intravenous bolus administration were prepared in the aseptic unit. Of these, 25 were prepared with a standard syringe hub cap and 25 were prepared with a SAL. The sample size was chosen based on the average of 25 cyclophosphamide syringes administered a week as an intravenous bolus in the oncology day unit. The cyclophosphamide dose prepared was 20 mg/mL with the preparation volume ranging from 5 mL to 50 mL. This sample size allowed for a suitable range of doses, administration nurses and administration techniques.
Cyclophosphamide was chosen as the drug of choice for this study, as it is one of the most common drugs administered as an intravenous bolus in the oncology day unit. It was chosen over doxorubicin and epirubicin because it is a clear liquid. In theory, this makes spills/leaks less easily identifiable, reducing the likelihood of safe handling on administration – representing the worst-case scenario. Cyclophosphamide was chosen over fluorouracil as it has a lower limit of detection. 63 At QCNW, the cyclophosphamide limit of detection was 7 pg/cm2 compared to 5-fluorouracil at 340 pg/cm2. 63 This ensured more accurate results on whether cytotoxic residue is present in low quantities on the sampling points.
The BD Luer Lock syringes and hub caps were selected as the equipment for this study based on current practice within the aseptic unit. SIMPLIVIA® SALs were chosen as the closed system device for this study, as other units within the University Hospitals Birmingham NHS Foundation Trust currently use these SALs at ward level.
The cyclophosphamide syringes were aseptically prepared in a negative pressure isolator in the aseptic unit as per standard operating procedures. The doses were prepared in the most appropriate syringe size for the volume and had either a standard syringe hub cap or a SAL added. The syringe sizes were chosen based on the maximum 85% filling of nominal capacity, as per the guidance provided by the PASG and the NHS Pharmaceutical Quality Assurance Committee.60,61 The syringe was wiped down with an isopropyl alcohol-impregnated wipe before being removed from the isolator to remove any cytotoxic residue present on the syringe. The syringes were labelled, sealed in clear packaging, final checked, and then sealed in opaque packaging before being delivered to the day unit. This is standard practice to contain any spills and provide protection from light for stability.
Cyclophosphamide residue sampling
A total of 8 nurses administered cyclophosphamide to patients at the oncology unit. Twenty-six doses were administered with the syringe pump (13 in each group) and 24 were administered with a hand push (12 in each group). Before intravenous syringes were administered via the SAL and complementary Luer Lock Adaptor (Simplivia Healthcare, Ltd), training on the devices was provided to the nurse administering the treatment. The Luer Lock Adaptor is screwed onto the female Luer lock injection point of an IV set and never removed. The nurses were supervised during the administration of all cyclophosphamide intravenous syringes included in this study. Figure 1 illustrates the reversible closed connection between the SAL and the Luer Lock Adaptor.
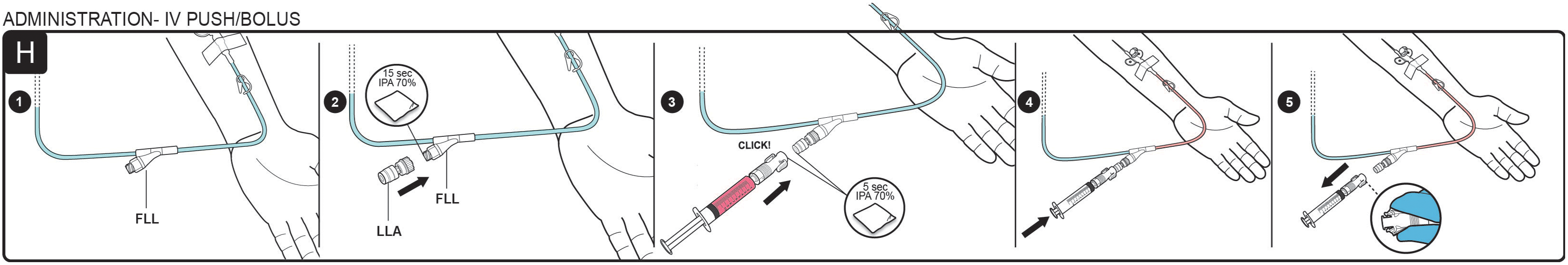
Instructions for use showing connection and disconnection of SAL and LLA for bolus injection. Reproduced with permission.
For the sampling process, the nurse wore a fresh pair of nitrile gloves and removed the cyclophosphamide syringe from the clear and opaque packaging. A swab was moistened with 0.25 mL methanol/water 50:50 using a plastic dropper. Prior to administration, the total surface area of the syringe, including the standard hub cap or SAL, was wiped with the moistened swab. The swab was inserted into the vial with the appropriate label.
Standard hub caps were removed from the syringe. The nurse held a swab under the Luer-connector of the syringe during connection and disconnection from the giving set. If administered as an intravenous bolus by hand, the swab was also held under the Luer-connector of the cyclophosphamide syringe during administration. If administered as an intravenous bolus via a syringe pump, the swab was stored in the vial during administration. The nurse swabbed the Luer-connectors on the syringe and giving-set post disconnection. The swab was then inserted into a vial for analysis.
The total surface area of the nurse's pair of nitrile gloves were also wiped with a swab moistened with 0.25 mL methanol/water 50:50, whilst the nurse was wearing them, and immediately prior to removing and discarding them. The swab was then inserted into a vial for analysis. Figure 2 illustrates the sampling process.
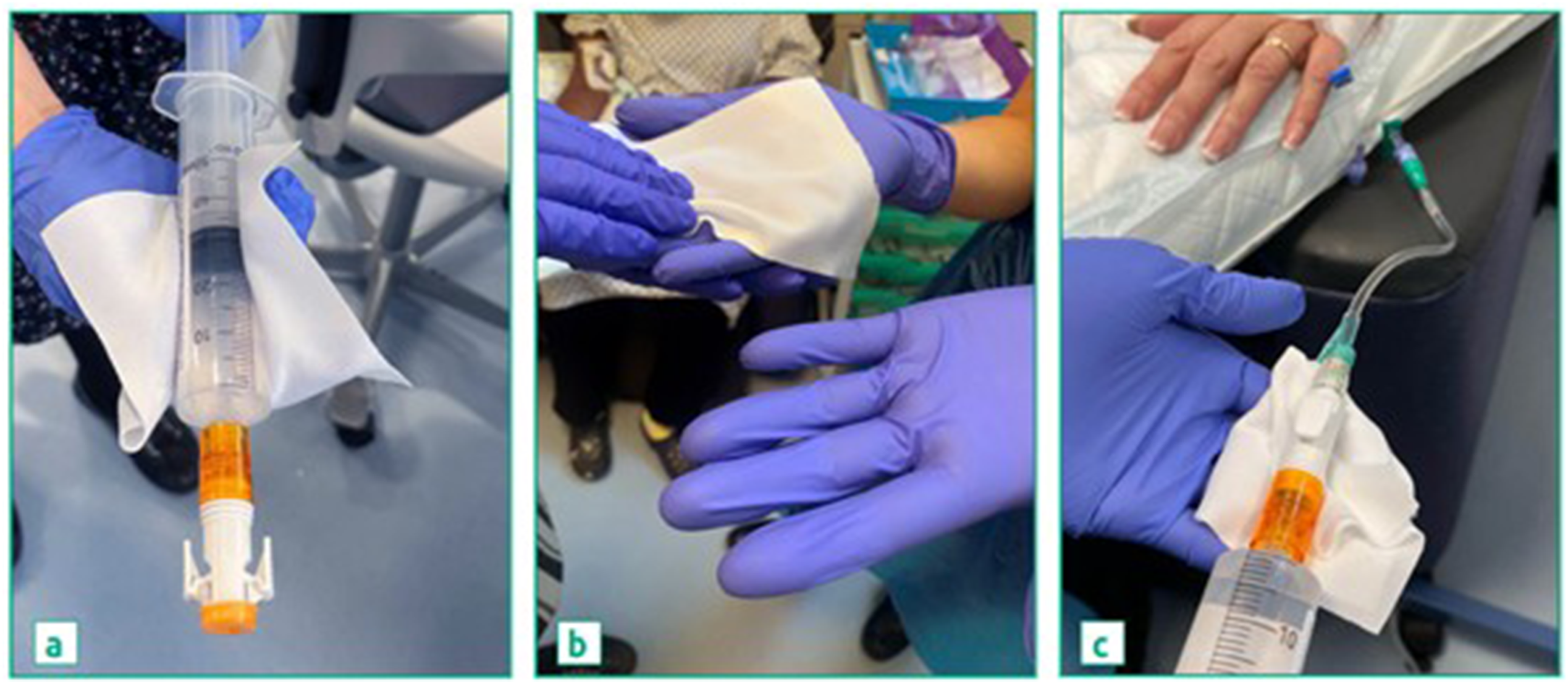
Sampling of (a) syringe surface, (b), gloves, and (c) gauze pads with SAL. The same procedure was performed with standard hub caps.
Two swabs directly placed into vials for analysis served as negative controls. Two swabs, on which a droplet of cyclophosphamide was directly placed, served as positive controls.
Analysis of cyclophosphamide samples
The vials containing the swabs were kept refrigerated and transported to QCNW within 72 h of sampling. At QCNW, 10 mL of 50% methanol were added to each vial containing a sample swab. The vials were then sealed and placed on a roller for 15 min at 45 rotations/minute. From each sample vial, 4 µL of liquid were injected into an Agilent 1260 Infinity II liquid-chromatography mass spectrometer (Agilent Technologies, Santa Clara, CA, USA) under the following conditions: column: Eclipse Plus C18 2.1 × 100 1.8 µ, 30°C; mobile phase: water 0.1% formic acid, methanol 0.1% formic acid; pump flow rate 0.4 mL/min. All samples were analyzed in replicates. This method followed QCNW standard operating procedures which complied with the British Pharmacopoeia guidance on liquid chromatography. 64
Surface area calculation
Cyclophosphamide contamination was further analyzed by surface area. Syringe surface area was estimated by measuring the surface area of the syringe including the extended part of the plunger for the filled syringe with the hub cap or SAL. The surface area of the gloves was based on the average size of male and female hands. 65 The surface area of the swabs was calculated according to their size.
Statistical analysis
Continuous variables were summarized using mean, standard error of the mean (SEM), minimum and maximum values. Categorical values were summarized by number and frequency. Comparisons between groups were performed using unpaired t-tests. A p value below 5% was considered statistically significant.
The following null hypotheses were tested:
There is no significant difference between the level of cyclophosphamide contamination on the syringes, between syringes with a standard hub cap and syringes with a SAL. There is no significant difference between the level of cyclophosphamide contamination on the swabs used for connect/disconnect between syringes with a standard hub cap and syringes with a SAL. There is no significant difference between the level of cyclophosphamide contamination on the nurses’ gloves, between syringes with a standard hub cap and syringes with a SAL.
Ethics considerations
Based on the outcome of the United Kingdom Research and Innovation Medical Research Council and NHS Health Research Authority questionnaire, NHS Research Ethics Committee approval was not required for this study.
Results
Syringe integrity testing with SALs
The Luer lock syringes with the SALs passed the syringe integrity testing validation, as no microbial contamination was observed in any of the broth-filled syringes, while the positive controls yielded microorganism growth.
Cyclophosphamide contamination on the syringes
The results for the cyclophosphamide contamination on the syringes with standard syringe hub caps are displayed in Table 1.
Cyclophosphamide contamination following use of syringes with standard hub caps compared to syringes with SIMPLIVIA® SALs.
Abbreviations: CI = confidence interval, LOD = limit of detection, SAL = Syringe Adaptor Lock, SEM = standard error of the mean; LOD = 7 pg/cm2 (based on sampling 10 cm × 10 cm surface area).
As shown in Table 1, statistically significant reductions in the cyclophosphamide contamination on the syringes, on the swabs used for connect/disconnect and on the nurses’ gloves were observed when SALs were used compared to standard hub caps. Therefore, all null hypotheses were rejected.
Cyclophosphamide contamination was further analyzed by the surface area of the syringes, gloves and swabs used in the study. The surface area of gloves was calculated as 421.51 cm2 and 307.06 cm2, for male and female hands, respectively. 65 Each swab had a surface area of 50 cm2. The syringes’ surface areas are shown in Table 2.
Surface area of syringes.
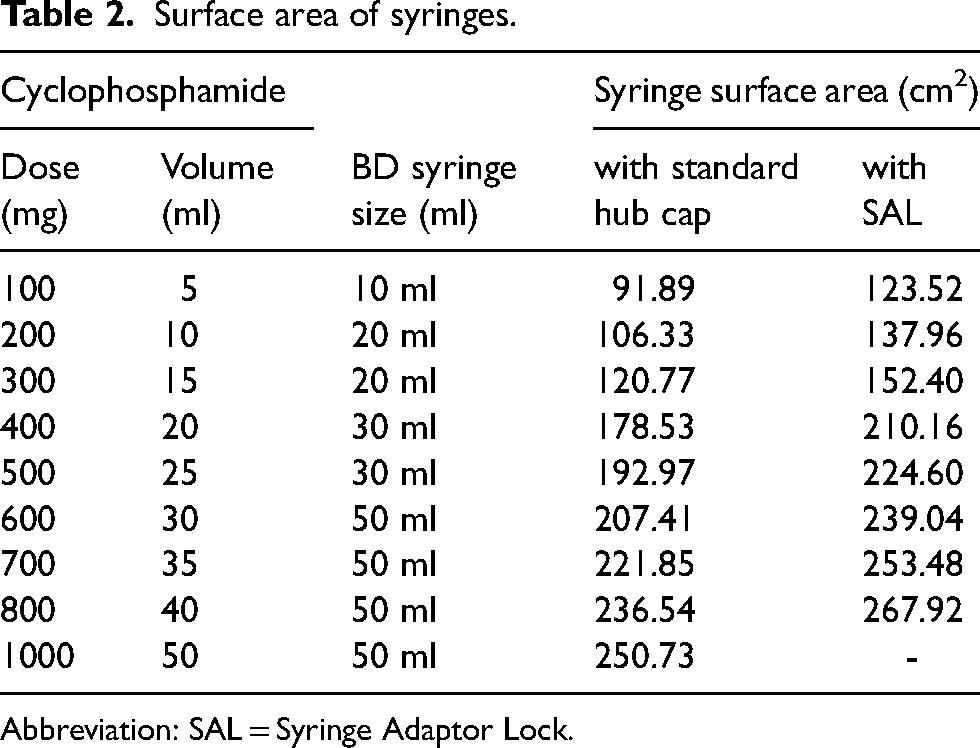
Abbreviation: SAL = Syringe Adaptor Lock.
This analysis of contamination by surface area showed that 12 of 25 samples (48%) analyzed for cyclophosphamide contamination on the surface of syringes with the standard syringe hub cap, 19 of 25 samples (76%) analyzed for contamination of the swab used during connect/disconnect with the standard syringe hub cap and 2 of 25 samples (8%) analyzed for contamination of nurses’ gloves following use of syringes with the standard hub cap showed cyclophosphamide residues above the European Biosafety Network recommended limit of 0.1 ng/cm2, regardless of whether a male or female hand size was used for the calculations. 10 In contrast, no contamination above this limit was observed on samples following the use of the SAL (Table 3).
Percent of samples with cyclophosphamide contamination above the European Biosafety Network recommended limit of 0.1 ng/cm2 following use of syringes with standard hub caps compared to syringes with SALs.

Abbreviations: SAL = Syringe Adaptor Lock.
Discussion
This study has shown that the addition of the SIMPLIVIA® SALs to syringes in the isolator reduces cytotoxic residue following intravenous administration, compared to the addition of standard hub caps. Statistically significant reductions of cyclophosphamide contamination on the surface of syringes, nurses’ gloves and on connect/disconnect were observed. Furthermore, cyclophosphamide contamination levels were below the European Biosafety Network recommended threshold of 0.1 ng/cm2. 10 This threshold was established based on a study conducted in the Netherlands that displayed negative urine samples for cyclophosphamide when the facilities had contamination levels below this level. 66 Therefore, the results indicate that SALs are beneficial over standard syringe hub caps in reducing cytotoxic residue and therefore reducing cytotoxic drug exposure to nurses administering intravenous syringes. The SALs also have a unique patented lock system for Luer lock syringes that cannot be opened once they are locked. This can help prevent accidental exposure to cytotoxic drugs. Furthermore, when the SAL replaces the syringe hub as the terminal closure device, there is full closure integrity of the final container system. This can ensure the safe storage and handling of the prepared cytotoxic drugs.
At the time of this study, the older generation SIMPLIVIA® SAL devices were available in the UK. This version was chemically compatible with most hazardous drugs, with the exception of drugs containing N,N-dimethylacetamide. Recently, however, the older line of devices was replaced with the updated Chemfort® CSTD, which is made of more broadly compatible materials and is approved for use with all known hazardous drugs. Thus, while cyclophosphamide was chosen as a model drug in this study, we expect that contamination reduction can be achieved for a wide range of hazardous drugs.
While other protective measures to reduce exposure to cytotoxic drugs should still be utilized, addition of the SIMPLIVIA® SAL in the isolator could potentially reduce the risks of CMR for nurses administering intravenous syringes containing cytotoxic drugs as well as for other healthcare workers in the wards, such as sanitation workers, who may come into contact with medical waste.
Notably, in this study, SALs were applied to syringes previously filled with cyclophosphamide. This may explain low-level residual contamination on some syringes to which SALs were applied. Alternatively, SALs could be used in combination with additional CSTD components, such as vial adaptors and bag adaptors, to reduce the risk of surface contamination inside the isolator. Future work will include an exploration of whether applying a complete CSTD system for preparation in the isolator may further reduce contamination during administration. We further recommend performing similar studies with other brands of CSTDs to determine whether the same level of contamination reduction can be achieved.
Footnotes
Acknowledgments
The author acknowledges and thanks The University of Hospitals Birmingham NHS Foundation Trust for supporting this study. She also acknowledges and thanks project supervisors Rachel Allen and Sue Renn for their support throughout the study.
Data availability
The data supporting the analysis and findings of this study is available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Based on the outcome of the United Kingdom Research and Innovation Medical Research Council and NHS Health Research Authority questionnaire, NHS Research Ethics Committee approval was not required for this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B. Braun Medical and Simplivia Healthcare, Ltd provided funding for the cytotoxic residue testing and supplied the SIMPLIVIA® Syringe Adaptor Locks and Luer Lock Adaptors free of charge.
