Abstract
Prophylactic anticoagulation (PAC) is used to reduce the risk of thrombosis in patients with nephrotic syndrome (NS), yet the optimal agent remains uncertain. This systematic review and meta-analysis compare the efficacy and safety of direct oral anticoagulants (DOACs) versus standard-of-care (SOC; heparins or cumarins) for thromboprophylaxis in NS. We systematically searched PubMed, Cochrane Library, Web of Science, and MEDLINE from inception to June 2025. Five cohort studies involving 399 adult patients were included. Pooled analysis indicated that while DOACs were associated with a higher risk of thrombotic events, they demonstrated a superior safety profile by significantly reducing the overall risk of bleeding events. The risk of major bleeding was comparable between the two strategies. These findings underscore the paramount importance of individualized risk-benefit assessment in clinical practice. The current evidence, derived from observational studies, calls for further validation from randomized controlled trials.
Keywords
Introduction
Nephrotic syndrome (NS) is commonly characterized by significant proteinuria (exceeding 3.5 g/day), hypoalbuminemia, edema, and hyperlipidemia, and frequently manifests in various glomerular diseases. Individuals with NS exhibit an elevated risk of thromboembolic events compared to the general population, with incidences reported at 26.7% in adults and 2.8% in children. 1 Thromboembolic complications frequently observed in NS patients include deep vein thrombosis, renal vein thrombosis, pulmonary embolism, and, less frequently, arterial embolism affecting the lower extremities, cerebral, and coronary circulations. 2
Prophylactic anticoagulation (PAC) has been proposed as a strategy to mitigate the prothrombotic condition in NS patients.3–7 While PAC has the potential to decrease the incidence, morbidity, and mortality associated with thrombosis, there remains a lack of consensus regarding the initiation criteria and choice of anticoagulant. The 2021 Kidney Disease: Improving Global Outcomes (KDIGO) guidelines provided a practice point with an algorithm for the prevention of venous thromboembolism (VTE) in adults with NS, recommending the use of PAC based on levels of albuminemia and the risk of bleeding. 8 Medjeral-Thomas et al reported that prophylactic-dose low molecular weight heparin (LMWH) or low-dose warfarin was effective in preventing venous VTE in patients with NS, with relatively few hemorrhagic complications. 9 Another study by Zou et al examined the role of PAC regimens based on LMWH or aspirin in patients with primary membranous nephropathy (PMN), demonstrating that aspirin effectively reduced the incidence of VTEs without affecting the occurrence of arterial thromboembolic events (ATEs). 10 Additionally, antiplatelet agents such as aspirin were employed in sequential therapy with heparin or warfarin when albumin levels improved to 20–30 g/L. 9 A randomized controlled trial (RCT) compared the efficacy and safety of indobufen with warfarin in patients with nephrotic syndrome, indicating that indobufen therapy provided comparable effects in preventing thromboembolic events to warfarin therapy, while a low dose of indobufen was associated with a reduced risk of bleeding events. 11
However, recent studies have suggested a potential role for direct oral anticoagulants (DOACs) in this context.12–14 The initial case reports of DOAC use for therapeutic anticoagulation in NS began to appear in 2014. 15 Kelddal et al and colleagues published a case series indicating the absence of new thrombotic events and the occurrence of only minor bleeding complications in NS treated with DOACs. 12 A study by Tijani et al demonstrated that the use of DOACs for VTE prophylaxis resulted in a clinically relevant, albeit statistically non-significant, reduction in major bleeding rates compared to warfarin. 16 Conversely, Arches et al posited that DOACs were both safer and more effective than traditional anticoagulant strategies for primary and secondary prophylaxis in NS patients. 17 DOACs may serve as an alternative to warfarin and LMWH under exceptional circumstances, particularly in cases of failure or intolerance of these agents. 8
Consequently, we have undertaken this systematic review and meta-analysis to rigorously assess the efficacy and safety of DOACs compared to conventional anticoagulants (heparins/coumarins) for prophylactic anticoagulation (PAC) in patients with NS.
Materials and Methods
This study was conducted and documented in accordance with the PRISMA guidelines, and its protocol was pre-registered on PROSPERO under the registration number CRD420251070072. The PRISMA checklist 18 is available in Supplemental Material 1.
Literature Search
A comprehensive literature search was performed across several major English-language electronic databases, including PubMed, the Cochrane Database of Systematic Reviews, the Web of Science Core Collection, and MEDLINE, covering the period from their respective inceptions up to June 2025. Detailed search strategies for each database are provided in Supplemental Material 2. Furthermore, the reference lists of the retrieved articles were manually reviewed to ensure the inclusion of all relevant studies. Two independent reviewers screened all identified studies, resolving any discrepancies through discussion.
Identification of Eligible Studies
The following inclusion criteria were defined for study inclusion:
Study design: RCT, or cohort study. Types of participants: adult patients with nephrotic syndrome. Intervention: prophylactic anticoagulation with DOACs or warfarin/heparin/other coumarins (standard-of-care [SOC]). Reporting on more than one of the following outcome measures: thrombotic events (eg deep vein thrombosis [DVT]/pulmonary embolism [PE]/renal venous thrombosis [RVT]/artery thrombus embolism [ATE]), major bleeding (ISTH criteria),
19
clinically relevant nonmajor bleeding (CRNMB).
20
Providing sufficient data for calculating the weighted mean difference (WMD) and pooled odd ratio (OR).
Literature reviews, case series, letters, case-report, comments, conference summaries, studies with full texts unavailable, and studies reported and published in non-English were excluded.
Data Extraction
The data were extracted by two review authors independently, and the disagreements were settled by discussing with a third review author to make a consensus. The extracted contents were: name of the first author, date of publication, study period, nationality, study design, number of recruited patients, follow-up duration, length of intervention, characteristics of the participants (age, gender, histopathological type, serum creatinine, estimate glomerular filtration rate [eGFR], serum albumin [sAlb], urinary protein to creatinine ratio [UPCR]), and outcome measures (thrombotic events, major bleeding and CRNMB). As patients with eGFR <15 mL/min/1.73 m2 and those on dialysis might confound outcomes, we eliminated these patients if studies elaborated on relative information. For continuous data reported in the original studies as median (range interquartile), the mean with the standard deviation (SD) would be calculated via a validated mathematical method.18,21
Quality Assessment
The Newcastle–Ottawa Scale (NOS) was utilized to assess the quality of observational studies, with scores of 7–9 indicating “high quality”.22,23 Two independent reviewers conducted the quality assessment, with disagreements resolved through discussion.
Statistical Analysis
Review Manager (RevMan) 5.4 software (Cochrane Collaboration, Oxford, UK) and RStudio were adopted for data synthesis and meta-analysis. WMD and OR were applied as pooled statistics for continuous data and dichotomous data, respectively. The 95% confidential interval (95%CI) of each variable was provided. Chi-squared (χ2) test and I 2 statistic were adopted for testing the heterogeneity among the studies. 24 A p value ≤ 0.10 in χ2 test with an I 2 > 50% indicated that evident heterogeneity existed, and thus a random-effect model was used for meta-analysis. Otherwise (p value > 0.10 in χ2 test with an I 2 ≤ 50%), a fixed-effect model was applied. One-way sensitivity analysis was also conducted to detect the effect of each included study on the pooled results. A funnel plot was produced to reveal the existence of publication bias. Analytic code of R 4.5.2 was in Supplemental Material 3.
Results
Study Selection and Characteristics
The PRISMA flow diagram of the literature searching and screening process is provided in Figure 1.

PRISMA flow diagram of study screening and selection.
The searches in PubMed (n = 575), MEDLINE (n = 1121), Web of Science (n = 1331), and Cochrane Database (n = 11) retrieved 3038 articles. Following the process of duplicate-checking, the titles and abstracts of 1489 articles were browsed. A total of 5 studies meeting the inclusion criteria were included,16,17,25–27 involving 399 adult patients with nephrotic syndrome receiving DOACs, warfarin, other coumarins or heparin for prophylactic anticoagulation. Four patients undergoing dialysis (2 in DOACs and 2 in SOC), pointed out in study by Tijani et al, were eliminated from patients’ number of outcome measures. 16 Of these, one patient in the DOACs cohort was eliminated from patients’ number of bleeding events and major bleeding events. Five studies were all retrospective cohort-design. No RCTs were published. Characteristics and the score of quality assessment of the studies were presented in Table 1, anticoagulant treatment and patient outcomes were presented in Table 2.
Baseline Characteristics of Include Studies and Methodological Assessment.

DOACs = direct oral anticoagulants; SOC = standard-of-care; MN = membranous nephropathy; FSGS = focal segmental glomerulosclerosis; LN = lupus nephritis; DN = diabetic nephropathy; MCD = minimal change disease; AF.
Anticoagulant Treatment and Patient Outcomes.

MN = membranous nephropathy; sAlb = serum albumin; DOACs = direct oral anticoagulants; SOC = standard-of-care; INR = International Normalized Ratio.
The included cohort studies were graded as “high quality”, with a median score of 8.16,17,25–27 Detailed results of the quality assessment are presented in Supplemental Material 4.
Demographic Characteristics
Between the two groups, no evident differences were identified in age (WMD: −0.45; 95% CI: −6.38, 5.48; p = 0.88), gender (male/total, OR: 1.01; 95% CI: 0.49, 2.07; p = 0.98), eGFR (WMD: 3.22; 95% CI: −4.58,11.02; p = 0.42), sAlb (WMD: −0.29; 95%CI: −1.39,0.81; p = 0.61), and UPCR (WMD: 0.71; 95% CI: −0.59,2.00; p = 0.28) (Table 3).
Demographics and Clinical Characteristics of Included Studies.

eGFR = estimate glomerular filtration rate; sAlb = serum albumin; UPCR = Urine protein/creatinine ratio; WMD = weighted mean difference; OR = odds ratio; CI = confidence interval.
Thrombotic Events
Four studies documented the incidence of thrombotic events,16,17,25,26 encompassing a total of 374 patients, with 172 in the DOACs group and 202 in the SOC group. The pooled results indicated patients in the DOACs group exhibited a significantly higher risk of thrombotic events (OR: 5.83; 95% CI: 1.26, 26.92; p = 0.02) (Figure 2). There was no significant heterogeneity observed (I2 = 0%, p = 0.94) (Figure 2). The funnel plot appeared to be symmetric, suggesting no significant publication bias was detected (Figure 3A).

Forest plot of compared result between DOACs and SOC on thrombotic events.

Funnel plot of outcome measures (A) Thrombotic events, (B) Bleeding events, (C) Major bleeding events, (D) CRNMB events.
Bleeding Events
Five studies reported on the incidence of bleeding events,16,17,25–27 involving 395 participants, with180 in the DOACs group and 215 in the SOC group. The pooled analysis indicated that patients in the DOACs group experienced a significantly lower incidence of bleeding events (OR: 0.33; 95% CI: 0.16, 0.68; p = 0.002) (Figure 4). No significant heterogeneity was detected (I2 = 0%, p = 0.52) (Figure 3). Publication bias was confirmed among the studies in the funnel plot (Figure 3B) and showed no significant publication bias.
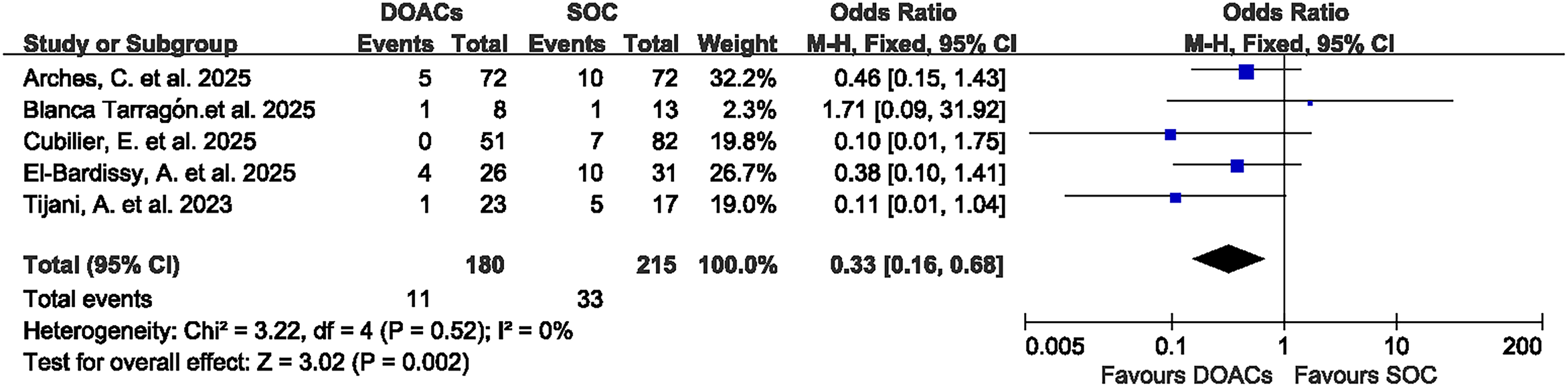
Forest plot of compared result between DOACs and SOC on bleeding events.
Major Bleeding Events
Five studies reported on the incidence of major bleeding events,16,17,25–27 involving a total of 395 patients, with 180 in the DOACs group and 215 in the SOC group. The pooled analysis revealed no significant difference in the incidence of major bleeding events between the two groups (OR: 0.41; 95% CI: 0.15, 1.17; p = 0.09) (Figure 5). There was no significant heterogeneity (I2 = 0%, p = 0.50) (Figure 5). The funnel plot appeared to be symmetric, suggesting no significant publication bias (Figure 3C).

Forest plot of compared result between DOACs and SOC on major bleeding events.
CRNMB Events
Regarding CRNMB events, four studies reported the incidence of CRNMB events,16,17,25,26 encompassing a total of 374 patients (172 in the DOACs group and 202 in the SOC group). The pooled results demonstrated that patients in the DOACs group exhibited a significantly lower incidence of CRNMB events (OR: 0.32; 95% CI: 0.11, 0.88; p = 0.03) (Figure 6). There was no significant heterogeneity (I2 = 0%, p = 0.92) (Figure 5), and no significant publication bias confirmed in the funnel plot (Figure 3D).

Forest plot of compared result between DOACs and SOC on CRNMB events.
Sensitivity Analysis
A one-way sensitivity analysis was conducted for the incidence of thrombotic events (Figure 7), bleeding events (Figure 8), major bleeding events (Figure 9), and CRNMB events (Figure 10) to assess the impact of each study on the pooled results. The sequential exclusion of individual studies did not alter the pooled OR for thrombotic events, bleeding events, major bleeding events, and CRNMB events. The p-value analysis revealed no significant differences in the incidence of thrombotic events when either the study by Cubilier et al or the study by Arches et al was excluded (Figure 7). Similarly, the p-value analysis for CRNMB events demonstrated no statistically significant differences when either the study by EI-Bardissy et al or the study by Cubilier et al was omitted (Figure 10).

Sensitivity analysis diagram of thrombotic events.

Sensitivity analysis diagram of bleeding events.

Sensitivity analysis diagram of major bleeding events.
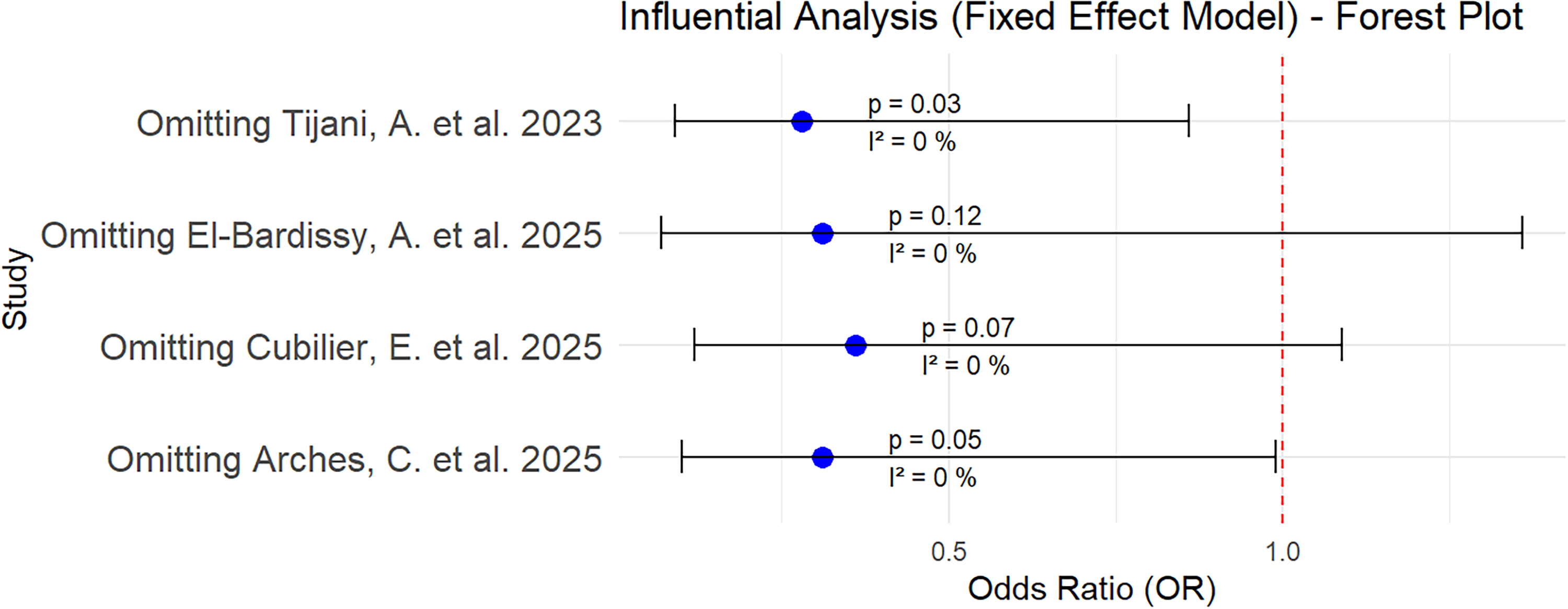
Sensitivity analysis diagram of CRNMB events.
Rivaroxaban Versus Apixaban in Bleeding and Thrombotic Events
To compare the differences in the incidence of bleeding events and thrombotic events in DOACs, we extracted the number of patients and events using rivaroxaban or apixaban from five studies (Table 2) and compared the risks in bleeding events and thrombotic events. Five studies reported the incidence of bleeding events and thrombotic events,16,17,25–27 49 patients in rivaroxaban and 126 in apixaban, excluding 2 patients in apixaban undergoing dialysis. The pooled analysis revealed no significant difference in the incidence of bleeding events (OR: 0.55; 95% CI: 0.13, 2.36; p = 0.42) (Supplement material 4, Figure 1) and thrombotic events (OR: 2.51; 95% CI: 0.50, 12.65; p = 0.26) (Supplement material 4, Figure 2) between the two groups. There was no significant heterogeneity in bleeding events (I2 = 11%, p = 0.34) (Supplemental material 5, Figure 1) and thrombotic events (I2 = 0%, p = 0.50) (Supplemental Material 5, Figure 2). Publication bias was confirmed among the studies in the funnel plot (Supplemental Material 5, Figure 3).
Discussion
The analysis included a total of five retrospective cohort studies,16,17,25–27 while three case series studies on DOACs were excluded.12–14 Among these five studies, three were multicenter, and two were single-center studies. The demographic characteristics, including age, gender, eGFR, sAlb, and UPCR, showed no significant differences across these studies, despite moderate heterogeneity in age (p = 0.05, I2 = 59%) and gender (p = 0.07, I2 = 55%) (Table 2). The pooled analysis suggests that patients with nephrotic syndrome receiving conventional anticoagulant strategies appear to have a superior outcome in preventing thrombotic events compared to those receiving DOACs (OR: 5.83, p = 0.02). Conversely, patients with NS treated with DOACs exhibit a reduced risk of bleeding (OR: 0.33, p = 0.002) and CRNMB (OR: 0.32, p = 0.03) events although the risk of major bleeding remains comparable (OR: 0.41, p = 0.09).
Sensitivity analyses revealed that the statistical significance of thrombotic events (Figure 7) and clinically relevant non-major bleeding (CRNMB) events (Figure 10) was lost upon the exclusion of certain studies (eg, Cubilier et al or Arches et al for thrombotic events; EI-Bardissy et al or Cubilier et al for CRNMB events), indicating a potential dependence of the results on the specific studies included. Although an I2 value of 0% suggests low statistical heterogeneity, clinical heterogeneity may still be present. All included studies enrolled patients with various types of kidney pathologies. In the intervention group, rivaroxaban and apixaban were the primary DOACs used, whereas coumarins were employed in the control group principally. Notably, Tijani et al excluded patients diagnosed with an acute venous thromboembolism (VTE) within the preceding six months, 16 whereas Arches et al included patients undergoing secondary prophylaxis for thrombotic complications. 17 Arches et al excluded patients undergoing dialysis or with an eGFR < 15 ml/min per 1.73 m2, 17 whereas other studies did not provide detailed information on these matters except Tijani et al. 16 Cubilier et al excluded indications for anticoagulation other than NS, 25 while Arches et al included patients with atrial fibrillation who developed NS during treatment. 17
DOACs are integral to anticoagulant strategies in patients with atrial fibrillation or VTE, demonstrating superiority or non-inferiority to vitamin K antagonists (VKAs) in mitigating thromboembolic risk while being associated with a lower incidence of bleeding.28–30 However, the application of DOACs in NS presents a complex scenario with potential safety concerns stemming from their pharmacokinetic and pharmacodynamic profiles. Notably, hypoalbuminemia has been linked to an elevated frequency of bleeding events in patients administered rivaroxaban. 31 Furthermore, hypoalbuminemia correlates with an increased risk of thromboembolic events in NS.3,32 The clearance of DOACs is reduced with worsening renal impairment, resulting in increased plasma exposure and enhanced pharmacodynamic effects. 33 Consequently, the anticoagulant efficacy and bleeding risk associated with DOACs may need to be stratified based on albumin levels in patients with nephrotic syndrome (NS). Unfortunately, the original studies included in our meta-analysis did not provide granular patient-level data to perform such subgroup analyses.
A significant confounding factor identified across the studies included in this analysis was the heterogeneity in anticoagulant dosing regimens. Specifically, some patients were administered prophylactic doses, such as apixaban 2.5 mg twice daily, whereas others received therapeutic doses, such as apixaban 5 mg twice daily (see Table 2). This variability in dosing could have impacted both efficacy and safety outcomes, as higher doses are likely to provide enhanced prophylaxis but may also elevate the risk of bleeding. The inability to stratify outcomes based on dosing regimens constitutes a major limitation, potentially obscuring a more detailed understanding of the risk-benefit profile of DOACs in nephrotic syndrome (NS). Future research should aim to standardize or systematically compare dosing strategies to elucidate their effects on clinical endpoints.
The influence of the underlying native kidney disease, such as membranous nephropathy compared to other conditions, was recognized as a significant factor affecting thrombotic risk in NS. 34 The histopathological types of patients in our meta-analysis included membranous nephropathy (MN), focal segmental glomerulosclerosis (FSGS), lupus nephritis (LN), diabetic nephropathy (DN), minimal change disease (MCD), IgA nephropathy, and amyloidosis. However, only two studies by Arches et al and Cubilier et al reported kidney pathology in patients who experienced events.17,25 Consequently, the absence of detailed data in the original studies prevented stratification based on these factors.
Furthermore, there was considerable variation in the duration of follow-up and exposure to anticoagulation across the studies. Specifically, the median follow-up period was 6 months in the study by El-Bardissy et al, compared to 60 months in the study by Blanca et al (see Table 1). In the study conducted by Cubilier et al, the median duration of intervention in the DOACs group was the shortest at 48 days, whereas it was 429 days in the SOC group as reported by El-Bardissy et al (see Table 2). These discrepancies may have influenced the detection of late-onset thrombotic or bleeding events. To improve comparability, future research should implement standardized follow-up periods.
This systematic review is subject to several limitations. Primarily, all included studies employed retrospective cohort designs. Although this meta-analysis consolidates the best available evidence, the absence of randomized controlled trials diminishes the robustness of causal inferences and exposes our findings to potential residual confounding and bias. A notable limitation impacting the clinical interpretability of our results is the inability to conduct subgroup analyses based on critical patient characteristics, such as serum albumin levels, histopathological subtypes of nephrotic syndrome (eg, membranous nephropathy vs others), or specific DOAC agents. Thrombotic risk in nephrotic syndrome is known to vary significantly with these factors; however, the lack of detailed data in the original studies prevented such stratification. As a result, our pooled estimates reflect an average effect that may not be directly applicable to all patient subgroups. Furthermore, the duration of follow-up and exposure to prophylactic anticoagulation varied considerably across the included studies, which may have affected the detection of outcomes, particularly for events with longer latency periods. Future large-scale, rigorously designed randomized controlled trials should endeavor to report outcomes stratified by these variables to facilitate the advancement of more individualized treatment algorithms.
Conclusion
In conclusion, this meta-analysis demonstrates a critical trade-off in prophylactic anticoagulation for nephrotic syndrome patients. Standard of care (SOC) was superior to DOACs in preventing thrombotic events; however, this benefit came at the expense of a significantly higher risk of overall bleeding and CRNMB events. While the incidence of major bleeding was not statistically different between the two strategies, the favorable safety profile of DOACs regarding overall and CRNMB events cannot be overlooked. There was no significant difference in the risks of bleeding events and thrombotic events between rivaroxaban and apixaban. These findings underscore the necessity of individualized decision-making, where the choice of anticoagulant should be guided by a careful assessment of the patient's inherent thrombotic and bleeding risks.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261442053 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-cat-10.1177_10760296261442053 for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis by Lihua Shi, Mengjin Jiang, Xiaoying Zhang, Congyao Wang and Wenze Yu in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296261442053 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-2-cat-10.1177_10760296261442053 for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis by Lihua Shi, Mengjin Jiang, Xiaoying Zhang, Congyao Wang and Wenze Yu in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-txt-3-cat-10.1177_10760296261442053 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis
Supplemental material, sj-txt-3-cat-10.1177_10760296261442053 for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis by Lihua Shi, Mengjin Jiang, Xiaoying Zhang, Congyao Wang and Wenze Yu in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-4-cat-10.1177_10760296261442053 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-4-cat-10.1177_10760296261442053 for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis by Lihua Shi, Mengjin Jiang, Xiaoying Zhang, Congyao Wang and Wenze Yu in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-5-cat-10.1177_10760296261442053 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-5-cat-10.1177_10760296261442053 for Efficacy and Safety of Direct Oral Anticoagulants Versus Standard Anticoagulation for Thromboprophylaxis in Nephrotic Syndrome: A Systematic Review and Meta-Analysis by Lihua Shi, Mengjin Jiang, Xiaoying Zhang, Congyao Wang and Wenze Yu in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Acknowledgments
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work supported by the Major Science and Technology Plan Project of Xiaoshan District (Project Number: 2022210) and the Zhejiang Provincial Medical and Health Science and Technology Plan (Project Number: 2025HY0780).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
