Abstract
Background
Peripheral artery disease (PAD) is a major contributor to cardiovascular morbidity and mortality, yet reliable biomarkers for early detection remain limited. Glycated hemoglobin (HbA1c) reflects long-term glycemia, but its relationship with PAD in the general population remains incompletely understood.
Methods
We analyzed 6284 adults aged ≥40 years from the 1999–2004 National Health and Nutrition Examination Survey (NHANES). PAD was defined as an ankle–brachial index ≤0.90. Weighted logistic regression, restricted cubic spline, and threshold models evaluated associations between HbA1c and PAD. To further examine causality, a two-sample Mendelian randomization (MR) using genome-wide association data on HbA1c (n = 389,889) and PAD (7098 cases, 206541 controls) was conducted.
Results
In NHANES, each 1% increment in HbA1c was linked to a higher likelihood of PAD (fully adjusted OR 1.18, 95% CI 1.04-1.34). Participants in the highest HbA1c tertile had more than a twofold greater risk compared with those in the lowest (OR 2.30, 95% CI 1.68-3.15). Restricted cubic spline analysis revealed a nonlinear relationship with a threshold at 6.11%, below which PAD risk rose steeply. In MR analysis, genetically predicted HbA1c was positively associated with PAD risk, showing that genetically predicted HbA1c was positively associated with PAD (IVW OR 1.23, 95% CI 1.10-1.37, p = 0.0002), with consistent results across sensitivity analyses.
Conclusion
Elevated HbA1c is independently and causally linked to PAD risk, with a nonlinear dose–response pattern. These findings support HbA1c as a potential tool for identifying individuals at higher PAD risk.
Keywords
Introduction
Peripheral artery disease (PAD) represents a common manifestation of systemic atherosclerosis and contributes substantially to cardiovascular morbidity and mortality. It affects an estimated 236 million individuals worldwide, with prevalence ranging from 4%–10% among U.S. adults over 40 years and exceeding 20% in those over 65. 1 Nearly 90% of cases are asymptomatic or unrecognized, particularly among women,2,3 leading to substantial underdiagnosis. Clinical presentations vary from intermittent claudication and cold extremities to ulcers and, in advanced stages, critical limb ischemia with risk of amputation. The development of PAD is driven by multiple risk factors, with age being one of the strongest determinants, 4 while smoking and diabetes nearly triple the risk, and hypertension and dyslipidaemia further accelerate disease progression.5,6 Therefore, identifying modifiable determinants is essential for improving early prevention and intervention strategies.
Glycated hemoglobin (HbA1c) reflects average blood glucose levels over the preceding two to three months and is widely used to diagnose diabetes, monitor glycemic control, and predict long-term complications. Elevated HbA1c has been consistently associated with increased risks of cardiovascular and limb events in individuals with diabetes and PAD, highlighting the vascular consequences of chronic hyperglycaemia.7,8 Importantly, a graded, independent association between HbA1c and PAD has also been observed in individuals without diabetes, 9 where even modest elevations (≥5.3%) may identify high-risk individuals who could benefit from earlier intervention. 10 Together, these findings suggest that HbA1c may function as a clinically relevant marker of PAD risk beyond traditional diabetes thresholds.
The National Health and Nutrition Examination Survey (NHANES) provides nationally representative data on the health and nutrition of U.S. adults. Prior NHANES-based analyses of HbA1c and PAD have predominantly stratified participants by diabetes status, while few have evaluated HbA1c as an independent, continuous biomarker of PAD risk. 11 Current evidence is also largely confined to observational associations, which cannot establish causality and remain vulnerable to confounding and reverse causation. Mendelian randomization (MR), which employs genetic variants as instrumental variables, provides a complementary approach for evaluating potential causal relationships in this context.12,13
In the present study, we combined a large-scale cross-sectional analysis of NHANES 1999–2004 with a two-sample MR approach to comprehensively assess the relationship between HbA1c and PAD. This integrative design aimed to determine whether elevated HbA1c levels contribute causally to the development of PAD, thereby providing additional insight for risk stratification and prevention strategies.
Methods
Study Population and Design
NHANES is a nationally representative program designed to evaluate the relationship between nutrition and health in the U.S. population. During the 1999–2004 survey cycles, 31126 participants were enrolled, of whom lower limb examination data were available for adults aged ≥40 years (n = 9970). For the present analysis, we included all eligible individuals who underwent lower limb disease assessments. We excluded participants with missing ankle–brachial index (ABI) data (n = 3040), participants with ABI > 1.4 (n = 103), and participants without glycated hemoglobin (HbA1c) measurements (n = 196). We further excluded individuals with incomplete covariate information (n = 347). After these exclusions, 6284 participants with complete data were included in the final analysis (Figure 1).
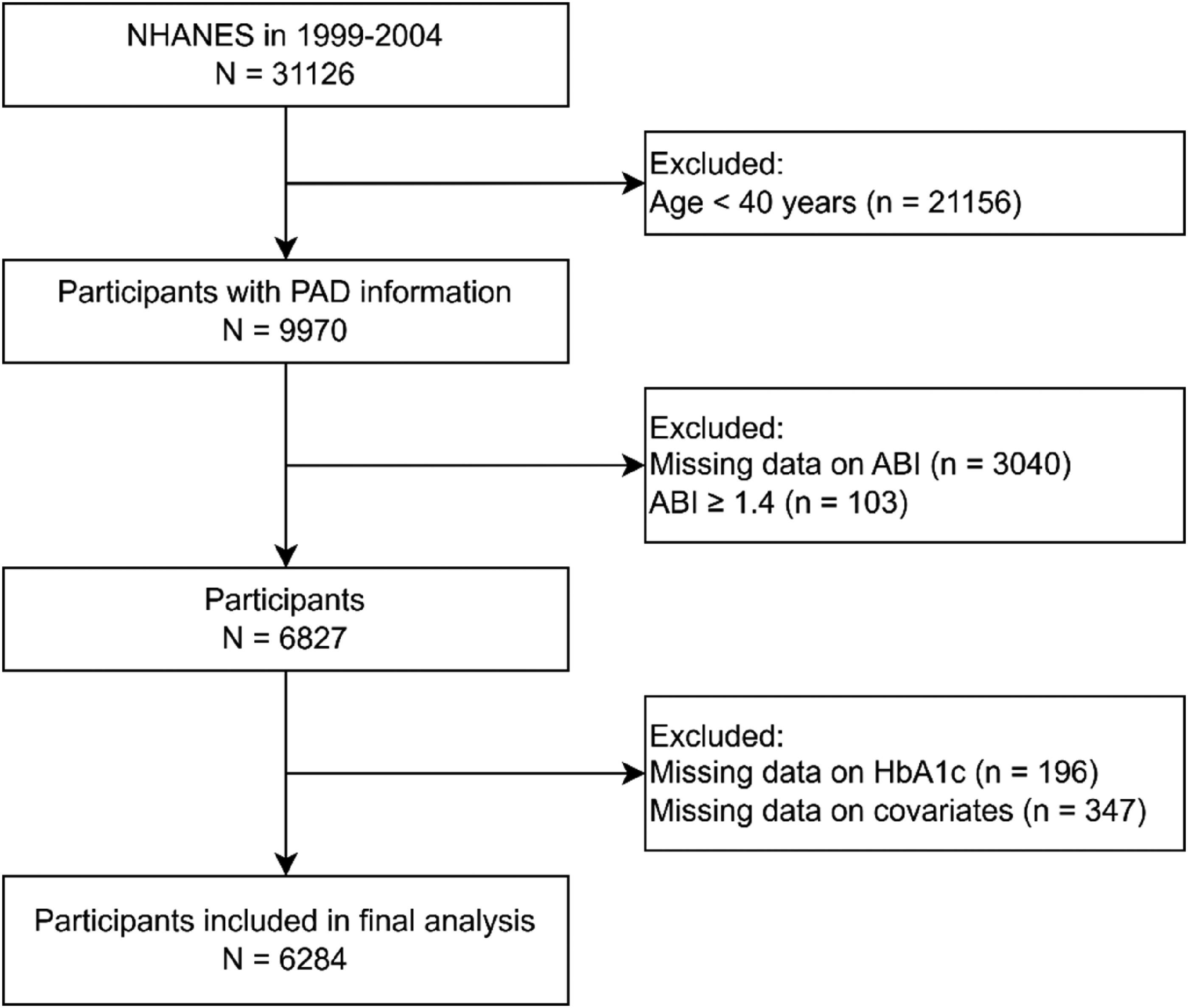
Flowchart of sample selection from NHANES 1999–2004. NHANES, National Health and Nutrition Examination Survey; PAD, peripheral arterial disease; ABI, ankle–brachial index; HbA1c, glycosylated hemoglobin; DM, diabetes.
Exposure and Outcomes
In this study, HbA1c was treated as the primary exposure. HbA1c was measured from whole blood samples using standardized laboratory protocols in NHANES. It was analysed both as a continuous variable and as a categorical variable. For categorical analyses, participants were divided into tertiles according to the distribution of HbA1c levels in the study population: T1 (3.3%-5.3%), n = 2312 (44.92%); T2 (5.3%-5.7%), n = 2253 (34.95%); and T3 (5.7%-18.8%), n = 1719 (20.13%).
PAD was defined using ABI according to established criteria applied in previous NHANES studies. ABI was calculated as the ratio of the average ankle systolic blood pressure (posterior tibial artery or dorsalis pedis artery) to the average brachial systolic blood pressure, measured in the right arm using a standardized protocol. PAD was defined as an ABI ≤0.90 in either leg, consistent with clinical and epidemiological criteria reported in previous research. Participants with an ABI >1.40, suggestive of non-compressible arteries, were excluded from the analysis.
Assessment of Covariates
To reduce potential confounding, we adjusted for demographic, lifestyle, and clinical variables defined in the NHANES protocol. Age (years) and sex were recorded at baseline. Race/ethnicity was classified as non-Hispanic White, non-Hispanic Black, Hispanic, or other. Educational attainment was categorized as less than high school, high school graduate, or college and above. Smoking status was defined as current (≥100 cigarettes in lifetime and still smoking), former (≥100 cigarettes in lifetime but not currently smoking), or never (<100 cigarettes in lifetime and not currently smoking). Alcohol consumption was categorized as never (no alcohol use both over the lifetime and in the past 12 months), former (lifetime alcohol use but none in the past 12 months), or current (≥12 drinks in lifetime and at least one drink in the past 12 months). Diabetes mellitus was defined as self-reported physician diagnosis, use of oral hypoglycemic agents or insulin, fasting plasma glucose ≥7.0 mmol/L, or HbA1c ≥ 6.5%. Hypertension was defined as self-reported physician diagnosis, current use of antihypertensive medications, mean systolic blood pressure ≥130 mm Hg, or mean diastolic blood pressure ≥80 mm Hg. Use of lipid-lowering therapy was determined from prescription records and self-report. Kidney function was assessed using the estimated glomerular filtration rate (eGFR), derived from the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. 14
Data Sources for MR
We conducted a two-sample Mendelian randomization analysis in accordance with the Strengthening the Reporting of Mendelian Randomization Studies (STROBE-MR) guidelines. 15 The GWAS summary statistics for HbA1c were obtained from the study by Mbatchou et al, which included a total of 389889 individuals. Summary statistics for PAD were retrieved from the FinnGen database (https://www.finngen.fi/en/access_results), comprising 7098 cases and 206,541 controls. 16
Instrumental Variable Selection
First, SNPs associated with HbA1c at genome-wide significance (p < 5e−08) were selected as instrumental variables. We subsequently applied a threshold of r2 less than 0.001 and excluded SNPs within a 10000 kb range that were in linkage disequilibrium (LD) and SNPs strongly associated with PAD (p < 5e−08) were removed. The strength of the selected SNPs as IVs was assessed via the F statistic, which was calculated as β2/SE2, where β represents the effect size of the SNP on the exposure, and SE is the standard error of the β value.17–19 We chose SNPs with an F statistic greater than 10. Selected SNPs were then extracted from the PAD GWAS dataset, and unavailable variants were excluded. The remaining SNPs were utilized as the final IVs for the MR analysis. Finally, SNP information from HbA1c and PAD datasets was harmonized for two-sample MR analysis.
Statistical Methods
All analyses were conducted in strict accordance with the NHANES analytic guidelines. Baseline characteristics were summarized according to HbA1c tertiles. Continuous variables were expressed as weighted means ± standard deviations and compared using the design-based Kruskal–Wallis test. Categorical variables were presented as weighted frequencies (percentages) and compared using Pearson's χ2 test with the Rao–Scott adjustment. To evaluate the association between HbA1c and PAD, four stepwise multivariable logistic regression models with survey weights were constructed. Model 1 was unadjusted. Model 2 adjusted for demographic factors (age, sex, race/ethnicity, and education level). Model 3 was further adjusted for lifestyle factors including smoking status, alcohol consumption, and BMI. Model 4 additionally adjusted for hypertension, use of lipid-lowering drugs, and eGFR. To assess the stability of the findings, stratified analyses were conducted. Restricted cubic spline (RCS) regression with survey weights was used to assess potential nonlinear relationships between HbA1c and PAD, and generalized additive models (GAMs) were applied to produce smooth curves. Threshold effect analysis was further performed using the segmented package to identify possible inflection points of HbA1c related to PAD risk.
We conducted a two-sample MR analysis using the harmonized SNPs. For exposures with ≥3 IVs, we applied six methods—Inverse Variance Weighted (IVW), MR Egger, Weighted Median, Simple Mode, Weighted Mode and MR-ConMix—for MR analysis. The primary estimate was derived from the IVW method. 20 First, we employed Cochran's Q test to assess hetero-geneity among IVs. If the p value was greater than 0.05, there was no heterogeneity among the IVs. Second, we used the MR‒Egger regression intercept to test the horizontal pleiotropy of IVs. If the p value is greater than 0.05, we assume that the MR results are not influenced by horizontal pleiotropy. Evidence of horizontal pleiotropy would indicate potential bias in the causal estimates. Therefore, regardless of the significance of MR results, the presence of horizontal pleiotropy suggests no association between exposure and outcome. Finally, we conducted a leave-one-out analysis to examine whether there were any IVs that exerted a substantial effect on the MR results. To further verify the validity of the causal direction between exposure and outcome, we conducted the Steiger directionality test. 7 This method evaluates whether the selected genetic instruments explain more variance in the exposure than in the outcome, thereby confirming the assumed causal direction of the MR analysis.
All analyses were conducted via R software version 4.3.2, using R software version 4.3.2, primarily with the TwoSampleMR package.
Results
Baseline Characteristics of Participants
The baseline characteristics of 6284 participants are presented in Table 1. The mean age was 55.80 ± 11.75 years, and 50.91% were women. Overall, 516 participants had PAD, corresponding to a prevalence of 5.37%. Across HbA1c tertiles, significant differences were observed in demographic characteristics, lifestyle factors, comorbidities, and medication use (all p < 0.05). Participants in the higher tertiles were more frequently non-Hispanic Black, had lower levels of education, and were more often current or former smokers as well as non-drinkers. They also had higher BMI, lower eGFR, and a greater prevalence of hypertension, diabetes, and use of lipid-lowering therapy.
Baseline Characteristics by HbA1c Tertile Group.
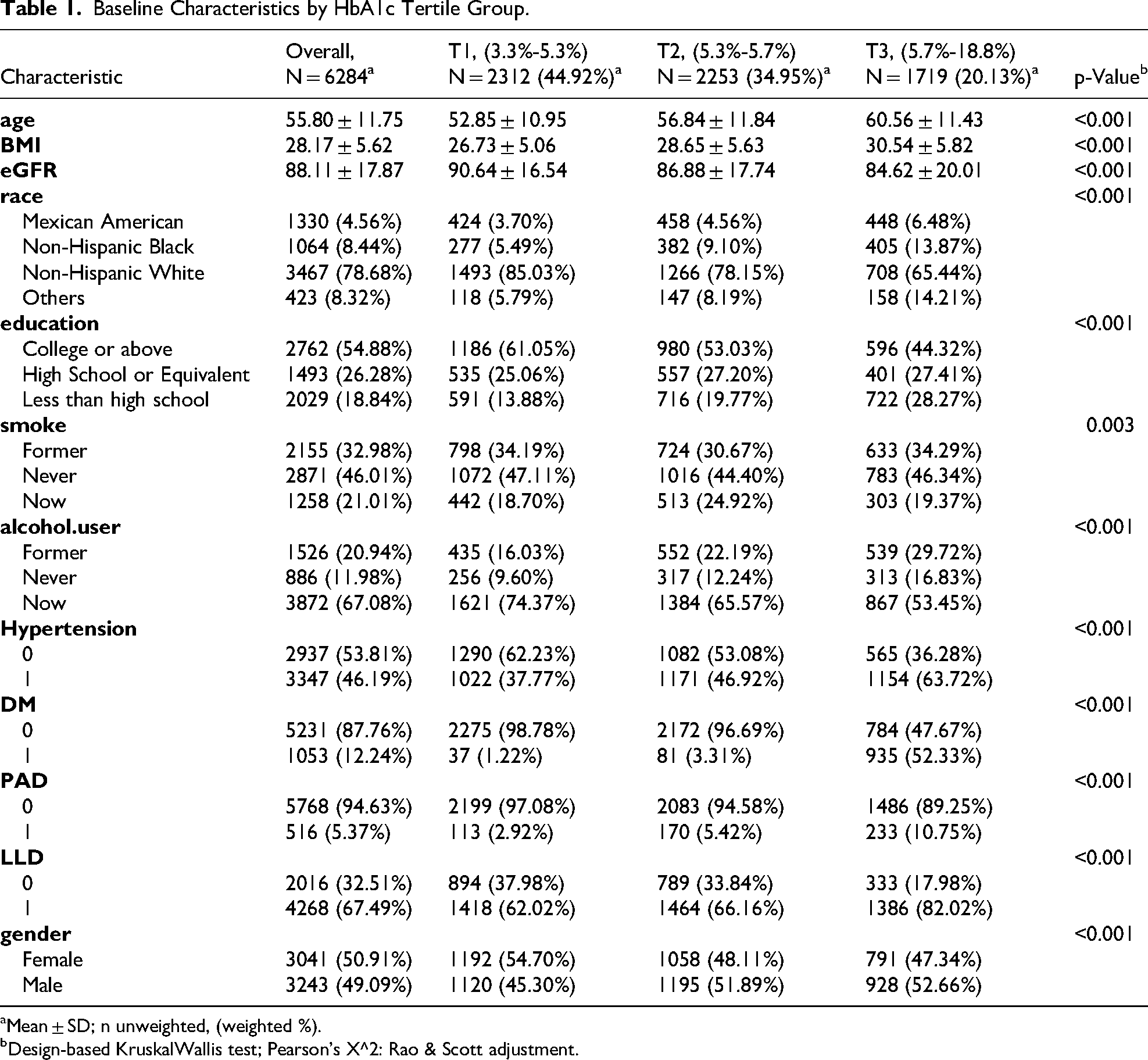
Mean ± SD; n unweighted, (weighted %).
Design-based KruskalWallis test; Pearson's X^2: Rao & Scott adjustment.
Association Between HbA1c and PAD in NHANES
We used weighted logistic regression models to assess the association between HbA1c and PAD. When HbA1c was categorized into tertiles, participants in the highest group (T3) consistently showed the highest odds of PAD. Compared with the reference group (T1), the ORs for T3 were 4.00 (95% CI: 2.98-5.37, p < 0.001) in Model 1 and 2.30 (95% CI: 1.68-3.15, p < 0.001) in Model 2. Although the estimates were attenuated after further adjustment, elevated HbA1c levels remained significantly associated with PAD (Model 3: OR 2.04, 95% CI: 1.45-2.86, p < 0.001; Model 4: OR 1.95, 95% CI: 1.39-2.73, p < 0.001). A significant linear trend across tertiles was observed in all models (p for trend < 0.001) (Table 2a).
Association Between HbA1c Tertiles and Risk of Peripheral Artery Disease (PAD) in Weighted Logistic Regression Models.

Abbreviations: CI = Confidence Interval, OR = Odds Ratio.
Model1: Crude
Model2: Adjust: race, education, gender, age
Model3: Adjust: race, education, smoke, alcohol.user, gender, age, BMI
Model4: Adjust: race, education, smoke, alcohol.user, Hypertension, LLD, gender, age, BMI, eGFR
When HbA1c was analyzed as a continuous variable, each 1% increase in HbA1c was associated with higher odds of PAD in the crude model (OR 1.29, 95% CI: 1.19-1.39, p < 0.001), and the association remained significant after progressive adjustment for demographic and clinical factors (Model 2: OR 1.23, 95% CI: 1.09-1.38, p = 0.001; Model 3: OR 1.19, 95% CI: 1.05-1.36, p = 0.009; Model 4: OR 1.18, 95% CI: 1.04-1.34, p = 0.012) (Table 2b).
Subgroup Analysis and Sensitivity Analysis
Stratified analyses based on fully adjusted models (Model 4) are presented in Figure 2. The association between HbA1c and PAD remained generally consistent across most subgroups. Stronger effects were observed among non-Hispanic Black (OR 1.10, 95% CI: 1.02-1.20) and non-Hispanic White participants (OR 1.31, 95% CI: 1.04-1.66), as well as in individuals with lower educational attainment (OR 1.25, 95% CI: 1.11-1.41). Current smokers (OR 1.25, 95% CI: 1.08-1.44) and current drinkers (OR 1.25, 95% CI: 1.00-1.56) also showed higher risks. The association appeared more evident in men and in participants without diabetes, although formal interaction tests reached only borderline significance. No significant interactions were detected for hypertension, lipid-lowering therapy, or alcohol status. Overall, the subgroup analyses were consistent with the main findings.

Subgroup analyses of the association between HbA1c and PAD risk in NHANES. Odds ratios were estimated from fully adjusted logistic regression models; error bars indicate 95% confidence intervals.
Nonlinear Trend and Threshold Effects
RCS analysis revealed a nonlinear association between HbA1c and PAD risk after accounting for all confounding factors (Figure 3). The nonlinearity was statistically significant (p value for nonlinearity = 0.0245). A threshold effect was observed, with an inflection point at 6.11% (p for log-likelihood ratio < 0.001) (Figure 4). Below this threshold, each 1% increase in HbA1c was associated with a nearly twofold higher risk of PAD (OR 1.99, 95% CI: 1.47-2.71, p < 0.001), whereas above this level the association was no longer significant (OR 1.02, 95% CI: 0.91-1.13, p = 0.739) (Table 3).

Restricted cubic spline regression showing the nonlinear association between HbA1c and PAD risk in NHANES. Models were fully adjusted for demographic, lifestyle, and clinical covariates; the shaded area represents the 95% confidence interval.

Threshold effect analysis of HbA1c and PAD risk using two-piecewise linear regression. An inflection point was identified at 6.11%, below which each 1% increase in HbA1c was significantly associated with higher PAD risk.
The Threshold Effect Analysis of the HbA1c on PAD Risk among Adults in NHANES.

Adjust: race, education, smoke, alcohol.user, Hypertension, LLD, gender, age, BMI, eGFR.
Potential Relationships Between HbA1c and PAD
A total of 292 instrumental variables suitable for Mendelian randomization (MR) analysis were selected (Supplemental Materials 1). In the MR analysis, the IVW method indicated that HbA1c was significantly associated with an increased risk of PAD (OR 1.23, 95% CI [1.101-1.370], P = 0.0002) (Figure 5). The forest plot clearly illustrated the effect of each SNP on the outcome (Figure 6a), while the scatter plot demonstrated the genetic association between HbA1c and PAD risk (Figure 6b).

Mendelian randomization analysis of the association between genetically predicted HbA1c and PAD.
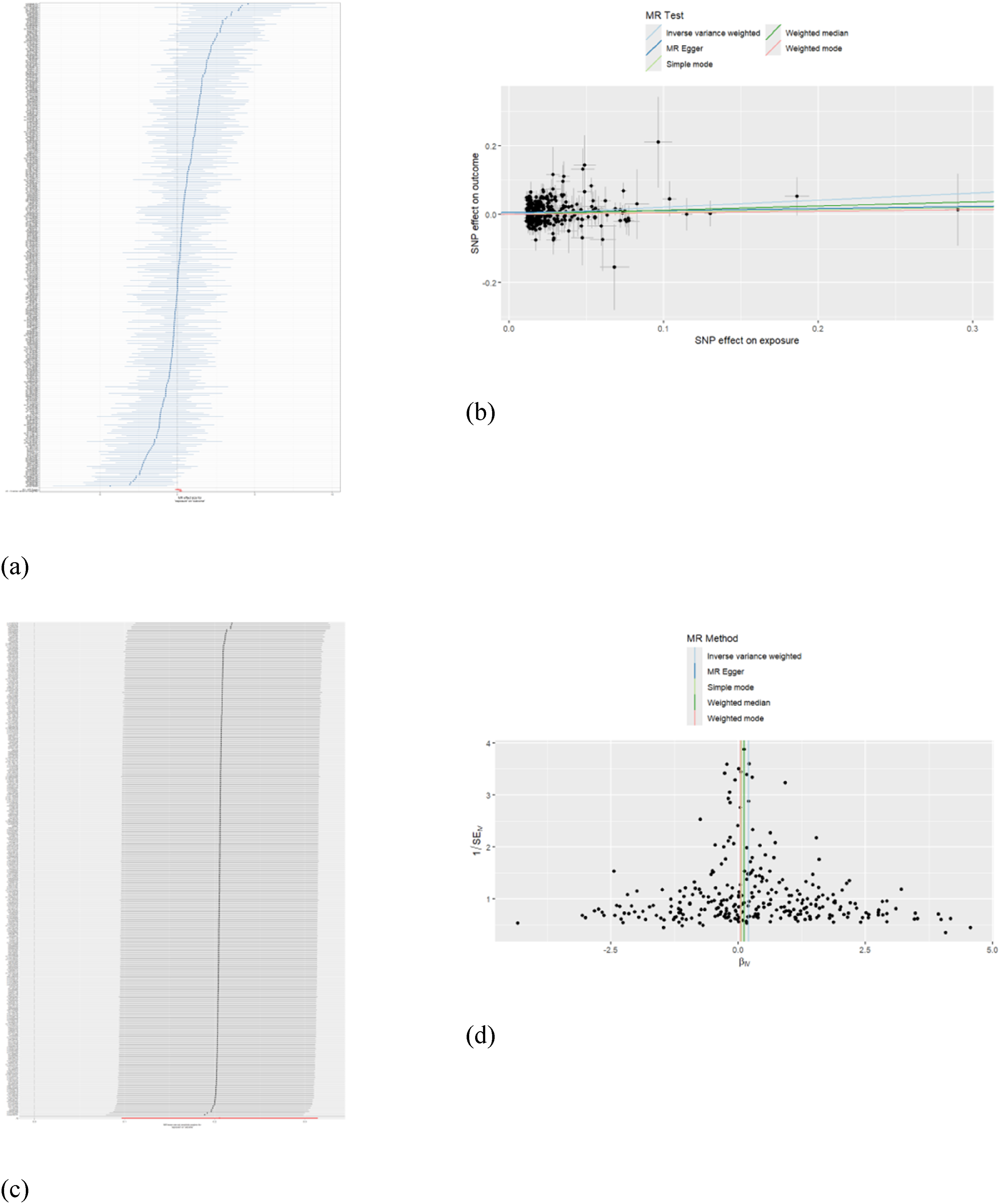
Mendelian randomization plots for the association between HbA1c and PAD risk scatter and forest plots illustrating the genetic association between HbA1c and PAD risk. (a) Scatter plot of SNP effects on HbA1c versus PAD. (b) Forest plot of individual SNP estimates. (c) Scatter plot of combined SNP effects. (d) Forest plot of combined estimates. Estimates are expressed as odds ratios (ORs) with 95% confidence intervals. Analyses were performed using IVW, MR-Egger, weighted median, and weighted mode approaches.
Sensitivity Analyses of MRs
Cochran's Q test suggested the presence of heterogeneity among the included SNPs (Table S1). The intercept terms from the MR-Egger regression yielded P values greater than 0.05, indicating that our results were not influenced by horizontal pleiotropy (Table S2). Leave-one-out analysis (Figure 6c) revealed that the absence of SNPs significantly influenced the results for both forward and reverse MRs. The funnel plot was used to assess potential heterogeneity among IVs (Figure 6d). The Steiger directionality test demonstrated that the majority of the instrumental variables explained greater variance in HbA1c than in PAD, supporting the hypothesized causal direction from HbA1c to PAD (Table S3). These results further supported the causal direction from HbA1c to PAD.
Discussion
Higher HbA1c levels were associated with an increased risk of PAD in both observational and genetic analyses. In NHANES, each 1% increase in HbA1c was associated with higher odds of PAD (OR 1.18, 95% CI 1.04-1.34), and participants in the highest tertile had more than a twofold greater risk compared with those in the lowest tertile (OR 2.30, 95% CI 1.68-3.15). The relationship was nonlinear, with spline models indicating a steeper rise in PAD risk at lower ranges and a plateau at higher HbA1c levels, and a threshold identified at 6.11%. In addition, Mendelian randomization analysis showed that genetically predicted HbA1c was also positively associated with PAD (IVW OR 1.23, 95% CI 1.10-1.37, p = 0.0002). The consistency between epidemiological and genetic evidence reduces concerns about residual confounding and supports a potential causal role of glycemic burden in PAD development.
Our findings are consistent with prior reports indicating that HbA1c level, rather than the diagnosis of diabetes itself, is closely tied to vascular risk.21,22 Several studies have demonstrated its value as an independent predictor of coronary artery disease in non-diabetic individuals, and even as a practical tool for identifying high-risk patients in primary care. 23 Beyond coronary disease, higher HbA1c has also been linked with stroke and cardiovascular mortality.24–26 Interestingly, some large cohorts have suggested a U-shaped association, where both very low and very high HbA1c levels are related to greater mortality risk.27–29 This pattern was also evident in a large registry of patients with established coronary artery disease undergoing revascularization, where even those with very low HbA1c faced increased mortality risk, underscoring the need to define optimal HbA1c targets in this population. 30 Together, these findings suggest that HbA1c may act as a continuous marker of vascular health and support our decision to examine PAD risk across HbA1c tertiles rather than grouping participants solely by diabetes status.
HbA1c is a simple and routinely used clinical measure that reflects average glycemia over the preceding 8–12 weeks, corresponding to the lifespan of circulating erythrocytes.31–34 This temporal stability reduces the influence of short-term fluctuations and underpins its role as a standardized diagnostic criterion for type 2 diabetes, as well as a complementary marker in the evaluation of other glycemic disorders. 35 Point-of-care HbA1c testing further enhances its utility by allowing rapid, on-site assessment, which is particularly valuable in resource-limited settings and in patients requiring frequent monitoring. 36 Importantly, the observed association was also evident among individuals without diagnosed diabetes, suggesting that HbA1c may capture vascular risk beyond the traditional diagnostic thresholds for diabetes.
In addition, we observed a nonlinear association between HbA1c and PAD, with an inflection point at 6.11%. This value is below the commonly used diagnostic threshold for diabetes (HbA1c ≥ 6.5%). The finding suggests that the vascular risk associated with glycaemic exposure may increase before the onset of clinically defined diabetes. Cardiovascular risk related to dysglycaemia often develops gradually rather than appearing only after diagnostic thresholds are reached. Our results therefore support the view that early elevations in HbA1c may already reflect vascular vulnerability.
The relationship between HbA1c and PAD is biologically plausible and may be explained through several interrelated mechanisms. Elevated HbA1c reflects chronic hyperglycemia, which initiates a cascade of metabolic and vascular disturbances. 37 Higher HbA1c levels have been linked to increased branched-chain amino acids and acylcarnitines, alongside reduced lysophosphatidylcholines. 38 These metabolic shifts promote insulin resistance and impaired glucose utilization, 39 and are accompanied by defective fatty acid oxidation and vascular injury,40,41 which may contribute to the increased risk of PAD. 42 Moreover, HbA1c has been identified as an independent determinant of blood viscosity and peripheral arterial blood flow,43,44 leading to reduced vascular compliance. Consistently, HbA1c levels have been independently associated with coronary atherosclerotic plaque burden even among non-diabetic individuals. 45
Our Mendelian randomization analysis provided genetic evidence supporting a potential role of HbA1c in PAD risk. Using the inverse-variance weighted approach, each genetically predicted 1% increase in HbA1c was associated with a 23% higher risk of PAD (OR 1.23, 95% CI 1.10-1.37, p = 0.0002). Consistent results were observed across complementary MR methods, including MR-Egger and weighted median, and sensitivity analyses suggested no evidence of horizontal pleiotropy or reverse causality. These findings are consistent with previous genetic studies linking HbA1c to cardiovascular outcomes, although relatively few MR analyses have specifically evaluated its association with PAD. By integrating nationally representative observational data from NHANES with genetic evidence, our study strengthens the overall evidence for an association between HbA1c and PAD. These findings suggest that HbA1c, beyond its role in glycemic monitoring, may also provide useful information for vascular risk assessment, particularly among individuals without diagnosed diabetes.
This study has several limitations. The cross-sectional nature of NHANES limits causal inference, although the consistency with MR findings strengthens confidence in the observed association. PAD was defined using ABI, which may underestimate subclinical disease. In addition, antihyperglycemic medications may influence HbA1c levels. Although diabetes status was adjusted for in the analyses, detailed information on specific glucose-lowering therapies was not incorporated into the models, and their potential impact on the association between HbA1c and PAD cannot be fully excluded. Finally, despite no evidence of pleiotropy in the MR analysis, differences in genetic architecture and clinical characteristics across populations may limit the generalizability of our findings.
Conclusion
In this population-based analysis, higher HbA1c levels were associated with a greater prevalence of PAD across observational and genetic analyses. The consistency between epidemiological findings and genetic evidence underscores the relevance of long-term glycemic exposure to PAD risk. These results suggest that HbA1c, beyond its established role in diabetes diagnosis and management, may provide additional information for vascular risk stratification.
Supplemental Material
sj-csv-1-cat-10.1177_10760296261437047 - Supplemental material for HbA1c as an Independent Predictor of Peripheral Artery Disease: Nonlinear Threshold and Causal Evidence from NHANES and Mendelian Randomization
Supplemental material, sj-csv-1-cat-10.1177_10760296261437047 for HbA1c as an Independent Predictor of Peripheral Artery Disease: Nonlinear Threshold and Causal Evidence from NHANES and Mendelian Randomization by Xixi Guo, Jianke Wang, Qiang Guo, Kai Zheng, Bin Ni, Xueqiang Fan and Qiangqiang Nie in Clinical and Applied Thrombosis/Hemostasis
Supplemental Material
sj-docx-2-cat-10.1177_10760296261437047 - Supplemental material for HbA1c as an Independent Predictor of Peripheral Artery Disease: Nonlinear Threshold and Causal Evidence from NHANES and Mendelian Randomization
Supplemental material, sj-docx-2-cat-10.1177_10760296261437047 for HbA1c as an Independent Predictor of Peripheral Artery Disease: Nonlinear Threshold and Causal Evidence from NHANES and Mendelian Randomization by Xixi Guo, Jianke Wang, Qiang Guo, Kai Zheng, Bin Ni, Xueqiang Fan and Qiangqiang Nie in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Abbreviations
Ethics Statement
This survey has received ethical approval from the NCHS Research Ethics Review Board, with all participants providing informed consent.
Informed Consent
The authors have nothing to report.
Funding
This work was supported by the National High Level Hospital Clinical Research Funding (2024-NHLHCRF-PY II-04).
Declaration of Conflicting Interests
The author(s) declare that financial support was received for the research and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
