Abstract
Keywords
Introduction
Over the past two decades, extensive studies have revealed significant contamination of hospital pharmacy environments by hazardous drugs (HDs).1–3 Occupational exposure to these substances poses numerous health risks, including an increased likelihood of developing leukemia (and other cancers), damage to vital organs, and complications in pregnancy affecting maternal and fetal health to healthcare professionals (HCPs) that handle, compound, administer, dispose of, manage drug waste (or HD-contaminated body fluids), clean equipment used for HD handling, storage, and administration. 4 Certain HDs are known to damage Deoxyribonucleic acid (DNA), which may raise the risk of specific cancer types, while others can adversely impact the liver, nervous system, and fertility. There is also a growing concern regarding the potential transmission of harmful effects from nursing mothers to their infants through breast milk. 4
In 2004, the National Institute for Occupational Safety and Health (NIOSH), a leading authority in occupational safety, issued a critical alert regarding handling antineoplastics and other hazardous drugs in healthcare settings, emphasizing the associated health risks, their increasing prevalence, and the persistent contamination of the healthcare environment. In response, new guidelines on the safe handling of cytotoxic and hazardous drugs have been developed by the American Society of Health-System Pharmacists (ASHP) and the International Society of Oncology Pharmacy Practitioners (ISOPP), organizations renowned for their contributions to healthcare safety and standards. 1 Despite the implementation of such guidelines, studies continue to report that healthcare workers remain at risk, with measurable concentrations of cytotoxic drugs found in their bodies and on workplace surfaces.5,6
A pivotal development in safeguarding healthcare personnel has been the introduction of Closed-System Transfer Devices (CSTDs). These devices are designed to eliminate or significantly reduce the exposure of healthcare workers to hazardous drugs by mechanically preventing the ingress of environmental contaminants while containing hazardous drug vapors. 1 When employed appropriately, CSTDs enhance the safety of healthcare personnel during the preparation and administration of hazardous medications by reducing accidental active drug analytes in workplace environments. 4 NIOSH recommends using CSTDs throughout the hazardous drug handling process, underscoring their importance in mitigating exposure risks. In 2012, the Food and Drug Administration (FDA) instituted a product code “ONB” for Closed Antineoplastic and Hazardous Drug Reconstitution and Transfer systems to protect healthcare personnel from chemotherapy agents. 7 Comprehensive standards for safely handling HDs were published in 2016 by the United States Pharmacopoeia (USP) and consist of what is currently known as USP Chapter <800> Hazardous Drugs – Handling in Healthcare Settings, recommending CSTDs as an additional engineering control during hazardous drug compounding and mandating their use during administration when the dosage form allows.8,9
In addition, CSTD configurations are designed with safety as a primary consideration. Each system features four interconnected components: (1) a vial adaptor, (2) a syringe adaptor, (3) an infusion bag spike, and (4) Luer-lock connectors. 9 The vial adaptor attaches securely to a vial and includes a pressure equalization system to prevent the escape of aerosolized drugs. Meanwhile, the syringe adaptor allows easy access to vial contents, supporting safe transfer to intravenous bags for dosage preparation.
A crucial element in ensuring a safe healthcare environment is the routine evaluation of hazardous drug contamination. Throughout the years, surface wipe testing for various antineoplastic agents has been utilized across multiple occupational settings for diverse purposes, including compliance, quality assurance, cleanliness verification, source identification, and the assessment of potential dermal exposure and associated health risks. Presently, the most widely accepted method for detecting surface drug contamination in healthcare settings is wipe testing, specifically for antineoplastic agents and other hazardous drug residues.4,10
The current USP <800> Section 6 stipulates that environmental wipe sampling for hazardous drug residues should be performed regularly, starting with an initial benchmark assessment and continuing every six months, or more frequently, if needed. Sampling should include the interior of the Compounding Pharmacy Environmental Control (C-PEC), pass-through areas, nearby staging and work areas, floors, dispensing areas, outside the HD buffer room, and patient administration areas. 11 Commonly assessed HD markers include cyclophosphamide, ifosfamide, methotrexate, fluorouracil, and platinum-containing drugs. For example, measurable contamination may be indicated by cyclophosphamide levels greater than 1.00 ng/cm2, which some studies suggest can lead to drug uptake in exposed workers. If any measurable contamination is detected, designated personnel must identify, document, and contain the source of contamination. Actions may include reevaluating work practices, re-training staff, conducting thorough deactivation, decontamination, cleaning, and enhancing engineering controls. Wipe sampling should be repeated to validate the effectiveness of the deactivation, decontamination, and cleaning procedures.
Currently, no studies confirm the effectiveness of specific numbers or sizes of wipe samples in measuring contamination levels. This creates a significant gap in the current understanding of contamination assessment, as the lack of empirical data makes it difficult to determine the most effective methods for accurately gauging HD residues. Without established guidelines, pharmacies struggle to develop effective sampling strategies, potentially leading to inconsistent results. Wipe sampling kits must be validated before use to ensure they can reliably recover known marker drugs from different surfaces. Additionally, there are no certifying agencies for these vendors, nor established standards for acceptable hazardous drug surface contamination limits. This absence of standardized protocols further complicates efforts to ensure safe handling of hazardous materials, emphasizing the need for continued research and development of best practices in contamination assessment.
The objective of this analysis was to evaluate hazardous drug surface contamination observed under routine clinical practice following implementation of a CSTD. Although pre-implementation (baseline) wipe testing data were not available for direct comparison, the analysis leverages a large, multiyear real-world dataset encompassing multiple hospital systems, care locations, and drug analytes. In this context, effectiveness is assessed as real-world performance, specifically, the ability of the CSTD to minimize detectable surface contamination during routine use, rather than under controlled experimental conditions. The observed low overall contamination rate, consistency of findings over time, and confirmation through independent statistical analysis collectively provide evidence of CSTD effectiveness in reducing hazardous drug surface contamination in real-world clinical settings.
Materials and methods
Data source and facility participation
This analysis utilized a preexisting database of HD surface wipe test results generated through an environmental monitoring program associated with implementation and use of a CSTD in U.S. Healthcare facilities. The database included multiple sample wipe tests collected using specific surface sampling ChemoAlert™ wipe kits after the implementation of a specific CSTD. Detailed procedures were used to train personnel on standardized procedures for the collection, receipt, preparation, and analysis of individual real-world antineoplastic HD surface wipes between 2018 to 2022. This real-world data (RWD) analysis of a pre-existing database of wipe test data assessed the type, frequency, and location(s) of HD contamination in hospitals and outpatient facilities in the US using a surface sampling kit after implementing a CSTD.
Data (wipe) collection – pooling of facilities and locations
To address variability and inconsistency in facility naming conventions and location descriptors within the real-world dataset, original data from 43 individual facilities were consolidated into 18 hospital systems based on documented affiliations. Similarly, 507 unique surface sampling locations were grouped into nine standardized location categories using common functional descriptors (e.g., Patient Administration Room, Infusion Area/Room, Pharmacy, Hood).
The pooling strategy was employed to enable meaningful aggregated analyses across a large, heterogeneous dataset by improving interpretability and reducing misclassification due to inconsistent nomenclature. However, this approach may attenuate site-specific variability and reduce resolution when comparing individual facilities. As such, pooled results should be interpreted as system-level and location-level trends rather than precise facility-level performance metrics.
Data (wipe collection) methods
Sites provided wipe test submissions every six months for three years following CSTD implementation. Up to five samples per submission were collected and analyzed for cyclophosphamide, ifosfamide, methotrexate, fluorouracil, and platinum-containing drugs. The threshold for detection was > 5 ng and results were typically available in 10 days. The various locations of sample collection were grouped as: (a) Biosafety Cabinet Hood, (b) Infusion Area/Room, (c) Nursing Area, (d) Patient Admin/Room, (e) Pharmacy, (f) Receiving room, (g) Staging room, (h) Unpacking Area/Room, and (i) Other.
While the sampling schedule and analytical methods were consistent, the selection of surface types, surface areas, and precise sampling locations was determined by each facility based on local workflows, risk assessments, and operational needs, resulting in variability across facilities in the types and sizes of surfaces sampled. Such variability reflects real-world clinical practice and is acknowledged as a limitation when interpreting differences between sites and locations.
Statistical analysis – outlier identification and Handling
During data review, a single contamination value (3430 ng) was identified as markedly higher than the remaining dataset, exceeding the mean contamination level among positive samples by more than 60-fold. Based on this extreme deviation and the potential for atypical factors such as inadequate surface cleaning or sampling error, this value was classified as an extreme outlier.
To assess its impact on overall results, sensitivity analyses were performed with and without the outlier included. Summary statistics are presented for both analyses to ensure transparency and allow readers to evaluate the influence of the outlier on reported contamination levels.
Statistical analysis – independent verification
Analyses were descriptive in nature and focused on summarizing the occurrence and magnitude of HD surface contamination across facilities, locations, and analytes. Contamination was summarized using frequency (percentage of positive wipes), mean contamination levels (ng), and distribution across pooled systems and locations. Comparative analyses by location were conducted to assess variability in contamination patterns.
Primary analyses were performed by the study sponsor, and the complete dataset was independently analyzed by an external statistical vendor using the same analytical approach on a de-identified dataset. Independent replication of the analyses was conducted to confirm consistency and validity of results and to ensure analytical independence. The external statistical vendor had no role in data collection, facility participation, or interpretation of the findings beyond reporting statistical outputs.
Results
Overall, this real-world dataset comprises 5531 wipe sample analyses, with the number of wipe analyses relatively equally distributed over the years 2018 to 2022. The lowest number of tests was collected in 2018, representing 6% of the total tests. The percentage of contaminated analyses ranged from 2% to 8% each year. The majority of contaminated analyses originated from two of the 18 systems (Health System 1 (52.4%) and Health System 2 (23.2%)) A summary of the wipe test collection is presented in Table 1.
Wipe test collection summary.
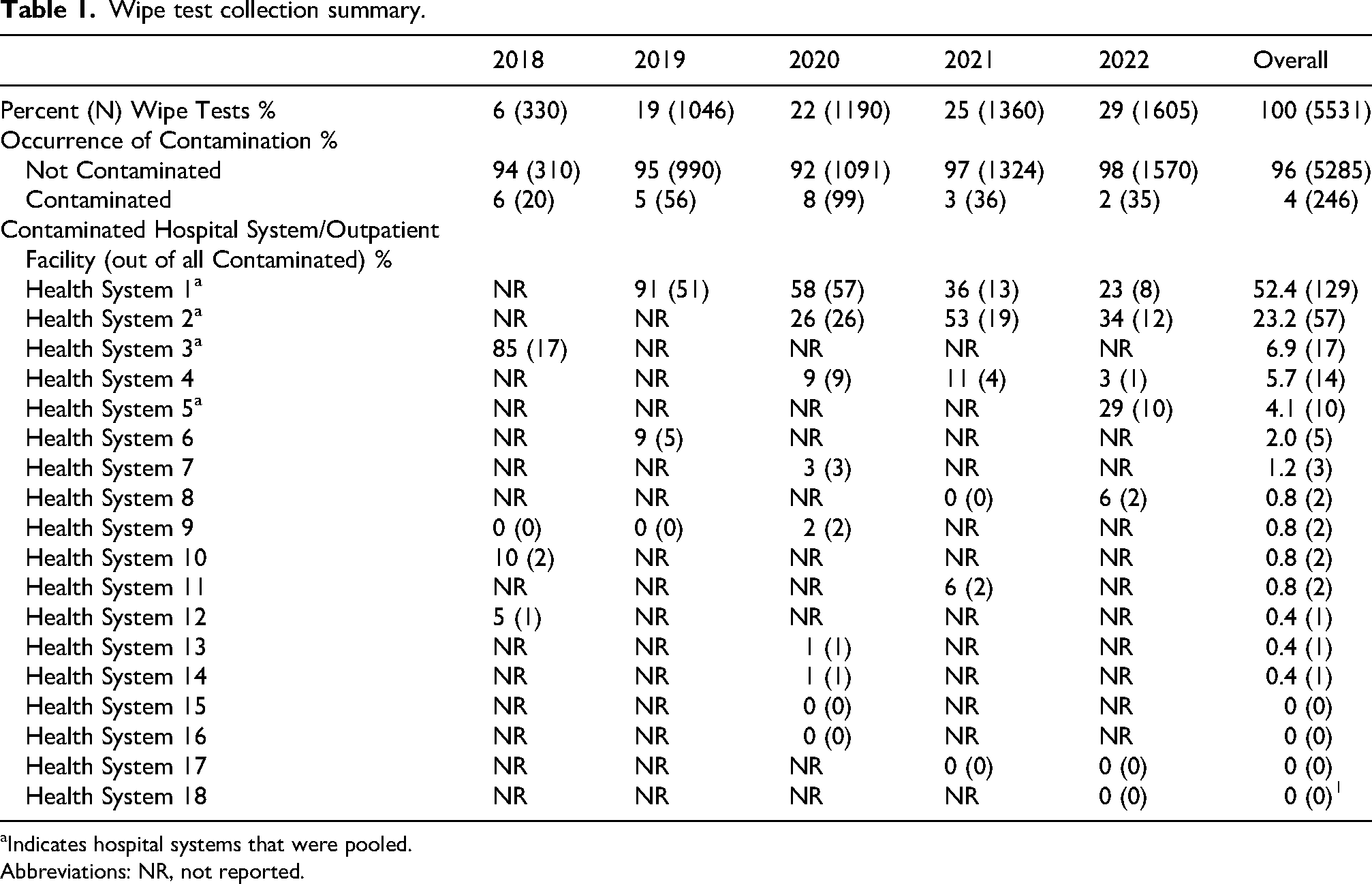
Indicates hospital systems that were pooled.
Abbreviations: NR, not reported.
Contamination overview
From 2018 to 2022, the overall occurrence of contamination was 4.45% (246 out of 5531; see Table 2 and Figure 1). The average level of contamination among the 246 positive contamination results was 54.8 ng, with a range from 5.1 ng to 3430 ng. The average time from collection to analysis for all tests was 11.4 days (range: 1–102 days), while for contaminated tests only, it was 13.9 days (range: 1–102 days). An investigation into the potential association between the level of contamination and the time from collection to analysis did not reveal a significant correlation.
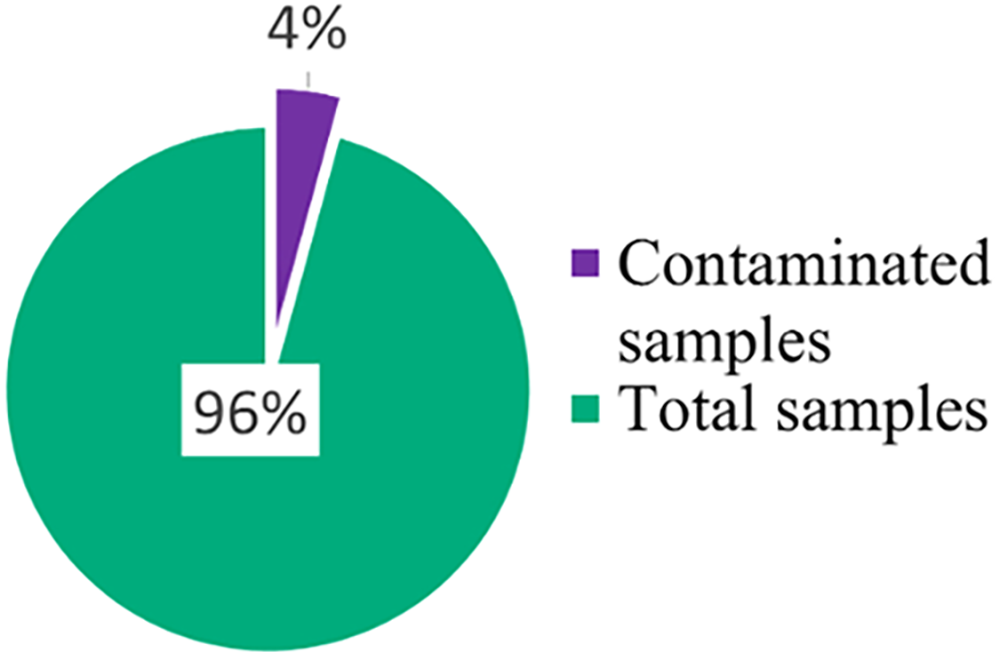
Overall contamination: From 2018 to 2022, the overall occurrence of contamination was 4.45%. This data was initially presented in a poster during the 2024 American Society of Healthcare Pharmacists Midyear Clinical Meeting & Exhibition on December 9, 2024.
Overall contamination with and without outliers.

Abbreviations: min, minimum; max, maximum; ng, nanogram.
Of the 5531 analyses, 12 analyses (0.02%) had contamination levels exceeding 200 ng. Due to the possibility that the highest level of contamination (3430 ng) might be attributed to improper cleaning or poor collection technique by the facility, analyses were performed again after excluding this outlier. After removing the outlier, the average level of contamination for the remaining 245 analyses was 41 ng, with a range of 5.1 ng to 653.0 ng (see Table 2).
Contamination by hospital system/outpatient facility
The occurrence of contamination by health system is detailed in Table 3, varying from 0% to 25% across hospital systems. Health System 6 exhibited the highest rate of contaminated wipe tests, with 25% of the analyses reporting some level of contamination, while 12.3% of analyses at Health System 2 also showed contamination above the threshold limit. The level of contamination ranged from 5.1 ng (at Health System 1) to the outlier of 3430 ng (at Health System 2). The highest mean contamination level was observed at Health System 2 (118.9 ng). It is notable that the majority of contaminated wipe test results originated from two hospital systems, which accounted for most contamination events observed across the dataset, providing important context for interpretation of the overall contamination rate. The concentration of contamination across two sites demonstrates that contamination was not uniformly distributed across participating facilities and may reflect system specific factors such as local practices, workflows, or hazardous drug utilization patterns. The occurrence of contamination by hospital system/outpatient facility is visually depicted in Figure 2.
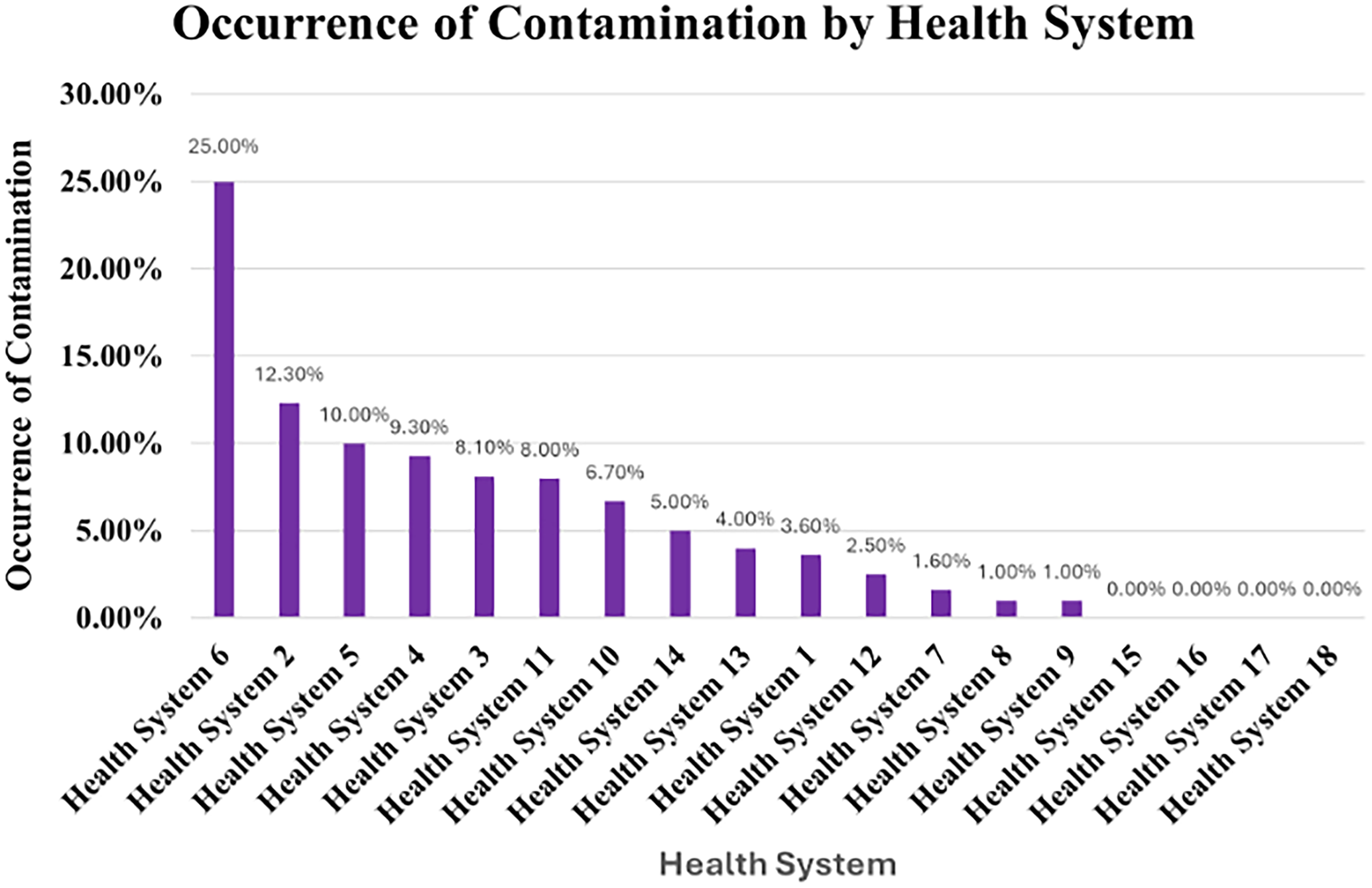
Occurrence of contamination by health system: Heath system 6 had the highest occurrence of contamination at 25.0%, while Health Systems 15–18 reported no contamination. This data was initially presented in a poster during the 2024 American Society of Healthcare Pharmacists Midyear Clinical Meeting & Exhibition on December 9, 2024.
Contamination by hospital system/outpatient facility.
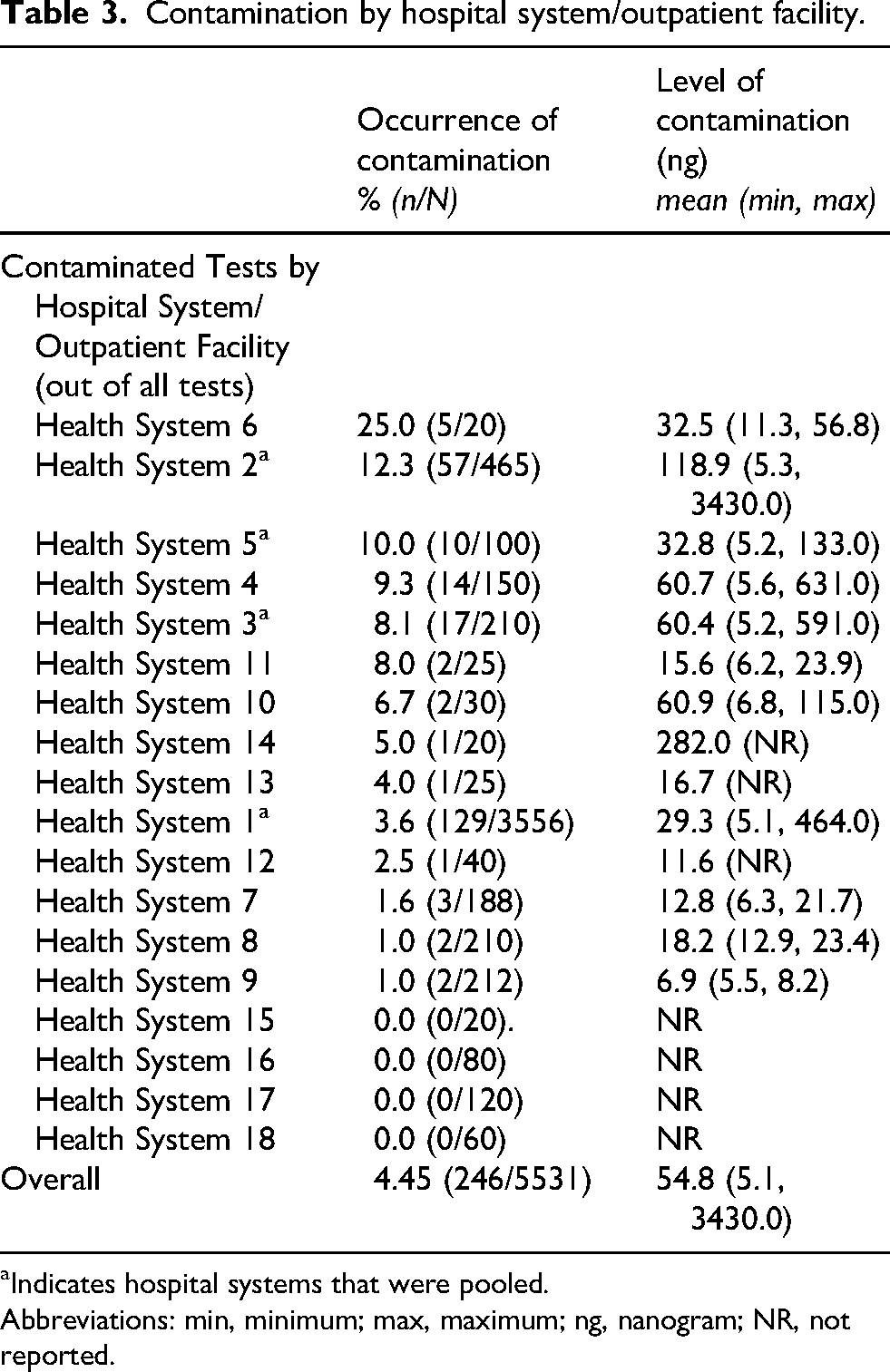
Indicates hospital systems that were pooled.
Abbreviations: min, minimum; max, maximum; ng, nanogram; NR, not reported.
Contamination by analyte
The highest occurrence of contamination was found among 5-Fluorouracil (5-FU) analytes, with 21.2% of the analyses displaying some level of contamination (see Table 4 and Figure 3). The average level of contamination was also highest for 5-FU (105.4 ng). After excluding the outlier, the occurrence of contamination remained highest for 5-Fluorouracil, with 20.9% of analyses showing contamination, whereas Paclitaxel exhibited the highest average level of contamination at 79.4 ng. The higher frequency of contamination observed for 5-FU may reflect local factors such as higher usage volume, preparation frequency, or administration practices rather than differences in device performance, and therefore should be interpreted within the context of this real-world analysis.
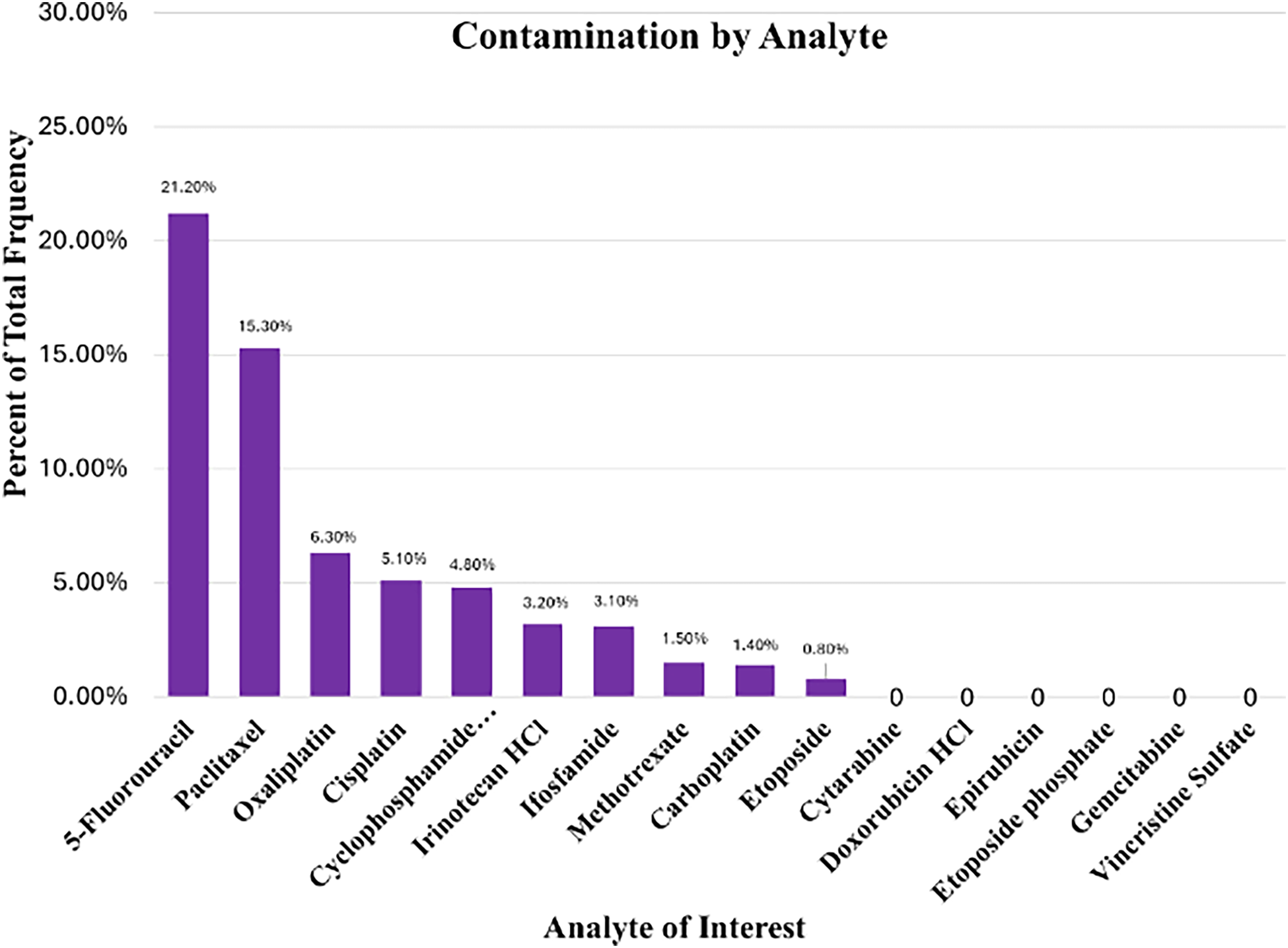
Contamination by analyte: After removing the outlier, the highest occurrence of contamination was reported for 5-fluorouracil (5-fu) analytes, with 20.9% (N = 53/254) of wipe tests showing a mean 42.7 ng level of contamination. The highest level of contamination measured for a single analyte was Paclitaxel at 79.4 ng. This data was initially presented in a poster during the 2024 American Society of Healthcare Pharmacists Midyear Clinical Meeting & Exhibition on December 9, 2024.
Contamination by analyte.
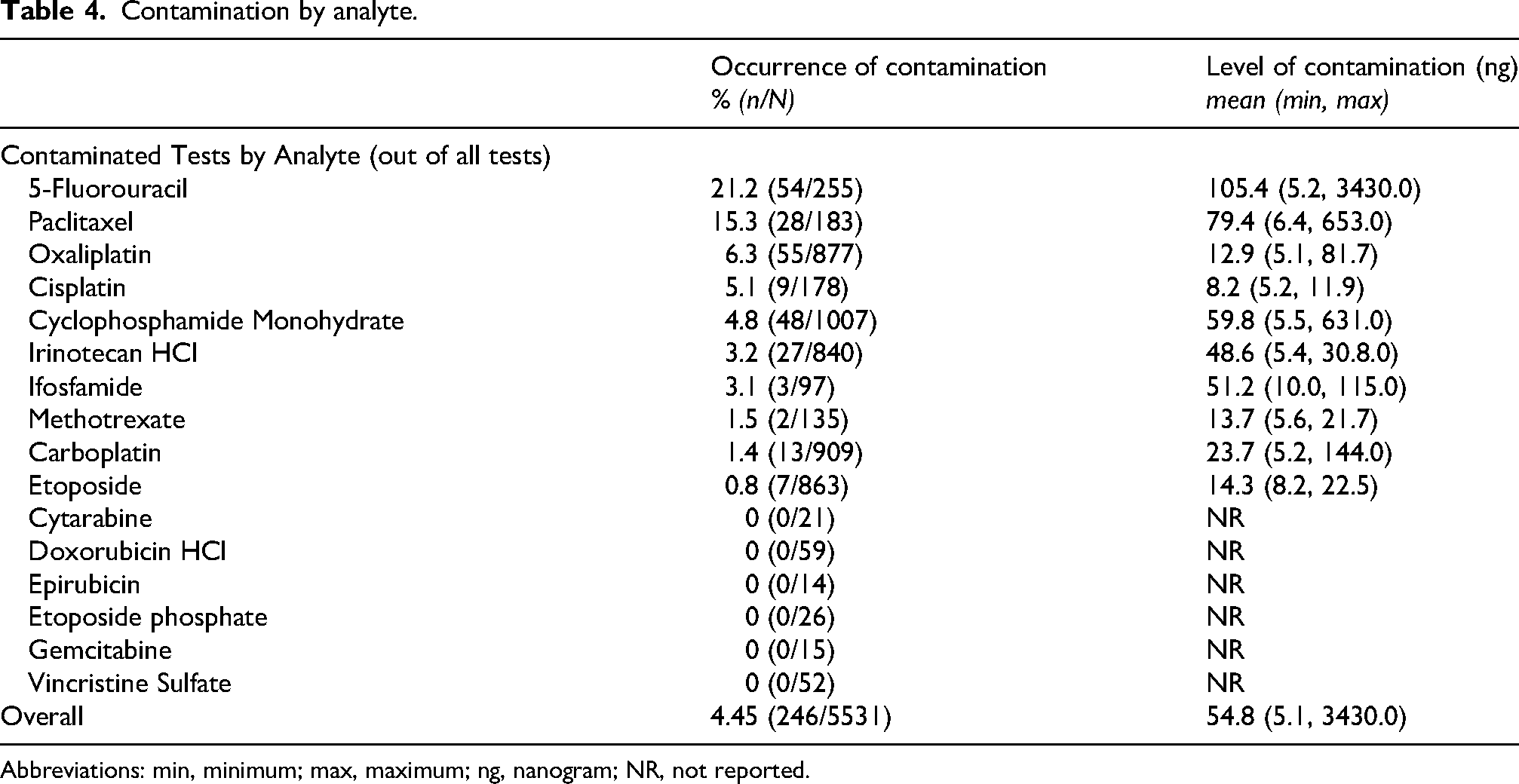
Abbreviations: min, minimum; max, maximum; ng, nanogram; NR, not reported.
An investigation into the time from collection to time of analysis was performed to determine if there was any association between the two. No contamination was detected for Cytarabine, Doxorubicin HCl, Epirubicin, Etoposide phosphate, Gemcitabine, and Vincristine Sulfate samples, regardless of the time from collection to analysis.
Contamination by location
The highest occurrence of contamination by location was reported in Patient Administration Rooms, where 28.8% of tests showed some level of contamination (see Table 5). The average level of contamination was also highest in the Patient Administration Rooms at 224.7 ng. (ranging from 5.3 to 3430) The frequency of contamination was lowest in Staging areas (0.6%), while the lowest mean level of contamination (6.6 ng) was observed in Unpacking Areas/Rooms. Higher contamination frequencies observed in Patient Administration Rooms and Infusion Areas are clinically meaningful given the patient-facing nature of these settings; however, these findings represent associations observed within this real-world dataset and should not be interpreted as outcomes directly attributable to CSTD use in the absence of pre-implementation baseline data. The frequency of contamination by location is visually represented in Figure 4.
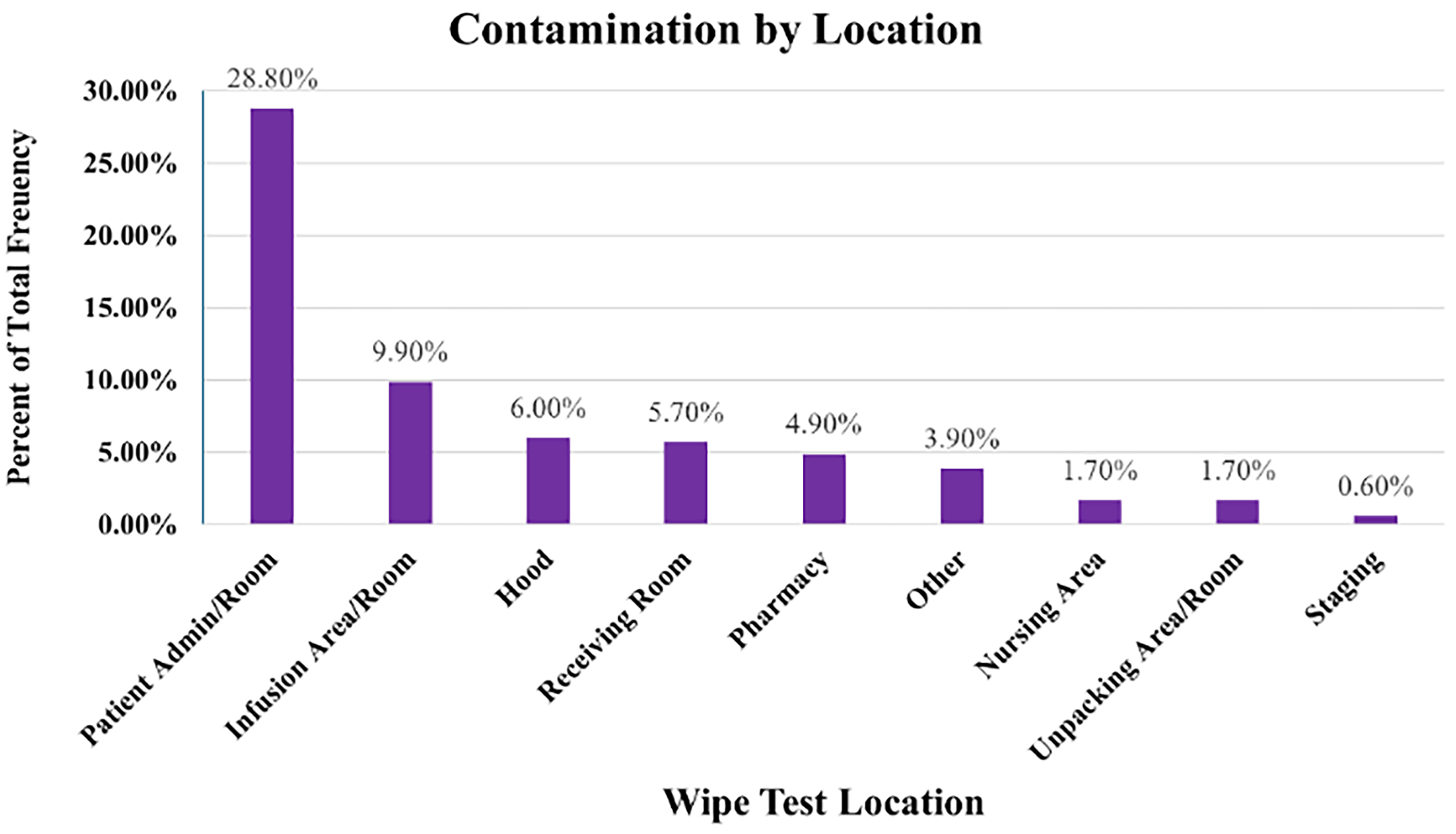
Contamination by location: The highest occurrence of contamination was reported in the patient admin/room, with 28.8% of the tests showing detectable contamination. The average level of contamination was also highest in the Patient Admin/Room (224.7 ng; range: 5.3, 3430.0 ng). The occurrence of contamination was lowest in Staging locations (0.6%), whereas the lowest mean level of contamination was observed in the Unpacking Area/Room (6.6 ng). This data was initially presented in a poster during the 2024 American Society of Healthcare Pharmacists Midyear Clinical Meeting & Exhibition on December 9, 2024.
Contamination by location.
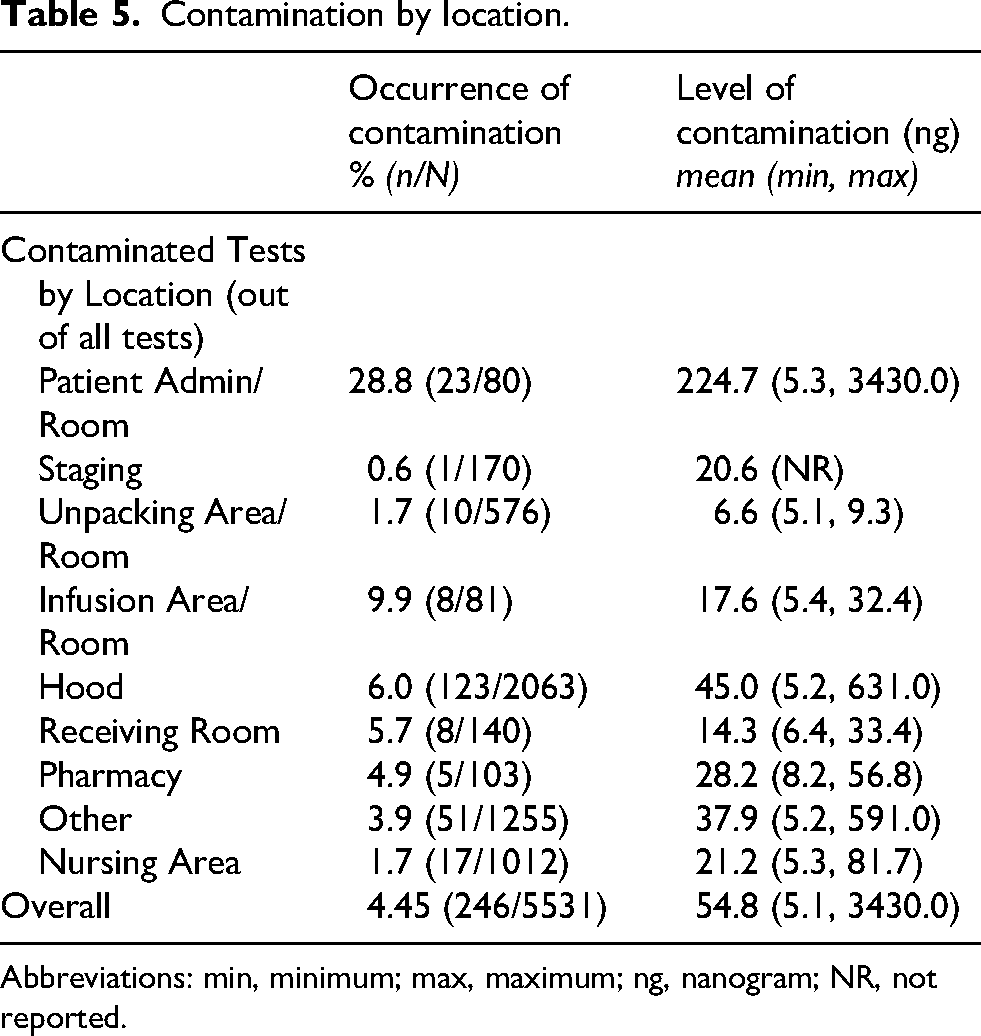
Abbreviations: min, minimum; max, maximum; ng, nanogram; NR, not reported.
Potential covariates
A simple linear regression model was fitted to determine whether a set of predictors (covariates) significantly explained the variance in the level of contamination. The results of the regression model indicated that pooled Hospital System/Outpatient Facility, Analyte, Year of Collection, and Time from Collection to Analysis were not significant predictors of the overall level of contamination (see Table 6). However, the highly significant P-value for Location (P < 0.0001) indicates that certain locations are predictive for HD contamination.
Simple linear regression results for level of contamination.
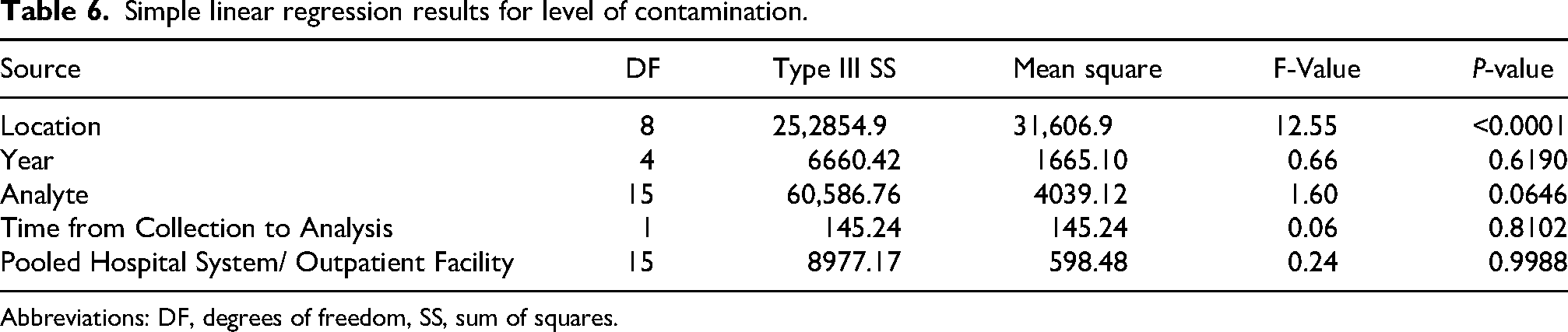
Abbreviations: DF, degrees of freedom, SS, sum of squares.
Discussion
Occupational exposure to HD substances poses numerous health risks to HCPs including, but not limited to, an increased likelihood of developing cancer such as leukemia, and complications in pregnancy with growing concerns regarding the potential transmission of harmful effects from nursing mothers to their infants through breast milk. 4 Currently, there are no certifying agencies nor established standards for acceptable HD surface contamination limits. However, after NIOSH issued a critical alert regarding handling antineoplastics and other HDs in healthcare settings in 2004, new guidelines on the safe handling of cytotoxic and hazardous drugs have been developed by the ASHP and ISOPP. 1 Despite the implementation of such guidelines, studies continue to report that HCPs remain at risk, with measurable concentrations of cytotoxic drugs found in their bodies and on workplace surfaces.5,6 This suggests that more effort is required to improve and harmonize drug handling and surface area cleaning practices. With this concern in mind, this RWD evaluation of HD wipe testing results was performed to better understand the current level of HD contamination at various US based Health Care Systems/Outpatient Facilities.
The use of CSTDs can significantly minimize HD contamination and potential exposure risks in healthcare settings. Results of this evaluation showed variability in surface level contamination across the 18 Hospital systems/Outpatient Facilities, as well as the nine individual locations within these facilities. Overall, of the 5531 wipe analyses, only 246 had measurable levels of contamination with a mean of 54.8 ng and a range from 5.1 ng to 3430 ng. The percentage of contamination by year ranged from 2% to 8% between 2018 to 2022. The Patient Administration Rooms had the highest level of contamination at 28.8%, with the Infusion Area/Room area following at 9.9%.
Higher contamination frequencies observed in Patient Administration Rooms and Infusion Areas may be influenced by several practice-related factors inherent to patient-facing environments. These areas often involve increased HD handling frequency, workflow steps outside controlled pharmacy settings, variability in cleaning responsibility between nursing and environmental services, and bedside administration activities, all of which potentially contribute to detectable surface contamination. These real-world factors highlight the importance of multidisciplinary practices and cleaning protocols in areas where hazardous drugs are administered.
The overall contamination rate observed in this assessment was 4.45%, indicating facilities employing CSTDs effectively minimize workplace surface contamination. The majority of the observed contamination occurred in only two hospital systems, suggesting that institutional cleaning procedures may contribute to overall contamination levels, regardless of implementation of CSTDs in drug handling. Unfortunately, we did not have any baseline data (pre CSTD use) that would have allowed for more precise quantification of the impact of CSTDs on reducing hazardous drug contamination over time, thus more effectively assessing the effectiveness of CSTDs. The concentration of contaminated wipe results within these two hospital systems suggests that institution specific practices, such as cytotoxic drug utilization volume, staffing models, local policies, or environmental cleaning procedures, may influence contamination outcomes. Conversely, the absence of detectable contamination in five hospital systems may reflect effective implementation of local practices, differences in sampling focus, or lower exposure opportunities, rather than absence of risk. As such, system level findings should be interpreted in the context of operational variability rather than uniform CSTD device performance.
Additionally, baseline data would help identify pre-existing problem areas or practices, allowing for targeted interventions and more effective use of CSTDs. Incorporating this baseline data collection into future studies would strengthen the evidence base for CSTD effectiveness and provide valuable insights for continuous improvement in hazardous drug handling practices.
A similar study involving a rigorous, standardized protocol at 13 cancer centers across the US demonstrated that CSTD use reduced surface contamination by cyclophosphamide and fluorouracil analytes during compounding and simulated administration with participants reporting that the device was easy to use, corroborating the role of the CSTD technology in enhancing safety. 5
Another study evaluated HD surface contamination in pharmacy and nursing areas, focusing on cleaning techniques and CSTDs. A total of 90 samples were collected over three timepoints (initial, 3 months, 6 months). Results showed that all samples were negative for HD residue, indicating effective contamination control. The study suggested standardized work practices and CSTDs help maintain reduced HD contamination. 11
Despite the apparent efficacy of CSTDs, variations in contamination levels across different facilities demonstrate the impact of inconsistent surface cleaning standards and HD handling practices. Facilities with robust and standardized cleaning protocols reported significantly lower contamination rates compared to those with less stringent practices. There is a need for clear, consistent, and effective cleaning procedures across all areas where HDs are handled. The healthcare industry would benefit from establishing clear standards for acceptable levels of HD contamination. Regular wipe testing and analysis should be implemented to identify and address contamination issues promptly. This emphasizes the urgent need for healthcare facilities to evaluate and revise their specific cleaning practices and HD handling processes in accordance with best practices.
Longitudinal studies such as those conducted by Sessink et al. 2023 highlight how standardized protocols and immediate cleaning after drug administration can further minimize contamination levels, stressing the importance of timing and technique in cleaning procedures. 12 In addition, the prevalence of contamination among specific HDs, such as 5-Fluorouracil, indicates that while CSTDs are effective, tailored cleaning strategies directed at specific HD analytes are necessary for comprehensive contamination management.
Because participating facilities had already established routine environmental wipe testing prior to CSTD implementation, the results may not be generalizable to hospitals that handle hazardous drugs but do not perform routine environmental monitoring. Consequently, findings may reflect practices at institutions with a higher emphasis on environmental monitoring and should be interpreted accordingly when considering broader generalizability.
Limitations
This RWD analysis was based on retrospective real-world data and therefore reflects inherent variability with routine clinical practice. Differences in local cleaning practices, environmental service responsibilities, and adherence to institutional protocols may have contributed to observed variability in contamination levels across facilities and locations. In addition, wipe sampling techniques were not fully standardized across sites; while analytical methods and testing intervals were consistent, the number of wipes collected, surface types sampled, and sampled surface areas varied according to facility-specific practices, which may impact comparability of results.
Furthermore, pre-implementation (baseline) or comparator wipe-testing data were not available. As a result, the analysis does not permit direct assessment of changes in contamination levels before and after CSTD implementation and thus limits causal inference. Nevertheless, the findings provide insight into hazardous drug surface contamination patterns observed during routine clinical use of a CSTD and support evaluation of real-world effectiveness under non-controlled practice conditions.
Conclusion
The low contamination rate of 4.45% identified in this assessment is promising and showcases the effectiveness of a proprietary CSTD during routine clinical use. Independent analyses conducted by both the sponsor and an external statistical vendor yielded consistent results, supporting the robustness of the findings. Although a wide range of contamination levels was observed across various facility locations, contamination was predominantly localized to Patient Administration Rooms and Infusion Areas. These observations highlight the importance of continued attention to handling practices and environmental controls in patient-facing administration locations.
Though pre-implementation baseline or comparator data were not available to permit direct causal assessment of change over time, the observed contamination patterns provide meaningful insight into HD surface contamination under real-world practice conditions. This assessment emphasizes the importance of CSTDs as a critical engineering control, along with continued vigilance and ongoing improvement in hazardous drug handling practices across the healthcare industry. By prioritizing a comprehensive approach that includes standardized wipe sampling protocols, clear cleaning guidelines, targeted interventions in higher risk areas, and continued environmental monitoring, healthcare systems can further enhance existing safety measures that reduce healthcare professional exposure to HD contamination.
While this assessment highlights the importance of CSTDs, continued efforts to standardize wipe sampling methodologies, optimize cleaning practices, and maintain routine environmental monitoring may further enhance existing safety measures aimed at reducing healthcare professional exposure to hazardous drugs. Consistent and appropriate use of CSTDs remains an important component of hazardous drug handling practices. Additional prospective studies are warranted to better characterize factors contributing to variability in surface contamination across different healthcare settings and to further inform best practices under routine clinical conditions.
Additional information
This work was presented in part as a poster at the 2024 American Society of Healthcare Pharmacists Midyear Clinical Meeting & Exhibition on December 9, 2024.
KEY POINTS
The 3 key point from this manuscript are:
This study analyzed over 5000 wipe test samples from US healthcare facilities to access hazardous drug (HD) contamination following the use of the OnGuard closed system transfer device (CSTD). Results showed an extremely low contamination of 4.45%, primary coming from 2 hospital system and from the Patient Admin/room location (28.8%). The utilization of a CSTD at the point-of-care could decrease HD exposure for healthcare workers.
Footnotes
Acknowledgments
Medical writing assistance was provided by Shelley Wells, MAS Clinical Research, of Fortrea Inc., and was funded by B. Braun Medical.
Ethical approval statement
In accordance with institutional policies and applicable regulations, this study did not require Institutional Review Board (IRB) approval, as it did not involve human subjects research as defined by federal regulations. All data were fully anonymized prior to analysis, and no identifiable information was accessed or used.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This evaluation was funded by B. Braun Medical Inc.
Declaration of conflicting interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B. Braun Medical Inc. funded the research. Akshar N. Patel, PharmD, RPH, Marie-Claude Gutekunst, MSN, RN, BCMAS, Diana Valencia, MD and Christopher R. Curtin BS, are employees of B. Braun Medical Inc. Kaisa Kivilaid is an employee of Fortrea. B. Braun Medical and Fortrea independently performed the analysis and were involved in the preparation of this manuscript.
Data availability statement
The data underlying this article cannot be shared publicly due to proprietary agreements with the participating facilities
