Abstract
Introduction
Opioid-induced constipation is a clinically relevant side effect and a cause of potentially avoidable drug-related hospital admissions.
Objectives
To describe the presence of laxative co-medication, the reasons for not starting laxatives and to evaluate changes in stool patterns of opioid initiators.
Methods
In this observational study community pharmacists evaluated the availability of laxative co-medication in starting opioid users and registered reasons for non-use. Two opioid initiators per pharmacy were invited to complete questionnaires (‘Bristol stool form scale’ and ‘Rome III Diagnostic Questionnaire for the Adult Functional Gastrointestinal Disorders’) on their defecation prior to and during opioid use. Descriptive statistics and Chi square tests were used to analyse reasons for non-use of laxatives and changes in defecation patterns.
Results
Eighty-one pharmacists collected data from 460 opioid initiators. Of those, 344 (74.8%) used laxatives concomitantly. Main reason not to use laxatives was that either prescribers or patients did not consider them necessary. Sixty-seven (89.3%) of the 75 opioid starters with two questionnaires completed were not constipated at opioid start. Eleven of them (16%) developed constipation during opioid use (Chi square p=0.003). At follow-up within laxative users 10.6% were constipated compared to 20.7% in subjects without laxatives.
Conclusion
One in four opioid starters did not dispose of laxative co-medication, mainly because they were not considered necessary by either the prescriber or the patient. The prevalence of constipation doubled during opioid use. A watchful waiting strategy for the use of laxative co-medication might include a monitoring of defecation patterns with validated questionnaires.
Introduction
Opioids (e.g. morphine, oxycodone and fentanyl) are effective analgesics, but may cause side effects, such as drowsiness, nausea, decreased gastrointestinal (GI) motility, increased GI transit time and subsequent constipation.1,2 Opioid-induced constipation (OIC) has been reported in approximately 90% of opioid users,2–5 after even a single dose of morphine. 2 According to the Rome III Diagnostic Criteria for Functional Constipation, this condition includes a number of symptoms, such as hard stools, infrequent stools (<3 per week), incomplete bowel evacuation or unsuccessful defecation and the need for excessive straining. 6 Several risk factors to develop OIC have been reported as more medical comorbidities or daily medications, dehydration, co-medication reducing bowel activity, physical abnormalities, decreased physical mobility, low fibre and fluid intake and reduced privacy.1,3,7–9 Age might not be a factor, as a recent study showed that within the first week after initiating opioid therapy for chronic non-cancer pain, constipation occurred comparably in younger and older patients with 32.8 and 35.9%, respectively. 10
OIC was reported as an important cause of potentially avoidable drug-related hospital admissions for between 4.6 and 6.6% of all potentially preventable drug-related hospital admissions.11–13 Moreover, OIC has been associated with an increase in overall work impairment. 4 To avoid serious side effects and as treatment of an already manifest OIC is more difficult, concomitant prophylactic use of laxatives is advised from the start and throughout the duration of opioid treatment.3,14–18 The practice of prescribing opioids without concomitant laxatives as a prophylaxis and for symptom control is regarded as inappropriate.1,14,19 From laxatives available, osmotic agents (e.g. lactulose and macrogol) are the preferred choice to prevent OIC.7,20
As OIC is preventable, healthcare providers should take care of prophylaxis against constipation when opioid therapy is prescribed. Consequently, prescribers as well as pharmacists are expected to counsel patients on symptoms and potential consequences of opioid side effects and to advocate laxative use in combination with opioids. Pharmacist-led interventions were shown to increase the degree of laxative co-medication. 21
Nevertheless, a cross sectional study in the United States reported a monthly prevalence of concurrent opioid and laxative use of only 55% in opioid users. 1 This percentage equals the mean percentage of opioid users with concomitant laxatives measured as a quality indicator by national pharmacy dispensing data in the Netherlands. 22 Since the start of measurements in 2008 the score for laxative co-medication in opioid users remained constantly low. 22 This was in contrast to the scores of other quality indicators such as the degree of gastro-protection in susceptible users of non-steroidal anti-inflammatory drugs, which improved to above 80% in the same period. 22 Community pharmacists gave a number of reasons for their low scores on this quality indicator such as chronic diarrhoea which made the use of laxatives superfluous. Or they explained their low scores with laxatives from other sources than from their community pharmacy dispensing the opioid. According to a questionnaire among 211 GPs in the Netherlands nearly half of them did not prescribe a laxative because the patient refused their use. 13 One-third of the GPs stated that the patients could not take laxatives. 13 However, this does not meet the evidence and recommendations from literature. 3 One-fifth of the GPs answered that they would reconsider laxatives later during the treatment, although all of them were aware of the guideline recommendations to start prophylaxis immediately. 13 Information is lacking whether patients were informed on opioid side effects and the benefits of laxatives to take a well-informed decision. Reasons not to use laxatives have not been evaluated in community-dwelling patients at the start of opioid treatment. Additionally, data on changes of defecation patterns in community-dwelling patients after opioid initiation are not available.
Therefore, this study evaluates the prevalence of laxative co-medication at opioid treatment initiation with reasons not to dispense or use laxatives and assesses the change in defecation patterns of opioid initiators in relation with laxative co-medication.
Methods
Study design
This study is a prospective observational study on the use of laxative co-medication in patients with a first opioid dispensing of a community pharmacy in the Netherlands. A first opioid dispensing was defined as no dispensing of any opioid to a patient within the previous 12 months.
All 1981 community pharmacies in the Netherlands were invited to participate in this study by means of the weekly electronic newsletter of the Royal Dutch Pharmacists Association and the website. Additionally, during the national pharmacists' congress in October, pharmacists were personally invited by leaflets and personal contact. The researchers also invited pharmacists from their own network.
During the study period of in total two weeks, either in November or in December 2016, participating community pharmacists checked the presence of a concomitant laxative prescription for opioid initiators. If no laxative was prescribed together with the opioid, the availability of a laxative at home was questioned and registered, otherwise laxative use was advised. Reasons for not following this advice were noted. Two of these patients per pharmacy were invited to fill questionnaires on their defecation patterns prior and during opioid use.
Setting
In the Netherlands, pharmacists register all dispensed medicines in their pharmacy information system.
Due to their legal and professional shared responsibility for the safe and effective use of medication, 23 pharmacists regularly contact prescribers when prescriptions may not be in accordance with prescription. The majority of GPs and community pharmacists regularly discusses prescribing guidelines in pharmacotherapy audit meetings. 24
In the Netherlands, patients generally get their medication from one pharmacy. 25 Moreover, patient medication records are exchanged between pharmacies if patient's consent was provided. Thus, community pharmacists generally dispose of comprehensive files on the medication of their patients. To support clinical risk management, in 2015 an automated clinical rule (CR) concerning the use of opioids without laxatives was developed and implemented in the pharmacy information systems. A decision tree within the CR provides automated advice on relevant process steps and the possibility to register the choices taken.
Ethical approval
The Medical Research Involving Human Subjects Act did not apply to this study, according to the Medical Research and Ethics Committee Nijmegen (‘2016-2918’). The Institutional Review Board for practice research of the University of Utrecht also evaluated the study and granted approval (UPF1613).
Data collection
Degree of laxative co-medication and reasons not to dispense or use it
Pharmacists were asked to collect information on all first opioid dispensing for two weeks either during November or the first two weeks of December 2016. These weeks were chosen at the beginning of the internship of the first author and just before expected extra work load before year's end. According to national dispensing data, on average three subjects started opioids weekly per community pharmacy. Consequently, on average six starters per pharmacy were expected for the two weeks lasting study period.
For data collection, participating pharmacists received a printed decision tree within the structure of the automated CR of the pharmacy medication surveillance system (Figure 1). This decision tree is implemented in the daily workflow of community pharmacies and known to the pharmacy team. Beside the CR additional information was collected on patients' age, sex and the dispensed opioid (chemical substance, dosage). If a laxative was dispensed, information on the type of laxative and dosage was registered. In case of no laxative dispensing, pharmacists could register the reason for this by choosing between a number of predefined options and with the possibility to add other reasons.
Decision tree to register laxative use in starting opioid users.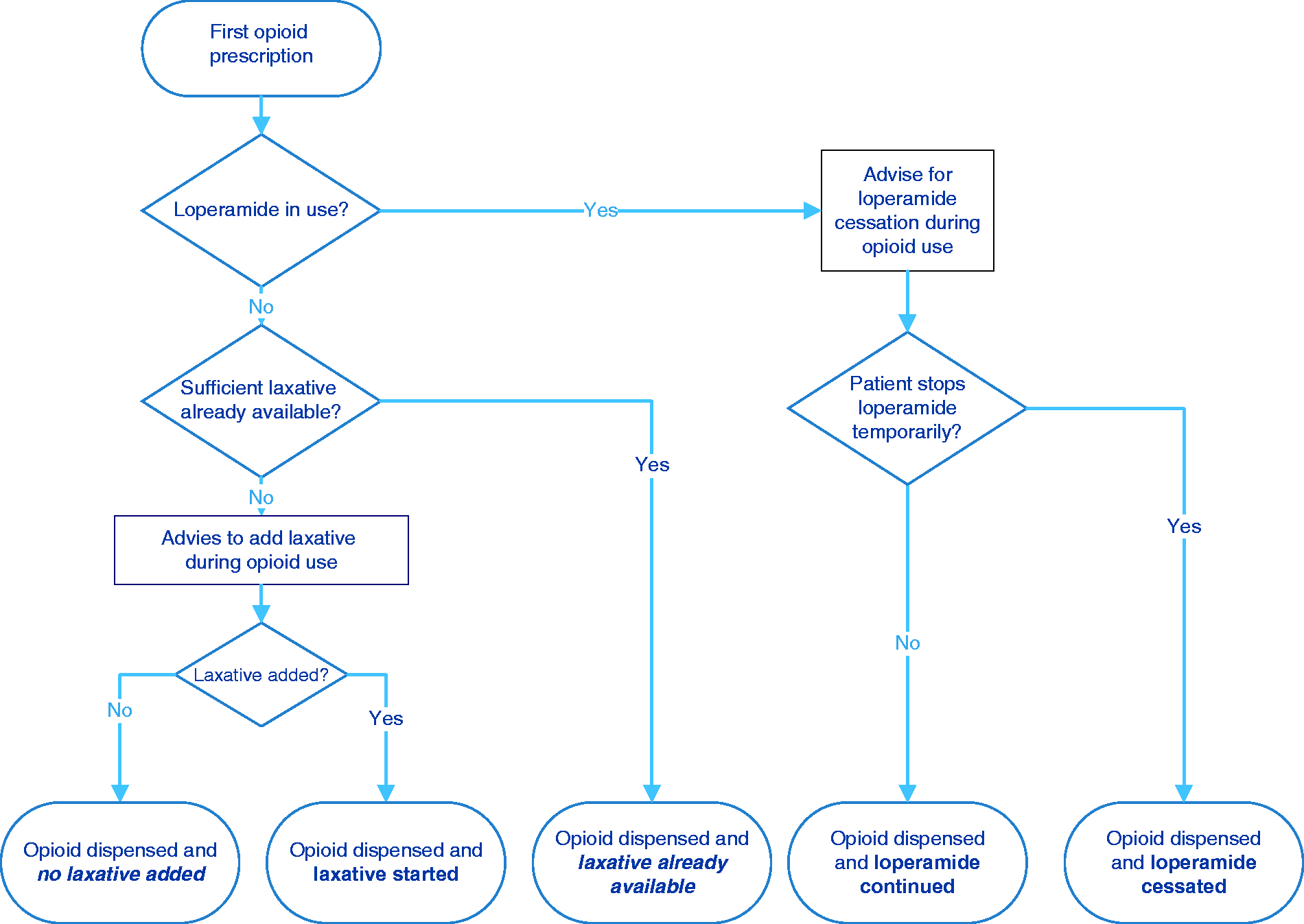
Changes in constipation patterns after opioid initiation
Starting opioid users of 18 years and older, able to understand a questionnaire in Dutch and with opioid use for at least three consecutive days, were invited to complete questionnaires on their defecation patterns. Pharmacists invited patients until two patients per pharmacy were willing to consider participation, following the sequence of offering a prescription in the community pharmacy. Patients were provided with oral and written information on the study, an informed consent form and two paper questionnaires. Participants had to complete two constipation questionnaires. The first one had to be completed before the start of opioid treatment. The second one had to be completed at the end of at least three days or after a maximum of two weeks of subsequent opioid use.
Beside general patient information on age, sex and the specific opioid initiated, the questionnaires were composed of the ‘Bristol stool form scale’ and the ‘Rome III Diagnostic Questionnaire for the Adult Functional GI Disorders’.6,26 The ‘Bristol stool form scale’ is an instrument with images of stool types ranging from the hardest (type 1) to the softest (type 7). Type 1 and type 2 are described as hard stools (constipation) and type 6 and 7 as abnormally loose stools (diarrhoea). Type 3, 4 and 5 are considered as the most normal stool forms. 26 The ‘Rome III Diagnostic Questionnaire for the Adult Functional GI Disorders’ comprises specific questions on aspects related to constipation such as the frequency and composition of defecation and the need to press. 20
Constipation as the main outcome for the change in defecation patterns was determined as being constipated as well according to the criteria of the ‘Bristol stool form scale’ as to the ‘Rome III Diagnostic Questionnaire for the Adult Functional GI Disorders’. This implied that a patient had to score ‘Type 1’ or ‘Type II’ in The Bristol stool forms scale and at least two of the following criteria from the Rome III questionnaire: (a) Straining during at least 25% of defecations, (b) lumpy or hard stools in at least 25% of defecations, (c) sensation of incomplete evacuation for at least 25% of defecations, (d) sensation of anorectal obstruction/blockage for at least 25% of defecations, (e) manual manoeuvres to facilitate at least 25% of defecations (e.g. digital evacuation, support of the pelvic floor) and (f) fewer than three defecations per week. 6 As both questionnaires are validated on existing tools, a pilot on their use was not necessary.
Data analysis
Degree of laxative co-medication and reasons not to use laxatives
Descriptive statistics were used for patient characteristics, opioid use and reasons for not dispensing laxative co-medication. The proportion of opioid users with laxative co-medication was calculated and stratified for different opioids, administration route and prescribed daily dose (PDD) of the opioid dispensed.
Change in patient defecation patterns before and during opioid use
Constipation was defined as being constipated according to as well the Rome III criteria as the Bristol stool form scale.6,26 Chi square tests were used to test on changes in defecation patterns before and after opioid initiation. Numbers and percentages of patients with constipation at study start and during follow-up were calculated, stratified by the use of laxatives (yes/no).
Results
Characteristics of starting opioid users and reasons for refusing laxative co-medication.

Handling of co-dispensing laxatives in opioid initiators was registered by 81 community pharmacists.
For 116 opioid starters refusing laxative co-medication in total 143 reasons for refusal were recorded.
Table 1 shows that 137 (30%) of the 460 patients initiating opioids already disposed of a laxative, sufficient to cover the period of opioid use. For 48 patients (10%), the use of laxative medication was unknown from pharmacy records, and thus patients used laxatives from other sources than the community pharmacy, that dispensed the opioid.
Of the remaining opioid starters, 207 (45.0%) received a laxative concomitantly. In total 344 (74.8%) of the starting opioid users had a laxative available as co-medication during their opioid use, either due to laxatives already at home or due to a new dispensing together with the opioid. Consequently, one in four starting opioid users did not dispose of a concomitant laxative during the first period of opioid use. From these 116 subjects (25.2% of all patients included) without a laxative, a total of 125 reasons not to use it were collected and shown in Table 1. Main reasons to refuse laxatives were patients' doubts on their necessity (44.1 %) or because of a waiting policy of patients on signs of OIC before starting a laxative (32.4%). In 20.0% of the patients the prescribers judged a laxative as not necessary because of short term or on demand use. Co-medication with loperamide was the first question in the CMR decision tree as an indication for existing diarrhoea and a possible reason for no laxative dispensing. This was only mentioned by one patient (0.2%). Detailed information on laxative co-medication stratified for opioid substance, administration route and mean PDD of the opioid is shown in supplement 1. Users of fixed combinations of different opioids and users of fentanyl tablets or nasal spray, buprenorphine tablets or morphine suppositories all disposed of laxative co-medication (100%). However, the absolute user numbers were small with 1–3 users per opioid formulation. Only the users of higher dosages of morphine tablets (from 24 mg) or oxycodone tablets (from 60 mg) or fentanyl transdermal patches (from 180 mcg) all used laxatives. In users of lower dosages of these opioid preparations, percentages of laxative co-medication were lower, being lowest with 62.5% for users of morphine tablets between 5 and 20 mg. The lowest percentage was detected in users for morphine drops with only half of them using laxative co-medication.
Defecation patterns prior to opioid initiation and during opioid use.

Patients were included by 50 community pharmacists.
BSF: Bristol Stoel Form.
Change in patients' defecation patterns during opioid use.
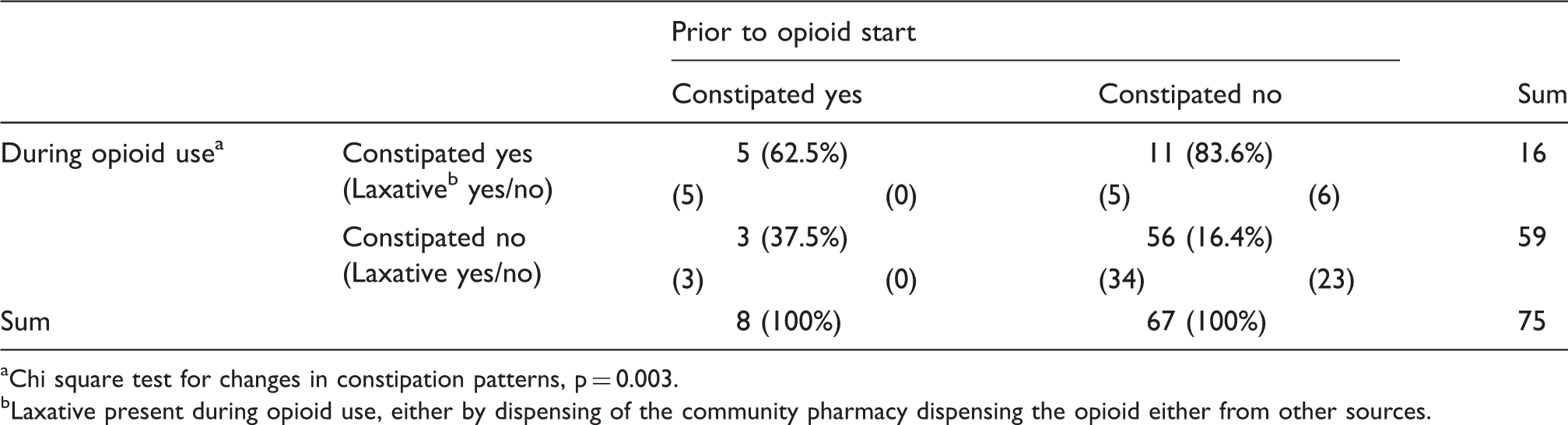
Chi square test for changes in constipation patterns, p = 0.003.
Laxative present during opioid use, either by dispensing of the community pharmacy dispensing the opioid either from other sources.
Discussion
Our study showed that one in four starting opioid users did not dispose of a concomitant laxative during the first period of opioid use. The corresponding percentage of 75% opioid users with concomitant laxative is higher than the 55% reported by dispensing or prescription data only,1,22 but still seems too low to meet the recommendations of current guidelines. Registrations of participating pharmacists revealed that 77% of the opioid starters without laxative co-medication did not regard laxatives as necessary and decided to start opioids without preventive co-medication.
To meet the guidelines recommendations, there seems to be room for the education of prescribers, pharmacists and patients on opioid side effects. However, our results also show that only 20.7% of the opioid users without laxative actually developed constipation. Thus, a watchful waiting strategy might be an option provided that the stool patterns are regularly monitored for constipation development. Both prescribers as well as pharmacists have a professional task in patient counselling to prevent OIC development and related hospital admissions. In this the use of constipation questionnaires might help patients to monitor a deterioration of their defecation and signalize the development of constipation. In our study, two validated constipation questionnaires, the ROME III and the BSF criteria, were used to provide specific results for OIC. Our results showed statistically significant worsening of defecation patterns during opioid use from 10.7% being constipated before opioid intake to 21.3% with constipation during opioid use.
The effectiveness of laxatives has already been shown before by large studies.27–30 However, in our population within laxative users during opioid use still 10.7% were obstipated. This might be due to non-adherence to laxatives or a suboptimal dosage of the laxative.
Some limitations have to be taken into account when interpreting our study results. Only 4% of the community pharmacies in the Netherlands participated. Although available communication sources were used intensely, no higher inclusion than 81 pharmacies could be achieved. Participation was voluntary, and it is possible that participating pharmacists were more motivated to prevent OIC and the percentage of laxative co-medication reported here might be higher compared to other pharmacies. Indeed, the percentage of starting opioid users together with a laxative we found (75%) was much higher than the 55% measured by the annual national assessment of quality indicators from all community pharmacies in the Netherlands. 22 Although 10% in difference were due to laxative availability not covered from dispensing data, this could not fully explain the higher scores of the pharmacists participating in this study. For the changes in defecation patterns, 75 patients returned constipation questionnaires before and during opioid use. The expectation to include two patients per participating pharmacy was not reached, and only 50 (62% from 81 participating) community pharmacies managed to include patients for answering the defecation questionnaires. Patient participation was also voluntary. As patients had to be able to read and understand the Dutch language, patients with low health literacy were missed. This group might be at a higher risk for opioid side effects because they cannot afford laxative co-medication or might have poor fibre intake. Thus, our findings might underestimate the risk of OIC development during opioid initiation. Additionally, terminally ill patients were not reached by our study, and thus our findings do not cover these patients. Furthermore, the scores on the constipation scales were self-reported and estimation might differ between patients. However, constipation prior to and during opioid use was compared on patient level, and a bias for changes during opioid use seems to be limited.
A strength of our study was the high amount of CR decision trees returned from participating pharmacies. The 460 forms returned nearly met the expected numbers of six starters during a fortnight per pharmacy (95%). As participating pharmacies came from all over the Netherlands, we have no selection bias on region or urbanity.
In conclusion one in four opioid starters did not dispose of a concomitant laxative, mainly because they were not considered necessary by patients. The prevalence of constipation increased during opioid use and was higher in subjects without laxative co-medication. A watchful waiting strategy for the use of laxative co-medication might include a monitoring of defecation patterns with validated questionnaires.
Key notes
One in four opioid starters did not dispose of a concomitant laxative. Main reasons were patients' and prescribers' doubts on the necessity of a laxative and preference of a waiting policy. The prevalence of constipation doubled during opioid use. A delay for the start of laxative co-medication could be justified by the monitoring of defecation patterns.
Supplemental Material
Supplemental material for Laxative co-medication and changes in defecation patterns during opioid use
Supplemental material for Laxative co-medication and changes in defecation patterns during opioid use by Frans de Bruin, Karin Hek, Jan van Lieshout, Monique Verduijn, Pim Langendijk, Marcel Bouvy and Martina Teichert in Journal of Oncology Pharmacy Practice
Footnotes
Acknowledgements
We would like to thank all participating community pharmacists and patients for their contribution to this pharmacy practice study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
