Abstract
Objective
To identify factors associated with pulmonary embolism (PE) in gastrointestinal cancer patients with isolated distal deep vein thrombosis (IDDVT) and to develop a nomogram-based predictive model for PE.
Methods
From January 2021 to January 2024, a total of 204 patients with gastrointestinal malignancies diagnosed with IDDVT were retrospectively analyzed. Pulmonary embolism was confirmed by computed tomography pulmonary angiography. Logistic regression analysis was performed to identify relevant risk factors, and a nomogram-based risk prediction model was subsequently established.
Results
Among the 204 patients with IDDVT, 86 (42.2%) developed PE. Univariate analysis showed that age ≥60 years, diabetes mellitus, coronary heart disease, tumor stage, and glucocorticoid use were significantly associated with PE (P < .05). Multivariate logistic regression identified age ≥60 years, coronary heart disease, and glucocorticoid use as factors independently associated with PE (P < 0.05). A nomogram was constructed based on these variables to predict PE occurrence. The area under the receiver operating characteristic (ROC) curve was 0.77 (95% CI 0.71–0.83), with a sensitivity of 64.2% and specificity of 76.6%.
Conclusion
This study observed a substantial frequency of PE among gastrointestinal cancer patients with IDDVT. Age ≥ 60 years, coronary artery disease, and lack of glucocorticoids were independently associated with PE in patients with IDDVT. With these risk factors in mind, we developed a nomogram risk prediction model to help detect concurrent PE early.
Keywords
Introduction
Deep vein thrombosis (DVT) and pulmonary embolism (PE), as manifestations of venous thromboembolism (VTE) at different anatomical sites and stages, pose serious threats to patients’ health and survival. 1 In most cases, PE originates from lower-extremity DVT, among which isolated distal DVT (IDDVT), although accounting for a considerable proportion of DVT cases, has traditionally been regarded as self-limiting, with its clinical significance remaining unclear. 2
In patients with gastrointestinal malignancies, owing to both disease-related and treatment-associated factors, IDDVT accompanied by PE is not uncommon. 3 However, the exact occurrence and risk factors for PE in the context of IDDVT remain poorly defined, creating challenges for clinical prevention and management. At present, there is a lack of practical risk prediction models specifically designed for evaluating the occurrence of PE in gastrointestinal cancer patients with IDDVT.
Previous studies have reported that advanced age, comorbidities such as cardiovascular disease and diabetes mellitus, tumor burden, surgical intervention, chemotherapy, and use of glucocorticoids are associated with an increased risk of venous thromboembolism in patients with gastrointestinal malignancies. 4 However, data specifically addressing factors associated with the progression from isolated distal deep vein thrombosis (IDDVT) to pulmonary embolism remain limited.
This study retrospectively analyzed clinical data from gastrointestinal cancer patients with IDDVT to identify factors associated with the development of pulmonary embolism and to construct a predictive model. The goal was to facilitate early identification of high-risk patients and support individualized clinical decision-making in this population.
Methods
Study Population
We retrospectively collected data on patients diagnosed with gastrointestinal malignancies complicated by IDDVT between January 2021 and January 2024. This was a retrospective observational study conducted at a single tertiary medical center. According to whether PE occurred, patients were classified into the IDDVT-only group and the IDDVT with PE group.
Data Collection
General demographic and clinical data were collected for eligible patients, including sex, age, body mass index (BMI), and comorbidities; tumor characteristics (primary site, tumor stage); surgical history; and details of IDDVT and PE (clinical symptoms, location, and type).
Gastric and colorectal cancers were staged according to the eighth edition of the American Joint Committee on Cancer (AJCC) TNM staging system.
5
Diagnostic criteria for lower-extremity DVT: examinations were performed by two experienced ultrasound physicians using venous compression techniques. A diagnosis of lower-extremity DVT was made when echogenic solid material of varying density was observed within the venous lumen, with absent or reduced color Doppler flow signals and no lumen collapse upon compression. Diagnostic criteria for PE: patients presenting with unexplained dyspnea, shortness of breath, chest pain, syncope, or hypotensive shock were diagnosed with PE if confirmed by computed tomography pulmonary angiography.
6
Classification of DVT: thrombosis involving the popliteal vein, anterior tibial vein, or posterior tibial vein was classified as axial vein type, whereas thrombosis in the peroneal vein, gastrocnemius vein, or soleal vein was classified as muscular vein type.
Lower-extremity venous ultrasonography was performed during hospitalization, either when patients presented with clinical symptoms suggestive of venous thromboembolism (such as limb swelling or pain) or when they were considered at high thrombotic risk due to active malignancy, perioperative status, or reduced mobility. This timing was selected to allow early detection of IDDVT and timely evaluation of potential progression to pulmonary embolism, thereby supporting appropriate clinical decision-making.
Statistical Analysis
All statistical analyses were performed using SPSS version 26.0. Categorical variables were presented as frequencies and percentages and compared using the χ2 test or Fisher's exact test, as appropriate. Variables with P < 0.05 in univariate analysis or deemed clinically relevant were included in a multivariable logistic regression model to identify factors independently associated with pulmonary embolism. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, and a two-sided P value < 0.05 was considered statistically significant.
Results
Baseline Characteristics of the Study Population
A total of 204 patients with gastrointestinal malignancies and IDDVT were included in the study. Among them, 118 (57.8%) were classified into the IDDVT-only group and 86 (42.2%) into the IDDVT with PE group, as shown in Table 1.
Comparison of Clinicopathological Characteristics Between Patients with IDDVT Only and Those with IDDVT Combined with PE, and Univariate Analysis.

In the IDDVT with PE group, 54 patients (62.8%) were aged ≥60 years, 37 (43.0%) were male, 5 (5.8%) had a BMI ≥28, 32 (37.2%) had hypertension, 39 (45.3%) had diabetes, 19 (22.1%) had coronary heart disease, and 9 (10.5%) had atrial fibrillation. Surgical treatment was performed in 48 patients (55.8%), 27 (31.4%) were at stage I/II, 7 (8.1%) received neoadjuvant chemotherapy, and 16 (18.6%) used glucocorticoids.
In the IDDVT-only group, 47 patients (39.8%) were aged ≥60 years, 56 (47.5%) were male, 10 (8.5%) had a BMI ≥28, 40 (33.9%) had hypertension, 46 (39.0%) had diabetes, 10 (8.5%) had coronary heart disease, and 14 (11.9%) had atrial fibrillation. Surgical treatment was performed in 72 patients (61.0%), 55 (46.6%) were at stage I/II, 15 (12.7%) received neoadjuvant chemotherapy, and 68 (57.6%) used glucocorticoids.
Thrombus Characteristics
As shown in Table 2, thrombi involved the axial veins in 41 patients (20.1%), muscular veins in 163 patients (79.9%), and both axial and muscular veins in 25 patients (12.3%).
Thrombus Characteristics in Patients with IDDVT Only and Those with IDDVT Combined with PE.
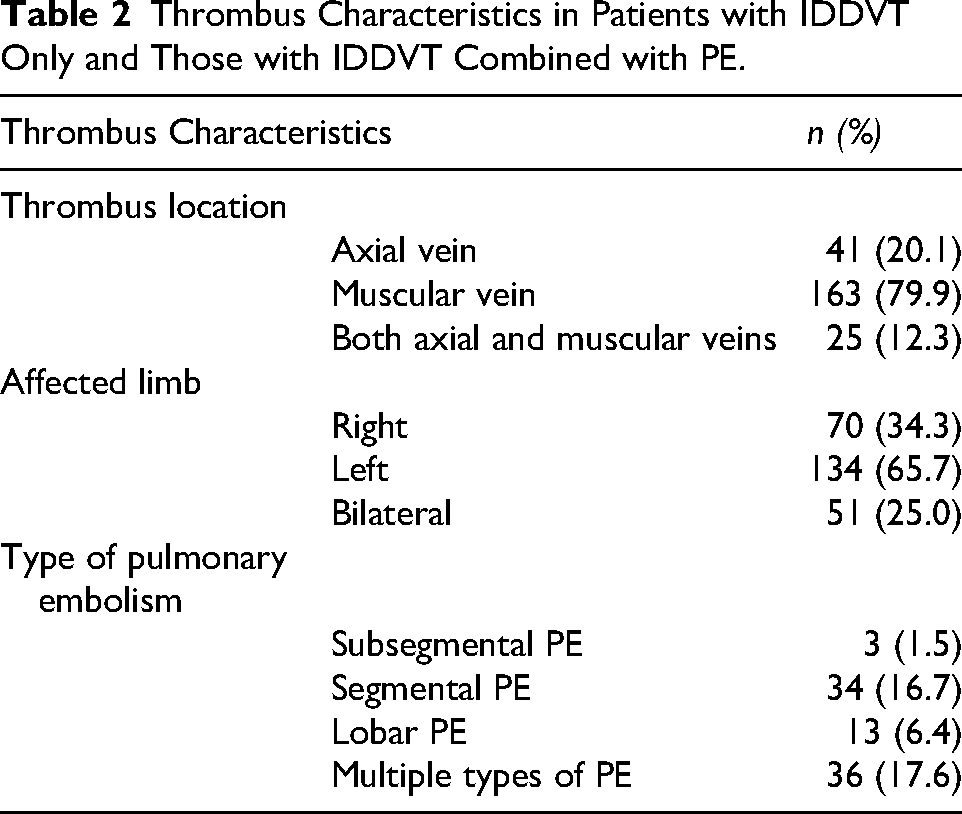
Regarding the affected limb, thrombi were limited to the right limb in 70 patients (34.3%), the left limb in 134 patients (65.7%), and both limbs in 51 patients (25.0%).
For PE type, 3 patients (1.5%) had subsegmental PE, 34 (16.7%) had segmental PE, 13 (6.4%) had lobar PE, and 36 (17.6%) had multiple types of PE concurrently.
Risk Factor Analysis for Pulmonary Embolism
Comparison of baseline characteristics between the IDDVT with PE group and the IDDVT-only group revealed that age ≥60 years, diabetes, coronary heart disease, tumor stage, and glucocorticoid use were significantly associated with the occurrence of PE in gastrointestinal cancer patients with IDDVT in univariate analysis (P < 0.05) (Table 1).
These five variables were entered into a binary multivariate logistic regression model, which identified age ≥60 years, coronary heart disease, and glucocorticoid use as factors independently associated with PE in this patient population (P < 0.05) (Figure 1).

Multivariate logistic regression analysis of clinical variables in the two patient groups.
Construction of the Risk Assessment Model and ROC Curve Analysis
Independent predictors identified in the multivariate analysis were incorporated into a nomogram to construct the risk assessment model (Figure 2).

Nomogram for predicting pulmonary embolism in gastrointestinal cancer patients with IDDVT.
Point allocation in the nomogram was based on the magnitude of regression coefficients derived from the multivariate logistic regression model. Age <60 years was assigned 0 points and age ≥60 years 60 points; absence of coronary heart disease was assigned 0 points and its presence 60 points; glucocorticoid non-use was assigned 100 points, whereas use was assigned 0 points. The maximum total score was 240 points. Although age and coronary heart disease had different regression coefficients, similar point values were assigned to enhance visual interpretability while preserving their relative contribution within the model.
The AUC of the nomogram was 0.77 (95% CI 0.71–0.83) (Figure 3), which was higher than the AUCs of models constructed from individual predictors, indicating that the developed risk assessment model demonstrated acceptable discrimination and potential clinical applicability.

ROC curves of age, coronary heart disease, and glucocorticoid use for predicting pulmonary embolism in gastrointestinal cancer patients with IDDVT.
Discussion
Gastrointestinal malignancies, particularly gastric cancer, are well recognized to be associated with a hypercoagulable state. Tumor cells can activate the coagulation cascade through the release of procoagulant factors, inflammatory cytokines, and interactions with host vascular endothelium, thereby significantly increasing the risk of venous thromboembolism. 7 This prothrombotic milieu provides a strong biological rationale for the increased occurrence of DVT and pulmonary embolism observed in patients with gastrointestinal cancer.
Pulmonary embolism, as a severe manifestation of VTE, carries a high mortality and disability rate in patients with gastrointestinal malignancies complicated by IDDVT. The occurrence of PE not only worsens patient prognosis but also increases healthcare resource utilization. In the present study, PE was observed in 42.2% of gastrointestinal cancer patients with IDDVT, highlighting the clinical relevance of careful monitoring and timely evaluation in this population. At present, the diagnosis of PE mainly relies on imaging examinations such as computed tomography pulmonary angiography; however, some asymptomatic patients may be missed due to a lack of timely imaging. Therefore, establishing an effective risk prediction model is of great significance for the early identification of high-risk patients and the implementation of preventive measures. 8 Previous studies have demonstrated that malignancy is widely recognized as a major contributor to VTE risk, which has become the second leading cause of death in cancer patients. 9 Furthermore, the risk of VTE in cancer patients is 4–7 times higher than in non-cancer patients. 10 Accurate identification of high-risk patients and appropriate use of prophylactic anticoagulation may reduce VTE risk in cancer patients; however, this benefit must be weighed against the elevated bleeding risk inherent to this population. Cancer patients often have tumor-related mucosal injury, chemotherapy-induced thrombocytopenia, and frequent exposure to invasive procedures, all of which increase bleeding susceptibility. Gastrointestinal malignancies, in particular, may directly predispose patients to gastrointestinal bleeding. Therefore, careful individualized evaluation of thrombotic and hemorrhagic risks is essential when considering anticoagulation therapy.
Although IDDVT has traditionally been regarded as a relatively benign and self-limiting condition, its clinical significance in patients with gastrointestinal malignancies should not be underestimated. These patients represent a high-risk population for venous thromboembolism due to cancer-related hypercoagulability, surgical trauma, chemotherapy, and immobilization. Early identification of IDDVT in this specific clinical context may facilitate risk stratification, closer surveillance, and timely diagnostic evaluation for pulmonary embolism. Therefore, investigating the progression of IDDVT to PE and establishing a practical risk prediction model may provide meaningful clinical guidance for individualized prevention and management strategies.
In our study, we observed a considerable proportion of PE cases among patients with gastrointestinal cancer and IDDVT, supporting the notion that distal thrombosis in this population is not always clinically benign.11,12 The progression from IDDVT to PE may be influenced by multiple factors, including thrombus size, location, and extent of involvement. Thrombus extension to axial veins or bilateral limb involvement may further increase the likelihood of embolic events. In addition, tumor-related hypercoagulability and systemic inflammatory responses in cancer patients may exacerbate thrombus propagation and embolization. 13 For gastrointestinal cancer patients, perioperative stress, chemotherapy exposure, and other cancer-related treatments may further contribute to thrombotic progression. 14
Currently, commonly used thrombosis risk assessment tools such as the Caprini risk score, 15 the Padua prediction score, 16 and the Khorana score 17 have certain clinical utility in evaluating the risk of VTE. However, their accuracy in predicting PE in gastrointestinal cancer patients with IDDVT is limited. 18 These scoring systems may not adequately account for the unique characteristics of gastrointestinal cancer patients, such as tumor stage and surgical approach, which may influence the risk of PE. 19 Furthermore, for IDDVT- a specific type of thrombosis- current scoring systems may not be able to accurately assess its risk of progression to PE. Therefore, it is necessary to develop a risk prediction model more suitable for this patient population. Studies have shown that different cancer populations have varying risks and patterns of VTE, and the predictive performance of existing models varies across patient groups.20,21
Compared with previous research, this study focuses specifically on the risk prediction of PE in gastrointestinal cancer patients with IDDVT. While some studies have emphasized the overall VTE profile, 14 our work analyzes this distinct subgroup of patients with concurrent IDDVT and PE. We identified age ≥60 years, coronary heart disease, and glucocorticoid use as factors independently associated with PE in this population, findings that are consistent with some reports, 22 although other studies have not identified these factors as primary independent predictors. The nomogram-based risk assessment model constructed in this study demonstrated good accuracy and clinical utility, providing a novel predictive tool for clinicians. Previous evaluations of VTE risk prediction models in cancer patients have found substantial variability in model performance across different patient populations. 23
The association between glucocorticoid use and pulmonary embolism observed in our study warrants careful interpretation. Glucocorticoids have been reported to promote a prothrombotic state by increasing coagulation factor levels, impairing fibrinolysis, and inducing endothelial dysfunction. 24 In cancer patients, glucocorticoids are frequently administered for anti-inflammatory purposes, management of chemotherapy-related adverse effects, or treatment of tumor-related complications, often in the context of more intensive clinical monitoring. Interestingly, non-use of glucocorticoids was associated with a higher risk of PE in our cohort. This apparent inconsistency may reflect confounding by indication or differences in disease severity and clinical management. Therefore, glucocorticoid exposure in this study should be interpreted as a marker associated with thrombotic risk within a specific clinical context, rather than as evidence of a direct protective or causal effect.
Nonetheless, this study has several limitations. First, some pulmonary embolism cases may have been asymptomatic and therefore not diagnosed by pulmonary CTA, potentially resulting in an underestimation of the true frequency of PE . In clinical practice, the diagnosis of venous thromboembolism in cancer patients is often delayed because symptoms are not consistently recognized, which may further contribute to underdetection. 25 Second, as a single-center retrospective observational study, the findings may be subject to selection bias and limited generalizability. In addition, thrombus-related characteristics such as size, extent, axial vein involvement, and bilaterality were not included in the multivariate analysis, partly due to incomplete documentation and limited sample size, which may have reduced the robustness of additional subgroup analyses. Given the retrospective design, the identified factors should be interpreted as associations rather than causal determinants. Therefore, multicenter prospective studies with larger sample sizes and standardized imaging assessments are warranted to further validate and refine the predictive model.
Conclusion
This study developed a risk prediction model for PE in gastrointestinal cancer patients with IDDVT and identified age ≥60 years, coronary heart disease, and lack of glucocorticoid use as factors independently associated with PE. The proposed nomogram may assist in early identification of high-risk patients and support clinical decision-making in gastrointestinal cancer patients with IDDVT.
Footnotes
Acknowledgements
The authors acknowledge all investigators who contributed to the trial.
Ethical Approval
This study was approved by the Ethics Committee of Beijing Chao-Yang Hospital, Capital Medical University. Due to the retrospective nature of the study, the requirement for written informed consent was waived by the ethics committee.
Author Contributors
Study design: Chai Qi and Liu XY. Data collection: Chai Qi and Liu XY. Data analysis: Chai Qi and Liu XY. Manuscript preparation: Chai Qi and Liu XY. Critical review and revision: Chai Qi and Liu XY. Final approval of the article: Chai Qi and Liu XY
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor
Qi Chai.
