Abstract
Introduction
The purpose of this series of three studies was to identify the limitations of the 2015 NIOSH Vapor Containment Performance Protocol for Assessing Closed System Transfer Devices (CSTDs) as well as the variables that may impact results when testing CSTDs.
Methods
Two different lots of Equashield® CSTDs were tested in accordance with Task 2 of the protocol, using 70% isopropyl alcohol (IPA) as a challenge agent and either a Gasmet™ DX5000 Terra multigas FTIR analyzer or MIRAN® SapphIRe XL Infrared analyzer to measure IPA vapor concentrations. All studies were conducted in NIOSH-designed, custom-built testing chambers. Variables assessed to determine impact on results included: user technique, brand of IV bag, brand of IV administration line, where (inside or outside the chamber) IV bags, IV lines, and y-site adapter CSTDs were assembled, CSTD manufacturing dates, and analyzer type.
Results
The variables that impacted testing results included user technique and experience, use or non-use of bulky gloves during y-site adapter-to-IV-line assembly, and analyzer. Variations in the brands of IV bags and IV administration lines and manufacturing dates of CSTDs did not impact the results.
Conclusion
Based on the known limitations of the 2015 NIOSH protocol, an improved testing protocol – with a better hazardous drug (HD) surrogate than IPA – is needed for assessing the true effectiveness of CSTDs in containing HDs. In the meantime, this protocol can be used to help CSTD manufacturers and/or end users isolate and mitigate potential weaknesses in their CSTD products and/or the compounding techniques used.
Keywords
Introduction
The handling of hazardous drugs (HDs) – from compounding through administration – poses health risks to the pharmacy staff and nurses who regularly use them.1–7 However, several engineering and hygiene controls, such as biosafety cabinets (BSCs), personal protective equipment (PPE), and closed system transfer devices (CSTDs) are commonly used by healthcare personnel when handling HDs and are quite effective in minimizing exposure to HDs.1,7–11
CSTDs are drug transfer devices that “mechanically prohibit the transfer of environmental contaminants into the system and the escape of the hazardous drug or vapor concentrations outside the system”. 12 These devices are designed with different engineering mechanisms for containing liquid, aerosol, and vapor forms of HDs. The primary types of CSTDs are physical barrier CSTDs, which use membranes to contain HDs within the system, and air-cleaning CSTDs, which use various types of filters to contain HDs.12–16
During compounding of HDs, vial adapter, syringe adapter, and IV bag adapter CSTDs are used in combination for the secure transfer of HDs from vials to syringes and syringes to IV bags. For HD administration, syringe adapters, IV bag adapters, and IV administration line y-site adapters are typically involved.
Assessing the effectiveness, or true closedness, of CSTDs is a challenging proposition though. Many studies have been performed to measure the amount of actual HD found in a drug preparation area or hood after compounding such drugs. These tests have demonstrated that CSTDs do indeed decrease the amount of HD contamination found in a compounding area after drug preparation, but these studies aren’t without limitations. These tests, which involve actual HDs such as cyclophosphamide, 5-fluorouracil, or gemcitabine,10,11,17–19 pose exposure risks to the researchers themselves, do not allow for the differentiation of liquid, aerosol, and vapor release, and don’t assess environmental contamination during HD administration. They also often do not provide specifics on where or how the HD is escaping.
In attempts to develop a safer protocol for assessing the closedness of CSTDs, several groups have investigated potential HD surrogates (or challenge agents) and testing methods. Early efforts to identify an effective HD surrogate included use of titanium tetrachloride (TiCl4), radioactive technetium [99mTc], and/or fluorescein; however, none of these agents proved specific enough to elucidate points of CSTD leakage while also quantifying the amount of surrogate leaked.20–22
In 2015, the National Institute of Occupational Safety and Health (NIOSH) developed a draft Vapor Containment Performance Protocol for Assessing CSTDs using a 70% isopropyl alcohol (IPA) as a HD surrogate and a MIRAN® SapphIRe XL Infrared analyzer for measuring the concentration of IPA vapor, released either as headspace gas or as a liquid, which vaporizes upon release. 12 While IPA is inexpensive, readily available, and relatively safe to use, this protocol cannot differentiate between vapor, aerosol, and liquid release during CSTD use and IPA is widely considered not to be an effective surrogate for testing air-cleaning types of CSTDs due to its high volatility and small size.12,14
Since the release of the draft 2015 NIOSH protocol, there have been numerous efforts to identify a better HD surrogate than IPA for testing CSTDs.23–24 These tests, which include testing surrogates such as phenoxyethanol (2-POE), propylene glycol methyl ether (PGME), 1,1,3,3-tetraethylurea (TEU), and methyl tert-butyl ether (MTBE) as well as varying the analytical methods used (e.g., Fourier transform infrared spectroscopy [FTIR], automated thermal desorption gas chromatography mass spectrometry [ATD-GC-MS], and selected ion flow tube mass spectrometry [SIFT-MS]),20,25–28 have not yet yielded an effective universal HD surrogate or protocol for testing both barrier and air-cleaning types of CSTDs, quantifying the amount of surrogate released, locating points of leakage, or differentiating between vapor, aerosol, and liquid release. However, the NIOSH 2015 draft protocol is still used as an industry standard, despite its limitations. 15
Studies that have tested CSTDs against the 2015 NIOSH Vapor Containment Performance Protocol with IPA and a MIRAN® SapphIRe XL Infrared analyzer have shown that some brands (i.e., Equashield®, BD Phaseal™) are able to contain IPA below the predefined testing criteria, defined as a mean maximum background-adjusted, zero-corrected (BG-0max) vapor concentration measurement (in parts per million [ppm]) less than the limit of quantification (LOQ) for the MIRAN® analyzer (LOQ = 1.0 ppm), while other brands have not.29–31 The brands that meet this testing criteria for both Task 1 and Task 2 of the 2015 NIOSH protocol are considered to be validated closed systems per the protocol.12,29
In contrast, a recent study – wherein seven non-vented CSTDs were tested according to the 2015 NIOSH protocol but with a Gasmet™ DX5000 Terra multigas FTIR analyzer instead of a MIRAN® analyzer – demonstrated that only one CSTD brand maintained an IPA vapor concentration below the Gasmet's LOQ of 0.04 ppm for IPA; all other brands did not. 32 However, it was noted in that study that IPA vapor measurements exceeding either analyzer's LOQ (Gasmet™ or MIRAN®) does not necessarily indicate that a CSTD is unable to contain HDs in clinical practice. Additionally, no studies have been performed to date to determine the correlation between amount of IPA released and amount of HD exposure; therefore, there is no clear threshold for declaring “failure” of a CSTD when using this protocol (with IPA), regardless of analyzer used.
The collective results from these IPA studies highlight a few important questions: what threshold of IPA vapor concentration is an acceptable safety performance metric when testing CSTDs according to the NIOSH protocol? And, how much IPA release is analogous to an unsafe amount of HD release? The lack of such clarity is another limitation of the existing NIOSH protocol.
The purpose of this series of three studies was to better understand the limitations of the 2015 NIOSH Vapor Containment Performance Protocol as well as the variables beyond CSTD engineering design that may impact results. This goal was achieved by assessing and comparing the effectiveness of two different sets of Equashield® CSTD products (older lots and newer lots) in combination with different brands of IV bags and IV administration lines in containing IPA during pharmaceutical compounding and administration processes following Task 2 of the protocol. A Gasmet™ DX5000 Terra multigas FTIR analyzer was used to measure IPA vapor concentrations in Studies 1 and 3 to directly compare results with our previous study. 32 A MIRAN® SapphIRe XL Infrared analyzer was used in Study 2 to allow for comparisons with Gasmet™-generated data as well as with historical data generated using the same type of MIRAN® analyzer.29–31
Methods
In a series of three different studies, Equashield® CSTDs were tested to determine their effectiveness in containing the challenge agent, 70% IPA, while following Task 2 of the 2015 NIOSH Vapor Containment Performance Protocol. Task 2 simulates a series of pharmaceutical compounding and administration steps and includes vial to syringe, syringe to IV administration line, and IV administration line to IV bag transfers. 12
The steps of Task 2 consist of transferring 45 mL of IPA from one vial to another vial using a 60 mL syringe connected to a syringe adapter, followed by two 45 mL transfers of IPA from the second vial to an IV administration line connected to a 500 mL, 0.9% sodium chloride IV bag. The IPA is injected twice into the IV administration line's y-site via a y-site adapter using two syringes with adapters. The injected IPA then flows up through the IV line and IV bag adapter into the IV bag. The CSTD components evaluated in each run of Task 2 included two vial adapters, two syringe adapters, one bag adapter, and one y-site adapter. Of note, unlike most CSTD brands on the market, Equashield® syringe adapters are pre-bonded to Equashield®-specific 60 mL syringes.
The testing was conducted in a laboratory setting equipped with a vented fume hood. All experiments were conducted at ambient room temperature (approximately 22–25°C) within a custom-built environmental test chamber, built per the specifications provided by the 2015 NIOSH protocol. 12 IPA vapor release was detected and measured in real time by a Gasmet™ DX5000 Terra multigas FTIR analyzer (Gasmet Technologies; Oy, Finland), operated with Calcmet software for real-time data acquisition and analysis for Studies 1 and 3. A MIRAN® SapphIRe XL Infrared analyzer (Thermo Scientific; Franklin, MA) was used for Study 2. Each analyzer was calibrated specifically for IPA detection according to each analyzer's instructions for use (IFU). The MIRAN® analyzer's limit of detection (LOD) for IPA is 0.3 ppm (LOQ = 1.0 ppm) while the Gasmet™ analyzer's LOD for IPA is 0.012 ppm (LOQ = 0.04 ppm) when 3-min sampling times are used. 33 The LOD for each analyzer was provided by the analyzer's manufacturer and the LOQ values were calculated by using a simple formula of 3.33 x LOD, as prescribed by the NIOSH protocol. 32 For the study using the MIRAN analyzer (Study 2), the chamber set-up described in the NIOSH protocol was used; however, a modified testing chamber – one that functioned with the Gasmet™ analyzer as a closed-loop system – was used for Studies 1 and 3. (Figure 1). 32 Additionally, for the Gasmet™ analyzer, airflow was maintained at a consistent rate of 2 liters per minute during testing and high purity nitrogen gas was used for purging and zeroing after each test per the analyzer's IFU. Three different technicians conducted the testing in these studies, each with 16–20 h of hands-on protocol training and CSTD testing, and each with proper technique verified by the PI, prior to conducting these experiments.

Modified NIOSH custom-built environmental test chamber connected to a Gasmet™ DX5000 Terra multigas FTIR analyzer.
Prior to each run of Task 2, two vials containing 50 mL of 70% IPA were prepared using the following process: 50 mL of room-temperature 70% IPA was transferred into a 100 mL glass vial using a 100 mL graduated cylinder. A 20 mm septum cap and a 20 mm aluminum crimp seal were placed on top of the vial and sealed using a 20 mm crimping tool. After the vial was sealed, the outside of the vial was wiped down with a nonalcoholic cleaner to ensure there was no residual IPA on the outside of the vial. Preparation of the vials was performed in a clean fume hood at room temperature. All other materials for the tasks (e.g., CSTDs, IV bags, IV administration lines) were kept at room temperature, in sealed packages, prior to the testing period. The packaged materials needed for each run were opened immediately before the start of the run.
All steps were performed in accordance with the CSTD manufacturer's IFU. The key steps for Task 2, after which analyzer readings were recorded upon reaching equilibrium, are listed in Table 1. Of note, air was not drawn into the syringes prior to withdrawing IPA from the vials, as was prescribed by steps 5 and 15 in the NIOSH protocol, 12 based on the Equashield® adapter's IFU.
NIOSH protocol steps for task 2.

Within the 2015 NIOSH protocol, vials and syringes were numbered 1 and 2 for Task 1; 3 and 4 for Task 2.
All Equashield® syringe adapters were pre-bonded to Equashield®-specific 60 mL syringes; air was not drawn into the syringe prior to withdrawing IPA from the vial, based on the Equashield® adapter's IFU.
The amount of IPA vapor detected by the Gasmet™ analyzer was recorded after the completion of each key step after 3 min (180 s) of continuous sampling to allow for equilibrium to be reached. This sampling time was a deviation from the 2015 NIOSH protocol which states to “pause for 30 s or until the IPA detector stabilizes” prior to recording IPA vapor concentration readings. 12 This deviation was based on the differences between the MIRAN® and Gasmet™ analyzers and internal testing performed to determine the time-to-equilibrium for the Gasmet™-chamber system.
The amount of IPA vapor detected by the MIRAN® analyzer was recorded after the completion of each key step after 30 s of continuous sampling or until the IPA concentration readings stabilized, as directed by the NIOSH protocol. The IPA vapor concentration in the chamber was reported and recorded in ppm for each of these steps. Please see the NIOSH protocol for the full stepwise test procedure. 12
For Study 1, which was exploratory in nature, Task 2 was run 15 times using the same set of Equashield® CSTD lots with variations in the technician running the test, the brand of IV bag used, brand of IV administration line used, and where (inside or outside the chamber) IV bags, IV lines, and y-site adapter CSTDs were assembled (Table 2). For Study 2, Task 2 was run 6 times, and for Study 3, Task 2 was run 6 times for each lot tested based on precedent set in previous studies.
Combined results from studies 1-3.
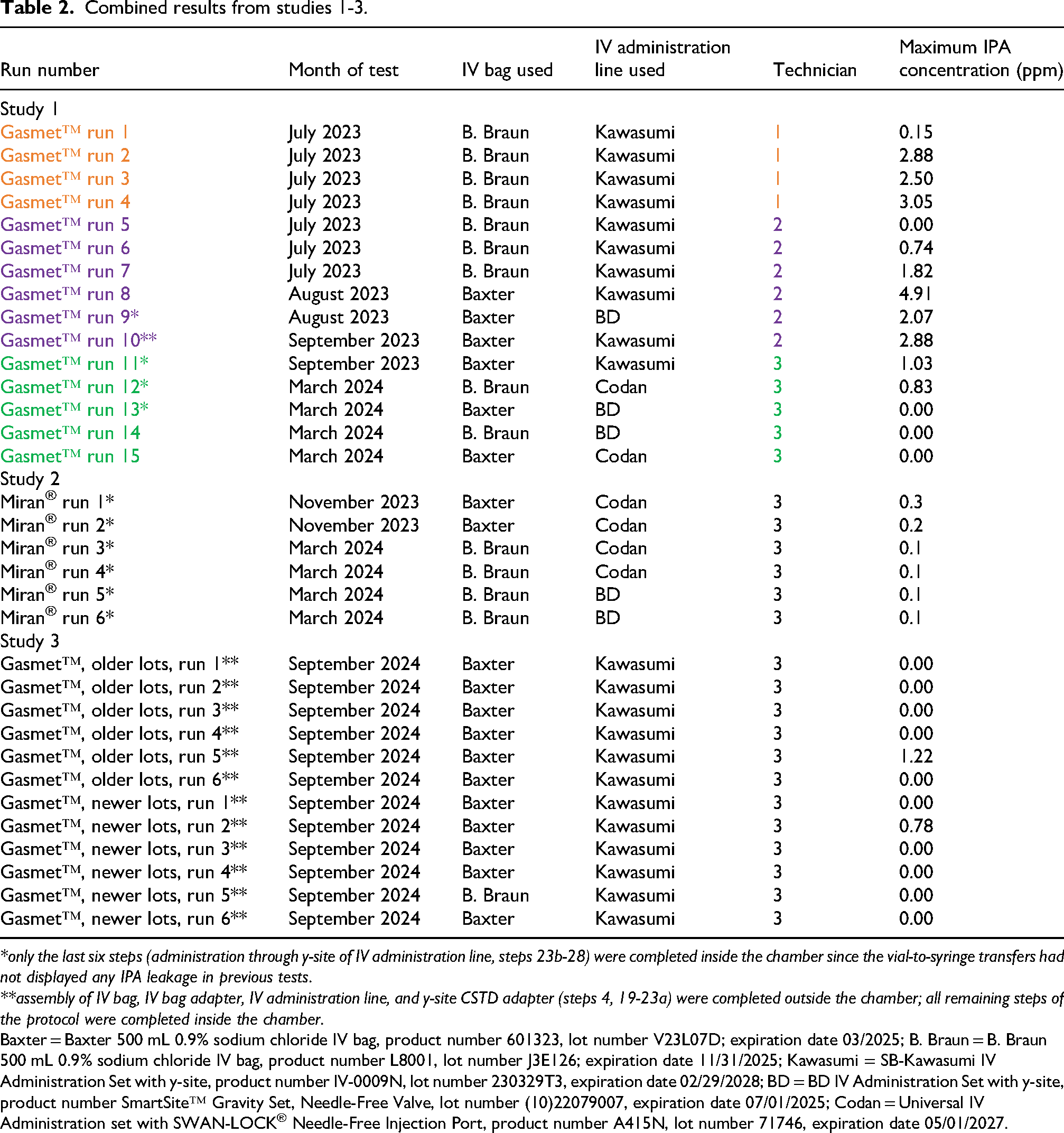
*only the last six steps (administration through y-site of IV administration line, steps 23b-28) were completed inside the chamber since the vial-to-syringe transfers had not displayed any IPA leakage in previous tests.
**assembly of IV bag, IV bag adapter, IV administration line, and y-site CSTD adapter (steps 4, 19-23a) were completed outside the chamber; all remaining steps of the protocol were completed inside the chamber.
Baxter = Baxter 500 mL 0.9% sodium chloride IV bag, product number 601323, lot number V23L07D; expiration date 03/2025; B. Braun = B. Braun 500 mL 0.9% sodium chloride IV bag, product number L8001, lot number J3E126; expiration date 11/31/2025; Kawasumi = SB-Kawasumi IV Administration Set with y-site, product number IV-0009N, lot number 230329T3, expiration date 02/29/2028; BD = BD IV Administration Set with y-site, product number SmartSite™ Gravity Set, Needle-Free Valve, lot number (10)22079007, expiration date 07/01/2025; Codan = Universal IV Administration set with SWAN-LOCK® Needle-Free Injection Port, product number A415N, lot number 71746, expiration date 05/01/2027.
To test the impact of the thick chamber gloves on the ease of CSTD manipulation, the following steps were completed outside the chamber for the runs marked in Table 2:
Runs marked with an *: only the last six steps (administration through y-site of IV administration line, steps 23b – 28) were completed inside the chamber since the vial-to-syringe transfers (steps 1 – 23a) had not displayed any IPA leakage in previous tests. The purpose of this change in protocol was to try and locate the point(s) of IPA leakage during the syringe-to-IV line-to-IV bag transfers in a time-efficient manner.
Runs marked with **: assembly of IV bag, IV bag adapter, IV administration line, and y-site CSTD adapter (steps 4, 19 – 23a) were completed outside the chamber and all remaining steps of the protocol were completed inside the chamber. These changes were made to test the impact of the use of bulky gloves on results. Completing steps 4 and 19 – 23a outside the chamber was a deviation from the NIOSH protocol; however, no IPA transfer was involved during these steps:
The CSTD bag adapter was attached to the IV bag (Step 4) The roller clamps on the IV administration tubing were closed (Step 19) The cover on the spike of the IV administration tubing was removed and the infusion port on the bag adapter was opened (Step 20) The tubing spike was inserted into the port of the bag adapter (Step 21) The IV bag was gently squeezed to verify there was a secure connection and flow into the drip chamber (Step 22) The IV y-site adapter was attached to the y-site of the IV line (Step 23a)
All vapor measurement data points collected were background-corrected prior to analysis and the maximum value observed during each run of Task 2 was averaged to give the mean maximum BG-0max concentration values (in ppm) for those sets of experiments. The 95% confidence interval (CI) was also calculated for the mean BG-0max values with the upper 95% CI of the mean BG-0max serving as the overall task performance metric, as prescribed by the NIOSH protocol. 12 All calculations were performed using Microsoft® Excel® software (Microsoft 365 MSO Version 2401 Build 16.0.17231.20236). Effective containment of IPA was defined as a measurable vapor concentration less than the analyzer's LOQ (LOQ = 0.04 ppm for the Gasmet™; 1.0 ppm for the MIRAN®), also in alignment with the NIOSH protocol.
Results
For Study 1, wherein Equashield® CSTDs were tested according to Task 2 of the 2015 NIOSH protocol using a Gasmet™ analyzer, mixed results were observed. During these 15 runs, the technician; brand of IV bag; brand of IV line; and location of IV bag, IV line, and y-site adapter assembly varied between runs. Four out of 15 runs (26.7%) had maximum readings below the instrument's LOQ of 0.04 ppm. The remaining 11 runs produced maximum IPA vapor concentration readings of 0.15 to 4.91 ppm. Variations in technician and location of IV bag, IV line, and y-site adapter assembly were the only variables that impacted the results (Table 2, Figure 2). The average maximum IPA concentration for all 15 runs was 1.52 ppm (95% CI = 0.79–2.26) (Table 3).
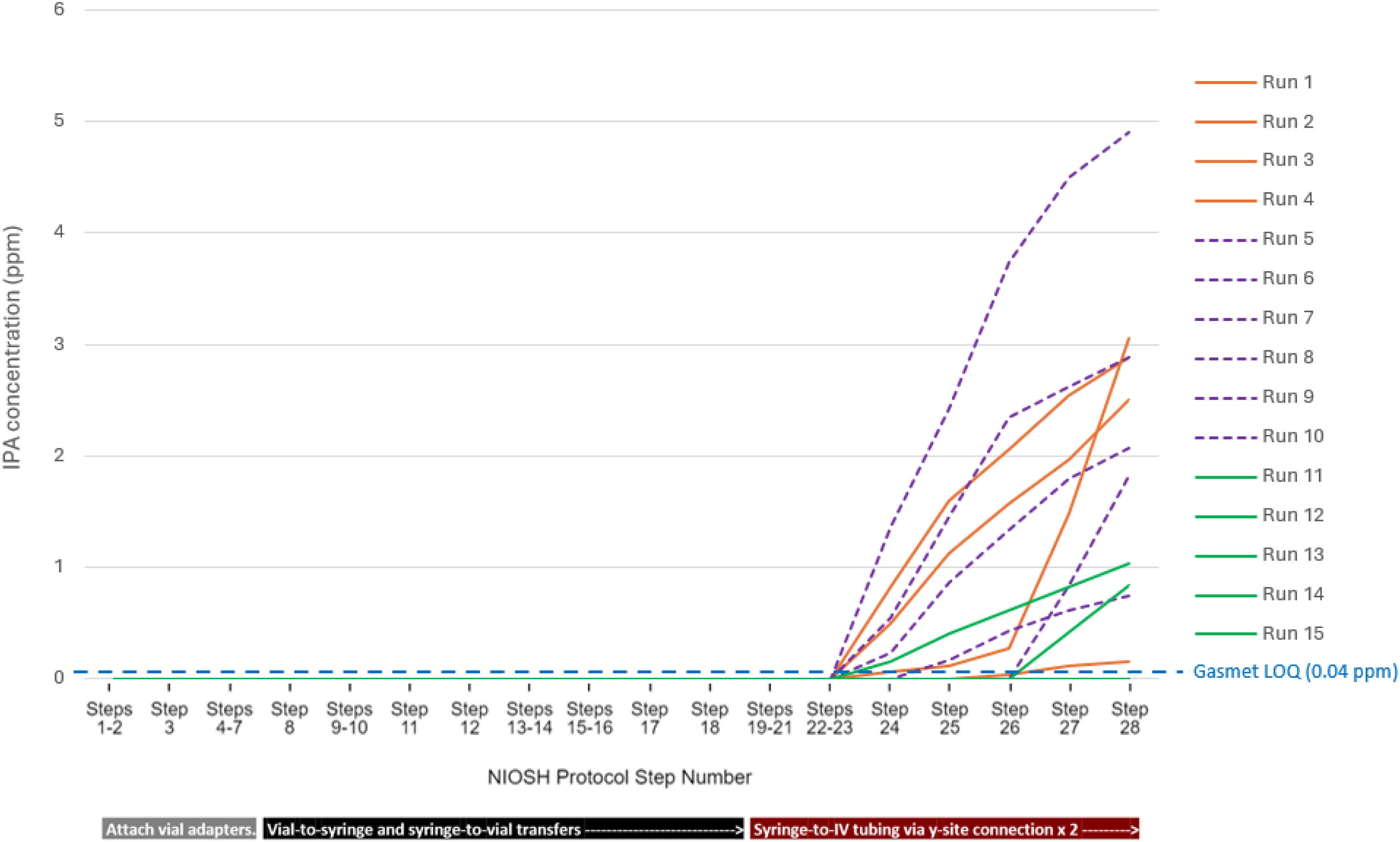
Task 2 results with a Gasmet™ analyzer (Studies 1 and 3).
Task 2 results from studies 1-3.
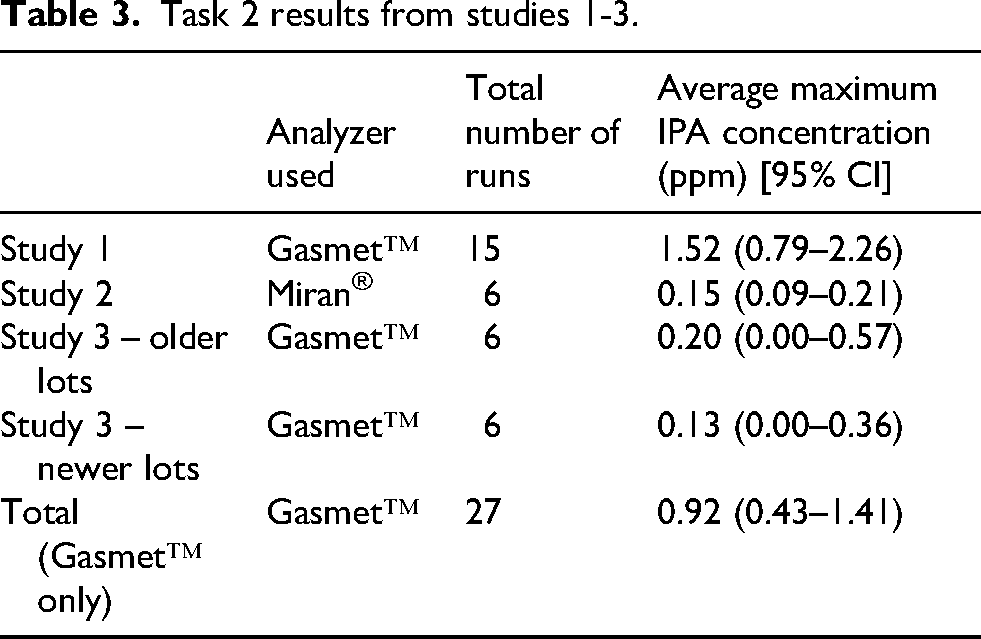
For Study 2, when a Miran® analyzer was used for IPA vapor concentration measurements, six out of six runs of Task 2 (100.0%) had maximum readings below the instrument's LOQ of 1.0 ppm and an average maximum IPA concentration of 0.15 ppm (95% CI = 0.09–0.21) (Tables 2 and 3). As in Study 1, the brand of IV bag and brand of IV line were varied between runs, with no noticeable impact on the results.
In Study 3, sets of older and newer lots of Equashield® CSTDs were tested according to Task 2 of the protocol using a using a Gasmet™ DX5000 Terra multigas FTIR analyzer. Five out of six runs (83.3%) for each set of lots tested (older and newer) had a maximum IPA vapor concentration measurement of 0.00 ppm (Table 2, Figure 2). However, one run out of six for each set tested displayed some IPA vapor leakage (up to 1.22 ppm) after vial adapters were attached to vials containing IPA. The average maximum IPA vapor concentration for both sets of lots were above the Gasmet™ analyzer's LOQ of 0.04 ppm (older lots: 0.20 ppm [95% CI = 0.00–0.57]; newer lots: 0.13 ppm [95% CI = 0.00–0.36]) (Table 3) but consistent with previously published results, which range from 0.30 to 0.60 ppm with a MIRAN® analyzer.3–5
Discussion
To determine what variables beyond CSTD engineering design may impact results obtained when using the 2015 NIOSH Vapor Containment Performance Protocol, a series of three studies were conducted with Equashield® CSTDs. In Study 1, variations in the technician running the test, the brand of IV bag used, brand of IV administration line used, lots of the CSTDs (with different manufacturing dates), and where (inside or outside the chamber) IV bags, IV lines, and y-site adapter CSTDs were assembled were tested to determine which of those factors, if any, impacted the results. This study provided evidence that changes in technician and location of IV-line assembly had an impact on the results.
For Study 2, the analyzer used to measure IPA vapor concentration levels was changed from a Gasmet™ DX5000 Terra multigas FTIR analyzer to a MIRAN® SapphIRe XL Infrared analyzer. The brand of IV bag and brand of IV line were also varied between runs. The overall IPA vapor concentrations detected using the MIRAN® analyzer were lower than when the Gasmet™ analyzer were used (Studies 1 and 3). Internal testing by this group also found that IPA readings for equivalent volumes of IPA were consistently higher with the Gasmet™ analyzer than with the MIRAN®, which would be expected given that the Gasmet™-chamber set up was a closed-loop system and the MIRAN®-chamber system was not. The results obtained in Study 2 also provided further evidence to that gathered in Study 1 that changes in the brand of IV bag and IV line did not impact results.
The results for Study 3, wherein IPA leakage was observed when vial adapters were attached to vials containing IPA for two of the 12 runs (16.7%), indicate a potential issue with the user's technique when attaching the vial adapters. There were no differences observed between the older and newer lots tested thereby indicating that potential variations in manufacturing processes at different points in time did not play a role in the observed results. Furthermore, all assembly of the IV bags, IV lines, and y-site adapter occurred outside of chamber for this study and no leakage from that assembly was observed during testing. Therefore, assembling the IV bag, IV line, and y-site adapter outside the chamber seemed to positively impact results.
Previous studies that evaluated CSTDs according to the 2015 NIOSH protocol with a MIRAN® analyzer showed that Equashield® CSTDs adequately contained IPA during Task 2 by maintaining a maximum IPA vapor measurement below the predefined testing criteria of 1.0 ppm (the Miran® analyzer's LOQ for IPA) on a consistent basis.29–31 Our results from Study 2 align with this historical data.
However, during Studies 1 and 3, Equashield® CSTDs did not fully contain IPA during Task 2, producing a maximum IPA vapor measurement above the Gasmet's LOQ of 0.04 ppm. When IPA leakage occurred in Study 1, it was observed after IPA was injected through the y-site adapter (Figure 2). It was noted by the users at the time of the testing that the Equashield® y-site adapter was difficult to attach to the IV administration line's y-site when using the bulky gloves of the chamber (see Figure 1). When the y-site adapter was attached to the IV administration line outside of the chamber prior to testing, without the encumbrance of the gloves (runs 9–15, Table 2), IPA vapor concentration measurements were lower than what was observed for earlier runs. The data also show that with time and user experience (i.e., from September 2023 through September 2024, Table 2), maximum IPA vapor concentration levels decreased.
Further supporting the theory that different users, length of user experience, and use or non-use of bulky gloves during line assembly were key contributing factors to the results observed, was that additional variables were evaluated over the course of the 33 runs of the three studies and none of those variables seemed to contribute to IPA leakage (Table 2). Specifically, different brands of IV bags and IV administration lines were tested to assess if either factor contributed to the observed IPA leakage or if the IPA leakage could be coming from the IV bag adapter to IV bag connection (Figure 3, A) or the IV administration line to IV bag adapter connection (Figure 3, B). However, no correlation between IPA leakage and variations in IV bag or IV administration line brand were noted (Figure 2, Table 2), leaving the y-site adaptor to IV administration line connection (Figure 3, C) as the likely point of leakage.
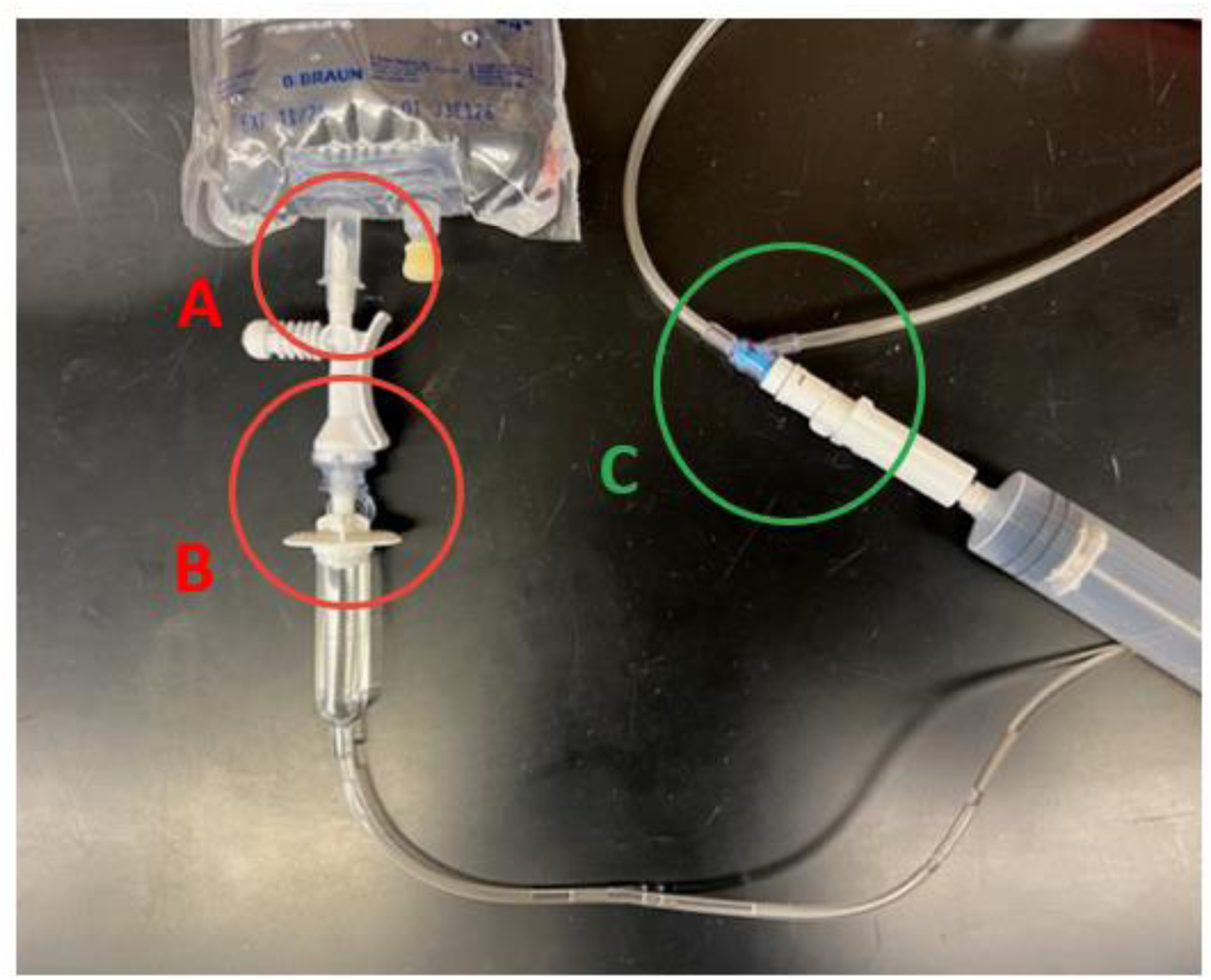
IV bag adapter to IV bag (A) and IV administration line to IV bag adapter connections (B) vs syringe adapter to y-site adapter to IV administration line connections (C).
In summary, the results from these three studies suggest that variations in the users’ technique as well as the use of bulky gloves during the assembly of the IV bags, IV lines, and y-site adapters contributed to the IPA vapor leakage observed. Additionally, the different analyzers used (one operating in open system [MIRAN®] and the other in a closed-loop system [Gasmet™]) measured the amount of IPA released to different degrees, meaning their results cannot be compared 1:1. The results measured by the Gasmet™ were higher than when the MIRAN was used. Changes in the brand of IV bags, brand of IV administration lines, and manufactured lots of CSTDs tested did not impact the results.
In considering these results, especially in light of the different analyzers used, it is important to reiterate that IPA vapor measurements exceeding the Gasmet™ or MIRAN® analyzer's LOQ do not necessarily indicate that a CSTD is unable to contain HD vapors in clinical practice. IPA, due to its higher volatility, smaller molecular size, and distinct chemical properties compared to HDs, is not an ideal surrogate for evaluating CSTD effectiveness.15,21 Furthermore, no existing data correlates the extent of exceeding the IPA vapor LOQ of the Gasmet™ or MIRAN® with actual HD occupational exposure. Such excursions merely indicate that a given CSTD cannot be validated as completely closed under the protocol as used, since detectable and quantifiable amounts of the surrogate agent (i.e., IPA) were observed during testing.
Some of the limitations of this series of studies include the small number of runs with each combination of variables and the limited understanding of the differences in sensitivity and specificity between the two analyzers used. More studies are warranted to fully elucidate those differences. The differences in user technique and relatively short period of user training prior to testing can also be considered limitations. However, variability of user proficiency and skill is to be expected with real-world use of these devices, so these limitations are minimal, rendering these results generalizable to everyday use.
These results highlight some limitations of the 2015 NIOSH protocol for testing the closedness of CSTDs. In addition to this protocol not being useful in testing air-cleaning CSTDs,12,14 the inadequacy of IPA as a HD surrogate, and the inability to derive clinically significant meaning from the results, there are issues with the details of the protocol itself. Namely, the use of thick chamber gloves does not adequately reflect real-world use and adds unnecessary encumbrance during CSTD manipulations. Additionally, the injection of IPA through an IV-line y-site towards an IV bag does not reflect actual practice and should be adjusted in any future protocol updates.
Conclusion
This series of studies, in conjunction with a previous study assessing Equashield® CSTDs performance during Task 2 of the 2015 draft NIOSH Vapor Containment Performance Protocol, 32 demonstrated variations in the CSTDs’ performance by user and user experience. However, the most significant variable in the performance of the Equashield® CSTDs, especially the Equashield® y-site adapters, was the use or non-use of bulky gloves when attaching a y-site adapter to an IV administration line. The bulky gloves in question are a part of the custom-made testing chamber recommended by NIOSH.
In addition, this series of studies isolated the likely point IPA leakage when leakage was observed during injections of IPA “doses” through a y-site adaptor and IV administration line into an IV bag via an IV bag adapter. This likely point of leakage was identified as being at the y-site-to-IV-line connection.
Based on the results of this study, we suggest that the protocol be adapted such that the y-site adapter be attached to the IV administration line before the line is introduced into the testing chamber, thereby eliminating the encumbrance of the bulky gloves during that manipulation and better reflecting real-world use of the device. Additionally, injections administered through an IV line's y-site should be directed towards the patient end of the line instead of towards the IV bag at the top of the line, again to better reflect real-world use.
Variations in the brand of IV bag, IV administration line, and lots of Equashield® CSTDs used did not have a noticeable impact on the performance of the CSTDs during Task 2.
Further study is required to develop an improved testing protocol and to identify a surrogate more representative of HDs than IPA for assessing the ability of CSTDs to contain HDs during compounding and administration processes (e.g., compounds more similar to HDs in molecular weight, vapor pressure, and chemical properties). However, until a better CSTD testing protocol can be developed, CSTDs that demonstrate the least amount of IPA vapor leakage should be preferred to minimize HD exposure, thereby maximizing patient and staff safety. Results using this protocol can also be used to help companies and/or end users isolate and mitigate potential weaknesses in their CSTD products and/or the compounding techniques used.
Key points
Variables that impacted CSTD testing results using the 2015 NIOSH protocol included user technique, use of bulky gloves during y-site adapter-to-IV-line assembly, and analyzer used. Other known limitations of this protocol include the challenge agent used (isopropyl alcohol) and lack of clarity on the clinical significance of observed results. Based on the known limitations of the 2015 NIOSH protocol, an improved testing protocol is needed for assessing the effectiveness of CSTDs in containing HDs.
Footnotes
Acknowledgements
The authors would like to thank Megan Earnhart and Lauren Hairston, PharmD Candidates at the UNC Eshelman School of Pharmacy for their assistance in conducting the experiments.
Ethical considerations
Not applicable.
Author contributions
SFE and LTA: conceived the study and were involved in protocol development. LTA: data collection, researched literature, performed data analysis, and wrote the first draft of the manuscript. All authors reviewed and approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Equashield® and BD provided funds to support this study.
Declaration of conflicting interests
Equashield® and BD provided funds to support this study. However, neither company participated in the conduct or reporting of this research. SFE has received honoraria from BD in the past for speaking engagements. The authors declare no additional potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
