Abstract
Peripheral artery disease (PAD), and subsequent chronic limb-threatening ischemia (CLTI), are frequently encountered among patients with end-stage renal disease (ESRD). Their coexistence is less favorable in comparison to patients with ESRD alone. We sought to investigate trends, comorbidities, determinants for cost, and prognostic outcomes in patients with concomitant ESRD and PAD. A retrospective analysis was performed using data from the National Inpatient Sample database from the years 2005-2014. ICD-9 codes were used to identify patients with diagnoses of PAD, CLTI, and ESRD. Pearson’s Chi-square, T-test, ANOVA, and multivariate binary logistic regression were used in this analysis. 7,214,843 patients with ESRD were identified. Of these, 123,499 patients were diagnosed with PAD and 102,447 with CLTI. Compared to ESRD alone, mortality rates increased with PAD and CLTI (5.7% vs. 13.9% vs. 15.9%, P < 0.001). Length of stay in days (7.3 vs. 10.2 vs. 11.1, P < 0.001) and in-hospital costs (59,872 vs. 85,866 vs. 89,016, P < 0.001) were higher with PAD and CLTI, respectively. CLTI demonstrated the highest independent predictor of mortality [OR = 6.93 (6.43-7.46), P < 0.001]. A decreasing trend in the rate of PAD (2005: 1.9% vs. 2014: 1.4%, P < 0.001) and CLTI (2005: 1.6% vs. 2014: 1.1%, P < 0.001) was noted. The presence of coexisting PAD, and furthermore CLTI, in patients with ESRD significantly raised in-hospital mortality, cost, and length of stay. A negative trend in rates of PAD and CLTI were observed. Proactive identification of this high-risk population may lead to accurate diagnosis and tailored therapeutic strategies.
Introduction
Peripheral artery disease (PAD) is a frequent diagnosis with high prevalence in the United States. An estimated 8-12 million Americans and 202 million people worldwide currently suffer from PAD. 1 The disease incites significant economic burden, increased early-age mortality, high morbidity rates, and low quality of life due to the symptoms it causes. 2 Although the most common symptom among patients with PAD is intermittent claudication, 3 the majority of patients are asymptomatic or present with atypical leg pain as the main complaint, 4 further delaying its diagnosis. The progression of PAD into chronic limb-threatening ischemia (CLTI) is an ominous clinical deterioration with high morbidity and mortality. CLTI is a clinical syndrome defined by the presence of PAD in combination with rest pain, gangrene, or a lower limb ulceration for greater than 2 weeks duration. 5
PAD, and later CLTI, is a frequent comorbidity of patients with chronic kidney disease (CKD) and end stage renal disease (ESRD), and is present in more than 24% of patients with an estimated glomerular filtration rate (eGFR) less than 60 ml/min/1.73 m2. 6 ESRD is defined as individuals with an eGFR less than 15 ml/min/1.73 m2, or those requiring hemodialysis regardless of eGFR. 7 In patients with ESRD, lower extremity amputations due to PAD occur 10 times more often than in those with adequate renal function. 8
Despite the frequency of these coexisting conditions, there is limited data describing the demographic characteristics, clinical outcomes, financial costs, and temporal trends in annual rates of these concomitant conditions. Taking advantage of a large database of hospitalized patients, we aim to describe such data.
Methods
Using the National Inpatient Sample (NIS) database, we identified patients greater than 18 years old with diseases of interest utilizing the ninth International Classification of Disease (ICD9) diagnostic codes from the years 2005 to 2014. This time frame was chosen to provide the maximum number of years for the analysis while maintaining the same version of ICD code. To better account for national estimates, our sample data was weighted using the provided discharge weights (DISCWT) from the NIS database. This weighting scale provides a sample of discharges from all hospitals nationally within the Healthcare Cost and Utilization Project (HCUP). Patients were included when the ICD9 codes of interest were listed as the first primary diagnosis, indicating a primary diagnosis at hospital admission.
Patients with ESRD were identified using code 585.6. A diagnosis of PAD was determined with inclusion of any of the following ICD9 codes: 440.20, 440.21, 440.22, 440.23, 440.24, and 440.29. A diagnosis of CLTI was determined with inclusion of diagnostic codes 440.2, 440.23 or 440.24 signifying disease with rest pain, ulceration, or gangrene, respectively. Cases that included more than one PAD diagnostic code within the provided diagnoses were excluded from this study.
Demographic data, including gender, age, race, hospital type, and household income were collected for analysis and provided by the NIS database. The Clinical Classifications Software (CCS) is a diagnosis and procedure code utilized by the NIS database to identify populations for a disease of interest. Comorbidities associated with patients of interest were analyzed utilizing these provided comorbidity variables extracted from the AHRQ co-morbidity severity data (congestive heart failure, chronic pulmonary disease, liver disease, metastatic cancer, obesity, diabetes mellitus, coronary artery disease). To identify patients with multi-vascular disease, all patients with any of the following codes qualified: myocardial infarction (CCS code = 100), cerebrovascular disease (CCS code = 109), or visceral atherosclerosis (CCS code = 114). Patient outcomes were assessed using data provided directly by the NIS database including in-hospital mortality, total costs billed during hospitalization, and length of stay. In-hospital mortality was defined as the patient dying during hospitalization. Charges accrued during hospitalization do not include professional fees and non-covered charges. If the source provided total charges with professional fees, then the professional fees are removed from the charge during HCUP processing.
Of the 13 demographic variables provided by the NIS database, 8 variables were missing data, and 3 variables were missing more than 5% of the original data. No variables were missing greater than 7% of data values. Hence, imputation of missing data was performed using the mean of nearby values in the original dataset. Results of this analysis were calculated using the complete data set after imputation techniques were implemented. Descriptive statistics between patients were analyzed using Pearson’s chi-squared tests. Interval data was analyzed utilizing parametric T-tests and ANOVA. To determine independent predictors of in-hospital mortality, odds ratios were calculated by univariate, followed by multivariate analysis using binary logistic regression for each comorbidity. Variables with statistical significance in univariate analysis (P < 0.20) were included in multivariate analysis to identify independent risk factors for mortality. Descriptive and numerical statistics, imputation, and logistic regression were calculated using SPSS software version 25 (IBM Corp., Armonk, New York, USA). We present continuous variables as mean with standard deviation and odds ratios with the 95% confidence interval. Approval of this study by institutional review board (IRB) was not required due to its retrospective design with de-classified data.
Results
A total of 7,214,843 hospitalizations were identified with ESRD between 2005 and 2014; 47.9% female and 52.1% male with a mean age of 62.7 ± 12.8 years. Of the patients diagnosed with ESRD, more than half were diagnosed with a form of cardiovascular disease. Yet, only a small subset of these patients were diagnosed with PAD (Table 1). There were 123,499 (1.7%) with ESRD who had concomitant diagnosis of PAD. Of these 123,499 hospitalizations, 102,447 (1.4%) were diagnosed with CLTI. Common comorbidities found in the ESRD cohort were coronary artery disease (51.9%), multi-vascular disease (43.8%), diabetes (42.5%), and congestive heart failure (22.5%).
Demographic and Prognostic Outcome Data Between Patients With ESRD Only, ESRD With PAD, and ESRD With CLTI.

*Continuous variables presented as mean (SD).
Compared to ESRD alone, in-hospital mortality rates were higher with concomitant PAD and subsequently with CLTI (5.7% vs. 13.9% vs. 15.9%, P < 0.001). Hospital length of stay in days was also shortest in the ESRD only group in comparison to ESRD-PAD and ESRD-CLTI groups (7.3 vs. 10.2 vs. 11.1, P < 0.001). When reviewing in-hospital costs, we found similar trends with higher costs in US dollars for patients with concomitant PAD and CLTI respectively (59,872 vs. 85,866 vs. 89,016, P < 0.001).
Figure 1 presents odds ratios of mortality from common comorbidities seen with ESRD. Comparing the present comorbidities, concomitant CLTI was the strongest independent predictor of mortality in patients with ESRD

Mortality logistic regression analysis.
There was a decreasing trend in the rate of PAD (2005: 1.9% vs. 2014: 1.4%, P < 0.001) and CLTI (2005: 1.6% vs. 2014: 1.1%, P < 0.001) during the study period which was statistically significant (Figure 2, Figure 3, respectively).
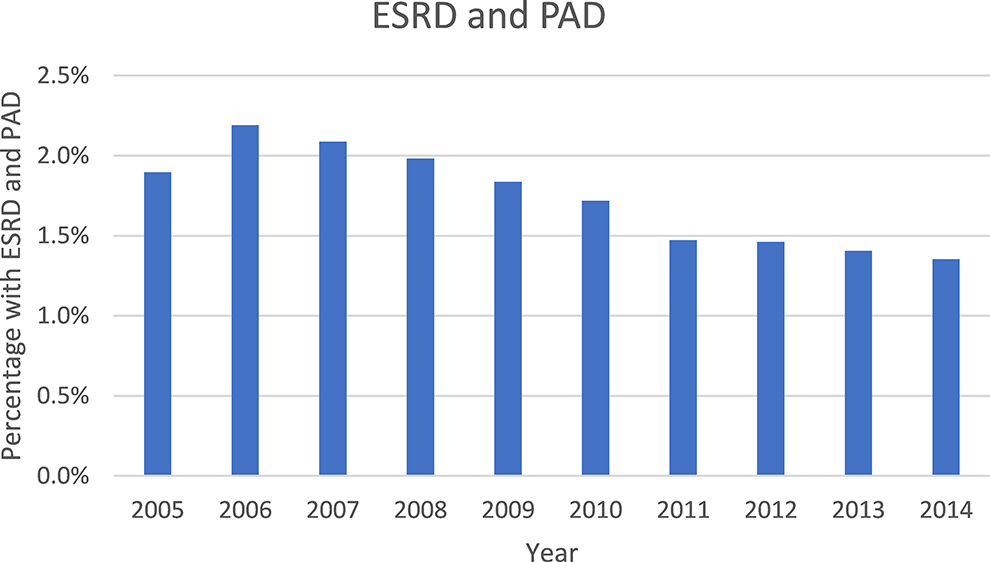
Yearly trend of ESRD and PAD (2005-2014).

Yearly trend of ESRD and CLTI (2005-2014).
Discussion
Patients with ESRD and concomitant PAD, or subsequent CLTI, were found to have longer hospital stays and increased hospital costs in comparison to ESRD alone. In our cohort of patients with ESRD, the diagnosis of CLTI had the strongest independent predictor of mortality, followed by metastatic malignancy and PAD, among other evaluated comorbidities. Surprisingly, a decreasing trend for PAD and CLTI was found from 2005 to 2014. It is plausible that a decreasing trend in diagnosis of PAD may be consequential of underdiagnosis. For CLTI, diagnosis of ESRD at a younger age or diabetic medications with better efficacy could explain these findings. A more in-depth investigation into these trends is necessary for a better explanation of these findings.
PAD represents a clinical spectrum ranging from asymptomatic individuals to patients with CLTI requiring amputation. The symptoms of PAD lead to decreased activity levels which leads to faster functional decline and morbidity. 9,10 Treatment of patients with PAD constitutes a large financial burden in the United States healthcare system with an average cost of $794,000 for 1 patient post-amputation. 11 Furthermore, approximately 35%-45% of patients are indicated to undergo lower extremity amputation at the time of diagnosis with CLTI. 12 Quality-adjusted life years is an economic measurement used to evaluate the effectiveness of an intervention in respect to the years of life lived post-intervention adjusted to the quality of life within those years. 13 One study by Barshes et al found that patients undergoing primary amputation for CLTI gained only 2.56 QALY post-amputation. 14 Current data demonstrates the 2-year mortality rate post-amputation is 38%. 15
The literature supports our findings that PAD is largely prevalent and a significant predictor of mortality in patients with ESRD. 16,17 Our study also investigated and quantified the increased risk of mortality with progression of PAD into CLTI. In individuals with ESRD, mortality is over 2 times higher when concomitant PAD is present and even higher at the stage of CLTI. Interestingly, most patients in our cohort with PAD were identified as CLTI, reinforcing the concept that most patients are diagnosed when the disease stage is too advanced. Although a decreasing trend for PAD and CLTI were observed, this in part may be a consequence of underdiagnosis rather than a true decrease in the number of cases. This assumption is based on 2 observations from the literature. First, most patient with PAD and ESRD are minimally symptomatic or present with atypical leg complaints rather than the classic claudication. 4 Hence, it is not uncommon to overlook PAD if other primary complaints are a reason for medical consultation in our practices. Second, the prevalence of other cardiovascular comorbidities, including coronary artery disease, was more than 50% in the same cohort. It is known that coronary artery disease and PAD share very similar risk factors and pathophysiologic processes that cannot be isolated to a single vascular bed.
Our findings highlight the strong, ominous impact that PAD has on individuals with ESRD. Taking advantage of a very large database of hospitalized individuals in the U.S., this data can be used to raise awareness as a pilot for change. As an example, a systematic review of the effect on patient awareness and education in patients with type 2 diabetes found an average absolute reduction of 0.57% in hemoglobin A1c in comparison to patients without education. 18 Understanding the morbid implications of PAD for those with ESRD can provide the groundwork for deeper investigation, proactive diagnosis, and early treatment.
Our study is limited by the intrinsic nature of retrospective population database review. However, the very large sample used decreases the likelihood of the findings being a consequence of chance. The NIS database is constructed from patient data exclusively from hospitals in the United States. While the sample size was adequate for interpretation, it is fair to say that demographic and prognostic outcomes may not be generalizable to other populations outside of the United States. This database does not provide patient data such as laboratory values, diagnostic imaging, or method of hemodialysis. This data would be useful to further investigate our patient cohort for severity of vascular disease. Furthermore, the database accuracy is relied upon by medical records acquired at hospital admission. It is possible that reporting error and misclassification bias occur with databases relying on patient sourcing.
With that in mind, it is plausible that proactive evaluation in the ESRD population may lead to earlier diagnosis and adequate treatment of PAD to halt or slow disease progression.
Conclusion
In patients with ESRD, the coexistence of PAD, and furthermore CLTI, are associated with a higher mortality, longer hospitalization time, and increased healthcare-associated costs. Furthermore, CLTI is the strongest independent predictor of mortality in the ESRD population, followed by metastatic cancer and PAD. Although negative trends for PAD and CLTI were observed in the study period, this is possibly a result of underdiagnosis. Increasing awareness of the futility of comorbid PAD/CLTI in the ESRD population can be a trigger for proactive diagnosis and early therapy. Prospective studies evaluating the effects of early intervention are needed.
Footnotes
Authors’ Note
The findings of this paper were presented at the 2020 Arteriosclerosis, Thrombosis, and Vascular Biology (ATVB) conference in Chicago, Illinois.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
