Abstract
Introduction
Peripherally acting
Methods
This is a retrospective study of 79 pediatric patients with opioid induced constipation. Patients were administered methylnaltrexone during their inpatient stay. Data on bowel activity after methylnaltrexone was obtained from the hospital information system.
Results
Out of the 79 patients who received methylnaltrexone, there were seven patients from whom data could not be analyzed. Of the 72 patients whose data was available, 38% (
Conclusion
Methylnaltrexone appears safe in the pediatric population and produces bowel movements in more than a third of pediatric patients. It is a feasible and safe option for opioid induced constipation in pediatric patients.
Introduction
Opioid induced constipation (OIC) is a distressing side effect that is experienced by greater than 50% of patients taking opioids.1,2 It is even more distressing in pediatric patients as treatment options are limited given that the safety and efficacy studies of most agents for OIC have not been established in such a vulnerable patient population.3,4 Most side effects experienced by patients on opioid therapy tend to subside over time, but constipation persists. 5 Pharmacotherapeutic options used to treat OIC include methylnaltrexone, alvimopan, naldemedine, and naloxegol.6–8
Methylnaltrexone is a peripherally acting opioid antagonist that has an approved indication by the Food and Drug Administration (FDA) for opioid agonist induced constipation in adults for both non-cancer pain and in advanced illness.5,9–12 There are currently no safety and efficacy data in children and adolescents, although it has been used in this population in clinical practice. The purpose of this retrospective study is to evaluate methylnaltrexone use in children and adolescents on opioid agonist therapy admitted inpatient at the pediatric unit of a cancer center.
The objectives of this study are to determine the efficacy and safety of methylnaltrexone for opioid agonist induced constipation in pediatric patients with cancer. The primary outcome is the efficacy of methylnaltrexone for opioid agonist induced constipation measured by the production of a bowel movement within 24 h after administration of the first dose of methylnaltrexone. Other secondary outcomes include the production of a bowel movement after successive administrations of methylnaltrexone and documentation of types of medication related adverse effects.
Methods
This study was in the inpatient setting of a pediatric unit at an academic hospital. Patients were ≤ 18 years and were administered methylnaltrexone for opioid-induced constipation. The Institutional Review Board at the academic hospital approved the study.
A search in the hospital information system was programmed to run a query in the computer information system to identify pediatric patients ordered and administered methylnaltrexone from 1/1/2015 to 5/31/2019. Patient charts were retrospectively reviewed to collect oral morphine equivalents (OME) of opioids administered, methylnaltrexone administration dates, doses administered, number of bowel movements, medications for constipation prophylaxis and/or constipation (e.g. senna, docusate, lactulose, and polyethylene glycol) and adverse reactions. The number of bowel movements produced within 24 h after methylnaltrexone administration was collected for all patients. Patients who did not have a bowel movement within 24 h after the first dose of methylnaltrexone, and who received consecutive doses on each additional day were monitored for bowel movements within 24 h of the new dose.
Statistical analysis
The patients’ characteristics and outcomes were described using median (range) for continuous variables, and the frequency
Results
A total of 79 patients were administered weight-based methylnaltrexone doses during the period of interest. All patients had received constipation prophylaxis medications or medications to treat existing constipation: senna, docusate, lactulose, and polyethylene glycol. The median age was 14.0 years (range, 1.1 to 18.0 years, Table 1). Most of the patients in this cohort received 0.15 mg/kg, although not all, as some patients received doses that were higher or lower than the planned 0.15 mg/kg (see Supplemental Table 1 for detailed data on doses). The median administered dose was 8.00 mg (range, 0.75 to 12.00). OME range for patients was 3–1791 mg. OME range for responders was 10.4–960 mg and for non-responders was 3–1791 mg. In the 24 h following methylnaltrexone administration, 27/72 patients had a bowel movement, resulting to an estimated rate of 38% (95% CI: 26 to 50%). The information on bowel movements was not retrieved for seven patients as it was not available in the patient's chart, but information regarding adverse events was recorded.
Patient characteristics.

a Median (Range);
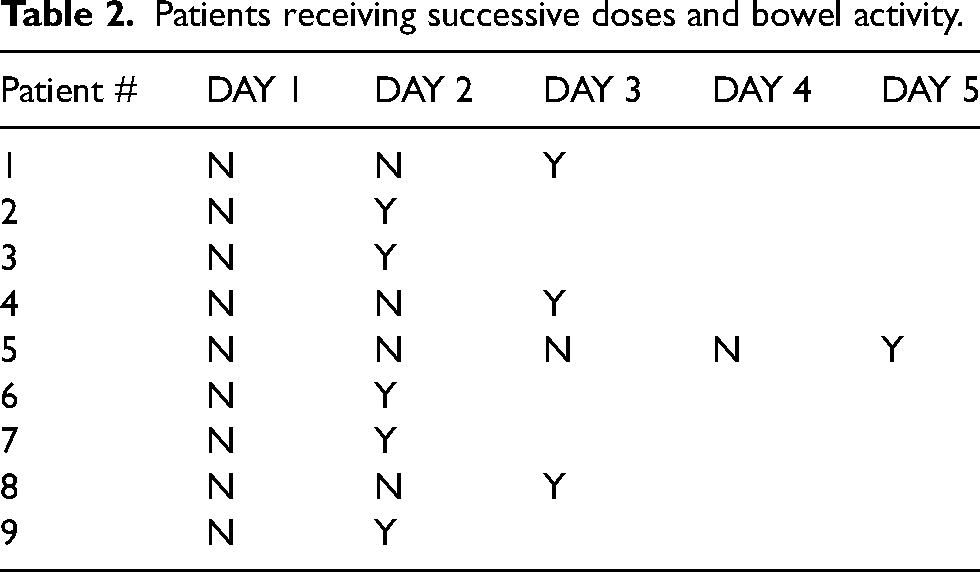
A subset of nine subjects who did not have a bowel movement after the first dose of methylnaltrexone, received repeated doses of methylnaltrexone, which eventually resulted in bowel movements by each subject (Table 2). All patients who received successive doses of methylnaltrexone (Table 2) ultimately had a bowel movement within 5 days of successive doses daily. Out of the nine subjects, 5 (56%) experienced a bowel movement after receiving a second dose of methylnaltrexone on the second day after the previously administered methylnaltrexone. One subject received two doses of methylnaltrexone on the same day 6 hours apart. This subject did not have a bowel movement; however, they were administered methylnaltrexone daily for two more days and recorded having a bowel movement on the third day from first administration of methylnaltrexone (patient #8 in Table 2). Therefore overall, out of the 72 patients, 4 (6%) needed 3 or more consecutive daily doses of methylnaltrexone in order to experience a bowel movement.
Patients receiving successive doses and bowel activity.
There was no evidence of a difference in the distribution of the dose in mg between patients with and without bowel movement (Mann–Whitney–Wilcoxon test,
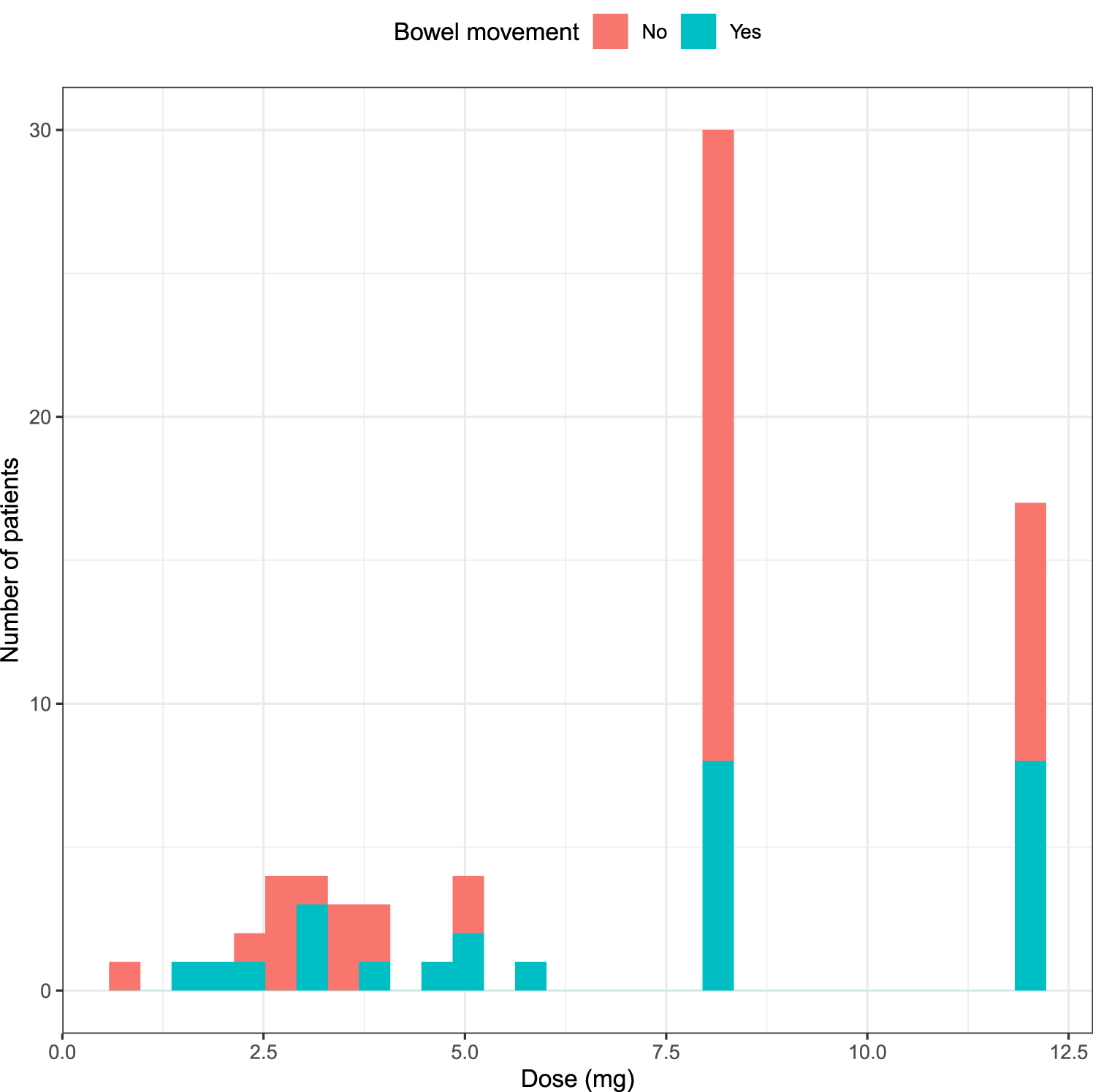
Distribution of dose according to bowel movement.
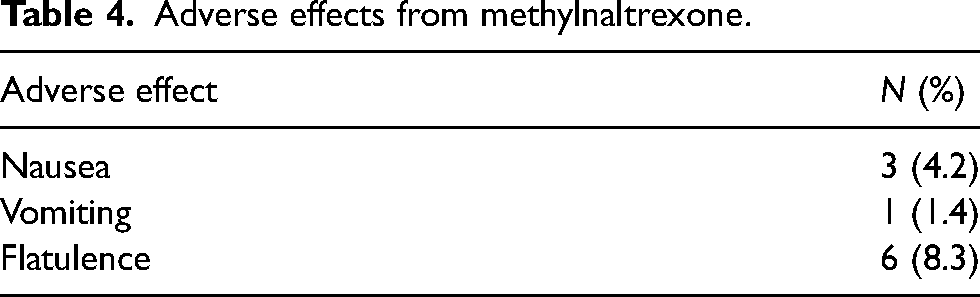
Overall, only 10 adverse events that maybe related to methylnaltrexone were observed, in 9/79 (13%, 95% CI: 6 to 22%) patients.
Discussion
There have been a few small studies on the use of PAMORA's in pediatric patients.13–18 There was the report of a case of an 8-year-old female with neuroblastoma who was successfully treated with subcutaneous methylnaltrexone for OIC. She had a bowel movement within 10 min of administration of methylnaltrexone. 13 A severe case of OIC was resolved using enteral methylnaltrexone in a 4-year-old male with epidermolysis bullosa related pain. 14 Another retrospective study reviewed methylnaltrexone use in nine pediatric patients with OIC and determined it to be safe and efficacious: 78% (seven patients) achieved laxation after methylnaltrexone administration with the first dose and 71% who responded had a continuous response to repeated doses. 15 Another retrospective study reviewed methylnaltrexone in 15 pediatric patients and determined that bowel movement occurred within 4 hours in 70% of doses administered. 16
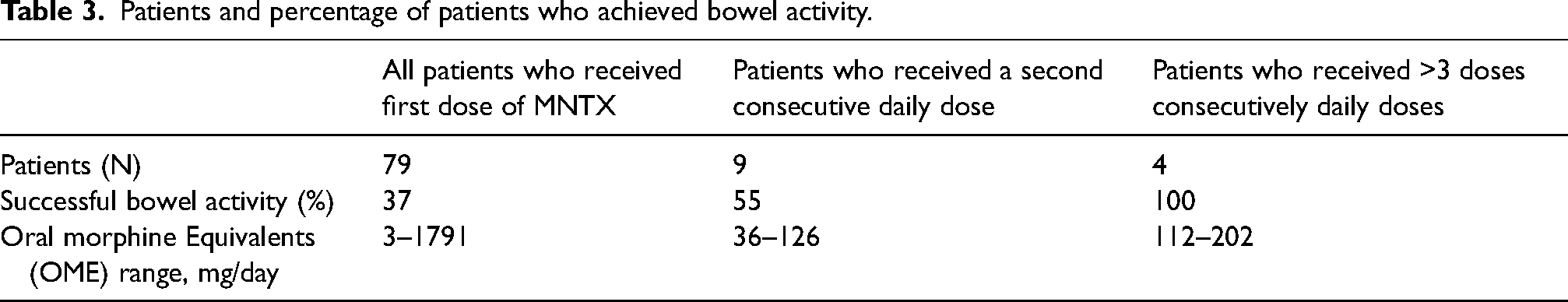
To the best of our knowledge, this is the largest study with 79 pediatric patients diagnosed with opioid-induced constipation who received subcutaneous methylnaltrexone during their inpatient admission stay. Seven patients were not included in the efficacy analysis because there was no information available about their response after methylnaltrexone was administered. In the 24 hours after receiving one dose of methylnaltrexone, 37.5% of patients were documented as having a bowel movement. We did not find a correlation with the dose. Nine patients who did not have a bowel movement after the first dose but received additional successive daily doses of methylnaltrexone demonstrated that bowel activity may be reasonably expected in the majority of patients with at least three daily doses (Tables 3 and 4). This occurrence may indicate that patients with OIC with no bowel obstructions may benefit from successive doses of methylnaltrexone in clinical practice after failure to respond after only one dose.
Patients and percentage of patients who achieved bowel activity.
Adverse effects from methylnaltrexone.
Prior to receiving methylnaltrexone, all patients suffering from OIC were on a bowel regimen (constipation prophylaxis) comprising of agents in the following classes i.e. stool softeners (docusate sodium), stimulant laxatives (senna), osmotic laxatives (lactulose and polyethylene glycol). Patients remained on these agents when methylnaltrexone was initiated. These agents were not further titrated after initiation of methylnaltrexone.
Adverse effects reported were minimal (Table 4). Nausea reported in one subject was persistent prior to the methylnaltrexone administration and post administration of methylnaltrexone. Four subjects were reporting nausea prior to administration of methylnaltrexone. The causes of nausea are multifactorial and may be from, but not limited to disease state, constipation, and methylnaltrexone. 17
As mentioned earlier, the recommended dose of methylnaltrexone in the literature and in drug databases does not include dosing for pediatric patients because safety and efficacy data has not been established in this population. The patients in this study tolerated the dose of methylnaltrexone with minimal adverse effects. For dosing, some studies in the literature that used methylnaltrexone dosing in pediatric patients used 0.15 mg/kg. 18 This weight-based dosing of 0.15 mg/kg was observed to have been used for most study patients.
Daily opioid use varied amongst study subjects (range of 3–1791.36 mg oral morphine equivalents (OME) per day). The equianalgesic calculations were approximated using equianalgesic ratios from the National Comprehensive Cancer Network Adult Cancer Pain Guidelines. 19 The amount of OME was based on a patient's long-acting opioids only to offer an approximate dose of opioids that were consistently being consumed by patients.
Some limitations of this study include the fact that the patients administered methylnaltrexone were followed 24 h post dose for a bowel movement. Therefore, bowel movements that occurred after 24 h post dose were not collected and documented. Additionally, while all study patients were administered constipation prophylaxis medications or medications to treat constipation, the exact doses of these medications were not measured.
Based on our study results, methylnaltrexone use in pediatric cancer patients with opioid induced constipation appears safe and effective. Some patients may need to receive consecutive doses of methylnaltrexone to achieve bowel activity and movement. We would recommend future prospective studies in pediatric patients on the use of PAMORAs to confirm those observations.
Supplemental Material
sj-docx-1-opp-10.1177_10781552231163540 - Supplemental material for The safety and efficacy of methylnaltrexone in pediatric oncology patients: A single center experience
Supplemental material, sj-docx-1-opp-10.1177_10781552231163540 for The safety and efficacy of methylnaltrexone in pediatric oncology patients: A single center experience by Lauren Koranteng, Audrey Mauguen, Miriam Butler, Shirley Qiong Yan and Damani Taylor in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
We wish to thank member of the DigITs- Technology Division for assistance with access to records for research purposes.
Author contributions
LK conceived the study. SY and LK worked on protocol development and ethical approval. LK wrote the first draft of the manuscript. All authors were involved in data analysis, review, editing and final approval of manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Health, (grant number CA 008748).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
