Abstract
Valuable data on deep vein thrombosis (DVT) patients with coexisting pulmonary embolism (PE) is scarce. This study aimed to identify risk factors associated with these patients and develop logistic regression models to select high-risk DVT patients with coexisting PE. We retrospectively collected data on 150 DVT patients between July 15, 2022, and June 15, 2023, dividing them into groups based on the presence of coexisting PE. Univariate and multivariate logistic regression analyses were performed to identify significant risk factors and construct predictive models. Discrimination and calibration statistics evaluated the validation and accuracy of the developed models. Of the 130 patients analyzed, 40 (30.77%) had coexisting PE. Univariate analysis revealed four significant predictors of DVT patients with coexisting PE: sex (OR 3.83, 95% CI: [1.76; 8.59], P = 0.001), body mass index (BMI) (OR 1.50, 95% CI: [1.28; 1.75], P < 0.001), chronic disease (OR 5.15, 95% CI: [2.32; 11.8], P < 0.001), and high-density lipoprotein (HDL) (OR 0.03, 95% CI: [0.01; 0.20], P < 0.001). Additionally, BMI > 24 kg/m2 (OR 9.70, 95% CI: [2.70; 67.5], P < 0.001) and BMI > 28 kg/m2 (OR 4.80, 95% CI: [2.15; 11.0], P < 0.001) were associated with concurrent PE. Three multiple regression models were constructed, with areas under the receiver-operating characteristic curves of 0.925 (95% CI: [0.882; 0.968]), 0.908 (95% CI: [0.859; 0.957]), and 0.890 (95% CI: [0.836; 0.944]), respectively. Sex, BMI, chronic disease, and HDL levels are significant predictors of DVT patients with coexisting PE.
Introduction
Deep vein thrombosis (DVT) and pulmonary embolism (PE) are two distinct clinical manifestations of venous thromboembolism (VTE). Studies have consistently reported a high incidence of DVT.1–3 In the United States, the incidence of first-time DVT was 4.8 per 1000 individuals. 4 Meanwhile, a population-based study in Norway found an incidence rate of 0.93 per 1000 person-years (95% CI: 0.85-1.02) for DVT. 5
Normally, PE has been viewed as originating from venous thrombosis of the lower extremities, as initially described by a German physician, and considered a consequence of DVT. 6 A systematic review reported a 1.6% risk of PE at 6 months of anticoagulation therapy in patients with thrombus above the popliteal vein. 7 Another review summarized 21 papers and found the rate of non-fatal PE ranged from 0% to 6.2% in patients with isolated calf deep vein thrombosis (C-DVT). 8
While several reviews have investigated the incidence of PE in DVT patients, adverse outcomes and clinical severity remain inadequately reported and controversial. This lack of evidence hinders the development of clinical decision-making guidelines. Moreover, given the high mortality of PE, the high cost of DVT treatment, and the lack of in-depth research on therapies for PE in DVT patients,9,10 these factors may influence the selection of anticoagulation therapy.
This study analyzed DVT patients with or without coexisting PE, aiming to identify potential risk factors for DVT patients with coexisting PE. Predictive models were constructed to screen patients at high risk for concurrent PE, thereby assisting clinicians in selecting appropriate anticoagulation strategies for DVT patients.
Methods
Study Population
This retrospective study included 150 consecutive DVT patients from Shanxi Bethune Hospital, a large referral general hospital in Shanxi Province, between July 15, 2022, and June 15, 2023. Exclusion criteria were age < 18 years, pregnancy, chronic pulmonary diseases (eg, COPD), malignant tumors, heart failure, and fatal PE that requires immediate thrombolysis. Patients with COPD were not included in this study because several studies suggested that patients with COPD are more likely to develop PE. Inclusion criteria were admission > 3 days, objectively proven DVT or PE and complete clinical data. Of the 130 eligible DVT patients, two groups were formed: non-PE and PE. The patient selection process is depicted in Figure 1. The study received approval from the Ethics Committee of Shanxi Bethune Hospital.
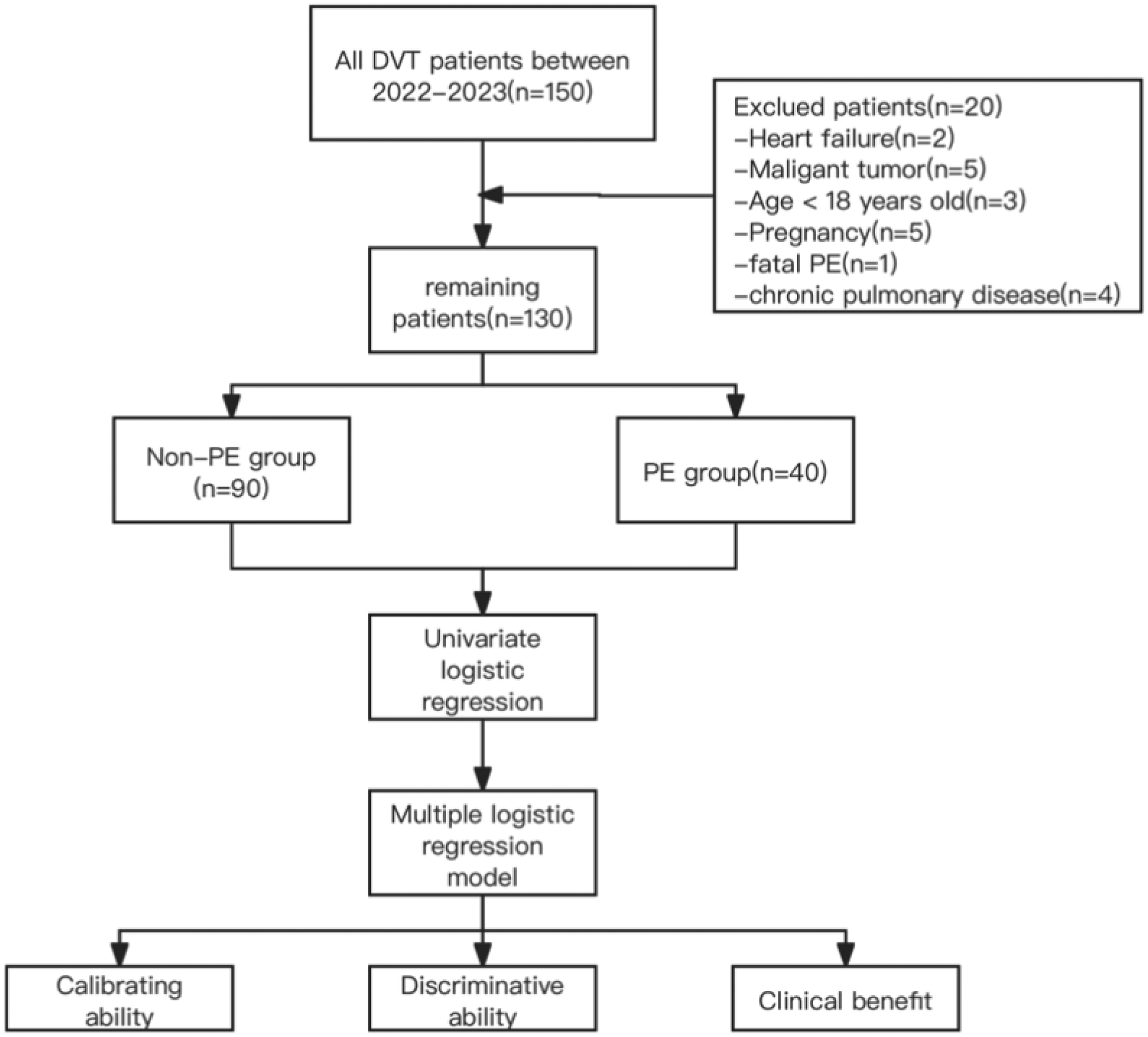
The procedure of the study.
Data Collection
Demographic data, clinical characteristics, and laboratory test results were collected within 3 days of admission. DVT and PE diagnoses were objectively confirmed by compression ultrasonography and computed tomography pulmonary angiography, respectively. When necessary, Doppler ultrasound at the junction of the common femoral vein and great saphenous vein assessed the presence of more proximal venous thrombosis. All scans were evaluated centrally by at least one vascular surgeon and one radiologist blinded to other results. Patients in the PE group were diagnosed with both DVT and PE upon hospital admission. DVT limb was defined as the lower extremity side with blood clots, including left, right, or bilateral. Proximal DVT was defined as thrombus extending above the calf vein levels, while distal DVT was isolated to the calf veins.
Statistics Analysis
Continuous variables were presented as mean ± standard deviation or median (IQR), while categorical variables were reported as frequencies (%). Normality was assessed using the Kolmogorov-Smirnov test. Continuous variables were compared between groups using the t-test or Mann-Whitney U test, as appropriate. Categorical variables were analyzed using the Chi-square test. All analyses were performed using SPSS software 21.0 and R version 4.3.2, with a two-tailed P-value < 0.05 considered statistically significant.
Models’ Development and Validation
Because the dependent variable in our study is a binary variable (yes or no), we chose to build logistic regression model. Multivariate binary logistic regression models were constructed based on univariate logistic regression analysis results. Model performance was evaluated using discrimination, expressed as the area under the receiver-operating characteristic curve (AUC), and calibration, presented through calibration plots. Decision curve analysis visualized clinical effectiveness. Internal validation was conducted using bootstrapping with 1000 repetitions.
Results
Patients’ Characteristics
During the study period, 130 patients with DVT were identified, of whom 40 (30.77%) were diagnosed with PE. The cohort comprised 52 (40%) men, with a mean age of 62.62 ± 18.14 years. Most patients (65/130, 50%) had both proximal and distal DVT. Chronic diseases were present in 44 (33.85%) patients, including 20 (45.45%) with hypertension and 24 (54.55%) with diabetes. Anticoagulation therapy during hospitalization was administered to 72 (55.38%) patients. Demographic and clinical characteristics are shown in Table 1.
The Demographic and Clinical Characteristics of the Patients.
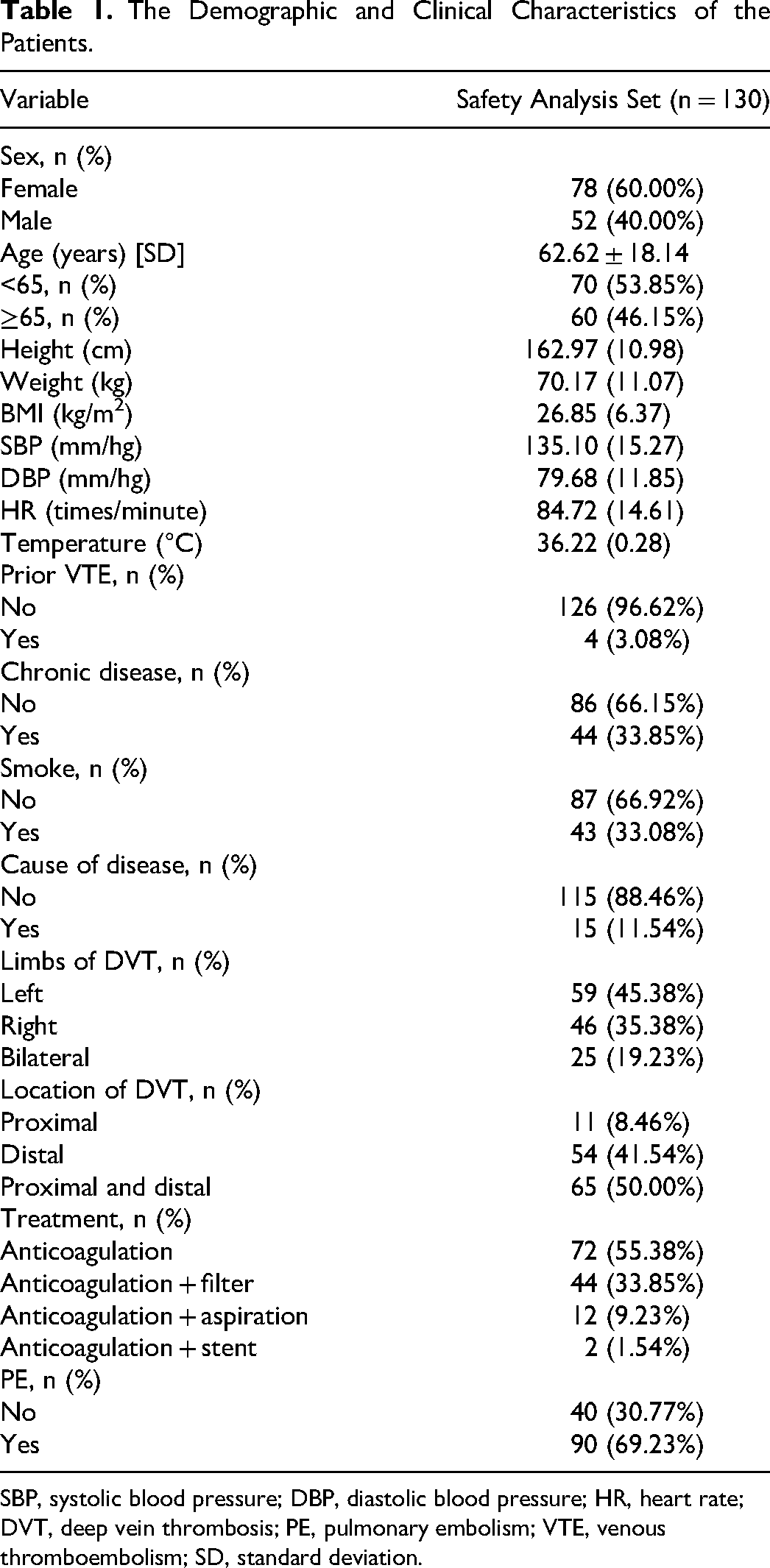
SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; DVT, deep vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism; SD, standard deviation.
Patient baseline characteristics are shown in Table 2. Sex (P = 0.001), body mass index (BMI) (P = 0.001), BMI > 24 kg/m2 (P = 0.001), BMI > 28 kg/m2, (P < 0.001), limbs affected by DVT, and chronic disease differed significantly (P ≤ 0.001) between non-PE and PE groups. Common risk factors like smoking and prior VTE did not differ significantly. Table 3 presents the differences in laboratory test results between groups. D-dimer and high-density lipoprotein (HDL) levels differed significantly (P < 0.001) between groups.
Baseline Characteristics of Two Groups.
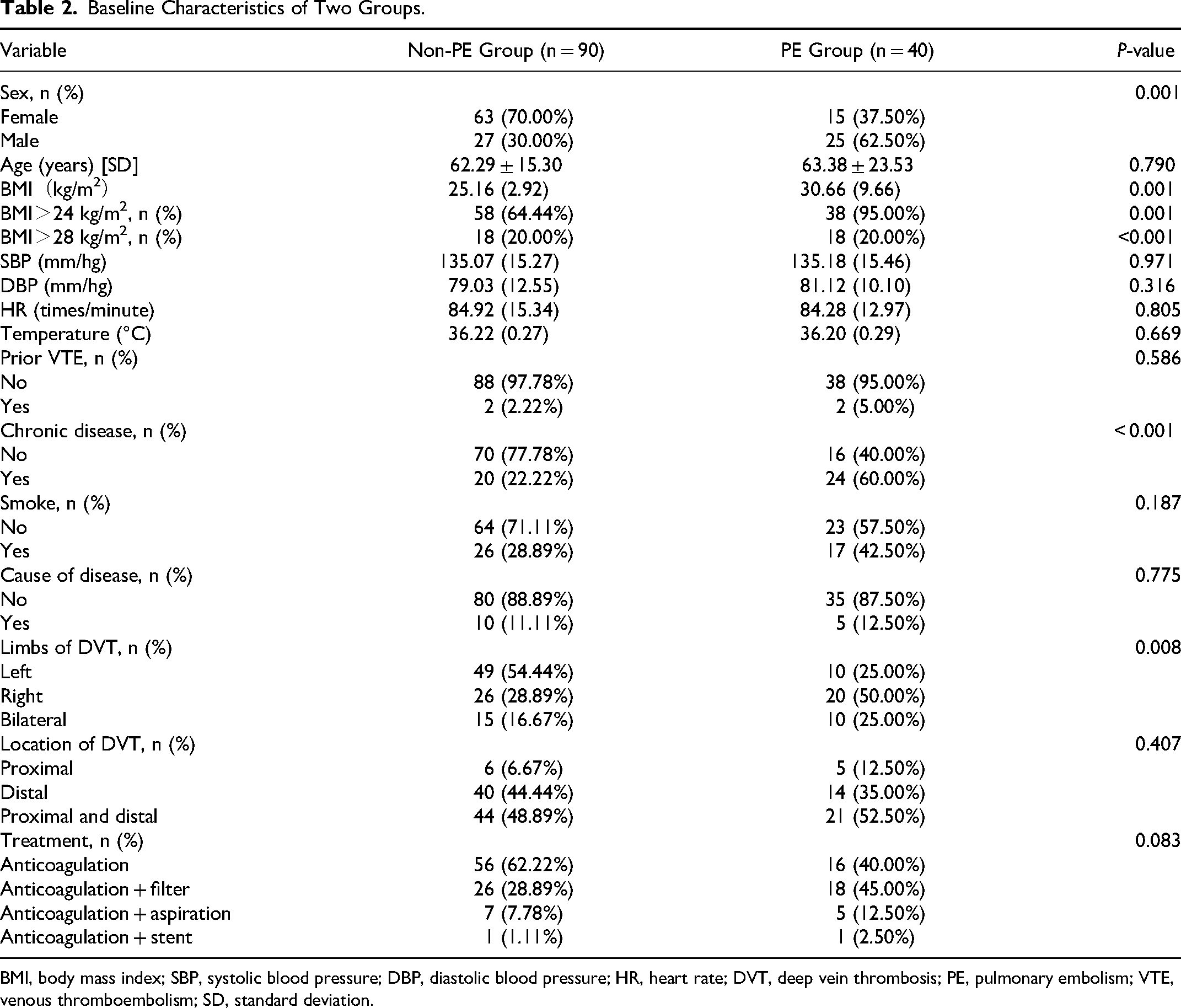
BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; HR, heart rate; DVT, deep vein thrombosis; PE, pulmonary embolism; VTE, venous thromboembolism; SD, standard deviation.
The Laboratory Test Results of the Patients.
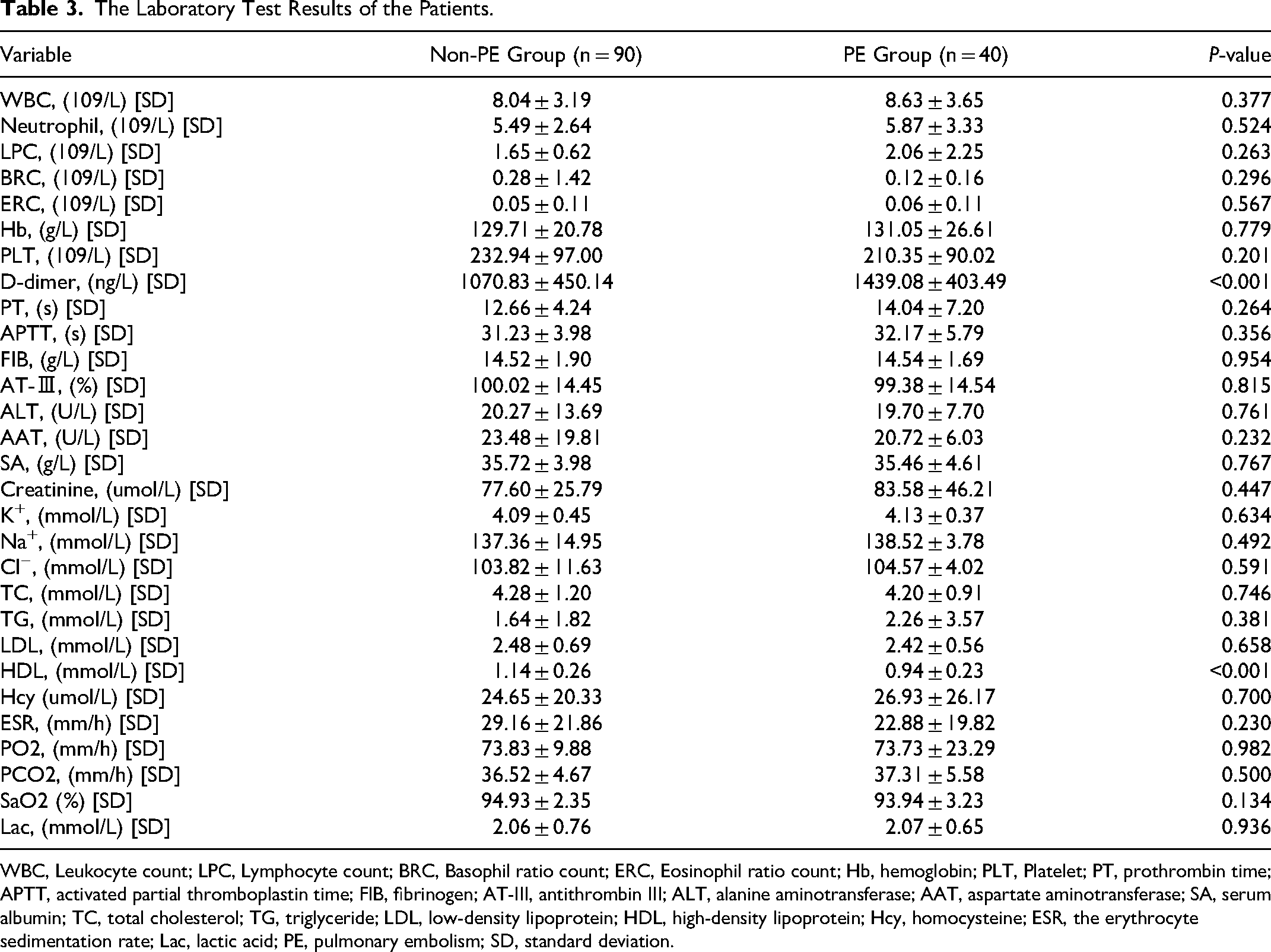
WBC, Leukocyte count; LPC, Lymphocyte count; BRC, Basophil ratio count; ERC, Eosinophil ratio count; Hb, hemoglobin; PLT, Platelet; PT, prothrombin time; APTT, activated partial thromboplastin time; FIB, fibrinogen; AT-III, antithrombin III; ALT, alanine aminotransferase; AAT, aspartate aminotransferase; SA, serum albumin; TC, total cholesterol; TG, triglyceride; LDL, low-density lipoprotein; HDL, high-density lipoprotein; Hcy, homocysteine; ESR, the erythrocyte sedimentation rate; Lac, lactic acid; PE, pulmonary embolism; SD, standard deviation.
Risk Factors and Multiple Logistic Regression Models
Univariate logistic regression analysis (Table 4) identified five significant risk factors for concomitant PE. Male gender in DVT patients was associated with higher odds of concomitant PE (OR 3.83, 95% CI: 1.76-8.59, P = 0.001). Higher BMI (OR 1.50, 95% CI: 1.28-1.75, P < 0.001), chronic disease (OR 5.15, 95% CI: 2.32-11.8, P < 0.001), D-dimer (OR 1.00, 95% CI: 1.00-1.00, P < 0.001), and lower HDL (OR 0.03, 95% CI: 0.01-0.20, P < 0.001) were associated with increased odds of concomitant PE. The relationship between BMI>24 kg/m2, BMI>28 kg/m2, and concomitant PE was further explored. The reason we chose these two BMI ranges is because overweight and obesity are defined as BMI > or = 24 kg/m2 and BMI > or = 28 kg/m2, respectively, in the Chinese criteria. As shown in Table 4, they were also associated with concomitant PE.
The Univariate Logistic Regression Analysis.

BMI, body mass index; HDL, high-density lipoprotein.
Three multivariate binary logistic regression models were constructed, incorporating BMI, BMI>24 kg/m2, and BMI>28 kg/m2 separately, to predict concomitant PE incidence in DVT patients (Figures 2–4). The AUCs were calculated for the models (Figure 5). The respective AUCs were 0.925 (95% CI: 0.882-0.968), 0.908 (95% CI: 0.859-0.957), and 0.890 (95% CI: 0.836-0.944). Calibration curves (Figures 6–8) demonstrated good predictive performance. Decision curves (Figures 9–11) exhibited the clinical utility of these models.
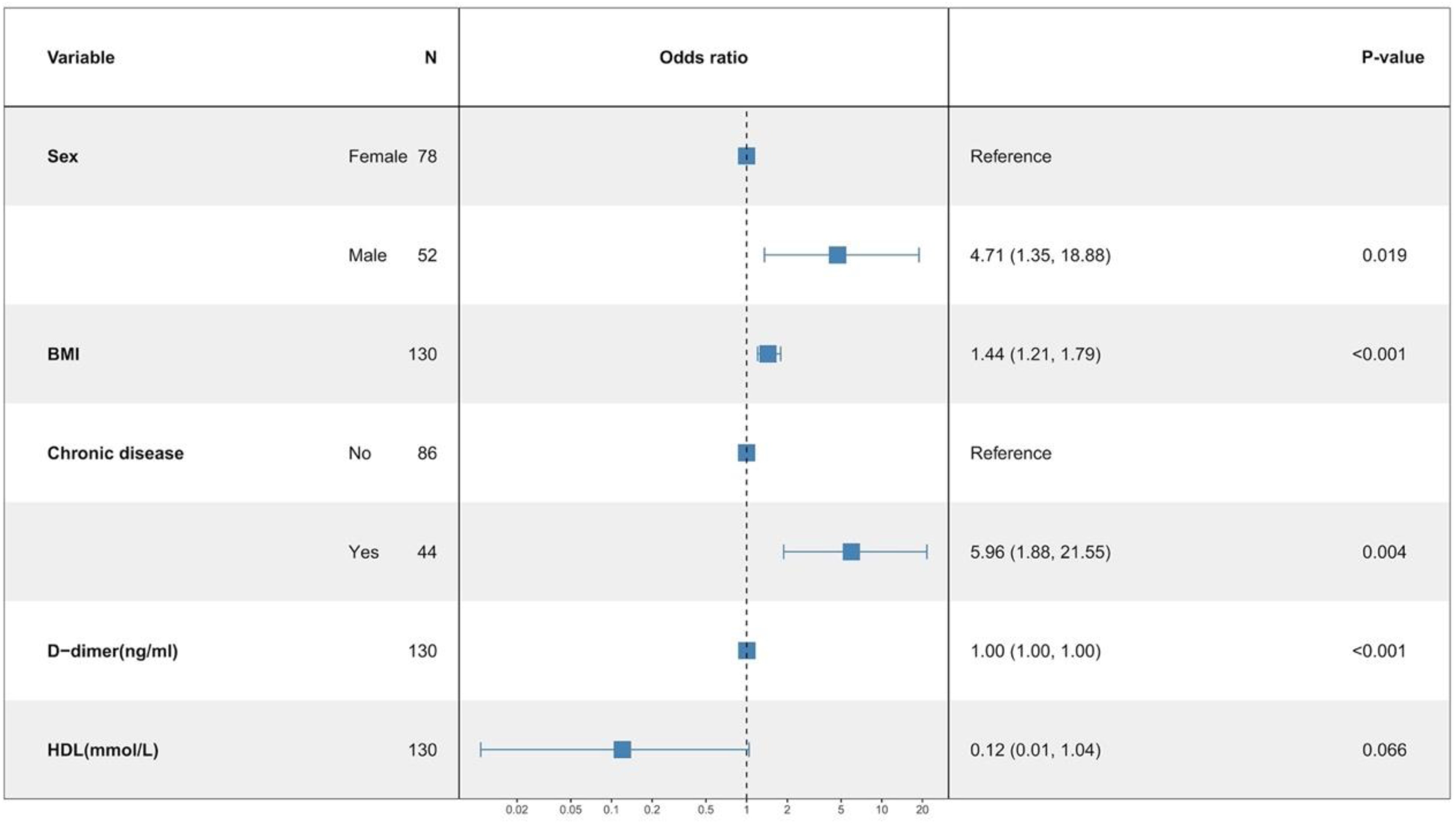
The multiple logistic regression model 1.
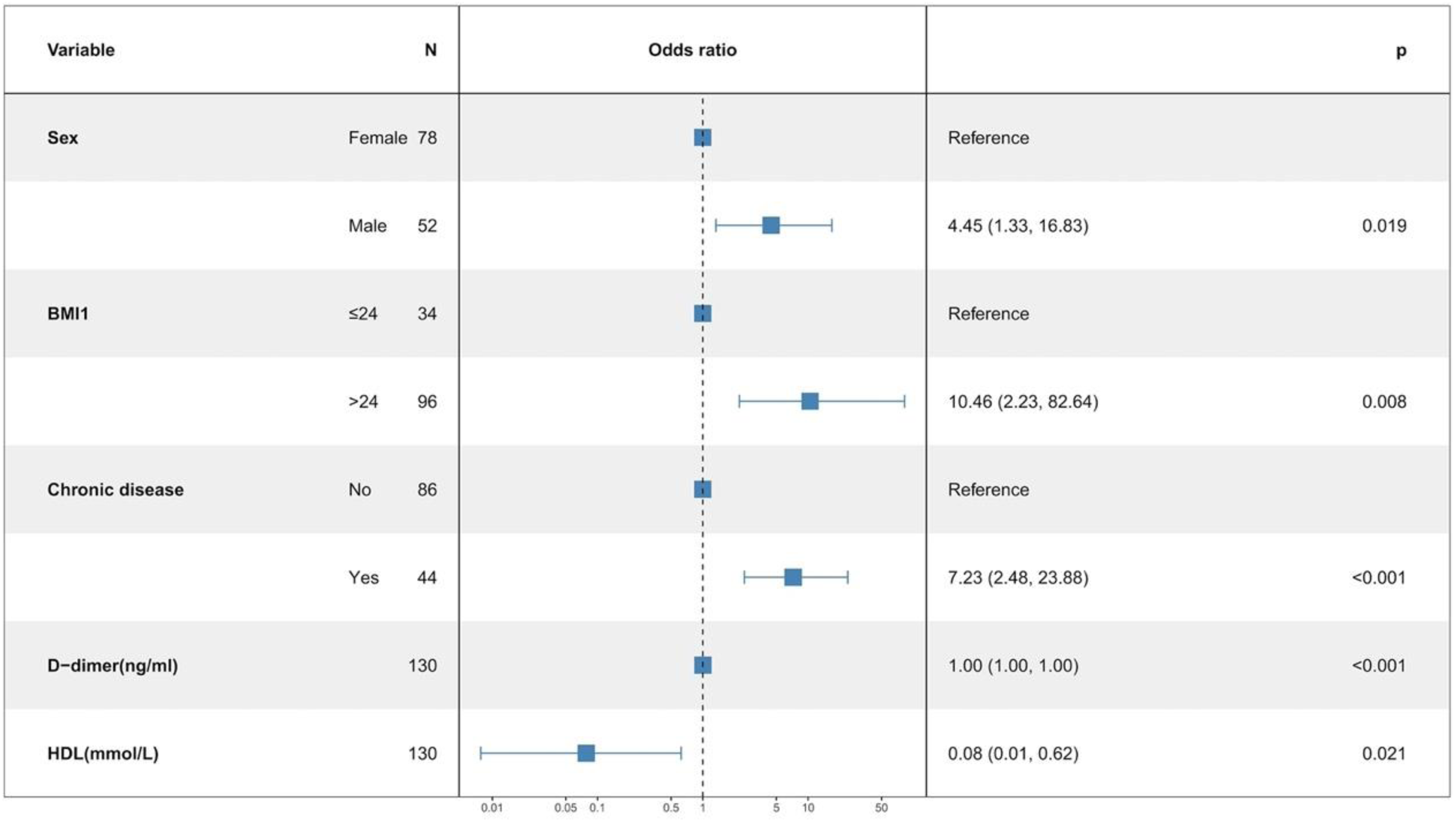
The multiple logistic regression model 2.
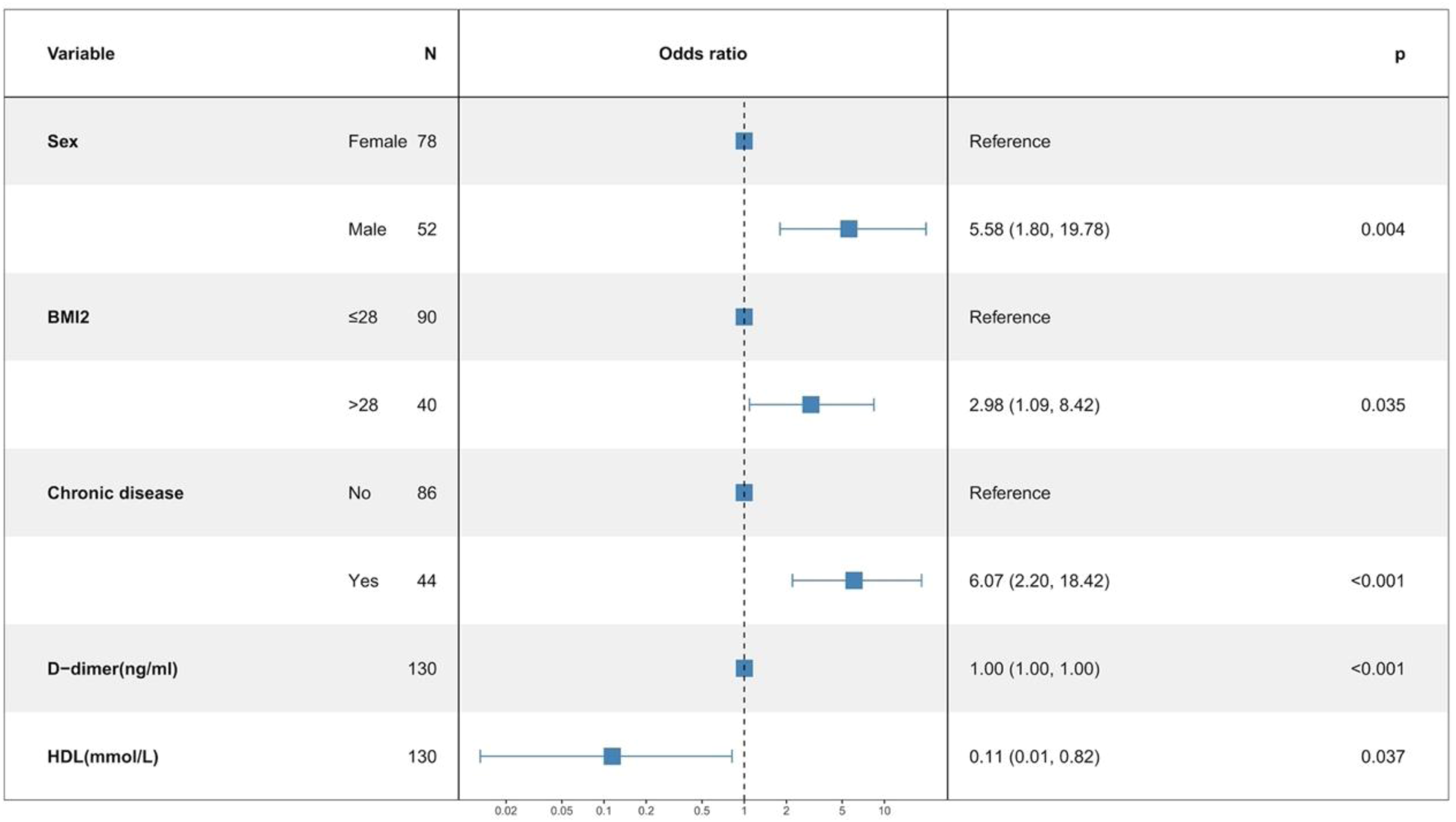
The multiple logistic regression model 3.
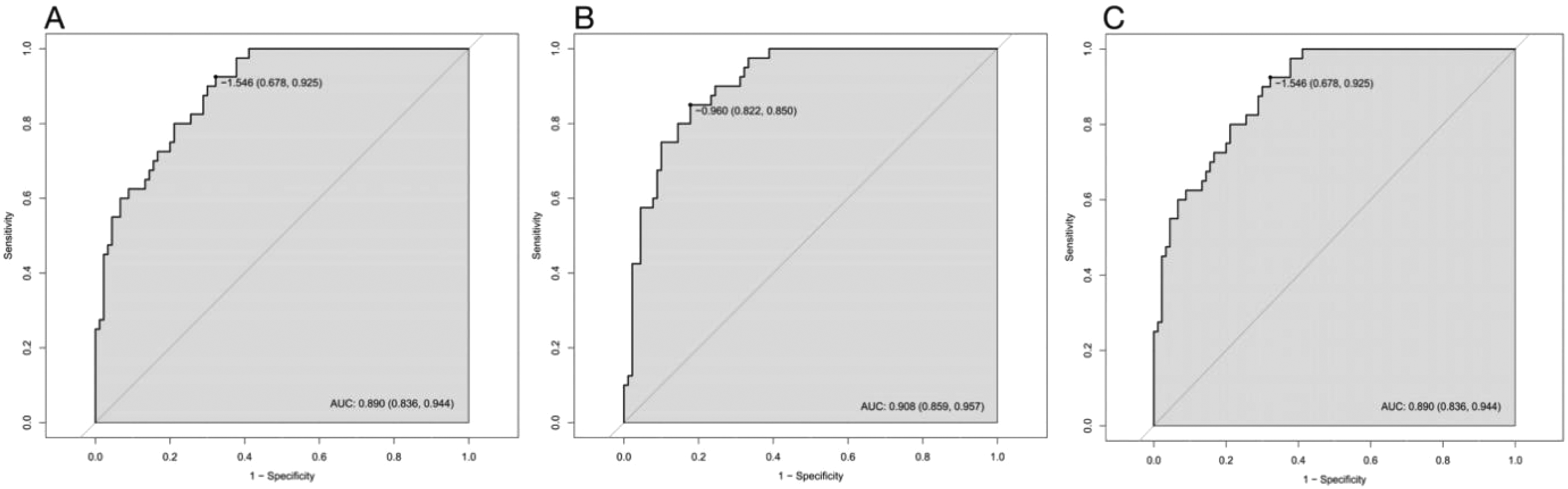
The AUCs of multiple logistic regression models. (a) The AUC of model 1; (b) the AUC of model 2; and (c) the AUC of model 3.
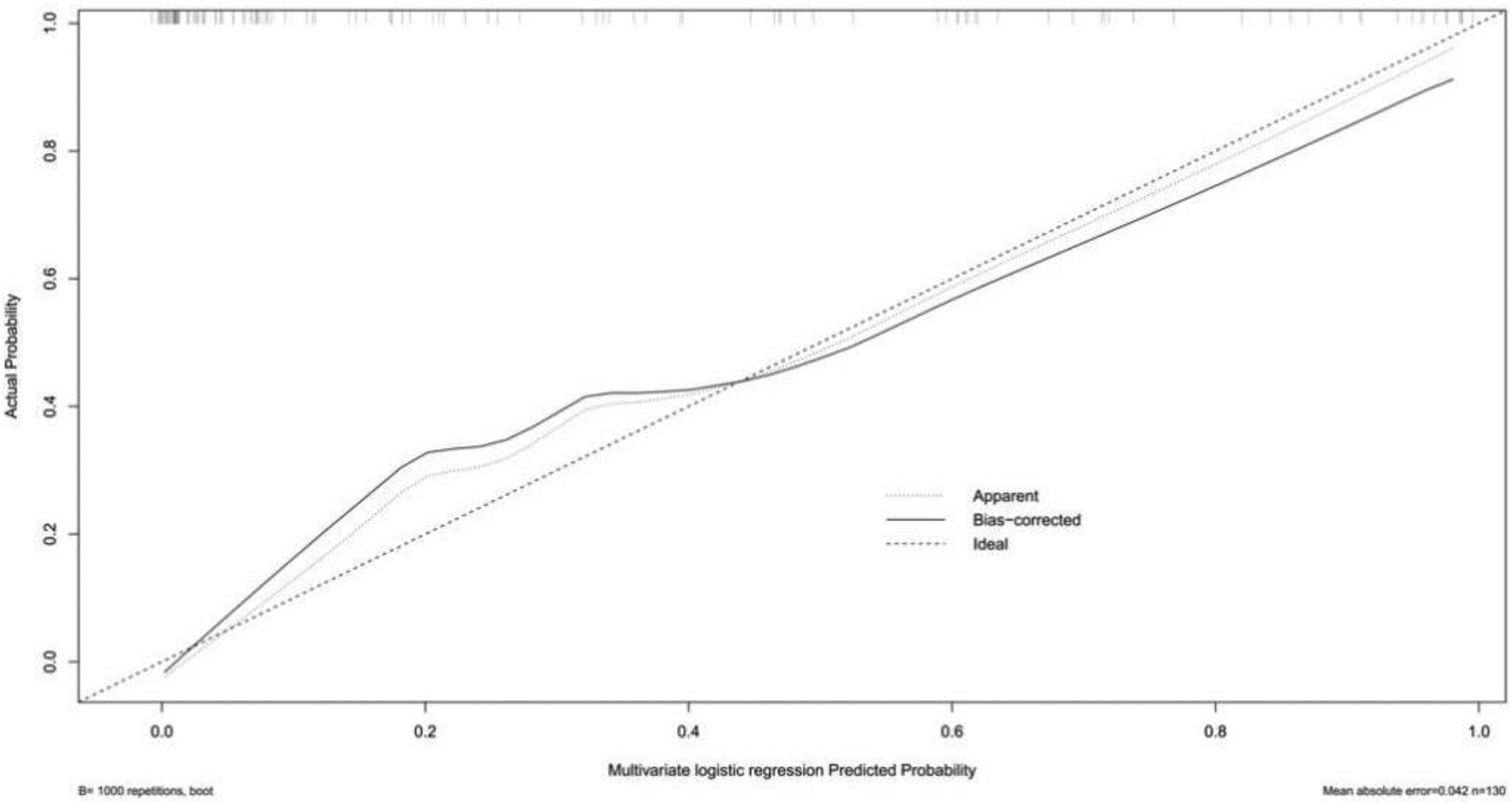
The calibration curve of multiple logistic regression model 1.
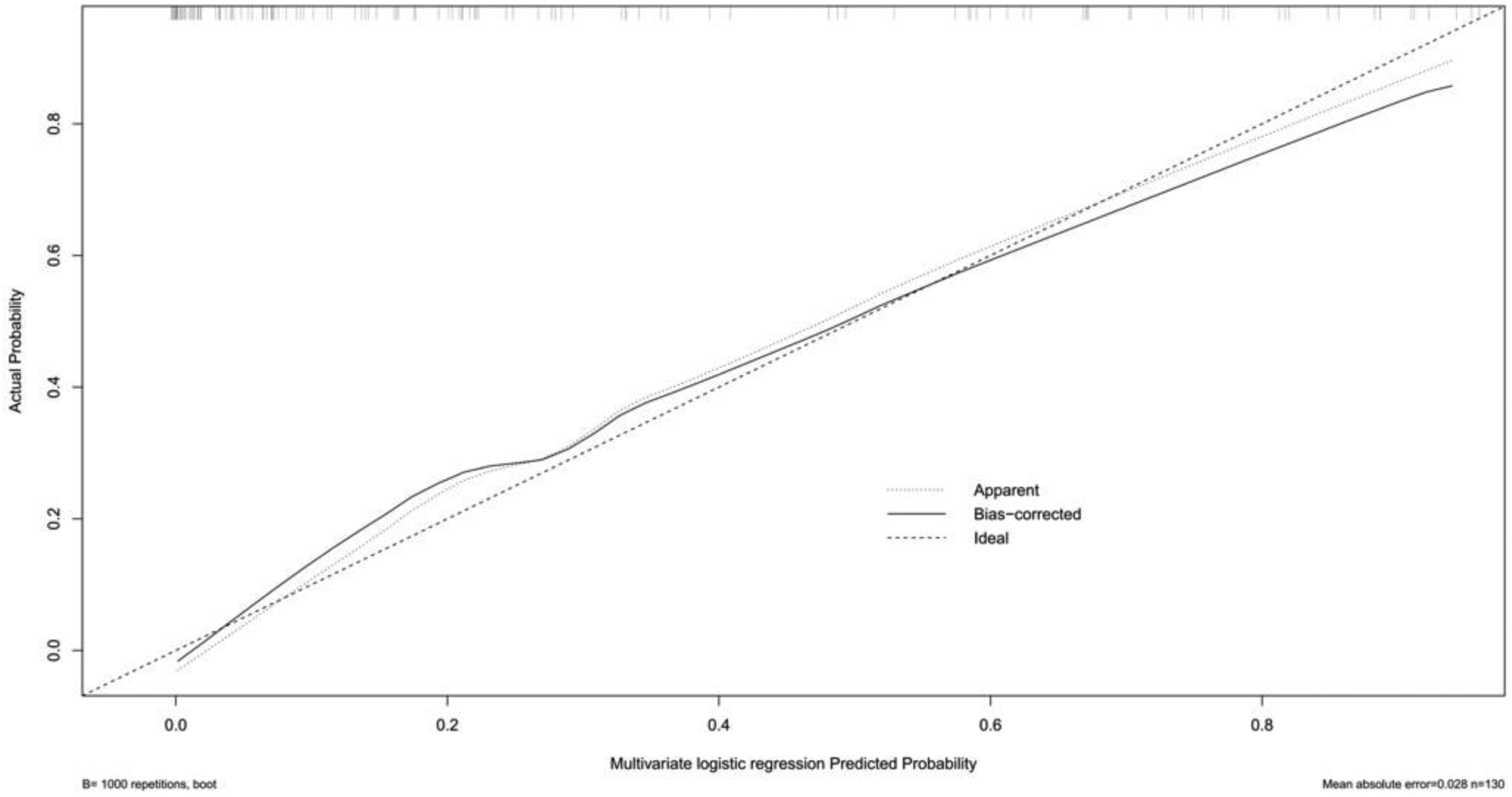
The calibration curve of multiple logistic regression model 2.
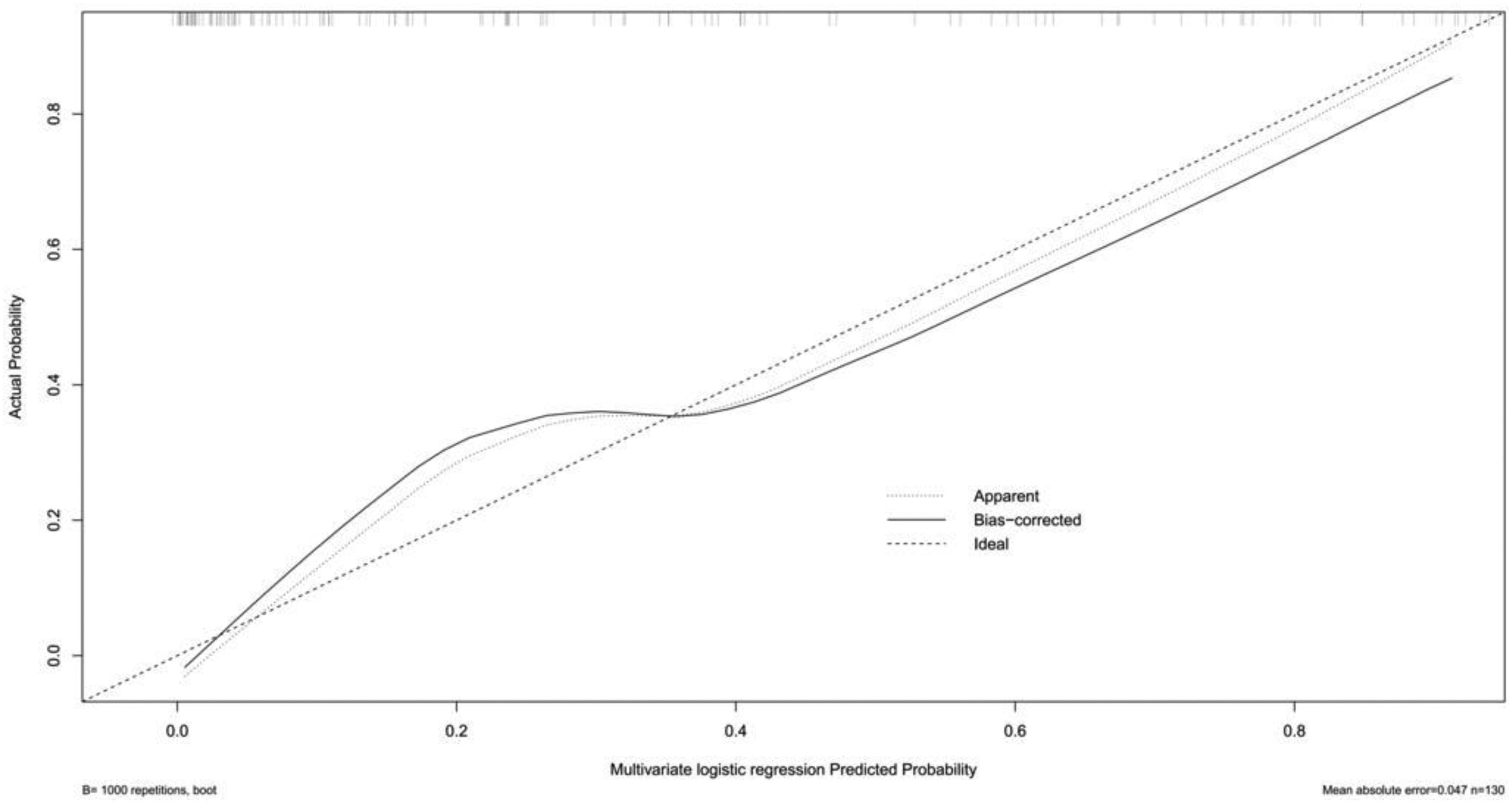
The calibration curve of multiple logistic regression model 3.
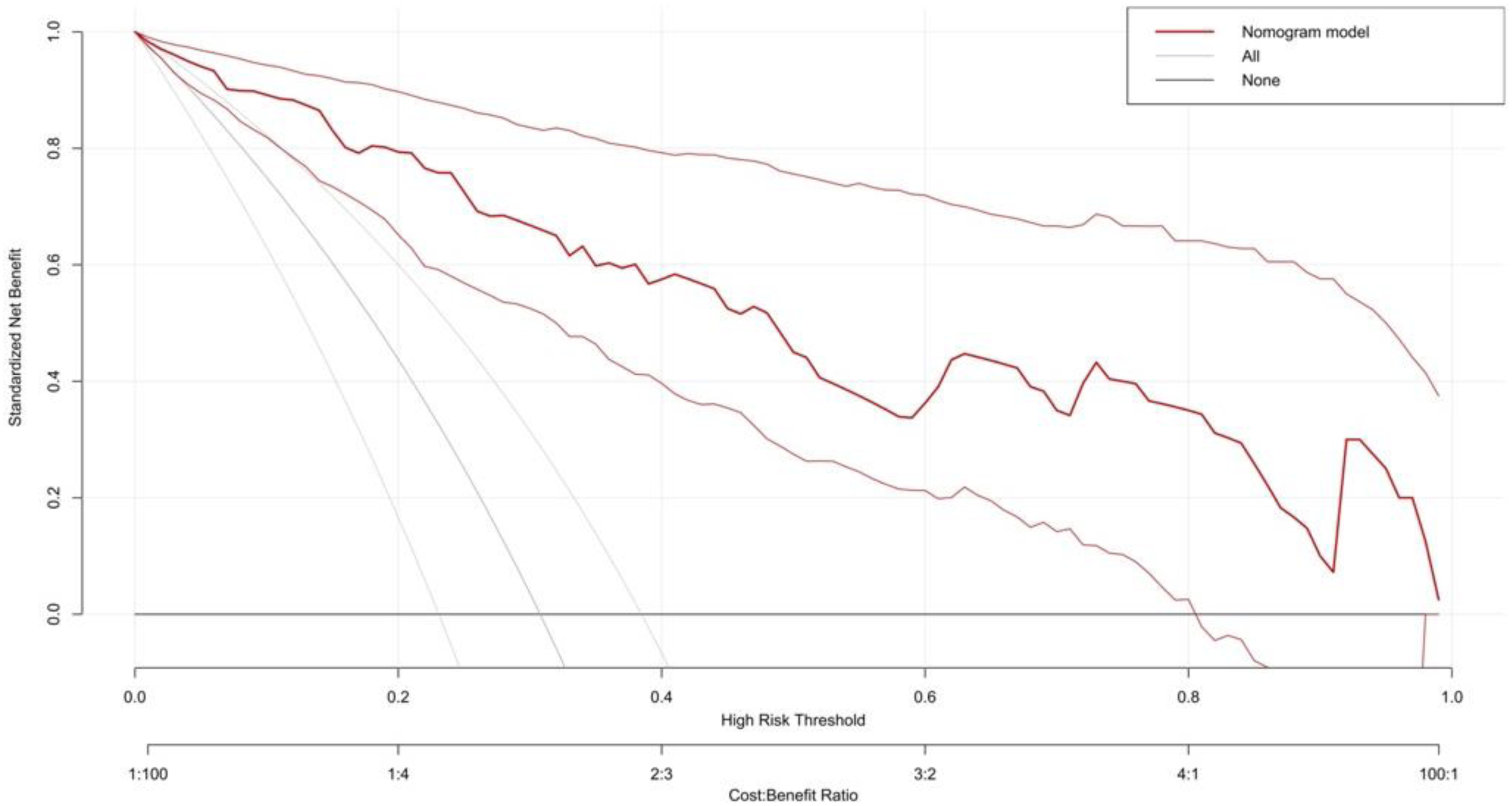
The decision curve of multiple logistic regression model 1.
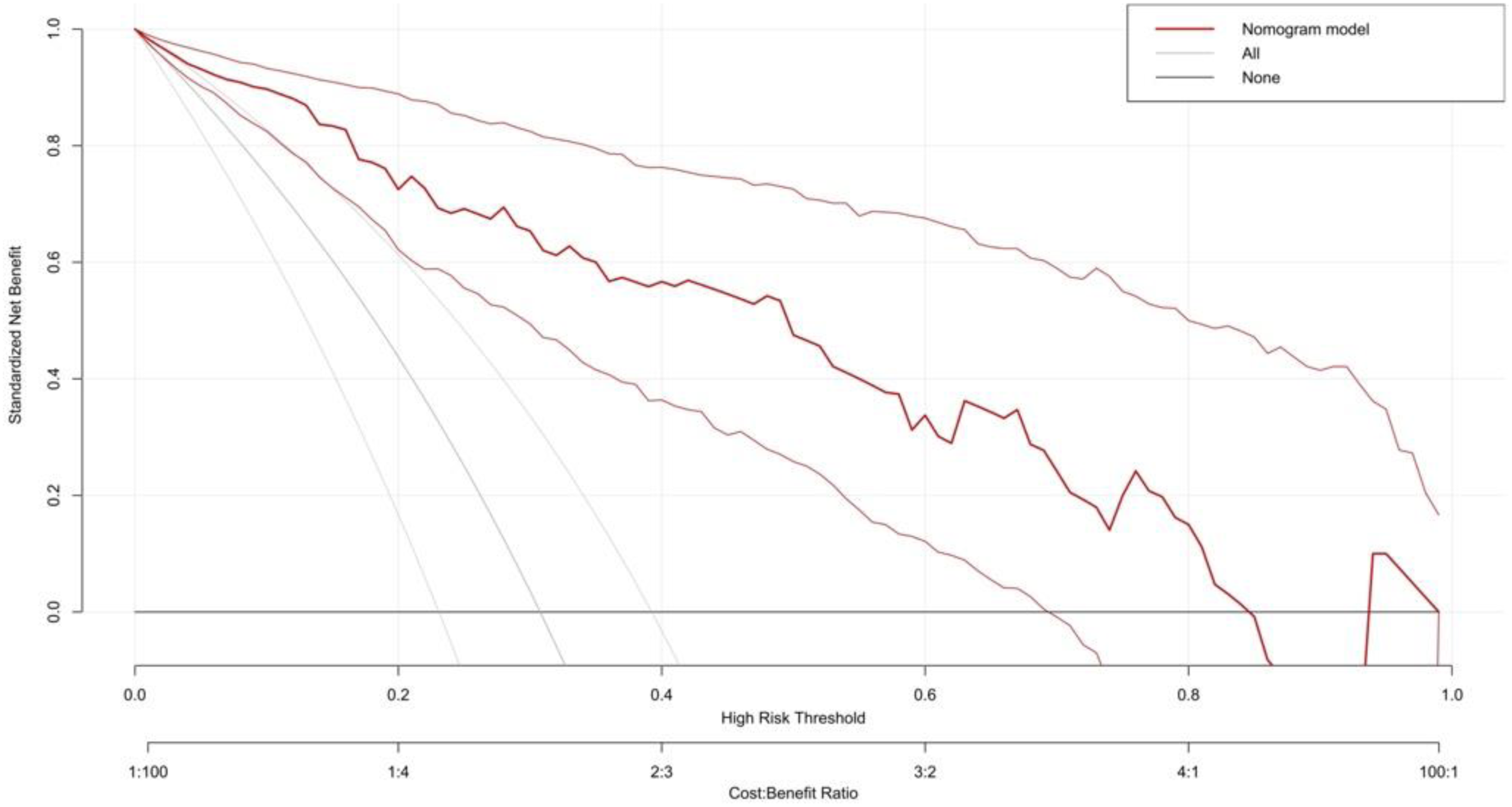
The decision curve of multiple logistic regression model 2.
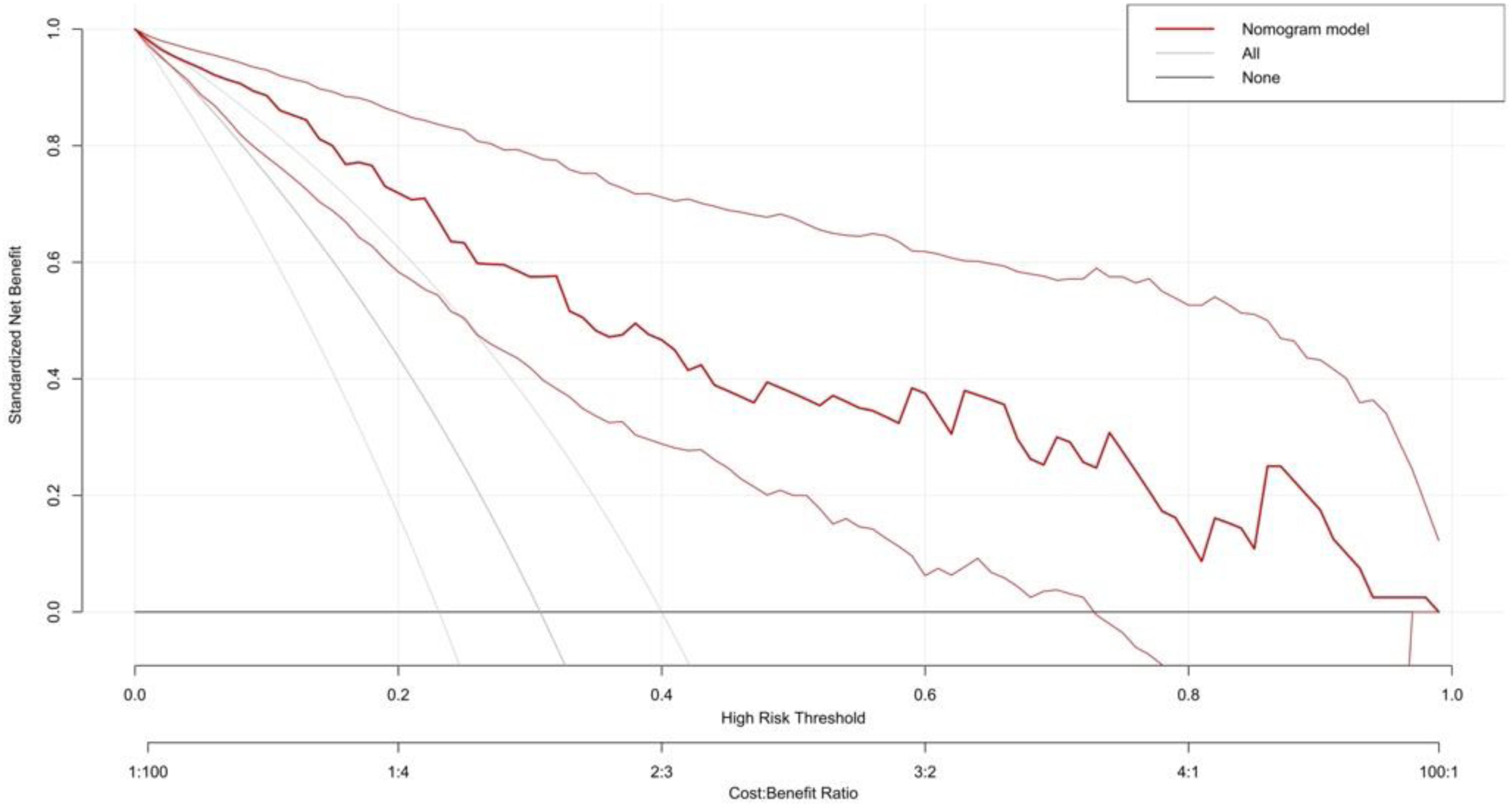
The decision curve of multiple logistic regression model 3.
Discussion
Our study predominantly involved isolated DVT cases without fatal PE events. Sex, BMI, chronic disease, D-dimer, and HDL levels were identified as risk predictors for concomitant PE in DVT patients. The proportion of concomitant PEs (40/130, 30.77%) in our population exceeded that reported in several previous studies.7–9 The higher incidence rate may be attributable to the inclusion of both proximal and distal DVT patients.
Numerous studies have confirmed the association between sex and VTE. A Swedish study demonstrated a higher risk of VTE in men compared to women. 11 Another retrospective cohort study reported a twofold higher risk of first-time VTE in men, 12 consistent with previous studies on recurrent VTE.13,14 Our study similarly indicated a higher likelihood of concomitant PE in male DVT patients. However, some studies have come to the opposite conclusion from ours. One comparative study reported a higher prevalence of PE in older females. 15 A Korean study by Choi et al found the same thing. Among patients older than 50 years, the rate of PE was higher in women (0.15%; 0.11-0.19%) than in men (0.08%; 0.05-0.11%; P < 0.01). 16 A systematic review, involving 93 eligible studies, also summarized that the risk of PE in older women (especially > 40 years old) was higher than that in men of the same age. 17 Jarmen et al 18 suggested a different result that the age adjusted incidence of PE is similar in men and women. This discrepancy may be attributable to the pattern of PE occurrence tended to change with age for both men and women after age adjustment.
Multiple studies have demonstrated BMI as a risk factor for cardiovascular disease. Hu et al 19 supported BMI as an independent risk factor for VTE, DVT, and PE, consistent with our findings. A systematic review analyzing 15 high-quality studies concluded that obesity more than doubles the risk of venous embolism. 20 An Italian study and a case-control study also observed the same association between obesity and thrombosis. Another review concluded a weak relationship between overweight and VTE, despite potential increased risks. 21 These inconsistent findings warrant further in-depth analysis of overweight patients.
Several studies have investigated the potential association between VTE and chronic diseases like hypertension and diabetes.22–24 A meta-analysis reported increased VTE risk for diabetes (OR 1.42, 95% CI: 1.12-1.77) and hypertension (OR 1.51, 95% CI: 1.23-1.85) compared to control. Our study yielded a similar result (OR 5.15, 95% CI: 2.32-11.8). However, Mahmoodi et al 25 and Lind et al 11 reported a lower VTE risk in hypertensive patients and could not reproduce these findings. These discrepancies are likely attributable to residual confounding factors.
Previous studies have demonstrated D-dimer as an important VTE biomarker, consistent with our findings. Elevated D-dimer levels, resulting from the degradation of cross-linked fibrin, reflect hemostasis, hypercoagulable state, and hyperfibrinolysis.26,27 D-dimer is suggested for predicting VTE incidence and recurrence risk.28,29 D-dimer been incorporated in several nomograms for predicting VTE risk in cancer patients.30,31 Additionally, the combination of D-dimer cut-off value with pretest clinical probability assessed by some scores, which is a common diagnostic algorithm for ruling out PE, is used to improve the efficiency of diagnosing and reduce unnecessary imaging. One study in 2006 resulted that PE can be ruled out when the Wells score of patients is equal or less than four and D-dimer is ≤ 500 μg/L. 32 A systematic review found that Wells rule combined with age-adjusted D-dimer, defined as the age multiplied by 10 μg/L in patients older than 50 years, can increase the efficiency to 33% and keep its failure rate less than 3% in three subgroups compared to fixed D-dimer. 33 Another prospective study also has the same result while it used revised Geneva score, or the 2-level Wells score to assess clinical probability. 34
Several studies have confirmed an association between elevated HDL levels and decreased VTE risk.35–37 A meta-analysis reported 2.86 mg/dL (95% CI: 1.38-4.34) lower HDL levels in VTE patients compared to non-VTE patients. 38 This suggests that elevated HDL is a protective factor against VTE and venous thrombosis, consistent with our results. The potential mechanism involves the anti-atherosclerotic thrombotic properties of HDL, including suppression of platelet aggregation, inhibition of tissue factor and PAI-1 activity, reduced viscosity, and enhanced factor Va inactivation via activated protein C. 39
To our knowledge, no predictive model exists for concomitant PE in DVT patients, and anticoagulation management in this population remains uncertain. We constructed three intuitive multiple logistic regression models to facilitate early identification of high-risk patients and guide clinical management.
Nevertheless, our study has certain limitations: first, the retrospective nature of the study and limited patient inclusion. Second, the odds ratios for D-dimer in the three models were 1.00 (95% CI: 1.00-1.00), despite a P-value <0.001. This result suggests no statistically significant association between D-dimer and concomitant PE. However, this may be due to limited sample size, and a larger sample may reveal a significant association. Finally, a direct comparison between overweight and normal-weight patients was not conducted.
Conclusion
We constructed three multiple logistic regression models for predicting concomitant PE risk in DVT patients. These models demonstrated high discrimination and clinical validity.
Footnotes
Acknowledgments
The authors have no acknowledgments in this article.
Author Contributions
TY designed this research. JHZ and CHS collected and analyzed the data. JHZ wrote and revised this article. All authors contributed to the article and approved the submitted version.
Data Sharing Statement
All data generated and analyzed during the study are included in this article. Further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This is a retrospective and observational study. Ethical approval was not required for the study involving humans in accordance with the local legislation and institutional requirements. All patients in this study gave written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
