Abstract
Current guidelines recommend the standard-of-care anticoagulation (vitamin K antagonists or low-molecular-weight heparin) in patients with cerebral venous thrombosis (CVT). Herein, we performed a meta-analysis of randomized clinical trials (RCTs) to assess the efficacy and safety of direct oral anticoagulants (DOACs) compared with the current standard of care in patients with CVT. We systematically searched the PubMed and Embase databases up to December 2023 to identify clinical trials on the effect of DOACs in patients with CVT. A Mantel-Haenszel fixed effects model was applied, and the effect measures were expressed as the absolute risk differences (RDs) and 95% confidence intervals (CIs). A total of 4 RCTs involving 270 participants were included. In the pooled analysis, DOACs and standard of care had low incidence rates of recurrent VTE and all-cause death, and similar rates of any recanalization (78.2% vs 83.2%; RD = −4%, 95%CI:−14% to 5%) and complete recanalization (60.9% vs 69.4%; RD = −7%, 95%CI:−24% to 10%). Compared with the standard of care, DOACs had non-significant reductions in the rates of major bleeding (1.2% vs 2.4%; RD = −1%, 95%CI: −6% to 3%), intracranial hemorrhage (1.9% vs 3.6%; RD = −2%, 95%CI:−7% to 3%), clinically relevant non-major bleeding (3.8% vs 7.4%; RD = −4%, 95%CI:−9% to 2%), and any bleeding (17.3% vs 21.4%; RD = −4%, 95%CI:−16% to 8%) in patients with CVT. DOACs and standard of care showed similar efficacy and safety profiles for the treatment of CVT. DOACs might be safe and a convenient alternative to vitamin K antagonists for thromboprophylaxis in patients with CVT.
Introduction
Cerebral venous thrombosis (CVT) is a rare thrombotic disorder and often affects younger females. Current guidelines recommend anticoagulation therapy for 3 to 12 months in patients with CVT to attain cerebral venous recanalization and decrease the risk of venous thromboembolism (VTE).1,2 The current standard-of-care anticoagulation in these patients includes vitamin K antagonists (VKAs) or low-molecular-weight heparin (LMWH). However, the use of VKAs such as warfarin poses certain drawbacks such as the need for frequent dose monitoring and adjustments, and interactions with other medications or food. On the other hand, low LMWHs, while effective in preventing and treating blood clots, come with their own set of inconveniences, namely the need for subcutaneous administration. Furthermore, LMWHs can cause adverse events such as heparin-induced thrombocytopenia and decreased bone mineral density. Regarding these disadvantages, direct oral anticoagulants (DOACs) may be considered for certain patients.
DOACs including dabigatran, rivaroxaban, apixaban, and edoxaban have been demonstrated to have more net clinical benefits compared with VKAs in selected populations who need anticoagulation therapy for thrombosis prophylaxis.3–7 Currently, DOACs have been commonly prescribed for stroke prevention among adult patients with atrial fibrillation,8,9 or in the acute treatment and secondary prevention of VTE including pulmonary embolism and deep vein thrombosis. 10 In addition, DOACs also have been studied in the specified adult population such as stage 4-5 chronic kidney disease or dialysis, 11 as well as children with cardiac disease12,13 or VTE.14,15
However, the efficacy and safety of DOACs in patients with CVT remains unclear. Considering these, recent randomized clinical trials (RCTs) such as RE-SPECT CVT (Safety and Efficacy of Dabigatran Etexilate Versus Dose-Adjusted Warfarin in Patients With CVT) 16 and SECRET (Study of Rivaroxaban for Cerebral Venous Thrombosis) 17 have examined the use of DOACs compared with standard of care in patients with CVT. Therefore, we performed a systematic review and meta-analysis of clinical trials to evaluate the efficacy and safety outcomes of DOACs when compared with the current standard of care in adult patients with CVT.
Methods
Literature Search
This meta-analysis was conducted based on the Cochrane Handbook for Systematic Reviews of Interventions version 6.2. 18 We systematically searched the databases of PubMed and Embase up to December 2023 to identify relevant studies, which focused on the effect of DOACs in patients with CVT. Three kinds of search terms were applied as follows: (i) “cerebral venous thrombosis”; AND (ii) “non-vitamin K antagonist oral anticoagulants” OR “direct oral anticoagulants” OR “novel oral anticoagulants” OR “dabigatran” OR “rivaroxaban” OR “apixaban” OR “edoxaban”; AND (iii) “vitamin K antagonists” OR “warfarin” OR “coumadin” OR “acenocoumarol” OR “phenprocoumon”. We applied no restrictions on the publication languages. The prespecified literature search strategies are presented in Supplemental Table 1. In addition, we also checked the reference lists of the prior reviews19–22 for additional studies.
Study Eligibility
Our meta-analysis included studies that met specific criteria as follows: (i) RCTs reported that the efficacy and safety outcomes between DOACs (dabigatran, rivaroxaban, apixaban, or edoxaban) and standard of care (VKAs, UFH, or LMWH) in adult patients with CVT; and (ii) the efficacy outcomes included cerebral venous recanalization including any recanalization and complete recanalization, recurrent VTE, and all-cause death, whereas the safety outcomes included major bleeding, intracranial hemorrhage (ICH), clinically relevant non-major bleeding, and any bleeding.
Observational studies and post-hoc analyses of RCTs were excluded in this meta-analysis. We did not include RCTs that reported the efficacy and safety of DOACs in pediatric patients (aged < 18 years) with CVT. In addition, we also did not include single-arm studies with no study controls.
Study Selection and Data Extraction
At the beginning of the study, 2 reviewers who relied on each other's decisions analyzed and assessed the titles and abstracts of the studies that were obtained from the PubMed and Embase databases. In this step, based on the predefined criteria, any observational studies or specific types of studies such as case reports, reviews, meta-analyses, comments, and conference abstracts were automatically excluded. Following the initial screening, the full texts of the remaining studies were thoroughly reviewed, and eligible studies were selected finally. In the event of any disagreements or discrepancies between the reviewers’ assessments, we sought to resolve them through discussions and consultations with senior experts.
The baseline characteristics of included studies were extracted as follows: first author and publication year, study eligibility, registry number of trials, study participants (sample size, age, and sex), study design, data source, dose and type of DOAC or VKAs, assessment of recanalization status, efficacy and safety outcomes, treatment time, and follow-up duration. The number of events and sample size were collected from each study. If there were different follow-ups in the included study, we would include the data with the longest follow-up.
Risk of Bias Assessment
The Cochrane Risk of Bias Tool 2.0 (RoB 2) 23 was used to assess the bias risk in randomized trials. This tool mainly included 5 domains, namely bias from the randomization process, bias due to deviations from intended interventions, bias due to incomplete outcome data, bias in the measurement of the outcomes, and bias in selectively reported results. The bias risk was scored “low risk,” “some concerns,” or “high risk” in each domain. The approach of the overall risk-of-bias judgment could be found in the publication of Sterne et al. 23
Statistical Analysis
We performed a heterogeneity test using statistical methods, specifically the P-value of the Cochrane Q test and the I² value. A P-value of less than 0.10 in the Cochrane Q test or an I² value of greater than 50% would indicate that there is significant heterogeneity among the included studies.24,25 In our pooled analysis, we used the Mantel-Haenszel random-effects model if the I² value was greater than 50%, indicating substantial heterogeneity. If the I² value was 50% or less, we used the fixed-effects model. The effect measures that were used to compare DOACs with the standard of care were the absolute risk differences (RDs) along with their 95% confidence intervals (CIs).
In the sensitivity analysis, we excluded the study on dabigatran and focused on examining the effect of rivaroxaban compared to the standard of care in patients with CVT. In addition, we also assessed the efficacy and safety outcomes between DOACs and VKAs in this specific population. However, due to the limited available data, we were unable to conduct any further subgroup analysis. To check for publication bias, we utilized the funnel plots and visually inspected for any asymmetry. However, according to the Cochrane Handbook, it is important to interpret the results of this analysis with caution because of the limited number of included studies in the publication bias analysis.
All statistical analyses for this meta-analysis were conducted using the Review Manager version 5.4 software (the Cochrane Collaboration 2014, Nordic Cochrane Centre Copenhagen, Denmark). We considered a value of P less than .05 to be statistically significant.
Results
Study Selection and Baseline Characteristics
The process of the study selection of this meta-analysis is presented in Figure 1. A total of 293 studies were found in the PubMed (n = 37) and Embase (n = 257) databases using the literature search strategies. The titles and abstracts of these studies were all screened. We excluded 288 studies in this process because they were observational studies, single-arm studies, reviews, or other types of non-randomized studies. Among the remaining 6 studies that were included in the full-text screening process, we further excluded 2 studies because (1) pediatric patients with CVT (n = 1), 26 and (2) post-hoc analysis of the RE-SPECT CVT trial (n = 1). 27 Finally, a total of 4 RCTs16,17,28,29 involving 270 participants were included in our current meta-analysis.
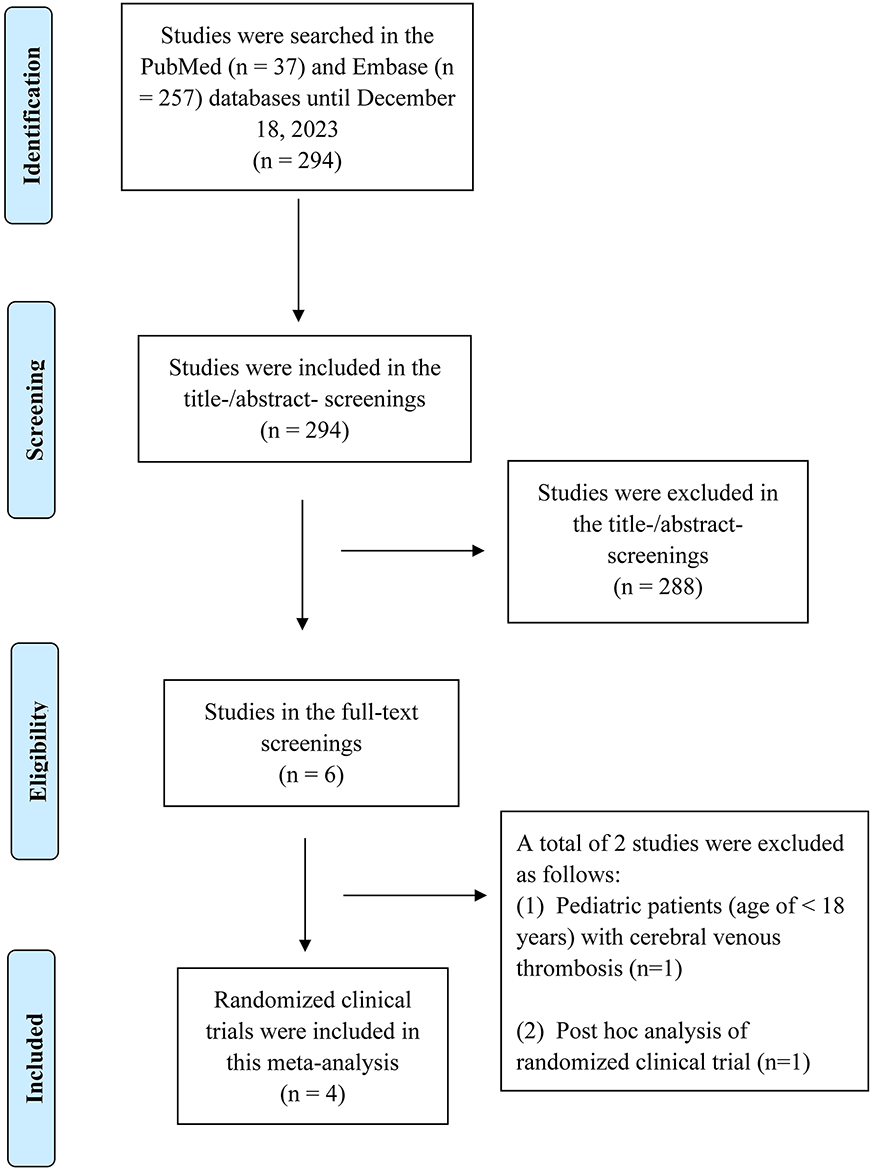
The process of the study selection of this meta-analysis.
The baseline characteristics of the included studies are shown in Table 1. A total of 3 RCTs reported the effect of rivaroxaban,17,28,29 whereas the remaining one provided the data of dabigatran. 16 Data on apixaban and edoxaban were lacking in current RCTs. Warfarin was reported in the control group of these 4 trials. Ferro et al, 16 Maqsood et al, 28 and Khorvash et al 29 included adult patients with CVT who were treated with parenteral LMWH or unfractionated heparin (UFH) before randomization to 2 treatment groups (DOACs vs warfarin). In the SECRET trial by Field et al, 17 55 adult patients within 14 days of a diagnosis of CVT were enrolled and were randomized to rivaroxaban compared with the standard of care (warfarin or LMWH). Among the 4 RCTs, the dose of warfarin was adjusted to maintain an international normalized ratio of 2 to 3. Most of the study participants were females, ranging from 55% to 82% across studies. Other characteristics such as location, sample size, age, treatment duration, efficacy and safety outcomes, and follow-up time are presented in Table 1. The risk of bias assessment of the 4 RCTs is presented in Supplemental Table 2.
Baseline Characteristics of Randomized Clinical Trials in our Meta-Analysis.
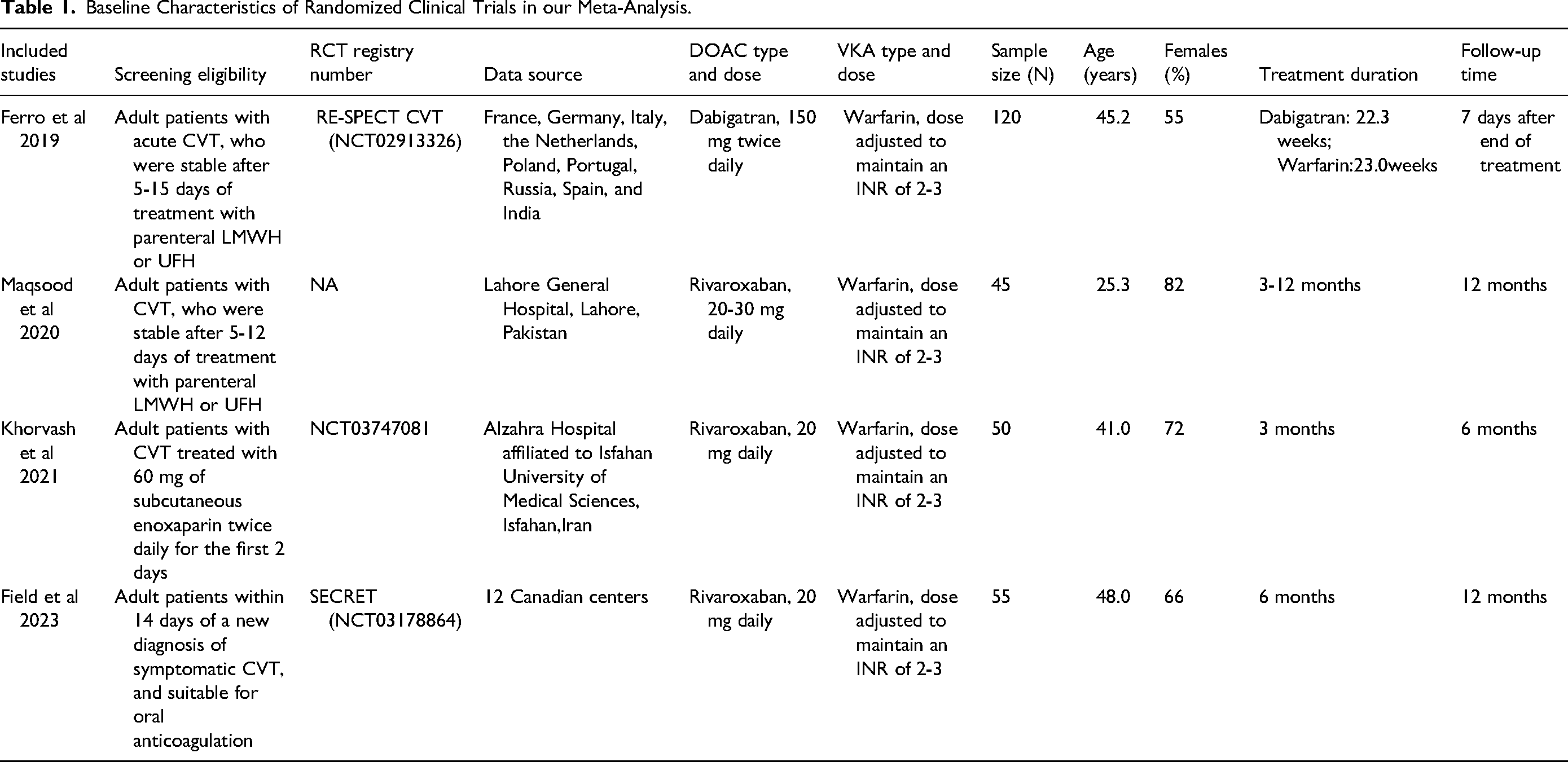

Abbreviations: CVT, cerebral venous thrombosis; RCT, randomized controlled trial; DOAC, direct oral anticoagulant; VKA, vitamin K antagonist; LMWH, low molecular weight heparin; UFH, unfractionated heparin; RE-SPECT CVT, Safety and Efficacy of Dabigatran Etexilate Versus Dose-Adjusted Warfarin in Patients With CVT; SECRET, Study of Rivaroxaban for Cerebral Venous Thrombosis; INR, international normalized ratio; VTE, venous thromboembolism; mRS, Modified Rankin Scale; NIHSS, National Institute of Health Stroke Scale; NA, not available.
Efficacy and Safety Outcomes Between DOACs and Standard of Care
Efficacy Outcomes
We applied the fixed effects model in the pooled analysis due to the low heterogeneity across studies. As shown in Figure 2, compared with standard of care, DOACs showed similar rates of any recanalization (partial or complete) (78.2%[79/101] vs 83.2%[84/101]; RD = −4%, 95%CI: −14% to 5%, P = .45) and complete recanalization (60.9%[28/46] vs 69.4%[34/49]; RD = −7%, 95%CI: −24% to 10%, P = 0.41) in patients with CVT. In addition, the incidence rates of recurrent VTE (0.9%[1/107] vs 0%[0/111]; RD = 1%, 95%CI: −3% to 4%, P = 0.61) and all-cause death (0%[0/132] vs 0.7%[1/136]; RD = −1%, 95%CI: −4% to 2%, P = 0.66) were relatively low in both study groups.
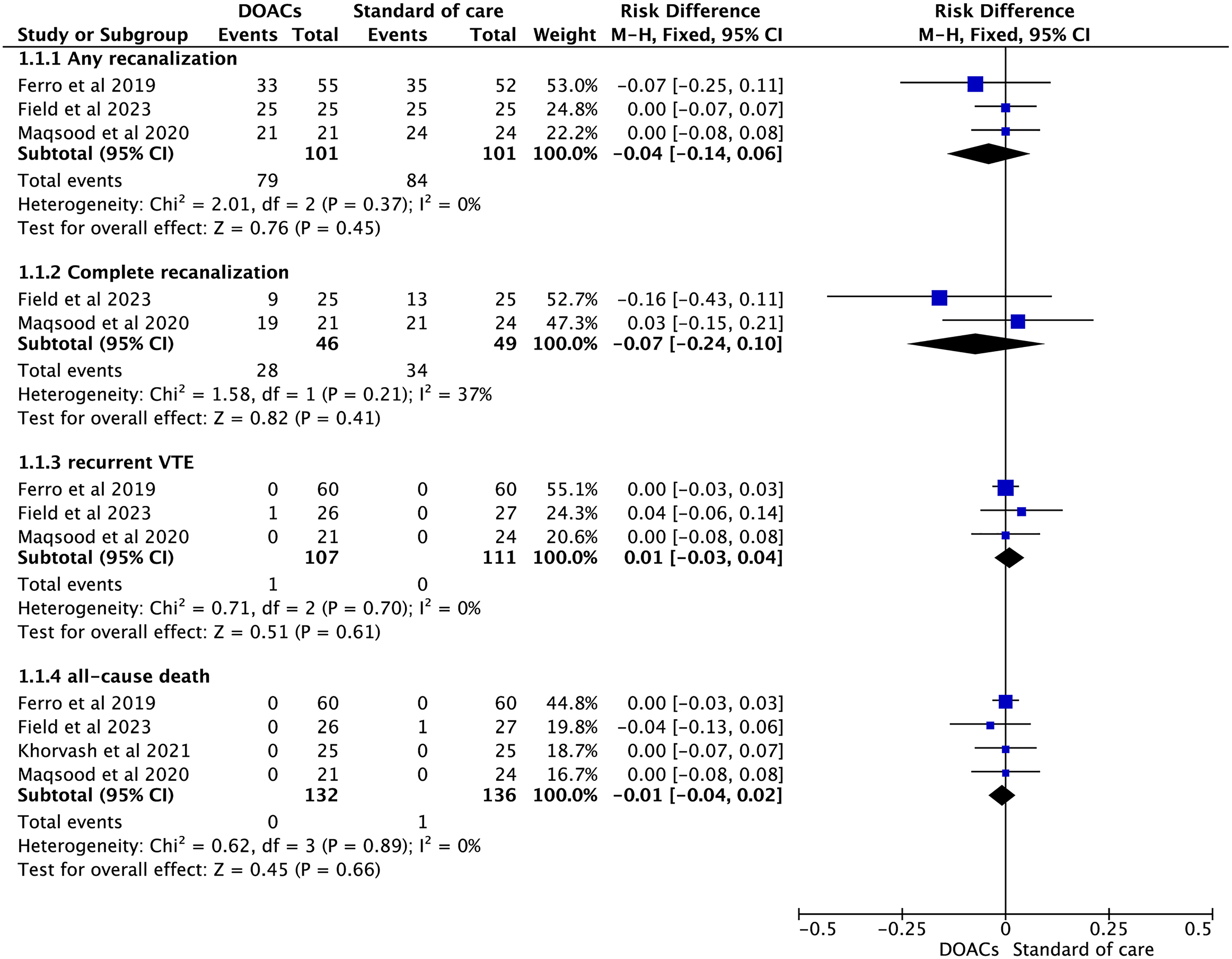
Efficacy outcomes of direct oral anticoagulants compared with standard of care in patients with cerebral venous thrombosis.
Safety Outcomes
The pooled results by a fixed effects model (Figure 3) showed that compared with standard of care, the use of DOACs had non-significant reductions in the occurrence rates of major bleeding (1.2%[1/81] vs 2.4%[2/84]; RD = −1%, 95%CI: −6% to 3%, P = 0.61), ICH (1.9%[2/107] vs 3.6%[4/111]; RD = −2%, 95%CI: −7% to 3%, P = 0.50), clinically relevant non-major bleeding (3.8%[5/132] vs 7.4%[10/136]; RD = −4%, 95%CI: −9% to 2%, P = 0.19), and any bleeding (17.3%[14/81] vs 21.4%[18/84]; RD = −4%, 95%CI: −16% to 8%, P = 0.49) in adult patients with CVT.
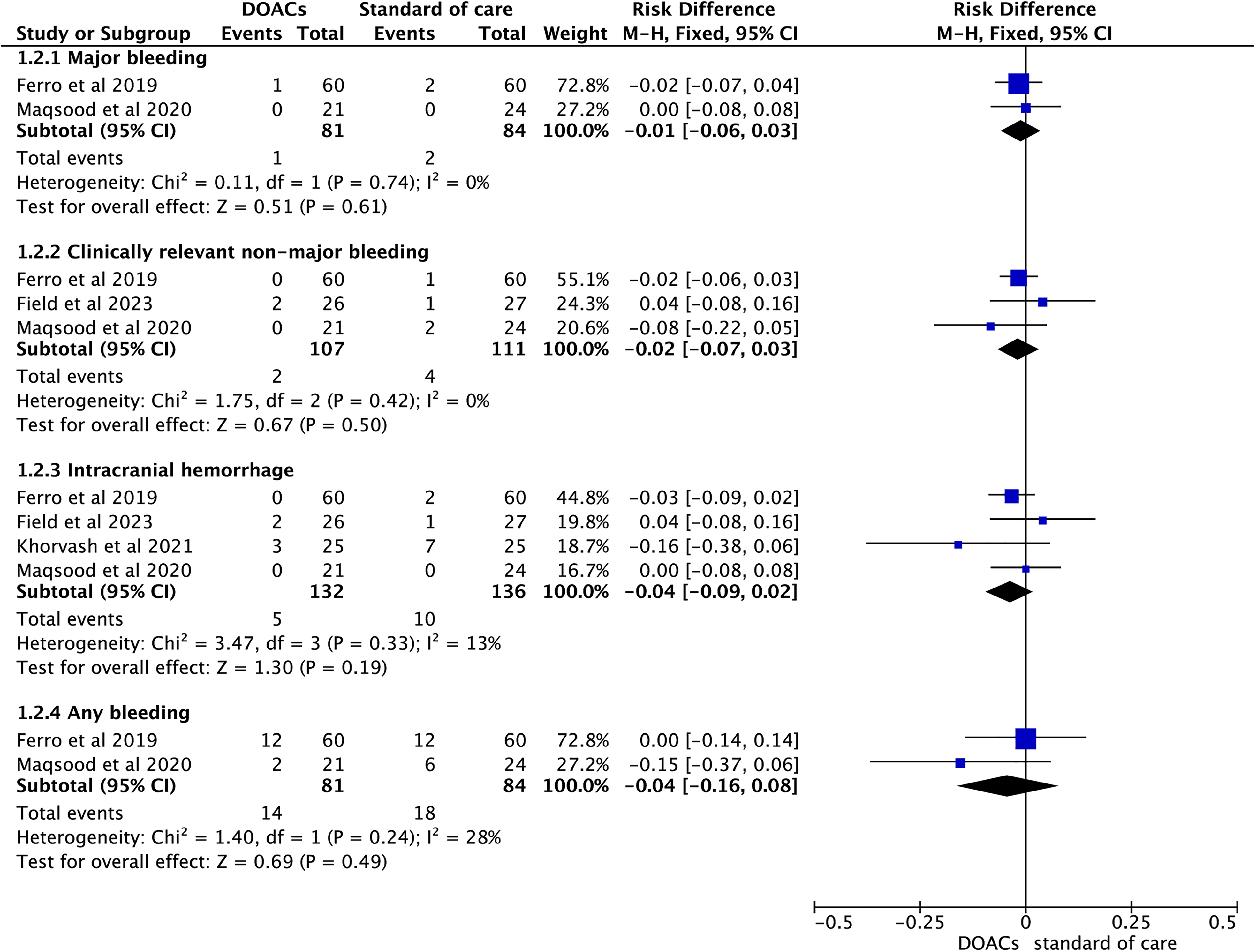
Safety outcomes of direct oral anticoagulants compared with standard of care in patients with cerebral venous thrombosis.
Sensitivity Analysis
In the sensitivity analysis, we excluded the study of Ferro et al, 16 and examined the efficacy and safety outcomes of rivaroxaban compared with standard of care. In addition, we also assessed the efficacy and safety outcomes between DOACs and warfarin after deleting the study of Field et al. 17 As shown in Supplemental Figure 1-4, the sensitivity analyses presented similar results to the main analysis mentioned above.
Publication Bias
The publication bias analyses using the funnel plots suggested no obvious publication bias for the studied efficacy (Supplemental Figure 5) and safety outcomes (Supplemental Figure 6).
Discussion
Based on our current analysis of data from 4 RCTs, we found that both groups of DOACs and standard of care had relatively low incidence rates of recurrent VTE and all-cause death. In addition, the rates of recanalization (partial or complete) and complete recanalization were similar between the 2 groups. Furthermore, when comparing DOACs to standard of care, we observed non-significant reductions in the occurrence rates of major bleeding, ICH, clinically relevant non-major bleeding, and any bleeding. This suggests that DOACs may have a comparable safety profile to standard anticoagulation treatments, which include VKAs. Overall, although we did not detect statistically significant differences, the efficacy and safety profiles of DOACs and standard anticoagulation treatments for CVT were similar. Therefore, DOACs could be considered a safe and convenient alternative to VKAs for thromboprophylaxis in patients with CVT.
RCT Evidence of DOACs in Patients With CVT
DOACs have been increasingly used in clinical practice. The first study to assess DOACs in patients with CVT was the exploratory RE-SPECT CVT trial by Ferro et al 16 in 2009. A total of 120 adult patients with CVT were enrolled after 5 to 15 days of treatment with parenteral LMWH or UFH, who were randomized to groups dabigatran (n = 60) and dose-adjusted warfarin (n = 60). They found that both treatment groups had a low risk of recurrent VTE and death, and showed similar rates of efficacy and safety outcomes. Subsequent 2 small trials by Maqsood et al 28 and Khorvash et al 29 examined the effects of rivaroxaban compared with dose-adjusted warfarin, suggesting that rivaroxaban was an efficacious and safe medication in treating CVT. The lead-in parenteral anticoagulation was applied in these 3 RCTs before randomization to 2 treatment groups of DOACs and warfarin.
In the latest SECRET trial by Field et al, 17 55 CVT patients were enrolled who were randomized to rivaroxaban compared with standard anticoagulation (warfarin or LMWH). We noted that the lead-in parenteral anticoagulation was used in the SECRET trial, and its comparator (ie, warfarin or LMWH) was also distinct from that (ie, warfarin only) in the aforementioned 3 trials.16,28,29 Functional outcomes such as modified Rankin Scale, quality of life, headache, mood, fatigue, and cognition were also prospectively assessed among the study groups in the SECRET trial, 17 providing valuable insights into the impact of DOACs on the patient's physical abilities and their subjective experiences during the treatment. Our current meta-analysis by pooling these 4 RCTs suggested that DOACs and standard of care (including VKAs) had similar efficacy and safety in the treatment of CVT. Moreover, Connor et al 26 performed a post-hoc analysis of the EINSTEIN-Jr (Oral Rivaroxaban in Children With Venous Thrombosis) pediatric VTE trial involving 117 children with acute CVT comparing rivaroxaban versus standard anticoagulation, suggesting consistent findings in pediatric patients with CVT. Future studies with larger sample sizes and better study quality could potentially strengthen the reliability of our findings and provide a more comprehensive understanding of the subject matter.
Observational Evidence of DOACs in Patients With CVT
RCTs may balance the findings between the treatment groups and acquire a fair assessment of the trial treatment effect. However, the study participants enrolled in RCTs are often not equal to those in observational studies. Whether similar findings of DOACs compared with standard anticoagulation (warfarin or LMWH) for the treatment of CVT in RCTs could be found in real-world settings remains unknown. Previously, several observational studies30–41 have investigated the use of DOACs in patients with CVT, although the sample sizes were relatively small and all of them were retrospective. These inconsistencies in study quality may introduce uncertainties in their results and should be taken into account when interpreting the implications of these studies.
Prior systematic reviews and meta-analyses19,20 of observational retrospective studies suggested that DOACs and warfarin had comparable efficacy and safety in patients with CVT, consistent with the findings of our current meta-analysis based on RCTs. Nevertheless, data on the large-scale international prospective real-world studies are still lacking. Ongoing high-quality multi-center observational cohort studies (eg, the DOAC-CVT study 42 ) may provide new insights into the effectiveness and safety of DOACs compared with VKAs for the treatment of CVT in real-world settings.
Limitations
We acknowledged that our present systematic review and meta-analysis, despite its valuable insights, came with certain limitations that should be considered. First, one of the notable limitations was the small sample size of the RCTs included in our pooled analysis. The relatively limited number of participants in these trials might restrict the generalizability of our findings to a larger population. Second, we should address the concern surrounding the quality of the included studies. It was possible that the methodological limitations or biases within these trials could have influenced the reliability and validity of our findings. Third, apixaban and edoxaban might have better efficacy and safety profiles than dabigatran and rivaroxaban in patients with atrial fibrillation.24,43 However, data on apixaban and edoxaban for the treatment of CVT are lacking in current RCTs, which needs further examination. In addition, there were still no studies assessing the effects of different DOACs in patients with CVT. Fourth, the limited data prevented us from conducting subgroup analyses to further examine the impact of baseline characteristics such as age, sex, and type and dose of DOACs on the outcomes. Finally, due to the limited data availability, we were unable to evaluate the functional outcomes and patient-reported outcomes. In addition, the cost-effectiveness of NOACs for the treatment of CVT needs further investigation.
Conclusions
Based on our analysis of the combined data from 4 RCTs, we have found that DOACs and the standard method of care including VKAs have demonstrated similar effectiveness and safety in the treatment of CVT. This implies that DOACs could potentially serve as a secure and more convenient alternative to VKAs for thromboprophylaxis when managing CVT patients in clinical practice. Nevertheless, to provide more robust and reliable evidence, further studies of superior quality are warranted to affirm our findings.
Supplemental Material
sj-docx-1-cat-10.1177_10760296241256360 - Supplemental material for Efficacy and Safety of Direct Oral Anticoagulants in Cerebral Venous Thrombosis: Meta-Analysis of Randomized Clinical Trials
Supplemental material, sj-docx-1-cat-10.1177_10760296241256360 for Efficacy and Safety of Direct Oral Anticoagulants in Cerebral Venous Thrombosis: Meta-Analysis of Randomized Clinical Trials by Xi Chen, Linjuan Guo and Meiming Lin in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Author Contributions
Xi Chen and Meiming Lin performed the literature search by searching various databases and online platforms, retrieving relevant studies. After the literature search, Xi Chen and Meiming Lin carefully selected the studies based on specific inclusion and exclusion criteria. Xi Chen and Linjuan Guo extracted data from the selected studies, meticulously collecting and organizing necessary information such as study design, participant characteristics, intervention or exposure, outcome measures, and results. Furthermore, Xi Chen and Meiming Lin conducted a comprehensive quality assessment of the included studies to evaluate their methodological rigor and internal validity. Next, Xi Chen critically reviewed and revised the draft written by Meiming Lin and Linjuan Guo. Meiming Lin also extensively edited the manuscript to ensure the correct usage of standard English grammar, proper sentence structure, and appropriate academic language. Finally, Meiming Lin and Linjuan Guo performed a final edit of the manuscript prior to submission.
Availability of Ddata and Mmaterials
All relevant data and materials are presented in the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sanming City Science and Technology Joint Funding Project(2022-S-56).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
